The clinical management of the individual requiring pacemaker therapy occurs across a range of settings. Frequently, the patient's need for a pacemaker is identified when the patient presents to a physician's office, ambulatory care setting, or emergency department with a complaint of frequent dizziness, syncopal or near-syncopal episodes, unexplained falls, or increasing signs of heart failure. Implantation of the pacemaker occurs in an acute care setting, where the patient's clinical status and pacemaker function are closely monitored for a short period. Long-term follow-up generally occurs in physicians' offices or ambulatory care settings. Using the recommendations published by the American College of Cardiology Foundation and the American Heart Association, medical indications will be outlined. Preoperative, immediate postoperative, and long-term follow-up care for patients undergoing permanent pacemaker implantation will be discussed. Emphasis will be placed on patient education. Topics that will be covered include safe use of home appliances and the issues of electromagnetic interference from sources such as airport security, commercial antitheft devices, cellular phones, and others. The use of biventricular pacemakers/cardiac resynchronization therapy (CRT) for persons with severe heart failure will be explored. Finally, application of the content will be provided through detailed case studies.
- INTRODUCTION
- PACEMAKER FUNCTION
- PACEMAKER CODING
- TYPES OF ANTIBRADYCARDIA PACEMAKERS
- ANTIBRADYCARDIA PACING
- DIAGNOSIS AND EVALUATION FOR ANTIBRADYCARDIA PACEMAKER IMPLANTATION
- PACEMAKER IMPLANTATION
- MONITORING PACEMAKER FUNCTION
- DISCHARGE EDUCATION
- FOLLOW-UP CARE AFTER ANTIBRADYCARDIA PACEMAKER IMPLANTATION
- TROUBLESHOOTING ANTIBRADYCARDIA PACEMAKER FUNCTION
- PACEMAKER SYNDROME
- BIVENTRICULAR PACING
- EMERGING TRENDS IN PACEMAKER THERAPY
- CLINICAL CASE STUDIES
- CONCLUSION
- RESOURCES
- GLOSSARY OF TERMS
- Works Cited
- Evidence-Based Practice Recommendations Citations
This course is designed for physicians, nurse practitioners, and nurses practicing in acute or adult healthcare settings.
Pacemakers are being used with increasing frequency in the United States, and healthcare professionals are likely to encounter patients for whom pacemakers may be indicated. The purpose of this course is to provide basic information on pacemaker therapy, indications for implantation, preprocedure and postprocedure care, identification and management of pacemaker malfunctions, and patient education.
Upon completion of this course, you should be able to:
- Describe the incidence, history, and trends in permanent pacemaker therapy for adults.
- Review normal cardiac conduction.
- Describe the basic components of an antibradycardia pacemaker system.
- Outline the basic and advanced pacemaker functions.
- Discuss five-digit pacemaker coding systems.
- Discuss the features of single and dual chamber atrial and ventricular demand pacemakers.
- Review symptomatic bradycardia and sinus node dysfunction indications for permanent pacemaker implantation in adults.
- Discuss second- and third-degree atrioventricular (AV) block as indications for permanent pacemaker implantation.
- Identify other potential indications for permanent pacemaker implantation.
- Describe key aspects of patient history that should be included in the diagnosis and evaluation of a patient for permanent pacemaker implantation.
- Discuss diagnostic tests used in the evaluation of a patient for permanent pacemaker implantation.
- List factors that should be considered in the selection of a specific type of pacemaker for an individual patient.
- Briefly describe the preprocedure care and implantation procedure for permanent pacemakers.
- Identify key points involved in monitoring pacemaker function in the postimplantation period.
- List key components of discharge education and follow-up care for a patient following permanent pacemaker insertion.
- Discuss identification and management of common pacemaker problems including end-of-battery life indications, failure to sense, failure to fire, and failure to capture.
- Review pacemaker syndrome, including signs and symptoms, underlying cause, and management.
- Describe the management of patients requiring biventricular pacemakers for cardiac resynchronization therapy (CRT), including indications for use and patient monitoring.
Karen Majorowicz, RN, is currently employed in the Cardiac Intermediate Care Unit at Shands Healthcare at the University of Florida, Gainesville. She received her Master's in Medical-Surgical Nursing in 1978 from the University of Maryland. Karen has created numerous instructional manuals on Medicare and has conducted educational programs on cardiovascular assessment.
Contributing faculty, Karen Majorowicz, RN, has disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
Ronald Runciman, MD
Mary Franks, MSN, APRN, FNP-C
The division planners have disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
Sarah Campbell
The Director of Development and Academic Affairs has disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
The purpose of NetCE is to provide challenging curricula to assist healthcare professionals to raise their levels of expertise while fulfilling their continuing education requirements, thereby improving the quality of healthcare.
Our contributing faculty members have taken care to ensure that the information and recommendations are accurate and compatible with the standards generally accepted at the time of publication. The publisher disclaims any liability, loss or damage incurred as a consequence, directly or indirectly, of the use and application of any of the contents. Participants are cautioned about the potential risk of using limited knowledge when integrating new techniques into practice.
It is the policy of NetCE not to accept commercial support. Furthermore, commercial interests are prohibited from distributing or providing access to this activity to learners.
Supported browsers for Windows include Microsoft Internet Explorer 9.0 and up, Mozilla Firefox 3.0 and up, Opera 9.0 and up, and Google Chrome. Supported browsers for Macintosh include Safari, Mozilla Firefox 3.0 and up, Opera 9.0 and up, and Google Chrome. Other operating systems and browsers that include complete implementations of ECMAScript edition 3 and CSS 2.0 may work, but are not supported. Supported browsers must utilize the TLS encryption protocol v1.1 or v1.2 in order to connect to pages that require a secured HTTPS connection. TLS v1.0 is not supported.
The role of implicit biases on healthcare outcomes has become a concern, as there is some evidence that implicit biases contribute to health disparities, professionals' attitudes toward and interactions with patients, quality of care, diagnoses, and treatment decisions. This may produce differences in help-seeking, diagnoses, and ultimately treatments and interventions. Implicit biases may also unwittingly produce professional behaviors, attitudes, and interactions that reduce patients' trust and comfort with their provider, leading to earlier termination of visits and/or reduced adherence and follow-up. Disadvantaged groups are marginalized in the healthcare system and vulnerable on multiple levels; health professionals' implicit biases can further exacerbate these existing disadvantages.
Interventions or strategies designed to reduce implicit bias may be categorized as change-based or control-based. Change-based interventions focus on reducing or changing cognitive associations underlying implicit biases. These interventions might include challenging stereotypes. Conversely, control-based interventions involve reducing the effects of the implicit bias on the individual's behaviors. These strategies include increasing awareness of biased thoughts and responses. The two types of interventions are not mutually exclusive and may be used synergistically.
#90804: Antibradycardia Pacemakers
The use of pacemaker therapy in the clinical management of persons with heart disease is well-established. It is estimated that more than 250,000 persons in the United States have a pacemaker implanted each year, and more than 700,000 pacemakers are implanted worldwide [1]. As the American population continues to age, the incidence of elderly persons requiring permanent pacemaker therapy is likely to increase, as more than 80% of patients requiring pacemaker therapy each year are 60 years of age or older [2].
Pacemakers were originally developed for use in emergency situations to aid cardiopulmonary resuscitation efforts. One of the earliest experimental pacemakers was developed in the 1930s; it consisted of a flashlight battery to provide an electrical power source, a mechanical timer to regulate the frequency of the electrical discharge, and a switch to allow control of the duration of each burst of electrical current. In the 1930s, cardiac resuscitation was highly controversial, and this early work never proceeded significantly past the experimental stage. In the 1950s, definitive progress was made in the development of a permanent implantable pacemaker. The first implantable pulse generator was designed and implanted, and transvenous pacemaker leads were developed and tested. These early pacemakers were very limited in function. Unlike today's pacemakers, the early devices could pace only in the ventricle and only at a preset rate; they were unable to sense the heart's own electrical activity and were not capable of altering the pacing rate when the person's activity level and metabolic demands changed. Early pulse generators were considerably larger than today's models and could be difficult to implant in some individuals. Battery life in the earliest pulse generators was very short, and often the end of the battery life occurred abruptly, creating an emergency situation for the pacemaker-dependent patient.
In the 1960s and 1970s, tremendous advances in pacemaker therapy occurred; key achievements included the development of a pulse generator that could both pace the heart and sense the heart's electrical activity and the creation of a pacemaker capable of sensing and pacing in both the atria and the ventricles at the same time.
Technologic advances in pacemaker therapy continue rapidly. Pacemakers have additional complex functions to help them function more like the heart's normal conduction system. These include a rate responsive feature that allows the pacemaker to modify the pacing rate in response to changes in a person's activity level or metabolic demands and an automatic mode switching feature that allows the pacemaker to switch from one type of pacing mode to another when the patient's heart rhythm changes. In addition, pulse generators have gotten smaller and more compact in size, and battery life and reliability have improved markedly [3,4,5].
As pacemaker technology has advanced, the clinical indications for implantation of a permanent pacemaker have expanded. Consider these examples:
Patient A, a man 83 years of age, receives a permanent pacemaker capable of sequentially pacing the atria and the ventricles. Patient A has a history of repeated falls with injury caused by diminished cerebral blood flow that occurs when his heart rate drops to 30 beats per minute (bpm).
Patient B, a woman 56 years of age, receives a permanent ventricular demand rate-responsive pacemaker following the radiofrequency ablation of her atrioventricular (AV) node for management of persistent refractory atrial fibrillation (AF) with rapid ventricular response.
Patient C, a woman 60 years of age, receives a permanent pacemaker for management of persistent symptomatic bradycardia caused by second-degree heart block following an anterior wall myocardial infarction (MI). Because her sinoatrial (SA) node is functional, Patient C's pacemaker is programmed to monitor her SA node functioning and pace the ventricles at the same rate.
Patient D, a man 72 years of age, receives a permanent pacemaker after he experiences a four-second pause accompanied by a severe drop in systolic pressure during a tilt-table test.
Patient E, a man 45 years of age with nonischemic cardiomyopathy, has a biventricular pacemaker placed to facilitate improved coordinated contraction of his right and left ventricles, to maximize his cardiac output and help relieve his symptoms of congestive heart failure.
Patient F, a woman 46 years of age with a history of documented ventricular tachycardia, undergoes implantation of a combination automatic internal defibrillator and permanent pacemaker for management of her tachyarrhythmias.
As indicated by these examples, implantation of a permanent pacemaker has become a mainstay in the management of persons with symptomatic bradycardia caused by such arrhythmias as sick sinus syndrome, first-, second-, and third-degree AV block, and atrial fibrillation with slow ventricular response. Permanent pacemakers have also been found to be effective in the management of persons with hypersensitive carotid sinus syndrome. Additionally, device manufacturers have produced an implantable device that combines the capabilities of an automatic internal cardiodefibrillator and permanent pacemaker. This therapy is used in the management of patients with some forms of tachyarrhythmias. Finally, medical research has shown that biventricular pacemakers are effective in improving quality of life and reducing symptoms for persons with severe congestive heart failure and some forms of cardiomyopathy [3,4,7,8,9,28].
The purpose of a pacemaker is to assist the heart to maintain an effective heartbeat and, if possible, a heart rhythm that is as close to normal as possible. Before examining basic pacemaker functions in detail, a brief review of the normal conduction system in the heart is necessary.
In the normal heart, the heartbeat is initiated by the SA node, which is located in the upper part of the right atrium. The SA is the primary pacemaker of the heart and normally fires at a rate of 60 to 100 impulses per minute. The electrical impulse travels from the SA node through both right and left atria, causing depolarization of the atrium. Complete depolarization takes approximately 80–100 milliseconds (ms). Atrial depolarization is followed by atrial contraction and atrial repolarization. The electrical impulse travels from the atria to the atrioventricular (AV) node, located in the bottom part of the right atrium. The speed of conduction slows in the AV node to allow the atria time to contract and complete ventricular filling. From the AV node, the electrical impulse travels through the bundle of His, located in the septum of the heart between the right and left ventricles. The bundle of His divides into the right and left bundle branches. These branches divide further into the smaller fibers of the Purkinje system. Electrical conduction through the His-Purkinje system is rapid, causing depolarization of both right and left ventricles. Depolarization of the ventricular cells spreads from the apex of each ventricle to the base and moves from the inside layer of the heart (i.e., the endocardium) to the outer layer (i.e., the epicardium). The entire ventricle depolarizes in approximately 80–100 ms. Ventricular depolarization is followed by ventricular contraction and ventricular repolarization [10,11,12].
When an electrical impulse stimulates a cardiac cell, a series of events are initiated that cause the cell to depolarize and repolarize. This generates an action potential that allows the electrical impulse to continue to travel. Ultimately, this process results in the contraction of the cells in the heart [10]. The major events are outlined in this section.
When an electrical impulse stimulates a cardiac cell, the cell depolarizes. Positively charged sodium ions from the extracellular space flood rapidly into the intracellular space. This increases the total number of positively charged ions in the intracellular space, and the charge in the intracellular space becomes less negative. The potential or voltage in the cell increases. This is phase 0 of the development of the action potential.
The flood of sodium ions into the intracellular space stops very quickly. It is followed by a brief and incomplete period of repolarization. This period is mediated by a temporary movement of potassium ions from the intracellular to the extracellular space. This brief period of repolarization is referred to as phase 1 of the action potential.
Phase 2 of the action potential is characterized by a balance of inward and outward movement of ions. Calcium ions move slowly through select channels into the intracellular space while potassium ions move out through multiple channels into the extracellular space. This initiates a slow repolarization and creates a plateau in the action potential. Cardiac contraction is mediated by phase 2.
In phase 3, the calcium channels close. The process of repolarization is accelerated. In phase 4, electrical diastole occurs. Except for the SA node, the heart rests. The SA node begins the process of initiating the next electrical impulse.
After the cardiac cell has depolarized, there is a period of time that the cell cannot generate an action potential in response to another electrical impulse. This is referred to as the absolute refractory period. As the cell continues to repolarize, an effective refractory period occurs, in which the cell can transiently depolarize in response to an electrical impulse but generally will not develop enough of an action potential to propagate the impulse to surrounding cells. As repolarization nears completion, the cell is said to be in a relative refractory period; in this period, a strong electrical stimulus can trigger the cell to depolarize and create another action potential [13].
Electrical events in the cardiac cycle are reflected in the electrocardiogram (ECG) waveform. The P wave represents atrial depolarization. The P-R interval reflects the amount of time from the beginning of atrial depolarization until the onset of ventricular depolarization; it represents the amount of time the electrical impulse takes to travel from the SA node through the AV node. The QRS complex represents the amount of time it takes the ventricles to depolarize. In normal conduction, ventricular depolarization occurs rapidly. This rapid conduction is reflected in a narrow QRS complex. The T wave represents ventricular repolarization. The Q-T interval represents the amount of time that it takes the ventricles to depolarize and repolarize and is measured from the beginning of ventricular depolarization (i.e., start of the QRS complex) to the end of repolarization (i.e., end of the T wave). During the early part of the Q-T interval, the ventricles are completely refractory and unable to respond to another electrical impulse. During the latter part of the interval, the ventricles are only partially refractory and may respond to some impulses. When changes occur in the normal cardiac cycle, the normal ECG waveform is altered to reflect them. For example, prolonged repolarization is reflected in a prolonged Q-T interval. A slowing of conduction from the SA node through the AV node may be reflected in a prolonged P-R interval. Abnormal conduction of the electrical impulse through the ventricles results in a QRS that is wider than usual or bizarre in shape, such as seen with bundle branch block (BBB). Careful analysis of the changes in a patient's ECG can provide valuable information in the diagnosis and treatment of an arrhythmia [10,14].
Permanent pacemaker systems are composed of several main parts: the pacemaker generator, the power source, the electronic circuitry, the pacemaker lead(s), and the external programmer.
The pacemaker generator is a small titanium case that contains the electronic circuitry and a power source for the pacemaker. Most generators are small and lightweight. They may range in weight from 1–1 ¾ ounces with average dimensions of 2 inches x 2 inches x ¼ inch. To protect the pacemaker's electronic circuitry from outside interference and to protect the patient from the risk of battery leakage, the components of the generator are sealed in layers of airtight materials [15,16].
Pacemaker Power Source
The power source for pacemaker generators is usually a lithium-iodide battery. Depending on use, a lithium battery typically lasts from 5 to 10 years. As lithium batteries near the end of their battery life, they show predictable, identifiable characteristics that serve as an early warning that the battery is becoming depleted. The early warning allows the physician to schedule the battery change procedure in a timely (as opposed to urgent or emergent) fashion [3,4,15,16,17,18].
Electronic Circuitry
The electronic circuitry that controls the functioning of the pacemaker is also housed in the generator. The circuitry is programmed by the physician when the pacemaker is initially implanted. The programming may be noninvasively changed at later intervals as needed. Four different types of circuits are present: a sensing circuit, a pacing or output circuit, a timing circuit, and a transceiver. The sensing circuit monitors the electrical activity in the heart. The output circuit transforms the electrical energy generated by the pacemaker battery into an electrical pulse that is delivered to the heart to initiate a heartbeat. The strength (or voltage) of the electrical impulse is carefully programmed to provide the proper amount of stimulus to the heart to stimulate depolarization. The timing circuit controls the timing of the artificial electrical impulses that are delivered to the heart by the pacemaker. It is programmed at the time that the pacemaker is implanted; its programming may be adjusted noninvasively as needed to maximize pacemaker function for the individual patient. The transceiver is an electronic device that allows the attending or monitoring physician to "talk" to the electronic circuitry in the pacemaker by way of a specialized programmer. This permits the physician to retrieve data about the pacemaker's current settings and stored data about the pacemaker's functioning. It also allows one to change the programmed settings in the generator noninvasively [3,4].
The pacemaker lead connects the pacemaker generator to the heart. A pacemaker lead serves two main functions: to deliver the pacing impulse created by the pacemaker generator directly to the heart muscle (pacing function), and to relay information about the heart's electrical activity back to the electronic circuitry in the generator (sensing function).
A pacemaker lead consists of an extremely thin, flexible wire that is coated with suitable insulation, usually either silicone or polyurethane. The extreme flexibility of the wire allows the lead to withstand normal body movements without breaking. On one end of the pacemaker lead is a connector pin; this pin attaches the pacemaker lead firmly to the connector block that is located on the outside of the generator. On the other end of the pacemaker's lead is the pacing electrode that is used to deliver the electrical impulse directly to the heart. To work effectively, this electrode must be in direct contact with the heart. A second electrode is also located on the pacemaker lead. It is a sensing electrode designed to monitor and relay information about electrical activity back to the generator.
Pacemaker leads may be attached to the epicardial (outer) or endocardial (inner) surface of the heart. Most permanent pacemaker leads are attached to the endocardium. To hold the pacemaker lead in position inside the heart, the pacemaker lead has a small fixation device at the tip. There are two major types of fixation devices: active fixation and passive fixation. Active fixation devices have a small screw that extends from the end of the pacemaker lead into the endocardium to hold the lead in place. Passive fixation devices have tines (like the tines of a fork) on the end; these tines are designed to become entangled in the trabeculae inside the atria or ventricle to hold the lead in place. In addition to a fixation device, pacemaker leads have a small amount of steroids in a reservoir at the tip of the lead. When the lead is positioned and attached, the steroid is released. The steroid, through its anti-inflammatory action, reduces the amount of inflammation and scar tissue that develop around the tip of the lead. Minimizing scar tissue allows the pacing electrode at the end of the lead to deliver electrical impulses more effectively [3,17,18,19].
In order for the pacemaker to deliver an effective electrical impulse to the heart, a complete electrical circuit must be present. A complete circuit requires both a positive electrode (anode) and a negative electrode (cathode). To provide the complete circuit, pacemaker leads may be designed as either unipolar or bipolar. A unipolar lead contains only a negative electrode, located on the lead tip. The metal casing of the generator acts as the positive pole or anode. A bipolar lead, on the other hand, contains both a positive and a negative electrode. The negative electrode again is located on the tip of the lead; the positive electrode is located several centimeters behind the negative electrode. Because the electrodes are relatively close together on a bipolar lead, the electrical circuit is smaller and is less likely to be disturbed or disrupted by outside electrical interference than a unipolar lead. However, some sources suggest that bipolar leads are more fragile than unipolar leads and more likely to break [16,20].
Depending on the type of pacemaker, one or more pacemaker leads may be used. Single chamber pacemakers, which pace and sense only in the ventricle or only in the atrium, use a single pacemaker lead. Dual chamber systems that are capable of sensing and pacing in both atria and ventricles use two leads. The newer biventricular pacemakers may use three leads: one positioned in the right atrium, a second in the right ventricle, and a third in the left ventricle [20].
In addition to the generator and pacemaker leads, which are implanted inside the person's body, the proper functioning of a permanent pacemaker requires the intermittent use of an external device called a programmer. A pacemaker programmer consists of a relatively portable specialized computer. The technology in the programmer allows the computer to communicate with the pacemaker via the transceiver housed in the pacemaker generator. This process of noninvasive communication is called "interrogation" of the pacemaker. To interrogate an implanted pacemaker, a physician or other person trained in the use of pacemaker programmers holds a wand directly over the generator site. Using remote telemetric communication technology, the wand can transmit data from the pacemaker generator to the programmer. The information retrieved usually includes the current pacemaker settings and some information about past pacemaker function. In addition, data about the status of the battery and remaining battery life may also be obtained. The retrieved data may be printed for later analysis and/or documentation in the patient's medical record. Changes in pacemaker settings may also be transmitted from the pacemaker programmer through the wand to the generator. This permits the physician to adjust settings based on analysis of the pacemaker's past functioning and the assessment of the patient's current status [17,18].
For a permanent pacemaker to act effectively to initiate a heartbeat when the heart's own conduction system fails to do so requires the interaction of three basic functions: pacing, sensing, and timing.
When the heart's normal rhythm is interrupted, the pacemaker can initiate a heartbeat by pacing the heart. Using power from its battery, the pacemaker generator creates a tiny electrical signal or impulse. This impulse is sometimes called a pacing pulse, pacing impulse, or pace. The impulse is conducted through the pacing lead to the electrode on the tip. The impulse is discharged through the electrode directly into the surrounding cells of the heart. If delivered at the right time and the proper voltage, the impulse causes the surrounding cells to depolarize, thus initiating a heartbeat. As discussed, the output circuit in the pulse generator is responsible for generating an impulse that has enough voltage to trigger depolarization. At the time of implantation, the amount of electrical energy needed to depolarize the heart is carefully determined. This is sometimes referred to as the pacing threshold or the stimulation threshold. Once the pacing threshold has been established and programmed into the pacemaker generator, the pacemaker pacing function is tested to ensure that the amount of voltage delivered in each pacing impulse can successfully trigger depolarization. If the pacing impulse successfully triggers depolarization and initiates a heartbeat, the pacemaker is said to have effectively achieved "capture." Depending on the patient's specific conduction abnormality, pacing impulses may be delivered to the atria, the ventricles, or both. The rate and timing of the pacing impulses will vary with the pacemaker's programmed settings. The frequency of paced beats depends on the patient's underlying rhythm. In some instances, when the patient has a severe conduction abnormality, the pacemaker will pace the heart 100% of the time. If the patient's conduction problem is only intermittent, the pacemaker will fire only when needed. The ability of the pacemaker to pace the heart only when needed is referred to as "demand" pacing. Demand pacing prevents the pacemaker from competing with the heart's own intrinsic rhythm and reduces the risk that the pacing impulse will fall at a vulnerable point in the cardiac cycle and throw the heart into a chaotic rhythm. Demand pacing enables the heart to maintain a minimum rate and prevents long pauses without an effective heartbeat, maintaining cardiac output and cerebral circulation [17,19,20].
For the pacemaker to deliver a pacing impulse on demand, the generator must recognize or sense when the beat is needed. Sensing occurs when the pacemaker monitors and recognizes electrical activity in the heart. As previously noted, the pacemaker lead is equipped with both a sensing function and a pacing function. The sensing function monitors for depolarization in a specific region of the heart; if depolarization occurs, the sensing electrode transmits that information back through the pacemaker lead to the sensing circuit in the generator. The generator processes the information and responds according to its programmed parameters. For the pacemaker to recognize electrical activity in the heart correctly, the sensing threshold must be carefully established and programmed. The sensing threshold is defined as the largest electrical signal (ventricular or atrial) that can be consistently recognized by the sensing circuit. Sensing thresholds that are set too high or too low will result in improper functioning of the pacemaker's pacing function. Depending on the type of pacemaker, sensing may occur only in the atria, only in the ventricles, or in both the atria and the ventricles. The sensing function prevents the pacemaker from competing with the heart's own rhythm. It also permits the pacemaker to perform the more complex functions required by dual chamber and rate responsive pacemakers [17,18,19,21].
The timing function works interactively with the pacing and sensing functions of the pacemaker to ensure that a pacing impulse is delivered to the heart when it is needed, according to the pacemaker's programmed parameters. The timing function is very complex; it controls a variety of intervals that are involved in a pacing cycle. The specific intervals that are programmed depend on the type of pacemaker and its parameters. The timing function common to all types of demand pacemakers is the low rate limit setting. The low rate limit setting is the rate at which the pacemaker will pace the heart when the heart fails to initiate its own beat. This setting is usually expressed in terms of bpm and is often set at a rate of 50–70 bpm. To program the low rate setting limit, a low rate setting timing interval is used. This timing interval defines the maximum length of time that may occur between two paced beats, an intrinsic beat and a paced beat, or two intrinsic beats. The interval is based on the low rate limit; if the low rate limit is set at 60 bpm, a heartbeat (either paced or intrinsic) should occur approximately every second. If the low rate limit is set at 50 bpm, a heartbeat should occur approximately every 1.2 to 1.3 seconds. If the end of the timing interval is reached and no heartbeat occurs, the generator sends a pacing impulse to the heart. The sequence of events is [20]:
The timing circuit in the generator is activated.
The sensing circuit monitors for a heartbeat to occur before the end of the programmed low rate limit timing interval.
If a heartbeat occurs before the end of the programmed interval, the pacing function is inhibited, and the pacemaker generator does not deliver a pacing impulse to the heart.
The timing circuit resets.
Following a brief refractory period, the sensing circuit monitors for the occurrence of another heartbeat within the programmed interval.
If another normal heartbeat does not occur before the end of the programmed interval, the pacing function is not inhibited, and the pacemaker delivers a pacing impulse through the pacemaker lead to the heart.
The timing circuit again resets.
Following a brief refractory period, the sensing circuit monitors to determine if another heartbeat occurs before the end of the programmed interval.
This cycle repeats continuously.
In addition to the basic functions required by demand pacing, modern pacemakers may be equipped with advanced features and programmable options. These include a rate adaptive or rate responsive feature and magnet mode.
In the past, a drawback of many permanent pacemakers was the inability of the devices to increase the rate of pacing to meet increased metabolic demands of the body. Only one pacemaker, the dual chamber pacemaker (coded DDD), had the ability to vary pacing rates. However, in order for the dual chamber pacemaker to increase its rate, the patient's SA node had to be functioning normally. Because many people who required pacemaker therapy did not have intact SA node function, this DDD capability was of limited value. To provide an artificial substitute for the chemical, neural, and hormonal factors that normally increase the heart rate by stimulating the SA node, researchers developed a rate adaptive or rate responsive feature for permanent pacemakers. With the rate responsive feature, the pacemaker can increase or decrease its pacing rate in response to an identifiable parameter that reflects increased activity or increased metabolic demands. In an attempt to identify the most accurate and stable indicator for rate changes, researchers initially explored a wide variety of indicators, including minute ventilation, body temperature, changes in pH, changes in venous oxygen saturation in the body, increased stroke volume, changes in force of contraction, changes in respiratory rate, changes in skeletal muscle activity, and increased body motion. Activity is the most commonly employed rate indicator. The activity sensor is a piezoelectric crystal (i.e., vibration sensor) or accelerometer (i.e., acceleration sensor) that senses vibration from motion. When the activity sensor detects increased activity, the generator responds by increasing the pacing rate according to programmed parameters. When the activity sensor detects that activity has decreased, the generator is programmed to respond by lowering the rate to a preset level. Careful programming is a critical task to ensure that the rate responsive feature functions appropriately. To facilitate optimal programming, the software in newer generators allows for initial programming of the rate response along with subsequent automatic adjustments of the parameters. These generators also have the capability for physicians to retrieve data from the pacemaker to assess the adequacy/functioning of the rate responsive programming. In addition to a low rate limit and a low rate interval setting, a rate responsive pacemaker is programmed with a high rate limit setting. This high rate limit is the highest or fastest rate at which the pacemaker will pace the heart in response to data from the rate responsive sensor that an increase in heart rate is needed. In addition, the rate responsive feature is programmed with tiered rates correlated to activity levels. This often includes the resting rate and the average daily living rate. The rate responsive feature can be included in most types of demand pacemakers (single or dual chamber). Inclusion of the rate responsive feature involves additional costs, both for the sensor and for the programming. However, the benefits of rate responsive pacing as compared to nonrate responsive function include an improved ability to complete activities of daily living, provision of a heart rate that is appropriate (i.e., neither too fast nor too slow) for a given activity level, increased quality of life for the person who requires permanent pacing, and increased activity tolerance [18,22,23].
Pacemakers are now available with a programmable feature referred to as the "magnet mode." When programmed "on," the magnet mode allows the programmed functioning of the pacemaker to be altered temporarily by the direct, noninvasive application of a magnet over the pacemaker generator [15]. A pacemaker's specific response to the application of a magnet is established by the pacemaker manufacturer and will vary among different pacemaker types and brands. Generally, the expected response of a given pacemaker to application of a magnet should be known before the magnet is applied. Application of a magnet to a single chamber pacemaker always results in a change in pacing mode from demand to asynchronous pacing. In asynchronous mode, the sensing function is temporarily turned off, and the pacemaker generator is programmed to deliver a pacing impulse through the pacing lead at a regular interval and rate that do not vary. The term "magnet rate" is sometimes used to describe the rate at which the pacemaker paces the heart during the application of the magnet. The magnet rate may be used to evaluate the status of the pacemaker battery. As the battery voltage declines, the magnet rate will change. It is important to know the magnet rate for each specific pacemaker, as magnet rates vary greatly from manufacturer to manufacturer and even from one device to another produced by the same manufacturer [15,20,21].
Many types of permanent pacemakers are available. To facilitate and standardize communication about the types of pacemakers and pacemaker functions, a standardized coding system was developed through the work of the North American Society of Pacing and Electrophysiology and the British Pacing and Electrophysiology Group (NASPE/BPEG) [24].
The NASPE/BPEG system, commonly referred to as the NBG code, consists of up to five letter positions. The first three letter positions describe the antibradycardic functions of a pacemaker, the fourth addresses the rate responsive feature, and the fifth letter position designates the presence and location of multisite pacing [15,24,25].
In the clinical setting, the first three letters are commonly used alone to describe antibradycardia pacemakers. If the pacemaker has additional features, the fourth and fifth letters are added [15,24,25].
The first letter in the pacemaker code (i.e., the "first position") identifies which chamber(s) of the heart are paced by the pacemaker. "A" refers to atrium, "V" to ventricle, and "D" to both atrium and ventricle (or "dual") [15,25].
The second letter refers to the chamber in which, if any, the pacemaker can sense electrical activity. The same notation applies: "A" for atrial sensing, "V" for ventricular sensing, "D" for dual sensing in both atrium and ventricle, and "O" to indicate no sensing if the pacemaker does not sense in any chamber of the heart [15,25].
The third letter of the code describes the pacemaker's response to sensed electrical activity in the heart: "O" for no response, "I" for inhibited, "T" for triggered, and "D" for dual triggered and inhibited response. These modes of response will be explained in more detail in the sections on specific types of pacemakers [15,25].
The fourth letter is commonly used to indicate the presence of a rate modulation or rate responsive feature in the pacemaker. If the pacemaker is capable of increasing heart rate based on the body's metabolic demands, the fourth letter is coded "R" for rate responsive. If the pacemaker is not capable of increasing the paced heart rate in response to the body's demands, the fourth letter may be omitted or coded "O" [15,17,25].
In its 2002 update of the NBG code, the task force changed the fifth letter position to designate the location or the absence of multisite pacing. The notation is [15,24,25]:
"O" if the pacemaker does not have multisite pacing
"A" if there is: A pacing stimulation site present in both right and left atrium
More than one pacing stimulation site present in either the right or left atrium
Pacing stimulation sites present in both atria with more than one pacing stimulation site present in either the right or left atrium or both
"V" if there is: A pacing stimulation site present in both right and left ventricles
More than one pacing stimulation site present in either the right or left ventricle
Pacing stimulation sites present in both ventricles with more than one pacing stimulation site present in either the right or left ventricle or both
"D" if multisite pacing is present in both the atria and ventricles in any combination of A and V multi-site pacing
The NBG code will be used throughout the remainder of this course. Take a few minutes to readTable 1. Then, review the information by completing Practice Exercise 1.
TYPES OF PACEMAKERS
| Code | Type | Paces Atrium | Senses in Atrium | Paces Ventricle | Senses in Ventricle | Rate Responsive | Response to Sensed Electrical Activity |
|---|---|---|---|---|---|---|---|
| AAI | Single chamber atrial | YES | YES | NO | NO | NO | Sensed atrial electrical activity inhibits generator; no atrial pacing impulse is discharged. When the pacing lead senses no atrial electrical activity occurring within a programmed interval, the generator is not inhibited and a pacing impulse is delivered through pacing lead to right atrium. |
| AAIR | Single chamber atrial | YES | YES | NO | NO | YES | Sensed atrial electrical activity inhibits generator; no atrial pacing impulse is discharged. When the pacing lead senses the absence of electrical activity in atrium within the preset interval, the generator is not inhibited and a pacing impulse is delivered through pacing lead to right atrium. Responds to patient's activity. |
| AOO | Single chamber atrial | YES | NO | NO | NO | NO | Asynchronous mode is the generally programmed response to application of magnet to AAI/AAIR pacemaker. Asynchronous pacing is never used for permanent pacing. With asynchronous pacing, the device does not sense or respond to electrical activity in the atria. Continuously paces atria at fixed, preprogrammed rate. |
| VOO | Single chamber ventricular | NO | NO | YES | NO | NO | Asynchronous mode is generally the programmed response to application of magnet to VVI/VVIR pacemaker. Asynchronous pacing is never used for permanent pacing. With asynchronous pacing, the device cannot sense or respond to intrinsic electrical activity in the ventricles, but continues to pace ventricles at fixed, preprogrammed rate. |
| VVI | Single chamber ventricular | NO | NO | YES | YES | NO | Sensed electrical activity in ventricles inhibits generator; no ventricular pacing impulse is discharged. When the ventricular lead senses no electrical activity occurring in the ventricles within a programmed interval, the generator is not inhibited, and the pacemaker delivers a pacing impulse through the pacemaker lead. |
| VVIR | Single chamber ventricular | NO | NO | YES | YES | YES | Sensed electrical activity in ventricles inhibits generator; no ventricular pacing impulse is discharged. When the ventricular lead senses no electrical activity occurring in the ventricles within a programmed interval, the generator is not inhibited, and the pacemaker delivers a pacing impulse through the pacemaker lead. Responds to patient's activity. |
| DDD | Dual chamber | YES | YES | YES | YES | NO | Sensed electrical activity in the atria inhibits delivery of atrial pacing impulse. Absence of electrical activity in the atria triggers the generator to deliver an atrial pacing impulse. Sensed ventricular activity within the programmed AV interval inhibits delivery of a ventricular pacing impulse. Absence of electrical activity in the ventricles within the preprogrammed AV interval triggers delivery of a ventricular pacing impulse. |
| DDDR | Dual chamber | YES | YES | YES | YES | YES | Sensed electrical activity in the atria inhibits delivery of atrial pacing impulse. Absence of electrical activity in the atria triggers the generator to deliver an atrial pacing impulse. Sensed ventricular activity within the programmed AV interval inhibits delivery of a ventricular pacing impulse. Absence of electrical activity in the ventricles within the preprogrammed AV interval triggers delivery of a ventricular pacing impulse. |
| VDD | Modified: single chamber pacing; dual chamber sensing | NO | YES | YES | YES | NO | Sensed ventricular activity within the programmed AV interval inhibits delivery of a ventricular pacing impulse. Absence of electrical activity in the ventricles within the preprogrammed AV interval triggers delivery of a ventricular pacing impulse. |
A VVIR pacemaker can sense in which chamber(s) of the heart?
What does the "R" stand for in the code DDDR?
Can an AOO pacemaker sense in any chamber of the heart?
What chamber(s) of the heart can a DDD pacemaker pace?
What is the response of an AAI pacemaker to a sensed intrinsic electrical beat?
What chamber(s) can a VDD pacemaker pace? What chamber(s) can it sense?
What is the code for a pacemaker that paces and senses in both the atrium and in the ventricles, has dual triggered and inhibited response to sensed electrical activity, does not have a rate adaptive mechanism, and has pacing stimulation sites in both the right and left ventricles?
Does a pacemaker coded DDDOO have a rate responsive capability or multisite pacing capability?
A VVIR pacemaker senses electrical activity only in the ventricle.
An R in the fourth letter position indicates that the pacemaker has a rate responsive capability.
An AOO pacemaker is a fixed rate pacemaker; it cannot sense any intrinsic electrical activity.
A DDD pacemaker can pace the atrium, the ventricle, or both.
When an AAI pacemaker senses an intrinsic electrical beat in the atrium, it inhibits the generator from firing.
A VDD pacemaker can only pace in the ventricles; it can sense electrical activity in both the atrium and the ventricle.
This pacemaker would be coded DDDOV.
A DDDOO would not have a rate responsive or a multi-site pacing capability.
The most common types of antibradycardia pacemakers in use are atrial demand pacemakers, ventricular demand pacemakers, dual chamber pacemakers, and ventricular-paced/dual-sensing single chamber pacemakers.
Coded AAI or AAIR, a single chamber atrial pacemaker consists of a generator and one pacing/sensing lead. The lead is positioned in the right atrium. A single chamber atrial pacer is used when the SA node is not firing properly but the rest of the heart's conduction system is working correctly. Single chamber atrial demand pacemakers are capable of sensing and responding to electrical activity in the atrium. The single lead positioned in the atrium is capable of sensing the heart's electrical activity and delivering a pacing shock to the muscle of the right atrium to trigger depolarization. The AAI/AAIR pacemaker is programmed to sense when the heartbeat drops to less than the programmed low rate setting. When needed, the pacing lead delivers a pacing impulse to the right atrium; the right atrium depolarizes, and the wave of depolarization spreads to the left atrium and down to the AV node, just above the ventricles. The impulse is conducted normally through the AV node and the remainder of the conducting system; depolarization of the ventricles occurs, followed by normal contraction. When the AAI/AAIR pacemaker senses a normal atrial impulse initiated by the SA node, the generator is inhibited and refrains from delivering an atrial pacing impulse. Because the atrial demand pacemaker does sense electrical activity in the atria, it is not an appropriate choice to use for persons with chaotic atrial rhythms, such as atrial fibrillation and atrial flutter. Chaotic atrial activity confuses the pacemaker's sensor and results in either inappropriate pacing or inappropriate inhibition of pacing. A single chamber atrial pacemaker also cannot be used if the patient's AV node is functioning abnormally. The pacemaker will stimulate the atrium to depolarize; however, if there is impaired conduction in, around, or below the AV node, the impulse will not be conducted through the AV node and bundle branches. The ventricles will not depolarize, and no effective contraction will occur [17,18,22]. The atrial demand pacemaker may include the rate responsive feature.
In addition to the pacing and sensing thresholds, there are several key settings that must be programmed appropriately for an AAI pacemaker to work correctly. These include [20]:
Low rate limit setting
Timing interval for the low rate limit setting
Upper rate limit setting for rate adaptive response if rate responsive feature present
Normal AAI/AAIR pacing patterns
Normal pacing patterns that may be seen with an AAI pacemaker include:
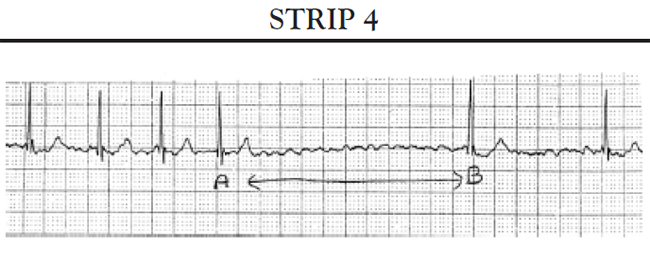
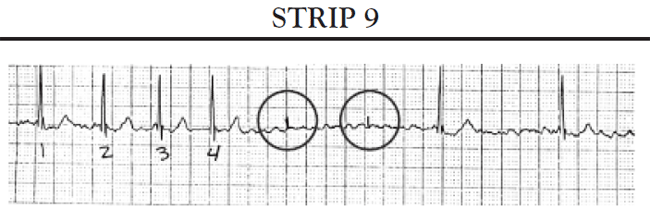
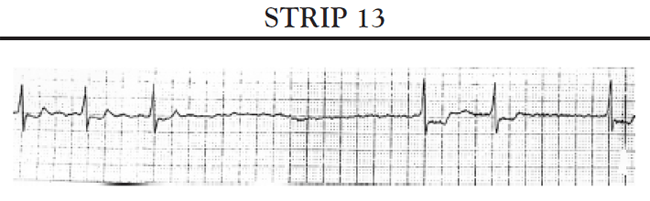
Absence of paced beats: The patient's ECG tracing reflects the patient's intrinsic rhythm. The patient's heart rate remains at or above the low rate setting.
Intermittent paced beats: The patient's ECG tracing shows a mixture of the patient's beats and paced beats. All paced beats come at the correct time, as determined by the low rate setting. No intervals (or pauses) occur that are longer than the low rate interval setting limit.
Completely paced (100%) rhythm: The ECG tracing shows that all beats are atrially paced. For an AAI pacemaker, the patient's heart rate remains at the low rate setting. For AAIR pacemakers, the paced rate may vary between the low rate setting and the upper rate limit for rate adaptation. The paced rate does not exceed the upper rate limit.
A single chamber ventricular pacemaker consists of a generator and a single pacing/sensing lead. The pacemaker lead is usually passed transvenously into and through the right atrium into the right ventricle, where it is attached to the wall of the right ventricle. VVI/VVIR pacemakers are capable of sensing and responding to electrical activity in the ventricles. The single lead positioned in the right ventricle is capable of sensing the heart's electrical activity and of delivering a pacing shock to the muscle of the right ventricle to trigger depolarization. The VVI/VVIR pacemaker is programmed to sense when the heartbeat drops to less than the programmed low rate setting. When needed, the pacing lead delivers a pacing impulse to the right ventricle; the right ventricle depolarizes, and the wave of depolarization spreads to the left ventricle. Depolarization of the ventricles is followed by normal contraction. When the VVI/VVIR pacemaker senses a normal ventricular depolarization impulse, the generator is inhibited and refrains from delivering a pacing impulse to the ventricles. Because a VVI/VVIR pacemaker neither paces nor senses in the atrium of the heart, there can be a loss of the normal AV synchrony. Remember that in normal conduction, the impulse coming from the SA node is slowed at the AV node to permit the atria to contract and complete ventricular filling. With a VVI/VVIR pacemaker, atrial contraction is not synchronized with ventricular contraction, and active ventricular filling may not occur. Cardiac output can drop. For persons with congestive heart failure, the loss of AV synchrony can increase signs of reduced cardiac output and heart failure. Because VVI/VVIR pacemakers do not sense or pace in the atrium, they are often the pacemaker of choice for persons with atrial fibrillation or atrial flutter (i.e., chaotic atrial arrhythmias), which could confuse the pacemaker. VVI/VVIR pacemakers are rarely used if the patient's SA node is functioning due to loss of AV synchrony. The rate responsive feature may be included in a ventricular demand pacemaker [3,4,22].
In addition to the pacing and sensing thresholds, programmable settings for VVI/VVIR pacemaker include [20]:
Low rate setting
Timing interval for the low rate setting
Upper rate limit for rate adaptation if rate responsive feature is present
Normal pacing patterns that the patient may have with VVI/VVIR pacemaker include:
Total absence of paced beats: The ECG tracing reflects the patient's intrinsic rhythm. The patient's heart rate remains at or above the low rate limit setting of the pacemaker.
Intermittent paced beats: The patient's ECG tracing shows a mixture of the patient's beats and paced beats. All paced beats come at the correct time (as determined by the low rate setting). No pauses occur that are longer than the low rate interval setting limit.
Completely paced (100%) rhythm: The ECG tracing shows that all beats are ventricularly paced. For a VVI pacemaker, the patient's heart rate remains at the low rate setting. For VVIR pacemakers, the paced rate may vary between the low rate setting and the upper rate limit for rate adaptation. The paced rate does not exceed the upper rate limit.
Dual chamber pacemakers are capable of pacing and sensing in both the atria and the ventricles. Designed to more closely mimic the normal electrical activity of the heart, the dual chamber pacemaker preserves the synchrony between atrial and ventricular depolarization, permitting the atria to contract and finish ventricular filling before the ventricles contract. Coded DDD, the dual chamber pacemaker consists of a generator and two pacing leads. The electronic circuitry in the generator is more complex than the circuitry in a single chamber pacemaker. Two complete sets of circuitry are included: one for pacing and sensing in the atria and one for pacing and sensing in the ventricles. One pacing lead is positioned in the right atrium, and one in the right ventricle. The functioning of the DDD pacemaker is also more complex. The normal sequence of events in dual chamber pacing is:
The sensing electrode in the atrium monitors for normal atrial electrical activity.
If it senses normal electrical activity, it relays that information to the generator. The atrial pacing circuit is inhibited, and the pacemaker does not deliver a pacing impulse through the atrial pacing lead.
If the atrial sensing electrode does not sense normal electrical activity within the preprogrammed interval, the generator delivers a pacing shock through the atrial pacing lead to the right atrium to stimulate depolarization.
Once the atrium has been depolarized by a pacing impulse or a normal SA node firing, the sensing electrode in the ventricle monitors for depolarization of the ventricles within a preset interval. This interval is known as the AV interval and corresponds approximately to the P-R interval on a normal ECG tracing.
If the ventricles spontaneously depolarize within the preset AV interval, the ventricular sensing electrode relays that information back to the pacemaker generator; the pacemaker generator is inhibited and does not deliver a pacing impulse to the right ventricle by way of the ventricular pacing lead.
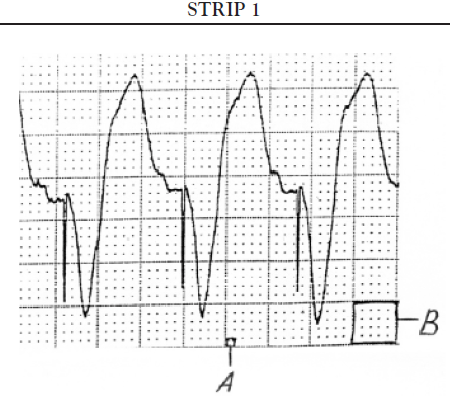
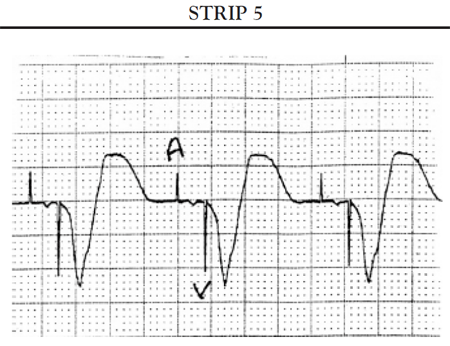
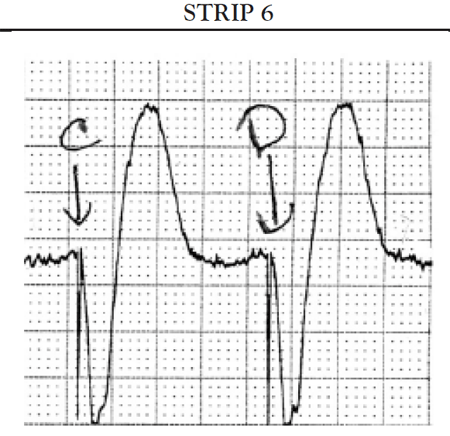
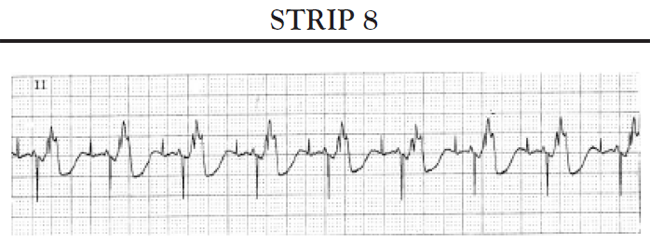
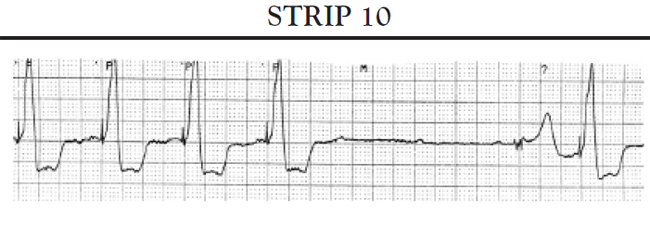
If the ventricles do not spontaneously depolarize within the preset AV interval, the pacemaker generator is triggered to deliver a pacing impulse through the ventricular pacing lead to the right ventricle to initiate timely depolarization of the ventricles. The ECG tracing shows a pacer spike in front of the QRS complex.
If the patient's SA node is functioning properly, a DDD pacemaker has the capability to track the SA node firing and pace the ventricles at the same rate. This capability allows the pacemaker to increase or decrease the ventricular pacing rate in response to changes in the SA node firing rate. For persons whose SA node is not working properly, a DDD pacemaker may come with the rate responsive feature. The rate responsive version is capable of increasing the atrial-pacing rate when indicated.
Dual chamber pacemakers are more expensive to implant than single chamber pacemakers. The additional cost is related to the need for a second pacing lead and the additional time needed to program the circuitry properly. However, it provides the most flexible options and has become the most common type of pacemaker implanted. Dual chamber pacemakers have several advantages as compared to single chamber pacemakers [26]:
As discussed earlier, the DDD pacemaker permits sequential pacing of the atria and ventricles, thereby preserving AV synchrony.
For persons whose SA nodes are intact, the DDD pacemaker permits the pacemaker to track the patient's intrinsic SA node firing and pace the ventricles in response. This permits a "natural" rate responsive feature very similar to the heart's normal conducting system.
As DDD pacemakers track electrical activity in the atria, they traditionally had not been implanted in persons with chronic chaotic atrial rhythms, such as atrial fibrillation or atrial flutter. Because the pacemaker can track atrial activity and pace the ventricles in response, the chaotic atrial activity may confuse the pacemaker or lead to inappropriately high ventricular responses. However, newer DDD pacemakers have an additional feature that may be programmed. Called the mode-switching feature, this programming aspect automatically causes the pacemaker to revert to a VVI mode when the circuitry recognizes the presence of atrial fibrillation or flutter. When atrial rhythm returns to normal, the programming automatically returns the pacemaker to DDD mode. The automatic mode switching function makes the DDD pacemaker a useful pacemaker for persons who have paroxysmal, infrequent episodes of atrial fibrillation.
Normal pacing patterns seen with a DDD pacemaker include:
Total absence of paced beats: The patient's heart rate remains at or above the low atrial rate limit setting.
Intermittent paced beats: The patient's ECG tracing shows a combination of paced beats and intrinsic beats. The precise configuration of the paced beats may vary depending on what chamber of the heart is being paced. The patient's heart rate remains at or above the low rate limit setting. No pauses occur that exceed the timing interval for the low rate limit setting.
Completely paced (100%) rhythm: The ECG shows all beats paced; the precise configuration of the paced rhythm may vary depending on which chamber(s) are paced. The heart rate should not drop to less than the low atrial rate limit setting. If the patient's SA node is functioning, the heart rate may vary between the low and upper rate limit settings. If the rate adaptation is present, the rate may vary between the low rate limit setting and upper limit for rate adaptation.
Settings that are programmed for the DDD/DDDR pacemaker include [4,17,18,22]:
Low rate setting for atrial pacing
Low rate setting interval (i.e., length of time permitted between one atrial depolarization and the next)
The AV interval (i.e., length of time permitted between atrial depolarization and initiation of ventricular depolarization)
Upper rate limit setting for ventricular tracking of SA node firing
If rate adaptive feature is present, high rate limit for rate responsive pacing
The single-lead, atrial-sensing ventricular pacemaker is a relatively recent development in pacemaker technology. Coded VDD, it is capable of pacing the ventricles only, but it can sense electrical activity in both the atrium and the ventricle. Like other single chamber pacemakers, it consists of a generator and one pacing/sensing pacemaker lead. It is appropriate for use in people whose sinus node is intact and who do not have atrial hypertrophy. Atrial hypertrophy is a significant risk factor for the eventual development of atrial fibrillation or atrial flutter [27]. A VDD pacemaker provides AV synchrony. The tip of the pacing lead is positioned in the right ventricle; it has the ability to sense electrical activity in the ventricle and to deliver a pacing impulse to the ventricular wall. The pacing lead also contains a pair of electrodes that are capable only of sensing electrical activity. When the tip of the pacing lead is properly positioned in the right ventricle, these sensing electrodes fall within the right atrium. They sense atrial depolarization but cannot fire a pacing impulse to the atrium. The response of the VDD to sensed electrical events is both triggered and inhibited. When the sensing electrodes in the atrium sense atrial depolarization, they relay that information to the generator. The generator is programmed to wait a preprogrammed interval to determine if the atrial impulse triggers ventricular response. If the sensing electrode in the ventricle does not pick up a ventricular response within the defined interval, the generator triggers the discharge of a pacing impulse back through the pacing lead to the right ventricle. If the sensing electrode in the ventricle picks up a ventricular response to the atrial impulse, the generator is inhibited and no pacing impulse is fired. A VDD pacemaker provides for a physiologic rate increase. Because it senses in the atrium, it can increase the rate of ventricular pacing as needed to keep up with an increase in SA node depolarizations. If the patient's own SA node firing fails to occur, the VDD pacemaker will pace the ventricles at the low rate setting. Because it only contains a single lead, it is easier to implant than a dual chamber device. VDD pacemakers do not come with a rate responsive feature. Because the VDD pacemaker does sense and respond to atrial activity, it is not an appropriate pacemaker to implant for a patient with chaotic atrial electrical activity, such as atrial fibrillation or atrial flutter.
Normal pacing patterns seen with a VDD pacemaker include:
Total absence of paced beats: The patient's ECG reflects the patient's intrinsic heart rhythm, and the patient's heart rate remains at or above the pacemaker's low rate setting.
Intermittent paced beats: The patient's ECG tracing reflects a combination of paced beats and intrinsic rhythm. No pause occurs that lasts longer than the programmed timing interval set determined by the low rate limit setting. The patient's heart rate remains at or above the low rate setting.
Complete paced (100%) rhythm: The ECG tracing reflects all paced beats. The heart rate may vary but should not drop to less than the low rate limit setting. The heart rate should not exceed the upper rate limit.
Programmed settings for a VDD pacemaker include [4,20,22]:
Low rate setting for ventricular pacing
Timing interval for the low rate setting
AV interval (i.e., length of time permitted between atrial depolarization and initiation of ventricular depolarization)
Upper rate limit setting for ventricular tracking of patient's SA node firing
In 2012, the American Heart Association (AHA) and the American College of Cardiology Foundation (ACCF) published guidelines for the implantation of cardiac pacemakers [22]. The guidelines were updated in 2018 [28]. Both sets of guidelines are based on a review of clinical findings from large, multicenter randomized clinical trials; well-designed analysis of data from smaller, nonrandomized studies or observational data registries; and the consensus opinion of experts. The 2012 guidelines focused on the medical indications for pacing and selection of the appropriate type of pacemaker. The focus of the 2018 guidelines is providing guidance to clinicians for the management of patients with bradycardia, which includes recommendations for the indications for both temporary and permanent pacing [28]. The task force that developed the guidelines separated the recommendations into categories based on the strength of the supporting evidence available. In brief, indications listed as Class I are supported by evidence or general agreement that the use of pacemaker therapy for these patients is beneficial, useful, and effective. Class II indications are those that have conflicting evidence and/or a difference in expert opinion about the usefulness and efficacy of permanent pacemaker therapy. Class III lists conditions that, according to available evidence and/or expert opinion, would not benefit from implantation of a permanent pacemaker. In 2014, the ACCF, AHA, and Heart Rhythm Society (HRS) issued a consensus statement about the appropriateness of implantable cardiac pacemakers for specific subsets of patients not included in or represented by clinical trials and who, therefore, are not covered by formal practice guidelines [29].
The most common Class I indication for implantation of a permanent pacemaker is symptomatic bradycardia. Symptomatic bradycardia is defined as a documented bradyarrhythmia that significantly reduces cerebral flow and results in symptoms of syncope, near syncope, light-headedness, dizziness, or confusion [28]. Patients with symptomatic bradycardia are often diagnosed after they seek medical attention following a fall, "black-out" spell, or recurrent episodes of dizziness or near fainting [4,22].
Consider these typical patient examples:
Patient G, a man 71 years of age, is found unconscious in his bathroom by his daughter. He is bleeding from a scrape/abrasion on his forehead. His daughter calls 911, and Patient G is transported via emergency medical support (EMS) to the local hospital emergency department. On arrival in the emergency department, Patient G is awake, alert, and oriented. His blood pressure is slightly elevated, but his heart rate and respirations are within normal limits. A blood sugar analysis shows his blood sugar is within normal limits. He is admitted to the inpatient unit for work-up of a syncopal episode.
Patient H, a woman 68 years of age, presents to her primary care physician for a routine check-up. She admits to having spells of feeling very dizzy and "almost fainting." She tells the physician that these spells are coming more and more often.
Patient I, a man 54 years of age, goes to see his physician with complaints of palpitations and a pounding sensation in his chest that occur intermittently. He also complains of periods of dizziness and light-headedness and reports "almost passing out" while working in his yard earlier that week. Patient I's vital signs are normal. However, due to his symptoms and near syncopal episode, a 24-hour Holter monitor is ordered to evaluate his cardiac rhythm. Data from the Holter monitoring shows that Patient I has episodes of atrial tachycardia at a rate of 180 bpm that alternate with periods of extreme bradycardia. The patient's history shows that he has experienced symptoms of near syncope and light-headedness during both the tachycardic and bradycardic episodes. He is admitted to the local hospital for evaluation and treatment.
EMS brings Patient J, a woman 78 years of age, to the emergency department. The patient had been out for dinner with her family in a local restaurant when she lost consciousness. When EMS arrived, she had a palpable pulse with a rate of 30 bpm, a blood pressure of 85/32 mm Hg, and was breathing spontaneously. She returned to consciousness en route to the emergency department but was "groggy." Patient J is admitted to the hospital for evaluation of her syncopal episode.
EMS is summoned to the home of Patient K, a man 82 years of age, by the patient's daughter, who lives out of state. She had called Patient K; when he answered the telephone, he was very confused and not completely coherent. He did not seem to know where he was, what day it was, and did not recognize his daughter's voice. The daughter contacted EMS who transported Patient K to the emergency department. On arrival to the emergency department, ECG monitoring showed that Patient K was in a ventricular escape rhythm at a rate of approximately 30 bpm.
As noted, the major signs and symptoms of symptomatic bradycardia are syncope, near syncope, confusion, light-headedness, and/or dizziness during a bradyarrhythmia. In addition to the major definitive symptoms of cerebral hypoperfusion, persons with symptomatic bradycardia may also experience hypotension, fatigue, signs of congestive heart failure, shortness of breath, and exercise intolerance. Specific diagnosis of symptomatic bradycardia can be challenging. The bradyarrhythmia and the symptoms should occur simultaneously, and their simultaneous occurrence should be clearly documented through appropriate diagnostic tests [3].
Symptomatic bradycardia may be caused by a wide range of conduction disorders involving the SA node, the conduction of the impulse from the SA node through the atria, the conduction of the impulse through the various areas of the AV node, or the conduction of the impulse through the bundle of His and the bundle branches. The major arrhythmias responsible for symptomatic bradycardia are sick sinus syndrome and second- and third-degree AV block.
Sinus node dysfunction occurs when the SA node is unable to pace the heart normally. Referred to as sick sinus syndrome, sinus node dysfunction encompasses a group of disorders that include [30]:
Chronotropic incompetence, or the inability of the sinus node to increase the rate of firing in response to increased metabolic demands or activity
Reduced ability of the SA node to generate an action potential and depolarize
Slowed or blocked conduction of the electrical impulse from the SA node to the specialized conducting pathways in the atria
Slowed or blocked conduction of the sinus impulse through the atria to the AV node
Signs and symptoms of sinus node dysfunction include [30,31]:
Sinus bradycardia: Sinus bradycardia in sinus node dysfunction can become quite severe; the heart rate may drop to less than 40–50 bpm and create symptoms of decreased cardiac output and decreased cerebral perfusion. Sinus bradycardia in sick sinus syndrome is refractory to any kind of medication therapy.
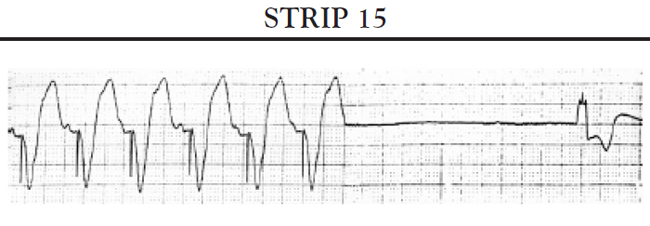
Tachy-brady syndrome: In addition to sinus bradycardia, persons with sinus node dysfunction syndrome often experience periods of atrial tachycardia that alternate with periods of profound bradycardia. Persons with tachy-brady syndrome experience signs and symptoms of decreased cardiac output during both the episodes of tachycardia and the bradycardic periods. The most common cause of the tachycardia in sinus node dysfunction is atrial fibrillation with a rapid ventricular response. The heart rate may range as high as 160 bpm during tachycardic episodes and drop to less than 40 bpm during bradycardic periods.
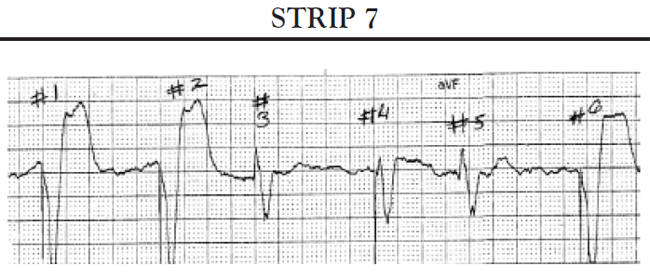
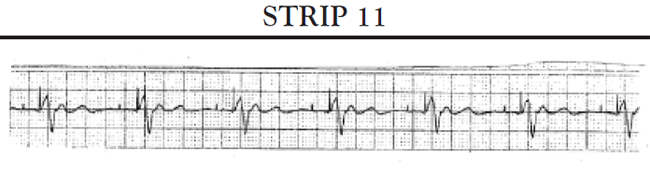
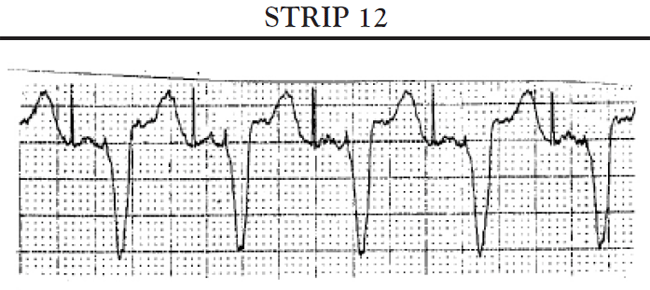
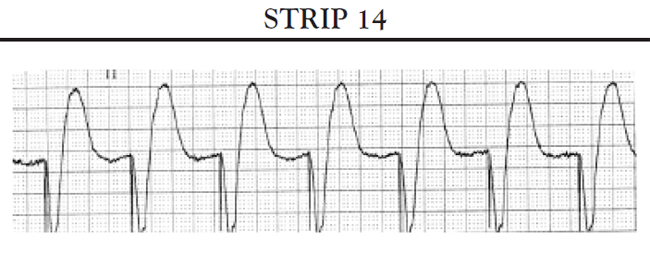
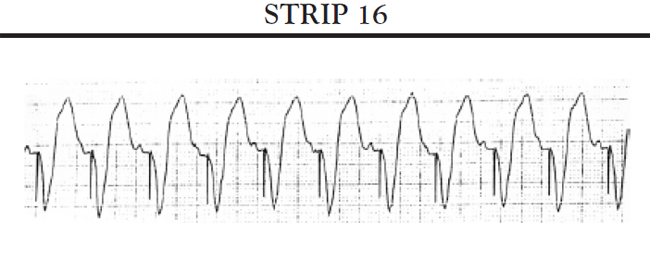
Sinus pauses: A sinus pause occurs when the sinus node fails to depolarize (sinus arrest) or when the impulse from the SA node fails to activate the atrial conduction system so that the impulse does not depolarize the atria and is not conducted to the AV node (SA block). Short pauses of less than three seconds rarely cause symptoms; however, longer pauses can cause symptoms associated with decreased cardiac output and reduced cerebral blood flow. If the patient's SA node stops functioning altogether, the patient may develop an escape rhythm in an attempt to maintain cardiac output. If conduction both at the level of the AV node and below is normal, the patient will develop a junctional escape rhythm; the AV node acts as the pacemaker for the heart and paces the heart at approximately 60 bpm. If, in addition to sick sinus syndrome, the patient also has some conduction abnormalities of the AV node, the patient may develop an escape rhythm that is paced by a site somewhere lower in the ventricles. This is called a ventricular escape rhythm; it is characterized by a rate of less than 40 bpm and a widened, bizarre-appearing QRS complex.
Associated clinical signs and symptoms include [22,27,31,32,33]:
Palpitations, angina, new or worsening congestive heart failure, muscle aches, and disturbed sleep, in addition to the symptoms frequently associated with symptomatic bradycardia (i.e., syncope, heart rate <60 bpm, dizziness, light-headedness, and confusion)
Failure to increase heart rate during exertion or stress
A heart rate that either stays relatively fixed or fails to rise during an exercise treadmill test
Failure of the sinus node to respond to the administration of intravenous atropine. With normal sinus node function, a person's heart rate should increase to approximately 100 bpm following the administration of atropine. With sinus node dysfunction, the sinus node does not respond to the administration of atropine; the heart rate rarely increases to greater than 90 bpm.
Underlying Causes
One of the most common causes of sinus node dysfunction is the normal aging process. As a person ages, a decrease occurs in the number of specialized cells that are present in the SA node and able to depolarize spontaneously and initiate a heartbeat. At the same time, an increase occurs in the amount of collagen tissue that is unable to generate or conduct an electrical impulse in the SA node. As a result, the ability of the SA node to act effectively as the pacemaker of the heart decreases. Another major cause of sinus node dysfunction is inferior wall MI. Cardiomyopathy, a condition in which the heart's ability to pump blood adequately is impaired, may also lead to sinus node dysfunction. Other cardiovascular diseases associated with sinus node dysfunction include hypertension, valvular heart disease, and coronary artery disease. Noncardiac causes of sinus node dysfunction include hypothyroidism, sleep apnea, and electrolyte imbalances. More rarely, conditions such as muscular dystrophy and amyloidosis may cause sinus node dysfunction [3,31].
Class I Indications for Pacemaker Implantation in Sinus Node Dysfunction
For persons with sinus node dysfunction, class I indications for permanent pacemaker implantation according to the updated ACCF/AHA guidelines include [28]:
Sinus node dysfunction with symptomatic bradycardia
Symptomatic sinus bradycardia resulting from required drug therapy for medical conditions
Symptomatic chronotropic incompetence with sinus node dysfunction was previously considered a class I indication but was downgraded to class II in the 2018 guidelines update [28].
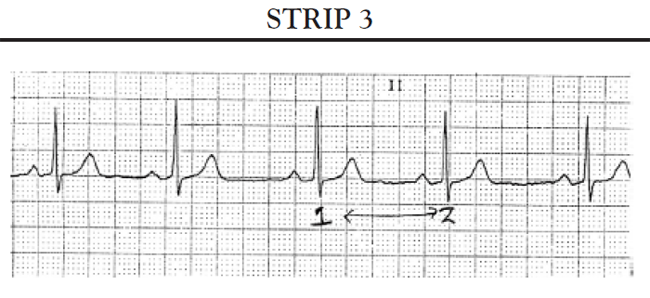
Another common cause of symptomatic bradycardia is third-degree (or complete) AV block. Third-degree AV block occurs when none of the impulses generated by the SA node are conducted through the AV node. Sinus node function is often normal; however, conduction abnormalities at the level of AV junction or below the AV junction in the bundle of His/bundle branch region prevent the sinus impulse from reaching the ventricles [34]. The ventricles are paced by an escape rhythm. The type and effectiveness of the escape rhythm depends on the location of the conduction abnormality. If the problem occurs high in the AV node, a junctional escape rhythm may pace the ventricles. Junctional escape rhythms are characterized by a normally configured QRS complex and a heart rate of 45–60 bpm [34]. Junctional escape rhythms are often effective in maintaining an adequate cardiac output. If the conduction abnormality occurs lower in the ventricular conducting system (i.e., at the level of the bundle branches or below), a ventricular escape rhythm may emerge as the pacemaker of the heart. Ventricular escape rhythms are characterized by a widened, bizarre-appearing QRS complex and a heart rate of 30–40 bpm [34]. Persons frequently develop symptoms of decreased cardiac output and diminished cerebral blood flow with a ventricular escape rhythm. The defining ECG characteristics of complete AV block include [34]
A regular atrial rate, indicated by a P wave to P wave interval that is constant (or almost constant) when measured
A regular ventricular rate, as evidenced by a constant (or almost constant) R to R interval
A complete absence of relationship between P waves and QRS complexes
In addition to identifiable ECG changes, signs and symptoms associated with complete heart block include [34]:
Exacerbation of underlying cardiovascular problems, including increased angina and increased signs of congestive heart failure
Activity intolerance
Shortness of breath
Signs associated with symptomatic bradycardia, including syncope, near syncope, light-headedness, and confusion
Underlying Causes
A major cause of complete AV block is MI. A branch of the right coronary artery usually provides the blood supply for the AV node and the bundle of His. An inferior wall MI can result in damage and impaired conduction through the node and the bundle of His. A branch of the left anterior descending (LAD) artery provides blood supply to the right and left bundle branches; an anterior wall MI may result in blocked conduction through this part of the ventricular conducting system. The development of chronic AV block has been linked with other cardiac causes, including valvular heart disease, congenital heart disease, coronary artery disease, myocarditis, and cardiomyopathies. Complete heart block may develop following cardiac surgery. Antiarrhythmic medications, such as digoxin, calcium channel blockers, beta blockers, and amiodarone, may also cause complete heart block. Older adults have an increased risk of developing heart block. Although the AV node is relatively unaffected by the aging process, both the bundle of His and the bundle branches gradually lose the ability to conduct impulses effectively as a person ages. In addition, elderly individuals are often more susceptible to the effects of antiarrhythmic medications and other medications known to influence cardiac rhythm. Finally, complete heart block may be caused by radiofrequency ablation (RFA). RFA of the AV node is an interventional procedure performed to manage a variety of tachyarrhythmias that are refractory to pharmacologic therapy. With RFA, the conducting cells in the AV node are destroyed and complete heart block results [3,34,35].
Class I Indications
According to the 2018 ACCF/AHA guidelines, the indications for permanent pacemaker implantation for the person with third-degree heart block include [28]:
Acquired second-degree Mobitz type II AV block, high-grade atrioventricular AV block, or third-degree atrioventricular AV block not attributable to reversible or physiologic causes, regardless of symptoms
Complete heart block associated with permanent atrial fibrillation and symptomatic bradycardia
Complete heart block caused by the necessary use of medications required to manage other arrhythmias or medical conditions
Heart block caused by neuromuscular diseases, such as myotonic muscular dystrophy
Second-degree heart block occurs when impulses from the SA node are intermittently blocked at the level of the AV node or below. Some atrial impulses are conducted, and some are not. There are two types of second-degree heart block: Mobitz type I and Mobitz type II. Mobitz type I, also known as Wenckebach, is characterized by a P-R interval that gradually lengthens until a P wave is blocked. The pattern of gradual lengthening of the P-R interval then repeats. Mobitz type I is associated with inferior wall MIs, digitalis toxicity, and administration of calcium channel blockers or beta blockers. It may also occur for a period of time following cardiac surgery. Mobitz type II is characterized by an intermittent block of the atrial impulse; however, unlike the pattern seen in Mobitz type I, the P-R interval for conducted beats remains the same. In Mobitz type II, the conduction block occurs in or below the bundle of His. It is associated with anterior wall MIs. When it occurs after an MI, it is often an indication of a poor long-term prognosis for the patient. Mobitz type II may progress to the development of complete heart block [35,36].
Signs and Symptoms
Clinical signs and symptoms depend in large part on how often the atrial impulses fail to be conducted to the ventricles. Blocked P waves that occur every second or third beat can result in a severe bradycardia, with resulting symptoms of decreased cardiac output and diminished cerebral circulation [35,36].
Class I Indications
According to the 2018 ACCF/AHA guidelines, patients with transient or reversible causes of AV block (e.g., Lyme carditis, drug toxicity) should have medical therapy and supportive care, including temporary transvenous pacing if necessary, before determination of the need for permanent pacing [28]. Permanent pacing is a class II recommendation [28].
Atrial fibrillation is an atrial arrhythmia characterized by an absence of normal P waves and an irregularly irregular ventricular response. In atrial fibrillation, the heartbeat is not initiated by the depolarization of the SA node. Instead, chaotic electrical activity is present in the atria. This chaotic electrical activity is reflected by fibrillation waves on the ECG tracing; they occur at excess of 400 bpm and are ineffective in depolarizing the atria. Some fibrillation waves are conducted to the AV node; some are conducted through the AV node, while others are blocked. Atrial fibrillation with slow ventricular response is defined as atrial fibrillation with an average ventricular response of less than 60 bpm. It may be induced by medications (e.g., antiarrhythmics) used to control tachyarrhythmia as well as catecholamine excess, hemodynamic stress, atrial ischemia, atrial inflammation, metabolic stress, and neurohumoral cascade activation [37]. Atrial fibrillation is present in 30% to 50% of patients undergoing valve surgery and is associated with reduced survival and increased risk of stroke [28]. Successful surgical correction of atrial fibrillation is associated with improved patient survival compared with patients who have recurrent atrial fibrillation. The 2018 ACCF/AHA guidelines address pacing after surgery for atrial fibrillation [28].
According to the 2018 ACCF/AHA guidelines, atrial fibrillation is an indication for permanent pacemaker implantation in patients who have new postoperative sinus node dysfunction, or AV block associated with symptoms, or hemodynamic instability that does not resolve after surgery for atrial fibrillation [28]. Permanent pacing is not indicated for the prevention of atrial fibrillation in patients without any other indication for implantation [28].
Hypersensitive carotid sinus syndrome is a noncardiovascular, neurogenic cause of syncope. It is an extreme reflex that occurs in response to carotid sinus baroreceptor stimulation. To understand the pathophysiology of the syndrome, it is important to understand the normal physiology related to carotid stimulation. Normally, when pressure is exerted on the carotid sinus, the baroreceptors in the wall of the carotid sinus are stimulated. Stimulation of the baroreceptors results in vagal stimulation and a subsequent slowing of the heart. This response is normal; however, some individuals may have an exaggerated response. There are three types of hypersensitive carotid sinus syndrome. The first, called the cardio-inhibitor reflex, comprises 70% to 75% of cases and results from increased parasympathetic tone and decreased sympathetic tone; the person may experience significant bradycardia and hypotension. The second type, vasodepressor carotid hypotension, comprises 5% to 10% of cases and is characterized by reduced peripheral vascular resistance caused by a decrease in sympathetic tone, resulting in hypotension. The heart rate may or may not drop. The third type is a mix of the first two and comprises 20% to 25% of cases [38]. Patients with the mixed type may experience hypotension caused both by a drop in peripheral vascular resistance and by significant bradycardia. Only the first and third types of hypersensitive carotid sinus syndrome may effectively be managed with the implantation of a permanent pacemaker. A group of international experts suggested that the classification of hypersensitive carotid sinus syndrome into three types should be revised. They recommend that all patients with the syndrome should be classified as "mixed" between vasodepression and cardio-inhibition because isolated cardio-inhibition does not occur [39]. Aging is a major risk factor for the development of hypersensitive carotid sinus syndrome [19,20,21,22,38].
Clinical signs and symptoms indicative of hypersensitive carotid sinus syndrome include syncope, blurred vision, and lightheadedness or confusion when the patient sits or stands. These symptoms may be most pronounced during movement of the patient's head or neck. Some persons may be able to identify factors that trigger the onset of an episode, including turning the head, wearing tight clothing around the neck, shaving, and hyperextending the neck. Other patients are unable to identify any specific precipitating event. Syncopal episodes are transient, lasting only several minutes. A basic screening measure for hypersensitive carotid sinus syndrome is carotid sinus massage, which may cause/precipitate the patient's symptoms. More definitive testing is done using a tilt-table test [19,20,21,22,38]. The 2018 ACCF/AHA guidelines contain no class I indications for permanent pacing in persons with carotid sinus hypersensitivity syndrome [28].
Antiarrhythmic medications are commonly prescribed for the management of a wide variety of tachyarrhythmias, including atrial fibrillation, atrial flutter, tachy-brady syndrome associated with sinus node dysfunction, and ventricular ectopy. However, antiarrhythmic medications, even when used in appropriate dosage, may cause additional conduction abnormalities. When an antiarrhythmic medication causes new or worsened arrhythmias, it is said to be "proarrhythmic." Proarrhythmic abnormalities such as sinus node dysfunction or AV block may occur. These abnormalities may result in symptomatic bradycardia [19].
Examples of medications commonly associated with the development of conduction abnormalities are outlined in Table 2. When possible, drug-induced arrhythmias are treated by changing the dosage and/or type of antiarrhythmic medication prescribed. In some cases, dosage reduction or a switch to another medication corrects the conduction abnormality. In other cases, however, it is not possible to find an antiarrhythmic agent (or combination of agents) that is effective in managing the patient's arrhythmia and does not cause additional conduction problems and significant bradycardia. Use of a permanent pacemaker is often indicated in these patients to counteract the bradycardia caused by the antiarrhythmic therapy. Other noncardiovascular medications, especially when given in conjunction with cardiovascular medications, may cause arrhythmias. Cardiovascular medications given in management of coronary artery disease or hypertension may also have proarrhythmic side effects [19,40].
MEDICATIONS THAT CAN CAUSE CONDUCTION ABNORMALITIES
| Medication | Indications | |||
|---|---|---|---|---|
| Digoxin |
| |||
| Metoprolol |
| |||
| Propranolol |
| |||
| Atenolol |
| |||
| Verapamil |
| |||
| Diltiazem |
| |||
| Disopyramide | Control of ventricular arrhythmias, ventricular tachycardia | |||
| Quinidine |
| |||
| Flecainide |
| |||
| Amiodarone |
| |||
| Sotalol | Ventricular arrhythmias | |||
| Amitriptyline | Depression | |||
| Doxepin | Depression | |||
| Imipramine | Depression | |||
| Chlorpromazine |
| |||
| Prochlorperazine |
| |||
| Lithium | Bipolar disorders |
Indications for permanent pacemaker implantation for the management of persons who have drug-induced conduction abnormalities include sinus node dysfunction with symptomatic bradycardia induced by necessary long-term pharmacologic therapy for which there is no acceptable alternative [28].
As pacemaker technology continues to advance, and as medical research continues to refine knowledge of the link between normal cardiac conduction and the successful management of cardiovascular disease and its symptoms, the indications for permanent pacemaker implantation will also continue to advance. The ACCF/AHA note that there are certain conditions that permanent pacemaker implantation may benefit; however, the guidelines caution that expert opinion and strength of clinical evidence to support the use of permanent pacemakers in the management of these conditions are controversial. Medical conditions that may benefit from permanent pacemaker implantation include [28]:
Tachy-brady syndrome and symptoms attributable to bradycardia
Symptomatic chronotropic incompetence
Symptomatic second- or third-degree AV block in patients on chronic stable doses of medically necessary antiarrhythmic or beta-blocker therapy
Second- or third-degree AV block associated with cardiac sarcoidosis (in patients with expected meaningful survival longer than one year)
Exertional symptoms (e.g., chest pain, shortness of breath) and first- or second-degree Mobitz type I AV block at rest; an exercise treadmill test is reasonable to determine whether the patient may benefit from permanent pacing
An infiltrative cardiomyopathy (e.g., cardiac sarcoidosis, amyloidosis) and second-degree Mobitz type II AV block, high-grade AV block, or third-degree AV block (in patients with expected meaningful survival longer than one year)
In patients with lamin A/C gene mutations, with a PR interval greater than 240 ms and left bundle branch block, permanent pacing (with additional defibrillator capability if needed and meaningful survival longer than one year is expected), permanent pacing is reasonable
In patients with marked first- or second-degree Mobitz type I (Wenckebach) AV block with symptoms that are clearly attributable to the AV block, permanent pacing is reasonable
Neuromuscular diseases (e.g., myotonic dystrophy type 1), with a PR interval greater than 240 ms, a QRS duration greater than 120 ms, or fascicular block (in patients with expected meaningful survival of longer than one year)
Kearns-Sayre syndrome and conduction disorders (in patients with expected meaningful survival of longer than one year)
Anderson-Fabry disease and QRS prolongation greater than 110 ms (in patients with expected meaningful survival of longer than one year)
New persistent left bundle branch block after transcatheter aortic valve replacement
Asymptomatic adults with congenital complete AV block
Epilepsy associated with severe symptomatic bradycardia where antiepileptic medications are ineffective
The 2018 ACCF/AHA guidelines note that implantation of a permanent pacemaker is not indicated for the management of some medical conditions. Conditions for which pacemaker therapy has not been found to be beneficial, useful, or effective, and which may cause harm include [28]:
Asymptomatic sinus bradycardia or sinus pauses secondary to physiologically elevated parasympathetic tone
Sleep-related sinus bradycardia or transient sinus pauses that occur during sleep
Asymptomatic sinus node dysfunction
Symptomatic or asymptomatic first-degree AV block or second-degree Mobitz type I believed to be at the level of the AV node
Acute AV block attributable to a known, reversible, and nonrecurrent cause, with complete resolution of the AV block and treatment of underlying cause
Asymptomatic vagally-mediated AV block
Asymptomatic patients with isolated conduction disease and 1:1 AV conduction
Acute MI and transient AV block that resolves
Acute MI and new bundle-branch block or isolated fascicular block in the absence of second- or third-degree AV block
Patients with indications for permanent pacing but also with significant comorbidities that make pacing therapy unlikely to provide meaningful clinical benefit, or if patients goals of care strongly preclude pacemaker therapy
The decision to implant a permanent pacemaker and the selection of the specific type of pacemaker device are based on a thorough assessment and evaluation of the patient's status. Two key components of the evaluation process are the patient history and selected diagnostic tests.
Thorough assessment of the patient's symptoms is necessary to determine if the symptoms are correlated with the occurrence of the arrhythmia. Begin by identifying the primary reason that the patient has sought medical care. Listen for a history of a recent fall, recurrent unexplained falls, fainting spells, or periods of "black out." Ask the patient's family if he or she has had any unexplained periods of confusion. Determine when the symptom occurred, what the person was doing, what (if anything) appeared to trigger the event, how long it lasted, and what (if anything) relieved the symptom. Also question the patient about any associated symptoms, including increased fatigue with no change in daily activities, shortness of breath, activity intolerance, or other signs of congestive heart failure. Determine how often symptoms occur: daily, weekly, or less frequently. Also obtain data about the patient's "usual" lifestyle; inquire about the patient's normal activity level, exercise tolerance, and ability to perform normal activities of daily living. Also determine if the patient has recently experienced any exacerbation or worsening of previous symptoms; examples include increasing frequency of angina attacks in a patient with coronary artery disease or decreasing activity tolerance in a person with congestive heart failure.
Specifically question the patient about the current medications that he or she is taking, with special attention to any cardiovascular medications. As noted, many antiarrhythmic medications prescribed to control tachyarrhythmias may cause a symptomatic bradycardia. In addition, medications that are prescribed to manage other cardiovascular conditions, such as hypertension and coronary artery disease, can affect the electrical conducting system in the heart and cause symptomatic bradycardia. Also inquire about any other medications the patient may be taking. A wide variety of medications, such as tricyclic antidepressants, lithium, and phenothiazines, can interact with cardiovascular medications or antiarrhythmic medications to create bradyarrhythmias. Also assess the patient's medication history. Determine if any other cardiovascular medications have been prescribed for the patient at one time and why those medications were changed or discontinued. If indicated, consider obtaining serum drug levels for medications such as digoxin or procainamide to determine if toxicity is present. Toxic levels of some antiarrhythmics may result in bradycardia [19].
Finally, obtain information about the patient's medical history. Specifically inquire about any history of cardiovascular diseases, including coronary artery disease, previous MI, congestive heart failure, cardiomyopathy, hypertension, valvular heart disease, and any previous history of cardiac conduction problems. Determine if the patient has any history of interventional cardiovascular procedures, such as coronary angioplasty, coronary artery bypass surgery, valvular heart surgery, or electrophysiology interventions such as radiofrequency ablation. The presence and type of concurrent cardiovascular disease may influence the type of pacemaker selected and its programming.
When a patient does not speak the same language as the clinician, a professional interpreter should be consulted to ensure accurate communication. A systematic review of the literature has shown that the use of professional interpreters provides better clinical care than the use of "ad hoc" interpreters, with the former improving the quality of care for patients with limited English language skills to a level equal to that for patients with no language barriers [41]. Use of professional interpreters has been associated with improvements in communication (errors and comprehension), utilization, clinical outcomes, and satisfaction with care [41]. Individuals with limited English language skills have indicated a preference for professional interpreters rather than family members [42].
Generally, in order for a patient to meet criteria for the implantation of a permanent pacemaker, the patient's symptoms should be clearly correlated with episodes of bradycardia. Commonly used diagnostic tools include an ECG (12-lead or rhythm strip) and ambulatory electrocardiographic monitoring (AECG).
The traditional method of assessing a patient's heart rhythm is a 12-lead ECG. It is inexpensive, noninvasive, and readily performed in any healthcare setting or physician's office. Unfortunately, for persons with symptomatic bradycardia, this diagnostic tool has limited value. Typically, an ECG only records about 12 seconds of data. Because symptoms may be transient and occur intermittently, the ECG may not capture the patient's heart rhythm at the time that the symptoms are occurring. The patient may or may not have symptoms during an office or clinical visit. The ECG is considered a passive diagnostic tool; it only records the rhythm occurring at the time the tracing is done. It does nothing to actively precipitate an occurrence of arrhythmia [18,27].
To facilitate documentation of the patient's heart rhythm at the exact time that the patient is experiencing symptoms, researchers have developed another type of ECG monitoring called ambulatory electrocardiography, or AECG. This type of monitoring may be done over a longer period of time and in a setting outside the hospital, clinic, or physician's office. Three types of AECG monitoring are available: Holter monitors, event monitors, and implanted event monitors. The earliest AECG monitor developed, the Holter monitor, is a small, portable, battery-powered device that is worn by the patient for a period of 24 to 48 hours. Electrodes connected to the monitor are attached and taped to the patient's chest. A belt or shoulder strap holds the monitor in position. The patient is not allowed to bathe or shower during the period of the test. Otherwise, the patient is encouraged to perform his or her usual daily activities. The patient is instructed to keep a diary to document the occurrence, type, and time of any symptoms that he or she experiences [43]. In some cases, if the patient is elderly or might otherwise have problems completing a written log, a family member is asked to assist the patient to document the occurrence of any symptoms. Holter monitoring is a noninvasive procedure with little to no associated risk to the patient. The device may be applied in a physician's office, outpatient clinic, or inpatient hospital unit. A Holter monitor may be useful for persons who experience symptoms on a daily (or more frequent) basis; it is also useful for persons whose symptoms are severe enough to cause loss of consciousness or severe disorientation [43]. Persons with these severe symptoms would not be able to operate some of the more complex forms of AECG. The disadvantage to Holter monitoring is that it may fail to capture a symptomatic period or a period of bradyarrhythmia. Many people's symptoms occur less frequently than every day; longer periods of ambulatory monitoring are required to capture the necessary period of time [18]. Wireless Holter monitors have a longer recording time than standard Holter monitors. Using wireless cellular technology, these monitors record the heart's electrical activity for a preset amount of time and then send the recorded data to the patient's physician. Wireless Holter monitors work for days or even weeks, until signs or symptoms of a heart rhythm problem occur. They are usually used to detect infrequent symptoms. The downside to wireless monitors is that the patient must remember to write down the time symptoms occurred so that his or her physician can match it to the recording. These monitors also have a short battery life and are more expensive than a Holter monitor or an event monitor [43].
Implantable cardiac monitors are also available to serve as long-term Holter monitors [44]. These monitors allow long-term ECG monitoring without external leads connected to a recording device. They use a wireless, implantable system to help diagnose cardiac arrhythmias and the causes of syncope. Implantable cardiac monitors can be programmed to automatically record when defined triggers are activated or can be activated manually by the patient [44].
Advances in technology have resulted in the development of an AECG device known as an event recorder. An event recorder may be used for monitoring the patient's heart rhythm over a one- to two-month period; it is helpful in documenting arrhythmias and episodes of symptoms that occur more infrequently (such as weekly or monthly). There are several types of event recorders. Post-event recorders are among the smallest event monitors—about the size of a thick credit card. They can be worn by the patient like a wristwatch or carried in a pocket. They have no wires connecting them to chest sensors. The patient must hold the device to the chest when a symptom is felt. Post-event recorders only record what happens after started by the patient, so they may miss a heart rhythm problem that occurs before and during the onset of symptoms. Additionally, it may be difficult for the patient to start the device while experiencing symptoms [43,45]. Autodetect recorders are also small—about the size of the palm of the hand. They do have wires connecting them to chest sensors. These recorders need not be started by the patient. They detect abnormal heart rhythms and automatically record and send the data to the patient's physician [43,45]. Pre-symptom memory loop recorders (also called continuous loop event recorders) are the size of a pager. They are worn constantly, either clipped to a belt or carried in a pocket. A loop recorder records the patient's heart rhythm continuously, but it only stores and saves the data when "instructed" to do so by the patient [43,45]. When the event monitor is applied, the patient is told to depress a switch on the recorder any time he or she feels symptoms. When the switch is activated, the recorder automatically stores a few minutes of data from before, during, and after the onset of symptoms, making it possible for the patient's physician to see even very brief changes in heart rhythm [43,45]. This type of event recorder is indicated for persons whose symptoms are so brief and fleeting that they would disappear before any type of recording device could be attached. The loop recorder may also be used for persons whose symptoms cause loss of consciousness (or near loss of consciousness) as long as the person is not severely disoriented upon returning to consciousness and can still depress the event switch on the outside of the recorder. The data obtained by the event recorder can be readily transferred to a physician via telephone for analysis.
Another type of external event recorder is only attached when the patient experiences symptoms. During a clinic visit, physician appointment, or prior to discharge from an inpatient hospital stay, the patient is given the recorder and instructed on how to apply the electrodes. Commonly, the electrodes attach as bracelets, finger clips, or patches applied under the arms. When the device is attached to the patient, it records and stores ECG data. This type of event recorder is indicated for persons whose symptoms last long enough to permit attachment of the device; it is not indicated for persons who experience loss of consciousness or near loss of consciousness during an episode. To capture the data, the patient should be capable of finding and applying the device during symptomatic episodes. As with the loop recorder, data recorded and stored by this type of event monitor may be readily transmitted to a physician via telephone for analysis [17,18,46].
An event monitor that may be implanted under the skin has also been developed. Implantation of this device is a minor procedure that takes approximately 20 minutes and may be performed under local anesthesia. To implant the event recorder, a physician trained in the procedure makes a short (roughly ¾ inch) incision in the subclavian area and creates a small pocket to hold the monitor. Risks include the usual risks associated with a minor surgical procedure (i.e., primarily the risk of infection and reactions to the local anesthesia) [45]. No significant morbidity has been associated with use of the actual device. The implanted event monitor may remain in place for up to 14 months, until the necessary data is obtained or the end of battery life occurs. Implanted event recorders are continuous loop recorders. Like the external loop recorders, the implanted device continuously records the patient's ECG rhythm. In some instances, the device may be programmed to automatically store ECG information during a fainting or syncopal episode. Additionally, the patient may be instructed to depress a switch when he or she feels symptoms. When the switch is activated, a small amount of data about the heart rhythm at that time is stored. Family members may be instructed on how to activate the switch in the event that the patient is unable to do so. The implanted recorder can only store a certain amount of recorded data; when it is "full," the physician may clear the stored information. In general, the patient should be instructed to contact his or her physician for an appointment shortly after he or she has experienced a symptomatic episode.
In general, patients may continue with their usual daily activities during the period of time that the event monitor is implanted. However, there are a few precautions that the patient should know about at the time of implantation. These include [17]:
Always carry the identification card that shows you have an implanted event monitor.
Airport screening devices will detect the metal and sound an alarm; before going through the screening device, let the security agent know that you have a heart device and show your identification card to the security agent. Walk through the screening device at a normal pace and move away if you feel rapid heartbeats or dizziness.
Use caution when using a cellular telephone. Keep the phone at least 6 inches away from the implantation site even when it is not in use. Hold the phone to the ear opposite from the implantation site when talking. Do not carry a cellular telephone in a pocket directly over the device implantation site.
Notify any healthcare personnel that you see that you have an implanted monitor.
Antitheft devices, such as those used in department stores, should not damage the event monitor but may disrupt its function. Walk through the device at a steady pace; do not linger around the devices.
The functioning of the device should not be affected by the normal use of most household electrical equipment, spark ignited internal combustion engines, or machine shop tools.
Mobile cardiac telemetry (MCT) monitoring represents a development in cardiac monitoring. The MCOT patch system consists of a sensor that the patient attaches to replaceable patches (placed near the heart) and a monitoring device that resembles a cell phone. The system conducts a beat-by-beat analysis of the patient's heart activity. Cardiac events are detected even if the patient is asymptomatic. The patient can also record symptoms as felt using the monitor. The recordings are transmitted to a central surveillance center where certified cardiac technicians perform real-time analysis. Patients are monitored 24 hours per day, seven days per week. Clinical reports are made available to the patient's healthcare professional both during and at the end of the monitoring period [47,48,49].
Outcomes from AECG Monitoring
Optimally, the data from AECG monitoring would clearly confirm the need for a pacemaker or definitively rule it out. In reality, several different outcomes may occur:
The ECG recording shows the presence of a bradyarrhythmia during the precise time that the patient is experiencing one or more of the cardinal symptoms associated with symptomatic bradycardia. This finding supports the need for implantation of a permanent pacemaker and may provide data to guide the selection of the most appropriate type of pacemaker.
The ECG recording shows no arrhythmias during the precise time that the patient is experiencing symptoms. This finding is also helpful; it rules out cardiac conduction abnormalities as a cause for the patient's symptoms. Evaluation of the patient can then turn to other potential causes.
The ECG documents the presence of a bradycardia during a time that the patient remains asymptomatic. This is an equivocal finding; usually, additional testing is indicated to determine if the patient's symptoms are actually due to conduction abnormalities.
The ECG shows no episodes of arrhythmias, and the patient experiences no symptoms during the period that the event monitor is used. This finding is the least helpful; it neither supports nor refutes the presence of conduction abnormalities or the need for a permanent pacemaker.
Due to the costs involved in the use of AECG monitoring, the ACCF, the AHA, and the HRS have established national guidelines for its use [46,50]. According to these guidelines, AECG is indicated for persons with documented or suspected bradycardia or conduction disorders [50]. Cardiac rhythm monitoring is useful to establish a correlation between heart rate or conduction abnormalities with symptoms, with the specific type of cardiac monitor chosen based on the frequency and nature of symptoms, as well as patient preferences [50]. The intermittent nature of most symptomatic bradycardia and conduction disorders often necessitates a more prolonged form of electrocardiographic monitoring to correlate rhythm disturbances with symptoms. For patients with daily symptoms, a 24- or 48-hour continuous ambulatory ECG (Holter monitor) is appropriate and, in active individuals, may help identify the presence or absence of chronotropic incompetence. Less frequent symptoms are best evaluated with more prolonged AECG that can be accomplished with a broad array of modalities, as previously discussed. The yield of ambulatory monitoring for significant bradyarrhythmias varies according to the population studies but is typically less than 15% [50]. The guidelines support the trend toward the development and use of AECG devices that permit longer periods of monitoring to facilitate correlation of symptoms with arrhythmias for persons with infrequent episodes [46,50].
Electrophysiology testing (EPS) is not generally the first diagnostic method of choice when evaluating a patient for the presence of symptomatic bradycardia. However, under certain conditions, EPS may be performed in order to aid in the management of the patient, such as patients with tachy-brady syndrome or when the initial noninvasive evaluation is nondiagnostic; EPS can document the mechanism and location of the tachyarrhythmia [50]. It can also determine if ablation of some area involved in the tachyarrhythmia can help control the rapid heart rate. EPS may be used to evaluate a patient for the presence of retrograde conduction. As will be discussed further, retrograde conduction (i.e., conduction that spreads from the AV node back into the atria) can interfere with optimal pacemaker functioning, and certain types of pacemakers are contraindicated in the presence of retrograde conduction. EPS may also be used to evaluate the extent of bundle branch block present in selected patients [19].
When hypersensitive carotid sinus syndrome is thought to be responsible for syncopal episodes, a tilt-table test may be used to confirm the diagnosis. Tilt-table tests are often performed in the cardiac catheterization laboratory. The patient is placed on a table that can tilt up to 80 degrees upright; safety straps are applied to prevent the patient from slipping and injuring him/herself during the test. An IV access is started to permit the rapid administration of IV medications or fluids should those be indicated during the test. Continuous ECG and blood pressure monitoring are performed, and the use of continuous intra-arterial blood pressure monitoring via placement of an arterial line is recommended. The purpose of the tilt test is to create the physiologic environment required to trigger vasovagal syncope. The patient is tilted from a supine position to a 70-degree angle. Patients who suffer from vasovagal syncope experience a decrease in venous return followed by a decrease in the left ventricular filling pressure when they are tilted to an upright position. The sequelae can include hypotension, bradycardia, and syncope. In addition to confirming the presence of a vasovagal syncope, a tilt-table test can also help to differentiate the major contributing cause. Permanent pacemaker implantation is indicated for syncope due to a mixed cardio-inhibitory/vasodepressor response [17,21].
After determining that permanent pacemaker implantation is indicated, it should then be determined which type of pacemaker system will be the most effective. A number of factors should be considered in the decision, including patient factors, technical aspects, and procedural issues. Patient factors include [28,50]:
What is the patient's specific medical indication for pacemaker implantation? Some types of pacemakers are contraindicated for certain conduction abnormalities, and some pacemakers are significantly more effective in managing a specific type of problem than other pacemaker types. Table 3 summarizes recommendations for the use of different types of pacemakers.
Does the patient require the rate responsive feature? How active is the patient's lifestyle? Is chronotropic incompetence present? Does (or will) an inability to increase the heart rate in response to increased activity create a problem for this patient?
Is the patient likely to develop increasing conduction problems that may require additional or more complex pacing functions? If so, a pacemaker that has the capability to be reprogrammed for more complex pacing functions when needed is indicated.
What is the patient's clinical status? Does the patient have a functioning SA node? A functioning AV node? Does the patient require AV synchrony to maintain adequate cardiac output? Does the patient have a history of chronic or paroxysmal atrial tachyarrhythmias?
What is the cost of the pacemaker system? Initial costs include the cost of the generator and leads, implantation costs, and the cost of the initial programming. Subsequent costs include the cost of follow-up and generator replacement, when needed. In general, dual chamber pacemakers are more expensive to implant and maintain. Use of the rate responsive feature also increases implantation and programming costs.
How might a pacemaker improve the patient's quality of life? Among patients with indications for permanent pacemaker implantation, the quality of life has been shown to improve substantially after pacemaker implantation. However, based on several trials and small crossover studies, the benefits of different pacing modes (i.e., dual chamber pacing versus single pacing) are inconsistent.
ACC/AHA/HRS RECOMMENDATIONS FOR TYPE OF PERMANENT PACING FOR MANAGEMENT OF BRADYCARDIA ATTRIBUTABLE TO SINUS NODE DYSFUNCTION
| Class I Recommendations | |||||
| |||||
| Class II Recommendations | |||||
|
Technical and procedural considerations include:
The type of pacemaker leads (i.e., unipolar versus bipolar; active versus passive fixation) and the type of insulation used on the pacemaker lead
Capabilities of the available pacemaker programmer
Locally available technical support
Choice of specific rate responsive sensor (if rate responsive feature is desired)
Ease of implantation of system
Ability to implant multiple leads
Careful analysis of all factors facilitates the selection of the pacemaker system and programmed settings that are the most likely to achieve the desired optimal outcome for the patient. The following clinical examples illustrate the pacemaker selection process.
Patient L is a man, 54 years of age, who is diagnosed with symptomatic bradycardia; the underlying conduction abnormality is atrial fibrillation with a slow ventricular response. Patient L has a history of atrial fibrillation with a rapid ventricular response. Pharmacologic therapy required to control the tachycardic episodes has resulted in the development of a severe bradycardia with pauses up to four seconds. Patient L's medical history is positive for coronary artery disease and percutaneous transluminal coronary angioplasty for treatment of significant blockages in branches of his left coronary artery. He also has a history of hypertension. The patient is currently employed full time as the sales representative of a major company. He is married, with teenage children; his hobbies include hiking and swimming. He describes himself as "perpetually on the go." Patient L's insurance will cover the cost of either a single or dual chamber pacemaker; it will also cover the cost of the rate adaptive feature. Based on a thorough assessment of Patient L's conduction abnormality and lifestyle, the physician chooses to implant a VVIR pacemaker.
Patient M is a woman, 68 years of age, who experiences a near syncopal episode in a local restaurant. Thorough evaluation of the patient shows that she has intermittent episodes of third-degree heart block. During these episodes, a ventricular escape rhythm occurs at a rate of 30 bpm. Patient M experiences dizziness, near-syncope, and some confusion during these bradycardic episodes. She has a history of a major MI involving the anterior wall of the heart; she also has congestive heart failure. Although her sinus node appears to fire at a regular rate, the physician notices that Patient M experiences increased symptoms of activity intolerance and congestive heart failure during stress or exertion. Testing shows that the patient has sinus node chronotropic incompetence. Patient M lives alone; she describes her lifestyle as "active." She volunteers frequently for community service organizations and participates regularly in her local church. Based on assessment of Patient M and her needs, the physician decides to implant a DDDR pacemaker. Use of a DDDR pacemaker maintains AV synchrony and provides for an increase in pacing rate when indicated by an increase in activity.
In patients with indications for permanent pacing but also with significant comorbidities such that pacing is unlikely to provide meaningful clinical benefit, or if patient goals of care strongly preclude pacemaker therapy, implantation or replacement of a pacemaker should not be performed [50].
After the decision is made to implant the pacemaker and the specific type of pacemaker is selected, the patient is scheduled for pacemaker implantation. The length of stay for pacemaker implantation is generally very short and often involves only an overnight stay in an acute care facility. Some institutions are exploring the option of implanting pacemakers on an outpatient basis; however, this practice is not yet widespread [51]. Pacemaker implantation may be safely performed in either the cardiac catheterization laboratory or the operating room. The selection of the specific site is based on an evaluation of which site will provide the best outcome and most cost-effective method for the individual patient. Some patients, such as patients who are confused, uncooperative, or have special medical needs, require the use of general anesthesia; pacemaker implantation for these patients should occur in the operating room. Implantation for patients who do not require general anesthesia but who can be managed with moderate sedation/analgesia may be performed in the cardiac catheterization lab [19,22].
Unless the patient's medical status requires hospital admission for management prior to pacemaker implantation, the patient may be admitted to the hospital on the morning of the procedure. The patient should be NPO for six to eight hours prior to the procedure. Basic preimplantation laboratory work is obtained, including a complete blood count, serum electrolytes, blood urea nitrogen (BUN), creatinine, prothrombin time (PT), partial thromboplastin time, and a urinalysis. A baseline 12-lead ECG, alveolar pressure (PA), and lateral chest x-ray are also obtained. Baseline vital signs and physical assessment are performed and documented. In the past, if the patient had been taking warfarin or similar anticoagulant drugs, the patient would have been instructed to discontinue taking them for several days in advance of the procedure to permit his/her' PT and international normalized ratio (INT) to return close to normal values in order to reduce the risk of excessive bleeding during the implantation. Patients at high risk for thromboembolic events might have been admitted to the hospital for temporary IV heparin therapy. However, a strategy of implanting devices during uninterrupted warfarin therapy appears to have a lower bleeding risk than a strategy of temporarily discontinuing warfarin and bridging with heparin [52].
The planned site for the pacemaker generator (usually the right or left subclavicular area) is scrubbed and shaved according to institutional protocol. The patient's dominant arm is identified; generally, pacemaker generators are implanted on the nondominant side. Informed consent should always be obtained. The patient may receive a mild sedative prior to leaving for the catheterization lab or operating room. Additional sedation and/or analgesics will be administered during the procedure. Generally, the patient is instructed to continue taking his/her routine medications prior to implantation. Due to the patient's NPO status, doses of insulin or oral hypoglycemic agents may need to be temporarily reduced or held.
Education for the patient and his or her family begins in the preoperative period and continues throughout the patient's life. The focus of patient education in the preoperative period is on addressing the patient's immediate questions and concerns and providing the patient and family with information about what to expect during implantation and in the period immediately following implantation. Many patients are very anxious about undergoing pacemaker implantation. They may express fears of "being dependent on a machine" to stay alive. Some may have heard a variety of "old wives' tales" about pacemakers from friends or relatives. Or, they may have known of a family member "years ago" who had a pacemaker implanted that did not work "right," and the person died. Some people have specific concerns about how a permanent pacemaker may interfere with their normal activities or ability to use certain types of equipment. Responding to any concerns or questions that the patient has during the preoperative period is very important. Providing specific information about what the patient can expect before, during, and immediately after pacemaker implantation can also help to reduce the patient's anxiety. The following topics are often helpful to cover in the preoperative period:
A brief explanation of why the patient needs a pacemaker. (Consider the use of a diagram or model to illustrate a brief explanation of normal conduction, the patient's specific conduction problem, and how the implantation of a pacemaker will correct that problem.)
What a pacemaker generator and pacemaker leads look like. (Pacemaker manufacturers are a good source of illustrations and diagrams of their products.)
Where the pacemaker generator will be implanted
How leads will be attached
Use of general anesthesia or moderate sedation during the procedure to minimize discomfort
Details of the usual preoperative preparation
Allow time for the patient and family to ask questions and clarify concerns. The AHA, the Heart Rhythm Society, and various patient education sites provided by pacemaker manufacturers may be recommended as helpful educational sources (Resources).
After informed consent is obtained and the preoperative preparation is complete, the patient is taken to the operating room or cardiac catheterization lab. IV access is obtained, and continuous monitoring of ECG rhythm, oxygen saturation, and blood pressure is initiated. Depending on the implantation site and the physician's judgment, the patient will receive either moderate sedation or general anesthesia. Commonly used agents for sedation include midazolam and fentanyl. Once the patient is sedated, a small surgical incision is made, approximately 2–3 inches in length, beneath the patient's clavicle. Preferably, the patient's nondominant side is chosen for placement of the incision and pacemaker generator; use of the nondominant side lessens the risk that the pacemaker leads will be dislodged by repetitive arm and shoulder movements. The incision site is positioned to provide the physician with ready access to either the subclavian or cephalic vein for transvenous insertion. Depending on the type of pacemaker, one or two pacemaker leads are inserted transvenously and passed through the vessels into the heart. Fluoroscopy is used to monitor the passage of the pacing lead through the vessels. Generally, an atrial pacemaker lead will be positioned inside the right atrial appendage; a ventricular pacemaker lead is positioned in the apex of the right ventricle. Once it is determined that the lead is in the correct position, the lead tip is attached to the endocardial surface of the heart. When the leads are properly positioned, a small area is opened beneath the skin in the region of the original incision. This area, called a "pacemaker pocket," is designed to hold the pulse generator. In most people, the pacemaker pocket is created in the subcutaneous tissue located on top of the pectoralis muscle. For extremely thin individuals, the pacemaker pocket may be created beneath the pectoral muscle to provide adequate support and stability for the pacemaker generator. The connector end of the leads is firmly attached to the connector block on the outside of the pulse generator and the screw tightened to ensure good contact and stable position. The pacemaker generator is then inserted into the pacemaker pocket, and the skin is sutured shut. A dressing is applied. In some institutions, a pressure dressing may be applied. Antibiotics are frequently administered to reduce the risk of infection. The exact routine varies from institution to institution; some administer a single dose before or during the procedure, while others administer IV antibiotics for a 24-hour period following implantation. Extreme care is taken throughout placement of the generator and leads to avoid impairing the range of motion of the upper extremity on the affected side. Care is also taken to make sure that the generator and leads are well protected by body tissue to prevent the accidental erosion of either the leads or pacemaker generator through the skin. As previously noted, most pacemakers contain a small amount of steroid at the endocardial end of the lead to reduce the probability of inflammation causing abnormalities in function soon after implantation. Finally, the initial pacemaker settings are programmed, and pacemaker function is tested [3,4,18,19,20].
After the procedure is completed, the patient is generally moved to a recovery area until he/she has recovered from the general anesthesia or moderate sedation. Once the patient is awake and stable, he/she is transferred back to the general unit. The goals of care in the immediate 24- to 48-hour postoperative period are to:
Monitor the patient for signs of postoperative complications.
Manage the patient's postprocedure pain.
Monitor both the patient and the pacemaker for proper pacemaker functioning and efficacy of pacemaker therapy in relieving preimplantation symptoms.
Begin patient discharge education and address patient/family concerns.
Complications that may develop following permanent pacemaker implantation include bleeding into the pacemaker generator pocket, hemothorax, pneumothorax, and dislodgment of the pacemaker leads. Upon return to the floor, the patient's vital signs are monitored periodically. Continuous ECG monitoring is initiated. Spot checks of the patient's oxygen saturation level may be performed as needed. The pacemaker pocket site is assessed frequently for signs of excessive bleeding or swelling; if these are present, the physician should be notified immediately. A PA and lateral chest x-ray is taken to evaluate the positioning of the pacemaker leads as well as to rule out the presence of either a hemothorax or a pneumothorax [18].
Patients often experience pain at the pacemaker implantation site. Depending on multiple factors, patients may describe pain as mild to very severe. The patient's pain level should be routinely assessed using a 10-point pain scale (1 for least, 10 for worst). The physician's orders should include analgesic medications; prescribed medications may range from acetaminophen to stronger narcotic analgesics. The effectiveness of pain medication should be assessed and documented. Any complaint of increasing pain despite adequate analgesic medication should be carefully investigated; increased pain in the pacemaker pocket may be an indication that bleeding is occurring into the pocket.
Early complications associated with permanent pacemaker implantation include infection of either the incision site or the pacemaker pocket and accidental dislodgment of the pacemaker leads. To lessen the risk of infection, the patient should avoid getting the incision site wet. The site should also be routinely inspected for signs of redness, warmth, or drainage. During the patient's hospital stay, a dressing should be maintained over the incision site as ordered by the physician; if the dressing becomes dislodged or saturated with blood during this initial period, it may be changed using sterile technique. To lessen the risk of lead dislodgment, the patient should be instructed to limit movement of the affected arm and cautioned to avoid extending the arm above the level of the shoulder. Use of a sling on the affected arm can serve to promote comfort and remind the patient to limit use of that arm [3].
Monitoring pacemaker function is an important part of initial postimplantation care. Comprehensive evaluation of pacemaker function requires the specialized skills of a physician trained in pacemaker therapy. However, nurses working directly with the patient are in an excellent position to identify early clues indicative of potential pacemaker problems. Monitoring pacemaker function involves two key areas:
Assessment of pacemaker performance
Assessment of the patient's response to the pacemaker therapy
To assess the performance of a specific pacemaker, begin by obtaining information about the type of pacemaker that was implanted and its settings. Specific details to determine include:
What type of pacemaker was implanted? Is it dual chamber? Single chamber atrial? Single chamber ventricular?
Does the pacemaker have rate responsive capability? Is the rate responsive feature turned on? If so, what is the upper rate limit?
What is the low rate setting?
If the pacemaker is a dual chamber, what is the high rate setting? What is the programmed AV interval?
This information may be obtained from the pacemaker data documented in the progress notes in the chart. Knowing the type of pacemaker and the applicable programmed settings is critical to an accurate evaluation of the pacemaker's function [19].
Assessing the patient's heart rate is a basic method for screening for proper pacemaker function that can be readily performed and requires no specialized ECG interpretation skills. If a permanent pacemaker is functioning correctly, the patient's heart rate should never drop to significantly less than the low rate setting on the pacemaker. A drop of one to two beats is not considered "significant" and may occur normally following pacemaker implantation; however, a rate that drops significantly may indicate a pacemaker malfunction [20].
Practice Exercise: Evaluating Heart Rates
Using the provided information regarding pacemaker codes and low heart rate settings, evaluate the heart rates in the following case examples.
Patient Z has a VVI pacemaker implanted for the management of chronic atrial fibrillation with a slow ventricular response. The low rate setting on the pacemaker is 60 bpm. When Patient Z's heart rate is checked, a reading of 58 bpm is obtained. Does this represent normal pacemaker function for this patient?
Answer and Rationale: Yes. The low rate setting on Patient Z's pacemaker is 60 bpm; a 1–2 bpm drop in heart rate may occur normally following the implantation and initial programming of the pacemaker. Patient Z's heart rate falls within the expected parameters for normal functioning of his pacemaker.
Patient Y also has a VVI pacemaker implanted for management of infrequent long pauses. The low rate setting for Patient Y's pacemaker is 50 bpm. The nursing assistant records a heart rate of 44 bpm for Patient Y. Does this represent normal functioning of Patient Y's pacemaker?
Answer and Rationale: No. Patient Y's pacemaker is programmed to deliver a pacing impulse as needed to maintain her heart rate at a rate of at least 50 bpm. A heart rate of 44 bpm is significantly less than the low rate setting programmed for the pacemaker and indicates a possible pacemaker problem.
Patient X also has a VVI pacemaker implanted for management of intermittent severe bradycardia. His underlying rhythm is atrial fibrillation. The low setting on his pacemaker is 55 bpm. The nursing assistant records his heart rate as 76 bpm. Does this represent normal pacemaker functioning?
Answer and Rationale: Yes. Patient X has only intermittent periods of bradycardia. A heart rate of 76 bpm most likely reflects his intrinsic heart rate and rhythm when he is not experiencing bradycardia. To confirm this assessment, his rhythm on telemetry is checked and it is clear that Patient X is indeed in his usual baseline rhythm with only intermittent paced beats present.
When a patient has a pacemaker that is capable of increasing the heart rate in response to changes in activity or metabolic demands, the assessment of the patient's heart rate as an indicator of pacemaker function becomes more complex. Appropriate assessment involves both counting a pulse rate and analyzing the patient's ECG image. General guidelines to follow when evaluating a heart rate for proper upper rate limit functioning include:
The patient's heart rate should never drop more than one to two beats less than the low rate setting of the pacemaker.
When the patient's rhythm is 100% paced, the paced rate should not exceed the upper rate limit setting.
When the patient's rhythm is a combination of paced beats and intrinsic rhythm, the patient's heart rate may exceed the upper rate limit setting.
Pacemakers that have the capability of increasing the pacing rate in response to the body's needs include any pacemaker with the rate responsive feature as well as dual chamber pacemakers in persons with intact sinus node function [20].
Practice Exercise
Using this information on low and upper rate limit settings and pacemaker coding, evaluate the heart rates for the following patients.
Patient W has an AAIR pacemaker implanted to manage severe bradycardia associated with sick sinus syndrome. The low rate setting on the pacemaker is 60 bpm, and the high rate setting is 120 bpm. Patient W's resting heart rate is 60 bpm, but his heart rate increases to 96 bpm during ambulation. According to Patient W's telemetry, he is 100% paced when he is at rest and when he is ambulating. Does this represent normal pacemaker function?
Answer and Rationale: Yes. At rest, Patient W's heart rate matches the low rate setting programmed for his pacemaker. An AAIR pacemaker is programmed to increase the rate of pacing with changes in activity. When patient W increases his activity by walking, his paced rate increases accordingly. The paced rate with activity does not exceed the programmed upper rate limit setting.
Patient U has dual chamber pacemaker implanted for management of third-degree AV block; her sinus node function is normal. The rate limit settings are 60 bpm (low) and 120 bpm (high). When Patient U is sitting out of bed reading, her heart rate is 64 bpm. When she works with physical therapy, her heart rate is 110 bpm. Telemetry monitoring shows that her rhythm is consistently 100% paced. Does this reflect normal pacemaker function for this patient?
Answer and Rationale: Yes. For a person with intact sinus node functioning, the dual chamber pacemaker is programmed to track the patient's SA node firing and pace the ventricles at the same rate as long as the sinus node fires at or above the programmed low rate setting on the pacemaker. The paced rate should not exceed the upper rate limit setting. A paced rate of 64 bpm at rest and a paced rated of 110 bpm with activity fall within normal functioning for this dual chamber pacemaker.
Patient V has a VVIR pacemaker implanted. The low rate setting is 60 bpm with a high rate of 120 bpm for rate responsive pacing. Patient V contacts a nurse in the middle of the night and states that she feels like her "heart is pounding." Her heart rate is 110 bpm. Patient V's blood pressure, heart rate, and temperature are normal. Patient V shows no clinical signs that would indicate an increased metabolic demand or a need for an increased heart rate. Telemetry shows that her heart rhythm is 100% paced at a rate of 110 bpm. Does this reflect normal pacemaker function for this patient?
Answer and Rationale: No. The rate responsive programming is designed to increase the patient's heart rate in response to activity or increased metabolic demands. At rest, Patient V's paced rate should not be less than 60 bpm but not markedly greater than 60 bpm. A resting paced rate of 110 bpm indicates a possible problem with the rate responsive sensor or programming.
Patient T has a DDDR pacemaker placed for management of sick sinus syndrome with chronotropic incompetence. Patient T is a man, 84 years of age, who has a history of coronary artery disease and moderate congestive heart failure. The low rate setting on the pacemaker is 60 bpm with an upper rate limit for activity response set at 120 bpm. Patient T's heart rate at rest is 60 bpm. When he walks in the hall, his heart rate increases to 120 bpm and he complains of dizziness, shortness of breath, and chest pain. Telemetry shows that his rhythm is consistently 100% paced. Is Patient T's pacemaker functioning properly?
Answer and Rationale: Yes. With a DDDR pacemaker, the pacemaker should pace the patient at the low rate setting when the patient is at rest. Patient T's low rate setting is 60 bpm and his heart rate at rest is also 60 bpm. With activity, a DDDR pacemaker should increase its rate of firing but should not exceed the programmed high rate setting. Patient T's high rate setting is 120 bpm, and his heart rate with activity is also 120 bpm. The pacemaker is functioning normally as programmed; however, because Patient T experiences symptoms with the high rate setting, the programming may need to be adjusted. Elderly patients with underlying cardiovascular disease may require a lower rate for the high rate setting.
In addition to heart rate monitoring, rhythm strip assessment can provide valuable clues about the functioning of the pacemaker. It is important to remember that full evaluation of pacemaker function can be very complicated, require specialized training skills, and (often) the use of specialized equipment. However, like heart rate assessment, evaluation of rhythm strips can provide clues to possible pacemaker malfunctions. The following components are part of rhythm strip assessment:
Calculation of the heart rate recorded on the rhythm strip
Assessment of the strip for the presence of pacemaker spikes when and where expected
Assessment of the strip to see if all pacemaker spikes are followed by the depolarization of the appropriate chamber of the heart
Assessment of the strip for the presence/absence of random pacemaker spikes
To calculate a heart rate from a rhythm strip, you should first recall a few basic facts about ECG recordings. ECG tracings are recorded on grid paper that is laid out in small and large squares. Vertical lines measure amplitude or voltage. Horizontal lines measure time. When ECGs are recorded at the standard speed of 25 mm/sec, the large and small squares may be used to calculate heart rates, measure intervals, and compute the length of a pause between two beats. On ECG recording paper, each small square is 1 mm on the longitudinal axis. This represents 0.04 seconds (or 40 ms); see "A" on Strip 1. Each large square is made of five small squares and is 5 mm on the X and Y axes. On the horizontal line, each large square represents 0.20 sec (or 200 ms); see "B" on Strip 1. Five large squares on the horizontal line represent one second.
There are several ways to estimate a heart rate from a rhythm image.
Method 1
Identify a six-second period of time on the image. Some ECG recording machines place a "hash mark" on top of the grid at three-second intervals. You can also identify a six-second period by counting off large squares on the recording paper. Five large squares measure one second, and therefore, 30 large squares make a six-second strip (Strip 2).
Start at the beginning of the six-second period and count the number of complete QRS complexes that fall within that time period. Multiply the number of QRS complexes in six seconds by 10. This gives you an estimated heart rate for one minute.
In Strip 2, there are almost 15 complete QRS complexes within the six-second period. Fourteen to fifteen complexes multiplied by 10 equals a heart rate of approximately 140–150 bpm.
Method 2
If the heart rate is regular, it may be calculated by using "the rule of 300." This method also works well for extremely slow or extremely fast heart rates. It is also useful to estimate rates when the patient's rhythm and rate change abruptly in a brief period of time. To estimate a heart rate using the rule of 300, follow these steps:
Count the number of large squares between two beats using the R-R interval.
Divide the number of large squares into 300; this gives an estimate of the heart rate.
Using the example in Strip 3, the R-R interval between 1 and 2 contains five large squares. Three hundred divided by five gives an estimated rate of 60 bpm.
To determine if pacemaker spikes are occurring when and where they should, several basic steps should be followed. Based on the type of pacemaker, determine if a pacemaker spike should occur before the P wave, the QRS complex, or both:
A beat generated by an AAI/AAIR pace-maker should have a pacemaker spike before the P wave.
A beat generated by a VVI/VVIR pacemaker should have a spike just before the QRS complex.
DDD/DDDR pacemakers may have a spike before the P wave, before the QRS complex, or both.
Carefully analyze each paced beat present on the ECG tracing to determine if the pacemaker spike occurs at the correct place. Low voltage spikes may be difficult to identify on a rhythm image. Adjusting the settings on the monitor, changing monitoring leads, or adjusting the position of the monitoring electrodes on the patient may be necessary to make the spikes more readily visible on a rhythm image.
Carefully analyze each pacer spike present on the ECG tracing to ensure that the spike is followed by depolarization of the appropriate chamber of the heart. Atrial pacing spikes should be followed by a paced P wave, and ventricular pacing spikes should be followed by a paced QRS complex. Paced beats often have a different configuration from normal beats. Depending on the location of the atrial pacing lead, a paced P wave may appear almost normal or it may have a flattened, abnormal configuration. Because the ventricular pacing lead is generally positioned in the right ventricle, paced QRS complexes generally show a left bundle branch block configuration. This occurs because the wave of depolarization from a paced impulse begins in the right ventricle and spreads across to the left ventricle and down toward the base of the heart. A pacer spike that is not followed by appropriate depolarization may indicate a problem in the pacemaker's ability to trigger depolarization. On Strips 1, 5, and 6, note the presence and the configuration of paced P waves and paced QRS complexes.
Carefully assess the ECG strip for the presence of random pacemaker spikes. Spikes are considered random if they appear anywhere in the tracing besides the programmed location. Random pacing spikes may indicate a problem with the sensing function of the pacemaker.
Finally, carefully assess the ECG strip to verify that none of the intervals between any two consecutive beats exceeds the timing interval for the low rate setting. An interval or a pause that exceeds the programmed timing interval may indicate a problem with the sensing or pacing function of the pacemaker.
When a patient is experiencing abnormally long pauses between effective heartbeats, it is often useful to calculate how long the pause lasts. By counting the squares between the R waves of two beats separated by a pause, the length of the pause in seconds or milliseconds can be determined. Find the beats marked A and B in Strip 4. Note that there are approximately 11.5 large squares between those two beats. According to this strip, the patient went for a little more than 2 seconds (11.5 x 0.20 seconds is 2.3 seconds) without an effective heartbeat.
When evaluating normal pacemaker function as well as normal cardiac conduction, it is necessary to measure various intervals. Generally, because most intervals are very short, the small squares are used to estimate any given interval. This can include:
P-R interval. The P-R interval reflects the length of time from the beginning of atrial depolarization until the onset of ventricular depolarization. The normal P-R interval is 120–200 ms (or 0.12 to 0.20 seconds).
AV interval. The AV interval is a programmed setting on a dual chamber pacemaker that most closely corresponds to the P-R interval in normal conduction. The AV interval is the length of time following atrial depolarization that the pacemaker waits to pace the ventricle. In Strip 5, the atrial pacing spike is labeled "A" and the ventricular spike is "V;" note that there are four small squares in between points A and B. Four squares multiplied by 0.04 seconds equals 0.16. The AV interval for this pacemaker is approximately 0.16 second.
Low rate timing interval. This is a programmed interval that tells the pacemaker when it needs to pace the heart. It is determined by the low rate setting on the pacemaker. Locate the pacemaker spikes labeled "C" and "D" for the two beats in Strip 6. This pacemaker's low rate setting is 75 bpm. If the pacemaker is functioning correctly, there should be approximately 0.8 second between the two paced beats, as determined by the four large squares in between the two spikes.
Before moving on to assessing the patient's response to pacemaker therapy, take a few minutes and complete the following practice exercise.
Example 1
Patient S has a VDD pacemaker implanted. The low rate setting is 60 bpm; the high rate setting is 100 bpm. Analysis of the rhythm strip indicates that the heart rate is 85 bpm. Pacer spikes are presently in front of each QRS complex. Each QRS complex is wider than normal and reflects a left bundle branch block pattern. There is no pause between any two beats that exceeds the programmed timing interval for the low rate setting. Sinus P waves are present; all P waves are followed by a paced QRS complex. All pacer spikes are followed appropriately by a QRS complex, and there are no random spikes. Does this data reflect normal VDD pacemaker function?
Answer and Rationale: Yes. A paced heart rate of 85 bpm falls within the programmed parameters of this patient's pacemaker. A VDD pacemaker is programmed to sense atrial activity and pace the ventricles in response (up to the limit of the high rate setting). The presence of sinus P waves followed by appropriately paced QRS complexes indicates that the pacemaker is sensing and pacing as programmed.
Example 2
Patient R has a DDD pacemaker implanted for management of intermittent second-degree heart block. The low rate setting is 60 bpm; the high rate setting is 110 bpm. Patient R's rhythm strip indicates a heart rate of 72 bpm. Sinus P waves are present at a regular interval. Some P waves are followed by a ventricular pacing spike and a paced QRS complex; other P waves are followed by a normal QRS complex. There are P waves in front of every QRS; every P wave is followed by either the patient's normal QRS complex or a paced QRS complex. The paced QRS complexes are broad and show a left bundle branch block pattern. The native QRS complexes are narrow. There are no random pacemaker spikes present in this tracing; every ventricular spike is followed by a paced QRS. Does this reflect normal DDD pacemaker function for this patient?
Answer and Rationale: Yes. With second-degree heart block, Patient R has normal sinus node function. The conduction abnormality lies in the AV node. The DDD pacemaker is programmed to sense the patient's sinus node activity and pace the ventricles in correct sequence at that same rate. A heart rate of 72 bpm falls within the programmed rate parameters for this pacemaker.
Example 3
Patient Q has a VVI pacemaker implanted for the management of symptomatic bradycardia caused by atrial fibrillation with a slow ventricular response. The low rate setting is 60 bpm. Does Strip 7 represent normal VVI pacemaker functioning for this patient?
Answer and Rationale: Yes. A VVI pacemaker is programmed to pace the heart when the heart's rate is either too slow or too irregular. With a low rate setting of 60 bpm, the interval between any two beats should not exceed approximately one second (or five squares). In this strip, beats 1 and 2 are paced beats at a rate of 60 bpm; beat 3 is the patient's beat, followed by a paced beat 1 second later. The fifth beat comes before the end of the programmed timing interval and is not paced; the sixth beat is paced and occurs at the end of one second.
Assessment of the patient's response to pacemaker therapy is a crucial component in the evaluation of pacemaker function. The patient's response may be the first clue that the pacemaker is not functioning properly or that the programmed settings require adjustment to optimize pacemaker function. Additionally, assessment of the patient's response may provide clues about how well the type of pacemaker and its programmed settings "match" the patient's symptoms and needs. A pacemaker may be functioning normally according to its programmed parameters but fail to meet desired clinical goals, such as increasing the patient's ability to perform routine activities of daily living, improving the patient's quality of life, or resolving the patient's troublesome symptoms. In some instances, the patient may actually develop new or worsening symptoms following the implantation of a permanent pacemaker. Patient assessment includes [20]:
Questioning and monitoring the patient for any recurrence of preimplantation symptoms. A recurrence of preimplantation symptoms may indicate a pacemaker malfunction.
Questioning and monitoring the patient for the development of new symptoms, including hypotension, dyspnea, increased fatigue, decreased activity tolerance, palpitations, or other symptoms that the patient did not have prior to pacemaker implantation. The development of new symptoms may indicate a problem known as "pacemaker syndrome," which occurs when the patient loses AV synchrony during pacing and will be discussed in detail later in this course. New or worsening symptoms indicate that the programmed settings need to be adjusted.
Observing the patient's activity tolerance and ability to perform activities of daily living without symptoms. The patient's response to activity or exercise is especially important for persons whose pacemaker has a rate responsive feature. Symptoms of activity intolerance may indicate that the rate responsive programming should be adjusted. For persons who cannot increase their heart rate when needed and who do not have a rate responsive pacemaker, signs of activity intolerance may indicate the need to revise the pacemaker to one with rate responsive capabilities.
In some cases, unraveling an apparent pacemaker malfunction requires skills and great attention. For example, Patient O is admitted for overnight monitoring following a percutaneous coronary angioplasty (PTCA) with stent placement. Patient O has a history of previous coronary artery bypass surgery and radiofrequency ablation with permanent pacemaker placement for management of atrial fibrillation refractory to medications. According to Patient O's medical records, his pacemaker is a VVIR. Following his admission to the hospital, Patient O's vital signs remain stable and he shows no signs of complications from the PTCA. Telemetry monitoring shows his heart rhythm is 100% paced at a rate that varies from 70–100 bpm depending on his activity. That evening, Patient O complains of chest pain and shortness of breath. His heart rate is 50 bpm, but his blood pressure is unchanged. The telemetry monitor indicates that Patient O's rhythm is 100% paced at a rate of 50 bpm. There is no evidence of pacemaker malfunction; there are no random pacemaker spikes or long pauses. Because Patient O is symptomatic, the physician is notified. By the time the physician arrives, the patient's paced rate has increased to 98 bpm. The physician interrogates the pacemaker generator to determine the programmed settings. The interrogation confirms that the pacemaker is programmed for a low rate limit setting of 70 bpm and a high rate responsive limit of 110 bpm. However, the interrogation produces an additional piece of information: at the time of insertion, the pacemaker had been programmed with a "sleep mode." With a sleep mode setting, the pacemaker was programmed to reduce the paced rate at a specific time in the evening and resume the more rapid rate at a preset time in the morning. The sleep mode rate is 50 bpm, and its programmed time of onset is 8:30 p.m. Because the patient experienced symptoms of decreased cardiac output at a heart rate of 50 bpm, the physician programs the sleep mode to "off." The patient experiences no further problems. What is the lesson? Not all apparent pacemaker malfunctions are due to problems with the generator or the pacing leads. Sometimes the "problem" is not a pacemaker malfunction, but a mismatch between the patient's needs and the programmed settings.
Discharge education begins prior to the implantation of a permanent pacemaker and continues through the patient's brief hospital stay, the initial follow-up appointment following discharge, and the long-term follow-up period. The major emphases of patient education during the patient's hospitalization include:
Measures to prevent infection in the incision or pacemaker pocket
Precautions to reduce the risk of injury to the pacemaker's leads or generator
Signs and symptoms that should be reported to the physician or nurse
Answers to any specific concerns or questions that the patient or family may have
During the immediate and long-term follow-up, the focus of patient education is on:
Assessment of the patient's/family's understanding of pacemaker function and management
Correction of any misperceptions, reinforcement of previously taught material, and additional explanations as indicated by patient/family assessment
Addressing specific patient/family questions and concerns that have arisen since the last follow-up appointment
Because the hospital stay following pacemaker implantation is often very short, patients need careful instructions on incision care and prevention of infection. The following points should be covered [45,51]:
Avoid getting the incision wet for the first five to seven days after you get home. You may sponge bathe but not shower during this time or until your first follow-up visit.
Keep the incision clean and dry.
After bathing, carefully pat the incision line dry. Do not rub. Do not apply creams, lotions, ointments, or powders to the incision line.
Look at the incision line every day, using a mirror if necessary. Examine the area for any signs of redness, drainage, or swelling around the incision line. Notify a nurse or physician immediately if any of these signs are present or if you develop a fever (>101°F or 38°C).
During the four to six weeks following pacemaker implantation, dislodgment of the pacemaker leads is a common complication. To reduce the risk of accidental dislodgment of the pacemaker leads, instruct the patient to follow these precautions [18,45,51]:
Avoid excessive arm movement on the side that pacemaker was implanted for at least four weeks.
Avoid vigorous activities (including golf, tennis) for four to six weeks.
Do not lift, push, or pull more than 10 pounds for the first four weeks. Activities that should be restricted include carrying groceries in from the car, carrying laundry baskets, and pushing vacuum cleaners.
Do not put pressure on the pacemaker generator site. Patients who hunt should be instructed to avoid placing the rifle stock on the same shoulder as the pacemaker pocket.
Three basic safety measures to cover with the patient and family include the pacemaker identification card, notification of other physicians and healthcare providers, and monitoring pulse rate.
At the time that a pacemaker is implanted, a temporary identification card is issued. This card lists specific information about the patient's pacemaker, including the type of pacemaker, manufacturer, date of insertion, and healthcare provider information. All patients should receive the card before discharge. Explain that the device manufacturer will mail a permanent card to the patient within a few weeks following discharge. Instruct the patient to always carry the pacemaker identification card and encourage a family member to keep and carry an additional copy. Explain that the card contains vital information that a physician would need to know to accurately assess the patient and the pacemaker function in the event of a problem [51].
Instruct the patient and family to let all other medical healthcare providers, including dentists, know that the patient has a permanent pacemaker. Some medical and dental procedures may temporarily affect the functioning of the pacemaker and will need to be modified. Other procedures, such as magnetic resonance imaging (MRI), are contraindicated for a person with a permanent pacemaker (Table 4) [53,54].
ELECTROMAGNETIC INTERFERENCE AND MEDICAL/DENTAL PROCEDURES
| No Known Risk | Possible Interference | Generally Contraindicated | |||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|
| |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||
The patient or a family member should be taught how to check the patient's pulse. When teaching a patient to check his/her pulse, consider covering these points:
Check your pulse regularly, at least once a week (or more often as directed by your physician).
Count your pulse for a full minute.
Each time that you count your heart rate, record the rate and the date in a notebook. Bring this notebook to your follow-up appointments.
Find out from your physician what your lowest acceptable heart rate is. Notify the physician if your heart rate abruptly drops to significantly less than that level.
If your pacemaker has an upper rate limit, determine what your highest acceptable heart rate is. If your heart rate dramatically increases beyond that limit, call your physician.
Your pulse might feel a little irregular. As long as your pulse rate falls within the acceptable range established by your physician, do not worry about an occasional irregularity.
Instruct the patient to notify the physician if there are any signs of infection or any other symptoms that may indicate a problem with the pacemaker's functioning, including:
Fever or signs of infection at the pacemaker pocket site (i.e., warmth, redness, drainage, excessive swelling, or pain not relieved with pain medications)
Occurrence (or recurrence) of dizziness or fainting spells
Development or increase in shortness of breath
A sudden weight gain of 3–5 pounds over 24 to 48 hours
Swelling in fingers, ankles, or feet
Swelling that occurs in the arm on the same side as the pacemaker generator
Continuous rhythmic hiccoughing
Pulse rate that falls significantly outside the acceptable heart rate range established by the physician
Patients with permanent pacemakers and their families have many concerns and questions about what type of equipment they can use and what devices might interfere with the proper functioning of the patient's pacemaker.
Many tools and electronic devices use both electricity and magnets. The combination creates an electromagnetic field around the device. The size and strength of the field varies with the device. A large piece of equipment, such as an MRI machine or an arc-welding device, generates a very strong, powerful electromagnetic field. Household items, such as microwave ovens, mixers, and electric blankets, generate a very weak electromagnetic field. When the functioning of one electronic device disrupts or interferes with the normal functioning of another device, electromagnetic interference is said to occur. A strong electromagnetic field can disrupt the functioning of a permanent pacemaker. When electromagnetic interference occurs, the settings on the pacemaker may be altered. Sensing function may be lost, and the pacing rate may revert to the magnet mode rate. The patient may experience signs and symptoms, such as dizziness, light-headedness, or a rapid heart rate. Electromagnetic interference does not permanently damage pacemaker function. To relieve the symptoms and restore normal pacemaker function, persons with pacemakers may simply move away from the offending equipment and sit down and rest until symptoms disappear [53,54].
Many patients and their families have heard a mix of accurate and inaccurate information about the impact of electromagnetic interference on the functioning of a pacemaker [53,54]. Others may be unaware of the potential risk of electromagnetic interference from the use of relatively common items. Careful, precise instructions can both help relieve the patient's anxiety and prevent unnecessary disruption of pacemaker function from electromagnetic interference. Some pacemakers are called "MR conditional," "MRI conditional," or "MRI ready" [54]. This means that they are designed to allow the patient to safely undergo an MRI scan under certain conditions, due to safeguards in the system's design that protect it from the risks of electromagnetic interference [54].
Three major areas to cover with the patient and the family are: use of cell phones, use of normal household equipment, and airport security and commercial antitheft screening devices.
Some studies have shown that cell phones may not adversely affect pacemaker functioning. However, as technology continues to advance and the use of digital technology grows, it is prudent to instruct patients to use some precautions when using or carrying a cell phone, including maintaining a distance of 6 inches between cell phones and the device. Consider covering the following points [53,54]:
Use the ear on the opposite side from the pacemaker generator when talking on a cell phone.
Carry the cell phone in a pocket away from the pacemaker pulse generator; do not carry the phone in the pocket right over pacemaker generator site.
Cordless household telephones are not the same as cellular telephones and should not interfere with pacemaker function.
Generally, a person with a permanent pacemaker can safely use normal household electrical devices as long as those devices are in good repair and properly grounded. A few devices that, when used as intended, carry no known risk to implanted cardiac devices include [23,53,54]:
Household microwaves. Pacemaker and microwave technology have advanced enough that pacemaker functioning is not usually disrupted by microwaves.
Cordless household telephones (do not place directly over device)
Laptop/desktop computers
Electrical home appliances (e.g., stove, oven, washing machine, dryer, vacuum cleaners)
Electric blankets
Electric razors and hair dryers (do not place directly over device)
AM/FM radios
Televisions, DVD players, video game consoles
Remote controls (e.g., TV, garage door, stereo, video game, camera/video equipment)
Office electrical equipment, such as printers, copy and fax machines
Lawn mowers (keep 12 inches from device), leaf blowers, snow blowers
As noted, electrical equipment in poor condition or improperly grounded may produce electromagnetic interference, and these devices are contraindicated in patients with implanted cardiac devices. The use of magnetic mattresses or chairs is also contraindicated [54].
With the heightened emphasis on security checks at airports, patients and families may have concerns about getting through airport security. Studies have shown that while airport security devices do not damage pacemakers, the security devices will detect the implanted metal and trigger the alarm. To facilitate clearance through airport security, instruct patients to [53,54]:
Always carry their pacemaker identification card.
Present the pacemaker identification card to the security agent before walking through a security screening device.
Request a hand search. Ask the security guard to avoid holding the handheld screening device directly over the pacemaker generator for a prolonged period. Handheld screening devices contain a magnet and may interact with pacemaker if held directly over pacemaker generator pocket for more than a brief period.
Commercial antitheft devices are commonly used in many department stores in the United States. As noted, these devices have the potential to temporarily disrupt the functioning of the pacemaker. Instruct the patient to walk through the security-screening device at a reasonable pace; also instruct the patient to avoid lingering in the area around the antitheft devices [53,54].
Many medical and dental diagnostic procedures will not interfere with the proper functioning of a person's pacemaker; however, some procedures have the potential to disrupt normal pacemaker functioning (Table 4). Instruct the patient to be sure that all healthcare providers know that he/she has a pacemaker before a procedure is performed. If the procedure has a risk of disrupting the pacemaker's functioning, the patient should discuss the risks and benefits of having that procedure done with the healthcare provider. In some instances, specific precautions may be taken to reduce the risk that a given procedure will disrupt the pacemaker's function. Pacemaker manufacturers also recommend that the pacemaker be interrogated to verify proper programming following performance of some procedures that have risk to generate electromagnetic interference and disrupt pacemaker programming or functioning [17,54].
The evolution of pacemaker technology has led to the development of specialized follow-up clinics staffed by trained physicians, nurses, and allied health professionals. The ideal follow-up regimen should be determined by the physician and based on the patient's needs. Factors to consider may include the patient's preferences, underlying medical conditions, geographic isolation from direct follow-up, and costs [60].
Initial follow-up goals are to:
Evaluate thoroughly the status of the pacemaker generator pocket
Assess basic pacemaker function and adjust pacing and sensing thresholds as needed
Obtain and document required patient and pacemaker data
Establish the schedule for transtelephonic monitoring (TTM) and direct evaluation appointments
The pacemaker pocket site should be carefully assessed for indications of impaired healing or signs of complications. Complications that may occur include infection in the pacemaker pocket, development of a hematoma in the pocket site, or erosion of the generator or the pacemaker lead through the tissue and skin [15]. Persons with diabetes are more at risk to develop an infection in the pacemaker pocket. Persons taking anticoagulants are at risk to form a hematoma in the pacemaker pocket [15,19]. Signs of impaired healing may include:
Subjective complaint of increased pain at generator site
Increased swelling present in pacemaker pocket area
Elevated temperature or other signs of systemic infection
Purulent drainage from incision
Failure of incision line to heal
Warmth or redness around the incision line or pacemaker pocket
While not common, infection, hematoma, and erosion are serious complications that may require surgical revision or removal of the pacemaker [19]. Erosion also generally requires systemic antibiotics. In many cases, hematomas may be treated with direct pressure and observation [15].
A chest x-ray is performed to verify that the pacemaker leads remain correctly positioned. A 12-lead ECG with and without a magnet is done to evaluate pacemaker function. The pacemaker generator may be interrogated, and data obtained. Often, the sensing and pacing thresholds are tested. By the time of the first follow-up visit, the inflammation associated with implantation of the leads has resolved, and the programmed thresholds may need to be revised [19].
During either the initial follow-up visit or a subsequent visit, the patient's underlying rhythm should be determined and the extent of the patient's dependence on the pacemaker should be assessed. A person is considered "pacemaker dependent" if he/she requires the proper functioning of the pacemaker in order to perform activities of daily living. Persons who are pacemaker dependent often have very little effective underlying or intrinsic heart rhythm. Abrupt cessation of pacing often results in significant hemodynamic compromise. Persons who are pacemaker dependent may require more frequent follow-up, as pacemaker failure would have severe consequences requiring emergency intervention [21].
Adequate clinical documentation is vital for the effective long-term management of a person with a permanent pacemaker. Accurate evaluation of the pacemaker's function can only occur when the pacemaker type and programming are known. Current patient demographics are needed in the event that a device recall occurs, and patient notification is required. Recommended information to document in the patient's file includes [19]:
Demographic data: Name, age, address, telephone number, and email address (update with each visit)
Pacemaker data: Generator model and serial number; pacemaker lead(s) model and serial number
Operative note with implant data
Examination of incision
Chest x-ray findings
12-lead ECG
Rhythm strips
Interrogation data from pulse generator
Measurement of sensing and pacing thresholds
Measurement of sensor related data
Printout of final values/current programming
Before the end of the first follow-up visit, the patient and family should receive detailed instructions about long-term follow-up. This should include the schedule for both TTM and direct evaluation visits. The patient should receive the equipment required to perform TTM at home. In addition, the patient and a family member should be instructed in the procedures for contacting the TTM facility and performing the monitoring. Written instructions should supplement verbal instructions and demonstration of the equipment. Contact numbers for both the TTM center and the follow-up pacemaker clinic should be provided.
Ongoing follow-up of the patient after permanent pacemaker implantation is crucial to the achievement and maintenance of optimal clinical results for the patient from the pacemaker therapy. The focus of postimplantation follow-up is not merely on device surveillance but, more importantly, is also on the interface and interaction of the patient and pacemaker. A policy statement developed and published by the HRS notes that appropriate medical follow-up begins with patient and family education before a pacemaker is implanted and continues throughout the patient's life (as opposed to the life of the device). Major goals of follow-up include optimization of pacemaker function, ongoing patient and family education, early identification of potential problems, and timely identification of the need for battery and/or generator replacement. Although some follow-up may be provided by the patient's primary physician, the HRS recommends that scheduled follow-up also be performed by physicians and support personnel trained in the management and evaluation of pacemaker therapy. Two major types of follow-up are generally used to evaluate and manage the patient with a permanent pacemaker: TTM and direct evaluation [19,20,22]. A third method—remote monitoring—has emerged as a complement to routine in-office care [61].
TTM has been used for more than 30 years in the follow-up and management of persons with permanent pacemakers. TTM provides a relatively inexpensive, convenient method for monitoring battery function and screening pacemaker function. There are several advantages/benefits of TTM. It permits monitoring of pacemaker function in patients who live a considerable distance from an appropriate pacemaker follow-up center. Also, studies have shown that patients are more likely to use TTM than to make an appointment at a follow-up center if they are concerned about a possible pacemaker malfunction. Another advantage is the cost of TTM; it is less costly than a direct evaluation. TTM has been found to be effective in detecting major pacemaker problems such as failure to sense and failure to pace (i.e., output failure). It can also monitor basic battery status and evaluate the battery function for indications that the battery is becoming depleted. Interrogation of the pacemaker generator cannot be done using TTM; data about the remaining voltage in the battery, programmed settings, and other specific information may only be obtained by telemetry interrogation using a programmer in a direct evaluation visit. Although TTM is useful in screening for major problems, it has limited usefulness in correction of the problem. A person-to-person appointment in a pacemaker clinic or follow-up center is usually required to correct problems discovered during TTM.
Performance of TTM is not difficult, but some training and specialized equipment are required. Reasonable hand dexterity and adequate hearing are required. For older persons or those with impaired coordination, hearing, or sight, it is helpful to train a family member to assist the patient in TTM. To perform TTM, the patient dials the monitoring facility, which may be either a pacemaker clinic or a commercial company hired to do pacemaker monitoring. A specific series of events take place when the monitoring facility answers, although the exact sequence of events may vary from facility to facility.
Trained personnel at the monitoring facility briefly interview the patient to assess the patient's overall status and identify any potential problems or concerns that the patient has about the pacemaker's function. When instructed, the patient places the telephone receiver over the transmitting equipment and ensures that the ECG electrodes are properly attached to his/her chest, fingers, or wrist. To properly transmit, the electrodes must be in good contact with the patient's skin. An ECG tracing is transmitted via the telephone to the monitoring center. The monitoring equipment in the patient's home takes the electrical signal from the ECG recording and transforms it into sound waves that can be transmitted via conventional telephone lines. Specialized equipment at the monitoring center receives the signal and converts the sound waves back into an ECG tracing.
The initial transmission lasts approximately 30 seconds. This transmission is called a "free-running" ECG; this means that the system transmits whatever the patient's ECG rhythm is at the time. Depending on the patient's degree of pacemaker dependency or frequency of pacing, this transmission may or may not show any paced beats.
Following the first 30-second transmission, the patient is cued to place a magnet directly over the generator site. This places the generator in its "magnet mode." The response of the pulse generator to a magnet application will vary depending on the manufacturer. Persons in the monitoring facility are responsible for knowing the type of pacemaker and its distinctive response to a magnet application. The most common response to application of a magnet is the inhibition of the sensing function. When the sensing function is inhibited, the pacemaker delivers pacing impulses at a preset, fixed rate. This allows for evaluation of the pacemaker's ability to fire and to capture the heartbeat. Magnet rate (i.e., pacing rate of pacemaker during application of the magnet) also provides information about the status of the battery. As the battery ages and declines in voltage, there is a decline in magnet rate. To evaluate the magnet response, the personnel at the monitoring facility should know the specific magnet response for each pacemaker.
The magnet is applied for approximately 30 seconds and then removed. A free-running tracing is then briefly transmitted. The monitoring center confirms the transmission and the patient's next TTM appointment, and the call ends. The transmitted data are stored and reviewed. The free-running tracing is analyzed to assess the patient's underlying rhythm; it also is evaluated for any signs of improper sensing by the pacemaker. The magnet tracing is used to evaluate capture, magnet rate, and pulse width (i.e., measure of the pacemaker spike in time). For dual chamber pacemakers, capture and the pulse width for both atrial and ventricular pacing are analyzed. If specific problems are identified on analysis, the patient is immediately notified by telephone and instructed to contact the pacemaker facility and/or his or her physician.
Although TTM generally works well, there are some problems associated with its use [20,21]:
TTM is limited in its ability to pick up low voltage pacemaker spikes. The transmitted ECG tracing may fail to reflect low voltage spikes, or it may incorrectly add spikes where none have occurred.
Atrial pacing is more difficult to evaluate by TTM because atrial spikes and atrial depolarization generate less voltage than ventricular pacing spikes and depolarization.
Adequate TTM transmission resulting in good tracings may be difficult. Patient factors that affect transmission include impaired coordination, which may make it difficult for the patient to apply the electrodes correctly, and impaired hearing that interferes with the patient responding to cues to start the transmission, apply the magnet, and remove the magnet. Equipment factors include the current variation in available phones. Cordless telephones may not transmit the signal as clearly as corded telephones and are at greater risk for accidental disconnection during the transmission. Speaker phones and telephones with changed volume controls can create difficulties with adequate transmission. Any source of electromagnetic interference near the telephone during transmission can cause a faulty transmission and inadequate tracing.
Frequency of TTM
In general, published recommendations for follow-up are based on clinical experience with the management of various types of pacemakers and the reimbursement guidelines for follow-up care developed by the Centers for Medicare and Medicaid Services (CMS) for Medicare reimbursement [62]. Factors that impact the prescribed frequency of TTM include:
Whether the pacemaker is a single or dual chamber pacemaker. At points during the life of the pacemaker, a dual chamber pacemaker requires more frequent follow-up than a single chamber due to the complexity of the programming and component parts.
The age of the pacemaker. More frequent follow-up is indicated in the period immediately after the pacemaker is implanted and again in the period toward the end of the generator life.
Whether the patient is pacemaker dependent. Persons who are pacemaker dependent may require more frequent follow-up.
The documented clinical track record of the specific pacemaker (including the longevity and reliability of the battery). For pacemakers that have documented clinical evidence to support that they meet certain reliability and longevity criteria, the frequency of monitoring may be decreased during part of the life of the pacemaker.
Whether the patient has a history of pacemaker problems.
The patient's medical status. Patients who develop severe underlying heart disease or whose heart disease worsens during the life of the pacemaker may benefit from increased frequency of monitoring.
In addition to regularly scheduled monitoring appointments, additional TTM is indicated when:
The patient experiences symptoms indicative of a pacemaker malfunction
The patient experiences troublesome symptoms that may or may not be directly linked to the functioning of the pacemaker
The patient undergoes a medical procedure that has the potential to disrupt the functioning or programming of the generator
Drug therapy is initiated that may affect the stimulation threshold
Table 5 outlines a commonly recommended schedule for TTM frequency based on guidelines for Medicare reimbursement established by CMS [19,20].
RECOMMENDED SCHEDULE FOR TTM
| Time Since Implantation | Monitoring Frequency: Pacemaker Meets Reliability and Longevity Documentation Criteria | Monitoring Frequency: Pacemaker Does Not Meet Reliability and Longevity Documentation Criteria | ||
|---|---|---|---|---|
| Single Chamber | Dual Chamber | Single Chamber | Dual Chamber | |
| First month following implant | Every 2 weeks | Every 2 weeks | Every 2 weeks | Every 2 weeks |
| Months 2–6 | Every 3 months | Every 3 months | Every 2 months | Monthly |
| Months 7–12 | Every 3 months | Every 3 months | Every 2 months | Every 2 months |
| Months 13–24 | Every 3 months | Every 2 months | Every 2 months | Every 2 months |
| Months 25–30 | Every 3 months | Every 3 months | Every 2 months | Every 2 months |
| Months 31–36 | Every 3 months | Every 2 months | Every 2 months | Every 2 months |
| Months 37–48 | Every 3 months | Every 2 months | Monthlya | Monthlya |
| Months 49–72 | Every 2 months | Monthlya | Monthly unless already replaced | Monthly unless already replaced |
| Months 73 and beyond | Monthlya | Monthly unless already replaced | Monthly unless already replaced | Monthly unless already replaced |
| aNeed for elective replacement of generator likely to occur for these pacemakers during this time frame. | ||||
In addition to TTM, appropriate follow-up for persons with pacemakers includes direct evaluation at regularly scheduled intervals by a physician trained in pacemaker therapy. The recommended frequency of direct evaluation is [19,20]:
Twice within the first six months following implantation for both single and dual chamber pacemaker recipients
For persons with single chamber pacemakers, annually after the first six months
For persons with dual chamber pacemakers, biannually after the first six months
Additional direct evaluation may be indicated if TTM indicates problems with the pacemaker's functioning.
The purposes of direct evaluation are to assess the patient's status and to perform a comprehensive evaluation of the pacemaker's functioning and its efficacy in managing the patient's preimplantation symptoms. The major components of the direct patient assessment include patient history, pocket site assessment, physical assessment, demographic update, and pacemaker function.
The major purpose of the patient history is to determine if the patient is experiencing any symptoms related to a decrease in cardiac output (e.g., syncope, presyncope, confusion, dizziness), symptoms similar to those the patient experienced prior to implantation, or new symptoms that might indicate a pacemaker malfunction or need to alter pacemaker programming. Obtaining an accurate history may present a challenge. Some symptoms associated with pacemaker malfunction are subtle and may be difficult to elicit from the patient. Patients' expectations of pacemaker therapy as well as their general perceptions of health and well-being will influence what symptoms they perceive and report as problematic.
The patient should be assessed for any signs of infection in the pacemaker pocket, including complaints of fever, chills, recurrent respiratory illness, or increased pain at the pacemaker generator site. The incision and pacemaker pocket should be inspected for any evidence of swelling, drainage, tenderness, or signs of erosion of the leads or the generator through the skin.
Attention is given to vital sign measurement, assessment for neck vein distention, auscultation of heart sounds, assessment of arm on same side as generator pocket for edema, and auscultation of breath sounds. Physical assessment data may indicate that the patient has developed new or worsening cardiovascular problems; if this occurs, the patient should be referred to his/her primary care provider. Worsening cardiovascular disease may also indicate a need to alter pacemaker programming to help the patient achieve as optimal a functional status as is possible.
Direct evaluations provide an excellent opportunity to provide additional or ongoing patient/family education as needed, as well as an opportunity to address the patient's or family's specific concerns related to the pacemaker therapy. It is also important to use the follow-up as an opportunity to update the patient's demographic information. In the event that a device recall occurs involving the patient, updated, current information makes it easier to notify the patient.
Emphasis should be placed on the evaluation of the pacemaker's sensing, pacing, and rate adaptive functions, the integrity of the pacemaker leads, and the status of the battery. Table 6 lists tests that may be performed to assess the pacemaker's functioning. Continued evaluation of the pacemaker-patient match is also essential. This includes evaluation of the effectiveness of the pacemaker therapy in relieving the patient's preimplantation symptoms and identification of any new or worsening symptoms that have developed since the last direct evaluation [19,20,21].
COMPONENTS OF PACEMAKER EVALUATION
| Exam | Purpose | Comments | ||||||
|---|---|---|---|---|---|---|---|---|
| Chest x-ray |
| Current pacemaker generators have unique identifiers that may be obtained radiographically; this provides a method for identifying the pacemaker manufacturer when other information (such as a pacemaker identification card) is not available. | ||||||
| 12-lead ECG without magnet |
| If patient's pacemaker only paces the heart infrequently, evaluation of the pacemaker's ability to pace and capture from a 12-lead ECG without magnet will be difficult. However, the appropriate absence of pacemaker spikes provides some clues that the sensing function is working properly. | ||||||
| ECG with magnet |
| Magnet responses vary among manufacturers. Before a magnet is applied, the physician should know the programmed magnet response of the specific pacemaker. | ||||||
| Telemetry device interrogation |
| Telemetry interrogation requires use of manufacturer-specific programmers and software/modules. To interrogate a pacemaker, the physician should know the device manufacturer and model number. Measured data on lead functioning can provide indications of possible lead fracture or problems with lead insulation. | ||||||
| Establishment of underlying rhythm | Determine if patient is pacemaker dependent | Patients who are pacemaker dependent may experience severe consequences in event of pacemaker malfunction; more frequent evaluation may be indicated. Underlying rhythm may be determined by temporarily reprogramming pacemaker to different mode and very low rate. | ||||||
| Evaluation of sensing threshold |
| Pacemaker sensitivity is set in a range from 0.5–8.0 mV/cm; the smaller the number, the more sensitive the setting. If the sensing function is too sensitive, it will mistake other stimuli for electrical activity and inappropriately inhibit pacemaker firing. If the sensing function is too insensitive, it will fail to recognize electrical activity and the pacemaker may fire inappropriately. | ||||||
| Evaluation of pulsing output | Determines lowest amount of output from pulse generator that is needed for pacemaker impulse to effectively depolarize myocardium | If the electrical output from the pulse generator is too little, the pacemaker will fire at the appropriate time but the impulse will fail to trigger the required depolarization. Optimal setting is defined as the threshold or minimum amount of output needed to trigger depolarization consistently. | ||||||
| Sensor evaluation for rate responsive feature | Determines that the rate responsive feature responds as programmed to changes in activity | A simple screening device to assess the rate adaptive response. The patient is instructed to walk two flights down and up stairs with ECG continuous rhythm strip monitoring or telemetry. Physician can evaluate rate response and adequacy of upper rate limit. | ||||||
| Treadmill stress test | Evaluates rate adaptive response for persons who engage in vigorous activity or exercise | Examines patient's and pacemaker's response to activity under controlled conditions. |
Most manufacturers now produce pacemakers that are compatible with automated, web-based remote monitoring systems [44]. Using radio transmissions, the remote monitoring system can download patient data from the pacemaker to a base unit, typically located next to the patient's bed. The base unit automatically dials a telephone-based Internet connection and transmits the data to the physician's office. The system can be programmed to download data at specific dates and times (typically while the patient is sleeping), at regular follow-up intervals, or if specific triggers (e.g., settings out of range) are activated. Remote monitoring systems can relay heart rhythm changes, device malfunction, fluid retention in the lungs, and other "interesting events," as defined by the physician and patient. Known as "exception-based management," this allows the physician to monitor and act on rhythms that are the exception to normal heart function [44].
Although not all pacemakers are equipped with the remote monitoring function, studies have demonstrated advantages to patients for whom remote monitoring is part of their long-term follow up, including high patient satisfaction, lower costs, and improved survival rates [61]. Remote monitoring has also been shown to be effective in detecting and managing clinical events in pacemaker patients, with lower consumption of a clinic's time and resources [44,63].
Pacemaker malfunctions may occur at any point during the life of a pacemaker. Malfunctions may be detected either through routine TTM, remote monitoring, or during extensive evaluation of pacemaker function during an annual or biannual direct evaluation visit. Indications of pacemaker malfunctions may also be detected initially during routine medical appointments or hospitalizations for the management of conditions not related to the pacemaker. The most common pacemaker problems include end of battery life, failure to capture, failure to pace, and failure to sense.
With early pacemakers, the end of battery life often occurred abruptly and unpredictably, resulting in a dramatic, detrimental reduction in pacing rate and output voltage. Emergent replacement of the generator was required. With the advances in pacemaker technology, the end of battery life is a gradual and expected event. Through the use of TTM and pacemaker generator interrogation, the physician is able to watch the progression of battery usage and measure specific indicators that point to the need to replace the generator and battery within a few weeks to months before problems develop. These indicators have been termed "elective replacement indicators." Elective replacement indicators are device specific; information about the indicators for a specific device should be obtained from the manufacturer. Various indicators may be used. The most common is a gradual decline in the rate of the pacemaker when a magnet is applied. Other indicators include a change in pacing mode; for example, a VVIR may change to VVI when the battery is nearing depletion. Even with the new technology and improved pacemaker batteries, unexpected catastrophic battery failure is possible. Catastrophic battery failure results in gross, complete malfunctioning of the pacemaker and severe patient compromise for those who are pacemaker dependent. Emergent treatment with an external or temporary pacemaker is usually required until the pacemaker generator can be replaced [4,19,64].
Failure to capture occurs when a correctly delivered pacing impulse fails to depolarize the heart. On ECG tracing, the pacemaker spike appears at the proper time, but it is not followed by the expected P wave or QRS complex. In the initial period following pacemaker implantation, failure to capture may be caused by dislodgment of the pacemaker leads or poor positioning of the electrodes on the lead tip. Later in the life of the pacemaker, failure to capture may be caused by [15]:
An increase in fibrotic or scar tissue at the site of the lead implantation. Because scar tissue conducts electrical impulses poorly, an increase in voltage or output may be required for capture to occur.
Fracture of the pacemaker lead. Although pacemaker leads are designed to be extremely flexible and to withstand considerable motion and twisting, the wires may still fracture. Fracture of a pacemaker lead prevents the impulse produced by the generator from being transmitted to the electrode at the lead tip.
The connection between the pacemaker lead and the generator may have become loosened. The end of the pacemaker lead fits into the connector block on the outside of the generator. If this connection loosens, the electrical impulse will not effectively be transmitted to the electrode on the tip of the lead.
Damage to the insulation on the lead. A break in the insulation also interferes with the effective delivery of the electrical impulse to the myocardium.
Battery failure. This results in the generation of an impulse that is too low in voltage to depolarize the heart effectively.
Effects of metabolic changes or medications that change the effective threshold for capture and require an increase in voltage for depolarization to occur. Metabolic abnormalities that can affect capture threshold include hyperkalemia, hypercarbia, acidosis, alkalosis, severe hyperglycemia, or hypoxemia. Drugs associated with the development of an increased capture threshold include beta-adrenergic blockers, flecainide, quinidine, amiodarone, and procainamide. Reprogramming the capture threshold may correct the problem; however, in some cases, the metabolic abnormality should be corrected before the capture threshold can be effectively reprogrammed.
Myocardial ischemia and MI.
Depending on the cause, failure to capture may occur infrequently, intermittently, or continuously. The effects of the failure to capture depend on how frequently the pacemaker fails to capture, the extent to which the patient is pacemaker dependent, and the effectiveness of the patient's underlying rhythm in maintaining cardiac output. Persons who are not pacemaker dependent or who have only infrequent failure to capture may experience symptoms such as bradycardia, shortness of breath, hypotension, dizziness, weakness, or reoccurrence of preimplantation symptoms. Persons who are pacemaker dependent may experience severe bradycardia or asystole. Emergency measures such as external noninvasive or temporary transvenous pacing may need to be employed until the specific cause of the pacemaker malfunction can be identified and corrected. Often, revision of the pacemaker leads and/or generator is required to correct the causes of failure to capture. An apparent failure to capture may also be caused by inappropriate sensing or inappropriate timing intervals. If the pacemaker fires during the refractory period, the pacing impulse will not produce depolarization. Reprogramming the timing intervals or troubleshooting the sensing function is indicated [15,20,21,23].
Failure to pace (also referred to as failure to output) occurs when the pacemaker should deliver a pacing impulse but does not. On ECG tracing, failure to pace is characterized by a prolonged interval between two beats that exceeds the timing interval for the low rate limit. Clinically, failure to pace can result in pauses and a bradycardic rate that may precipitate symptoms of decreased cerebral perfusion, including syncope, near syncope, confusion, and dizziness. Common causes of failure to pace include battery failure, fracture or dislodgment of the pacemaker lead, a loose connection between the pacemaker generator and the pacemaker lead, generator or circuitry failure, and oversensing. Oversensing occurs when the sensing circuit inappropriately interprets some other stimulus as cardiac depolarization and inhibits delivery of a pacing impulse. A common cause of oversensing is electromagnetic interference. Failure to pace may occur intermittently, or it may occur 100% of the time. The effect on the patient depends on the frequency of the failure to pace and the patient's degree of dependence on the pacemaker to maintain an effective cardiac output. If the failure to pace is caused by oversensing, the problem may be temporarily corrected by putting the pacemaker into magnet mode. In an emergency, this may be performed through TTM. When the pacemaker is placed in magnet mode, the sensing function is turned off and the pacemaker fires asynchronously at a fixed rate. Permanent correction of failure to pace may require noninvasive reprogramming of the pacemaker's settings or the replacement of the pacemaker generator or leads [15,21,23].
Failure to sense (i.e., undersensing) occurs when the pacemaker fires randomly at any point during the cardiac cycle instead of at the indicated or appropriate time. In failure to sense, the pacemaker fails to detect previous electrical activity and the pacemaker fires inappropriately. The ECG tracing shows random pacemaker spikes appearing throughout the ECG tracing. Failure to sense causes the pacemaker to compete with the heart's intrinsic rhythm. The presence of random atrial spikes does not usually present a major problem; these spikes may interfere with the optimal functioning of the pacemaker, but because they are low in voltage, they do not usually precipitate the development of a chaotic electrical rhythm in the heart. The presence of random ventricular spikes presents more of a problem. Under certain circumstances, such as in the presence of myocardial ischemia or acute MI, random pacemaker spikes falling at a vulnerable point in the cardiac cycle may trigger an erratic, ineffective rhythm. Failure to sense may be caused by multiple factors; some of these are not true malfunctions but are the result of pacemaker programming. For example, a paced beat is normally followed by a period in which the pacemaker is refractory and does not sense any intrinsic electrical activity. If a premature beat occurs during the pacemaker's refractory period, the pacemaker may not sense it and may appear to fire "early." The presence of multiple or frequent pacemaker spikes that occur randomly throughout a tracing are an indication of a true malfunction. Causes of failure to sense include poor electrode position or dislodged electrode, break in the pacemaker lead's insulation, battery failure, inappropriate programming of sensitivity of the pulse generator, a nondocumented change in programming to asynchronous mode, or MI. If failure to sense is caused by programming problems, noninvasive reprogramming of the pacemaker's settings may correct the problem. A failure to sense caused by problems with the generator, circuitry, or leads requires replacement of the broken component [21,23,64].
Using your knowledge of normal antibradycardia pacemaker function, assess the following cases:
The patient has a DDD pacemaker implanted. The low rate setting is 50 bpm, the high rate setting is 120 bpm (see Strip 8). Based on the information provided, Strip 8 shows normal DDD pacemaker function. There is a pacemaker spike in front of each P wave; each paced P wave is followed by a paced QRS complex. The rate on the strip is 80 bpm and falls within the programmed parameters.
This patient has a VVI pacemaker; the low rate setting is 75 bpm (see Strip 9). Strip 9 shows a failure to capture. Following beat 4, there are two pacemaker spikes that are not followed by ventricular depolarization.
This patient has a VVI pacemaker; the low rate setting is 75 bpm (see Strip 10). With a VVI pacemaker set at 75 bpm, the patient should not experience a prolonged pause or an interval between any two beats that exceeds 0.8 second (or four large squares). On Strip 10, the patient has a pause of a little more than two seconds followed by some type of escape beat. The pacemaker has failed to fire a pacing impulse as programmed.
First identified in the 1970s, pacemaker syndrome is a complex of signs and symptoms that occurs when AV synchrony is lost during pacing and relieved when AV synchrony is restored. Once thought to occur solely with VVI pacing, pacemaker syndrome has also been shown to occur under certain circumstances with other types of pacemakers [65]. Pacemaker syndrome should be suspected any time the patient experiences a recurrence of preimplantation symptoms in the presence of a well-functioning pacemaker. The definition of pacemaker syndrome varies; some define it in terms of the presence of syncope, presyncope, or malaise. Others suggest that pacemaker syndrome may be present in a patient who fails to achieve his or her optimal functional status following pacemaker implantation. Pacemaker syndrome may be defined as a constellation of specific symptoms that occur in the setting of a temporary or permanent pacemaker and result from loss of physiologic timing of atrial and ventricular contractions [65]. The exact incidence of pacemaker syndrome is unclear; estimates range widely from 7% up to 83% depending on which definition of pacemaker syndrome is applied [19,65].
The primary cause of pacemaker syndrome appears to be the loss of AV synchrony (i.e., AV dyssynchrony) that occurs with pacing [65]. When AV synchrony is lost, atrial contraction and ventricular contraction are not coordinated. Active ventricular filling that normally occurs when the atria contract just before ventricular depolarization cannot occur; left ventricular filling is decreased, and cardiac output drops. It is estimated that up to 20% of an individual's cardiac output comes from active ventricular filling. With the drop in cardiac output, the patient may experience signs and symptoms similar to those he/she experienced prior to implantation. These may include syncope, presyncope, confusion, dizziness, or light-headedness. In addition, when AV synchrony is lost, atrial contraction may occur during the time that the mitral and tricuspid valves are closed. Atrial contraction against closed valves results in an increase in volume and pressure in the atria, which in turn may trigger a vagal response resulting in syncope, presyncope, and hypotension. The elevation in atrial and pulmonic pressures also may result in symptoms of congestive heart failure [19,20,21,65].
Symptoms associated with pacemaker syndrome are linked to either the drop in cardiac output or the elevation in atrial and pulmonic pressures. Common signs and symptoms include [19,20,21,65]:
Syncope or near syncope
Confusion
Malaise or fatigue
Weakness
Light-headedness
Dizziness
Shortness of breath at rest or with exertion
Orthopnea
Paroxysmal nocturnal dyspnea
Uncomfortable sense of "fullness" in neck or chest
Palpitations
Chest discomfort
Cough
Intermittent or persistent hypotension
Fluctuating neck vein distention accompanied by giant ("cannon") A waves
Crackles in lung fields
Peripheral edema
Orthostatic hypotension present during pacing
As previously noted, pacemaker syndrome was originally thought to occur solely with ventricular (VVI/VVIR) pacing. Because the VVI/VVIR pacemaker does not sense electrical activity in the atrium, ventricular pacing is not coordinated with atrial activity and AV synchrony is lost. However, it has been determined that pacemaker syndrome may also occur in dual chamber pacing under certain circumstances [65]. One cause of pacemaker syndrome in dual chamber pacemakers is an inappropriately programmed setting for the atrioventricular delay. If this delay is too long, it may interfere with coordinated atrial and ventricular contraction. Another cause is the presence of retrograde pathways from the AV node back to the atria. With retrograde conduction, impulses travel from the ventricles back across the AV node to the atria and trigger atrial depolarization. Because retrograde conduction causes atrial depolarization to occur after ventricular depolarization, loss of AV synchrony occurs [65]. In addition, atrial contraction may occur against closed mitral and tricuspid valves. Inappropriate mode switching in DDD/DDDR pacing can also cause pacemaker syndrome. Inappropriate mode switching from dual chamber pacing to ventricular pacing can occur when the pacemaker interprets some type of artifact (e.g., muscle artifact) as the onset of atrial fibrillation. Pacemaker syndrome may also occur with AAI/AAIR pacing when the pacemaker paces the atria rapidly in the presence of AV block. A rapid atrial rate may interfere with optimal filling of the atria so that ventricular filling and cardiac output are reduced. In the presence of AV block, this is further complicated by the slowed ventricular rate that can occur [65]. Finally, pacemaker syndrome may occur with rate responsive pacing in persons with underlying cardiac disease who cannot tolerate the increase in pacing rate. Patients at risk to develop pacemaker syndrome include [65]:
The elderly, as older persons are more likely to develop symptoms from the loss of AV synchrony
Persons with retrograde conduction
Persons with left ventricular disease
Patients with sinus rhythm, as persons who are normally in sinus rhythm are more likely to feel the effects from the drop in cardiac output caused by the loss of AV synchrony
Patients with single chamber ventricular pacemakers
Symptoms of pacemaker syndrome may be hard to elicit from a patient; some persons have the misperception that feeling less than well is an unalterable result of having a permanent pacemaker implanted. Careful questioning about specific symptoms is usually necessary. Pacemaker syndrome is diagnosed by correlating symptoms with pacing periods. The diagnosis may be confirmed by linking a fall in systolic blood pressure and pulse pressure with periods when the patient's rhythm is paced. Another confirmatory sign is the presence of huge A waves in the jugular veins during periods of paced rhythm. The presence of these clinical signs is usually sufficient to confirm the diagnosis of pacemaker syndrome; however, the clinical symptoms alone may be considered inadequate to justify reimbursement for a second operative procedure and new pacemaker by third party payers. Some reimbursement guidelines require substantial documentation that the normally functioning pacemaker is indeed creating hemodynamic limitations for the patient. Additional tests may be required to document the presence of compromised cerebral perfusion during pacing intervals. To facilitate reimbursement, the medical record should clearly document the test performed and the pertinent findings [19,20,21,65].
Pacemaker syndrome caused by VVI/VVIR pacing may be treated by reprogramming the low rate setting so that the paced beats occur less frequently or the pacing periods are shorter in length. Although decreasing the rate or frequency of VVI pacing may be helpful, the decrease in rate may result in decreased activity tolerance. More often, effective treatment of pacemaker syndrome requires replacing the VVI/VVIR pacemaker with a dual chamber device. For pacemaker syndrome caused by the programmed AV delay in VDD or DDD pacemakers, reprogramming the delay to a shorter interval may relieve the symptoms. If automatic mode switching is the culprit, reprogramming may also help. For pacemaker syndrome caused by an intolerance of a high atrial rate or high upper rate setting, the rate may be reduced to relieve the symptoms [19,20,21]. Alteration of the patient's medication regimen may also be indicated. Supportive care related to possible heart failure, hypotension, tachycardia, tachypnea, and oxygenation deficit may also be necessary [65].
Heart failure continues to be a major healthcare concern in the United States [68]. Management of heart failure is a major consumer of healthcare resources; it is estimated that more Medicare dollars are spent on the management of heart failure than any other diagnosis. For the individual, heart failure results in a decreased quality of life, frequent hospital admissions, and poor overall prognosis. Heart failure worsens as the disease progresses and prescribed medications become less and less effective. Other treatment options, such as heart transplant and ventricular assist devices, are available to only a small percentage of people [8,69,70].
As noted, treatment varies depending on the progression and stage of heart failure. The New York Heart Association (NYHA) uses a s simple classification system, based on limitations during physical activity, to determine the extent of heart failure. The NYHA classification system, which informs several authoritative guidelines, outlines the stages of heart failure as [67]:
Class I: No symptoms and no limitation in ordinary physical activity (e.g., shortness of breath when walking, climbing stairs, etc.)
Class II: Mild symptoms (mild shortness of breath and/or angina) and slight limitation during ordinary activity
Class III: Marked limitation in activity due to symptoms, even during less-than-ordinary activity (e.g., walking short distances [20—100 m]). Patient is comfortable only at rest.
Class IV: Severe limitations. Experiences symptoms even while at rest. Mostly bedbound.
Advances in pacemaker technology have provided a new therapy option for patients with severe heart failure. Cardiac resynchronization therapy (CRT), also referred to as atrial-synchronized biventricular pacing or simply biventricular pacing, has been found to be effective in persons with NYHA Class III or Class IV heart failure who meet certain criteria [7,71,72,73]. Consider the following clinical example:
Patient N, a man 66 years of age, presents to his physician's office complaining of increased shortness of breath, rapid weight gain over the last three to four days, and an inability to perform his usual daily activities due to fatigue. Patient N has a history of MIs, coronary artery bypass surgery, acute coronary syndrome, multiple stent placements, and heart failure. Until recently, Patient N's heart failure has been well managed through the use of medications and diet. However, over the last month, Patient N has been admitted to the local hospital twice for management of his heart failure symptoms.
This patient's experience is not unique. Heart failure is a progressive and debilitating disease that results in worsening symptoms, declining quality of life, and eventually death. In the past, medical therapies focused on the goal of improving blood flow through the heart to control or improve symptoms. Medications such as vasodilators, diuretics, and digoxin were prescribed along with diet modification and lifestyle changes. These measures were often effective for a time in managing symptoms but did not halt the progression of the disease. Later research showed that a number of neurohormonal processes are activated by the body when the heart begins to fail. Briefly, when the heart is not able to provide blood flow to the body to meet its metabolic needs, neurohormonal processes are activated that cause the ventricles in the heart to change shape and structure. These changes, which are often referred to as ventricular remodeling, damage the heart muscle further and result in the worsening signs of heart failure, declining quality of life, and eventually death. Medications, such as angiotensin-converting enzyme inhibitors (ACE inhibitors) and angiotensin II receptor blockers (ARBs), are frequently used to counteract the neurohormonal processes and slow the destructive processes of ventricular remodeling [8,69,71].
In the last several years, researchers have noted that one of the changes that can occur with ventricular remodeling is disruption of the normal coordinated contraction of the left and right ventricles. This uncoordinated contraction, known as ventricular dyssynchrony, causes a further drop in cardiac output, results in increased symptoms of heart failure, and triggers processes that result in still more destructive ventricular remodeling [8,69]. Researchers hypothesized that if ventricular synchrony could be restored so that coordinated contraction of the ventricles could occur, the symptoms of heart failure would improve, and ventricular remodeling might be slowed, stopped, or even reversed.
Biventricular pacing has shown great promise as a therapy to restore coordinated contraction of the right and left ventricles [74]. With biventricular pacing, carefully timed pacing impulses are sent to both the right and left ventricles to stimulate coordinated contraction. In several large clinical trials, the use of biventricular pacing in select populations with NYHA class III and class IV heart failure has been shown to be effective in reducing symptoms, increasing activity tolerance, and improving quality of life (Table 7) [8,69,70,75,77,88]. According to a published analysis of clinical trial data, biventricular pacing is equally effective in the management of both men and women with heart failure who meet the criteria for implantation of a biventricular device [76].
CARDIAC RESYNCHRONIZATION THERAPY AND RELATED CLINICAL TRIALS IN PATIENTS WITH CLASS III AND CLASS IV HEART FAILURE
| Clinical Trial | Focus | Overview of Study Population | Brief Summary of Findings | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PATH-CHF Trial (Pacing Therapies for Congestive Heart Failure Trial) | Designed to evaluate the acute hemodynamic effects and long-term clinical benefits of right ventricular, left ventricular, and biventricular pacing in target population |
| Showed trend toward improvement in functional status, quality of life, and maximum oxygen consumption | |||||||||||
| MUSTIC study (Multisite Stimulation in Cardiomyopathy study) | Designed to evaluate CRT in patients with heart failure |
|
| |||||||||||
| MIRACLE Trial (Multicenter InSync Randomized Clinical Evaluation Trial) |
|
| Patients randomized to CRT group showed reduced hospitalization and improvement in NYHA functional class, quality of life, and in 6-minute walk. | |||||||||||
| COMPANION Trial (Comparison of Medical Therapy, Pacing, and Defibrillation in Heart Failure Trial) | Multicenter prospective randomized trial that compared drug therapy alone to drug therapy plus CRT with or without an implantable cardiodefibrillator (ICD) |
| Patients receiving CRT alone or CRT with ICD therapy showed a decrease in all-cause mortality and all hospitalization |
A biventricular pacing system consists of two parts [81]:
A pulse generator that holds the power source (i.e., battery) and the electronic circuitry needed to generate and deliver pacing impulses to the heart
Pacemaker leads that can sense electrical activity and deliver pacing impulses to the heart as programmed. Unlike a traditional dual chamber pacemaker, which uses two pacemaker leads (one in the right atrium and one in the right ventricle), a biventricular system uses three leads: one for the right atrium, one for the right ventricle, and one for the left ventricle.
Placement of a biventricular pacemaker is, in many ways, similar to the placement of a standard antibradycardia pacemaker. The pacemaker generator is generally implanted in a pacemaker pocket created in the subcutaneous tissue located on top of the pectoralis muscle. The right atrial and right ventricular leads are inserted using a standard transvenous approach and attached to the endocardial surface of the right atrium and right ventricle respectively [81].
The major difference in implantation of a biventricular pacemaker is the placement of the left ventricular lead. Left ventricular leads are technically more difficult to place than leads in either the right atrium or right ventricle; proper positioning of the left ventricular lead is crucial to effective biventricular pacing [82]. Left ventricular leads are not placed on the endocardium. Instead, they are inserted transvenously and passed to the coronary sinus, a structure that drains blood from the epicardial surface of the heart [81]. The left ventricular lead is passed through the coronary sinus into one of the venous branches lying over the left ventricle. It may be helpful to obtain a venogram of the area to direct the selection and placement of this lead [81]. The left ventricular pacing lead is typically secured either with active fixation (tines) or with passive fixation using the multiple curves of the lead to fit it tightly in the target vein. An active-fixation unipolar lead is available whose tines can be extended from the body of the lead and retracted if repositioning is necessary [81]. In some patients, transvenous placement of the left ventricular lead in the proper position cannot be achieved. The physician may offer these patients the option of undergoing a surgical procedure (i.e., minithoracotomy) for placement of the left ventricular lead [83].
Two independent, randomized prospective studies have demonstrated promising outcomes when left ventricular lead placement was targeted away from the scar and toward sites of the latest mechanical activation. The Speckle Tracking Assisted Resynchronization Therapy for Electrode Region trial was designed to test whether improved CRT outcomes would be gained by using echo-guided transvenous left ventricular lead placement at the site of latest mechanical activation [84]. The primary endpoint was first hospitalization for heart failure or death. Of 187 patients with NYHA class II to IV, 110 were randomized to echo-guided approaches and 77 to routine strategies. Exact or adjacent lead placement was achieved in 85% of the echo-guided group and was associated with improved event-free survival [84].
The Targeted Left Ventricular Lead Placement to Guide Cardiac Resynchronization Therapy (TARGET) study was conducted to assess the impact of targeted left ventricular lead placement on CRT outcomes [85]. A total of 220 patients were randomized into two groups. In group 1, the lead was positioned at the latest scar-free site of peak contraction using speckle-tracking echocardiography. Compared with standard unguided CRT, the use of speckle-tracking echocardiography to target lead placement yielded significantly improved CRT response and lower rates of combined death and heart failure-related hospitalization [85].
For biventricular pacing to be effective in producing coordinated contraction of both right and left ventricles, the pacemaker must achieve 100% capture in both ventricles. Initially, the sensing electrode in the right atrium monitors for normal atrial electrical activity. If it senses normal electrical activity, it relays that information to the generator. The atrial pacing circuit is inhibited, and the pacemaker does not deliver a pacing impulse through the atrial pacing lead. If the atrial sensing electrode does not sense normal electrical activity within the preprogrammed interval, the generator delivers a pacing shock through the atrial pacing lead to the right atrium to stimulate depolarization.
After the atria have been depolarized, the generator delivers carefully timed pacing shocks to both the right and left ventricles to stimulate coordinated contraction. Two programmed settings are crucial to produce coordinated contraction. The first is the timing of the electrical stimulation to each ventricle. Initially, electrophysiologists programmed biventricular pacemakers to stimulate both ventricles simultaneously. The newest generation of biventricular pacemakers can be programmed to stimulate one ventricle slightly before the other. The optimal timing of ventricular contraction has not yet been determined. Electrophysiologists may use echocardiography to determine the best timing sequence for each patient [83].
The second vital setting is the length of the atrioventricular delay. Atrioventricular delay is a programmable setting; it determines the interval of time that elapses between atrial and ventricular depolarization. To help ensure 100% capture of both right and left ventricular depolarization, the programmed atrioventricular interval should be shorter than the heart's intrinsic atrioventricular delay [83]. Echocardiography can be used to guide the programming of the atrioventricular delay setting for each person [83].
Normal pacing patterns for biventricular pacemakers are different than the pacing patterns for traditional dual chamber antibradycardia pacemakers. In biventricular pacing, a P wave should be present before each beat. This P wave might be the patient's intrinsic P wave, or it might be paced with a pacemaker spike present before the P wave. Every P wave (either paced or intrinsic) should be followed by a paced QRS complex. Depending on the model of biventricular pacemaker, there may be a single ventricular spike before the QRS complex, or there may be two spikes. Strip 11 is an example of a biventricular pacing pattern. Note the atrial and ventricular pacing spikes.
Based on data from major clinical trials, a patient should meet the following criteria to be eligible for biventricular pacemaker implantation [7,8,71,81]:
A left ventricle ejection fraction of 35% or less
NYHA class III functional or class IV ambulatory heart failure
Persistently symptomatic despite optimal drug therapy
Presence of ventricular dyssynchrony, defined as a QRS duration that is greater than or equal to 0.12 sec (or 120 ms)
The use of biventricular pacing for persons who meet these criteria has received a class I recommendation in the ACCF/AHA heart failure guidelines as well as in the ACCF/AHA/HRS pacemaker implantation guidelines [22,28,86]. In light of accumulating evidence, the ACCF/AHA/HRS modified the NYHA class I indications for CRT in 2012 to include patients with class II symptoms and left-bundle-branch block with a QRS duration of 150 ms or less [88,89,90,91]. Additionally, guidelines from the European Society of Cardiology, developed in collaboration with the European Heart Rhythm Association, indicate that decisions to use CRT should be influenced by specific clinical manifestations of bradyarrhythmias in patients with [92]:
Persistent bradycardia (including sinus node disease and AV block)
Electrocardiographically documented intermittent bradycardia; and
Suspected (i.e., electrocardiographically undocumented) bradycardia
Initially, biventricular pacemakers were only approved for use with persons who were in normal sinus rhythm and did not require antibradycardia pacing or device therapies for ventricular tachycardia or ventricular fibrillation. Devices are now approved for biventricular and antibradycardia pacing and may be combined with an implantable cardiodefibrillator (ICD) [28].
In addition to the criteria established by the clinical trial data, some electrophysiologists also require that the patient show evidence of ventricular dyssynchrony on echocardiogram [93].
Preoperative preparation of the patient for transvenous insertion of a biventricular pacemaker is very similar to the preparation previously discussed for antibradycardia pacemakers. The same precautions and patient education are applicable in this case.
In addition to the usual risks associated with pacemaker implantation, implantation of a biventricular pacemaker carries additional risks specifically related to the left ventricular lead placement, including dissection or perforation of the coronary sinus [81]. Although these complications have been rare, their occurrence results in significant morbidity and mortality [7].
Care of the patient following successful biventricular pacemaker implantation is similar to that for patients who have undergone antibradycardia pacemaker implantation. Vital signs are monitored, and the pacemaker pocket is assessed for signs of bleeding or hematoma formation. The patient's ECG is continuously monitored to evaluate the pacemaker's function. Due to the sophistication of today's pacemakers and the complexity of the programming involved, the assessment and evaluation of biventricular pacing is the responsibility of a specially trained physician [71]. Nurses caring for the patient in the postoperative period should be alert to signs and symptoms that indicate a potential problem with the pacemaker, its leads, or its programming. Pertinent signs and symptoms include [69,83,94]:
Complete absence or intermittent absence of a ventricular pacing spike. To be effective in restoring ventricular synchrony, a biventricular pacemaker should achieve and maintain 100% ventricular capture. Loss of the ventricular pacing spike may indicate a problem with the pacemaker's programming and the atrioventricular delay interval.
Intermittent or complete loss of ventricular capture. As always, the loss of capture may indicate problems with one or more of the leads.
Decrease in duration of QRS complex. Effective biventricular pacing should shorten the amount of time it takes for the ventricles to depolarize, resulting in a decrease in QRS duration.
Sudden increase in QRS duration back to the patient's preimplantation baseline. A sudden increase in the duration of the QRS may indicate that synchronized ventricular contraction is no longer occurring. Loss of synchronization in the immediate postimplantation period is usually due to a loss of left ventricular pacing. Causes may include fracture or dislodgment of the left ventricular lead, a loose connection between the pacemaker generator and the pacemaker lead, or circuitry failure.
Practice Exercise
Patient AA has had a dual chamber biventricular pacemaker implanted. The patient required both antibradycardia pacing for sick sinus syndrome and biventricular pacing for CRT. The programmed rate parameters are 50 bpm (low rate setting) to 120 bpm (high rate setting). The device does not include an ICD. Patient AA is placed on telemetry, and it is determined that she is 100% AV paced. Several hours later, Strip 12 is obtained. Does this strip show normal biventricular pacing function? Why or why not? What action should be taken?
Answer: This strip does not show appropriate biventricular pacing. Pacer spikes in front of the P wave indicate that the atria are being paced. However, there are no pacemaker spikes in front of the QRS complexes; the ventricles are not being paced. In biventricular pacing, the ventricles should be paced 100% of the time. The physician should be notified. Evaluation of the programmed settings and pacemaker functioning is indicated.
When Left Ventricular Lead Placement Is Unsuccessful
On occasion, the physician will be unable to successfully position the left ventricular lead through a transvenous approach. Medical management options include:
Re-attempting transvenous placement of the left ventricular lead at a later time
Referring the patient to the appropriate surgical service for surgical placement of the left ventricular lead
Recommending that no further attempts be made to implant the left ventricular lead
Prior to the patient's discharge, the physician will evaluate the effectiveness of the pacemaker's programmed settings. An echocardiogram may be performed to assist the physician in identifying and programming optimal settings for each patient [71].
Discharge Education
Key points in discharge education following biventricular pacemaker implantation include [69,71,94]:
Incision care and temporary restrictions
Basic safety measures
Adherence to medication therapy and diet restrictions. Stress to the patient that biventricular pacing does not replace other therapies for the management of heart failure; adherence to prescribed medications and diet restrictions is still necessary.
Self-monitoring of symptoms. Persons who respond to CRT will experience gradual improvement in activity tolerance, quality of life, and activities of daily living. Instruct the patient to note his or her current symptoms and current activity level. Encourage the patient to note even small improvements in his/her symptoms and exercise tolerance. Instruct the patient to notify his/her physician if symptoms either abruptly worsen or return to preimplantation levels. An abrupt increase in symptoms may indicate a loss of resynchronization. Loss of resynchronization can be caused by lead failure or lead displacement (most commonly the left ventricular lead), device malfunction, or problems with the programmed intervals.
Long-term follow-up focuses on device surveillance and monitoring of the patient's clinical status. The biventricular pacemaker should be interrogated on a regular basis to verify battery status, lead status, and appropriate device functioning. Sources differ in the recommended frequency of monitoring. Some recommend interrogation as often as every three months; others recommend a six-month interval [15,71,83].
During follow-up visits, the patient's symptoms and overall disease progression should be assessed. Indications that the patient is responding favorably to the CRT include increased exercise tolerance, increased ability to participate in activities of daily living, improved quality of life, fewer unscheduled physician or urgent care visits, fewer hospitalizations or emergency department visits for symptom management, and shortened length of stay when hospitalization is required. The patient's adherence to prescribed medical therapies, including medications, diet, and lifestyle changes, should also be assessed [70].
For patients who do not respond to CRT as expected or who are responding very slowly, additional medical interventions may be employed. The physician may choose to increase the prescribed doses of beta blockers and/or ACE inhibitors. Patients with biventricular pacemakers may be able to tolerate increased doses of these medications; the increased doses will further block the neurohormonal processes that cause worsening symptoms. As a result, the patient may experience a gradual reduction in symptoms and an increase in activity tolerance. The physician may prescribe measures to increase physical reconditioning. Some patients, particularly those with NYHA Class IV heart failure, are debilitated and require very slow, gradual increases in physical activity to reverse the deconditioning effects of their disease [70].
Patients should be assessed for any problems or symptoms that indicate a loss of ventricular resynchronization. These symptoms can be overt, such as an obvious worsening of heart failure symptoms and decrease in exercise tolerance, or they may be more subtle, consisting of vague complaints of increasing weakness and fatigue. If symptoms are present, assessment of pacemaker function, including the left ventricle pacing threshold and left ventricle capture, should be performed. The programmed AV interval should be evaluated to see if it is too long, thus allowing the patient's intrinsic conduction to trigger ventricular depolarization. The patient's ECG should be assessed for the presence of increasing premature ventricular contractions (PVCs). Frequent PVCs can inhibit the pacemaker from firing and will result in a loss of ventricular synchrony. If increasing PVCs are present, the patient's overall status should be assessed to determine the cause. The physician may consider prescribing antiarrhythmic medications to limit or stop the PVCs. Increasing symptoms of heart failure can also be an indication that the patient's disease is continuing to progress toward its end stage [7].
Biventricular pacing remains a major focus in pacemaker research. Other areas that have been identified for further research include [5,28,71,75,78,79,80,87,93]:
Use of echocardiography to determine presence of ventricular dyssynchrony in patients with a narrow QRS complex
Use of CRT in persons with NYHA class II heart failure to improve ejection fraction and reverse destructive left ventricular remodeling
Better identification of persons who will respond to CRT
Exploration of the effects of right ventricular pacing on left ventricular function and the development of left ventricular dyssynchrony
Improvement of pacemaker design, function, lead systems, and battery life
Pacing in the bundle of His
The role of pacing among patients with transient bradycardia with reflex-mediated syncope
The role of CRT in patients with an LVEF of >35%
In 2014, the FDA cleared for sale two new CRT devices that are thinner and smaller than other implantable devices [6]. In 2016, the FDA approved the first leadless pacemaker to treat heart rhythm disorders; expanded FDA approval was later granted for the novel use in the stimulation of the bundle of His (in 2018) and for patients with AV block (in 2020) [55,56,66]. The device works like other pacemakers to regulate heart rate; however, the device is self-contained, only 1 inch long, and is implanted directly in the right ventricle of the heart. In 2023, the FDA approved the first dual-chamber leadless pacemaker that allows wireless communication and synchronization between two leadless pacemakers that are smaller than a AAA battery [57]. Additionally, genetic therapy and devices not requiring surgery or incisions are being investigated in the United States [58,59].
Patient BB is a woman, 74 years of age, who presents to her physician's office with complaints of dizziness and recent unexpected falls. She reports that she experiences symptoms once every week or so. When questioned about her falls, she states that she thinks she "blacks out for a second" but "comes to fast" and "knows where she is." She denies any injury from the falls. On admission, her blood pressure is 154/86 mm Hg, her heart rate is 68 bpm and irregular, and her respiratory rate is 14. Patient BB is alert and oriented, and does not appear to be in any distress. A 12-lead ECG shows that the patient is in atrial fibrillation at a controlled rate. A review of Patient BB's medical record shows that she has persistent atrial fibrillation. Past attempts at pharmacologic and electrical cardioversion have failed, and Patient BB remains on low dose antiarrhythmic therapy for rate control and on warfarin for anticoagulation. She has a history of hypertension, coronary artery disease, and percutaneous transluminal angioplasty (PTCA) for placement of two stents. She also has a history of a mitral valve replacement with an artificial valve. Patient BB lives alone in a senior citizen apartment complex; she participates in the social life of the community and takes frequent walks outside in good weather. Her favorite hobby is window-shopping at the mall.
Comments and Rationale: Complaints of dizziness and frequent, unexplained falls are symptoms commonly associated with symptomatic bradycardia. Symptomatic bradycardic episodes are often transient and difficult to capture either on 12-lead ECG or during routine vital sign checks. Due to Patient BB's age, she is at risk to develop symptomatic bradycardia as a result of age-related changes in the cardiac conduction system. She is also more likely to develop conduction abnormalities from antiarrhythmic therapy and medications prescribed to manage hypertension.
The physician orders AECG monitoring for Patient BB. A continuous loop event recorder is selected, and the patient is instructed on its use. She is also instructed to contact the physician when she has experienced one of her symptomatic episodes.
Comments and Rationale: A continuous loop event recorder may be used to document arrhythmias that occur relatively infrequently. This type of AECG is worn continuously; the patient is instructed to depress a switch when he or she feels symptoms. Although Patient BB appears to lose consciousness briefly during her episodes, she is not severely disoriented when she returns to consciousness and would be able to depress the switch in time to signal the device to save the data. Data from the AECG may be transmitted via telephone for analysis.
Approximately 10 days after Patient BB begins wearing the AECG, she experiences another symptomatic episode. Following instructions, she depresses the switch on the recorder. When the episode has ended, she contacts her physician and follows instructions to transmit the data from the AECG via telephone. Analysis of the transmitted data shows that Patient BB is experiencing episodes of severe bradycardia; her ECG tracing shows that she has pauses that last several seconds without an effective heartbeat; during these episodes, her heart rate drops to well less than 60 bpm. See Strip 13.
A diagnosis of symptomatic bradycardia caused by atrial fibrillation with a slow ventricular response is established. Patient BB is referred for pacemaker implantation. Based on a thorough assessment, the physician chooses to implant a VVIR pacemaker.
Comments and Rationale: The definitive diagnosis of symptomatic bradycardia requires that the patient's symptoms of decreased cerebral blood flow and bradyarrhythmia be correlated and documented. For some persons with atrial fibrillation and a slow ventricular response, modifying the prescribed antiarrhythmic therapy may be sufficient to resolve the bradycardic episodes. However, this patient's antiarrhythmic therapy is already low dose and cannot be effectively reduced or altered. Implantation of a permanent pacemaker is indicated. A VVI (or VVIR) is the pacemaker of choice for persons with chronic atrial fibrillation; because a VVI pacemaker does not sense or pace in the atria, it cannot be confused by the chaotic atrial activity that occurs in atrial fibrillation. As Patient BB has an active lifestyle, the rate adaptive feature could be indicated. This will allow the pacemaker to pace her heart at a higher rate when she is engaged in increased activity.
The physician instructs Patient BB to discontinue taking her warfarin two days in advance of her admission to the hospital. When the patient is admitted to the hospital, her INR remains elevated. A continuous heparin drip is initiated and partial thromboplastin times (PTTs) are monitored; the heparin drip is adjusted to maintain the PTT in the desired range. Preoperative lab work, ECG, and chest x-ray are completed. Preoperative education is begun. Two days after admission, Patient BB's INR has dropped to the desired level, and pacemaker implantation is scheduled for the next day. Preoperative preparation is completed; Patient BB is made NPO overnight, and her heparin drip is discontinued. The following morning, Patient BB is taken to the cardiac catheterization lab. Under sedation and local anesthesia, the pacemaker is implanted. The low rate setting is programmed at 60 bpm, and the upper rate limit for rate adaptation is set at 120 bpm.
Comments and Rationale: To reduce the risk of bleeding during and after implantation, warfarin is discontinued prior to the procedure, and the patient's INR is allowed to return to near normal. However, because of Patient BB's chronic atrial fibrillation and artificial heart valve, she is at high risk for thromboembolic events. To reduce this risk, she is admitted to the hospital several days before the implantation is scheduled and placed on a heparin drip. To limit the risk of bleeding during the procedure, the heparin drip is discontinued four to six hours before the procedure. However, as previously stated, a strategy of implanting devices during uninterrupted warfarin therapy appears to have a lower bleeding risk than a strategy of temporarily discontinuing warfarin and bridging with heparin. Because the patient has no special needs, the cardiac catheterization lab is an appropriate site for the pacemaker implantation.
Following Patient BB's return to her room, she is placed on continuous telemetry monitoring. Her vital signs are monitored periodically, and the pacemaker pocket site is inspected for signs of excessive swelling or bleeding. She is instructed to limit use of the arm on the same side as the pacemaker generator, and she receives analgesic medications as prescribed for pain management. A chest x-ray is done, and a 12-lead ECG (with and without magnet) is completed. Patient BB's vital signs remain stable; the pacemaker generator site shows no signs of excessive bleeding or swelling. Telemetry monitoring obtains Strip 14.
Comments and Rationale: The most common complications in the immediate postoperative period are bleeding or swelling in the pacemaker pocket and accidental dislodgment of the pacemaker lead. A chest x-ray is performed to verify lead position; a 12-lead ECG is used to evaluate pacemaker function. Strip 14 shows normal VVIR pacemaker function. The strip shows a heart rate of approximately 70 bpm; this falls within the lower and upper rate limits of Patient BB's pacemaker. Ventricular pacer spikes are present in front of each QRS complex. The QRS complex shows the typical configuration for a paced QRS. Each pacer spike is followed by a paced QRS; there are no random pacemaker spikes or excessive pauses between beats.
Prior to Patient BB's discharge, she receives information on incision line care and precautions to consider. She receives her temporary pacemaker identification card and is instructed to carry the identification card at all times. She is taught how and when to count her pulse and when to notify the physician. The nurse provides the patient with additional written materials, including the patient education handbook supplied by the pacemaker manufacturer. Patient BB expresses concerns about using cellular phones and flying; the nurse provides specific information about the use of cellular phones and how to manage airport security. Patient BB is discharged home; she has a follow-up appointment one week following discharge.
Comments and Rationale: The major focus of patient education just prior to discharge is on prevention of infection and prevention of accidental dislodgment of the pacemaker leads. Specific patient and family concerns should be addressed. Because patients and families frequently think of additional questions following the patient's discharge, the use of written materials provides a resource for them to use at home. Additional patient education will occur during the follow-up visit.
Patient CC, a man 56 years of age who has a DDD pacemaker, calls the TTM center to report that his heart rate is 30 bpm and that his preimplantation symptoms of dizziness, shortness of breath, and light-headedness have reoccurred. Patient CC's pacemaker was implanted approximately two years earlier for management of third-degree heart block. The rate settings on the DDD pacemaker are 60 bpm (low) and 120 bpm (high). Up to this point, the patient has experienced no problems with his pacemaker. Following instructions from the TTM center, Patient CC transmits a recording of his heart rhythm without application of a magnet.
Comments and Rationale: A heart rate that drops to dramatically less than the low rate setting coupled with a return of preimplantation symptoms is indicative of pacemaker malfunction. Malfunctions that may occur during the mid-life of a pacemaker include damage to the pacemaker lead or its insulation. TTM may be used to identify indications of pacemaker malfunction.
The TTM center receives Strip 15. The center immediately notifies Patient CC to report to the emergency department of his local hospital.
Comments and Rationale: Strip 15 shows a pause of approximately three seconds. The pause is followed by an escape beat. As there are no pacemaker spikes present during the pause, the pacemaker malfunction appears to be failure to fire. Patient CC should obtain immediate medical care.
Upon Patient CC's arrival in the emergency department, he is placed on continuous telemetry monitoring. The monitoring initially shows a heart rate of 60 bpm. Telemetry interrogation of the pacemaker shows indications of probable lead fracture. A chest x-ray confirms the presence of a lead failure. The patient is admitted to a telemetry unit for monitoring and revision of his pacemaker. On the acute care unit, Patient CC is again placed on continuous telemetry monitoring. Telemetry monitoring shows a paced rhythm with intermittent missed beats and an isolated two- to three-second pause. An external noninvasive pacemaker is made available. Preoperative lab work is completed, and the patient is kept NPO. Patient CC is taken to the catheterization lab, and the broken lead is replaced.
Comments and Rationale: Telemetry interrogation can retrieve data that reflects the functioning of the pacemaker leads. Fracture of a pacemaker lead may often be seen on a PA and lateral chest x-ray. Fracture of a lead may result in the pacemaker's inability to deliver a pacing impulse to the heart when needed. Due to the length of Patient CC's pauses and the bradycardic rate of his escape rhythm, equipment for externally pacing the heart should be readily available. Replacement of the pacemaker lead is indicated to correct the pacemaker malfunction.
Following revision of the lead, the patient returns to the inpatient unit. His vital signs are monitored periodically, and the generator pocket site observed for bleeding or swelling. He is placed on continuous telemetry monitoring, and his rhythm is monitored for signs of proper pacemaker functioning. His rate settings remain at 60 bpm (low) and 120 bpm (high). Strip 16 is obtained and documented.
Comments and Rationale: Strip 16 shows normal DDD pacemaker functioning. The patient's SA node is firing normally, and the pacemaker is pacing the ventricles at the same rate. The heart rate is approximately 100 bpm, which is somewhat high but within the rate limit setting of the pacemaker. Each pacemaker spike falls at the appropriate place, and each spike is followed by a QRS complex. Each QRS complex shows the typical configuration for a paced QRS complex. There are no random pacer spikes, and there are no pauses that exceed the interval allowed by the timing interval for the low rate limit setting.
As previously discussed, Patient N, a man 66 years of age, presented to his physician's office complaining of increased shortness of breath, rapid weight gain in the last three to four days, and an inability to perform his usual daily activities due to fatigue. Patient N has a history of MIs, coronary artery bypass surgery, acute coronary syndrome, multiple stent placements, and heart failure. Until recently, Patient N's heart failure has been well managed through the use of medications and diet. However, in the last month, the patient has been admitted to the local hospital twice for management of his heart failure symptoms. Based on an assessment of Patient N's status, the physician decides to admit him to the hospital for further management of his heart failure and evaluation for possible biventricular pacemaker implantation.
Comments and Rationale: Heart failure is a progressive, debilitating disease characterized by increasing symptoms, loss of function, and frequent admissions to the hospital for symptom management. Research has shown that neurohormonal mechanisms in the body contribute to this pattern of increased symptoms and worsening disease. Biventricular pacing has been found to be an effective therapy for the management of heart failure in selected patients.
Patient N's admitting ECG reveals that he is in normal sinus rhythm, with a QRS duration of 150 ms. An echocardiogram shows an ejection fraction of 25% as well as abnormal findings consistent with ventricular dyssynchrony. Based on the patient's symptoms, the physician determines that he is in NYHA functional class III heart failure. A review of Patient N's medications shows that he is receiving optimal doses of recommended medications for the management of heart failure. The physician recommends the implantation of a biventricular pacemaker ICD.
Comments and Rationale: Patient N meets the eligibility criteria for implantation of a biventricular pacemaker: an ejection fraction of 35% or less, NYHA Class III to Class IV heart failure, symptomatic despite optimal medical therapy, and a QRS duration of greater than 120 ms. In addition, the patient's ECG demonstrates additional clinical findings that are indicative of ventricular dyssynchrony. Patient N also meets criteria, based on clinical trial data, for the implantation of an ICD: has a history of ischemic heart disease, is at least 40 days post MI, has a left ventricle ejection fraction less than or equal to 30%, NYHA class II or III symptoms despite optimal medical therapy, and can reasonably be expected to survive with a good functional status for more than one year.
Patient N undergoes successful transvenous implantation of a biventricular pacemaker ICD in the cardiac catheterization lab. Upon return to the floor, his vital signs are stable. He is drowsy but responds appropriately to verbal stimuli. The pacemaker pocket shows no signs of bleeding or hematoma formation. Continuous telemetry monitoring is initiated and Strip 17 is obtained.
Comments and Rationale: Strip 17 does not show any evidence of pacing problems; each beat is initiated by a P wave, and each beat is followed by a paced QRS complex. Every QRS is paced, and the rate falls within the rate setting programmed for the pacemaker.
Key points of care following implantation of a biventricular pacemaker ICD include assessment of vital signs, continuous monitoring of ECG rhythm, and assessment of the pacemaker pocket for signs of bleeding or hematoma formation. Although definitive diagnosis of pacing problems requires the expertise of a specially trained physician, continuous telemetry monitoring can identify clues that suggest a problem might exist.
About four hours after Patient N's return from the catheterization lab, he indicates that he has become extremely short of breath. His status is assessed. His blood pressure remains within his usual range. His heart rate is 100 bpm, and he remains 100% ventricularly paced on telemetry. His oxygen saturation has fallen to 88% on 2 liters nasal cannula. He has increased crackles and wheezes bilaterally in his lung bases. The physician is notified and orders a portable chest x-ray and a dose of IV furosemide. Patient N verbalizes distress regarding the recurrence of his symptoms, asking if this means "the pacemaker is not going to help."
Comments and Rationale: Shortness of breath following pacemaker implantation in a patient with severe heart failure may be an indication that a pneumothorax has developed as a complication of the procedure, or it may indicate that the patient is experiencing some volume/fluid overload. A chest x-ray can be used to confirm or rule out the presence of a pneumothorax. IV furosemide is a treatment of choice of an acute episode of volume overload. Volume overload postimplantation in a patient with heart failure may occur from a combination of factors, including physiologic stress of the procedure, anxiety, disruption in normal medication times, and the administration of even small amounts of IV fluid during the procedure. It is appropriate to reassure the patient that the occurrence of his symptoms at this point in his hospitalization is not an indication that CRT will be ineffective.
In response to the IV furosemide, Patient N diureses well and his acute episode of dyspnea resolves. He develops no further acute episodes and is discharged home two days later. In addition to the usual discharge instructions for patients with newly implanted pacemakers, Patient N is instructed to adhere to his prescribed medication therapy, diet restrictions, and lifestyle changes. He is advised to identify the level of his symptoms and activity; it is suggested that he keep a diary noting even small improvements. He is instructed to report increasing signs and symptoms of heart failure to his physician and to make sure to keep his follow-up appointments for the evaluation of his device. Because Patient N also has an ICD, specific ICD discharge instructions are also included.
Comments and Rationale: Persons with biventricular pacemakers should understand that the pacemaker does not replace previously prescribed medical therapies, and that just because their symptoms decrease, they cannot stop taking their medications or adhering to their diet restrictions and recommended lifestyle changes. Improvement in activity tolerance and symptom reduction may occur very gradually. Instructing the patient to note his/her baseline level of symptoms/activity and to record every improvement, no matter how small, can encourage the patient that CRT is working. Recurring or increased signs of failure can be an indication that resynchronization has been lost and should be reported immediately to the physician.
At Patient N's six-month follow-up appointment, he tells the physician that he has noticed little improvement in the amount of activity that he can do. With questioning, the patient admits that he has not been re-hospitalized for management of his heart failure symptoms in the last six months and that his ICD has not fired. The physician interrogates the device and obtains an echocardiogram to evaluate the effectiveness of the programming. The physician also reviews Patient N's current medications, with special emphasis on the dosages of his beta blockers and ACE inhibitors. The physician also questions the patient about his daily activities and what steps he has taken to gradually improve his physical conditioning. Based on this assessment data, the physician decides to increase Patient N's ACE inhibitor dose and to prescribe a structured activity program designed to increase his exercise tolerance.
Comments and Rationale: Some people respond more slowly than others to biventricular pacing. Measures that can improve the patient's response include "fine tuning" the device's programming, decreasing symptoms by increasing neurohormonal blockade through increased medication doses, and employing measures to help the patient slowly, carefully increase his/her activity in small steps.
Due to research and advances in pacemaker technology, pacemaker therapy may be successfully used in the management of persons who have symptomatic bradycardia and in persons with heart failure who meet criteria for ventricular dyssynchrony. For antibradycardia pacing, definitive diagnosis requires the correlation of the bradyarrhythmia with the occurrence of symptoms of decreased cerebral blood flow, such as syncope, presyncope, dizziness, and confusion. Ambulatory ECG monitors provide valuable assistance in the documentation and correlation of symptoms with episodes of bradyarrhythmias. Pacing may also be used to provide CRT for persons with heart failure who have developed a loss of normal coordinated ventricular contraction as a result of the ventricular remodeling that occurs in heart failure. Selection of a specific type of pacemaker is based on an evaluation of patient factors and technical considerations. Follow-up care, including ongoing patient and family education, is vital in the effective management of the patient with a permanent pacemaker.
| American Heart Association |
| 1-800-242-8721 |
| https://www.heart.org |
| Heart Rhythm Society |
| 202-464-3400 |
| https://www.hrsonline.org |
| American College of Cardiology |
| 1-800-253-4636 |
| https://www.acc.org/about-acc |
Asynchronous mode: pacing mode in which the sensing function is turned off. The pacemaker delivers a pacing impulse at a set rate without regard for the heart's intrinsic electrical activity. It is the default setting for magnet mode in some pacemakers.
AV interval: programmable setting on DDD and VDD pacemakers that defines the length of time that should occur between atrial depolarization and delivery of a ventricular pacing impulse. Roughly corresponds to the P-R interval in normal condition.
Capture: ability of the pacemaker to consistently trigger depolarization in the heart chamber paced.
Chronotropic incompetence: inability of the SA node to increase its rate of firing in response to exertion or stress.
Demand pacing: pacing mode that delivers a pacing impulse only when needed (as defined by its programmed parameters).
Inhibit: response of the pacemaker to sensed electrical activity. When the pacemaker senses electrical activity, it inhibits the pacing function and does not deliver a pacing impulse.
Pacing: the ability of the pacemaker to deliver an electrical impulse through the pacemaker lead to the heart to stimulate depolarization.
Sensing: the ability of the pacemaker to recognize electrical activity in the heart.
Sensing threshold: the largest electrical signal (ventricular or atrial) that can be consistently recognized by the sensing circuit.
Stimulation threshold: the amount of electrical energy needed to depolarize the heart.
Triggered: pacemaker response to sensed electrical activity. When the pacemaker recognizes electrical activity, it triggers the pacemaker to deliver a pacing impulse at a preprogrammed interval.
2. Mandawat A, Curtis JP, Mandawat, A, et al. Safety of pacemaker implantation in nonagenarians: an analysis of the Healthcare Cost and Utilization Project, Nationwide Inpatient Sample. Circulation. 2013;127(14):1453-1465.
3. Boyle J, Rost MK. Present status of cardiac pacing: a nursing perspective. Crit Care Nurs Q. 2000;23(1):1-19.
5. Kurtz SM, Ochoa JA, Lau E, et al. Implantation trends and patient profiles for pacemakers and implantable cardioverter defibrillators in the United States: 1993–2006. Pacing Clin Electrophysiol. 2010;33(6):705-711.
6. U.S. Food and Drug Administration. July 2014 PMA Approvals. Available at https://wayback.archive-it.org/7993/20170112093613/http://www.fda.gov/MedicalDevices/ProductsandMedicalProcedures/DeviceApprovalsandClearances/PMAApprovals/ucm414958.htm. Last accessed August 1, 2023.
7. Abraham WT, Hayes DL. Cardiac resynchronization therapy for heart failure. Circulation. 2003;108(21):2596-2603.
8. Gura MT, Foreman L. Cardiac resynchronization therapy for heart failure management. AACN Clin Issues. 2004;15(3):326-339.
9. Hunt SA, Abraham WT, Chin MH, et al. 2009 focused update incorporated into the ACCF/AHA 2005 guidelines for the diagnosis and management of heart failure in adults: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2009;53(15):e1-e390.
10. Woods SL, Froelicher ES, Motzer SA, Bridges EJ. Cardiac Nursing. 7th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2021.
11. National Heart, Lung, and Blood Institute. How the Heart Works. Available at https://www.nhlbi.nih.gov/health/heart. Last accessed August 1, 2023.
12. National Heart, Lung, and Blood Institute. Atrial Fibrillation. Available at https://www.nhlbi.nih.gov/health/atrial-fibrillation. Last accessed August 1, 2023.
15. Beyerbach DM. Pacemakers and Implantable Cardioverter-Defibrillators. Available at https://emedicine.medscape.com/article/162245-overview. Last accessed August 23, 2023.
16. Mallela VS, Ilankumaran V, Rao NS. Trends in cardiac pacemaker batteries. Indian Pacing Electrophysiol J. 2004;4(4):201-212.
17. Medtronic. Living with a Heart Device: Overview. Available at https://www.medtronic.com/us-en/patients/treatments-therapies/living-with-heart-device.html. Last accessed August 1, 2023.
18. Heart Rhythm Society. Atrial Fibrillation (AFib). Available at https://www.upbeat.org/heart-rhythm-disorders/atrial-fibrillation-afib. Last accessed August 1, 2023.
19. Ellenbogen KA, Kaszala K (eds). Cardiac Pacing and ICDs. 7th ed. Hoboken, NJ: John Wiley & Sons; 2020.
20. Hayes DL, Asirvatham SJ, Friedman PA (eds). Cardiac Pacing, Defibrillation and Resynchronization: A Clinical Approach. 3rd ed. Hoboken, NJ: Wiley-Blackwell; 2013.
21. Moses HW, Mullin JC. A Practical Guide to Cardiac Pacing. 6th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2007.
22. Epstein AE, DiMarco JP, Ellenbogen KA, et al. 2012 ACCF/AHA/HRS focused update incorporated into the ACCF/AHA/HRS 2008 guidelines for device-based therapy of cardiac rhythm abnormalities: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2013;61(3):e6-e75.
23. Sarko JA, Tiffany BR. Cardiac pacemakers: evaluation and management of malfunctions. Am J Emerg Med. 2000;18(4):435-440.
24. Bernstein AD, Daubert JC, Fletcher RD, et al. The revised NASPE/BPEG generic code for antibradycardia, adaptive-rate, and multisite pacing. Pacing Clin Electrophysiol. 2002;25(2):260-264.
26. Ouali S, Neffeti E, Ghoul K, et al. DDD versus VVIR pacing in patients, ages 70 and over, with complete heart block. Pacing Clin Electrophysiol. 2010;33(5):583-589.
27. Dharmendrakumar AP, Lavie CJ, Milani RV, Shah S, Gilliland Y. Clinical implications of left atrial enlargement: a review. Ochsner J. 2009;9(4):191-196.
28. Kusumoto FM, Schoenfeld MH, Edgerton JR, et al. 2018 ACC/AHA/HRS guideline on the evaluation and management of patients with bradycardia and cardiac conduction delay: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines, and the Heart Rhythm Society. Heart Rhythm. 2019;16(9):e129-e226.
29. Kusumoto FM, Calkins H, Boehmer J, et al. HRS/ACC/AHA expert consensus statement on the use of implantable cardioverter-defibrillator therapy in patients who are not included or not well represented in clinical trials. Circulation. 2014;130:94-125.
30. Heart Rhythm Society. Sick Sinus Syndrome (SSS). Available at https://www.upbeat.org/heart-rhythm-disorders/sick-sinus-syndrome. Last accessed August 1, 2023.
32. Livingston MW. Sinus Bradycardia. Available at https://emedicine.medscape.com/article/760220-overview. Last accessed August 1, 2023.
33. MedlinePlus. Sick Sinus Syndrome. Available at https://medlineplus.gov/ency/article/000161.htm. Last accessed August 1, 2023.
34. Agrawal A. Third-Degree Atrioventricular Block (Complete Heart Block). Available at https://emedicine.medscape.com/article/162007-overview. Last accessed August 1, 2023.
35. Paul S. Understanding advanced concepts in atrioventricular block. Crit Care Nurse. 2001;21(1):56-66.
36. Sovari AA, Gaeta TJ Kocheril AG, Levine MD. Second-Degree Atrioventricular Block. Available at https://emedicine.medscape.com/article/161919-overview. Last accessed August 1, 2023.
37. Rosenthal L. Atrial Fibrillation. Available at https://emedicine.medscape.com/article/151066-overview. Last accessed August 1, 2023.
38. Wijetunga MN. Carotid Sinus Hypersensitivity. Available at https://emedicine.medscape.com/article/153312-overview. Last accessed August 1, 2023.
39. Wieling W, Krediet CT, Solari D, et al. At the heart of the arterial baroreflex: a physiological basis for a new classification of carotid sinus hypersensitivity. J Intern Med. 2013;273(4):345-358.
40. Vallerand AH, Sanoski CA. Davis's Drug Guide for Nurses. 18th ed. Philadelphia, PA: FA Davis Company; 2022.
41. Karliner LS, Jacobs EA, Chen AH, Mutha S. Do professional interpreters improve clinical care for patients with limited English proficiency? A systematic review of the literature. Health Serv Res. 2007;42(2):727-754.
42. Ngo-Metzger Q, Massagli MP, Clarridge BR, et al. Linguistic and cultural barriers to care: perspectives of Chinese and Vietnamese immigrants. J Gen Intern Med. 2003;18(1):44-52.
43. Merck Manual. Continuous Ambulatory Electrocardiography. Available at https://www.merckmanuals.com/home/heart-and-blood-vessel-disorders/diagnosis-of-heart-and-blood-vessel-disorders/continuous-ambulatory-electrocardiography. Last accessed August 1, 2023.
44. Diagnostic and Interventional Cardiology. An Introduction to Remote Monitoring Systems for Implantable EP Devices. Available at https://www.dicardiology.com/article/introduction-remote-monitoring-systems-implantable-ep-devices. Last accessed August 1, 2023.
45. National Heart, Lung, and Blood Institute. Heart Tests. Available at https://www.nhlbi.nih.gov/health/heart-tests. Last accessed August 1, 2023.
46. Crawford MH, Bernstein SJ, Deedwania PC, et al. ACC/AHA guidelines for ambulatory electrocardiography: executive summary and recommendations: a report of the American College of Cardiology/American Heart Association task force on practice guidelines. Circulation. 1999;100(8):886-893.
47. MCOT Patch. Available at https://www.myheartmonitor.com/wp-content/uploads/sites/2/2020/02/MCOT-Patch.pdf. Last accessed August 1, 2023.
48. Medtronic. Remote Monitoring for Implanted Heart Devices. Available at https://www.medtronic.com/us-en/patients/treatments-therapies/remote-monitoring.html. Last accessed August 1, 2023.
49. Cardiac Monitoring.com. Mobile Cardiac Telemetry. Available at http://cardiacmonitoring.com/mobile-cardiac-telemetry. Last accessed August 1, 2023.
50. Kusumoto FM, Schoenfeld MH, Barrett C, et al. 2018 ACC/AHA/HRS guideline on the evaluation and management of patients with bradycardia and cardiac conduction delay. J Am Coll Cardiol. 2019;74(7):e51-e156.
51. National Heart, Lung, and Blood Institute. What are Pacemakers? Available at https://www.nhlbi.nih.gov/health/pacemakers. Last accessed August 1, 2023.
52. Ghanbari H, Phard WS, Al-Ameri H, et al. Meta-analysis of safety and efficacy of uninterrupted warfarin compared to heparin-based bridging therapy during implantation of cardiac rhythm devices. Am J Cardiol. 2012;110(10):1879-1913.
53. Boston Scientific. Pacemaker Device Support: Medical and Dental Procedures. Available at https://www.bostonscientific.com/en-US/patients-caregivers/device-support/pacemaker/medical-dental.html. Last accessed August 1, 2023.
54. Abbott. Electromagnetic Interference. Electromagnetic Interference: Guide to Electromagnetic Compatibility and Your Cardiac Device. Available at https://www.cardiovascular.abbott/us/en/patients/cardiovascular-device-patient-services/emi.html. Last accessed August 1, 2023.
55. Medtronic. Medtronic First to Receive FDA Approval for Pacing the Heart's Natural Conduction System. Available at https://news.medtronic.com/2022-10-17-Medtronic-First-to-Receive-FDA-Approval-for-Pacing-the-Hearts-Natural-Conduction-System. Last accessed August 1, 2023.
56. Lewis AJM, Foley P, Whinnett Z, et al. His bundle pacing: a new strategy for physiological ventricular activation. JAHA. 2019;8(6):e010972.
57. Abbott. Abbott Receives FDA Approval for World's First Dual Chamber Leadless Pacemaker. Available at https://abbott.mediaroom.com/2023-07-05-Abbott-Receives-FDA-Approval-for-Worlds-First-Dual-Chamber-Leadless-Pacemaker. Last accessed August 1, 2023.
58. Manley D. Logan Woman Becomes First Utah Patient to Get Tiny New Pacemaker. Available at https://archive.sltrib.com/article.php?id=58276080&itype=CMSID. Last accessed August 1, 2023.
59. Reardon S. Gene Therapy Creates Biological Pacemaker. Available at https://www.nature.com/news/gene-therapy-creates-biological-pacemaker-1.15569. Last accessed August 1, 2023.
60. Wilkoff BL, Auricchio A, Brugada J, et al. HRS/EHRA expert consensus on the monitoring of cardiovascular implantable electronic devices (CIEDs): description of techniques, indications, personnel, frequency, and ethical considerations. Heart Rhythm. 2008;5(6):907-925.
61. Zeitler EP, Piccini JP. Remote monitoring of cardiac implantable electronic devices (CIED). Trends Cardiovas Med. 2016;26(6): 568-577.
62. Centers for Medicare and Medicaid Services. Medicare Claims Processing Manual. Chapter 35—Independent Diagnostic Testing Facility (IDTF). Available at https://www.cms.gov/Regulations-and-Guidance/Guidance/Manuals/downloads/clm104c35.pdf. Last accessed August 1, 2023.
63. Ricci RP, Morichelli L, D'Onofrio A, et al. Effectiveness of remote monitoring of CIEDs in detection and treatment of clinical and device-related cardiovascular events in daily practice: the HomeGuide Registry. Europace. 2013;15(7):970-977.
64. Ghosh P. Pacemaker Malfunction. Available at https://emedicine.medscape.com/article/156583-overview. Last accessed August 1, 2023.
65. Beyerbach DM, Cadman C. Pacemaker Syndrome. Available at https://emedicine.medscape.com/article/159706-overview. Last accessed August 1, 2023.
66. U.S. Food and Drug Administration. FDA Approves First Leadless Pacemaker to Treat Heart Rhythm Disorders. Available at https://www.fda.gov/news-events/press-announcements/fda-approves-first-leadless-pacemaker-treat-heart-rhythm-disorders. Last accessed August 1, 2023.
67. The Joint Commission. Specifications Manual for Joint Commission National Quality Measures (v2018A): New York Heart Association (NYHA) Classification. Available at https://manual.jointcommission.org/releases/TJC2018A/DataElem0439.html. Last accessed August 1, 2023.
68. Centers for Disease Control and Prevention. Heart Failure. Available at https://www.cdc.gov/heartdisease/heart_failure.htm. Last accessed August 1, 2023.
70. Trupp RJ. Cardiac resynchronization therapy: optimizing the device, optimizing the patient. J Cardiovasc Nurs. 2004;19(4):223-233.
71. Flanagan J, Horwood L, Bolin C, Sample R. Heart failure patients with ventricular dysynchrony: management with a cardiac resynchronization therapy device. Prog Cardiovasc Nurs. 2003;18(4):184-189.
72. Tang WH Wilson, Francis GS. Cardiac resynchronization therapy in New York Heart Association Class IV heart failure: it is all about selection. Circulation. 2007;115:161-162.
73. Albouaini K, Egred M, Rao A, Alahmar A, Wright DJ. Cardiac resynchronization therapy: evidence-based benefits and patient selection. Eur J Intern Med.2008;19(3):165-172.
74. Agrawal, A, Guzman DB. Cardiac Resynchronization Therapy. Available at https://emedicine.medscape.com/article/1839506-overview. Last accessed August 1, 2023.
75. Narayan SM. Implantable defibrillators with and without resynchronization for patients with left ventricular dysfunction. Tex Heart Inst J. 2005;32(3):358-361.
76. Yarnoz MJ, Curtis AB. Sex-based differences in cardiac resynchronization therapy and implantable cardioverter defibrillator therapies: effectiveness and use. Cardiol Rev. 2006;14(6):292-298.
77. Linde C, Ellenbogen K, McAlister FA. Cardiac resynchronization therapy (CRT): clinical trials, guidelines, and target populations. Heart Rhythm. 2012;9(8):S3-S13.
78. Schuster P, Faerestrand S. Techniques for identification of left ventricular asynchrony for cardiac resynchronization therapy in heart failure. Indian Pacing Electrophysiol J. 2005;5(3):175-185.
79. Yu CM, Chan YS, Zhang Q, et al. Benefits of cardiac resynchronization therapy for heart failure patients with narrow QRS complexes and coexisting systolic asynchrony by echocardiography. J Am Coll Cardiol. 2006;48(11):2251-2257.
80. Quigley RL. A hybrid approach to cardiac resynchronization therapy. Ann Thorac Cardiovasc Surg. 2011;17(3):273-276.
81. Cleveland Clinic. Biventricular Pacemaker. Available at https://my.clevelandclinic.org/health/treatments/16784-biventricular-pacemaker. Last accessed August 1, 2023.
82. Kydd AC, Khan FZ, Watson WD, Pugh PJ, Virdee MS, Dutka DP. Prognostic benefit of optimum left ventricular lead position in cardiac resynchronization therapy: follow-up of the TARGET study cohort. JACC Heart Fail. 2014;2(3):205-212.
83. McAtee ME, Gawlinski A. Biventricular pacing: new treatment for patients with heart failure bears important nursing implications. Am J Nurs. 2002;102(5):4-7.
84. Saba S, Marek J, Schwartzman D, et al. Echocardiography-guided left ventricular lead placement for cardiac resynchronization therapy: results of the Speckle Tracking Assisted Resynchronization Therapy for Electrode Region trial. Circ Heart Fail. 2013;6(3):427-434.
85. Khan FZ, Virdee MS, Palmer CR, et al. Targeted left ventricular lead placement to guide cardiac resynchronization therapy: the TARGET study: a randomized, controlled trial. J Am Coll Cardiol. 2012;59(17):1509-1518.
86. Heidenreich PA, Bozkurt B, Aguilar D, et al. 2022 AHA/ACC/HFSA guideline for the management of heart failure: a report of the American College of Cardiology /American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2022;145(18):e895-e1032.
87. Bleeker GB, Schalij MJ, Holman ER, Steendijk P, van der Wall EE, Bax JJ. Cardiac resynchronization therapy in patients with systolic left ventricular dysfunction and symptoms of mild heart failure secondary to ischemic or nonischemic cardiomyopathy. Am J Cardiol. 2006;98(2):230-235.
88. Bristow MR, Saxon LA, Boehmer J, et al. Cardiac-resynchronization therapy with or without an implantable defibrillator in advanced chronic heart failure. N Engl J Med. 2004;350(21):2140-2150.
89. Young JB, Abraham WT, Smith AL, et al. Combined cardiac resynchronization and implantable cardioversion defibrillation in advanced chronic heart failure: the MIRACLE ICD Trial. JAMA. 2003;289(20):2685-2694.
91. Tracy CM, Epstein AE, Darbar D, et al. 2012 ACCF/AHA/HRS focused update of the 2008 guidelines for device-based therapy of cardiac rhythm abnormalities: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Circulation. 2012;126:1784-1800.
92. Glikson M, Nielsen JC, Kronborg MB, et al. 2021 ESC guidelines on cardiac pacing and cardiac resynchronization therapy: developed by the Task Force on cardiac pacing and cardiac resynchronization therapy of the European Society of Cardiology (ESC) with the special contribution of the European Heart Rhythm Association (EHRA). Eur Heart J. 2021;42:3427-3520 .
1. Kusumoto FM, Schoenfeld MH, Barrett C, et al. 2018 ACC/AHA/HRS guideline on the evaluation and management of patients with bradycardia and cardiac conduction delay: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines, and the Heart Rhythm Society. J Am Coll Cardiol. 2019;74(7):932-987. Available at https://www.hrsonline.org/clinical-resources/2018-accahahrs-guideline-evaluation-and-management-patients-bradycardia-and-cardiac-conduction. Last accessed August 30, 2023.
2. National Clinical Guideline Centre. Transient Loss of Consciousness ("Blackouts") in Over 16s. London: National Institute for Health and Clinical Excellence; 2014. Available at https://www.nice.org.uk/guidance/cg109. Last accessed August 30, 2023.
3. American Society of Anesthesiologists Task Force on Anesthetic Care for Magnetic Resonance Imaging. Practice advisory on anesthetic care for magnetic resonance imaging: an updated report by the American Society of Anesthesiologists Task Force on Anesthetic Care for Magnetic Resonance Imaging. Anesthesiology. 2015;122(3):495-520. Available at https://anesthesiology.pubs.asahq.org/article.aspx?articleid=2091587. Last accessed August 30, 2023.
4. Glikson M, Cosedis Nielsen J, Kronborg MB, et al. 2021 ESC Guidelines on cardiac pacing and cardiac resynchronization therapy: Developed by the Task Force on cardiac pacing and cardiac resynchronization therapy of the European Society of Cardiology (ESC) With the special contribution of the European Heart Rhythm Association (EHRA). Eur Heart J. 2021;42(35):3427-3520. Available at https://academic.oup.com/eurheartj/article/42/35/3427/6358547. Last accessed August 30, 2023.
Mention of commercial products does not indicate endorsement.