The unique and dynamic environment of the oral and maxillofacial complex is subject to a wide variety of pathological processes. Bacterial, fungal, and viral infections in these systems can range from a temporary inconvenience to a life-threatening emergency. Autoimmune disorders that target the oral mucosa can adversely affect the quality of life. Oral trauma from methamphetamine abuse ("meth mouth") and psychiatric disorders such as anorexia nervosa and bulimia nervosa can decimate both teeth and soft tissues. The purpose of this course is to highlight a select group of pathological entities of microbial, autoimmune, and behavioral origin that can cause a wide range of responses for the hard and soft tissues of the oral and maxillofacial complex. Although a complete discussion of all oral pathological issues is beyond the scope of this continuing education course, the participants should develop an appreciation that oral pathology can have diverse origins. Further, this course should serve as a reminder that there are numerous other sources, whether microbial, autoimmune, or behavioral, that can manifest as oral pathology issues. The oral and maxillofacial complex should be viewed as an important component to overall health, and any pathological process may compromise both.
- INTRODUCTION
- ORAL DISEASES OF BACTERIAL ORIGIN
- ORAL DISEASES OF VIRAL ORIGIN
- ORAL DISEASES OF FUNGAL ORIGIN
- MISCELLANEOUS ORAL LESIONS
- ORAL PATHOLOGY OF BEHAVIORAL AND PSYCHIATRIC ORIGIN
- CONSIDERATIONS FOR NON-ENGLISH-PROFICIENT PATIENTS
- CONCLUSION
- Works Cited
- Evidence-Based Practice Recommendations Citations
This course is designed for all dental professionals who care for patients who may have oral pathology.
The purpose of this course is to provide dental professionals with the information necessary to identify, assess, and treat or refer patients with a wide range of conditions of the hard and soft tissues of the oral and maxillofacial complex resulting from pathologic entities of microbial, autoimmune, and behavioral origin.
Upon completion of this course, you should be able to:
- Describe the etiology, pathogenesis, and treatment of oral diseases of bacterial origin.
- Differentiate between herpes simplex virus 1 primary and recurrent infections.
- Review the oral and maxillofacial manifestations of varicella-zoster infections.
- List the common oral and facial opportunistic infections caused by the human immunodeficiency virus (HIV).
- Differentiate among the varied forms of oral candidiasis.
- Cite the differences among various oral presentations of aphthous ulcers.
- List the oral presentations of erosive and non-erosive lichen planus.
- Discuss the oral ramifications of methamphetamine abuse ("meth mouth"), anorexia nervosa, and bulimia nervosa.
Mark J. Szarejko, DDS, FAGD, received his dental degree from the State University of New York at Buffalo in 1985. He received fellowship from the Academy of General Dentistry in 1994.
Contributing faculty, Mark J. Szarejko, DDS, FAGD, has disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
Sarah Campbell
The Director of Development and Academic Affairs has disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
The purpose of NetCE is to provide challenging curricula to assist healthcare professionals to raise their levels of expertise while fulfilling their continuing education requirements, thereby improving the quality of healthcare.
Our contributing faculty members have taken care to ensure that the information and recommendations are accurate and compatible with the standards generally accepted at the time of publication. The publisher disclaims any liability, loss or damage incurred as a consequence, directly or indirectly, of the use and application of any of the contents. Participants are cautioned about the potential risk of using limited knowledge when integrating new techniques into practice.
It is the policy of NetCE not to accept commercial support. Furthermore, commercial interests are prohibited from distributing or providing access to this activity to learners.
Supported browsers for Windows include Microsoft Internet Explorer 9.0 and up, Mozilla Firefox 3.0 and up, Opera 9.0 and up, and Google Chrome. Supported browsers for Macintosh include Safari, Mozilla Firefox 3.0 and up, Opera 9.0 and up, and Google Chrome. Other operating systems and browsers that include complete implementations of ECMAScript edition 3 and CSS 2.0 may work, but are not supported. Supported browsers must utilize the TLS encryption protocol v1.1 or v1.2 in order to connect to pages that require a secured HTTPS connection. TLS v1.0 is not supported.
The role of implicit biases on healthcare outcomes has become a concern, as there is some evidence that implicit biases contribute to health disparities, professionals' attitudes toward and interactions with patients, quality of care, diagnoses, and treatment decisions. This may produce differences in help-seeking, diagnoses, and ultimately treatments and interventions. Implicit biases may also unwittingly produce professional behaviors, attitudes, and interactions that reduce patients' trust and comfort with their provider, leading to earlier termination of visits and/or reduced adherence and follow-up. Disadvantaged groups are marginalized in the healthcare system and vulnerable on multiple levels; health professionals' implicit biases can further exacerbate these existing disadvantages.
Interventions or strategies designed to reduce implicit bias may be categorized as change-based or control-based. Change-based interventions focus on reducing or changing cognitive associations underlying implicit biases. These interventions might include challenging stereotypes. Conversely, control-based interventions involve reducing the effects of the implicit bias on the individual's behaviors. These strategies include increasing awareness of biased thoughts and responses. The two types of interventions are not mutually exclusive and may be used synergistically.
#58663: Oral Pathology Review
Pathology is the discipline of medicine concerned with the nature of disease and the structural and physiologic changes that accompany the disease process. Oral pathology is a specialty of dentistry recognized by the American Dental Association. The spectrum of its study includes diseases that originate in the oral and maxillofacial complex and those that are manifestations of systemic diseases. There is a vast array of pathologic entities that can afflict this region, with consequences ranging from a temporary and minor inconvenience to those with a high degree of morbidity and even death. This course will concentrate on the aspects of oral and maxillofacial pathology that are frequently encountered by the general population and conditions that occur infrequently but can have significant impact on overall patient health.
Diseases of bacterial, fungal, viral, autoimmune, and behavioral origin are the main causes of oral pathology. The etiology, pathogenesis, and treatment options of each disease will be highlighted with a particular emphasis on the impact on oral and systemic health. Upon completion of this course, practitioners should have a sense of appreciation for the multitude of pathogenic agents that contribute to oral disease and for the host response, which can limit the accompanying initial and long-term problems. Oral diseases can have clear effects on overall health, and early intervention for these problems can increase the quality of life and alleviate suffering.
Among the classifications of the varied inflammatory periodontal diseases, acute necrotizing ulcerative gingivitis/periodontitis (ANUGP) uniquely exhibits rapid onset and severe pain and can cause irreversible damage to the interdental papilla and even to the supporting alveolar bone.
Over time, this disease has been known by many names. The most descriptive term, trench mouth, originated as a description of this oral malady that afflicted soldiers confined to the trenches during World War I [1]. The lack of proper oral hygiene, the stress of battle, inadequate nutrition, and cigarette smoking combined to afflict many soldiers with this problem. Improved hygiene and oral health awareness have combined to minimize the occurrence of this disease. However, the emergence of human immunodeficiency virus/acquired immune deficiency syndrome (HIV/AIDS), poor nutrition, and a scarcity of dental care in developing nations have resulted in a resurgence of this acute oral problem.
The classic identifying lesion associated with ANUGP is a painful, necrotic ulceration that originates in the interdental papilla. The lesion is often accompanied by a grey-white pseudomembrane—a necrotic layer of debris. The removal of this pseudomembrane will yield an underlying layer of tissue that bleeds profusely and is extremely sensitive to any environmental stimulus. Anterior teeth are involved more than posterior teeth, with a variance in the number of teeth and surfaces involved [2]. Erythema and edema of the adjacent tissue is extensive, and a bright red margin is peripheral to the lesion. The process begins at the tips of the interdental papilla and can continue to involve the entire interdental gingiva. The necrotic tissue produces a foul odor (fetor oris) that is another diagnostic feature. Elevation in body temperature will occur in some, but not all, patients. Localized lymphadenopathy, increased salivation, and malaise may also accompany these outbreaks [3].
When the disease process extends to include the underlying alveolar bone, the condition progresses to acute necrotizing ulcerative periodontitis. This often occurs after several cycles of exacerbations and remission of the lesions [2]. The gingival lesions associated with this advanced stage are larger and can coalesce to involve several teeth. The alveolar bone involved becomes necrotic, and the resultant loss of osseous support can lead to the loss of teeth. Any patient, but especially those who are immunocompromised, can experience an extension of ANUGP beyond the mucogingival junction into oral mucosa and even into the skin. This condition is known as cancrum oris (noma) and can ultimately involve a gangrenous necrosis of the tissues that may result in death [4]. The process rarely extends to this level as the initial pain caused by ANUGP usually motivates patients to seek professional treatment.
The exact mechanism by which ANUGP develops in patients whose immunocompetence is unchallenged is unknown [5]. Patients with ANUGP have poor oral hygiene, stress, smoking, and poor nutrition as common factors. However, not all patients who share these features develop ANUGP.
Bacteria that are isolated from these lesions are constituents of the normal oral flora. The bacterial species associated with ANUGP are spirochetes such as Treponema vincentii and other species, including Fusobacterium spp. [5]. Also identified are Selenomonas spp. and gram-negative rods such as Prevotella intermedia [6]. An immune system weakened by HIV/AIDS, chemotherapy, radiotherapy, or systemic illness can allow these factors and bacteria to manifest as an opportunistic infection. A lack of proper function of polymorphonuclear (PMN) leukocytes, defective lymphocyte function, and a reduction in immunoglobulin production are features of decreased immunocompetence that can precede the development of ANUGP in susceptible individuals [7]. Stress may result in increased cortisol and catecholamine levels, which may compromise the host immune response and gingival microcirculation [7].
As stated, the acute pain and appearance of the gingival tissues usually prompts patients to seek treatment. Initial treatment is gentle mechanical debridement of the involved teeth along with adjunctive support from local or systemic antibiotic therapy [2,8]. The gingival tissues are extremely sensitive at the initial clinical presentation and usually cannot be manipulated. Analgesic medications and frequent rinses with solutions such as salt water, 3% hydrogen peroxide mixed with equal parts of water, or 0.12% chlorhexidine can serve to rinse away accumulated debris and decrease bacterial mass [9]. After the initial pain has subsided, definitive treatment, such as root planing and curettage, can be completed. Patients should be educated as to the combination of factors that were the basis of this problem. Smokers should be provided information about smoking cessation programs.
The prognosis for patients with ANUGP is variable and depends upon the patient's immunocompetence, prior periodontal condition, and the degree to which the necrosis has affected the gingival and/or osseous tissues. Initial outbreaks usually respond well to empiric therapy, but recurrences of this disease can cause cumulative damage to the periodontal architecture. The remodeling of the gingival and osseous support can leave contours that present a challenge for oral hygiene. When ANUGP affects the alveolar bone, periodontal surgery may be necessary to restore proper anatomic contours. Extractions are necessary if alveolar bone loss is of a magnitude that advanced mobility precludes periodontal intervention.
Osteomyelitis of the jaws (OMJ) begins as an inflammatory process of the medullary bone. Extension of this infection outward to the cortical bone and periosteum leads to destruction and necrosis of bone and osseous remodeling of the affected areas [10]. There are varied classifications for this condition that reflect origin, duration, and the presence or absence of suppuration from the involved area(s).
It is rare, but possible, for a pathogenic organism to cause OMJ via the hematogenous route. However, most cases develop by non-hematogenous means via an extension of a contiguous oral infection [11]. Acute osteomyelitis occurs for 30 days or less, while the chronic form continues for more than 30 days. Osteomyelitis that presents with an absence of intraoral or external suppuration or fistulas in the affected area is categorized as non-suppurative; the presence of these features indicates the suppurative form [11,12].
Most cases of OMJ originate from adjacent periapical or periodontal infections, fractures of the jaw that have inoculated pathogenic organisms deep within the bone, or infections that develop after recent oral surgery [11,13]. Predisposing factors include diseases or medications that cause systemic immunosuppression. Medical procedures such as radiotherapy to the face, head, or neck, which decreases vascularity to the irradiated bone, can also increase the potential for OMJ, especially in the mandible [10,11]. Organisms isolated from suppurative cases can vary according to the stage of the disease and age of the patient. Children 1 to 16 years of age with acute osteomyelitis have had Staphylococcus aureus, Streptococcus pyogenes, and Haemophilus influenzae recovered from cultures. Patients older than 16 years of age have had S. aureus, S. epidermidis, and gram-negative bacilli identified in their cultures. Pathogenic organisms associated with chronic osteomyelitis include S. aureus, S. epidermidis, S. pyogenes, enterococci, gram-negative bacilli, and anaerobes [10]. The amount and virulence of these organisms can vary, and other bacterial species may be involved.
The acute stage of OMJ begins when a pathologic stimulus causes an inflammatory process within the medullary bone. The increased blood flow and capillary permeability, with a commensurate influx of granulocytes, presents clinically with a minimum of swelling and an absence of suppuration [2]. Progression beyond the acute stage features the development of an abscess, the growth of which causes an increase in the intramedullary pressure. The perfusion of blood to the affected area decreases and ischemia results. The extension of the infection progresses through the cortical bone and periosteum with the development of oral and cutaneous fistulas [14]. Necrotic segments of bone called sequestra develop secondary to oxygen and nutrient deprivation caused by the ischemia. Because the blood supply to the maxillary arch is more copious than that to the mandibular arch, the mandible is more susceptible to the development of osteomyelitis.
Antibiotic therapy is the usual mode of treatment for the initial onset of acute osteomyelitis. A culture from the affected area should be developed to determine the antibiotic susceptibility of the offending pathogen. Antimicrobial treatment for OMJ is of a much longer duration than for that of the typical odontogenic infection. Patients may require the placement of a peripherally inserted central catheter (PICC) line, through which antibiotics may be systemically administered for several weeks. Bacterial resistance to antibiotics can impair healing and require a change to one or more additional medications [11].
When acute non-suppurative osteomyelitis develops into chronic suppurative osteomyelitis (CSO), the treatment usually involves surgery to remove bony sequestra that were too large for resorption by osteoclasts [15]. If large sections of necrotic bone are removed, bone and tissue grafts may be necessary to restore some of the soft tissue and osseous foundation. Teeth may be lost when the underlying bone becomes necrotic. The fabrication and use of partial or complete dentures can be a challenge even after reconstructive surgery. Patients with CSO may require surgical drains placed into the affected area for an extended interval to expedite the removal of suppurative material. Patients who have lost large segments of bone are at increased risk for pathologic fractures of the jaw.
Osteomyelitis of the jaws can be a challenging disease to treat. Some forms can be refractory to antibiotic and surgical treatment, with a cyclical course of remissions followed by possible recurrences. This is a disease that can have significant morbidity and occasionally serve as the origin of a fatal septicemia. Prompt initial treatment, continuing pharmacotherapeutic therapy, and surgery where needed are necessary to manage the disease properly.
Few acute diseases demonstrate the potentially fatal consequences of odontogenic infections as Ludwig angina, which was first described in 1836 by Wilhelm Friedrich von Ludwig [16]. By definition, this is a rapidly advancing cellulitis that features bilateral involvement of the submandibular and sublingual fascial spaces posteriorly and the submental fascial space anteriorly [17,18]. The origin of approximately 90% of Ludwig angina cases is odontogenic; usually, carious and/or periodontally involved lower molars are the source of the initial oral infection [19]. The clinical presentation is of diffuse cellulitis. Infection and edema from bilateral involvement of the submandibular space causes an indurated swelling of both sides of the neck above the hyoid bone [18]. When bilateral involvement of the sublingual spaces occurs, the posterior aspect of the tongue becomes enlarged. Edema and swelling in the floor of the mouth cause the tongue to be elevated above the level of the mandibular teeth [18,20]. When this occurs, swallowing saliva is difficult for patients and drooling becomes common. Speaking may also be difficult as basic movements of the tongue are severely restricted [18]. Despite the swelling, intraoral and facial suppuration is rarely seen. The areas involved in Ludwig angina can extend to the clavicles and even to the superior aspect of the chest. The skin overlying these sites can appear erythematous. The most critical concern for patients afflicted with Ludwig angina is airway compromise. Death is more common from airway obstruction than from systemic sepsis [21]. Many patients are febrile upon clinical presentation.
As noted, carious teeth that have become necrotic and abscessed and teeth with periodontal infection are responsible for the vast majority of Ludwig angina cases. Other sources include respiratory infections, traumatic infections, traumatic injury to the soft tissue, and mandibular fractures. The bacteria associated with these infections can vary [18,22]. No individual bacterial species has been identified as the sole etiologic agent of this disease. However, the more common bacterial species that have been isolated from the cultures from affected areas include both aerobic and anaerobic species, including streptococci, staphylococci, pseudomonas, and anaerobes (e.g., Bacteroides spp. and Peptostreptococcus spp.) among many others [2,19]. The unique oral flora of the host and the health of the host, especially as they relate to immunocompetence, are among the factors that can influence the bacterial composition and virulence of the infection.
It is imperative to understand that while odontogenic pathology is the usual origin of Ludwig angina, the condition constitutes a medical emergency. When this condition is suspected, the patient should be immediately referred to the emergency department, as swelling and edema around the trachea can obstruct and ultimately close a patent airway. Treatment for Ludwig angina combines surgical drainage of the affected fascial spaces and aggressive intravenous antibiotic therapy. It is often necessary to perform a tracheostomy to maintain a patent airway [19]. In these cases, purulent discharge can be present but may be embedded deep within the tissue such that surgical incisions usually must be made into the submental, sublingual, and submandibular spaces. A serosanguineous fluid is usually the clinical appearance of this exudate, from which bacterial cultures can be made [23]. Empiric antibiotic therapy is begun upon admission and may be changed pending the results of the cultures. Surgical drains are sutured into place to allow for continuous drainage until the infection has resolved, usually several days. Most patients remain in the intensive care unit during this period. The teeth from which the infection originated may be extracted at the same time as the surgical drains are placed. If not, extractions, periodontal therapy, or endodontic treatment should be performed when the infection has resolved. Most patients experience a full recovery.
When carious and/or periodontally involved teeth are the origin of Ludwig angina, patients should be educated as to the cause and effect relationship between the two. Problematic teeth that could not be extracted during the hospital admission should be extracted or restored as soon as safely possible. A lack of attention to this detail could allow for a relapse. Prompt emergency medical intervention, appropriate antibiotic therapy, surgery to drain and to maintain drainage of the affected spaces, and the extraction or restoration of the involved teeth allow for a good prognosis for most patients. Even small delays in the initiation of treatment can be the difference between life and death.
The oral and maxillofacial complex is subject to viral infections, the extent of which can range from site-specific to regional involvement, with durations being limited or for extended periods of time. The DNA-containing herpesvirus group is a viral family with well-known pathologic oral manifestations [24].
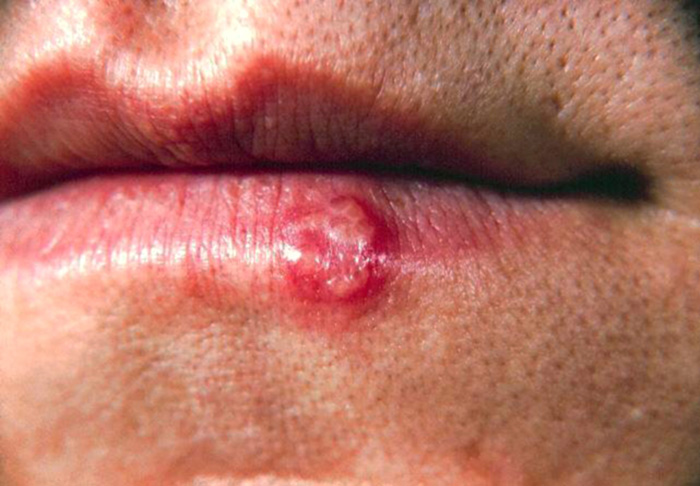
The initial infection with herpes simplex virus type 1 (HSV-1) can occur at any age, but it is most frequently seen in infants and young children [25]. Intraoral lesions are most prominent in this initial presentation, called primary herpetic gingivostomatitis. Occasionally, the skin around the lips also becomes infected. The oral lesions of primary herpetic gingivostomatitis affect both the keratinized and non-keratinized oral mucosa [26]. Vesicles may form but can rupture quickly to produce non-specific ulcers. The initial outbreak can be very painful, with accompanying fever, lymphadenopathy, and malaise. Swallowing can be difficult when palatal lesions are involved [25]. Palliative support with analgesics, antipyretics, and nutritional support usually allow for recovery within two weeks. The disappearance of these initial lesions is not equated with a complete eradication of this disease. Instead, the virus migrates to a regional nerve ganglion. The trigeminal ganglion is the usual reservoir for HSV-1, where it can lie dormant for years. Reactivation of HSV-1 can occur at any time with exposure to certain stimuli; stress, illness, sunlight, and immunosuppression are considered among the many factors that can end viral dormancy [25,27]. As opposed to the primary herpetic gingivostomatitis, recurrent lesions occur most often on the junction of the lips and skin and are known as recurrent herpes labialis. These lesions begin with formation of minute vesicles that coalesce to form several large lesions. Most patients experience a prodromal sensation characterized by itching, burning, tingling, or tightness of the skin in the area that will feature the herpetic outbreak [25]. The lesions of herpes labialis rupture, form scabs, and ultimately heal within two weeks without scar formation (Image 1). Patients should be advised not to touch these recurrent lesions as the vesicles contain a multitude of viral particles, and upon inoculation into the skin of a fingertip, the condition known as herpetic whitlow can develop. The virus is also highly contagious during outbreaks, and patients should take steps to prevent infecting close contacts. The affected area can be subjected to a lifetime of recurrent painful herpetic outbreaks.
Recurrent intraoral herpes simplex occurs infrequently. These recurrent lesions affect only the keratinized mucosa of the hard palate and attached gingiva [25,28]. As a result, intraoral lesions rarely have intact vesicles as seen with herpes labialis. Lesions usually heal within a two-week period.
Treatment for outbreaks of herpes labialis can be categorized as either preventative or palliative. Because this is a disease with recurrences at varying intervals, nothing can prevent all future outbreaks. However, some risk factors, such as exposure to sunlight and stress, can be modified [27]. Lip balms and sun blocks can be applied to the mucocutaneous junction near the lips to reduce the amount of exposure to sunlight and minimize, but not eliminate, the chance of a recurrent outbreak. Antiviral ointments or pills, such as acyclovir, can be taken or applied to emerging herpetic vesicles to decrease the duration or frequency of outbreaks [27]. Ointments should be applied with an applicator and never directly by the fingers to avoid the potential of herpetic inoculation of the fingers. Patients who wear complete or partial dentures should use care while inserting and removing these prostheses during herpetic recurrences. Vesicles with live viral particles can be stretched and ruptured and further inoculate the adjacent tissue or fingers. Patients with recurrent intraoral herpes may be unable to wear their prostheses. Fortunately, both herpes labialis and recurrent oral herpes heal without scarring after a period of approximately 14 days. Patients who are immunosuppressed will have a longer duration of herpetic recurrences and more tissue surface area will be involved. A higher degree of morbidity and even death can occur when a compromised immune system is challenged by this virus.
The varicella zoster virus (VZV) has a pathogenesis that is similar to that of HSV-1. The initial infection caused by this virus is chickenpox and is most frequently seen in children. After this infection resolves, VZV migrates to dorsal root and cranial nerve ganglia, where it can lie dormant forever or until its reactivation, the clinical presentation of which is known as shingles [4,29]. Although less common than cutaneous lesions, both chickenpox and shingles can have oral manifestations.
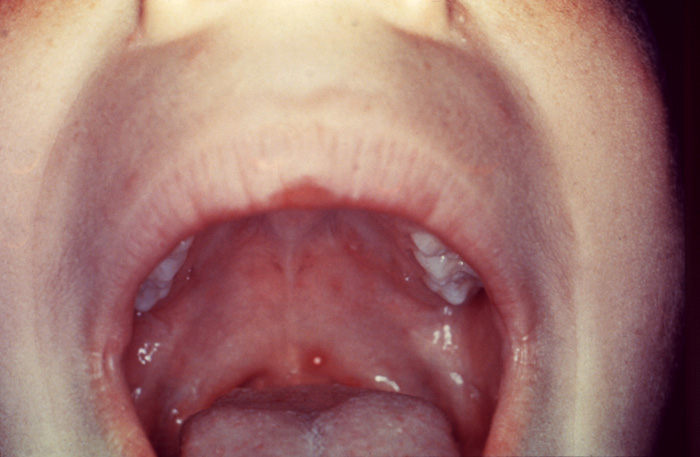
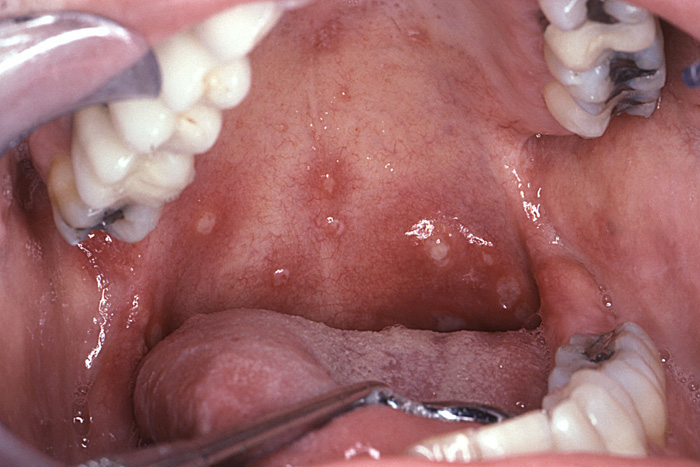
The initial infection of chickenpox is a highly contagious disease that is spread through direct contact of infected secretions such as respiratory droplets. A rash with macular, vesicular, and pustular stages is seen predominantly on the head, neck, and trunk. These lesions resolve approximately two weeks after their emergence [2]. Oral lesions are less common but can occur and feature vesicles that rupture quickly and form small, non-specific ulcers (Image 2). When these occur on the soft palate, swallowing and eating can be painful. Systemic analgesics and topical anesthetic preparations can palliate some of these symptoms. The primary infection of chickenpox resolves uneventfully for most patients. However, immunocompromised children and adults who contract this illness for the first time can have severe systemic involvement and even succumb from its complications.
Many years after the resolution of the initial outbreak of chickenpox, the reactivation of VZV causes the development of shingles. Similar to chickenpox, the head and thorax are the most common areas of involvement. The rash is similar in appearance to that of chickenpox, but the stabbing and radiating pain that accompanies these outbreaks distinguishes between these two diseases.
Shingles primarily affects older adults and the immunocompromised but can occur in any age group. When present, the oral and maxillofacial manifestations of shingles can be severe and cause intense, prolonged pain. Oral manifestations of shingles commonly involve the ophthalmic, maxillary, and mandibular division of the trigeminal nerve. Unilateral distribution is a classic feature, as the cutaneous and intraoral lesions of shingles do not cross midline [30,31]. Vesicles form and rupture to form painful ulcers in the affected areas, which can take two weeks or longer to heal. The pain that accompanies the intraoral lesions can make eating and swallowing extremely difficult. Topical anesthetic solutions may offer some relief from this pain. Systemic anti-inflammatory medications and narcotic analgesics are often needed to subdue the associated pain. Cutaneous lesions of the maxillofacial region are also very painful, and involvement of the ophthalmic division of the trigeminal nerve can produce corneal ulceration and resultant loss of vision [32]. Viral involvement of the facial nerve can cause Ramsay Hunt syndrome. This features cutaneous vesicle eruptions of the ear and facial nerve dysfunction with weakness and paralysis of the affected side [29,33]. VZV can even cause osteonecrosis of the alveolar bone, possibly the result of vasoconstriction with subsequent ischemia to the involved area. Tooth loss can also occur [34].
Although the oral and cutaneous lesions heal, many patients with shingles develop postherpetic neuralgia. This debilitating pain syndrome originates in the area affected by the vesicular rash and can persist for years after the area has healed. Approximately 50% of patients older than 60 years of age with shingles experience this problem [29]. Because many of these patients will have narcotic analgesics and/or antidepressant medications prescribed, healthcare professionals should use caution in dispensing any additional medication that could have harmful interactions.
For patients experiencing a shingles outbreak, oral antiviral medications such as acyclovir (Zovirax), valacyclovir (Valtrex), and famciclovir (Famvir) may be used; these medications are most effective if their administration begins within 72 hours of the initial outbreak [35,36]. Narcotic and non-narcotic analgesics can decrease the pain. A tapering dose of prednisolone may decrease the inflammation and its accompanying pain. While some patients heal from this disease and have no postherpetic neuralgia or recurrences, other patients are not so fortunate. Shingles can recur at any time, and for those who are immunocompromised, the widespread involvement can produce systemic viremia with a high degree of morbidity and potential mortality.
One of the most promising treatments for VZV is vaccination. In 1995, the chickenpox vaccine was approved for administration as part of the childhood immunization schedule [37]. Ten years later, a shingles vaccine was approved for use in patients 60 years of age and older [38,39]. In 2018, the recombinant zoster vaccine was added as the preferred vaccine for adults 50 years of age and older [40,41]. The impact of vaccination against varicella in childhood on the potential to develop shingles later in life has not yet been studied. Patients should consult with their physician to determine if the shingles vaccine may be appropriate for their unique health profile.
Another treatment for VZV, the varicella zoster immune globulin (VariZIG), is approved for administration to high-risk individuals (e.g., immunocompromised children and adults, infants) within 10 days (ideally within 4 days) of exposure to chickenpox [42,43]. Prevention of shingles would spare many patients an illness whose debilitating pain, chance for recurrence, and potential for postherpetic neuralgia adversely affect the quality of life.
The global epidemic of HIV/AIDS has changed the medical, political, and social spheres as few diseases have ever done. Since the discovery of HIV in 1984, the virus has infected millions of people throughout the world. In 2019, 36,801 people received an HIV diagnosis in the United States and dependent areas. The annual number of new diagnoses decreased 9% between 2015 and 2019 [44]. In the United States, the Centers for Disease Control and Prevention (CDC) estimates that, through 2019, the cumulative number of patients living with HIV is approximately 1.2 million. In 2019, more than 5,000 people died with HIV/AIDS as the underlying cause of death [45].
HIV selectively binds to CD4+ lymphocytes and, in the process of viral replication, destroys these cells critical to immune function and host defense. Additionally, dysfunction of B-lymphocytes, macrophages, and neutrophils occurs [46]. This process may take years before the increase in the HIV viral load causes a commensurate decrease in immune cells. At this point, opportunistic infections can occur, many of which have oral manifestations that fulfill the criteria as established by the CDC for AIDS-defining conditions.
Candida albicans is a normal inhabitant of the oral microflora and a subsequent section of this course will discuss the varied clinical presentations of this fungal organism in more detail. This is the most common oral fungal infection in patients with and without HIV/AIDS [47]. Although this condition will be discussed in detail later in this course, there are aspects of its clinical presentation and implications unique to patients with HIV/AIDS. When a candidal infection affects patients without immune system compromise, the typical presentation is that of pseudomembranous candidiasis. Regional and systemic dissemination is always possible, but confinement is usually within the oral cavity. Immunocompromised patients, such as those with HIV, have a high occurrence of erythematous candidiasis rather than the pseudomembranous form. This features an erythematous lesion that does not have a white to yellow-white covering. Either form is capable of extension into the esophagus and can induce systemic fungal involvement. Approximately 90% of patients with HIV experience clinically evident candidiasis, and its appearance indicates the progressive deterioration of the immune system. Patients who experience this fungal disease usually have a prominent indicator for progression from HIV infection to AIDS [48]. Candidal infections in patients with HIV can be refractory to conventional oral rinses such as nystatin or antifungal lozenges such as clotrimazole. Oral systemic antifungal medications such as ketoconazole, voriconazole, ravuconazole, fluconazole, or posaconazole may be necessary. Caution is required when prescribing these medications as they have multiple drug interactions [49]. The recurrence rate of oral candidiasis remains high in patients with HIV, and some fungal organisms have acquired resistance to these medications.
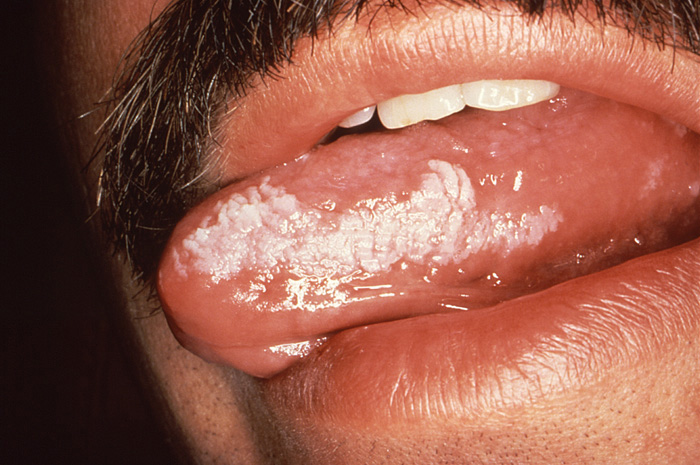
Oral hairy leukoplakia features bilateral areas of epithelial hyperplasia on the lateral and ventral surface of the tongue found upon oral examination (Image 3). Corrugated in texture, these lesions are adherent to the underlying tissue and are asymptomatic for the patient. The emergence of oral hairy leukoplakia in patients with HIV is caused by the reactivation of the Epstein-Barr virus as the efficacy of the immune system continues to decrease. While oral antiviral medication can resolve these lesions, they have a tendency to recur upon the cessation of therapy. Occasionally, the size of these lesions can be large enough to be traumatized during occlusion. Because these lesions appear when the CD4+ count has diminished to levels of approximately 300/mcL, this reflects continued deterioration of the immune system and a less favorable overall prognosis for the patient [50].
Gingivitis and periodontal disease are common oral problems for many patients. As the immune system of patients with HIV/AIDS becomes progressively weakened, pathogenic bacteria associated with gingivitis and periodontal disease can cause rapidly progressing and generalized destructive involvement of the periodontium.
An early manifestation of the gingival involvement is that of linear gingival erythema. Formerly called HIV-associated gingivitis, this problem has a unique clinical presentation in patients with HIV infection. This condition features an intense zone of inflammation that is finely demarcated and involves the free and attached gingiva and extends into the interdental papilla [51]. The tendency for gingival bleeding is increased. These areas reflect minimal or no resolution despite excellent oral hygiene and conventional periodontic treatment. Consideration has been given that this could represent an unusual distribution and chronic infection with C. albicans[48].
Some patients will progress to varying forms of periodontal disease, such as necrotizing gingivitis and periodontitis. More advanced stages feature aggressive progression of the pathogenic bacteria that can cause loss of tissue attachment and destruction of the supporting alveolar bone. Occasionally, the bacteria can extend into the alveolar and buccal mucosa and cause a necrotizing stomatitis [24]. The damage to the alveolar bone can be of such magnitude that extractions of the involved teeth are the only course of definitive treatment.
Patients with HIV/AIDS should maintain a prophylaxis schedule that reflects the extent of their periodontal involvement to decrease the progression of these destructive periodontal patterns. Because the inflammatory lesions of periodontal disease can provide systemic access for multiple pathogenic organisms, it is essential that periodontally involved areas are treated promptly. Teeth with a poor periodontal prognosis should be extracted to eliminate a source of local, regional, and systemic infection. Before surgical procedures are initiated, recent laboratory values, including updated CD4+ lymphocyte, neutrophil, and platelet levels, should be obtained. Some patients with HIV/AIDS will develop platelet dysfunction problems such as thrombocytopenia [52]. If the platelet level is low, problems with hemostasis can occur during and after basic periodontal procedures and especially oral surgery. Thus, patients that have progressed to AIDS may require surgical procedures to be completed in a hospital setting. Antibiotic prophylaxis is not an absolute requirement when surgical procedures are anticipated for patients with HIV/AIDS. However, patients should be referred to a specialist if the stage of this disease has left the patient with advanced immunosuppression.
Once a rare malignancy that most frequently occurred in elderly men of the Mediterranean region and certain regions of Africa, Kaposi sarcoma is now the most common malignant disease in patients with HIV/AIDS [53]. In patients without HIV, Kaposi sarcoma grows slowly and is usually seen as cutaneous lesions on the extremities. The lesions associated with HIV/AIDS tend to grow rapidly and can disseminate to cause multiple organ involvement. Lesions may arise within the endothelium of small blood vessels and cause cutaneous and intraoral lesions. Kaposi sarcoma was the first pathologic entity to be identified as an AIDS-defining illness [54,55].
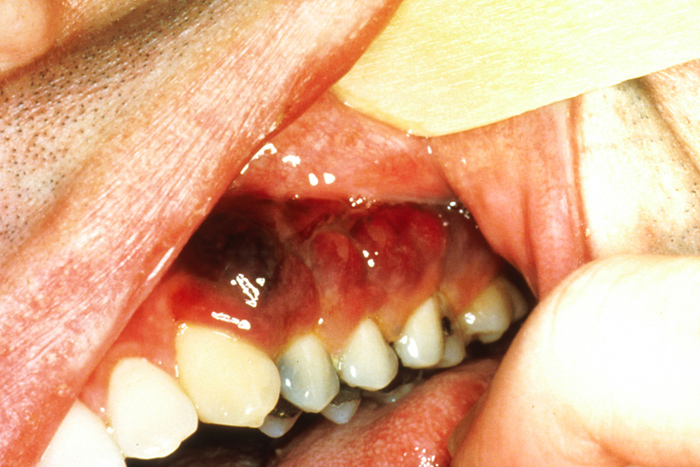
Although cutaneous lesions are more common, oral lesions of Kaposi sarcoma affect approximately one-third of patients [56]. Initial oral lesions are asymptomatic, and their red-to-violet color can resemble ecchymosis within the mucosal and gingival tissues (Image 4). An incisional biopsy must be completed to establish a definitive diagnosis. The most common intraoral site affected by Kaposi sarcoma is the hard palate [57]. However, lesions have been seen on all other intraoral sites. As the condition progresses, the flat appearance of the early lesions changes to a more raised appearance (in the case of a single lesion) or multiple lobules of a larger lesion. The color can vary and may be a composite of red, blue, purple, or brown. Enlargement of the lesions can cause difficulty in eating, speaking, and swallowing [57]. Partial or complete dentures may be difficult to fit and/or wear as these lesions will change tissue topography. If surgery is performed to remove these lesions, alterations in tissue contour would require that a new prosthesis be made. New Kaposi sarcoma lesions perpetuate this cycle, and patients may be unable to wear a prosthesis.
The exact cause of Kaposi sarcoma lesions has been suggestive of a virus that exerts its influence as the immunosuppression of these patients increases [57]. Viral DNA sequences of human herpesvirus 8 have been found in Kaposi sarcoma lesions in both HIV-seropositive and HIV-seronegative patients [57]. The role that this virus has in the development of Kaposi sarcoma is not certain. However, it appears that while the human herpesvirus 8 is required for the development of these lesions, it is not the sole etiologic agent [58].
Treatment and Prognosis
The use of a combination of medications for patients with HIV known as combination antiretroviral therapy (cART) aims to decrease viral replication and increase CD4+ levels. As such, opportunistic oral infections such as Kaposi sarcoma have a decreased incidence in patients that comply with this regimen [59]. Since the inception of cART, the treatment goals in Kaposi sarcoma have changed from short-term palliation to long-term remission and control [59]. When the size of the lesion has increased to become an obstruction to proper oral function or a cosmetic problem, varied techniques can allow for it to be removed or reduced in size. Radiation therapy is the most widely used and effective local therapy and can palliate bleeding, pain, or unsightly lesions. Responses occur in 80% to 90% of patients [60]. In patients with widespread skin involvement, extended-field electron beam radiation therapy is effective in controlling the disease [60]. Surgical removal or reduction can provide some relief for patients with small superficial lesions, but lesions commonly recur [60]. Direct injections of vinblastine into these lesions can cause sclerosis of the malignancy and palliate the associated symptoms. Responses occur in 60% to 90% of patients [60]. Side effects include changes in pigmentation, swelling, blistering, ulceration, and pain on injection [60]. Unfortunately, original lesions can recur and new Kaposi sarcoma lesions can develop. The overall prognosis for patients with HIV-associated Kaposi sarcoma is usually poor as these lesions emerge at time of increasing viral load and decreasing immunocompetence [59].
The variation in the response of each patient to HIV infection and to the development of AIDS is considerable. Compliance with recommended cART can also vary. Prescribed medications can have numerous side effects, which may discourage patients from continuing with their use. Thus, the disease process can occur more rapidly in some patients as compared to others. In a similar fashion, the oral manifestations of HIV/AIDS can differ widely among this patient population. All patients with HIV infection should have a comprehensive dental exam as early as possible in the course of their disease. Teeth with advanced caries or periodontal problems should be restored to proper function or extracted, as the odontogenic infections they can cause are more difficult to treat as systemic immunosuppression increases. Frequent recall appointments can promote oral and overall health and help to maintain a better quality of life.
Varied species of candidal fungal organisms normally inhabit the oral microflora. A healthy host and competition with numerous other oral microbes usually prevent the most common fungal organism, C. albicans, from causing oropharyngeal candidiasis. However, altered immune status, systemic antibiotic therapy, varied systemic diseases, chemotherapy, and radiotherapy can change the oral and pharyngeal environment to favor the proliferation of C. albicans and other fungal species. This leads to the development of the opportunistic infection oropharyngeal candidiasis.
The clinical presentation of oropharyngeal candidiasis can have four different appearances, the most common of which is pseudomembranous candidiasis [61]. The lesions typical of pseudomembranous candidiasis consist of white-to-yellow flecks or plaques with a smooth and slightly raised appearance. The surface of these lesions can be removed easily, leaving an erythematous base that may bleed. These plaques occur on any mucosal surface.
Erythematous (atrophic) candidiasis features red patches that can also occur on any mucosal surface [61,62]. When the tongue is affected by erythematous candidiasis, loss of the papillae can result in a burning sensation [61]. The plaques of hyperplastic (chronic) candidiasis have an appearance similar to those of pseudomembranous candidiasis. The distinction is that the lesions of hyperplastic candidiasis adhere to the mucosal surface and cannot be wiped away [61]. Pigments from food, beverages, and tobacco can also cause the plaques of hyperplastic candidiasis to darken over time.
The fourth presentation of candidal infections is that of angular cheilitis, often induced by denture wearing. This presents as cracks or fissures that radiate from the corners of the mouth and extend onto the adjacent skin [61]. Saliva laden with organisms from an intraoral fungal infection can be the origin of angular cheilitis. This is a mixed fungal and bacterial infection, with S. aureus as a frequent bacterial component [49]. A thin pseudomembrane can cover the fissures associated with angular cheilitis. Normal movement of the lips can easily disrupt this involved area and cause a cycle of bleeding and scabbing, which will delay the healing.
There are several factors that can result in C. albicans becoming an oral pathogen and manifesting in varied clinical presentations. Diseases such as HIV/AIDS that decrease the normal immune response allow a normal oral inhabitant such as C. albicans to flourish and can cause not only oropharyngeal candidiasis, but also esophageal candidiasis. Among patients who have advanced to AIDS, 90% will experience this fungal pathology [63]. Patients who undergo chemotherapy for systemic malignancies develop problems with granular and agranular leukocytes, both of which are essential components of a healthy immune system, and may subsequently present with symptoms of candidiasis [61].
Oropharyngeal candidiasis can develop quickly in immunocompromised patients and pose a serious problem if organisms are disseminated systemically. Because these patients may also be experiencing mucositis related to chemotherapy, the presence of denuded mucosal surfaces can facilitate systemic access for fungal organisms [61]. Systemic fungal infections that develop in this fashion have a fatality rate that is twice that of similarly disseminated bacterial infections [64].
Decreases in salivary production from medical procedures, such as radiotherapy for oral cancer, or autoimmune diseases, such as Sjögren syndrome, can cause a change in the oral environment that is favorable to the development of candidiasis. Both of these possible causes will result in long-term problems as radiation-induced damage to salivary glands is irreversible and most autoimmune disorders, including Sjögren syndrome, are incurable diseases. Saliva contains immunoglobulins that promote the proper function of B-lymphocytes, a component of the immunologic response toward C. albicans[65]. When the lubricating and cleansing action of the saliva are diminished, microorganism adherence to the mucosal surfaces is enhanced. Increased exposure time and decreased immune function allow for the development of opportunistic infections such as candidiasis. This is a problem that will resolve for patients undergoing chemotherapy after their regimen ends and the formed elements of human blood return to their appropriate levels.
Medications can be another source of changes to the oral environment [61]. Antibiotics such as amoxicillin or penicillin can alter the composition of the oral microflora. Because these medications are bactericidal, they will decrease the number of bacteria that would normally compete with C. albicans for the available nutrient supply. This decrease in competitive inhibition can allow for candidiasis to develop in susceptible individuals. Fungal infections triggered by antibiotic use usually resolve with the cessation of antibiotic therapy but can recur if the same or similar antibiotic regimen is used again. In addition, numerous medications can cause a decrease in salivary flow, which may also contribute to fungal overgrowth.
Medications used to decrease the risk of rejection of transplanted organs, such as azathioprine, decrease the function of T-lymphocytes. Similarly, systemic corticosteroids such as prednisone and prednisolone, which are used to treat a variety of systemic ailments, can alter the appropriate function of T-lymphocytes [61,66]. The diminished immune response can encourage the development of opportunistic infections such as candidiasis.
Patients who wear partial or complete dentures or orthodontic appliances can be subjected to an increased risk of candidiasis on the tissue-bearing surfaces of these appliances, known as denture-induced stomatitis [61]. This condition features an erythematous area below the outline of the prosthesis. Microscopic porosities within the acrylic can harbor fungal organisms and facilitate the development of this form of candidiasis. This most frequently occurs beneath upper dentures but can occur under any prosthesis.
Most episodes of oropharyngeal candidiasis are of short duration and are easily treated with antifungal medications. Nystatin, available as a swish-and-swallow regimen or as a cream applied to the tissue surface of a prosthesis, is an effective fungicidal medication for treatment of mild disease. Nystatin combined with triamcinolone as an ointment is the choice for angular cheilitis. Some medications, such as clotrimazole, are available as a lozenge formulation. These lozenges are to be dissolved slowly five times per day for 7 to 14 days [66,67]. Patients who have salivary hypofunction problems will not benefit from medications delivered via lozenge. Patients with prosthetic appliances should refrain from wearing them when medicated lozenges are being used. Moderate-to-severe disease should be treated with oral fluconazole at a dose of 100–200 mg daily for 7 to 14 days [66,67]. Treatment of denture-induced stomatitis begins with patient education. Patients should be instructed that dentures are not meant to be worn 24 hours per day and that, upon removal, the entire denture, including the tissue surface and the oral tissues that form the prosthetic foundation, should be cleaned thoroughly. When a patient presents with an existing case of denture-induced stomatitis, both the tissue and the prosthesis should be treated. Antifungal rinses such as nystatin can treat the Candida-laden tissue surface when the denture is removed. The acrylic surface of the prosthesis should also receive antifungal treatment to kill the fungal organisms that reside within the acrylic porosities; this should prevent a cycle of re-inoculation. An antifungal cream can be placed inside the cleansed denture as would a denture adhesive. Resistant candidal infections may require the use of systemic antifungal medications [67].
If the initial antifungal therapy does not eradicate the infection in patients who have been compliant with the required regimen, referral to another clinician or specialist may be needed. Resistance to the antifungal medication is a concern, but the immunocompetence of the patient is another. Systemic antifungal medications may be necessary if the initial occurrence of candidiasis does not resolve.
Medications with side effects that include immunosuppression and/or hyposalivation and systemic diseases that compromise the efficacy of the immune system subject patients to recurrent and more virulent outbreaks of candidiasis. When necessary, systemic medications such as ketoconazole, fluconazole, and itraconazole may be delivered in an intravenous form for fungal infections that have not resolved or for those that have systemic involvement [67]. The prognosis for patients with immunocompromising medical issues is variable. Most patients will recover but are susceptible to recurrent fungal infections. Unfortunately, some patients will succumb when systemic fungal involvement overwhelms the capability of a compromised immune system.
Recurrent aphthous stomatitis, ulcerative lesions also known as recurrent aphthous ulcers or more commonly as canker sores, features painful ulcerative lesions that generally heal without scarring in immunocompetent patients but can recur at any time (Image 5). The size is a demarcation for the categorization of these lesions. Minor aphthae are less than 1 cm at the greatest diameter; major aphthae exceed this dimension. Each type of lesion only involves nonkeratinized tissues, such as the buccal and labial mucosa, the soft palate, and the surface layer of the floor of the mouth. Movement of the mucosa affected by lesions of either size is painful and can interfere with speaking, eating, and swallowing. Major aphthae tend to have irregular shapes and borders and ulcers with more depth. The numbers of both can vary, although solitary lesions are uncommon. Lesions of aphthous stomatitis feature a shallow yellow base and an intense erythematous halo peripherally.
The incidence of these lesions varies from 20% to 60% and can reflect the actual population studied. Many patients develop the condition during the first two decades of life [68]. There has yet to be a bacterial, viral, or fungal etiology that can be attributed to the origin of these lesions. A genetic predisposition to the occurrence is possible. When both parents have a history of these lesions, there is a 90% chance that their children will be similarly affected [69]. Common predisposing factors include physical and emotional stress and sensitivity to ingredients in toothpaste and/or mouthwash, such as sodium lauryl sulfate and alcohol [70,71]. Tissue manipulation during dental or medical procedures, during which the oral tissues are stretched or manipulated by instruments, is a source of local trauma that can cause the development of these ulcers. Nutritional deficiencies in folic acid, iron, and vitamin B12 are other potential etiologies of these lesions [71]. Systemic chemotherapy and radiotherapy targeting malignant lesions of the oropharynx can cause the development of overlapping aphthous ulcers of considerable size that can serve as areas of systemic access of pathogenic organisms. Occasionally, these ulcers occur as part of a disease such as Behçet syndrome. In patients with Behçet syndrome, major aphthae are more frequent than minor aphthae and occur simultaneously on the nonkeratinized oral mucosa, uvula, and genitals [4,71]. Sensitivity to certain medications, including nonsteroidal anti-inflammatory drugs, angiotensin-converting enzyme inhibitors, and bisphosphonates, may produce an aphthous-like tissue reaction. Given the multitude of medications available and potential untoward effects that can occur in the patients who take them, this could be a potential issue for numerous medications.
The lesions of recurrent aphthous stomatitis usually revolve spontaneously. Minor aphthae resolve within 7 to 14 days from onset with no residual scarring. Major aphthae can take three weeks or longer to heal and usually do so with scar formation. These healing periods can be prolonged in patients with HIV/AIDS and those with systemic illnesses such as diabetes. Antibiotics are only needed if the involved areas become secondarily infected. Treatment is usually designed to avoid contact with these lesions as a traumatic incident will exacerbate the pain and prolong healing. Palliation of the symptoms associated with these lesions consists of topical anesthetic preparations and systemic analgesics, as needed. A diet of food and beverages that minimizes both acidity and extreme temperatures and avoids foods with sharp edges will facilitate proper nutritional support and minimize physical trauma to the affected areas [71]. Patients with removable prostheses or removable orthodontic retainers may refrain from using them if an aphthous ulcer is adjacent to their extensions.
As the name would suggest, these lesions have periodic recurrences. Because more serious lesions such as squamous cell carcinoma can appear in a multitude of different forms, patients should be advised that lesions that do not heal should be re-evaluated for a biopsy.
Lichen planus is a chronic autoimmune condition with cutaneous and oral manifestations [72]. Although the actual prevalence of the condition in the United States is unknown, lichen planus is estimated to affect approximately 1% to 3% of the adult population [72]. The classic presentation of lichen planus lesions on the oral mucosa are fine, well-demarcated, adherent white lines called Wickham striae that can involve varying amounts of the posterior buccal mucosa. Occasionally, lichen planus may involve the dorsum of the tongue and appear as adherent white plaques on the anterior two-thirds of the tongue. These forms of oral lichen planus are usually asymptomatic and are often discovered during a clinical dental or medical examination. However, exacerbations from this asymptomatic state to erosive lichen planus can occur. Patients with erosive oral lichen planus present with symptoms that range from minimal discomfort to a burning sensation [71]. The buccal mucosa, tongue, and gingiva can be involved in these outbreaks. The irregularly-shaped lesions of erosive lichen planus can involve large areas of tissue and appear as patchy white areas interspersed with erythematous zones of inflamed or denuded tissue. Secondary infection by C. albicans can increase discomfort and make the diagnosis and treatment of the involved areas a challenge.
The stimulus for the autoimmune origin of this disease remains unknown [72]. However, a similar oral pathologic entity called a lichenoid lesion has a more definitive origin. While these lesions share the appearance of white lines or striae with classic lichen planus, their distribution does not feature bilateral involvement. Numerous medications and repetitive tissue trauma are considered the leading causes of lichenoid lesions. Unlike a classic allergic reaction to medication, lichenoid lesions caused by a given medication can take months to develop. If these lesions develop when a patient is taking multiple medications, it can be difficult to identify the offending medication(s) [72]. However, after the appropriate medication is identified and halted, the lichenoid lesion can resolve over time [73].
Oral lichen planus is considered an incurable chronic disease [72]. Some patients will have extended symptom-free periods during which they require a minimum of palliative relief. Others may have exacerbations of pain, particularly with erosive lichen planus. For these patients, systemic analgesics and systemic steroids can alleviate the associated pain and inflammation. Antifungal medications may be necessary as an adjunctive medication if secondary infection with C. albicans is suspected.
All patients with oral lichen planus should have the involved areas evaluated at recall appointments. There is a low but present risk that a malignancy such as a squamous cell carcinoma will develop from oral lichen planus or a lichenoid lesion [72,74]. If oral lichen planus lesions change in their extent or have invasive qualities, a biopsy should be performed.
Despite numerous public campaigns to discourage and eliminate drug abuse, illicit drug abuse remains a prevalent global issue. Drug abuse crosses all social, economic, racial, and cultural boundaries. The devastating impact that drug abuse causes in terms of physical and psychologic suffering on individuals and families is immense. In the past decade, the abuse of methamphetamine has increased in incidence and has left a trail of destruction. In 2016, the National Institute on Drug Abuse reported that more than 16 million Americans 12 years of age and older had used methamphetamine at least once and that more than 1.2 million were current users [75]. The average age of the new user is 25.6 years [75,76]. Nationwide treatment admissions for methamphetamine use disorder dropped slightly, from 8.1% in 2005 to 7% in 2015 [77]. Emergency department admissions for methamphetamine abuse also declined, from 132,576 visits in 2004 to 103,000 visits in 2011 [76]. Despite the national trends showing declines, abuse remains noteworthy in Hawaii, the West Coast, and parts of the Midwest [76].
In addition to their illicit use, amphetamines have some clinical applications and have been used to treat attention deficit hyperactivity disorder in adults and children older than 6 years of age. Adults and children older than 12 years of age have had this drug prescribed to treat certain forms of obesity [66].
Beyond its pharmacotherapeutic realm, methamphetamine has gained popularity among drug abusers for several reasons. The manufacturing process of methamphetamine is inexpensive and uncomplicated. Once produced, it may be smoked, snorted, swallowed as a pill, or injected. The euphoria produced by methamphetamine can last approximately 12 hours, compared to the 1-hour duration for cocaine [78]. The neurophysiologic basis for this euphoric state is that methamphetamine blocks the re-uptake of the cerebral neurotransmitters such as dopamine, norepinephrine, and serotonin [79]. Repeated use of this drug can induce cerebral damage, including edema, hemorrhage, strokes, and persistent hallucinations. A potent stimulant, methamphetamine can also cause elevation of both systolic and diastolic blood pressure [80].
The teeth are not spared from the problems caused by the abuse of this drug. As use of this drug increased, an aggressive and rapidly progressing form of dental caries called "meth mouth" was identified. Use of this drug causes several physiologic changes and behaviors that result in this rampant form of caries.
As a stimulant of the alpha-adrenergic receptors within the salivary gland vasculature, methamphetamine causes vasoconstriction and a reduction in salivary flow. Repetitive abuse of this potent stimulant can result in hyposalivation [81]. A chronic lack of salivary flow causes a decrease in the mechanical cleansing of plaque from the teeth and an increase in the retention of cariogenic organisms. There is also a commensurate decrease in salivary immunoglobulins and bactericidal enzymes.
Behaviorally, methamphetamine induces a craving for sugar-laden carbonated beverages, and prolonged exposure of these beverages on the teeth promotes dental caries. Those who abuse this drug also have a tendency for clenching and grinding (bruxing) the teeth. These forces on already weakened teeth further fractures and cause weakening of the remaining tooth structure. After the enamel has fractured away, the softer underlying dentin is more prone to decay and less resilient to fracture.
During the extended periods of euphoria, dental problems often go unnoticed by the patient and progress beyond any hope of restoration. Similarly, oral hygiene is usually neglected while bacteria-laden plaque induces damage to the teeth and gingival tissues. The most common areas affected are the buccal surfaces of the teeth and the interproximal areas of the anterior teeth, both of which are easily accessed for basic oral hygiene techniques [82].
Demographically, methamphetamine use is most prevalent among men between 19 and 40 years of age [83]. However, the systemic and oral effects of methamphetamine abuse extend to all age groups. Unfortunately, most teeth involved with "meth mouth" have extensive caries that require extensive and expensive restorative efforts or are non-restorable and should be extracted. Dental professionals should be cautious in treating these patients. Vasoconstrictors such as epinephrine or levonordefrin can potentiate the stimulant effects of methamphetamine and precipitate a hypertensive crisis. Because most of these patients will require oral surgery to alleviate their dental problems, narcotic analgesics should be prescribed with caution or not at all due to their addictive potential. If a clinician suspects that a patient is abusing methamphetamine, a nonjudgmental discussion should place the overall health and welfare of the patient as the primary concern. Referral to a specialist or a substance abuse program should be a consideration. While the oral problems of "meth mouth" can be rectified even to the point of full-mouth extractions and the placement of dentures, the systemic effects of the continued abuse of methamphetamine have high morbidity and may result in death. An interdisciplinary team approach to treat the dental, medical, psychologic, and behavioral issues of the addictive behavior and the multitude of consequences associated with continued drug use is usually needed to rehabilitate the patient and restore the proper quality of life.
Anorexia nervosa and bulimia nervosa are two of the most widely known eating disorders. Patients with anorexia have body dysmorphia and an excessive fear of gaining weight; they are usually less than 85% of their ideal body weight [48]. In contrast, patients with bulimia try to restrict calorie intake through purging. Often, these patients will experience episodes of extreme binge eating. Those with bulimia may consume a large quantity of food in a short, definitive segment of time (e.g., two hours), but given their concerns about minimizing calorie intake, they induce vomiting and/or use laxatives to remove the food that was consumed during the binge [84]. Patients with anorexia nervosa rely more on severe limitation of food intake to achieve their goals and less so on induced vomiting.
Approximately 90% to 95% of eating disorder cases involve women, and most patients are 12 to 24 years of age [85]. There is no known etiology for anorexia or bulimia. A combination of genetics, psychiatric issues, and cultural standards coupled with an obsessive preoccupation with body size form some of the basis for these diseases.
The systemic health of patients with anorexia or bulimia can be severely compromised. Patients with anorexia become emaciated as a result of their disease, and the lack of proper nutrition can cause a multitude of problems, including bradycardia and cardiac arrhythmias. Individuals with bulimia who are involved in repetitive episodes of binge eating, self-induced vomiting, and use of diuretics and laxatives can experience a severe disruption of the electrolyte balance. Dehydration, problems with muscular contraction, and abnormalities in the conduction of nerve impulses are all potential issues when fluid and electrolyte levels are altered.
The teeth and soft tissues are adversely affected by the repetitive episodes of induced vomiting characteristic of bulimia. Erosion, the loss of tooth structure via a chemical process, can occur as gastric contents deliberately expelled after a binging episode contain hydrochloric acid. The repetitious episodes of binging and vomiting result in an extended cumulative exposure of tooth enamel to this acidic mixture. A unique pattern of erosion called perimylolysis develops.
The enamel of the lingual, occlusal, and incisal surfaces of the maxillary and mandibular teeth is gradually eroded by the gastric acid (Image 6). The erosion extends through the enamel and into the dentin and yields a surface that is smooth and glossy. Sensitivity to temperature and sweets can develop when the superficial dentinal tubules are uncovered. This erosive process can also undermine existing amalgam or bonded restorations. Exposed dentin is a softer material than enamel and is more prone to caries. Tooth structure that previously served as a contact point for occlusion can be minimized or lost. Changes in both the occlusal pattern and the vertical dimension of occlusion can develop. Tooth structure and tooth contour used for a partial denture clasp can change configurations that preclude the proper contact. This can make a partial denture difficult to wear or unusable. If patients have concurrent bruxing or other parafunctional habits, further erosion and abrasion can accelerate the loss of the remaining tooth structure. It may take two or more years for the purgative episodes to develop the erosive patterns [86]. Thickness of the enamel, oral hygiene, and the frequency of the purgative episodes can alter this interval.
The mucosa of the oropharyngeal region can also be traumatized when fingers or objects are used to induce vomiting. Frequent occurrences of these episodes can delay healing of the affected areas and increase the potential for localized infections. The soft palate and the pharynx are the areas most often traumatized.
The parotid glands frequently undergo bilateral enlargement in patients with bulimic and binge eating disorders ("chipmunk facies"). The massive quantities of food ingested during episodes of binge eating cause a commensurate increase in the amount of salivary production. Hypertrophy of the glandular elements causes an increase in the overall size of the gland that can alter the external facial appearance. The duct to the parotid gland remains patent, and salivary flow is unaltered [87].
The treatment of eating disorders such as bulimia and anorexia requires intervention from medical and psychiatric specialists. The prognosis can have considerable variation, as the complexities of the medical and psychiatric issues that underlie eating disorders make the treatment of and recovery from these disorders unpredictable.
Dental treatment can be complex for patients who have lost one or more surfaces of a tooth via perimylolysis. Multiple teeth are usually involved, and conservative and less expensive treatment such as bonded or amalgam restorations may not effectively restore the erosive defects created.
When crowns are needed, the cost of treatment can rise substantially. Patients may require more frequent recall appointments as underlying psychiatric issues and the loss of tooth structure can present a challenge for optimal oral hygiene. Some patients may also be taking antidepressant medications that cause hyposalivation, which can further complicate the ability to maintain optimal oral hygiene. Some antidepressants prescribed for these patients, such as amitriptyline, have the potential to interact with vasoconstrictors found in various concentrations in local anesthetics. An increase in the blood pressure can result. Alternately, medications such as pre-operative sedatives or narcotic analgesics can enhance depression of the central nervous system when taken in conjunction with amitriptyline or other antidepressant. Such medication compatibility issues require collaboration among all healthcare professionals involved in the treatment and care of such patients.
As a result of the evolving racial and immigration demographics in the United States, interaction with patients for whom English is not a native language is inevitable. Because patient history and education are such vital aspects of the prevention, diagnosis, and treatment of oral lesions, it is each practitioner's responsibility to ensure that information and instructions are explained in such a way that allows for patient understanding. When there is an obvious disconnect in the communication process between the practitioner and patient due to the patient's lack of proficiency in the English language, an interpreter is beneficial.
In this multicultural landscape, interpreters are a valuable resource to help bridge the communication and cultural gap between patients and practitioners. Interpreters are more than passive agents who translate and transmit information back and forth from party to party. When they are enlisted and treated as part of the interdisciplinary clinical team, they serve as cultural brokers who ultimately enhance the clinical encounter.
This course has focused on several of the microbial pathogens, autoimmune disorders, and behavioral/psychiatric disorders that contribute to oral pathology. There are numerous other pathogenic organisms, systemic diseases, benign and malignant lesions, and other sources from which a pathologic state of the oral and maxillofacial complex can arise. The diversity and extent of local, regional, and systemic problems that can develop in the oropharyngeal and facial region is considerable. Similarly, the morbidity, treatment, and prognosis of these varied conditions span a wide spectrum. Because many patients do not seek routine dental care, other members of the allied health professions may be the first to encounter patients with pathologic issues located in the oral and facial region. While some of these problems are benign and self-limiting, others can be life-threatening. It is incumbent that clinicians who initially identify the oral problem can provide the necessary treatment to restore optimal oral health or refer the patient to a practitioner who can. Patients should be educated partners in the course of their treatment and should understand the origin, extent, treatment, and prognosis of their specific oral pathologic issue. The efforts of the allied healthcare professionals involved in the treatment of these patients should be concentrated on an approach that restores oral health, promotes systemic health, and maintains an optimal quality of life.
1. Nichols CM, Flaitz CM, Hicks MJ. Treating Kaposi's lesions in the HIV-infected patient. J Am Dent Assoc. 1993;124(11):78-84.
2. Hupp JR, Ferneini EM. Head, Neck, and Orofacial Infections: An Interdisciplinary Approach. 1st ed. St. Louis, MO: Elsevier Science; 2016.
3. Rathe F, Chondros P, Chistodoulides N, Junker R, Sculean A. Necrotising periodontal diseases. Perio. 2007;4(2):93-107.
4. Scully C, Flint SR, Bagan JV, Porter SR, Moos KF. Oral and Maxillofacial Diseases. 4th ed. Boca Raton, FL: CRC Press; 2010.
5. MedlinePlus. Noma. Available at https://medlineplus.gov/ency/article/001342.htm. Last accessed December 7, 2021.
6. Enwonwu CO. Noma (Cancrum Oris). Available at https://www.uptodate.com/contents/noma-cancrum-oris. Last accessed December 7, 2021.
7. Johnson BD, Engel D. Acute necrotizing ulcerative gingivitis: a review of diagnosis, etiology and treatment. J Periodontal. 1986;57(3):141-150.
8. Mayo Clinic. Gingivitis. Available at https://www.mayoclinic.org/diseases-conditions/gingivitis/symptoms-causes/syc-20354453. Last accessed December 7, 2021.
9. Feller L, Lemmer J. Necrotizing periodontal diseases in HIV-serpositive subjects: pathogenic mechanisms. J Int Acad Periodontol. 2008;10(1):10-15.
10. Pincus DJ, Armstrong MB, Thaller SR. Osteomyelitis of the craniofacial skeleton. Semin Plast Surg. 2009;23(2):73-79.
12. Eyrich GK, Baltensperger MM, Bruder E, Graetz KW. Primary chronic osteomyelitis in childhood and adolescence: a retrospective analysis of 11 cases and review of the literature. J Oral Maxillofac Surg. 2003:61(5):561-573.
13. Yeoh SC, MacMahon S, Schifter M. Chronic suppurative osteomyelitis of the mandible: case report. Aust Den J. 2005;50(3):200-203.
14. Bevin CR, Inwards CY, Keller EE. Surgical management of primary chronic osteomyelitis: a long-term retrospective analysis.J Oral Maxillofac Surg. 2008;66(10):2073-2085.
15. Strumas N, Antonyshyn O, Caldwell C B, Mainprize J. Multimodality imaging for precise localization of craniofacial osteomyelitis. J Craniofac Surg. 2003;14(2):215–219.
16. Murphy SC. The person behind the eponym: Wilhelm Frederick von Ludwig (1790–1865). J Oral Pathol Med. 1996;25(9):513-515.
18. MedlinePlus. Ludwig Angina. Available at https://medlineplus.gov/ency/article/001047.htm. Last accessed December 7, 2021.
19. Winters M. Evidence-Based Diagnosis and Management of ENT Emergencies. Available at https://www.medscape.com/viewarticle/551650_4. Last accessed December 7, 2021.
20. Honrado CP, Lam SM, Karen M. Bilateral submandibular gland infection presenting as Ludwig's angina: first report of a case.Ear Nose Throat J. 2001;80(4):217-223.
21. Furst IM, Ersil P, Caminiti M. A rare complication of a tooth abscess-Ludwig's angina and mediastinitis. J Can Dent Assoc. 2001;67(6):324-327.
22. Hought RT, Fitzgerald BE, Latta JE, Zallen RD. Ludwig's angina: report of two cases and review of the literature from 1945 to January 1979. J Oral Surg. 1980;38(11):849-855.
23. Carter L, Lowis E. Death from overwhelming odontogenic sepsis: a case report. Br Dent J. 2007;203(5):241-242.
24. Tyring SK (ed). Mucosal Immunology and Virology. Singapore: Springer-Verlag London Limited; 2006.
25. Klatte JM. Pediatric Herpes Simplex Virus Infection. Available at https://emedicine.medscape.com/article/964866-overview. Last accessed December 7, 2021.
26. Axéll T, Liedholm R. Occurrence of recurrent herpes labialis in an adult Swedish population. Acta Odontol Scand. 1990;48(2):119-123.
27. Ayoade FO, Balan S, Todd JR IV. Herpes Simplex. Available at https://emedicine.medscape.com/article/218580-overview. Last accessed December 7, 2021.
28. Sheridan PJ, Hermann EC Jr. Intraoral lesions of adults associated with herpes simplex virus. Oral Surg Oral Med Oral Pathol. 1971;32(3):390-397.
29. Khan ZZ. Varicella-Zoster Virus. Available at https://emedicine.medscape.com/article/231927-overview. Last accessed December 7, 2021.
30. Nally FF, Ross IH. Herpes zoster of the oral and facial structures: report of five cases and discussion. Oral Surg. 1971;32(2):221-234.
31. Albrecht MA. Clinical Manifestations of Varicella-Zoster Virus Infection: Herpes Zoster. Available at http://www.uptodate.com/contents/clinical-manifestations-of-varicella-zoster-virus-infection-herpes-zoster. Last accessed December 7, 2021.
32. Vinzio S, Lioure B, Enescu I, Schlienger JL, Goichot B. Severe abdominal pain and inappropriate antidiuretic hormone secretion preceding varicella-zoster reactivation 10 months after autologous stem cell transplantation for acute myeloid leukaemia. Bone Marrow Transplant. 2005;35(5):525-527.
33. Jan AM, McGuire TP, Clokie CM, Sandor GKB. Unilateral facial swelling caused by Ramsay Hunt syndrome resembles odontogenic infection. J Can Dent Assoc. 2006;72(9):829-832.
34. Closmann JJ, Fielding CG, Pogrel MA. Prevention and management of trigeminal herpes zoster and postherpetic neuralgia.Gen Dent. 2008;56(6):563-566.
35. Galluzzi KE. Management strategies for herpes zoster and postherpetic neuralgia. J Am Osteopath Assoc. 2007;107(3 Suppl 1):S8-S13.
36. Mayo Clinic. Shingles: Diagnosis and Treatment. Available at https://www.mayoclinic.org/diseases-conditions/shingles/diagnosis-treatment/drc-20353060. Last accessed December 7, 2021.
37. Marin M, Güris D, Chaves SS, Schmid S, Seward JF. Prevention of varicella: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR. 2007;56(RR4):1-40.
38. Harpaz R, Ortega-Sanchez IR, Seward JF. Prevention of herpes zoster: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR. 2008;57(5):1-30.
39. Centers for Disease Control and Prevention. Vaccines and Preventable Diseases. What Everyone Should Know About Zostavax [archive]. Available at https://www.cdc.gov/vaccines/vpd/shingles/public/zostavax/index.html. Last accessed December 7, 2021.
40. Advisory Committee on Immunization Practices. Recommended Adult Immunization Schedule for Ages 19 Years or Older— United States, 2021. Available at https://www.cdc.gov/vaccines/schedules/downloads/adult/adult-combined-schedule.pdf. Last accessed December 7, 2021.
41. Dooling KL, Guo A, Patel M, et al. Recommendations of the Advisory Committee on Immunization Practices for Use of herpes zoster vaccines. MMWR. 2018;67(3):103-108.
42. Centers for Disease Control and Prevention. Updated recommendations for use of VariZIG—United States, 2013. MMWR. 2013;62(28):574-576.
43. Ready T. Varicella-Zoster Virus Globulin: New CDC Recommendations. Available at https://www.medscape.com/viewarticle/808220. Last accessed December 7, 2021.
44. Centers for Disease Control and Prevention. HIV. Basic Statistics. Available at https://www.cdc.gov/hiv/basics/statistics.html. Last accessed December 7, 2021.
45. Kaiser Family Foundation. HIV/AIDS. The HIV/AIDS Epidemic in the United States: The Basics. Available at https://www.kff.org/hivaids/fact-sheet/the-hivaids-epidemic-in-the-united-states-the-basics/#footnote-525108-2. Last accessed December 7, 2021.
46. Vergis EN, Mellors JW. Natural history of HIV-1 infection. Infect Dis Clin North Am. 2000;14(4):809-825.
47. Centers for Disease Control and Prevention. Candida Infections of the Mouth, Throat, and Esophagus. Available at https://www.cdc.gov/fungal/diseases/candidiasis/thrush/index.html. Last accessed December 7, 2021.
48. Little JW, Miller CS, Rhodus NL. Little and Falace's Dental Management of the Medically Compromised Patient. 9th ed. St Louis, MO: Mosby; 2017.
49. Wynn RL, Meiller TF, Crossley HL (eds). Drug Information Handbook for Dentistry. 24th ed. Hudson, OH: Lexi Comp; 2018.
50. Patton LL, Glick M (eds). Clinician's Guide to Treatment of HIV-Infected Patients. 3rd ed. Baltimore, MD: American Academy of Oral Medicine; 2001.
51. Friedman RB, Gunsolley J, Gentry A, Dinius A, Kaplowitz K, Settle J. Periodontal status of HIV-seropositive and AIDS patients.J Periodontal. 1991;62(10):623-627.
52. Najean Y, Rain JD. The mechanism of thrombocytopenia in patients with HIV infection. J Lab Clin Med. 1994;123(3):415-420.
53. Dodd CL, Greenspan D, Greenspan JS. Oral Kaposi's sarcoma in a woman as a first indication of HIV infection. J Am Dent Assoc. 1991;122(4):61-63.
54. Epstein JB, Silverman S Jr. Head and neck malignancies associated with HIV infection. Oral Surg Oral Med Oral Pathol. 1992;73(2):193-200.
55. American Cancer Society. What is Kaposi Sarcoma? Available at https://www.cancer.org/cancer/kaposi-sarcoma/about/what-is-kaposi-sarcoma.html. Last accessed December 7, 2021.
56. Thieringer FM, Cede J, Glatz K, et al. Oral Kaposi's sarcoma: a case report and literature review on treatment management. Craniomaxillofacial Trauma Reconstruction Open. 2021;6:1-7.
57. Mayo Clinic. Soft Tissue Sarcoma: Kaposi's Sarcoma. Available at https://www.mayoclinic.org/diseases-conditions/kaposis-sarcoma/cdc-20387726. Last accessed December 7, 2021.
58. Aoki Y, Tosato G. Pathogenesis and manifestations of human herpesvirus-8-associated disorders. Semin Hematol. 2003;40(2):143-153.
59. Katz J. Kaposi Sarcoma Treatment and Management. Available at https://emedicine.medscape.com/article/279734-treatment. Last accessed December 7, 2021.
60. Rios A, Hagemeister FB. The acquired immunodeficiency syndrome-related malignancies. In: Kantarjian HM, Wolff R (eds).TheMD Anderson Manual of Medical Oncology. 3rd ed. McGraw-Hill; 2016: 933-960.
61. Hidalgo JA. Candidiasis. Available at https://emedicine.medscape.com/article/213853-overview. Last accessed December 7, 2021.
62. Centers for Disease Control and Prevention. Fungal Diseases: Invasive Candidiasis. Available at https://www.cdc.gov/fungal/diseases/candidiasis/invasive/. Last accessed December 7, 2021.
63. Dentistry Today. New Clue to the Virulence of Candida albicans. Available at https://www.dentistrytoday.com/sp-614807553/. Last accessed December 7, 2021.
64. Naylor GD, Terezhalmy GT. Oral complications of cancer chemotherapy: prevention and management. Spec Care Dentist. 1988;8(4):150-156.
65. Dale RA, Harrison JS, Redding SW. Oral complications in cancer chemotherapy, cancer incidence, and mortality in the U.S. Gen Dent. 2004;52(1):64-71.
66. Lacy CF, Armstrong LL, Goldman MP, Lance LL (eds). Lexi-Comp's Drug Information Handbook 2011–2012. 20th ed. Hudson, OH: Lexi Comp; 2012.
67. Pappas PG, Kauffman CA, Andes D, et al. Clinical practice guidelines for the management of candidiasis: 2009 update by the Infectious Diseases Society of America. Clin Infect Dis. 2009;48(5):503-535.
68. Sciubba JJ. Oral mucosal diseases in the office setting-part I: aphthous stomatitis and herpes simplex infections. Gen Dent. 2007;55(4):347-354.
69. Ship II. Epidemiologic aspects of recurrent aphthous ulcerations. Oral Surg Oral Med Oral Pathol. 1972;33(3):400-406.
70. Porter SR, Scully C, Pedersen A. Recurrent aphthous stomatitis. Crit Rev Oral Biol Med. 1998;9(3):306-321.
71. Mirowski GW. Aphthous Stomatitis. Available at https://emedicine.medscape.com/article/1075570-overview. Last accessed December 7, 2021.
72. Thoppay JR. Oral Lichen Planus. Available at https://emedicine.medscape.com/article/1078327-overview. Last accessed December 7, 2021.
74. Moncarz V, Ulmansky M, Lustmann J. Lichen planus: exploring its malignant potential. J Am Dent Assoc. 1993;124(3):102-108.
75. Substance Abuse and Mental Health Services Administration. 2020 National Survey of Drug Use and Health Releases: Detailed Tables. Available at https://www.samhsa.gov/data/release/2020-national-survey-drug-use-and-health-nsduh-releases. Last accessed December 7, 2021.
76. National Institute on Drug Abuse. What is the Scope of Methamphetamine Abuse in the United States? Available at https://www.drugabuse.gov/publications/research-reports/methamphetamine/what-scope-methamphetamine-abuse-in-united-states. Last accessed December 7, 2021.
77. Substance Abuse and Mental Health Services Administration. Treatment Episode Data Set (TEDS) 2005–2015: National Admissions to Substance Abuse Treatment Services. Available at https://wwwdasis.samhsa.gov/dasis2/teds_pubs/2015_teds_rpt_natl.pdf. Last accessed December 7, 2021.
78. American Dental Association. Methamphetamine use and oral health. Am Dent Assoc. 2005;136(10):1491.
79. Klasser GD, Epstein J. Methamphetamine and its impact on dental care. J Can Dent Assoc. 2005;71(10):759-762.
80. Mouth Healthy. American Dental Association. Meth Mouth: How Methamphetamine Use Affects Dental Health. Available at https://www.mouthhealthy.org/en/az-topics/m/meth-mouth. Last accessed December 7, 2021.
83. U.S. Department of Justice Drug Enforcement Administration. Methamphetamine: A Growing Domestic Threat. Available at https://fas.org/irp/agency/doj/dea/product/meth/threat.htm. Last accessed December 7, 2021.
84. Goldman L, Schafer AI (eds). Goldman-Cecil Medicine. 25th ed. Philadelphia, PA: WB Saunders; 2015.
85. Majid SH, Treasure JL. Eating disorders. In: Wright P, Stern J, Phelan M (eds). Core Psychiatry. 3rd ed. Edinburgh: Elsevier; 2012.
1. Freedman MS, Ault K, Bernstein H. Advisory Committee on Immunization Practices recommended immunization schedule for adults aged 19 years or older—United States, 2021. MMWR. 2021;70:193-196.Available at https://www.cdc.gov/mmwr/volumes/70/wr/mm7006a2.htm. Last accessed December 9, 2021.
2. National Institutes of Health Office of AIDS Research. Guidelines for the Prevention and Treatment of Opportunistic Infections in HIV-Exposed and HIV-Infected Children. Washington, DC: Department of Health and Human Services; 2021. Available at https://clinicalinfo.hiv.gov/en/guidelines/pediatric-opportunistic-infection/candida-infections. Last accessed December 9, 2021.
Mention of commercial products does not indicate endorsement.