Pneumonia is a substantial healthcare concern, ranking among the most common reasons for emergency department and outpatient visits, hospitalizations, and deaths among both adults and children. Decreasing the incidence of pneumonia and its associated morbidity and mortality requires a multifaceted approach and a strategy that includes: a concerted effort to improve rates of pneumococcal and influenza vaccinations, especially among high-risk populations; better adherence to guideline-recommended treatment; systems-level approaches to improve the appropriate use of antibiotics; and performance improvement initiatives to reduce healthcare-associated infections.
- HISTORICAL BACKGROUND
- DEFINITIONS
- EPIDEMIOLOGY AND SCOPE
- GUIDELINE-DIRECTED MANAGEMENT AND PREVENTION OF PNEUMONIA
- PATHOGENESIS AND CLASSIFICATION OF PNEUMONIA
- COMMUNITY-ACQUIRED PNEUMONIA
- PREVENTION OF PNEUMONIA
- PNEUMONIA ASSOCIATED WITH HEALTHCARE FACILITIES
- SUMMARY
- Works Cited
- Evidence-Based Practice Recommendations Citations
This course is designed for all physicians, physician assistants, and nurses, especially those working in the emergency department, outpatient settings, pediatrics, nursing homes, and intensive care units.
The purpose of this course is to provide physicians, nurses, and other healthcare professionals who manage the care of patients with pneumonia a foundation for effective management strategies in order to improve outcomes and foster an interprofessional collaborative practice consistent with published guidelines.
Upon completion of this course, you should be able to:
- Discuss the epidemiology, scope, and classification of pneumonias.
- Predict the likely etiology (pathogens) in a given case of pneumonia, based on epidemiologic features, clinical setting, and risk factor assessment.
- Assess the diagnostic probability of pneumonia in a given patient, using careful history and clinical examination findings.
- Determine, by clinical criteria and severity of illness score, which patients with pneumonia require hospitalization or admission to an intensive care unit.
- Develop a management plan for community-acquired pneumonia, including selection of initial antibiotic therapy appropriate to clinical context and site of care, in accordance with established guidelines.
- Outline the diagnosis and management of community-acquired pneumonia in pediatric patients.
- Devise a strategy for prevention of community-acquired pneumonia, including risk factor reduction and recommended immunization protocols.
- Identify the epidemiology and risk factors of hospital-acquired, ventilator-associated, and nursing home-acquired pneumonia.
- Anticipate the likely pathogens and antibiotic-sensitivity patterns associated with pneumonia that arises in healthcare facilities.
- Initiate the management of patients with hospital-acquired or ventilator-associated pneumonia, including guideline-adherent selection of empiric antibiotic therapy.
- Develop a strategy to reduce the risk of pneumonia for patients in healthcare facilities.
Carol Whelan, APRN, has been working in nursing education since 2000. She received her Master's degree in psychiatric/mental health nursing from St. Joseph College in West Hartford, Connecticut, and completed post-graduate nurse practitioner training at Yale University. Ms. Whelan is an Associate Clinical Professor and Lecturer at Yale University and works as an APRN at the Department of Veterans' Affairs in Connecticut, where she also serves as the Vice President of Medical Staff. She has authored many articles, textbook chapters, and books.
John M. Leonard, MD, Professor of Medicine Emeritus, Vanderbilt University School of Medicine, completed his post-graduate clinical training at the Yale and Vanderbilt University Medical Centers before joining the Vanderbilt faculty in 1974. He is a clinician-educator and for many years served as director of residency training and student educational programs for the Vanderbilt University Department of Medicine. Over a career span of 40 years, Dr. Leonard conducted an active practice of general internal medicine and an inpatient consulting practice of infectious diseases.
Contributing faculty, Carol Whelan, APRN, has disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
Contributing faculty, John M. Leonard, MD, has disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
John V. Jurica, MD, MPH
Mary Franks, MSN, APRN, FNP-C
Randall L. Allen, PharmD
The division planners have disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
Sarah Campbell
The Director of Development and Academic Affairs has disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
The purpose of NetCE is to provide challenging curricula to assist healthcare professionals to raise their levels of expertise while fulfilling their continuing education requirements, thereby improving the quality of healthcare.
Our contributing faculty members have taken care to ensure that the information and recommendations are accurate and compatible with the standards generally accepted at the time of publication. The publisher disclaims any liability, loss or damage incurred as a consequence, directly or indirectly, of the use and application of any of the contents. Participants are cautioned about the potential risk of using limited knowledge when integrating new techniques into practice.
It is the policy of NetCE not to accept commercial support. Furthermore, commercial interests are prohibited from distributing or providing access to this activity to learners.
Supported browsers for Windows include Microsoft Internet Explorer 9.0 and up, Mozilla Firefox 3.0 and up, Opera 9.0 and up, and Google Chrome. Supported browsers for Macintosh include Safari, Mozilla Firefox 3.0 and up, Opera 9.0 and up, and Google Chrome. Other operating systems and browsers that include complete implementations of ECMAScript edition 3 and CSS 2.0 may work, but are not supported. Supported browsers must utilize the TLS encryption protocol v1.1 or v1.2 in order to connect to pages that require a secured HTTPS connection. TLS v1.0 is not supported.
The role of implicit biases on healthcare outcomes has become a concern, as there is some evidence that implicit biases contribute to health disparities, professionals' attitudes toward and interactions with patients, quality of care, diagnoses, and treatment decisions. This may produce differences in help-seeking, diagnoses, and ultimately treatments and interventions. Implicit biases may also unwittingly produce professional behaviors, attitudes, and interactions that reduce patients' trust and comfort with their provider, leading to earlier termination of visits and/or reduced adherence and follow-up. Disadvantaged groups are marginalized in the healthcare system and vulnerable on multiple levels; health professionals' implicit biases can further exacerbate these existing disadvantages.
Interventions or strategies designed to reduce implicit bias may be categorized as change-based or control-based. Change-based interventions focus on reducing or changing cognitive associations underlying implicit biases. These interventions might include challenging stereotypes. Conversely, control-based interventions involve reducing the effects of the implicit bias on the individual's behaviors. These strategies include increasing awareness of biased thoughts and responses. The two types of interventions are not mutually exclusive and may be used synergistically.
#94674: Pneumonia
Hippocrates first described the clinical picture of pneumonia in 400 B.C.E., including the presence of fever, chest pain, productive cough, rales, and dyspnea[1]. However, the disease was recognized even before Hippocrates' time. The disease has resulted in a serious public health and mortality burden over the years, with Osler referring to pneumonia as the "captain of the men of death" in the early 1900s. During this same period, pneumonia surpassed tuberculosis as a leading cause of death.
In the past century, dramatic advances in health care have greatly improved outcomes for patients with pneumonia. These advances include the introduction of effective antibiotics and immunization practices, improved clinical care, and safer surgical technique. Although morbidity and mortality from pneumonia has declined in developed countries, pneumonia remains a major health concern, and the emergence of multidrug-resistant organisms has led to renewed interest and research on this ancient disease.
Pneumonia is defined as a lower respiratory tract, parenchymal infection of the lung. The usual clinical presentation is that of acute- or subacute-onset fever, productive cough, pleuritic chest pain, localized rales and signs of consolidation, accompanied by pulmonary opacification(s) on chest radiograph. For clinical purposes, pneumonia in a nonhospitalized patient is designated as either community-acquired (CAP) or healthcare-associated (HCAP) depending on whether there has been significant exposure to a healthcare environment (e.g., hospital, nursing home, dialysis clinic) within the previous 90 days. Pneumonias that develop as a complication of hospitalization are termed "nosocomial" and are further divided into hospital-acquired pneumonia (HAP) or ventilator-associated pneumonia (VAP). These are important distinctions, as HCAP and nosocomial pneumonias carry a greater risk for less common, multidrug-resistant bacterial infection.
The term "pneumonia" is sometimes used in reference to other inflammatory conditions of the lung when a component of infection is known or suspected. An example is "aspiration pneumonia," whereby a focal chemical pneumonitis (lung injury) is followed rapidly by bacterial overgrowth and incipient infection (pneumonia).
Pneumonia is a substantial healthcare concern, ranking among the most common reasons for emergency department and outpatient visits, hospitalizations, and deaths among both adults and children [2,3,4,5,6]. The World Health Organization (WHO) lists pneumonia as the world's fourth leading cause of death, accounting for an estimated 2.5 million lives lost to lower respiratory infection in 2021 [228]. Collected data consistently demonstrate a bimodal distribution of mortality, with peaks in children younger than 5 years of age and adults older than 75 years of age. Worldwide, pneumonia was responsible for an estimated 808,000 deaths in children younger than 5 years of age in 2017, mainly in developing countries [228]. In the United States, pneumonia is the leading cause of death from infectious disease and the eighth most common cause of death overall. According to data from the National Center for Health Statistics, there were 41,108 deaths from bacterial and viral pneumonias in 2022, a rate of 12.3 per 100,000 population [234]. There is seasonal variation in the incidence of pneumonia, with most cases occurring in the winter months.
U.S. hospital discharge statistics show that the rate of hospitalization for pneumonia varies with age, being highest among adults 75 to 84 years of age. In recent decades, the rate of hospitalization for pneumonia has been relatively stable for adults younger than 65 years of age and has declined somewhat for adults older than 65 years (Table 1) [6]. In 2021, there were 1.4 million U.S. hospital visits with pneumonia due to infectious organism as the primary diagnosis [2].
The mortality rate for pneumonia and influenza combined has decreased substantially in the United States over the past 20 years, falling from 23.7 per 100,000 in 2000 to 12.3 per 100,000 in 2022 [234]. Two important public health factors, which may account for this trend, are the increased utilization of pneumococcal and influenza vaccines among adults and children and the decline in cigarette smoking [220,221].
Despite advances made in prevention, treatment, and clinical outcomes, the impact on healthcare delivery systems and the aggregate cost of caring for patients with pneumonia are expected to increase in years to come. This is because of an aging U.S. population, the very group in whom the rate of pneumonia is highest. Using a decision analytic model that assumes no targeted intervention, a population medicine study group projected the incidence of pneumococcal pneumonia in the United States will increase by 38% between 2014 and 2040, with hospitalizations for pneumococcal pneumonia increasing by 96% (from 401,000 to 790,000) in that same period. As a result, healthcare costs associated with pneumonia are expected to increase by $2.5 billion and demand for healthcare services for pneumonia is expected to double [14].
In the past two decades, clinical guidelines for the management of pneumonia have been developed by infectious disease and pulmonary medicine societies to improve outcomes and decrease the cost of care. Unfortunately, adherence to guideline-directed management protocols has been low, despite studies demonstrating that lack of adherence is associated with higher rates of adverse outcomes and inappropriate use of antimicrobials [15,16,17,18,20,21]. Attention to guidelines varies across hospitals, clinical settings, and specialty practices. Adherence rates tend to be lower among non-pulmonologists and in relation to patient variables such as presence or absence of comorbidities and recent use of antibiotics [20,22,23]. Several barriers to guideline adherence have been identified, including lack of familiarity, concern over the practicality and perceived cost of recommended antibiotics, limited documentation of improved outcomes, and potential conflict with other guidelines [23]. The time spent on continuing education activities appears to have a direct correlation with a positive attitude toward, and propensity to follow, published clinical guidelines.
Success in reducing the incidence of pneumonia relies on effective strategies to prevent disease. The primary strategy for prevention of CAP is immunization with influenza and pneumococcal vaccines, targeting high-risk groups (i.e., young children, older individuals, and people with compromised immune systems). Targeted immunization has been shown to decrease the rate of hospitalization for pneumonia and influenza and to decrease the risk of long-term morbidity and mortality [7,9,10,218]. However, vaccine utilization rates are low, especially the rate of pneumococcal vaccination among high-risk groups and influenza vaccination among children [6,11]. Only about one-quarter of adults have ever had a pneumococcal vaccine.
Prevention of HCAP focuses on care measures to preserve healthy pulmonary defense mechanisms and reduce transmission of healthcare-associated, often multidrug-resistant, bacterial pathogens. Adherence to guidelines for the prevention of pneumonia that arises in the hospital setting has been low, with approximately 39% to 66% of hospitals reporting full compliance and up to one-half of nurses reporting that they do not routinely adhere to recommended prevention practices [12,13].
Decreasing the incidence of pneumonia and its associated morbidity and mortality requires a multifaceted approach and a strategy that includes a concerted effort to improve rates of pneumococcal and influenza vaccinations, especially among high-risk populations; better adherence to guideline-recommended treatment; systems-level approaches to improve the appropriate use of antibiotics; and performance improvement initiatives to reduce healthcare-associated infections. This course is designed to assist healthcare professionals provide better care to their patients by highlighting guideline-recommended diagnosis, treatment, and prevention of pneumonia.
Pneumonia is an acute inflammatory condition within the parenchyma of the lung caused by infection that reaches the lower respiratory tract. In most cases, pneumonia develops as a consequence of bacterial colonization/infection of the upper respiratory tract, followed by microaspiration of infected secretions at a time of impaired host pulmonary defense mechanisms [217]. The prime host defenses against foreign particulate matter that reaches the lower respiratory tract are the cough reflex, tracheobronchial (mucociliary) clearance, and alveolar macrophage phagocytosis. Activation of the humeral (antibody) immune response provides augmentation of phagocytosis and the acute cellular response. One or more of these defense mechanisms may be impaired by a variety of factors, including underlying cardiopulmonary and neurologic disease, sedative medication, bronchial obstruction, concurrent active viral and mycoplasma bronchitis, and toxic/metabolic conditions such as alcohol excess, acidosis, and hypoxia. Individuals with an impaired immune system, such as occurs from immunosuppressive drugs, human immunodeficiency virus (HIV), chronic disease, or old age, are more susceptible to infection [4].
Clinically, pneumonia is often described in reference to suspected or established causative pathogens (i.e., viral, bacterial, fungal, or parasitic); however, the precise etiology cannot be identified in more than half the cases in which testing is done [9,24,25]. Classifying pneumonia according to setting in which it develops is more useful for clinical purposes because the most common pathogens, as well as clinical outcomes, are similar within distinct clinical settings [26,27]. Pneumonia was once broadly classified as either community-acquired (developing outside of a hospital or other healthcare facility) or nosocomial (developing 48 hours or more after hospital admission, usually postoperatively). In its 2016 guideline, the American Thoracic Society (ATS) and the Infectious Diseases Society of America (IDSA) noted three distinct categories within the broader classification of pneumonia associated with healthcare facilities: HAP, VAP, and HCAP (Table 2) [3,28]. These three categories of pneumonia are similar in that they often result from colonization, then infection, by resistant gram-negative bacilli and methicillin-resistant Staphylococcus aureus (MRSA), necessitating broader empiric antibiotic therapy than that commonly used for CAP [27].
TYPES OF PNEUMONIA
| Type | Definition | ||||
|---|---|---|---|---|---|
| Community-acquired | New infection in a patient residing in the community, with no recent exposure to a healthcare setting or antibiotics | ||||
| Hospital-acquired | New infection occurring more than 48 hours after hospital admission | ||||
| Ventilator-associated | New infection occurring more than 48 to 72 hours after endotracheal intubation | ||||
| Healthcare-associated |
|
As noted, the cause of pneumonia varies according to setting and patient age. Viruses are the most common cause in young children, whereas bacteria are the more frequent cause among older children and adults [29,30,31]. Studies have shown that respiratory viral pathogens play a greater role in the pathogenesis of pneumonia than once thought; many cases of pneumonia, both pediatric and adult, involve a combination of bacterial and viral pathogens or two or more viral pathogens [9,24,30,32]. The increase in the number of viral infections is thought to be related, in part, to better diagnostic testing methods, most notably, polymerase chain reaction (PCR)-based techniques [24,33,34].
Pyogenic bacterial infection is the cause of nearly all cases of HAP and VAP, and the distribution of pathogens varies among institutions [26,28,29]. Mixed infection appears to be common, as more than one pathogen is frequently isolated from sputum cultures in these cases [28]. Bacteria isolated from cases of early-onset HAP (within four days after admission) are usually sensitive to available drugs [28]. In contrast, late-onset HAP (i.e., more than five days after admission) is likely to be caused by multidrug-resistant pathogens, such as Pseudomonas spp., MRSA, and Acinetobacter spp. [26,35]. Viral and fungal pathogens rarely cause HAP or VAP [28].
Determining accurate incidence rates for CAP is challenging because "pneumonia" is not a reportable disease; moreover, case definition varies across studies and national databases often link pneumonia with influenza. Epidemiology of pneumonia relies primarily on data derived from community-based cohort studies and surveillance networks. Approximately 5 to 6 million cases of pneumonia are diagnosed annually, with about 1 million occurring in older adults [36]. Approximately 4.2 million adult outpatient visits are related to CAP every year, and the mortality rate is less than 1% for adults treated on an outpatient basis [37].
The burden of disease is considerably greater for patients hospitalized with pneumonia. A prospective cohort study of adult residents living in Louisville, Kentucky (population 587,000 adults), recorded 7,449 unique patients hospitalized with CAP between June 2014 and June 2016 [232]. The annual age-adjusted incidence was 649 patients hospitalized with CAP per 100,000 adults, which extrapolates to nearly 1.6 million annual adult CAP hospitalizations in the United States. The observed mortality during hospitalization was 6.5%. An earlier report placed the average overall mortality rate for hospitalized adults at 12%, but the rate is higher—about 30% to 40%—for adults who require admission to an intensive care unit (ICU) [37]. The estimated direct and indirect financial costs are $3.7 billion and $1.8 billion, respectively [38].
The burden of pneumonia is greatest among the elderly (65 years of age and older). In one study of 46,237 people 65 years of age and older, the overall rate of CAP was 18.2 cases per 1,000 person-years for people 65 to 69 years of age, increasing to 52.3 cases per 1,000 person-years for those 85 years of age or older [39].
The mortality rate for adults with pneumonia has decreased substantially over the past two decades. In a review of more than 2.6 million Medicare claims for pneumonia between 1987 and 2005, the age- and sex-adjusted mortality rate dropped from 13.5% to 9.7% [40].
The rate of pediatric outpatient visits for CAP has been reported to be 35 to 52 per 1,000 children 3 to 6 years of age and 74 to 92 per 1,000 children 2 years of age and younger [10]. The hospitalization rate for children up to 18 years of age is 201.1 per 100,000; the highest rate is for infants younger than 1 year of age (912.9 per 100,000) and lowest for teenagers (62.8 per 100,000) [4]. According to data from the Centers for Disease Control and Prevention (CDC), 343 infants (younger than 1 year of age) and 502 children (1 to 17 years of age) in the United States died as a result of pneumonia (or another lower respiratory tract infection) in 2021 [234].
The primary risk factors for CAP are age, smoking history, and chronic lung disease (e.g., chronic obstructive pulmonary disease [COPD]) and other comorbidities. Occupational dust exposure and history of childhood pneumonia have also been associated with an increased risk, as has male gender, unemployment, and single marital status [39,41]. As noted earlier, the risk for pneumonia is higher for individuals 65 years or older compared with younger adults, with the risk further increasing for those 85 years and older [39]. Alcoholism and chronic diseases, such as respiratory disease, cardiovascular disease, or kidney disease, also increase the risk for pneumonia, especially in the older population [3,42,43]. Conditions of frailty, dementia, alcohol use, and sedative medication all lead to diminished or ineffectual cough and the propensity for aspiration, thereby increasing the risk for pneumonia. Diseases or medications that suppress the immune system increase the risk among all ages [39,42]. In the pediatric population, very young children are at increased risk because their immune systems have not fully developed. Secondhand smoke exposure, particularly with two or more smokers in the home, is a significant risk factor for pneumonia in children [241].
Although molecular diagnostic techniques have identified a diverse pulmonary alveolar microbiota coexisting within the healthy lung, the pulmonary airways may be considered "sterile" in regard to pathogens associated with incident pneumonic infection. Respiratory tract microbiota may work in concert with pulmonary defense mechanisms, including mucociliary clearance and alveolar macrophage phagocytosis, to keep the lower respiratory tract free from bacterial colonization/infection [238]. Cigarette smoking and other chronic respiratory conditions eventually cause bronchial inflammation and disrupt host defense mechanisms to such an extent that "colonization" of the airways by microbial pathogens is established early in the course of many persons with COPD [44]. The pathogens most commonly implicated are adenovirus, Chlamydophila pneumoniae, Haemophilus influenzae, Moraxella catarrhalis, and Streptococcus pneumoniae. Bacterial colonization in this setting represents low-grade chronic infection, which, in combination with clinical exacerbations, augments airway inflammation, and contributes to pathogenesis and disease progression.
The use of proton pump inhibitors (PPIs) may increase the risk of developing pneumonia, but data are somewhat unclear. One study found that only treatment with PPIs within the past 30 days (and not long-term use) was associated with increased risk, but a later meta-analysis showed that the risk was increased among people taking PPIs or histamine2 receptor antagonists [44,45].
Among nursing home patients, older age and male sex are risk factors for pneumonia. Other risk factors for this population include swallowing difficulty, inability to take oral medications, profound disability, bedridden state, and urinary incontinence [42].
Given the right conditions, a great many micro-organisms are capable of infecting the lung. In general, however, the number of viruses and bacteria implicated in most cases of CAP in adults and children is relatively small. For a given case, the clinical setting and the patient's age, comorbidity, and risk factors are useful predictors of causation. Viral pneumonia (e.g., influenza) is most commonly linked to community outbreaks.
The most common cause of CAP is S. pneumoniae, identified in approximately one-third of all cases and 40% to 50% of all culture-confirmed bacterial pneumonia cases that require hospitalization [9,29,30,46]. The most common causative pathogen varies in relation to the patient's age, illness severity, and clinical context (Table 3) [29,30,47].
MOST LIKELY ETIOLOGIES OF COMMUNITY-ACQUIRED PNEUMONIA ACCORDING TO PATIENT AGE AND SETTING
| Age and/or Setting | Most Likely Pathogens | ||||||
|---|---|---|---|---|---|---|---|
| Adults | |||||||
| Outpatient |
| ||||||
| Inpatient, not intensive care unit |
| ||||||
| Intensive care unit |
| ||||||
| Children | |||||||
| Birth to 3 weeks |
| ||||||
| 3 weeks to 3 months |
| ||||||
| 4 months to 4 years |
| ||||||
| 5 to 15 years |
| ||||||
Clues to the etiology of the pneumonia can often be found in the patient's past medical history (Table 4). Persons with chronic bronchitis/COPD frequently have tracheobronchial colonization with S. pneumoniae, H. influenzae, or M. catarrhalis, and when pneumonia supervenes, it is usually with one of these pathogens. Heavy alcohol use carries the risk for anaerobic pleuropulmonary infection (e.g., lung abscess, empyema) and pneumococcal or gram-negative bacillary (e.g., Klebsiella pneumoniae, Proteus spp.) pneumonia.
COMORBIDITY AND EXPOSURE IN RELATION TO AT-RISK PATHOGENS
| Patient Characteristic | Suspect Pathogen(s) | ||||
|---|---|---|---|---|---|
| Alcoholism |
| ||||
| COPD, tobacco use |
| ||||
| Nursing home resident |
| ||||
| Poor dental hygiene | Oral anaerobes | ||||
| Recent exposure to contaminated plumbing or water | Legionella organisms | ||||
| Exposure to exotic birds and/or decaying bird nesting sites |
| ||||
| HIV infection |
| ||||
| Exposure to excreta of wild rodents | Sin nombre virus (hantavirus pulmonary syndrome) | ||||
| COPD = chronic obstructive pulmonary disease. | |||||
Other epidemiologic clues to the etiology of pneumonias include seasonal and geographic considerations. Influenza outbreaks are associated with a seasonal increase in secondary S. pneumoniae, S. aureus, and H. influenzae pneumonias. Legionellosis is acquired through inhalation of an aerosol arising from contaminated water; cases present sporadically or as cluster outbreaks related to a point source exposure such as a reservoir, water tower, or air conditioning system [229].
Bacterial causes of CAP predominate, accounting for at least half of all adult cases, including older individuals [9,42]. S. pneumoniae is the leading cause of CAP in any adult age-group, with or without comorbid conditions [6,7,10]. It is estimated that pneumococcal infection accounts for 20% to 60% of all hospitalized patients with pneumonia [6]. Common bacterial pathogens other than S. pneumoniae include H. influenzae type b, S. aureus, and gram-negative bacilli [25,26,29,48]. H. influenzae type b is a small, pleomorphic gram-negative rod known for causing pneumonia in older adults and patients with underlying lung disease.
Atypical pneumonia (and the pathogens associated with this syndrome) is so labeled because the onset of illness tends to be subacute and the clinical exam and radiographic features lack the classical findings seen with typical cases of pneumonia. The most common atypical pathogens are Mycoplasma pneumoniae and C. pneumoniae, followed by Legionella spp. [9]. M. pneumoniae is a tiny bacterium that lacks a rigid cell wall. It is spread by droplet nuclei, and transmission within a community proceeds slowly over many weeks. Mycoplasma infection is a disease of adolescence and young adulthood, and it is the most common cause of atypical pneumonia in those younger than 40 years of age [66]. Small cluster outbreaks of pneumonia have been observed in large families, schools, nursing homes, and other closed population. There are about 60 different species of Legionella, but most disease is caused by Legionella pneumophila, a gram-negative rod usually transmitted via inhalation of aerosolized water contaminated with the bacteria [229].
The distribution of etiologic agents varies in relation to illness severity and management setting. In cases of relatively mild illness that permit outpatient treatment, blood cultures are rarely positive and the diagnosis relies on sputum culture and/or serial serology. In a Canadian study of CAP in the ambulatory setting, designed to determine the frequency of usual and atypical bacterial pathogens, an etiologic diagnosis was established in 48% of patients examined [222]. Of the 419 patients who had blood cultures, 7 (1.4%) were positive, all for S. pneumoniae. The atypical pathogen group (M. pneumoniae or C. pneumoniae) accounted for 29% of cases, S. pneumoniae for 6%, and Haemophilus spp. for 5%. The etiologic role of viruses was not studied [222].
A similar distribution and frequency was observed in a well-studied series from Spain, comparing pneumonia microbial etiology in three clinical management settings: outpatient, inpatient on the general care ward, and inpatient admissions to the ICU [29]. Among outpatients with CAP, the most frequently identified etiology was the atypical pathogen group (36%), followed by S. pneumoniae (35%), viruses (9%), and mixed etiologies (9%). As the severity of illness increased, marked by admission to the hospital general ward and ICU, the likelihood of mycoplasma or chlamydia etiology decreased substantially (14%) and the frequency of S. pneumoniae (43%), mixed bacterial pathogens (22%), S. aureus, Pseudomonas, and other gram-negative bacteria infection increased.
S. aureus is an uncommon cause of CAP but should be suspected during influenza outbreaks and in any patient with sepsis syndrome and multifocal pulmonary infiltrates. The role of S. aureus, and MRSA specifically, was examined in an observational study of 627 CAP cases admitted to 12 university-affiliated hospitals during the winter months (influenza season) of 2006–2007 [49]. Of the 595 patients from whom blood and sputum cultures were collected, a bacterial pathogen was identified in 107 (17%). The most common pathogen identified was S. pneumoniae (57 cases), followed by S. aureus (23 cases, 14 of which were MRSA). Thus, S. aureus accounted for 5% of the total and 22% of the cases in which the etiology was identified. Of the 23 patients with staphylococcal pneumonia, blood cultures were positive in 39% and sputum culture in 89%. Clinical features observed to be highly associated with S. aureus infection were multiple pulmonary infiltrates, altered mental status, illness severity requiring ICU admission, and intubation [49].
Studies have indicated that 5% to 20% of adult CAP may be caused by a viral pathogen [50]. However, as noted earlier, the role of respiratory tract viral infection in pneumonia is complex and perhaps underestimated. Studies utilizing newer diagnostic methods such as PCR have demonstrated rates of viral infection as high as 39% in patients presenting with pneumonia [9,34]. Because these studies rely on specimens and washings taken from the nasopharynx, rather than directly from the lung, it is not clear to what extent viral isolates in this setting represent primary pneumonia pathogens or concomitant viral upper respiratory infection that may impair pulmonary defense mechanisms and thus predispose to bacterial pneumonia.
Clinical and pathologic studies of pneumonia during influenza seasons have demonstrated clearly that influenza virus (types A and B) is an important cause of primary viral CAP [25,47]. Seasonal respiratory syncytial virus (RSV), rhinovirus, adenovirus, and parainfluenza virus are also commonly associated with pneumonia in adults [31,34,47]. Since the advent of coronavirus disease 2019 (COVID-19) pandemic, SARS-CoV-2 has superseded the usual viral respiratory pathogens. RSV and rhinovirus are especially common among older adults and nursing home residents [31]. Clinical studies that utilize viral culture for case definition have found that RSV can be recovered from 3% to 10% of older adults with pneumonia [30]. The paramyxovirus hMPV, first isolated in 2001 from children hospitalized with acute respiratory infection, has now been reported in all age groups and stages of disease, from asymptomatic carrier state to severe bronchitis and pneumonia [30].
The clinical recognition of CAP in adults is challenging because its presentation is similar to other acute respiratory illnesses such as pulmonary embolism/infarction and congestive heart failure [3,51,52]. Diagnosis relies on clinical features combined with radiographic findings; however, both the clinical presentation and chest x-ray abnormalities are variable and in part nonspecific, particularly in the elderly [3,29]. Common presenting symptoms and signs are:
Productive cough, purulent sputum
Fever with rigors (shaking chills)
Dyspnea
Pleuritic chest pain
Tachypnea
Tachycardia
Hypoxemia
Signs of consolidation (e.g., crackles, bronchial breath sounds, egophony)
Signs of pleural effusion (e.g., absent fremitus, dullness to percussion, decreased breath sounds)
Pneumonia in the elderly may present without a history of chills or fever, little cough, and a paucity of findings on exam and chest x-ray. Often in such cases, some combination of tachypnea, tachycardia, and altered mental status is the only sign [31,42].
Physical examination should focus on the chest, with auscultation to detect localized crackles (rales), bronchial breath sounds, and other signs of consolidation or pleural effusion [47]. Pulse oximetry should also be done. The most clinically significant individual findings are (in descending order) egophony, bronchial breath sounds, and dullness on percussion [53].
When pneumonia is suspected on the basis of these clinical features, chest radiography is the standard for confirming the diagnosis, and posteroanterior and lateral radiographs are recommended [3,29]. Some degree of infiltrate is almost always demonstrated on chest radiographs of patients who have been ill longer than 24 to 48 hours, although the appearance may be subtle or absent on initial presentation [29,47]. Pneumonia is described according to its anatomic distribution on chest radiographs as either lobar, multifocal/lobar, bronchopneumonic, or interstitial. Chest radiography also provides clues to alternative diagnoses having similar signs and symptoms. Computed tomography (CT), which is more sensitive than chest radiographs, may be useful in selected patients where diagnostic considerations are complex and initial radiographic studies are negative or inconclusive.
The characteristic symptoms and signs, combined with radiographic findings of an infiltrate, establish the clinical diagnosis of pneumonia. One validated prediction tool commonly used assigns 1 point for each of five clinical features present in conjunction with an infiltrate on chest radiography [54]:
Temperature >37.8°C (100.04°F)
Heart rate >100 beats per minute
Crackles on auscultation
Decreased breath sounds
Absence of asthma
A score of 4 or 5 indicates a 25% to 50% probability of pneumonia; a score of 2 or 3 indicates a probability of 3% to 10%; and a score of 0 or 1 represents a probability of l% or less [29,54]. Neither clinical nor radiographic features can reliably differentiate primary viral from bacterial or combined viral-bacterial pneumonia [9,31,32]. There are some features that, if present, aid in making the distinction. The presence of a viral epidemic in the community, such as influenza or RSV, increases the likelihood of a viral etiology [32]. The patient's age can also help identify the most probable cause; as noted previously, viral infections have been found more often in young children and adults older than 60 years of age compared with younger adults [9,24]. Chest pain is significantly more frequent in adults with bacterial pneumonia than in those with viral pneumonia [9]. Radiographic findings are generally not useful in identifying a specific pathogen, although multilobar infiltrates suggest infection with S. pneumoniae, S. aureus, or L. pneumophila, and patchy, interstitial infiltrates suggest a viral or mycoplasmal etiology [47,49].
The first use of the term atypical pneumonia was in 1938 to describe a series of seven patients who had developed an unusual form of tracheobronchitis [65]. There had also been descriptions of outbreaks of pneumonia that behaved atypically in Europe in the 1920s. In general, these outbreaks were milder and had higher recovery rates than expected for the typical case of pneumonia.
At the present time, atypical pneumonia is encountered, and managed, primarily in the outpatient setting. The causative pathogen most commonly identified in such cases is M. pneumoniae. According to CDC estimates, Mycoplasma infections occur at the rate of 2 million cases each year and are responsible for between 1 and 10 of every 50 cases of CAP [66].
Atypical pneumonia syndrome, best represented by mycoplasma infection, presents with a subacute prodrome of malaise, low-grade fever, headache, myalgia, and non-productive cough. Symptoms progress slowly over days to weeks; often patients are thought to have an upper respiratory infection or bronchitis and appear less ill than those with typical bacterial pneumonia [65,66]. The physical examination usually reveals fine rales but no signs of lung consolidation. In the early stage, there may be maculopapular skin eruptions and, on examination of the ear canal, bullous myringitis of the tympanic membrane. Chest x-ray reveals patchy alveolar densities or inhomogeneous segmental infiltrates, often bilateral involving the middle lobe and lingual. The white blood cell count may be normal or only slightly elevated. Full recovery is expected with no residual effects in a previously healthy individual. However, the disease can be severe in those with sickle cell anemia, older adults, and those with immunosuppression [65].
In younger patients, C. pneumoniae (TWAR strain) infection may present as atypical pneumonia. Outbreaks tend to occur in communal settings such as military units and college dormitories [231]. The illness is similar to that seen with mycoplasma infection, except that laryngitis is a prominent feature and nonexudative pharyngitis is common [26]. Chest x-ray may show patchy consolidation, interstitial infiltrates, or funnel-shaped lesions. The white blood cell count is usually normal.
The first recorded outbreak of legionellosis occurred in 1976 at an annual convention of the American Legion in Philadelphia. A total of 182 of the delegates (many of whom were elderly) became ill, and 146 were hospitalized. The mortality rate was 16%. Because the conference ended prior to the development of significant symptoms in many patients, hospitals all over the United States admitted one or more of the patients who had attended the convention. Despite an outpouring of resources, it took six months to isolate the organism, later named L. pneumophila. The pneumonia caused by the organism is commonly known as Legionnaires' disease [65].
L. pneumophila is a small gram-negative bacillus, atypical in its clinical presentation and for its lack of susceptibility to ß-lactam antibiotics. There are about 60 identified species of Legionella, although L. pneumophila is the primary pulmonary pathogen [230]. Legionella accounts for an estimated 8,000 to 18,000 cases of pneumonia requiring hospitalization in the United States each year [229,230]. Suspicion for infection with Legionella organisms should be high in older adults, in those with chronic underlying disease, and in all patients with pneumonia severe enough to require hospitalization.
Legionella bacteria are found in common sources of freshwater but not usually in sufficient numbers to cause disease. However, in commercial water systems such as those found in large buildings, storage tanks, cooling towers, decorative fountains, or hot tubs, Legionella growth exceeds the threshold required for transmission to susceptible hosts via aerosolization [229]. Because hotels, resorts, and cruise ships often use large, complex water systems and other aerosol-generating devices, travel is a risk factor for disease. This is also true for hospitals and long-term care facilities.
The onset of infection is marked by dry cough, fever of 38.3°C–38.8°C (101°F–102°F), then progressive symptoms and signs of pneumonia accompanied by multi-organ involvement—vomiting, diarrhea, headache, and altered mental status. Chest x-ray reveals rapidly progressive, asymmetric infiltrates without signs of consolidation. Prompt diagnosis relies on clinical suspicion, urine antigen assay, and specialized culture techniques.
Recommendations for the laboratory diagnosis and treatment of pneumonia in adults were first developed independently by the ATS and the IDSA, and guidelines were published in the 1990s and early 2000s [36,63,64]. Although each guideline differed somewhat, the principles of care were the same [36]. In order to avoid confusion associated with separate guidance, the IDSA and ATS jointly developed the current guideline for CAP, published in 2007 and updated in 2019 [47,235]. The IDSA/ATS guideline focuses on decision making about site of care, laboratory testing, empirical selection of antibiotics, and issues in the delivery of antibiotics (e.g., timing of the first dose, transition from parenteral to oral antibiotics, duration of therapy). Treatment of symptoms associated with CAP is not addressed in the guideline. A systematic review published in 2012 found insufficient evidence to determine if there is benefit to over-the-counter medications (e.g., mucolytics, cough suppressants) for cough associated with acute pneumonia [67].
The challenge of diagnosis is complicated by the lack of cost-effective, reliable, and rapidly available tests to discriminate between viral and bacterial pneumonia [37]. The IDSA/ATS guideline notes that routine cultures of sputum and blood are not recommended for patients treated in the ambulatory setting, as results rarely impact management decisions [47]. The primary reason for cultures and serologic testing is to identify specific pathogens suspected on the basis of clinical and epidemiologic findings or cases in which the results of testing will substantially alter the empirical treatment of the patient [47]. Testing is recommended when there are symptoms or signs of severe CAP (e.g., multilobar infiltrates, respiratory insufficiency, sepsis, leukopenia), when drug-resistant or unusual pathogens are suspected (e.g., MRSA, Pseudomonas, Legionella), and when patients do not show clinical improvement within 72 hours after starting empiric treatment [235].
Blood Culture
Blood cultures are optional and not recommended as a routine diagnostic test for CAP managed in the ambulatory setting. The principle reason is that the yield is low, and studies show that a positive culture leading to a change in antimicrobial therapy occurs in about 3% or fewer cases [55,56,222]. The IDSA/ATS guideline recommends pretreatment blood cultures in patients managed in the hospital who are classified as severe CAP, or being empirically treated for MRSA or gram-negative bacilli, or have a history of hospitalization and antimicrobial therapy within the previous 90 days [235]. Blood and sputum cultures should also be obtained in patients hospitalized with CAP and any one of the following conditions:
Cavitary infiltrates
Leukopenia
Active alcohol abuse
Chronic liver disease
Asplenia
COPD
Pleural effusion
Illness severity requiring admission to the ICU
Blood cultures are indicated for patients who have severe CAP, as they are more likely to have infection with a pathogen other than S. pneumoniae [235].
The ATS and the American College of Emergency Physicians (ACEP) also note that blood cultures need not be obtained routinely in all patients admitted with CAP [57]. Similarly to IDSA/ATS, ACEP adds that blood cultures should be considered for patients at higher risk, such as persons who have compromised immune systems, significant comorbidities, severe disease, or another risk factor for infection with resistant organisms [57].
Sputum Culture and Gram Stain
Sputum Gram stain and culture are also considered optional and are not recommended for routine management of adult CAP in the ambulatory setting. Pretreatment Gram stain and culture of respiratory secretions are recommended in adults hospitalized with CAP, in accordance with the same decision criteria outlined above for obtaining blood cultures [235]. Examination and culture of respiratory secretions should be performed only on specimens that meet quality performance measures for collection, transport, and processing of sputum samples.
The diagnostic utility of sputum Gram stain and culture has been demonstrated in patients hospitalized with pneumococcal pneumonia confirmed by positive blood culture. In a series of 58 patients from whom good quality sputum specimens (>10 inflammatory cells per epithelial cell) were submitted before or within six hours after initiation of antibiotic therapy, pneumococci were identified by Gram stain in 63% and by culture in 89% of cases [224].
Newer Diagnostic Techniques
Assays for the detection of antigen and other components of bacterial and viral pathogens have become a useful adjunct for establishing the etiology of pneumonia. Among these is the detection of bacterial antigen in the urine of patients with CAP. In a clinical series report, an assay for S. pneumoniae cell wall polysaccharide in urine was positive in 64% of patients with pneumococcal pneumonia; the sensitivity increased to 88% in patients who were bacteremic [225].
In a meta-analysis of published studies, the assay for detection of Legionella antigen in the urine of patients with pneumonia has been shown to have excellent specificity (99%) but only modest sensitivity (74%) [226]. Thus, a urine Legionella antigen assay is very useful to "rule in" the diagnosis but does not rule it out—a negative result should be interpreted with caution. The ATS/IDSA guideline recommends against routinely testing urine for Legionella antigen in adults with CAP, except when indicated by epidemiologic factors, such as association with a Legionella outbreak or recent travel or in adults with severe CAP [235]. Isolation of Legionella from sputum can be accomplished on selective media. Serologic diagnosis requires acute and convalescent serum; it is useful to confirm a case, but of little value in early diagnosis.
Testing for Viruses
Viral culture remains the criterion standard for diagnosis of viral pneumonia, but because of limitations such as the need for prompt transportation, time needed for viral detection, and the lack of sensitivity for all viruses, rapid antigen testing is often done. In adults, rapid testing has a sensitivity of 50% to 60% and a specificity of at least 90% [31]. Testing of nasal swab specimens is slightly less sensitive than testing of wash specimens, but wash specimens can be difficult to obtain in frail or cognitively impaired adults. Rapid RSV tests are usually not useful for adults, as the level of virus titers shed is low [31]. Diagnostic testing (PCR) for SARS-CoV-2 by nasopharyngeal swab may be performed on patients presenting with CAP in areas experiencing COVID-19 epidemic activity, although lower respiratory symptoms typically begin as upper respiratory symptoms are resolving or have already resolved.
Molecular diagnostic testing of sputum holds promise for providing a rapid and accurate etiologic diagnosis. Studies show that real-time PCR is significantly more sensitive and specific for the detection of the common respiratory viruses that cause CAP, as well as M. pneumoniae and C. pneumoniae [24,33]. However, molecular assays are expensive and not currently widely available [31].
Biomarkers
Over the past several years, researchers have been evaluating biomarkers for their utility in diagnosis and for determining duration of empirical therapy for presumed bacterial pneumonia. Procalcitonin has been shown to be superior to other commonly used markers for its specificity for bacterial infection and its ability to distinguish CAP from asthma and COPD [58,59]. This marker has predictive value; however, no biomarker should be used on its own and, if used, should be considered within the context of clinical and laboratory findings [59]. The 2019 IDSA/ATS guidelines do not recommend the use of procalcitonin to determine need for initial antimicrobial therapy [235].
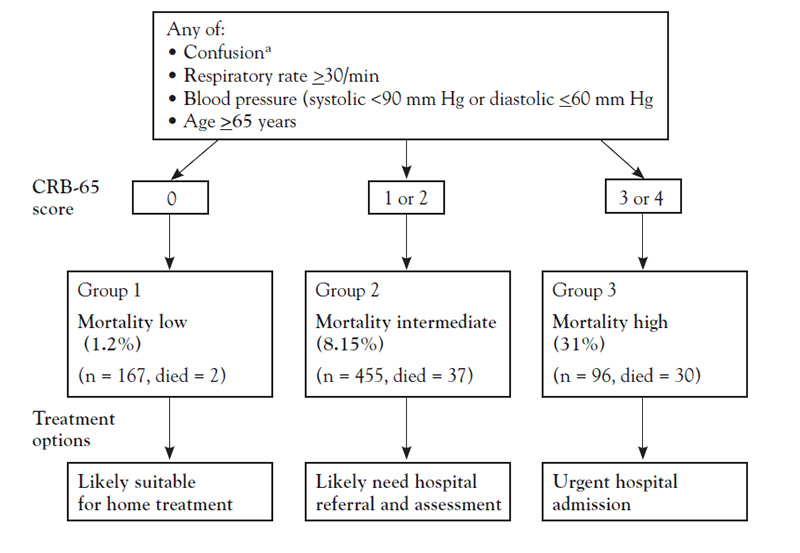
One of the most important decisions in the management of CAP is determining the site of care—that is, outpatient or inpatient and, if the latter, a general care floor or an ICU [68]. Many physicians admit patients to the hospital when they could be managed effectively on an outpatient basis [47]. This decision requires a careful evaluation of the severity of illness in the context of the personal and social well-being of the patient. Objective severity-of-illness scores and prognostic models can aid in identifying patients who may require hospitalization or admission to an ICU. The most widely used scales are the CRB-65 (confusion, respiratory rate, blood pressure, age 65 years or older) (Figure 1), the CURB-65 severity score (which adds urea level to the CRB-65 criteria), and the Pneumonia Severity Index (PSI) (Table 5). These assessment tools are recommended by the IDSA/ATS as an aid to clinical judgment in determining the site of care [47,69,70]. The scales have been compared, and they do not differ significantly in overall performance [71]. However, each scale has advantages and disadvantages, and none factor in all clinical considerations (such as comorbidities or social factors) [68]. CURB-65 and CRB-65 are easier to score as they have fewer variables and are more likely to correctly classify high-risk patients (i.e., high positive-predictive value) [72]. In contrast, the PSI is more sensitive and is better at determining which patients do not require hospitalization (i.e., low false-negative rate). About 30% to 60% of patients at low risk are unnecessarily admitted to the hospital according to the PSI score [68].
PNEUMONIA SEVERITY INDEX: POINT SCORING SYSTEM FOR STEP 2 OF THE PREDICTION RULE FOR ASSIGNMENT TO RISK CLASSES II, III, IV, AND V
| Characteristic | Points Assigneda | ||||
|---|---|---|---|---|---|
| Nursing home resident | +10 | ||||
| Demographic factor (age) | |||||
| Men | Age (yr) | ||||
| Women | Age (yr)-10 | ||||
| Coexisting illnessesb | |||||
| Neoplastic disease | +30 | ||||
| Liver disease | +20 | ||||
| Congestive heart failure | +10 | ||||
| Cerebrovascular disease | +10 | ||||
| Renal disease | +10 | ||||
| Physical-examination findings | |||||
| Altered mental statusc | +20 | ||||
| Respiratory rate ≥30 breaths/min | +20 | ||||
| Systolic blood pressure <90 mm Hg | +20 | ||||
| Temperature <35°C or ≥40°C | +15 | ||||
| Pulse ≥125 beats/min | +10 | ||||
| Laboratory and radiographic findings | |||||
| Arterial pH <7.35 | +30 | ||||
| Blood urea nitrogen ≥30 mg/dL | +20 | ||||
| Sodium <130 mmol/L | +20 | ||||
| Glucose ≥250 mg/dL | +10 | ||||
| Hematocrit <30% | +10 | ||||
| Partial pressure of arterial oxygen <60 mm Hgd | +10 | ||||
| Pleural effusion | +10 | ||||
| |||||
The PSI, CURB-65, and CRB-65 were developed to predict the risk of death. Because this risk does not always equate to the need for hospitalization and/or ICU admission, other scales have been developed. For example, SMART-COP provides a score based on a composite of systolic blood pressure, multilobar involvement on chest radiograph, albumin level, respiratory rate, tachycardia, confusion, oxygenation, and arterial pH [73]. SMART-COP was found to accurately predict the need for intensive respiratory or vasopressor support. Another tool, the Severe Community-Acquired Pneumonia (SCAP) score, includes points assigned to eight variables: arterial pH, systolic pressure, confusion, blood urea nitrogen level, respiratory rate, chest radiograph findings, pulmonary arterial oxygen tension (PaO2), and age (older than 80 years) [74]. SCAP has identified a larger proportion of patients as low risk compared with the PSI, CURB-65, and CRB-65, and is better than or as accurate as those scores at predicting adverse outcomes in hospitalized patients [74,75]. The IDSA/ATS guideline notes that the results of these objective criteria should always be accompanied by clinical judgment, including consideration of subjective factors, such as the availability of outpatient support resources and the patient's ability to safely and reliably take oral medication [47].
It is estimated that admission to an ICU is needed for 10% to 20% of patients hospitalized with CAP [76]. The IDSA/ATS guideline recommends two major and nine minor criteria to define severe pneumonia requiring ICU admission. [47]. The major criteria are septic shock requiring vasopressors or acute respiratory failure requiring intubation and mechanical ventilation. The presence of at least three of the following minor criteria suggests the need for ICU admission [47]:
Increased respiratory rate (≥30 breaths per minute)
Low PaO2/fraction of inspired oxygen ratio (≤250)
Multilobar infiltrates
Confusion/disorientation
Uremia (blood urea nitrogen level ≥20 mg/dL)
Leukopenia (white blood cell [WBC] count <4,000 cells/mm3)
Thrombocytopenia (platelet count <100,000 cells/mm3)
Hypothermia (core temperature <36°C [96.8°F])
Hypotension requiring aggressive fluid resuscitation
These criteria are based on empirical evidence from published studies and validated in pneumonia cohorts from different countries [235,77,78].
The goal of antibiotic treatment of pneumonia is to eradicate the infection or to reduce the bacterial load so the patient's own immune response is able to limit spread and speed recovery. The choice of regimen is based on consideration of known or suspected etiology, patient's age and severity of illness, comorbidities, and knowledge of resistance patterns in the community. When culture results are known, one should tailor therapy in accordance with antibiotic sensitivities and avoid unnecessarily prolonged treatment so as to minimize the potential for the development of resistance [37].
Pending results of cultures and serologic testing, an initial empiric treatment regimen is selected according to patient variables and clinical setting (Table 6) [47]. Patients with mild illness and no serious coexisting disease may be managed as outpatients. The 2019 ATS/IDSA guideline recommends amoxicillin 1 g three times daily, doxycycline 100 mg twice daily, or a macrolide (e.g., azithromycin 500 mg on first day then 250 mg daily or clarithromycin 500 mg twice daily) [235]. The macrolide monotherapy recommendation is conditional based on prevalence of local pneumococcal resistance (<25%) and provided the patient has not received antimicrobials within the previous three months [47]. S. pneumoniae resistance to macrolides is four times more likely in adult patients who have received this class of drug within the previous three months, in which case a fluoroquinolone or ß-lactam plus macrolide combination should be selected. Patients with comorbidities should receive broader spectrum treatment as they are more likely to harbor resistant pathogens and to be more vulnerable to poor outcomes if the initial regimen is inadequate. For outpatient adults with comorbidities, the ATS/IDSA guideline recommends one of the following options (in no order of preference) [235]:
Monotherapy: Respiratory fluoroquinolone (levofloxacin 750 mg daily, moxifloxacin 400 mg daily, or gemifloxacin 320 mg daily)
Combination therapy: Amoxicillin/clavulanate 500 mg/125 mg three times daily or a cephalosporin (cefpodoxime 200 mg twice daily or cefuroxime 500 mg twice daily) and doxycycline or a macrolide
RECOMMENDED EMPIRICAL ANTIMICROBIAL THERAPY ACCORDING TO 2019 IDSA/ATS GUIDELINE FOR THE MANAGEMENT OF COMMUNITY-ACQUIRED PNEUMONIA
| Site of Care and Patient Characteristics | Recommended Drug Class | Specific Drug Options | Level of Evidence | ||||
|---|---|---|---|---|---|---|---|
| Previously healthy outpatient, no exposure to antibiotics within past three months | Amoxicillin | — | Strong recommendation, moderate quality of evidence | ||||
| Tetracycline or macrolide | Doxycycline, azithromycin, or clarithromycin | Conditional recommendation, low-to-moderate quality of evidence | |||||
| Outpatients with comorbiditiesa or exposure to antibiotics within the previous three monthsb | Monotherapy with a respiratory fluoroquinolone | Moxifloxacin, gemifloxacin, or levofloxacin | Strong recommendation, moderate quality of evidence | ||||
| Combination therapy with a ß-lactam + macrolide or doxycycline | Amoxicillin-clavulanate or cephalosporin (cefpodoxime or cefuroxime) + azithromycin or clarithromycin or doxycycline | Conditional recommendation, low quality of evidence | |||||
| Inpatient (not ICU) | Respiratory fluoroquinolone | — | Strong recommendation, high quality of evidence | ||||
| ß-lactam + macrolide | — | Strong recommendation, high quality of evidence | |||||
| Inpatient (ICU) |
| Cefotaxime, ceftriaxone, or ampicillin-sulbactam | Strong recommendation, high quality of evidence | ||||
| |||||||
The choice of treatment option requires consideration of clinical and epidemiologic factors unique to patient and locale. As a rule, patients with recent exposure to one class of antibiotics should receive treatment with antibiotics from a different class to avoid possible bacterial resistance to the initial regimen. Fluoroquinolones should not be used routinely, as widespread use increases the possibility that resistance will develop.
The selection of a respiratory fluoroquinolone or a ß-lactam plus macrolide combination is recommended also for patients with CAP who are hospitalized on a general floor [235]. Patients with recent hospitalization and parenteral antibiotic treatment, as well as those with severe pneumonia usually requiring admission to an ICU, need empiric treatment for usual pathogens and consideration of coverage for S. aureus and gram-negative bacteria infection pending sputum and blood culture results. This is achieved with a regimen that combines a broad-spectrum ß-lactam (e.g., piperacillin/tazobactam) or a carbapenem with either azithromycin or a respiratory fluoroquinolone, adding vancomycin or linezolid to cover MRSA if there is clinical suspicion of S. aureus infection. Antibiotic selection for treatment of CAP in reference to specific pathogens is summarized in Table 7 [47].
ANTIBIOTIC SELECTION FOR SPECIFIC PATHOGENS ACCORDING TO IDSA/ATS GUIDELINE FOR THE MANAGEMENT OF COMMUNITY-ACQUIRED PNEUMONIA
| Pathogen | Preferred Antibiotic | Alternative Options |
|---|---|---|
| Streptococcus pneumoniae, not penicillin resistant | Penicillin G, amoxicillin | Macrolide, cephalosporins, clindamycin, doxycycline, respiratory fluoroquinolone |
| Streptococcus pneumoniae, penicillin resistant | Based on susceptibility (cefotaxime, ceftriaxone, fluoroquinolone) | Vancomycin, linezolid, high-dose amoxicillin |
| Haemophilus influenzae, non-ß-lactamase producing | Amoxicillin | Fluoroquinolone, doxycycline, azithromycin, clarithromycin |
| Haemophilus influenzae, ß-lactamase producing | Second- or third-generation cephalosporin, amoxicillin-clavulanate | Fluoroquinolone, doxycycline, azithromycin, clarithromycin |
| Mycoplasma pneumoniae/Chlamydophila pneumoniae | Macrolide, a tetracycline | Fluoroquinolone |
| Legionella spp. | Fluoroquinolone, azithromycin | Doxycycline |
| Pseudomonas aeruginosa | Antipseudomonalß-lactam plus ciprofloxacin or levofloxacin or aminoglycoside | Aminoglycoside plus ciprofloxacin or levofloxacin |
| Acinetobacter spp. | Carbapenem | Cephalosporin-aminoglycoside, ampicillin-sulbactam, colistin |
| Staphylococcus aureus, methicillin susceptible | Antistaphylococcal penicillin | Cefazolin, clindamycin |
| Staphylococcus aureus, methicillin resistant | Vancomycin or linezolid | Trimethoprim/sulfamethoxazole |
For adults who present with presumed viral CAP, it is unclear whether antibiotic treatment is beneficial. However, when the patient with CAP has epidemiologic, clinical, or laboratory evidence of active influenza, a neuraminidase inhibitor should be included in the treatment regimen [32,235]. During the COVID-19 pandemic, patients with upper respiratory tract infection progressing to clinical and/or radiographic signs suspicious for viral pneumonia should immediately receive SARS-CoV-2 diagnostic testing and consideration for COVID-19 treatment protocol. Updated guidance for managing patients with COVID-19 pneumonia is available at the IDSA website [94].
The time to the first dose of antibiotics for adults with CAP has engendered debate. A 2003 guideline developed by the IDSA recommended initiation of antibiotic therapy within four hours after hospitalization. Quality measures linked to this timeframe were developed by the Joint Commission and the Centers for Medicare and Medicaid Services [2,66,79,80]. Experts have criticized the timeframe requirement, with some noting that it has the potential to result in less-than-optimal care and others adding that diagnosis of pneumonia in the emergency department is challenging, especially in older patients who have an atypical presentation [51,52,79,80]. In a survey of 121 emergency physicians, 55% of the respondents said they had prescribed antibiotics to patients they did not believe had pneumonia in an effort to comply with the Centers for Medicare and Medicaid Services quality measure; 42% of these respondents said they had prescribed as such more than three times a month [80]. Sixty percent of the respondents said they did not believe that the guideline improves patient care. The results of a systematic review and a large-scale study have shown no decrease in mortality with a first dose administered within four hours [57,81,82].
As emphasized by the IDSA/ATS guideline committee, the recommendation at present is to begin antibiotic treatment promptly, without delay, administering the initial dose at the site of care (e.g., emergency department, clinic, office) where the diagnosis is first made [235].
With the availability of well-absorbed, effective oral antibiotics, hospitalized adults do not require intravenous antibiotics for the duration of treatment. Intravenous therapy can be changed to an oral regimen when the patient is hemodynamically stable, improving clinically, and able to take oral medications safely [235]. For patients on a general ward floor, this transition can often be made by the third hospital day; patients in the ICU usually reach this point within seven days. It is recommended that the oral antibiotic be either the same drug or within the same drug class as the intravenous antibiotic. Patients can be discharged from the hospital as soon as clinical stability has been achieved, provided they have no comorbidities requiring inpatient care and have a safe home environment and reliable follow-up. The IDSA/ATS note the following criteria for determining clinical stability [235]:
Temperature ≤37.8°C (100.04°F)
Heart rate ≤100 beats per minute
Respiratory rate ≤24 breaths per minute
Systolic blood pressure ≥90 mm Hg
Arterial oxygen saturation ≥90% or partial pressure of oxygen ≥60 mm Hg on room air
Ability to maintain oral intake
Normal mental status
The IDSA/ATS recommend that antibiotic therapy be given for a total of at least five days. The duration of therapy should be extended at least 48 to 72 hours beyond resolution of fever, assuming significant clinical improvement and no more than one pneumonia-associated active clinical sign [235]. A five- to seven-day course should suffice for most uncomplicated cases that show a prompt and satisfactory response to treatment.
The duration of treatment for gram-negative bacillary and staphylococcal pneumonia bears further comment. Unlike pneumococcal pulmonary infection, which usually heals without residual damage, these pathogens often cause destructive changes and small cavities in the lung, which clear slowly and heal by fibrosis. Thus, a more prolonged course of therapy (two to three weeks) should be considered, depending on severity of illness and response to therapy.
The clinical response to initial antibiotic therapy is unsatisfactory in approximately 15% of adults with CAP [47]. Failure to respond has no clear definition, and the IDSA/ATS guideline suggests using a systematic classification of cases, with attention to timing and character of response, as a guide to further evaluation and management. In general, treatment failures may be classified as persistent or non-responding, as a delay in achieving clinical stability, or as progressive pneumonia with clinical deterioration. Some clinical deterioration during therapy is not uncommon in the first 24 hours of treatment; as many as 45% of adults admitted to the hospital later require transfer to the ICU [47]. When the diagnosis of CAP is correct and guideline-recommended therapy has been used, the most common reason for treatment failure is an inadequate host response. For these patients, the appropriate management depends on individual case considerations, such as comorbidities, adequacy of pulmonary toilet, and whether the intravenous regimen has been reliably and consistently administered.
Guideline-directed management of CAP has been associated with many benefits. In one study, use of guideline-recommended antibiotics was associated with a significantly shorter time to clinical stability; clinical stability was achieved by seven days in 71% of patients treated with guideline-recommended antibiotics and in 57% of those treated with nonadherent regimens [15]. Adherence to recommendations guiding the selection of antibiotics was also associated with a significantly shorter length of stay (8 vs. 10 days) and a significantly lower overall in-hospital mortality rate (8% vs. 17%) [15]. In a Canadian study of adults (mean age: 51 years) who, in the main, had mild pneumonia, guideline-adherent selection of antibiotic treatment was associated with a lower mortality rate (1%) than that found when treatment selection that was not adherent to guidelines (6%) [83]. The mortality rate associated with the use of macrolides was also significantly lower than that with the use of fluoroquinolones (0.2% vs. 3%) [83]. In a large study of 54,619 patients who were hospitalized at 113 community hospitals (not in the ICU), use of guideline-adherent treatment was associated with a lower in-hospital mortality rate, lower rate of sepsis and renal failure, and shorter length of stay and duration of parenteral therapy [17]. Decreased mortality has also carried over to populations with more severe disease, with nonadherent therapy being associated with an increase in inpatient mortality (25% vs. 11%) among older adults (median age: 71 years) who were admitted to an ICU [16]. In addition to the higher rates of adverse outcomes, the low rate of adherence has also resulted in the inappropriate use of antimicrobials in at least half of cases [21].
Despite the benefits of guideline-directed treatment and the wide dissemination of the guidelines for management of pneumonia in adults, adherence has been low, especially with regard to antibiotic selection, with rates ranging from 9% to 82% [15,16,17,18,20]. In a study of more than 34,000 patients in a managed care organization, adherence to the 2003 IDSA guidelines in ambulatory settings was 52% for patients who were previously healthy and had not had recent exposure to antibiotics [20]. The rate of adherence was better (82%) for patients who had comorbidities and no recent exposure to antibiotics [20]. One study found that most cases of guideline-discordant use of antibiotics for older adults represent undertreatment [15]. The use of recommended antibiotics in the emergency department significantly increased from 1993 through 2008, but the percentage of patients receiving these drugs is still not optimal, with 60% to 70% of patients not receiving recommended antibiotics [84].
As the low rate of guideline adherence demonstrates, disseminating clinical practice guidelines alone is not enough to change practice. Physician education should address barriers to guideline adherence, including lack of familiarity, concerns about the practicality of recommended antibiotics, increased cost, lack of documented improved outcomes, and potential conflict with other guidelines [23]. Physician practices and healthcare systems should implement strategies that have changed physician behavior in other health condition settings, such as face-to-face educational outreach, use of local opinion leaders, and individualized audit with peer-comparison feedback [85]. In a study of six Dutch hospitals, significant increases in adherence to guideline-recommended care were achieved with an intervention that included the establishment of a local committee, a lecture by a respected opinion leader, feedback on performance, and critical care pathway pocket cards [86]. The intervention also included a second phase that focused on aspects of treatment in most need of improvement. In another study, weekly e-mail reminders listing performance data on antibiotic administration recommendation for individual emergency physicians helped to increase guideline adherence [87]. The use of a standardized evidence-based order set was associated with a decrease in mortality and was also cost-effective [88].
Evidence suggests that severe pneumonia is a cause of long-term morbidity and excess mortality among adults. In a population-based follow-up study of adults with CAP in Canada, conducted over a median of four years, the re-hospitalization rate for pneumonia was 16% to 72% for all causes [9].
The PSI classification and the time to clinical stability can both help predict adverse outcomes. Mortality has been reported to be higher for people originally classified as PSI class V than PSI classes I and II, with rates of 82% compared with 15% [9]. A time to clinical stability of more than 72 hours has been associated with a significantly higher rate of adverse outcomes than shorter times [90]. Overall, severe CAP has been associated with a 30-day re-hospitalization rate as high as 20%, a 30-day mortality rate as high as 23%, and all-cause mortality within one year as high as 28% [76].
These findings indicate that adults with severe pneumonia should be followed up closely to monitor for adverse events after discharge. The time to clinical stability is a useful guide for a follow-up plan; patients in whom clinical stability is not achieved until more than 72 hours after admission should be seen in follow-up soon after discharge [3,90]. Strategies to prevent influenza and pneumonia should also be emphasized for all hospitalized patients. When indicated, immunization against pneumococcal infection should be initiated before or shortly after discharge, as recommended by the Advisory Committee on Immunization Practices (ACIP) and others [47,91,227].
After a diagnosis of pneumonia has been made, patient education should include directions for use of the antibiotic and information on potential untoward effects of the drug. Follow-up instructions, depending on the clinical situation, may include 24-hour telephone contact or follow-up in the office after 24 to 48 hours. This will improve adherence to the prescribed therapy, provide an opportunity to address side effects of drug therapy, and allow progress to be monitored. The need for hospitalization should be assessed throughout the course of the illness. Education should also include instructions to drink plenty of fluids and to use an antipyretic to control fever and myalgias when needed. Use of cough suppressants should be avoided, as the cough reflex and sputum expectoration enhance removal of thick secretions. However, in the event of a constant, nonproductive cough, as found especially with mycoplasmal infection, a narcotic such as codeine at night may allow for more restorative sleep.
Provisions for patients with limited English language proficiency are required under federal law, and the U.S. Department of Health and Human Services and the Office of Civil Rights view a lack of adequate interpretation as discrimination, based on the Civil Rights Act of 1964 [19]. According to U.S. Census Bureau data, nearly 68 million Americans speak a language other than English at home, with more than 25 million (8.2% of the population) reporting that they speak English less than "very well" [127]. Immigrant patients with chronic illness may feel unable to return to their home countries due to a lack of available medical care. Changes in healthcare law restricting federal funding of services to only legal residents may cause significant problems for certain facilities, with conflicts arising from providing life-saving care for patients who have no means of reimbursement and no medical services waiting for them in their home countries.
A semi-retired man, 68 years of age, presents one Sunday morning to the emergency department with malaise, fever, productive cough, and right pleuritic chest pain of less than 24 hours duration. He has been active, works as a custodian, has never been hospitalized, takes no medications, and does not regularly see a physician. On review of systems, the patient states that he gave up smoking years ago, has a mild chronic cough and morning sputum production, and has noted mild dyspnea on exertion for the past six months. He drinks only beer, never after work, but every Saturday afternoon he likes to take a six-pack out into the backyard, where he relaxes in his lounge chair. When asked whether there was anything different about the Saturday before the onset of the illness, his wife relates that he consumed two six-packs and failed to come in that evening. She found him later, after dark, asleep in his lounge chair, and helped him in to bed. He awoke this morning with fever and chills. On exam, the patient's temperature is 102.6°F, blood pressure 154/80 mm Hg, pulse 94 beats per minute, and respiration 20 breaths per minute. He is alert, with signs of mild emphysema and crackles audible over the right lower posterolateral chest. The chest x-ray shows patchy alveolar opacification in the right lower lobe and slight cardiomegaly.
The working diagnosis here is CAP, likely caused by S. pneumoniae or H. influenzae, as the patient has no prodromal upper respiratory symptoms to suggest viral or mycoplasma infection.
Why is this happening now? COPD/chronic bronchitis appears to have developed in recent years. Such patients have damaged, poorly functioning mucociliary epithelium and rely on compensatory cough to promote tracheobronchial clearance. Moreover, they often have colonization with pneumococcus and H. influenzae. An additional risk factor in this patient may be mild heart failure with ambient alveolar edema in the basal segments of the lower lungs. Excessive beer consumption the evening before onset of illness made him somnolent and suppressed his cough reflex, thus rendering him vulnerable to aspiration and retention of upper tract secretions (if not gastroesophageal reflux and aspiration). Encumbered by alveolar edema, and perhaps impaired by the metabolic effects of alcohol, pulmonary macrophages in the basal segment of the right lung were simply overwhelmed.
What is the best site of care and treatment for this patient? While he does not meet the criteria for ICU admission, his age, comorbidities, degree of illness, and social situation taken together suggest the need for hospital admission, parenteral antibiotic therapy, and close observation, anticipating a short hospital stay. He was treated with a ß -lactam and macrolide, improved rapidly, and was discharged day ٣ on a matching oral regimen, to complete a ١٠-day course of therapy.
What preventive measures were taken to reduce the risk of this happening again? The 20-valent pneumococcal conjugate vaccine (PCV20) (Prevnar 20) was administered prior to discharge and arrangements were made for primary care follow-up. The patient and his wife were educated regarding the need for yearly influenza vaccination. The role of alcohol was discussed, as well as the importance of keeping the Saturday afternoon beer consumption within clearly defined limits.
Pneumonia is the leading infectious cause of death in children worldwide [228]. An estimated 808,000 children died of pneumonia in 2017, which accounts for 15% of all deaths in children younger than 5 years of age. Children and families in every country are affected, but childhood pneumonia is most prevalent in South Asia and sub-Saharan Africa. The most common etiologic agents are S. pneumoniae, H influenzae, RSV, and, in young children with HIV, Pneumocystis jiroveci. Childhood risk factors for contracting pneumonia include malnutrition, indoor air pollution (e.g., caused by cooking and heating with biomass fuels, parenteral smoking), and pre-existing illness such as active HIV infection and measles [228].
Viral pathogens are reported to be responsible for most cases of CAP in preschool-aged children and as many as 80% of cases in children younger than 2 years of age [30]. In children younger than 2 years of age, the most common viral pathogen, occurring in up to 40% of cases, is RSV; other viral pathogens include adenoviruses, bocavirus, human metapneumovirus, influenza A and B viruses, parainfluenza viruses, coronaviruses, and rhinovirus [9,29,30,32].
RSV infection is common in infants and young children; it is estimated that most children have had RSV by 2 years of age [31]. It is the leading cause of pneumonia in infants younger than 1 year of age, with 25% to 40% of those infected developing signs of pneumonia or bronchiolitis [29]. Premature birth, very young age, compromised immune system, and impaired lung or heart function are all risk factors for RSV-related pneumonia in infants. In contrast to preschool-aged children, the percentage of viral cases is much lower among older children and adolescents (10 to 16 years of age), and pneumonia caused by RSV is rare in this population.
In older children, viral and atypical bacterial infection account for most mild CAP managed in the ambulatory setting, while pyogenic respiratory bacterial infection is responsible for the majority of CAP in seriously ill, hospitalized children [30]. S. pneumoniae is the most common bacterial pathogen in school-aged children. Studies show that atypical pathogens account for 3% to 23% of cases, most commonly mycoplasma in older children and chlamydia in infants and young children [30]. A 2009 European study examining causative agents in hospitalized pediatric patients with radiographic evidence of pneumonia found bacterial infection in 53% of patients and viral pathogens in 67% of patients, with 33% of children in the study showing evidence of both [63]. S. pneumoniae was the most common bacterial pathogen (46%), followed by M. pneumoniae and C. pneumoniae. The primary viral pathogens identified were influenza A or B, parainfluenza, rhinovirus, RSV and, human metapneumovirus [63].
As with adults, severe CAP caused by S. aureus is encountered during outbreaks of influenza [223]. Legionella spp. and fungal pathogens are uncommon in children. A combination of viral and bacterial pathogens occurs in up to half of children with CAP [30,32].
The clinical presentation of CAP in children is similar to that in adults, but can vary according to age and developmental stage. For example, cough productive of purulent sputum may be elicited in older children, but nonproductive cough is common in young children and infants [30,60]. Nonspecific irritability and restlessness may be the primary symptoms in infants.
During the physical examination of pediatric patients, the clinician should look for signs of hypoxia and dehydration, as well as retractions, tachypnea, and use of accessory muscles of respiration [60]. The clinician should also evaluate the upper respiratory tract for evidence of rhinorrhea, otitis media, and pharyngitis [60]. Auscultation of the chest should be carried out, and the Pediatric Infectious Diseases Society (PIDS)/IDSA guideline recommends pulse oximetry for children with suspected hypoxemia [30].
One of the most common reasons for pediatric emergency room visits is fever, and fever is present in 88% to 96% of identified pneumonia cases in developed countries [70]. However, children with fever and wheezing commonly have either upper respiratory disease or reactive airway disease. As with pneumonia in adults, the accuracy of any one sign or symptom in predicting the likelihood of pneumonia is limited [61]. Nonspecific symptoms such as vomiting and abdominal discomfort are common. Careful attention should be given to the chest exam, as diminished breath sounds and fine end-inspiratory crackles are subtle, important clues to the presence of pneumonia in the pediatric patient. In one study, non-specific crackles were present in more than 90% of children with pneumococcal or mycoplasma pneumonia [70]. Infants with pneumonia commonly present with poor feeding and irritability as well as tachypnea, retractions, grunting, and hypoxemia; cough is rare [64].
Several clinical rules have been developed for predicting the likelihood of pneumonia in children on the basis of discernible clinical signs. The presence of at least two of the following signs—fever, tachypnea, and reduced oxygen saturation—is associated with a high probability of the disease; the absence of all three indicates a low probability [61]. Other signs of respiratory distress, such as cough, nasal flaring (in infants), rales, and decreased breath sounds, have also been found to be independent predictors of pneumonia in infants and children [60,62]. Bronchial breath sounds, rales, and dullness to percussion are more likely to occur in older children and adolescents [60].
Unlike diagnosis in adults, a chest radiograph is not the diagnostic standard to be applied for all CAP in children. The PIDS/IDSA guideline notes that routine chest radiographs are not necessary for children who can be treated as outpatients [30]. However, posteroanterior and lateral chest radiographs should be obtained when there is fever and respiratory distress suspected or documented hypoxemia, or illness severe enough to warrant hospitalization [30]. In a study of 99 children hospitalized with what was later determined to be pneumonia, the most common abnormal finding was "diminished" breath sounds; only 21% were described as having "normal" breath sounds. Radiographic evidence of pulmonary consolidation was present in 79% of patients, and correlation between diminished breath sounds and a positive chest x-ray was 60.2% [63].
Laboratory Tests
Unlike the situation in adults, titers of shed virus in children are high [31]. Thus, rapid antigen testing of nasal or throat swabs for influenza and other respiratory viruses should be done for infants and young children [30]. However, it should be noted that negative results of influenza virus on rapid antigen tests do not conclusively rule out infection with influenza virus. Testing for C. pneumoniae is not recommended.
Blood cultures are not routinely needed but should be obtained in children hospitalized for moderate-to-severe pneumonia that is presumed to be bacterial [30]. Urinary antigen detection tests often have false-positive results in children and are therefore not recommended for the diagnosis of pneumococcal pneumonia.
The PIDS/IDSA guideline, published in 2011, addresses the management of CAP in children 3 months of age and older who are otherwise healthy; the guideline does not provide guidance for neonates and infants younger than 3 months of age or children with comorbidities [30]. The guidelines were developed in an effort to decrease morbidity and mortality, as had been shown with the guideline for adults. Similar to the IDSA/ATS guideline, the management issues addressed in the PIDS/IDSA guidelines are site of care and selection and duration of antibiotic therapy, as well as adjunctive surgical and nonantibiotic treatment for complications. As with the guideline for adults, treatment of pneumonia-related symptoms is not included in the pediatric guideline. The discussion here is limited to site of care and antibiotic therapy.
Site of Care
To aid in making site-of-care decisions, the PIDS/IDSA guidelines recommend that a child or infant with CAP be hospitalized if any of the following factors are present [30]:
Moderate-to-severe illness, as defined by several features, including respiratory distress and hypoxia
Suspected or documented infection caused by a pathogen with increased virulence, such as community-associated MRSA
Uncertainty about care at home or availability for follow-up
Most children with pneumonia do not require care in an ICU. The guideline states that a child should be admitted to an ICU or a unit with continuous cardiorespiratory monitoring capabilities if the child [30]:
Requires invasive ventilation via a nonpermanent artificial airway (endotracheal tube)
Has impending respiratory failure or sustained tachycardia, inadequate blood pressure, or need for pharmacologic support of blood pressure or perfusion
Has altered mental status as a result of pneumonia
Has a pulse oximetry measurement <92% on inspired oxygen of ≥0.50
Requires acute use of noninvasive positive pressure ventilation
Selection of Antibiotics or Antivirals
The PIDS/IDSA guideline recommends empiric antibiotic therapy according to patient age, immunization status, and site of care. Among infants and children 3 months to 5 years of age, antibiotic therapy is not routinely recommended because viral infection is the predominate cause of CAP in this age group [30]. When influenza virus is the suspected cause of pneumonia, influenza antiviral therapy should be started as soon as possible, as maximal benefit is achieved when treatment begins within 48 hours of the onset of symptoms. (Treatment should not be delayed while waiting for the results of viral testing.) The PIDS/IDSA guideline recommends three U.S. Food and Drug Administration (FDA)-approved influenza antiviral therapies: oseltamivir (Tamiflu), zanamivir (Relenza), and amantadine (Symmetrel) [30]. A fourth antiviral therapy, rimantadine (Flumadine), is included in the guideline, with a note that the agent is FDA-approved for prophylaxis—not treatment—in children 1 year of age and older [30]. The guideline adds that data on the safety and efficacy of the agent for children 1 year of age and older have been published.
As in adults, S. pneumoniae is the most common bacterial cause of CAP in children; thus, if a bacterial pathogen is thought to be the cause, amoxicillin or amoxicillin/clavulanate is recommended as first-line therapy for mild-to-moderate illness in previously healthy children 3 months to 5 years of age who are up-to-date with immunization [30]. Several alternatives can be used for children who are allergic to amoxicillin (Table 8). Amoxicillin is also the preferred antibiotic for mild-to-moderate CAP in adolescents and children 5 years of age and older [30]. For children of all ages, especially children older than 5 years of age and adolescents, a macrolide is recommended if an atypical bacterial pathogen is thought (or documented) to be the cause.
EMPIRIC ANTIBIOTIC THERAPY FOR COMMUNITY-ACQUIRED PNEUMONIA IN CHILDREN ACCORDING TO PIDS/IDSA GUIDELINE
| Site of Care, Patient Characteristics | Presumed Bacterial Pneumonia | Presumed Atypical Pneumonia | ||||
|---|---|---|---|---|---|---|
| Outpatient | ||||||
| <5 years |
|
| ||||
| ≥5 years |
|
| ||||
| Inpatient (all ages) | ||||||
| Fully immunizedb and minimal local penicillin resistance in invasive strains of pneumococcus |
|
| ||||
| Not fully immunized and/or significant local penicillin resistance in invasive strains of pneumococcus |
|
| ||||
| ||||||
For fully immunized infants and school-aged children who are hospitalized, treatment with ampicillin or penicillin G is recommended when local epidemiologic data show a low level of penicillin resistance to S. pneumoniae [30]. For children who are not fully immunized or are hospitalized in an area with a high level of penicillin-resistant S. pneumoniae, treatment with a third-generation cephalosporin (ceftriaxone or cefotaxime) should be given intravenously. If M. pneumoniae or C. pneumoniae is strongly suspected, treatment should include a macrolide (orally or intravenously) with a ß-lactam and diagnostic testing should be done as soon as possible [30]. The PIDS/IDSA guideline also recommends antimicrobial treatment for specific pathogens; however, a discussion of all possible pathogens is beyond the scope of this course.
According to a systematic review, zinc supplementation in addition to standard antibiotic therapy was not shown to have significant benefit on clinical recovery of severe or nonsevere pneumonia in children 2 to 59 months of age [89].
Duration of Therapy
Most studies have evaluated 10-day therapy, and this duration is associated with good outcomes. However, a shorter duration may be equally as effective, especially for mild disease treated on an outpatient basis [30].
Benefits of Guideline Adherence
Because the PIDS/IDSA guideline for management of CAP in children is relatively recent, data are lacking on the benefits of guideline-adherent treatment in the pediatric population. One study did show that more children received appropriate antibiotics after the development of a clinical practice guideline based on the PIDS/IDSA guideline and an antimicrobial stewardship program [14]. It is assumed that more data will become available over time.
Late Complications
Data on the long-term effects of pneumonia during childhood are lacking. A systematic review demonstrated that severe pneumonia in children younger than 5 years of age is associated with long-term sequelae, with restrictive lung disease being the most common sequela [95]. Overall, major respiratory sequelae (e.g., restrictive lung disease, obstructive lung disease, bronchiectasis) occurred in 5.5% of children treated on an outpatient basis and in 13.6% of children hospitalized for treatment [95]. Sequelae occurred in approximately 54% of children who had pneumonia caused by adenovirus.
The primary preventive strategy for pneumonia is immunization with pneumococcal and influenza vaccines, especially for adults older than 65 years of age, young children, and other individuals in high-risk groups (Table 9) [91]. Additional preventive measures include improved hand hygiene compliance and adherence to healthy lifestyle behaviors, including cigarette smoking cessation.
HIGH-PRIORITY AND HIGH-RISK GROUPS FOR VACCINATION
| Vaccination | Priority Groups | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annual influenza vaccination |
| ||||||||||||
| Pneumococcal vaccination |
|
Pneumococcal vaccines have been improved over time by broadening the coverage of serotypes in the vaccine to include those that are causing the most common invasive infections. In the past, a single agent, the 23-valent polysaccharide vaccine (PPSV23) (Pneumovax), had been recommended for use in selected adults with conditions of impaired immunity, and for all adults older than 65 years of age [96]. This vaccine provided some protection against 85% to 90% of the pneumococcal serotypes that cause invasive disease in these populations [97]. In 2021, PCV15 (replacing PCV13) and PCV20 were introduced for adults [240].
The use of pneumococcal conjugate vaccines in the pediatric age group has been followed by a reduction in the incidence of pneumococcal disease among children, and, indirectly, among adults as well. By 2013, the incidence of invasive pneumococcal disease caused by serotypes represented in the PCV13 vaccine had declined in the adult population older than 65 years of age by approximately 50% compared with 2010 [227]. In 2012, upon approval by the FDA, the ACIP recommended the use of PCV13 for adults with immune deficits and other conditions that impose a heightened risk for invasive pneumococcal infection. After reviewing additional data in 2014, the ACIP extended its recommendation for PCV13 use to all adults older than 65 years age [227].
In 2021, the ACIP again amended its recommendation for PCV use in older adults, based on sharp declines in pneumococcal disease among adults since the advent of PCV13 use in children [227]. The ACIP now recommends a routine single dose of PCV20 for adults older than 65 years of age (Table 10). Alternatively, one dose of PCV15 may be administered followed by PPSV23 given at least one year after the PCV15 dose. A minimum interval of eight weeks between PCV15 and PPSV23 can be considered for adults with an immunocompromising condition, cochlear implant, or cerebrospinal fluid (CSF) leak to minimize the risk of invasive pneumococcal disease caused by serotypes unique to PPSV23 in these vulnerable groups [96,227,240].
IMMUNIZATION SCHEDULE RECOMMENDED BY THE ACIP
| Vaccination | Recommended Recipients | |||
|---|---|---|---|---|
| Influenza vaccination (annually)a | Adults and children 6 months of age and older | |||
| Pneumococcal vaccination (PCV20 OR PCV13 in series with PPSV23, 12 months apart)b,c | Adults 65 years of age and older | |||
| High-risk children and adults (5 to 64 years of age) | ||||
| Haemophilus influenzaeb(series of 4) | Infants at 2, 4, 6, and 12 to 15 months of age | |||
| Pneumococcal conjugate vaccine (series of 4) | Infants at 2, 4, 6, and 12 to 15 months of age | |||
| ||||
As of June 2024, ACIP recommends PCV21 as an option for adults aged 19 years of age or older who currently have a recommendation to receive a dose of PCV [239]. Current information, schedules, and guidance for adult immunizations is maintained at the CDC/ACIP website at https://www.cdc.gov/vaccines/schedules.
The influenza vaccine is developed each year to contain the three virus strains that are expected in the upcoming influenza season. The vaccine has traditionally been a trivalent inactivated vaccine (TIV), but in 2003, a trivalent live, attenuated influenza vaccine (LAIV) was introduced in the United States [97]. In 2010, a new high-dose formulation of TIV became available. The LAIV, which contains four times the amount of influenza antigens as other TIVs, is designed to induce a higher immune response in older people [97]. The LAIV is administered as a nasal spray.
The ACIP once recommended a risk-stratified approach to influenza vaccination, but it updated its recommendations to universal vaccination beginning in the 2010–2011 influenza season (Table 10) [91]. Routine annual influenza vaccination is recommended for all persons 6 months of age and older who do not have contraindications. The ACIP provides updated recommendations for the use of licensed and age-appropriate seasonal influenza vaccines in the United States. Current ACIP influenza vaccine recommendations are available at the CDC website [236].
In their guideline for the management of CAP, the IDSA/ATS make the following strong recommendations for prevention based on the ACIP recommendations [47]:
All persons 50 years of age and older, others at risk for influenza complications, household contacts of high-risk persons, and healthcare workers should receive inactivated influenza vaccine as recommended by the ACIP (level I evidence).
The intranasally administered LAIV is an alternative vaccine formulation for some persons 5 to 49 years of age without chronic underlying diseases, including immunodeficiency, asthma, or chronic medical conditions (level I evidence).
Pneumococcal vaccines are recommended for persons 65 years of age and older and for those with selected high-risk concurrent diseases, according to the current ACIP guideline (level II evidence).
The IDSA/ATS recommends that vaccination status be assessed in all patients with CAP at the time of hospital admission, especially in those with underlying medical conditions [47]. If vaccination is needed, it may be done either at hospital discharge or during outpatient treatment.
The PIDS and the IDSA also echo the ACIP recommendations in their guideline [30]:
Children should be immunized with vaccines for bacterial pathogens, includingS. pneumoniae, H. influenzae type b, and pertussis (strong recommendation, high-quality evidence).
All infants 6 months of age or older and all children and adolescents should be immunized annually with vaccines for influenza virus (strong recommendation, high-quality evidence).
Parents and caretakers of infants younger than 6 months of age, including pregnant adolescents, should be immunized with vaccines for influenza virus and pertussis to protect the infants from exposure (strong recommendation, weak-quality evidence).
High-risk infants should be provided immune prophylaxis with RSV-specific monoclonal antibody to decrease the risk of severe pneumonia and hospitalization caused by RSV (strong recommendation, high-quality evidence).
Declining rates of pneumonia and pneumonia-related deaths are thought to represent the effectiveness of influenza and pneumococcal vaccination [40,98,99]. In a study of a community-dwelling older population, influenza vaccination decreased the risk of hospitalization for pneumonia or influenza, as well as the risk of death, across 10 influenza seasons [7]. Systematic reviews and meta-analyses have shown that pneumococcal vaccination reduces the incidence of invasive pneumococcal disease in both older adults and children, although the findings are unclear for adults with chronic illness [100,101]. Other studies of adults have shown that pneumococcal vaccination is associated with benefit in terms of a lower risk of adverse outcomes associated with the disease. For example, in a study of nearly 3,500 older people (median age: 75 years) who were hospitalized for CAP, the rate of mortality or ICU admission was 40% lower among those who had received prior PPSV23 vaccination [8].
Among children, the introduction of the PCV7, PCV13, and later PCV15 has led to a substantial decrease in the rate of invasive pneumococcal disease, but the decrease in the rate of CAP has been less dramatic. Early studies showed substantial improvements in the hospitalization rate for CAP only among young children. In one study, the hospitalization rate decreased 39% for children younger than 2 years of age [98]. In another study, the decrease was substantial only for children younger than 1 year of age (22%) and was minimal for children 1 to 5 years of age; the rate increased for adolescents and children older than 5 years of age [4]. The rate of outpatient CAP visits has not changed significantly for this population [5,10].
Despite the wide distribution of the ACIP immunization schedule and public campaigns about the importance of vaccination, rates of both pneumococcal and influenza vaccination remain relatively low. According to CDC estimates, influenza vaccination coverage for the 2021–2022 season among adults 18 years of age or older was 49.4%, a decrease of 0.8 percentage points from the prior season [102]. Coverage among children 6 months through 17 years of age was 57.8%. Roughly half (51.4%) of all persons 6 months and older were vaccinated during the 2021–2022 season. Influenza vaccination coverage was lower among Hispanic (37.9%) and non-Hispanic Black (42.0%) adults than White (53.9%) adults [102]. Coverage among adults 65 years of age or older (73.9%) was higher compared with younger age groups (37.1% to 52.4%). Among healthcare personnel, influenza vaccine coverage during the 2021–2022 influenza season was 79.9%. When analyzed by setting, vaccine coverage was highest among healthcare personnel in ambulatory care and hospital settings with vaccine requirements (81.2% and 92.0%, respectively), and lowest in medical settings without vaccination requirements or recommendations (48.1%) [103].
The national rate of influenza vaccination among all adults has improved over the past decade, yet racial disparities persist. Comparing rates at five-year intervals from 2005 to 2015, the rate of vaccination more than doubled for adults younger than 65 years of age in all ethnic categories (Table 11) [113]. The rate disparity between white adults (44.2%) and that observed for black (36.7%) and Hispanics (31.2%) remains evident. Previous studies have also shown higher rates of vaccination for white older adults compared with black and Hispanic older adults [104,105,106,107]. Racial disparities have also been found when rates of pneumococcal and influenza vaccination for residents of long-term care facilities were compared, with substantially lower rates for black residents [108,109,110].
RATE OF INFLUENZA VACCINATION AMONG ADULTS ACCORDING TO AGE AND RACE/ETHNICITY, FIVE-YEAR INTERVALS, 2005–2015
| Age/Ethnicity | Rate | ||
|---|---|---|---|
| 2005 | 2010 | 2015 | |
| 18 to 44 years of age | 10.1% | 24.6% | 30.9% |
| 45 to 64 years of age | 20.2% | 37.8% | 45.1% |
| 65 years of age and older | 59.7% | 63.9% | 69.1% |
| White | 22.5% | 36.9% | 44.2% |
| Black | 15.5% | 28.1% | 36.7% |
| Hispanic | 12.0% | 26.5% | 31.2% |
In 2018, the estimated overall rate of pneumococcal vaccination coverage among adults older than 65 years of age was 69% [237]. The rate was substantially lower (approximately 23%) among younger adults in high-risk groups. Selected data from this report are summarized in Table 12[237].
In addition, adherence to recommendations for pneumococcal and influenza vaccination of older adults admitted to the hospital has been low. In a study of nearly 105,000 patients 65 years of age and older who had not received either vaccination before admission to the hospital, 99.4% did not receive the pneumococcal vaccine and 97.3% did not receive the influenza vaccine before hospital discharge [111].
Rates of both pneumococcal and influenza vaccination are higher among children than adults. Overall, approximately 83% of children 19 to 35 months of age have received at least four PCV13 doses [112]. The rate varies according to race/ethnicity, with the lowest rates among Asian and black children (Table 13) [112].
In its Healthy People 2030 initiative, the U.S. Department of Health and Human Services has set objectives for improving vaccination rates among adults and children [114]. To reach these targets, healthcare providers must address documented barriers to recommended vaccinations and gain a better understanding of other challenges to vaccination. Unequal access to health care appears to account for a low percent of racial disparities [105]. Rather, lack of awareness of the need for vaccination and misconceptions about vaccines have been reported as the primary barriers in several studies [104,105,106,115,116,117].
Among adults, misconceptions about vaccines range from the belief that healthy people do not need vaccinations to a fear of side effects [104,106,116]. Beliefs about vaccines vary by race/ethnicity, age, education, and gender. For example, in a survey of more than 6,700 older adults, lack of awareness that influenza vaccination was needed was more common among Hispanic (33%) and black individuals (25%) than among white individuals (21%) [105]. In contrast, concern about side effects was more common among white individuals (15%) than among black and Hispanic individuals (10% and 6%, respectively) [105]. The belief that vaccination would not prevent illness was consistent across the racial/ethnic groups. In other studies, lower rates of influenza vaccination among older black adults have been significantly associated with lower rates of positive attitudes about vaccination [105,118]. It is unclear whether the negative attitude represents mistrust of the vaccine itself or of healthcare/healthcare providers in general [105]. The findings of one study showed that, compared with white adults, more black and Hispanic adults believed that they had become sick from a previous influenza vaccination [106]. Language proficiency and level of acculturation have been associated with lower vaccination rates among older Hispanic adults [107,119].
Parental attitudes about vaccines are an important factor in vaccination rates among children. The primary attitude is concern about the safety and efficacy of the vaccine, including fear of adverse events, the discomfort associated with vaccination, distrust of advocates of vaccination, and belief that the vaccine should not be given when a child has a minor illness [117,120,121,122]. Difficulty remembering or confusion about the vaccination schedule for children is also a major challenge [120,122]. Changes in access to health care have been noted as a factor in the low rate of influenza vaccination among teenagers [117].
Healthcare provider-related factors should also be addressed. Slightly more than half of older adults have said that their healthcare provider did not recommend influenza vaccination, and this percentage has been consistent across races/ethnicities [105,106]. The lack of provider recommendation may be a misperception or may be a reality. It has been noted that nearly half of providers do not follow the ACIP recommendations for vaccination [116]. Provider recommendation is essential, as it has been found to be the strongest predictor of whether a person will receive vaccination, even among those who have negative attitudes toward vaccines [104,106,115,116,123]. Providers have said that the lack of an effective reminder system is a factor in low vaccination rates [116,123].
Strategies to improve rates of vaccination and other preventive measures rely on effective patient-clinician communication. Among the most important factors for effective communication across all healthcare settings are knowledge of the language preference of the patient and family; an awareness of the patient's and family's health literacy levels; and an understanding of and respect for the patient's and family's cultural values, beliefs, and practices [124,125,126]. These issues are significant, given the growing percentages of racial/ethnic populations. According to U.S. Census Bureau data from 2013, more than 60.3 million Americans speak a language other than English in the home, with more than 25.1 million of them (8.6% of the population) reporting that they speak English less than "very well" [127]. Clinicians should ask their patients what language is spoken at home and what language they prefer for their medical care information, as some patients prefer their native language even though they have said they can understand and discuss medical information in English [128]. When the healthcare professional and the patient speak different languages, a professional interpreter should be used. Studies have demonstrated that the use of professional interpreters rather than "ad hoc" interpreters (e.g., untrained staff members, family members, friends) facilitates a broader understanding, leads to better outcomes, and is better aligned with patient preferences [129,130,131].
Studies have indicated that as many as 26% of Americans have inadequate health literacy, which means they lack the ability to understand health information and make informed health decisions; an additional 20% have marginal health literacy [132,133,134]. Health literacy varies widely according to race/ethnicity, level of education, and gender. Clinicians are often unaware of the literacy level of their patients and family, but several instruments are available to test the health literacy level [126,135]. These instruments vary in the amount of time needed to administer and the reliability in identifying low literacy. Among the most recent tools is the Newest Vital Sign (NVS), an instrument named to promote the assessment of health literacy as part of the overall routine patient evaluation [136]. The NVS takes fewer than three minutes to administer, has correlated well with more extensive literacy tests, and has performed moderately well at identifying limited literacy [126,135]. Two questions have also been found to perform moderately well in identifying patients with inadequate or marginal literacy: "How confident are you in filling out medical forms by yourself?" and "How often do you have someone help you read health information?" [126]. Clinicians should adapt their discussions and educational resources to the patient's and family's identified health literacy level and degree of language proficiency and should also provide culturally appropriate and translated educational materials when possible.
Cultural competency is essential for addressing healthcare disparities among minority groups [124]. Clinicians should ask the patient about his or her cultural beliefs, especially those related to health, and should be sensitive to those beliefs.
Targeted evidence-based strategies can help clinicians improve vaccination rates (Table 14). Education about the importance of vaccination is the cornerstone of most strategies. Messages should be clear and emphasize the benefits of vaccination and the risks of not receiving vaccination. Acknowledging the risks of vaccines can help enhance patient trust [117]. Clinicians should give their patients a list of online resources that provide balanced information on vaccines (Table 15). Differences in beliefs about vaccines across racial/ethnic groups indicate that targeted messages developed for specific demographic subgroups may be useful [219]. In addition, language-specific educational resources may also help increase vaccination rates by enabling patients to better understand the need for vaccination and its safety.
BARRIERS TO OPTIMAL VACCINATION AND POSSIBLE SOLUTIONS
| Barriers | Solutions |
|---|---|
| Decreased knowledge about pneumonia and its seriousness | Provide education resources (language-specific, as appropriate) that highlight the potential severity of disease and the consequences of not receiving protection through vaccination. |
| Belief that vaccines are unsafe or will cause illness | Refer patient (or parent) to objective information about vaccines. |
| Lack of awareness for the need of vaccination | Take advantage of all visits (well and acute) to remind patients (or parents) about the need for vaccination, to administer vaccination, or to schedule appointment for vaccination. |
| Lack of provider recommendations | Identify high-risk patients and encourage them to receive vaccination. |
| Lack of effective practice systems | Implement effective reminder systems and standing orders. |
RESOURCES ABOUT VACCINATIONS FOR PATIENTS AND PARENTS
|
Education and provider recommendation are particularly important for people at high-risk for severe health impacts of pneumonia, as the lowest vaccination rates are reported for these patients [102,103]. One survey showed that provider recommendations for pneumococcal and influenza vaccination were low for this population; the rate of recommendation was lowest for people with a weakened immune system and those receiving radiation therapy or chemotherapy (Table 16) [116]. Clinicians should identify high-risk patients in their practice and take special steps to ensure that these patients receive appropriate vaccinations.
HEALTHCARE PROVIDER RECOMMENDATIONS FOR INFLUENZA AND PNEUMOCOCCAL VACCINATIONS BY PATIENT TYPE
| Patient Type | Influenza Vaccine | Pneumococcal Vaccine | ||||
|---|---|---|---|---|---|---|
| Physicians | PA/NP/RNs | Physicians | PA/NP/RNs | |||
| All adults | 39% | 59%a | — | — | ||
| Aged ≥50 years | 28%a | 15% | 4% | 18%a | ||
| Aged ≥65 years | 37% | 28% | 65% | 55% | ||
| Chronic lung disease | 45% | 40% | 68% | 55% | ||
| Diabetes mellitus | 31% | 25% | 44%a | 26% | ||
| Heart disease | 20% | 11% | 29%a | 12% | ||
| Chronic liver disease | 22% | 16% | 27% | 20% | ||
| Chronic kidney disease | 22% | 12% | 25% | 17% | ||
| Weak immune system | 17% | 20% | 24% | 29% | ||
| Radiation/chemotherapy | 14% | 9% | 17% | 10% | ||
| Asplenia | — | — | 27%a | 8% | ||
| Complications or risk from other illness | 25% | 17% | 28% | 23% | ||
| Smoker | — | — | 13% | 11% | ||
| Close contact with someone at high risk | 24% | 22% | 11% | 10% | ||
| ||||||
Missed opportunities represent another practice-related area in which clinicians can improve vaccination rates. Although many clinicians check immunization status during well visits, most do not check the status during acute visits, nor do they take advantage of the visit to administer the vaccination [105,115]. Healthcare providers can close the gap on missed opportunities for vaccination by taking advantage of every office visit to administer vaccinations, reminding their patients about the need for vaccination, or scheduling a future appointment for vaccination [105,115,117]. Educational fliers and pamphlets in the waiting room and examination rooms can engage patients and parents and help prompt discussions about vaccination [116].
Patient reminder and recall systems in primary care settings have been effective in improving vaccination rates. A meta-analysis found that rates among both children and adults increased up to 20% with several types of reminders, including postcards, letters, and phone calls [137]. The most effective reminder system was phone calls, but it was also the most expensive. Given that about 25% of primary care physicians currently use reminder systems, increasing the number of physicians who use such systems can in turn increase vaccination rates [123]. Standing orders for vaccinations have been shown to substantially increase vaccination rates, yet are used by only 20% to 33% of physicians [123,138]. Again, adopting this system results in improved vaccination rates.
Many people have turned to facilities outside of their primary healthcare provider to receive vaccinations. Health fairs, pharmacies, grocery stores, senior centers, and workplaces have become more common settings for vaccination because of their convenience and lower cost [123,138]. Clinicians can also help increase vaccination rates by participating in community events that provide vaccinations and by promoting these settings as alternative options.
Programs to provide vaccinations to high-risk patients in the emergency room have been successful at increasing vaccination rates [139,140]. In a three-week intervention program at one inner city emergency department, participants were provided appropriate immunizations when they were at high risk for specific diseases [139]. During the study period, rates of influenza and pneumococcal vaccinations increased from 16% to 83% and from 18% to 84%, respectively. Such programs can help healthcare systems adhere to guideline recommendations for vaccinating hospitalized patients.
Pneumonia associated with healthcare facilities encompasses the broad category of cases that arise in persons who reside in, or have had significant recent exposure to, facilities such as hospitals, nursing homes, dialysis clinics, and transfusion centers. Despite advances in clinical care and prevention, this category of pneumonia remains a serious cause of morbidity and mortality and a challenging, costly public health issue. The IDSA and the ATS subdivide and defines this category of pneumonia as follows:
HAP is hospital-acquired pneumonia that occurs 48 hours or more after admission and did not appear to be incubating at the time of admission.
VAP is a separate type of HAP that develops more than 48 hours after endotracheal intubation.
HCAP is defined as pneumonia occurring in a nonhospitalized patient with extensive healthcare contact, evidenced by one or more of the following:
Intravenous therapy/chemotherapy or wound care within the prior 30 days
Residence in a nursing home or other long-term care facility
Discharge from an acute care hospital or chronic care facility within the prior 90 days
Attendance at a hospital or hemodialysis clinic within the prior 30 days
HAP and VAP have been studied most often, and the bulk of data on causative pathogens comes from studies of VAP. All three categories of pneumonia carry an increased risk for drug-resistant infection, though the risk of multidrug-resistant infection has been more consistently applicable to HAP and VAP [28]. Within the category of HCAP, nursing home-acquired pneumonia is the type with the most published data and will be discussed in this course. The ATS and the IDSA have jointly published evidence-based recommendations, updated in 2016, for the diagnosis and treatment of HAP and VAP [28].
Approximately 3 to 10 cases of HAP occur per 1,000 hospital admissions [26]. Pneumonia as a complication of hospitalization increases length of stay (by more than one week), increases mortality risk, and adds an additional cost of care that can reach $40,000 per case [26].
The rate of VAP is higher than that for HAP, with a reported rate of 1 to 4 cases per 1,000 ventilator-days, and rates as high as 10 cases per 1,000 in some neonatal and surgical populations [12,28,141]. An estimated 10% of patients requiring mechanical ventilation will develop VAP, and the mortality rate directly attributable to VAP is estimated at 13% [28]. Excess cost of care resulting from prolongation of hospital stay is estimated to range from $30,000 to $40,000 per patient [28]. Pediatric VAP has not been as well studied as in adults. It occurs most commonly in children 2 to 12 months of age [142].
Pneumonia develops in approximately 2.3% of nursing home residents [1]. The mortality rate attributed to nursing home-acquired pneumonia is 10% to 30% [143].
Illness and injury requiring admission to a healthcare facility often confers an increased risk for infection. Multiple factors account for this, including weakness and debility, use of indwelling catheters, compromised immune function, and poor nutrition [26,144]. To these may be added sedating medication intended to promote sleep or permit invasive procedures; this in turn increases the risk for aspiration of nasopharyngeal secretions colonized with nosocomial bacterial pathogens.
The nasopharynx tends to become colonized by enteric gram-negative bacilli within a few days after admission to a hospital. Risk factors for colonization by multidrug-resistant pathogens include exposure to critical care units, prolonged hospital stay, prior antibiotic therapy, history of cigarette smoking, major surgery, multiple organ-system failure, and foreign bodies such as nasogastric and endotracheal tubes [26,144].
In a systematic review, the American College of Physicians found several patient-related and surgery-related factors that increased the risk of postoperative pulmonary complications. The most common patient-related factors were the presence of COPD and an age older than 60 years [145]. Other significant factors were an American Society of Anesthesiologists (ASA) class of 2 (defined as a patient with mild systemic disease) or higher, functional dependence, and congestive heart failure. Cigarette use was associated with a modest increase in risk, and obesity and mild or moderate asthma were not found to increase risk [145]. Use of a PPI or histamine2 receptor antagonist is also thought to be a risk factor [45]. Surgery-related factors included prolonged duration of surgery (i.e., more than three to four hours), emergency surgery, and surgical site, with abdominal surgery, thoracic surgery, neurosurgery, head and neck surgery, vascular surgery, and aortic aneurysm repair being associated with the greatest risks [145].
The risk for VAP appears to be greatest during the first week after intubation. In one study, the risk was estimated to be 3% per day during the five-day period following intubation, decreasing to 2% per day for days 5 through 10, and to 1% per day for longer durations [147]. In a population of children who had cardiothoracic surgery, pneumonia risk correlated with mechanical ventilation for longer than three days [144]. Nearly half of all cases of VAP develop within the first four days of mechanical ventilation [148].
Other identified risk factors among adults include prolonged placement of the patient's head in the supine position; use of a nasogastric tube, paralytic agents, or PPI or histamine2 receptor antagonist; advanced age; chronic lung disease; and head trauma [45,149]. Among children, VAP has been significantly associated with subglottic/tracheal stenosis, trauma, and tracheostomy [150]. In one study, VAP was most frequently associated with ICU admission diagnoses of postoperative care, neurologic conditions, sepsis, and cardiac complications [151].
The risk factors reported to be associated with nursing home-acquired pneumonia include profound disability, immobility, urinary incontinence, deteriorating health status, difficulty swallowing, and inability to take oral medications [42]. Older age, male gender, and antipsychotic and anticholinergic medications have also been reported to increase risk [23,42].
Gram-negative enteric bacilli and Pseudomonas spp. rarely colonize the upper respiratory tract of healthy individuals, but often do so in persons with an underlying disease, such as alcohol use disorder, and in those who are hospitalized or reside in nursing homes. Therefore, a history of recent hospitalization or nursing home residency should heighten suspicion for a gram-negative pathogen when such a patient presents with clinical signs of infection.
Most cases of pneumonia that develop in a healthcare facility are caused by aspiration of oropharyngeal or gastric secretions colonized with hospital bacterial flora [26,28]. Consequently, the prevalent causation as well as the antibiotic sensitivity pattern of resident pathogens will vary from region to region in relation to the type of facility and burden of antimicrobial usage. The selection of initial antibiotic therapy in these cases is based on the patient's risk factors for infection with a multidrug-resistant organism, such as MRSA, P. aeruginosa, K. pneumoniae, or Acinetobacter. The ATS/IDSA lists the following risk factors for multidrug-resistant pathogens in patients presenting with HAP or VAP [233,28]:
Prior intravenous antibiotic use within 90 days
Septic shock at time of VAP
ARDS prior to onset of VAP
High frequency of antibiotic resistance in the community of residence or the hospital unit of residence
Five or more days of hospitalization prior to onset of pneumonia
Home infusion therapy
Chronic dialysis within 30 days
Family member with multidrug-resistant infection
Immunosuppression
Viral and fungal pathogens are rare causes of HAP, VAP, and nursing home-acquired pneumonia in immunocompetent adults. Outbreaks of viral pneumonia may occur during influenza season, and influenza, parainfluenza, adenovirus, and RSV are involved in about 70% of those cases [28]. Candida spp. and Aspergillus fumigatus may cause pneumonia in patients who have had organ transplantation or who have a compromised immune system and neutropenia.
Among adults with no previous antibiotic exposure, the most common bacterial causes of HAP are S. pneumoniae, H. influenzae, Escherichia coli, K. pneumoniae, and S. aureus [26,28,35,148]. Gram-negative bacilli resistant to first-generation cephalosporins also frequently develop in late-onset HAP. For up to 40% of adults with previous antibiotic exposure, late-onset HAP is caused by potentially multidrug-resistant pathogens, including Pseudomonas aeruginosa, Acinetobacter baumannii, and MRSA [26]. In a study of more than 3,600 patients admitted to an ICU, Pseudomonas spp. was the cause of pneumonia in 25% of patients; MRSA in 18%; and Acinetobacter spp. in 6% [35]. Other studies have shown that S. aureus is common among patients who are in a coma or have diabetes or renal failure. P. aeruginosa is common among patients who have had a prolonged stay in the ICU, have received prior antibiotics or corticosteroids, or who have structural lung disease. Legionella is usually found in patients who have compromised immune systems [35].
The causes of HAP in children have not been well studied. However, outbreaks of pneumonia caused by RSV have been common in pediatric wards [28].
The most common pathogens associated with VAP in adults are S. aureus and P. aeruginosa, followed by Enterobacter spp., A. baumannii, and K. pneumoniae [26,148,152,153]. These bacteria are among those that have become resistant to antibiotics, and the frequency of infection with MRSA is increasing. Almost half of all cases are caused by infection with more than one pathogen [148]. Although bacteria are the primary causative agents, viruses and saprophytic fungi have also been implicated as well [154].
As with HAP, few data are available on the etiology of VAP in children. In one report, P. aeruginosa was the most common cause, accounting for 22% of cases [142].
The bacterial pathogens that cause pneumonia in residents of nursing homes (and other long-term care facilities) differ according to the severity of disease. S. pneumoniae and H. influenzae are the most common causes of mild-to-moderate pneumonia in long-term care facilities [155]. In cases requiring hospitalization, C. pneumoniae, S. aureus, and influenza virus are frequently observed as well. Patients with severe illness commonly are infected with methicillin-sensitive S. aureus or MRSA, gram-negative enteric pathogens, or P. aeruginosa [23,155].
The difficulty in recognizing HAP, VAP, or nursing home-acquired pneumonia has been well documented [28,147,156]. The clinical signs often resemble other, noninfectious conditions, and the specificity of clinical criteria is low [148]. According to the CDC definition, the diagnosis in adults is made on the basis of clinical signs and results of laboratory testing or imaging and must meet one of two criteria [157].
Criterion 1 is rales or dullness to percussion on physical examination of the chest and at least one of the following:
New onset of purulent sputum or change in character of sputum
Organisms cultured from blood
Isolation of an etiologic agent from a specimen obtained by transtracheal aspirate, bronchial brushing, or biopsy
Criterion 2 is chest radiograph that shows new or progressive infiltrate, consolidation, cavitation, or pleural effusion and at least one of the following:
New onset of purulent sputum or change in character of sputum
Organisms cultured from blood
Isolation of an etiologic agent from a specimen obtained by transtracheal aspirate, bronchial brushing, or biopsy
Isolation of virus from or detection of viral antigen in respiratory secretions
Diagnostic single antibody titer immune globulin M or fourfold increase in paired sera immune globulin G for pathogen
Histopathologic evidence of pneumonia
A set of clinical diagnostic criteria for HCAP includes the presence of a new and persistent (more than 48 hours) infiltrate in addition to one of the following [148]:
Radiographic evidence of cavitation or necrosis
Histopathologic evidence of pneumonia
Positive pleural or blood culture for the same micro-organism as that found in respiratory secretions
Plus two of the following signs:
Core temperature >38.3°C (100.94°F)
WBC count >10,000 cells/mm3
Purulent tracheal secretions
There are no compelling data to recommend a specific approach to diagnosing HAP and VAP. For patients who are not receiving mechanical ventilation, collection of a sputum specimen should be attempted before antibiotic therapy is begun [35,158]. Specimens for culture can be obtained by bronchoscopy with a protected specimen brush to limit contamination or by bronchoalveolar lavage. The latter method has been found to lead to higher rates of treatment than diagnosis based on the CDC definition, and one study showed that preferential sampling of the right lung (rather than the left) improved the diagnostic accuracy of bronchoalveolar lavage [35,159,160]. However, the invasive procedure has disadvantages, including high cost, need for technical expertise, and the potential for false-negative results [35,159].
The ATS/IDSA guideline recommends collecting specimens from the lower respiratory tract for culture, preferably by noninvasive techniques, and reliance on semiquantitative culture technique [28]. Noninvasive methods to obtain respiratory samples in patients with HAP (non-VAP) include spontaneous expectoration, sputum induction, nasotracheal suctioning (in a patient unable to produce a sample), and endotracheal aspiration in a patient with HAP who subsequently requires mechanical ventilation [28]. A 2012 meta-analysis found no evidence that the use of quantitative cultures of respiratory secretions resulted in decreased mortality, reduced time in ICU and on mechanical ventilation, or higher rates of antibiotic change compared with qualitative cultures in patients with VAP [161]. In addition, there was no difference in mortality whether invasive or noninvasive methods were used to obtain specimens.
The treatment of HAP and VAP is complicated by two divergent needs: the need for empiric therapy with a broad-spectrum antibiotic, to aid in reducing mortality rates, and the need to avoid the indiscriminate use of antibiotics, to avoid the development of resistance. To address this complex issue, the strategy of de-escalation therapy was developed. With this treatment approach, a broad-spectrum antibiotic targeted to likely pathogens is administered, and the antibiotic regimen is modified after the results of cultures are known [154,162]. This strategy has reduced the mortality rate while achieving an overall objective of a more judicious use of antibiotics [154,163]. In one study, de-escalation led to a significantly lower mortality rate compared with either escalation therapy or therapy that was neither escalated nor de-escalated (17% vs. 43% and 24%, respectively) [151].
The empiric treatment of nosocomial pneumonia in general, requires knowledge of the infection history (hospital flora) of the healthcare facility and of individual patient units [35,148,164]. The selection of an empiric antibiotic regimen for HAP and VAP should be guided by local antibiotic-resistance data. The ATS/IDSA recommend that all hospitals regularly generate and disseminate a local antibiogram, ideally one that is specific to their intensive care population(s), if possible [28].
In managing a case of HAP and VAP, the clinician should review in detail the guidance provided by the ATS/IDSA, and consider consultation with appropriate subspecialty colleagues [28]. Recommendations governing selected issues of initial management emphasize the following principles [28]:
Obtain sputum samples from the lower respiratory tract for culture before beginning antibiotic therapy. Do not delay initiation of therapy for critically ill patients in order to obtain specimens.
Begin treatment promptly, selecting an empiric antibiotic regimen that covers S. aureus, P. aeruginosa, and other gram-negative bacilli.
In selecting coverage for S. aureus, choose an agent active against MRSA (vancomycin or linezolid) for patients with risk factor(s) for antimicrobial resistance, treatment in hospital or units where >10% of isolates are methicillin-resistant, and patients in settings where the prevalence of MRSA is unknown.
In selecting coverage for P. aeruginosa, one antibiotic active against this pathogen is satisfactory if the patient has no risk factors for antimicrobial resistance and <10% of gram-negative isolates from the patient's unit are resistant to the agent chosen; otherwise, prescribe two antipseudomonal antibiotics from different classes.
Consider de-escalation of antibiotics after the results of cultures and sensitivities are known and the clinical response is satisfactory.
After an optimal antibiotic regimen is confirmed, a seven-day course of therapy is recommended, provided the rate of improvement of clinical, radiographic, and laboratory parameter is satisfactory.
For patients with HAP/VAP, it is suggested to use serum procalcitonin levels plus clinical criteria to guide discontinuation of antibiotic therapy, rather than clinical criteria alone.
Selection of specific antimicrobial therapy is influenced by the timing of onset of clinical signs, as well as the presence or absence of risk factors for infection with multidrug-resistant organisms. For early-onset pneumonia and/or patients with no such risk factors, limited-spectrum antibiotic therapy is recommended (Table 17) [28]. For late-onset pneumonia and/or patients at increased risk for multidrug-resistant organisms, a broad-spectrum antibiotic regimen is recommended.
RECOMMENDED ANTIBIOTIC THERAPY FOR HEALTH FACILITY-ASSOCIATED PNEUMONIA ACCORDING TO SITE OF CARE
| Site of Care | Recommended Regimen |
|---|---|
| Nursing home | Antipneumococcal fluoroquinolone or either a high-dose ß-lactam/ß-lactamase inhibitor or a second- or third-generation cephalosporin in combination with azithromycin |
| Hospital | Antipseudomonal cephalosporin, antipseudomonal carbapenem, or extended-spectrum ß-lactam/ß-lactamase inhibitor and antipseudomonal fluoroquinolone or aminoglycoside and anti-MRSA agent (vancomycin or linezolid) |
| Intensive care unit | Empiric MRSA and double coverage of Pseudomonas pneumonia |
VAP is often caused by MRSA and gram-negative bacilli such as Acinetobacter spp. and Pseudomonas. Vancomycin has been considered the first choice for treatment of MRSA infections [154]. However, the ATS/IDSA guidelines note that linezolid may have advantages over vancomycin for pneumonia caused by MRSA [28]. Linezolid has been compared with vancomycin for the treatment of pneumonia caused by MRSA in many studies, and linezolid has been found to improve survival and to be more cost-effective [147,165,166,167,168]. In a 2008 study, the rate of early microbiologic cure was not significantly higher for linezolid than for vancomycin, although there were trends favoring linezolid in several secondary clinical outcomes, such as clinical cure; duration of ventilation, hospitalization, and stay in ICU; survival time not on a ventilator; and overall survival [169]. The findings led the authors to suggest that the benefit of linezolid may be related to factors other than bacterial clearance.
For cases of VAP caused by gram-negative bacilli that are susceptible only to aminoglycosides or polymyxins, the ATS/IDSA suggests both inhaled and systemic antibiotics, rather than systemic antibiotics alone [28]. It is also reasonable to consider adjunctive inhaled antibiotic treatment as a last resort for patients who are not responding to intravenous antibiotics alone, whether or not the infecting organism is multi-drug resistant.
According to a meta-analysis, a short fixed-course (7 or 8 days) of antibiotic therapy may be more appropriate than a prolonged course (10 to 15 days) for patients with VAP not caused by nonfermenting gram-negative bacilli [170]. The short course reduced recurrence of pneumonia caused by multiresistant organisms without adversely affecting other outcomes. Among patients with nonfermenting gram-negative bacilli, recurrence was greater after the short course.
The 2019 ATS/IDSA guideline for CAP provides a rationale for choice of antibiotic therapy without specifying distinct protocols for nursing home-acquired pneumonia. The ATS/IDSA recommended abandoning the HCAP categorization for purposes of management decision. Instead, emphasis is placed on local epidemiology and validated risk factors to determine need for empiric MRSA or gram-negative bacillary coverage, followed by de-escalation of treatment if cultures are negative [235]. As indicated, the consistently strong individual risk factors for respiratory infection with MRSA, P. aeruginosa, or other gram-negative bacilli are prior isolation of these organisms, and/or recent hospitalization and exposure to parenteral antibiotics.
The lack of adherence to guideline-directed treatment of pneumonia cases associated with healthcare facilities is evidenced by wide variations in practice. For example, one study showed that more than 100 different antibiotic regimens had been prescribed as initial treatment and that de-escalation therapy was used for only 22% of patients [151]. Adherence rates for pneumonia associated with healthcare facilities have been reported to be lower than rates of adherence to guidelines for treatment of CAP. In one survey, guideline-recommended antibiotics were used 78% of the time for CAP, compared with 9% for HCAP [18]. This lack of adherence was not due to unfamiliarity or disagreement with the guidelines; 71% of the survey respondents said they were aware of the guidelines, and 79% said they agreed with and practiced according to them. In contrast, another survey showed that fewer than half of physicians were familiar with the ATS/IDSA guideline for treatment of nursing home-associated pneumonia [23]. It is reasonable to expect that strategies used to enhance adherence to guidelines in the setting of CAP would also be beneficial in the setting of pneumonia associated with healthcare facilities. Thus, feedback on performance, reminder systems, standardized order sets, and education emphasizing outcomes and cost-effectiveness would be valuable.
The CDC has published a guideline for the prevention of HAP and VAP, with a focus on strategies to decrease or eliminate modifiable risk factors for pneumonia associated with healthcare facilities [93]. These strategies are related to preoperative and postoperative care and measures to reduce the risk of transmission of etiologic pathogens. In addition, steps to prevent the spread of influenza virus are essential, especially during influenza season.
The prevention of postoperative pneumonia has long been a part of initiatives to decrease complications among patients undergoing surgery. The Respiratory Risk Index was developed to classify patients as being at low, medium, or high risk for postoperative respiratory failure [26]. The factors in the index include the complexity of the surgery, the ASA status, and comorbidities.
Smoking triples the risk for pulmonary complications after surgery, and smoking cessation for at least eight weeks before surgery, when possible, is recommended for current smokers [26]. The risk for complications in patients with respiratory disease or congestive heart failure can be ameliorated by optimum treatment before surgery (e.g., treatment with steroids for patients with COPD or asthma) [26].
Effective pain management after surgery also helps to decrease the risk of pulmonary complications. For postoperative patients who are not mechanically intubated, the ability to cough and clear secretions is important for preventing pulmonary complications [26]. The use of incentive spirometry and deep breathing exercises are recommended, especially for people at high risk for pulmonary complications, as are frequent coughing and early movement (in bed and/or walking) [26,93,145]. Fair evidence supports the selective (rather than routine) use of a nasogastric tube after abdominal surgery [145].
Two guidelines were developed to focus specifically on the prevention of VAP; one was jointly developed by the Society for Healthcare Epidemiology of America (SHEA) and IDSA, and the other was jointly developed by the Canadian Critical Care Trials Group and the Canadian Critical Care Society [149,171]. In addition, the CDC guideline addresses the prevention of HAP and VAP [93]. All of these agencies suggest a multicomponent strategy for prevention of pneumonia. Compliance with guidelines, however, has been slow; nursing surveys demonstrate rates of adherence to specific preventive measures ranging from 15% to 50% [12,172]. Education is beneficial, and training sessions are a proven means to enhance knowledge and practice among healthcare professionals caring for intubated patients [173].
The Institute for Healthcare Improvement (IHI) found that implementation of its ventilator bundle, a collection of five prevention strategies drawn from these guidelines, led to a 45% reduction in the incidence of VAP [174]. The bundle includes the following interventions [174]:
Assessment of readiness to extubate and daily interruptions of sedation
Elevation of the head of the bed
Daily oral care with chlorhexidine
Prophylaxis of peptic ulcer disease
Prophylaxis of deep venous thrombosis
The IHI how-to guide on preventing VAP provides several practical recommendations, and posting compliance with the ventilator bundle in a prominent place in the ICU can encourage and motivate staff (Table 18) [174].
PRACTICAL STEPS IN FOLLOWING GUIDELINES TO PREVENT VENTILATOR-ASSOCIATED PNEUMONIA
| Assessment of Readiness to Extubate and Sedative Interruptions | |||||
| |||||
| Elevation of the Head of the Bed | |||||
| |||||
| Oral Care with Chlorhexidine | |||||
| |||||
| Prophylaxis of Peptic Ulcer Disease | |||||
| |||||
| Prophylaxis of Deep Venous Thrombosis | |||||
|
Assessment of Readiness to Extubate
Because of the increasing risk of infection as the duration of ventilation increases, the primary goal is to extubate patients as early as possible. Thus, assessment of the readiness for extubation and weaning protocols are key aspects in the preventive approach [28,35]. Daily interruption of sedation until the patient is awake has been shown to significantly decrease the number of days on mechanical ventilation, from 7.3 days to 4.9 days in one study [175]. There are risks to this approach, including the potential for increased pain, anxiety, and desaturation [174]. However, sedation interruption has been further demonstrated to reduce the complications of prolonged mechanical ventilation [176]. The SHEA/IDSA guideline recommends daily assessment of the readiness to wean and the use of weaning protocols [171]. For children, daily assessment of readiness to extubate should be carried out, but sedation interruption is not recommended because of the high risk of unplanned extubation [177].
Elevation of the Head of the Bed
Reducing the risk of aspiration and contamination with gastric secretions also helps to prevent the development of pneumonia. Positioning the head of the bed at an angle of 30 to 45 degrees reduces the risk of aspiration significantly [149,178,179]. In one randomized, controlled trial, there were 18% fewer cases of VAP among intubated patients in the group assigned to the recumbent position (45 degrees) compared with the group assigned to the supine position [179]. In another study, elevation of the head of the bed to 30 degrees was the most effective measure among a group of preventive interventions, resulting in a 52% variance in the rate of VAP [180]. Both the ATS/IDSA and SHEA/IDSA guidelines recommend maintaining the head of the bed at a 30- to 45-degree angle [28,171]. An angle of 30 to 45 degrees is also recommended for infants and children, but a lower angle (15 to 30 degrees) should be used for neonates [177].
Daily Oral Care with Chlorhexidine
Oral care interventions have been suggested by some, in part because of an association between a high level of dental plaque and a high rate of colonization with aerobic pathogens, including S. aureus, gram-negative bacilli, and P. aeruginosa[181]. Research has shown that oral decontamination with chlorhexidine leads to a significant reduction in the colonization of pathogens in the oropharynx. In most studies, the intervention has not had a significant effect on the rate of VAP or associated mortality, but more recent studies have shown a significant decrease in the rate of pneumonia [180,182,183,184,185,186]. Brushing the teeth with chlorhexidine does not seem to add benefit [183]. Regular oral care with an antiseptic solution or chlorhexidine is recommended in the ATS/IDSA and SHEA/IDSA guidelines [28,171].
Prophylaxis of Peptic Ulcer Disease
Prophylaxis of peptic ulcer disease has evolved with some conflicting views. Antacids, histamine2 receptor antagonists, and sucralfate have been traditionally given to patients receiving mechanical ventilation to prevent the formation of stress ulcers. However, reducing the amount of gastric acid can increase the risk of colonization of gram-negative bacilli in the stomach. As a result, the WHO recommends avoiding the use of these agents [187]. The CDC notes that there was insufficient evidence on the use of peptic ulcer prophylaxis and includes no recommendations in this regard in its guideline [93]. The ATS/IDSA guideline states that the risks and benefits of prophylaxis should be weighed carefully [28]. The most recent guideline, developed by SHEA/IDSA, notes that histamine2 receptor antagonists and PPIs should be avoided in patients who are not at high risk for developing a stress ulcer or stress gastritis [171]. However, peptic ulcer prophylaxis is recommended for children, as appropriate for age and health status [177].
Prophylaxis of Deep Venous Thrombosis
There is no clear relation between prophylaxis of deep vein thrombosis and VAP pneumonia, but the American College of Chest Physicians reported a decrease in the rate of VAP when such prophylaxis was implemented as part of a package of interventions and included this measure in its clinical practice guideline [188]. This recommendation also applies to children, as appropriate for age and health status [177].
Other Measures
In addition to the interventions in the ventilator bundle, other measures have been recommended to help prevent VAP. One such measure is selective decontamination of the digestive tract, which involves the use of either topical antiseptic, oral antibiotics, or a brief course of systemic antibiotics [26]. A meta-analysis of 28 studies showed that selective decontamination of the digestive or respiratory tract with use of topical antiseptic or antimicrobial agents helped reduce the frequency of VAP in the ICU [146]. The estimate of efficacy in prevention was 27% for antiseptics and 36% for antibiotics. Neither had an effect on mortality. This intervention is recommended in the SHEA/IDSA guideline [171].
Other preventive measures are targeted primarily to the care and use of ventilator equipment and practices in direct patient care. Meticulous attention to aseptic care of the equipment is necessary, and all reusable components, such as nebulizers, should be disinfected or sterilized. Tubing circuits should be replaced after 48 hours or earlier if there are signs of malfunction or contamination [93]. Changes in the design of the endotracheal tube have also been evaluated; for example, a tube with a suction port above the cuff allows for continuous aspiration of subglottic secretions. Use of this specially designed endotracheal tube has led to significantly lower rates of VAP as well as shorter durations of ventilation and shorter stays in the ICU [189,190]. Among patients who had major cardiac surgery, the greatest benefit was found for patients who received ventilation for more than 48 hours [190]. Although the cost of the tube is higher than traditional tubes, the overall cost savings in preventing VAP more than compensates [189]. In one meta-analysis, subglottic secretion drainage was significantly associated with a decreased incidence of VAP, shorter time on mechanical ventilation, and longer time to the development of pneumonia [191]. The CDC, the ATS/IDSA, and the SHEA/IDSA guidelines recommend subglottic secretion drainage with this tube when possible [28,93,171].
The use of noninvasive ventilation is another measure that has reduced the incidence of VAP [93,192,193,194]. In one study, the incidence decreased from 20% to 8% when noninvasive techniques were used routinely for critically ill patients with acute exacerbation of COPD or severe cardiogenic pulmonary edema [192]. Again, the CDC, the ATS/IDSA, and the SHEA/IDSA guidelines recommend the use of noninvasive ventilation when possible [28,93,171].
Quality Improvement Initiatives and Enhanced Infection Control Strategies
Quality improvement and infection control initiatives and strategies have led to a substantial decrease in the rates of VAP since the early 2000s [195]. The use of physician-led multidisciplinary rounds with team decision-making, checklists, and a focus on the ventilator bundle has led to significant reductions in the risk for pneumonia [196,197,198]. Strong downward trends were also found for the average length of stay in the ICU and the financial costs per patient [196].
As with HAP, strategies to decrease or eliminate modifiable risk factors for nursing home-acquired pneumonia should be implemented. A multidisciplinary panel made three recommendations for prevention of pneumonia among nursing home residents [199]:
Pneumococcal vaccination of patients at admission, if indicated
Annual influenza vaccination for residents
Annual influenza vaccination for nursing facility staff
The vaccination status of healthcare workers has been found to have a direct effect on transmission of influenza virus to patients. Outbreaks of influenza in healthcare settings have been associated with low rates of vaccination among healthcare workers, and lower rates of nosocomial influenza have been related to higher vaccination rates among healthcare workers [200,201]. Because of these findings, the ACIP recommends annual influenza vaccination for all healthcare workers, and the IDSA/ATS guideline endorses this recommendation [47]. The ACIP notes that the TIV is preferred over LAIV for workers who are in close contact with severely immunosuppressed people requiring protective isolation [112]. In addition, the Joint Commission began including vaccination programs in its accreditation standards in 2007 [123].
Despite these recommendations, only 29% to 69% of healthcare workers receive the influenza vaccination each year [202,203,204]. Healthcare workers have given many reasons for not being vaccinated, and the reasons vary among professions. Across all categories, shortage of the vaccine is the primary reason for not being vaccinated; other reasons include concern about side effects, inconvenience, and forgetfulness [204].
Efforts to increase the vaccination rate among healthcare workers are ongoing. A CDC guideline includes four level I recommendations to help increase rates of vaccination [205]:
Offer influenza vaccine annually to all eligible healthcare workers.
Provide influenza vaccination to healthcare workers at the work site and at no cost as one component of employee health programs. Use strategies that have been demonstrated to increase influenza vaccine acceptance, including vaccination clinics, mobile carts, vaccination access during all work shifts, and modeling and support by institutional leaders.
Monitor influenza vaccination coverage and declination of healthcare workers at regular intervals during influenza season and provide feedback of ward-, unit-, and specialty-specific rates to staff and administration.
Educate healthcare workers about the benefits of influenza vaccination and the potential health consequences of influenza illness for themselves and their patients, the epidemiology and modes of transmission, diagnosis, treatment, and non-vaccine infection control strategies, in accordance with their level of responsibility in preventing healthcare-associated influenza.
Hand hygiene is the most important preventive measure in hospitals, and the Joint Commission mandates that hospitals and other healthcare facilities comply with the Level I recommendations in the CDC guideline for hand hygiene [206]. The CDC guideline states the specific indications for washing hands, the recommended hand hygiene techniques, and recommendations about fingernails and the use of gloves [207]. The guideline also provides recommendations for surgical hand antisepsis, selection of hand-hygiene agents, skin care, educational and motivational programs for healthcare workers, and administrative measures.
Despite the simplicity of the intervention, its substantial impact, and wide dissemination of the guideline, compliance with recommended hand hygiene has ranged from 16% to 81%, with an average of 30% to 50% [207,208,209,210,211,212]. Among the reasons given for the lack of compliance are inconvenience, understaffing, and damage to skin [207,210,213]. The development of effective alcohol-based handrub solutions addresses these concerns, and studies have demonstrated that these solutions have increased compliance [211,214,215]. The CDC guideline recommends the use of such solutions on the basis of several advantages, including [207]:
Better efficacy against both gram-negative and gram-positive bacteria, mycobacteria, fungi, and viruses than either soap and water or antimicrobial soaps (such as chlorhexidine)
More rapid disinfection than other hand-hygiene techniques
Less damaging to skin
Time savings (18 minutes compared with 56 minutes per eight-hour shift)
The guideline suggests that healthcare facilities promote compliance by making the handrub solution available in dispensers in convenient locations (such as the entrance to patients' room or at the bedside) and provide individual pocket-sized containers [207]. The handrub solution may be used in all clinical situations except for when hands are visibly dirty or are contaminated with blood or body fluids. In such instances, soap (either antimicrobial or nonantimicrobial) and water must be used.
However, there are many other reasons for lack of adherence to appropriate hand hygiene, including denial about risks, forgetfulness, and belief that gloves provide sufficient protection [207,210,213]. These reasons demand education for healthcare professionals to emphasize the importance of hand hygiene. Also necessary is research to determine which interventions are most likely to improve hand-hygiene practices, as no studies have demonstrated the superiority of any intervention [216]. Single interventions are unlikely to be effective.
A man, 73 years of age, with a history of coronary disease, COPD, benign prostatic hyperplasia, and type 2 diabetes is hospitalized on transfer from an assisted-living facility because of weakness, loss of appetite, and low-grade fever. He had been admitted elsewhere for similar symptoms six months earlier and was diagnosed with urinary tract infection and treated with an unknown antibiotic. On evaluation, the patient's temperature is 37.6°C (99.8°F) and his other vital signs are stable; his exam is unremarkable. The WBC is normal, and the urinalysis shows pyuria. The admission chest x-ray shows hyperlucent lung fields and flattened diaphragms indicative of emphysema, but no infiltrate. Empiric treatment with a first-generation cephalosporin is begun for presumed urinary tract infection. The patient has no further fever, and his appetite and strength improve over the next 48 hours. He does have periods of mild agitation and insomnia, which are treated with a benzodiazepine at bedtime.
On the fourth day, as plans for discharge were in place, the patient appears worse, with a cough and a temperature of 38°C (100.4°F). A repeat chest x-ray shows a small focal opacity in the left upper lobe, thought to represent "aspiration." No change in antibiotics is made, and he is observed. Over the next 36 hours, the patient's condition worsens; he now has a cough productive of purulent sputum, fever (102°F to 103°F), shortness of breath, and tachypnea. A follow-up chest x-ray now shows an extensive opacification/infiltrate in the left upper lobe, with signs suggestive of either central cavitation or consolidation high-lighting emphysematous blebs.
In this elderly, somewhat debilitated man with chronic lung disease, who may be at risk of aspiration, a rapidly progressive, necrotizing (hospital-acquired) pneumonia developed while he was being treated with an oral cephalosporin for urinary tract infection, and receiving a nightly sedative medication for sleep.
What are the etiologic considerations and how should the patient be managed? Within days of admission to a hospital, and especially if treated with antibiotics, many patients develop nasopharyngeal colonization by hospital flora (e.g. gram-negative bacilli and occasionally S. aureus). When pneumonia supervenes, it reflects this colonization; moreover, prior antibiotic therapy tends to select out resistant pathogens. Therefore, the selection of empiric antibiotic treatment for this patient is based on the presumption of hospital-acquired bacterial infection in the lung caused by one or more pathogens resistant to first-generation cephalosporins. Cultures of blood and sputum should be obtained; gram stain of the sputum is often helpful in cases such as this, as it may demonstrate a predominate pathogen and whether it is gram-positive or gram-negative. Empiric antibiotic therapy, following ATS/IDSA recommendations for HAP, should be started promptly. A good choice would be an extended-spectrum ß-lactam/ß-lactamase inhibitor or a carbapenem (e.g., piperacillin/tazobactam or imipenem) combined with a fluoroquinolone and vancomycin, pending culture results.
Gram stain of the patient's sputum shows many polys and gram-negative bacilli; the culture is positive for K. pneumoniae and P. aeruginosa. His management, including empiric antibiotic therapy followed by de-escalation (of vancomycin) after culture data are available, conforms to ATS/IDSA recommendations. The patient is treated for 10 days and recovers following a brief period in the ICU.
This case illustrates that the pathogenesis of adult bacterial HAP is essentially the same as for CAP; namely, nasopharyngeal and upper respiratory colonization by virulent bacteria combined with aspiration of infected secretions during a period of impaired host pulmonary defenses. The difference lies in the burden of vulnerability imposed by hospitalization, including the propensity for colonization by gram-negative bacilli and the likelihood of antimicrobial resistance—so uncommon in healthy individuals outside of healthcare facilities, but so prevalent among patients hospitalized longer than 48 hours.
Pneumonia-related mortality and morbidity have decreased since the late 1990s, but the disease still represents a substantial healthcare concern, especially for high-risk adults and children. Pneumonia is primarily classified according to the setting in which it develops, and the epidemiology, etiology, and risk factors vary according to setting. Diagnosis can be challenging because of differences in presentation and the lack of reliable, cost-effective, and rapidly available diagnostic testing methods. Specialty society guidelines for prevention, diagnosis, and treatment are available for CAP, HAP, and VAP. Guideline-directed treatment has been shown to improve the care of patients while promoting good antibiotic stewardship, minimizing exposure to inappropriate antibiotic treatment and reducing the emergence of antibiotic-resistant pathogens.
For CAP and nursing home-acquired pneumonia, determining the site of care is an important initial decision point. Guidelines from the IDSA/ATS, the PIDS/IDSA, and the ATS outline useful criteria for determining need for hospitalization and ICU care. These objective criteria are important factors in decision-making, but clinical judgment is also necessary for selecting the most appropriate site of care. Initial antibiotic treatment of all types of pneumonia is empirical. The selection is best made in relation to the most likely pathogens in a given clinical setting and to patient variables, such as comorbidities, recent exposure to antibiotics, and immunization status (for children). The timeliness of antibiotic treatment is also important; treatment should begin as soon as possible after diagnosis is made, administering the first dose promptly at the originating site of care.
Guideline-directed therapy of pneumonia has been shown to decrease morbidity and mortality, but adherence varies across settings and specialties and has been suboptimal. Physician practices and healthcare systems can improve adherence by implementing evidence-based strategies, such as standardized order sets, reminders, performance feedback, and easy-to-carry resources.
The incidence of pneumonia and its associated morbidity and mortality can be reduced further by adherence to effective preventive measures. Several guidelines are available for preventing specific types of pneumonia. The primary preventive strategy for CAP is immunization with influenza and pneumococcal vaccines, especially for individuals at high risk. These vaccinations have been shown to decrease the incidence and severity of pneumococcal pneumonia, as well as the risk of long-term morbidity and mortality. However, rates of vaccination vary across age, race/ethnicity, and risk. Two target populations with the lowest immunization rates are high-risk adults in need of pneumococcal vaccination and teenagers in need of influenza vaccination. Rates of vaccination among healthcare professionals are also low. Clinicians and healthcare systems should encourage vaccination and offer convenient access, especially during influenza season.
Lack of awareness about the need for vaccination, misconceptions about vaccines, and low level of knowledge about pneumonia have been reported to be the primary barriers to vaccination, especially among minority populations. Clinicians should promote practice strategies and public health efforts designed to target these barriers and address the populations in greatest need. Several strategies have been shown to increase vaccination rates, and education is the cornerstone. Clinicians should emphasize to patients the need and benefit of immunization, address concerns about the safety of vaccines, and incorporate routine immunization protocols into their practices. Provider recommendation is essential, as it is the strongest predictor of vaccination. System-related strategies such as automatic reminders and standing orders have also been effective.
Guidelines for prevention of HAP focus on measures to reduce pulmonary complications after surgery. Prevention of VAP relies on strategies to reduce the risk of transmission of etiologic agents. Use of a ventilator "bundle" (a set of interventions) has been shown to markedly reduce VAP. Although adherence to guidelines is suboptimal, healthcare facilities are increasingly implementing initiatives to help enhance adherence.
1. Hippocrates. On Regimen in Acute Disease. Available at http://classics.mit.edu/Hippocrates/acutedis.html. Last accessed August 21, 2024.
2. Centers for Disease Control and Prevention. Pneumonia. Available at https://www.cdc.gov/nchs/fastats/pneumonia.htm. Last accessed August 21, 2024.
3. Shorr A, Owens RC. Guidelines and quality for community-acquired pneumonia: measures from the Joint Commission and the Centers for Medicare and Medicaid Services. Am J Health Syst Pharm. 2009;66(12 Suppl 4):S2-S7.
4. Haessler S, Schimmel JS. Managing community-acquired pneumonia during flu season. Cleve Clin J Med. 2012;79(1):67-78.
5. Lee G, Lorch SA, Sheffler-Collins S, Kronman MP, Shah SS. National hospitalization trends for pediatric pneumonia and associated complications. Pediatrics. 2010;126(2):204-213.
6. National Center for Health Statistics. Health, United States, 2014. Hyattsville, MD: National Center for Health Statistics; 2015.
7. Nichol K, Nordin JD, Nelson DB, Mullooly JP, Hak E. Effectiveness of influenza vaccine in the community-dwelling elderly. N Engl J Med. 2007;357(14):1373-1381.
8. Johnstone J, Marrie TJ, Eurich DT, Majumdar SR. Effect of pneumococcal vaccination in hospitalized adults with community-acquired pneumonia. Arch Intern Med. 2007;167(18):1938-1943.
9. Johnstone J, Majumdar SR, Fox JD, Marrie TJ. Viral infection in adults hospitalized with community-acquired pneumonia: prevalence, pathogens, and presentation. Chest. 2008;134(6):1141-1148.
10. Grijalva C, Poehling KA, Nuorti JP, et al. National impact of universal childhood immunization with pneumococcal conjugate vaccine on outpatient medical care visits in the United States. Pediatrics. 2006;118(3):865-873.
11. O'Leary ST, Crane LA, Wortley P, et al. Adherence to expanded influenza immunization recommendations among primary care providers. J Pediatr. 2012;160(3):480-486.
12. Pogorzelska M, Stone PW, Furuya EY, et al. Impact of the ventilator bundle on ventilator-associated pneumonia in intensive care unit. Int J Qual Health Care. 2011;23(5):538-544.
13. Cason C, Tyner T, Saunders S, Broome L. Nurses' implementation of guidelines for ventilator-associated pneumonia from the Centers for Disease Control and Prevention. Am J Crit Care. 2007;16(1):28-37.
14. Wise PC, Finkelstein JA, Ray GT, et al. Aging population and future burden of pneumococcal pneumonia in the United States. J Infect Dis. 2012;205:1589-1592.
15. Arnold F, LaJoie S, Brock GN, et al. Improving outcomes in elderly patients with community-acquired pneumonia by adhering to national guidelines: Community-Acquired Pneumonia Organization International Cohort study results. Arch Intern Med. 2009;169(16):1515-1524.
16. Frei C, Attridge RT, Mortensen EM, et al. Guideline-concordant antibiotic use and survival among patients with community-acquired pneumonia admitted to the intensive care unit. Clin Ther. 2010;32(2):293-299.
17. McCabe C, Kirchner C, Zhang H, Daley J, Fisman DN. Guideline-concordant therapy and reduced mortality and length of stay in adults with community-acquired pneumonia. Arch Intern Med. 2009;169(16):1525-1531.
18. Seymann G, Di Francesco L, Sharpe B, et al. The HCAP gap: differences between self-reported practice patterns and published guidelines for health care-associated pneumonia. Clin Infect Dis. 2009;49(12):1868-1874.
19. Youdelman M, Perkins J. Providing Language Interpretation Services in Health Care Settings: Examples from the Field. Available at https://www.commonwealthfund.org/publications/fund-reports/2002/may/providing-language-interpretation-services-health-care-settings?redirect_source=/publications/fund-reports/2002/may/providing-language-interpretation-services-in-health-care-settings--examples-from-the-field. Last accessed August 26, 2024.
20. Wu J, Howard DH, McGowan JE Jr, Turpin RS, Henry Hu X. Adherence to Infectious Diseases Society of America guidelines for empiric therapy for patients with community-acquired pneumonia in a commercially insured cohort. Clin Ther. 2006;28(9):1451-1461.
21. Dellit T, Owens RC, McGowan JE Jr, et al. Infectious Diseases Society of America and the Society for Healthcare Epidemiology of America guidelines for developing an institutional program to enhance antimicrobial stewardship. Clin Infect Dis. 2007;44(2):159-177.
22. Menéndez R, Torres A, Zalacaín R, et al. Guidelines for the treatment of community-acquired pneumonia: predictors of adherence and outcome. Am J Respir Crit Care Med. 2005;172(6):757-762.
23. El-Solh A, Alhajhusain A, Saliba RG, Drinka P. Physicians' attitudes toward guidelines for the treatment of hospitalized nursing home-acquired pneumonia. J Am Med Dir Assoc. 2011;12(4):270-276.
24. Templeton K, Scheltinga SA, van den Eeden WCJFM, Graffelman AW, van den Broek PJ, Claas ECJ. Improved diagnosis of the etiology of community-acquired pneumonia with real-time polymerase chain reaction. Clin Infect Dis. 2005;41(3):345-351.
25. Davis B, Aiello AE, Dawid S, Rohani P, Shrestha S, Foxman B. Influenza and community-acquired pneumonia interactions: the impact of order and time of infection on population patterns. Am J Epidemiol. 2012;175(5):363-367.
26. Kieninger A, Lipsett PA. Hospital-acquired pneumonia: pathophysiology, diagnosis, and treatment. Surg Clin N Am. 2009;89(2): 439-461.
27. Anand N, Kollef MH. The alphabet soup of pneumonia: CAP, HAP, HCAP, NHAP, and VAP. Semin Respir Crit Care Med. 2009;30(1):3-9.
28. American Thoracic Society, Infectious Diseases Society of America. Management of adults with hospital-acquired and ventilator-associated pneumonia: 2016 clinical practice guidelines by the Infectious Diseases Society of America and the American Thoracic Society. Clin Infect Dis. 2016;63:1-61.
29. Catia C, Santiago E, Eva P, et al. Microbial aetiology of community-acquired pneumonia and its relation to severity. Thorax. 2011;66:340-346.
30. Bradley J, Byington CL, Shah SS, et al. The management of community-acquired pneumonia in infants and children older than 3 months of age: clinical practice guidelines by the Pediatric Infectious Diseases Society and the Infectious Diseases Society of America. Clin Infect Dis. 2011;53(7):e25-e76.
32. Ruuskanen O, Lahti E, Jennings LC, Murdoch DR. Viral pneumonia. Lancet. 2011;377(9773):1264-1275.
33. Johansson N, Kalin M, Tiveljung-Lindell A, Giske CG, Hedlund J. Etiology of community-acquired pneumonia: increased microbiological yield with new diagnostic methods. Clin Infect Dis. 2010;50(2):202-209.
34. Jennings L, Anderson TP, Beynon KA, et al. Incidence and characteristics of viral community-acquired pneumonia in adults. Thorax. 2008;63(1):42-48.
35. Flanders S, Collard HR, Saint S. Nosocomial pneumonia: state of the science. Am J Infect Control. 2006;34(2):84-93.
36. Niederman M, Mandell LA, Anzueto A, et al. Guidelines for the management of adults with community-acquired pneumonia: diagnosis, assessment of severity, antimicrobial therapy, and prevention. Am J Respir Crit Care Med. 2001;163(7):1730-1754.
37. File TMJ. The science of selecting antimicrobials for community-acquired pneumonia (CAP). J Manag Care Pharm. 2009;15(2 Suppl):S5-S11.
38. Weycker D, Strutton D, Edesberg J, Sato R, Jackson LA. Clinical and economic burden of pneumococcal disease in older U.S. adults. Vaccine. 2010;28(31):4955-4960.
39. Jackson M, Neuzil KM, Thompson WW, et al. The burden of community-acquired pneumonia in seniors: results of a population-based study. Clin Infect Dis. 2004;39(11):1642-1650.
40. Ruhnke G, Coca-Perrallion M, Kitch BT, Cutler DM. Marked improvement in 30-day mortality among elderly inpatients and outpatients with community-acquired pneumonia. Am J Med. 2011;124(2):171-178.
41. Farr B, Bartlett CL, Wadsworth J, Miller DL. Risk factors for community-acquired pneumonia diagnosed upon hospital admission. Respir Med. 2000;94(10):954-963.
43. Müllerova H, Chigbo C, Hagan GW, et al. The natural history of community-acquired pneumonia in COPD patients: a population database analysis. Respir Med. 2012;106(8):1124-1133.
44. Sethi S, Maloney J, Grove L, Wrona C, Berenson CS. Airway inflammation and bronchial bacterial colonization in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2006;173:991-998.
45. Eom CS, Jeon CY, Lim J-W, Cho E-G, Park SM, Lee K-S. Use of acid-suppressive drugs and risk of pneumonia: a systematic review and meta-analysis. CMAJ. 2011;183(3):310-319.
46. Jinno S, Jacobs MR. Pneumonia due to drug-resistant Streptococcus pneumoniae. Curr Infect Dis Rep. 2012;14(3):292-299.
47. Mandell L, Wunderink RG, Anzueto A, et al. Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community-acquired pneumonia in adults. Clin Infect Dis. 2007;44:S27-S72.
48. Thiem U, Heppner HJ, Pientka L. Elderly patients with community-acquired pneumonia: optimal treatment strategies. Drugs Aging. 2011;28(7):519-537.
49. Moran G, Krishnadasan A, Gorwitz RJ, et al. Prevalence of methicillin-resistant Staphylococcus aureus as an etiology of community-acquired pneumonia. Clin Infect Dis. 2012;54(8):1126-1133.
51. Metersky M, Sweeney TA, Getzow MB, Siddiqui F, Nsa W, Bratzler DW. Antibiotic timing and diagnostic uncertainty in Medicare patients with pneumonia: is it reasonable to expect all patients to receive antibiotics within 4 hours? Chest. 2006;130(1):16-21.
52. Fee C, Weber EJ. Identification of 90% of patients ultimately diagnosed with community-acquired pneumonia within four hours of emergency department arrival may not be feasible. Ann Emerg Med. 2007;49(5):553-559.
53. McGee SR (ed). Evidence-Based Physical Diagnosis. 3rd ed. Philadelphia, PA: Elsevier Inc.; 2012.
54. Heckerling P, Tape TG, Wigton RS, et al. Clinical prediction rule for pulmonary infiltrates. Ann Intern Med. 1990;113(9):664-670.
55. Campbell S, Marrie TJ, Anstey R, Ackroyd-Stolarz S, Dickinson G. Utility of blood cultures in the management of adults with community acquired pneumonia discharged from the emergency department. Emerg Med J. 2003;20(6):521-523.
56. Afshar N, Tabas J, Afshar K, Silbergleit R. Blood cultures for community-acquired pneumonia: are they worthy of two quality measures? A systematic review. J Hosp Med. 2009;4(2):112-123.
57. Nazarian D, Eddy OL, Lukens TW, et al. Clinical policy: critical issues in the management of adult patients presenting to the emergency department with community-acquired pneumonia. Ann Emerg Med. 2009;54(5):704-731.
58. Christ-Crain M, Opal SM. Clinical review: The role of biomarkers in the diagnosis and management of community-acquired pneumonia. Crit Care. 2010;14(1):203.
59. Berg P, Lindhardt BØ. The role of procalcitonin in adult patients with community-acquired pneumonia: a systematic review. Dan Med J. 2012;59(3):A4357.
60. Ostapchuk M, Roberts DM, Haddy R. Community-acquired pneumonia in infants and children. Am Fam Physician. 2004;70(5): 899-908.
61. Ebell M. Point-of-care guides: clinical diagnosis of pneumonia in children. Am Fam Physician. 2010;82(2):192-193.
62. Lynch T, Platt R, Gouin S, Larson C, Patenaude Y. Can we predict which children with clinically suspected pneumonia will have the presence of focal infiltrates on chest radiographs? Pediatrics. 2004;113(3 pt 1):e186-e189.
63. Cevey-Macherel M, Galetto-Lacour A, Gervaix A, et al. Etiology of community-acquired pneumonia in hospitalized children based on WHO clinical guidelines. Eur J Pediatr. 2009;168(12):1429-1436.
64. Mandell LA, Bartlett JG, Dowell SF, et al. Update of practice guidelines for the management of community-acquired pneumonia in immunocompetent adults. Clin Infect Dis. 2003;37(11):1405-1433.
65. Forgie S, Marrie TJ. Healthcare-associated atypical pneumonia. Semin Respir Crit Care Med. 2009;30(1):67-85.
66. Centers for Disease Control and Prevention. Mycoplasma pneumoniae Infection. Available at https://www.cdc.gov/mycoplasma/about/index.html. Last accessed August 26, 2024.
67. Chang C, Cheng AC, Chang AB. Over-the-counter (OTC) medications to reduce cough as an adjunct to antibiotics for acute pneumonia in children and adults. Cochrane Database Syst Rev. 2014;3:CD006088.
68. Niederman M. Making sense of scoring systems in community acquired pneumonia. Respirology. 2009;14(3):327-335.
69. Fine M, Auble TE, Yealy DM, et al. A prediction rule to identify low-risk patients with community-acquired pneumonia. N Engl J Med. 1997;336(4):243-250.
70. Lim W, van der Eerden MM, Laing R, et al. Defining community-acquired pneumonia severity on presentation to hospital: an international derivation and validation study. Thorax. 2003;58(5):377-382.
71. Chalmers J, Singanayagam A, Akram AR, et al. Severity assessment tools for predicting mortality in hospitalised patients with community-acquired pneumonia: systematic review and meta-analysis. Thorax. 2010;65(10):878-883.
72. Loke Y, Kwok CS, Niruban A, Myint PK. Value of severity scales in predicting mortality from community-acquired pneumonia: systematic review and meta-analysis. Thorax. 2010;65:884-890.
73. Charles P, Wolfe R, Whitby M, et al. SMART-COP: a tool for predicting the need for intensive respiratory or vasopressor support in community-acquired pneumonia. Clin Infect Dis. 2008;47(3):375-384.
74. Yandiola P, Capelastegui A, Quintana J, et al. Prospective comparison of severity scores for predicting clinically relevant outcomes for patients hospitalized with community-acquired pneumonia. Chest. 2009;135(6):1572-1579.
75. España P, Capelastegui A, Quintana, JM, et al. Validation and comparison of SCAP as a predictive score for identifying low-risk patients in community-acquired pneumonia. J Infect. 2010;60(2):106-113.
76. File TMJ, Marrie TJ. Burden of community-acquired pneumonia in North American adults. Postgrad Med. 2010;122(2):130-141.
77. Kontou P, Kuti JL, Nicolau DP. Validation of the Infectious Diseases of America/American Thoracic Society criteria to predict severe community-acquired pneumonia caused by Streptococcus pneumoniae. Am J Emerg Med. 2009;27(8):968-974.
78. Chalmers J, Taylor JK, Mandal P, et al. Validation of the Infectious Diseases Society of America/American Thoracic Society minor criteria for intensive care unit admission in community-acquired pneumonia patients without major criteria or contraindications to intensive care unit care. Clin Infect Dis. 2011;53(6):503-511.
79. Kanwar M, Brar N, Khatib R, Fakih MG. Misdiagnosis of community-acquired pneumonia and inappropriate utilization of antibiotics. Chest. 2007;131:1865-1869.
80. Nicks B, Manthey DE, Fitch MT. The Centers for Medicare and Medicaid Services (CMS) community-acquired pneumonia core measures lead to unnecessary antibiotic administration by emergency physicians. Acad Emerg Med. 2009;16(2):184-187.
81. Yu K, Wyer PC. Evidence behind the 4-hour rule for initiation of antibiotic therapy in community-acquired pneumonia. Ann Emerg Med. 2008;51(5):651-652.
82. Quattromani E, Powell ES, Khare RK, et al. Hospital-reported data on the pneumonia quality measure "time to first antibiotic dose" is not associated with inpatient mortality: results of a nationwide cross-sectional analysis. Acad Emerg Med. 2011;18(5):496-503.
83. Asadi L, Eurich DT, Gamble JM, Minhas-Sandhu JK, Marrie TJ, Majumdar SR. Guideline adherence and macrolides reduced mortality in outpatients with pneumonia. Respir Med. 2012;106(3):451-458.
84. Neuman M, Ting SA, Meydani A, Mansbach JM, Camargo CA Jr. National study of antibiotic use in emergency department visits for pneumonia, 1993 through 2008. Acad Emerg Med. 2012;19(5):562-568.
85. Simpson S, Marrie TJ, Majumdar SR. Do guidelines guide pneumonia practice? A systematic review of interventions and barriers to best practice in the management of community-acquired pneumonia. Respir Care Clin North Am. 2005;11(1):1-13.
86. Schouten JA, Hulscher ME, Trap-Liefers J, et al. Tailored interventions to improve antibiotic use for lower respiratory tract infections in hospitals: a cluster-randomized, controlled trial. Clin Infect Dis. 2007;44(7):931-941.
87. Weiner S, Brown SF, Goetz JD, Webber CA. Weekly e-mail reminders influence emergency physician behavior: a case study using the Joint Commission and Centers for Medicare and Medicaid Services Pneumonia Guidelines. Acad Emerg Med. 2009;16(7):626-631.
88. Fleming NS, Ogola G, Ballard DJ. Implementing a standardized order set for community-acquired pneumonia: impact on mortality and cost. Jt Comm J Qual Patient Saf. 2009;35(8):414-421.
89. Haider B, Lassi ZS, Ahmed A, Bhutta ZA. Zinc supplementation as an adjunct to antibiotics in the treatment of pneumonia in children 2 to 59 months of age. Cochrane Database Syst Rev. 2011;10:CD007368.
90. Aliberti S, Di Pasquale M, Zanaboni AM, et al. Stratifying risk factors for multidrug-resistant pathogens in hospitalized patients coming from the community with pneumonia. Clin Infect Dis. 2012;54(4):470-478.
91. Kroger AT, Sumaya CV, Pickering LK, Atkinson WL. General recommendations on immunization: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR. 2011;60(RR2);1-60.
92. Centers for Disease Control and Prevention. Recommended adult immunization schedule—United States, 2012. MMWR. 2012;61(4):1-7.
93. Tablan O, Anderson LJ, Besser R, Bridges C, Hajjeh R. Guidelines for preventing health-care-associated pneumonia, 2003: recommendations of CDC and the Healthcare Infection Control Practices Advisory Committee. MMWR. 2004;53(RR3):1-36.
94. Infectious Disease Society of America. COVID-19 Resource Center. Available at https://www.idsociety.org/covid-19-real-time-learning-network/infection-prevention/battling-health-care-associated-infections-in-the-covid-19-era-compendium-best-practices/#/+/0/publishedDate_na_dt/desc/. Last accessed August 26, 2024.
95. Edmond K, Scott S, Korczak V, et al. Long term sequelae from childhood pneumonia: systematic review and meta-analysis. PLoS One. 2012;7(2):e31239.
96. Murthy N, Wodi AP, Bernstein H, McNally V, Cineas S, Ault K. Advisory Committee on Immunization Practices recommended immunization schedule for adults aged 19 years or older—United States, 2022. MMWR. 2022;71(7):229-233.
97. Immunization Action Coalition. Pneumococcal: PCV. Available at https://www.immunize.org/vaccines/a-z/pneumococcal/. Last accessed August 26, 2024.
98. Grijalva C, Nuorti JP, Arbogast PG, Martin SW, Edwards KM, Griffin MR. Decline in pneumonia admissions after routine childhood immunisation with pneumococcal conjugate vaccine in the USA: a time-series analysis. Lancet. 2007;369(9568):1179-1186.
99. Klugman K, Chien YW, Madhi SA. Pneumococcal pneumonia and influenza: a deadly combination. Vaccine. 2009;27(Suppl 3): C9-C14.
100. Holden J, Tatham DP, Andrews RM. Vaccines for preventing pneumococcal infection in adults. Cochrane Database Syst Rev. 2013;1:CD000422.
101. Lucero M, Dulalia VE, Nillos LT, et al. Pneumococcal conjugate vaccines for preventing vaccine-type invasive pneumococcal disease and x-ray defined pneumonia in children less than two years of age. Cochrane Database Syst Rev. 2009;7(4):CD004977.
102. Centers for Disease Control and Prevention. Flu Vaccination Coverage, United States, 2021–22 Influenza Season. Available at https://www.cdc.gov/flu/fluvaxview/coverage-2022estimates.htm. Last accessed August 23, 2024.
103. Centers for Disease Control and Prevention. Influenza Vaccination Coverage Among Health Care Personnel—United States, 2021–22 Influenza Season. Available at https://www.cdc.gov/mmwr/volumes/71/wr/mm7142a2.htm?s_cid=mm7142a2_w. Last accessed August 23, 2024.
104. Santibanez T, Nowalk MP, Zimmerman RK, et al. Knowledge and beliefs about influenza, pneumococcal disease, and immunizations among older people. J Am Geriatr Soc. 2002;50(10):1711-1716.
105. Hebert P, Frick KD, Kane RL, McBean AM. The causes of racial and ethnic differences in influenza vaccination rates among elderly Medicare beneficiaries. Health Serv Res. 2005;40(2):517-537.
106. Winston C, Wortley PM, Lees KA. Factors associated with vaccination of Medicare beneficiaries in five U.S. communities: results from the racial and ethnic adult disparities in immunization initiative survey, 2003. J Am Geriatr Soc. 2006;54(2):303-310.
107. Haviland A, Elliott MN, Hambarsoomian K, Lurie N. Immunization disparities by Hispanic ethnicity and language preference. Arch Intern Med. 2011;171(2):158-165.
108. Marsteller J, Tiggle RB, Remsburg RE, Bardenheier B, Shefer A, Han B. Pneumococcal vaccination in nursing homes: does race make a difference? J Am Med Dir Assoc. 2008;9(9):641-647.
109. Bardenheier B, Wortley P, Ahmed F, Gravenstein S, Hogue CJ. Racial inequities in receipt of influenza vaccination among long-term care residents within and between facilities in Michigan. Med Care. 2011;49(4):371-377.
110. Bardenheier B, Wortley P, Shefer A, McCauley MM, Gravenstein S. Racial inequities in receipt of influenza vaccination among nursing home residents in the United States, 2008—a pattern of low overall coverage in facilities in which most residents are black. J Am Med Dir Assoc. 2012;13(5):470-476.
111. Bratzler DW, Houck PM, Jiang H, et al. Failure to vaccinate Medicare inpatients: a missed opportunity. Arch Intern Med. 2002;162(20):2349-2356.
112. Hill HA, Elam-Evans LD, Yankey D, et al. Vaccination coverage among children aged 19–35 months—United States, 2016. MMWR. 2017;66(43):1171-1177.
113. National Center for Health Statistics. Health, United States, 2016. Available at https://www.cdc.gov/nchs/data/hus/hus16.pdf. Last accessed August 23, 2024.
114. U.S. Department of Health and Human Services. Healthy People 2030: Vaccination. Available at https://health.gov/healthypeople/objectives-and-data/browse-objectives/vaccination. Last accessed August 26, 2024.
116. Johnson D, Nichol KL, Lipczynski K. Barriers to adult immunization. Am J Med. 2008;121(7 Suppl 2):S28-S35.
118. Lindley M, Wortley PM, Winston CA, Bardenheier BH. The role of attitudes in understanding disparities in adult influenza vaccination. Am J Prev Med. 2006;31(4):281-285.
119. Pearson W, Zhao G, Ford ES. An analysis of language as a barrier to receiving influenza vaccinations among an elderly Hispanic population in the United States. Adv Prev Med. 2011:298787.
120. Mills E, Jadad AR, Ross C, Wilson K. Systematic review of qualitative studies exploring parental beliefs and attitudes toward childhood vaccination identifies common barriers to vaccination. J Clin Epidemiol. 2005;58(11):1081-1088.
121. Niederhauser V, Markowitz M. Barriers to immunizations: multiethnic parents of under- and unimmunized children speak. J Am Acad Nurse Pract. 2007;19(1):15-23.
122. Luthy K, Beckstrand RL, Peterson NE. Parental hesitation as a factor in delayed childhood immunization. J Pediatr Health Care. 2009;23(6):388-393.
123. Nichol K. Improving influenza vaccination rates among adults. Cleve Clin J Med. 2006;73(11):1009-1015.
124. Office of Minority Health. Cultural and Linguistic Competency. Available at https://minorityhealth.hhs.gov/cultural-and-linguistic-competency. Last accessed August 26, 2024.
125. Paez K, Allen JK, Beach MC, Carson KA, Cooper LA. Physician cultural competence and patient ratings of the patient-physician relationship. J Gen Intern Med. 2009;24(4):495-498.
126. Powers B, Trinh JV, Bosworth HB. Can this patient read and understand written health information? JAMA. 2010;304(1):76-84.
127. U.S. Census Bureau. Selected Social Characteristics in the United States: 2019. Available at https://data.census.gov/cedsci/table?q=english&tid=ACSSPP1Y2019.S0201. Last accessed August 26, 2024.
128. Karliner L, Napoles-Springer AM, Schillinger D, Bibbins-Domingo K, Pérez-Stable EJ. Identification of limited English proficient patients in clinical care. J Gen Intern Med. 2008;23(10):1555-1560.
129. Karliner L, Jacobs EA, Chen AH, Mutha S. Do professional interpreters improve clinical care for patients with limited English proficiency? A systematic review of the literature. Health Serv Res. 2007;42(2):727-754.
130. Flores G. The impact of medical interpreter services on the quality of health care: a systematic review. Med Care Res Rev. 2005;62(3):255-299.
131. Ngo-Metzger Q, Massagli MP, Clarridge BR, et al. Linguistic and cultural barriers to care: perspectives of Chinese and Vietnamese immigrants. J Gen Intern Med. 2003;18(1):44-52.
132. Committee on Health Literacy Board on Neuroscience and Behavioral Health. Health Literacy: A Prescription to End Confusion. Washington, DC: The National Academies Press; 2004.
133. Paasche-Orlow M, Parker RM, Gazmararian JA, Nielsen-Bohlman LT, Rudd RR. The prevalence of limited health literacy. J Gen Intern Med. 2005;20(2):175-184.
134. Kutner M, Greenberg E, Jin,Y, Paulsen C, White S. The Health Literacy of America's Adults: Results from the 2003 National Assessment of Adult Literacy. Washington, DC: National Center for Education Statistics; 2006.
135. Shah L, West P, Bremmeyr K, Savoy-Moore RT. Health literacy instrument in family medicine: the "newest vital sign" ease of use and correlates. J Am Board Fam Med. 2010;23(2):195-203.
136. Weiss B, Mays MZ, Martz W, et al. Quick assessment of literacy in primary care: the newest vital sign. Ann Fam Med. 2005;3(6):514-522.
137. Jacobson V, Szilagyi P. Patient reminder and patient recall systems to improve immunization rates. Cochrane Database Syst Rev. 2005;(3):CD003941.
138. Traeger M, Say KR, Hastings V, Yost DA. Achievement of Healthy People 2010 objective for adult pneumococcal vaccination in an American Indian community. Pub Health Rep. 2010;125(3):448-456.
139. Rimple D, Weiss SJ, Brett M, Ernst AA. An emergency department-based vaccination program: overcoming the barriers for adults at high risk for vaccine-preventable diseases. Acad Emerg Med. 2006;13(9):922-930.
140. Martin D, Brauner ME, Plouffe JF. Influenza and pneumococcal vaccinations in the emergency department. Emerg Med Clin North Am. 2008;26(2):549-570.
141. Society of Healthcare Epidemiology of America Research Committee. Enhancing patient safety by reducing healthcare-associated infections: the role of discovery and dissemination. Infect Control Hosp Epidemiol. 2010;31(2):118-123.
142. Richards MJ, Edwards JR, Culver DH, Gaynes RP. Nosocomial infections in pediatric intensive care units in the United States: National Nosocomial Infections Surveillance System. Pediatrics. 1999;103(4):e39.
143. Mills K, Graham AC, Winslow BT, Springer KL. Treatment of nursing home-acquired pneumonia. Am Fam Physician. 2009;79(11):976-982.
144. Foglia E, Meier MD, Elward A. Ventilator-associated pneumonia in neonatal and pediatric intensive care unit patients. Clin Microbiol Rev. 2007;20(3):409-425.
145. Qaseem A, Snow V, Fitteman N, et al. Risk assessment for and strategies to reduce perioperative pulmonary complications for patients undergoing noncardiothoracic surgery: a guideline from the American College of Physicians. Ann Intern Med. 2006;144(8):575-580.
146. Pileggi C, Bianco A, Flotta D, Nobile CG, Pavia M. Prevention of ventilator-associated pneumonia, mortality and all intensive care unit acquired infections by topically applied antimicrobial or antiseptic agents: a meta-analysis of randomized controlled trials in intensive care units. Crit Care. 2011;15(3):R155.
147. Kollef MH. Antibiotic management of ventilator-associated pneumonia due to antibiotic-resistant gram-positive bacterial infection. Eur J Clin Microbiol Infect Dis. 2005;24(12):794-803.
148. Kollef MH. What is ventilator-associated pneumonia and why is it important? Respir Care. 2005;50(6):714-721.
149. Dodek P, Keenan S, Cook D, et al. Evidence-based clinical practice guideline for the prevention of ventilator-associated pneumonia. Ann Intern Med. 2004;141(4):305-313.
150. Bigham M, Amato R, Bondurrant P, et al. Ventilator-associated pneumonia in the pediatric intensive care unit: characterizing the problem and implementing a sustainable solution. J Pediatr. 2009;154(4):582-587.
151. Kollef MH, Morrow LE, Niederman MS, et al. Clinical characteristics and treatment patterns among patients with ventilator-associated pneumonia. Chest. 2006;129(5):1210-1218.
152. Hidron A, Edwards JR, Patel J, et al. Antimicrobial-resistant pathogens associated with healthcare-associated infections: annual summary of data reported to the National Healthcare Safety Network at the Centers for Disease Control and Prevention, 2006–2007. Infect Control Hosp Epidemiol. 2008;29(11):996-1011.
153. Kollef MH, Micek ST. Staphylococcus aureus pneumonia: a "superbug" infection in community and hospital settings. Chest. 2005;128(3):1093-1097.
154. Depuydt P, Myny D, Blot S. Nosocomial pneumonia: aetiology, diagnosis and treatment. Curr Opin Pulm Med. 2006;12(3):192-197.
155. El-Solh A, Niederman MS, Drinka P. Nursing home-acquired pneumonia: a review of risk factors and therapeutic approaches.Curr Med Res Opin. 2010;26(12):2707-2714.
157. Horan TC, Andrus M, Dudeck MA. CDC/NHSN surveillance definition of health care-associated infection and criteria for specific types of infections in the acute care setting. Am J Infect Control. 2008;36(5):309-332.
158. Porzecanski I, Bowton DL. Diagnosis and treatment of ventilator-associated pneumonia. Chest. 2006;130(2):597-604.
159. Miller PR, Johnson JC 3rd, Karchmer T, Hoth JJ, Meredith JW, Chang MC. National nosocomial infection surveillance system: from benchmark to bedside in trauma patients. J Trauma. 2006;60(1):98-103.
160. Zaccard CR, Schell RF, Spiegel CA. Efficacy of bilateral bronchoalveolar lavage for diagnosis of ventilator-associated pneumonia. J Clin Microbiol. 2009;47(9):2918-2924.
161. Berton D, Kalil AC, Teixeira PJ. Quantitative versus qualitative cultures of respiratory secretions for clinical outcomes in patients with ventilator-associated pneumonia. Cochrane Database Syst Rev. 2014;10:CD006482.
162. Micek ST, Heuring TJ, Hollands JM, Shah RA, Kollef MH. Optimizing antibiotic treatment for ventilator-associated pneumonia. Pharmacotherapy. 2006;26(2):204-213.
163. Rello J, Vidaur L, Sandiumenge A, et al. De-escalation therapy in ventilator-associated pneumonia. Crit Care Med. 2004;32(11):2183-2190.
164. Sligl W, Taylor G, Brindley PG. Five years of nosocomial gram-negative bacteremia in a general intensive care unit: epidemiology, antimicrobial susceptibility patterns, and outcomes. Int J Infect Dis. 2006;10(4):320-325.
165. Mullins D, Kuznik C, Shaya FT, Obeidat NA, Levine AR, Liu LZ, Wong W. Cost-effectiveness analysis of linezolid compared with vancomycin for the treatment of nosocomial pneumonia caused by methicillin-resistant Staphylococcus aureus. Clin Ther. 2006;28(8):1184-1198.
166. Shorr AF, Susla GM, Kollef MH. Linezolid for treatment of ventilator-associated pneumonia: a cost-effective alternative to vancomycin. Crit Care Med. 2004;32(1):137-143.
167. Grau S, Alvarez-Lerma F, del Castillo A, Neipp R, Rubio-Terres C. Cost-effectiveness analysis of the treatment of ventilator-associated pneumonia with linezolid or vancomycin in Spain. J Chemother. 2005;17(2):203-211.
168. Kollef MH, Rello J, Cammarata SK, Croos-Dabrera RV, Wunderink RG. Clinical cure and survival in gram-positive ventilator-associated pneumonia: retrospective analysis of two double-blind studies comparing linezolid with vancomycin. Intensive Care Med. 2004;30(3):388-394.
169. Wunderink RG, Mendelson MH, Somero MS, et al. Early microbiological response to linezolid vs vancomycin in ventilator-associated pneumonia due to methicillin-resistant Staphylococcus aureus. Chest. 2008;134(6):1200-1207.
170. Pugh R, Grant C, Cooke RP, Dempsey G. Short-course versus prolonged-course antibiotic therapy for hospital-acquired pneumonia in critically ill adults. Cochrane Database Syst Rev. 2015;8:CD007577.
171. Coffin SE, Klompas M, Classen D, et al. Strategies to prevent ventilator-associated pneumonia in acute care hospitals. Infect Control Hosp Epidemiol. 2008;29:S31-S40.
172. van Nieuwenhoven CA, Vandenbroucke-Grauls C, van Tiel FH, et al. Feasibility and effects of the semirecumbent position to prevent ventilator-associated pneumonia: a randomized study. Crit Care Med. 2006;34(2):396-402.
173. Tolentino-Delos Reyes AF, Ruppert SD, Shiao SY. Evidence-based practice: use of the ventilator bundle to prevent ventilator-associated pneumonia. Am J Crit Care. 2007;16(1):20-27.
174. Institute for Healthcare Improvement. How-to Guide: Prevent Ventilator-Associated Pneumonia. Available at https://cha.com/wp-content/uploads/2018/03/IHI-HowtoGuidePreventVAP-with-bundle.pdf. Last accessed August 26, 2024.
175. Kress J, Pohlman AS, O'Connor MF, Hall JB. Daily interruption of sedative infusions in critically ill patients undergoing mechanical ventilation. N Engl J Med. 2000;342(20):1471-1477.
176. Schweickert W, Gehlbach BK, Pohlman AS, Hall JB, Kress JP. Daily interruption of sedative infusions and complications of critical illness in mechanically ventilated patients. Crit Care Med. 2004;32(6):1272-1276.
177. Antalová N, Klučka J, Říhová M, Poláčková S, Pokorná A, Štourač P. Ventilator-associated pneumonia prevention in pediatric patients: narrative review. Children (Basel). 2022;9(10):1540.
178. Bearman GM, Munro C, Sessler CN, Wenzel RP. Infection control and the prevention of nosocomial infections in the intensive care unit. Semin Respir Crit Care Med. 2006;27:310-324.
179. Drakulovic MB, Torres A, Bauer TT, Nicolar JM, Nogué S, Ferrer M. Supine body position as a risk factor for nosocomial pneumonia in mechanically ventilated patients: a randomised trial. Lancet. 1999;354(9193):1851-1858.
180. Shay A, O'Malley P. Blue Ribbon Abstract Award: clinical outcomes of a ventilator associated pneumonia prevention program. Am J Infect Control. 2006;34(5):E19-E20.
181. El-Solh AA, Pietrantoni C, Bhat A, et al. Colonization of dental plaques: a reservoir of respiratory pathogens for hospital-acquired pneumonia in institutionalized elders. Chest. 2004;126(5):1575-1582.
182. Sona C, Zack JE, Schallom ME, et al. The impact of a simple, low-cost oral care protocol on ventilator-associated pneumonia rates in a surgical intensive care unit. J Intensive Care Med. 2009;24(1):54-62.
183. Munro C, Grap MJ, Jones DJ, McClish DK, Sessler CN. Chlorhexidine, toothbrushing, and preventing ventilator-associated pneumonia in critically ill adults. Am J Crit Care. 2009;18(5):428-437.
184. Fourrier F, Dubois D, Pronnier P, et al. Effect of gingival and dental plaque antiseptic decontamination on nosocomial infections acquired in the intensive care unit: a double-blind placebo-controlled multicenter study. Crit Care Med. 2005;33(8):1728-1735.
185. Pineda LA, Saliba RG, El Solh AA. Effect of oral decontamination with chlorhexidine on the incidence of nosocomial pneumonia: a meta-analysis. Crit Care. 2006;10(1):R35.
186. Bopp M, Darby M, Loftkin KC, Broscious S. Effects of daily oral care with 0.12% chlorhexidine gluconate and a standard oral care protocol on the development of nosocomial pneumonia in intubated patients: a pilot study. J Dent Hyg. 2006;80(3):9.
187. World Health Organization. Prevention of Hospital-Acquired Infections: A Practical Guide. 2nd ed. Geneva: WHO Press; 2002.
188. Geerts WH, Pineo GF, Heit JA, et al. Prevention of venous thromboembolism: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest. 2004;126(3 Suppl):338S-400S.
189. Dezfulian C, Shojania K, Collard HR, Kim HM, Matthay MA, Saint S. Subglottic secretion drainage for preventing ventilator-associated pneumonia: a meta-analysis. Am J Med. 2005;118(1):11-18.
190. Bouza E, Pérez MJ, Muñoz P, Rincón C, Barrio JM, Hortal J. Continuous aspiration of subglottic secretions in the prevention of ventilator-associated pneumonia in the postoperative period of major heart surgery. Chest. 2008;134(5):938-946.
191. Wang F, Bo L, Tang L, et al. Subglottic secretion drainage for preventing ventilator-associated pneumonia: an updated meta-analysis of randomized controlled trials. J Trauma Acute Care Surg. 2012;72(5):1276-1285.
192. Girou E, Brun-Buisson C, Taille S, Lemaire F, Brochard L. Secular trends in nosocomial infections and mortality associated with noninvasive ventilation in patients with exacerbation of COPD and pulmonary edema. JAMA. 2003;290(22):2985-2991.
193. Isakow W, Kollef MH. Preventing ventilator-associated pneumonia: an evidence-based approach of modifiable risk factors. Semin Respir Crit Care Med. 2006;27(1):5-17.
194. Osmon S, Kollef MH. Prevention of pneumonia in the hospital setting. Clin Chest Med. 2005;26(1):135-142.
195. Craven DE, Hjalmarson K. Prophylaxis of ventilator-associated pneumonia: changing culture and strategies to trump disease.Chest. 2008;134(5):898-900.
196. Jain M, Miller L, Belt D, King D, Berwick DM. Decline in ICU adverse events, nosocomial infections and cost through a quality improvement initiative focusing on teamwork and culture change. Qual Saf Health Care. 2006;15(4):235-239.
197. Stone MJ, Snetman D, O'Neill A, et al. Daily multidisciplinary rounds to implement the ventilator bundle decreases ventilator-associated pneumonia in trauma patients: but does it affect outcome? Surg Infect (Larchmt). 2011;12(5):373-378.
198. Cachecho R, Dobkin E. The application of human engineering interventions reduces ventilator-associated pneumonia in trauma patients. J Trauma Acute Care Surg. 2012;73(4):939-943.
199. Hutt E, Kramer AM. Evidence-based guidelines for management of nursing home-acquired pneumonia. J Fam Pract. 2002;51(8):709-716.
200. Salgado CD, Giannetta ET, Hayden FG, Farr BM. Preventing nosocomial influenza by improving the vaccine acceptance rate of clinicians. Infect Control Hosp Epidemiol. 2004;25(11):923-928
201. Carman WF, Elder AG, Wallace LA, et al. Effects of influenza vaccination on health-care workers on mortality of elderly people in long-term care: a randomized controlled trial. Lancet. 2000;355(9198):93-97.
202. Dash GP, Fauerbach L, Pfeiffer J, et al. APIC position paper: Improving health care worker influenza immunization rates. Am J Infect Control. 2004;32(3):123-125.
203. Centers for Disease Control and Prevention. Estimated influenza vaccination coverage among adults and children—United States, September 1, 2004–January 1, 2005. MMWR. 2005;54(12):304-307.
204. Christini AB, Shutt KA, Byers KE. Influenza vaccination rates and motivators among healthcare worker groups. Infect Control Hosp Epidemiol. 2007;28(2):171-177.
205. Pearson ML, Bridges CB, Harper SA. Influenza vaccination of health-care personnel. MMWR. 2006;55(RR02):1-16.
206. U.S. Food and Drug Administration. Reprocessing of Reusable Ultrasound Transducer Assemblies Used for Biopsy Procedures. Available at https://wayback.archive-it.org/7993/20170112170658/http://www.fda.gov/Safety/MedWatch/SafetyInformation/SafetyAlertsforHumanMedicalProducts/ucm151526.htm. Last accessed August 26, 2024.
207. Boyce JM, Pittet D. Guideline for hand hygiene in health-care settings. MMWR. 2002;51(RR16):1-44.
209. Leapfrog Group. Press Release: Eighty-Seven Percent of U.S. Hospitals Do Not Take Recommended Steps to Prevent Avoidable Infections. Available at https://www.infectioncontroltoday.com/view/leapfrog-group-survey-reveals-87-percent-us-hospitals-dont-take. Last accessed August 26, 2024.
210. Clark AP, Houston S. Nosocomial infections: an issue of patient safety: part 2. Clin Nurse Spec. 2004;18(2):62-64.
211. Pittet D, Hugonnet S, Harbarth S, et al. Effectiveness of a hospital-wide programme to improve compliance with hand hygiene. Lancet. 2000;356(9238):1307-1312.
212. Larson EL, Quiros D, Lin SX. Dissemination of the CDC's hand hygiene guideline and impact on infection rates. Am J Infect Control. 2007;35(10):666-675.
213. Weinstein R. Hospital-acquired infections. In: Kasper DL, Braunwald E, Fauci AS, Hauser SL, Longo DL, Jameson JL, Isselbacher KJ (eds.) Harrison's Principles of Internal Medicine. 16th ed. New York, NY: McGraw Hill; 2004.
214. Johnson PDR, Rhea M, Burrell LJ, et al. Efficacy of an alcohol/chlorhexidine hand hygiene program in a hospital with high rates of nosocomial methicillin-resistant Staphylococcus aureus (MRSA) infection. Med J Aust. 2005;183(10):509-514.
215. Gordin FM, Schultz ME, Huber RA, Gill JA. Reduction in nosocomial transmission of drug-resistant bacteria after introduction of an alcohol-based handrub. Infect Control Hosp Epidemiol. 2005;26(7):650-653.
216. Gould DJ, Moralejo D, Drey N, Chudleigh JH, Taljaard M. Interventions to improve hand hygiene compliance in patient care.Cochrane Database Syst Rev. 2017;9:CD005186.
217. Mason CM, Nelson S. Pulmonary host defenses and factors predisposing to lung infection. Clin Chest Med. 2005;26(1):11-17.
218. Johnstone J, Eurich DT, Jamumdar SR, Jin Y, Marrie TJ. Long-term morbidity and mortality after hospitalization with community-acquired pneumonia: a population-based cohort study. Medicine (Baltimore). 2008;87(6):329-334.
219. Santibanez T, Mootrey GT, Euler GL, Janssen AP. Behavior and beliefs about influenza vaccine among adults ages 50–64 years. Am J Health Behav. 2010;34(1):77-89.
220. Griffin M, Zhu Y, Moore M, Whitney C, Grijalva C. U.S. hospitalizations for pneumonia after a decade of pneumococcal vaccination. N Engl J Med. 2013;369:155-163.
221. Nuorti J, Butler J, Farley M, et al. Cigarette smoking and invasive pneumococcal disease. N Engl J Med. 2000;342:681-689.
222. Marrie T, Poulin-Costello M, Beecroft M, Herman-Gnjidic Z. Etiology of community-acquired pneumonia in the ambulatory setting. Respir Med. 205;99:60-65.
223. Kallen AJ, Reed C, Patton M, et al. Staphylococcus aureus community-onset pneumonia in patients admitted to children's hospitals during autumn and winter of 2006–2007. Epidemiol Infect. 2010;138:666-672.
224. Musher D, Montoya R, Wanahita A. Diagnostic value of microscopic examination of Gram-stained sputum and sputum cultures in patients with bacteremic pneumococcal pneumonia. Clin Infect Dis. 2004;39:165-169.
225. Gutiérrez F, Masiá M, Rodríguez JC, et al. Evaluation of the immunochromatographic Binax NOW Assay for detection of Streptococcus pneumoniae urinary antigen in a prospective study of community-acquired pneumonia in Spain. Clin Infect Dis. 2003;36(3):286-292.
226. Toshihiko S, MD, Yoshinori N, Jackson J, et al. Systematic review and meta-analysis: urinary antigen tests for legionellosis. Chest. 2009;136:1576-1585.
227. Matanock A, Lee G, Gierke R, et al. Use of 13-valent pneumococcal conjugate vaccine and 23-valent pneumococcal polysaccharide vaccine among adults aged >/= 65 years: updated recommendations of the Advisory Committee on Immunization Practices. MMWR. 2019;68:1069-1075.
228. World Health Organization. Fact Sheet: Pneumonia. Available at: https://www.who.int/health-topics/pneumonia. Last accessed August 26, 2024.
229. Centers for Disease Control and Prevention. Legionella (Legionaires' Disease and Pontiac Fever). Available at https://www.cdc.gov/legionella/index.html. Last accessed August 26, 2024.
230. Hicks LA, Garrison LE, Nelson GE, Hampton LM. Legionellosis—United States, 2000–2009. MMWR. 2011;60(32):1083-1086.
231. Centers for Disease Control and Prevention. Chlamydophila pneumoniae Infection. Available at https://www.cdc.gov/cpneumoniae/index.html. Last accessed August 26, 2024.
232. Ramirez JA, Wiemken TL Peyrani P, et al. Adults hospitalized with pneumonia in the United States: incidence, epidemiology, and mortality. Clin Infect Dis. 2017;65:1806-1812.
233. American Thoracic Society, Infectious Diseases Society of America. Guidelines for the management of adults with hospital-acquired, ventilator-associated and healthcare-associated pneumonia. Am J Respir Crit Care Med. 2005;171(4):388-416.
234. Centers for Disease Control and Prevention. Underlying Cause of Death 1999–2021 on CDC WONDER Online Database. Available at https://wonder.cdc.gov/. Last accessed August 21, 2024.
235. Metlay JP, Waterer GW, Long AC, et al. Diagnosis and treatment of community-acquired pneumonia in adults: an official clinical practice guideline of the American Thoracic Society and Infectious Diseases Society of America. Am J Respir Crit Care Med. 2019;200(7):e45.
236. Centers for Disease Control and Prevention. Influenza ACIP Vaccine Recommendations. Available at https://www.cdc.gov/acip-recs/hcp/vaccine-specific/flu.html. Last accessed August 23, 2024.
237. Lu P, Hung M, Scrivastav A, et al. Surveillance of vaccination coverage among adult populations—United States, 2018. MMWR. 2021;70(SS-3):1-26.
238. Faner R, Sibila O, Agusti A, et al. The microbiome in respiratory medicine: current challenges and future perspectives. Eur Respir J. 2017;49(4):1602086.
239. Centers for Disease Control and Prevention, Pneumococcal Vaccine Work Group. Pneumococcal Vaccines. Available at https://www.cdc.gov/vaccines/acip/meetings/downloads/slides-2024-06-26-28/01-Pneumococcal-Loehr-508.pdf. Last accessed August 26, 2024.
240. Centers for Disease Control and Prevention. Pneumococcal Vaccine Recommendations. Available at https://www.cdc.gov/vaccines/vpd/pneumo/hcp/recommendations.html. Last accessed August 26, 2024.
1. Kirsch J, Mohammed TH, Kanne JP, et al. ACR Appropriateness Criteria: Acute Respiratory Illness in Immunocompetent Patients. Reston, VA: American College of Radiology; 2018. Available at https://acsearch.acr.org/docs/69446/Narrative. Last accessed August 29, 2024.
2. Liu C, Bayer A, Cosgrove SE, et al. Clinical practice guidelines by the Infectious Diseases Society of America for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children. Clin Infect Dis. 2011;52:1-38. Available at https://academic.oup.com/cid/article/52/3/e18/306145. Last accessed August 29, 2024.
3. Metlay JP, Waterer GW, Long AC, et al. Diagnosis and treatment of adults with community-acquired pneumonia: an official clinical practice guideline of the American Thoracic Society and Infectious Diseases Society of America. Am J Respir Crit Care Med. 2019;200(7):e45-e67. Available at https://www.atsjournals.org/doi/full/10.1164/rccm.201908-1581ST. Last accessed August 29, 2024.
4. Kalil AC, Metersky ML, Klompas M, et al. Management of adults with hospital-acquired and ventilator-associated pneumonia: 2016 clinical practice guidelines by the Infectious Diseases Society of America and the American Thoracic Society. Clin Infect Dis. 2016;63(5):e61-e111. Available at https://www.thoracic.org/statements/resources/tb-opi/hap-vap-guidelines-2016.pdf. Last accessed August 29, 2024.
Mention of commercial products does not indicate endorsement.