This comprehensive course explores the appropriate use of antithrombotic agents for improved patient outcomes. Key topics include pharmacology of common antiplatelet and anticoagulant drugs, evidence-based indications for mono- and combination therapy, monitoring parameters for safe and effective use, and management of potential complications. Upon completion, you'll be equipped with essential knowledge and clinical pearls for delivering high-quality antithrombotic care.
This course is designed for physicians, nurses, and pharmacists whose patients benefit from anticoagulation and/or antiplatelet therapy.
The purpose of this course is to provide clinicians with clinical guidance on the appropriate use of antithrombotic agents in order to improve patient outcomes.
Upon completion of this course, you should be able to:
- Compare and contrast the mechanisms of action of commonly used anticoagulant and antiplatelet agents, including unfractionated heparin, low molecular weight heparins, vitamin K antagonists, direct thrombin inhibitors, factor Xa inhibitors, aspirin, and P2Y12 inhibitors.
- Identify best practice and evidence-based recommendations for initiating anticoagulant therapy for various conditions, including atrial fibrillation, acute coronary syndrome, prosthetic heart valves, and venous thromboembolism using risk stratification frameworks.
- Recognize clinical situations warranting antiplatelet therapy, either alone or in combination with anticoagulants, based on updated treatment guidelines for relevant vascular conditions, including acute coronary syndrome, coronary artery disease, acute ischemic stroke, and peripheral arterial disease.
- Select the most appropriate medication monitoring strategy based on specific anticoagulant or antiplatelet agent and therapeutic indication.
- Indicate safe strategies for transitioning between anticoagulant and antiplatelet agents to avoid therapy gaps or excessive overlap that can precipitate thrombotic complications or bleeding, based on indication timeframe, procedure guidance, and antithrombotic agent properties.
- Recognize special populations who may require additional monitoring of anticoagulant or antiplatelet therapies due to altered pharmacokinetics and pharmacodynamic parameters.
Justin Delic, PharmD, BCCCP, is a critical care clinical pharmacy specialist at Cooper University Hospital in Camden, New Jersey. He completed his first year of pharmacy residency at UPMC Mercy Hospital in Pittsburgh, Pennsylvania, followed by a second year of residency specializing in critical care medicine at University Hospitals Cleveland Medical Center in Cleveland, Ohio. Following residency, he went on to practice as an assistant professor of clinical pharmacy at the Philadelphia College of Pharmacy in Philadelphia, Pennsylvania, prior to transitioning into his current role as a clinical pharmacy specialist at Cooper University Hospital. His areas of expertise include pharmacology, neurocritical care, simulation education, and anticoagulation.
Contributing faculty, Justin Delic, PharmD, BCCCP, has disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
John M. Leonard, MD
Candace Pierce, DNP, RN, CNE, COI
Randall L. Allen, PharmD
The division planners have disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
Sarah Campbell
The Director of Development and Academic Affairs has disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
The purpose of NetCE is to provide challenging curricula to assist healthcare professionals to raise their levels of expertise while fulfilling their continuing education requirements, thereby improving the quality of healthcare.
Our contributing faculty members have taken care to ensure that the information and recommendations are accurate and compatible with the standards generally accepted at the time of publication. The publisher disclaims any liability, loss or damage incurred as a consequence, directly or indirectly, of the use and application of any of the contents. Participants are cautioned about the potential risk of using limited knowledge when integrating new techniques into practice.
It is the policy of NetCE not to accept commercial support. Furthermore, commercial interests are prohibited from distributing or providing access to this activity to learners.
Supported browsers for Windows include Microsoft Internet Explorer 9.0 and up, Mozilla Firefox 3.0 and up, Opera 9.0 and up, and Google Chrome. Supported browsers for Macintosh include Safari, Mozilla Firefox 3.0 and up, Opera 9.0 and up, and Google Chrome. Other operating systems and browsers that include complete implementations of ECMAScript edition 3 and CSS 2.0 may work, but are not supported. Supported browsers must utilize the TLS encryption protocol v1.1 or v1.2 in order to connect to pages that require a secured HTTPS connection. TLS v1.0 is not supported.
The role of implicit biases on healthcare outcomes has become a concern, as there is some evidence that implicit biases contribute to health disparities, professionals' attitudes toward and interactions with patients, quality of care, diagnoses, and treatment decisions. This may produce differences in help-seeking, diagnoses, and ultimately treatments and interventions. Implicit biases may also unwittingly produce professional behaviors, attitudes, and interactions that reduce patients' trust and comfort with their provider, leading to earlier termination of visits and/or reduced adherence and follow-up. Disadvantaged groups are marginalized in the healthcare system and vulnerable on multiple levels; health professionals' implicit biases can further exacerbate these existing disadvantages.
Interventions or strategies designed to reduce implicit bias may be categorized as change-based or control-based. Change-based interventions focus on reducing or changing cognitive associations underlying implicit biases. These interventions might include challenging stereotypes. Conversely, control-based interventions involve reducing the effects of the implicit bias on the individual's behaviors. These strategies include increasing awareness of biased thoughts and responses. The two types of interventions are not mutually exclusive and may be used synergistically.
#95310: Anticoagulation and Antiplatelet Therapy: Clinical Use Guidelines
Despite the wide availability of effective anticoagulant and antiplatelet medications, confusion on proper role, duration, transitions, monitoring, and real-world translation of evolving clinical practice guidelines drives the persistent over-, under-, and misuse of these agents. This gap between knowledge and appropriate evidence-based practice leads directly to worsened patient outcomes and increased health system costs. This course looks to provide consolidated, practical guidance tools that build provider competence and confidence in navigating nuanced prescribing decisions surrounding antithrombotic therapies to significantly narrow this gap tied to preventable patient harm.
The development of antithrombotics, including both anticoagulants and antiplatelets, has been one of the most significant advancements in the treatment and prevention of cardiovascular and thromboembolic disease states in history. Many of these agents have been around for decades, with the use of aspirin being traced back to 1897, and the oral anticoagulant warfarin first being used in humans in 1954 [1,2]. These discoveries provided groundbreaking treatment options for patients and prescribers alike; however, they also brought along with them concerns for an increased risk of bleeding. Over the past two decades, this landscape has continued to change with the approval of the direct oral anticoagulants (DOACs) and other antiplatelet agents within this therapeutic category. Although many of these newer agents provide an improved safety profile, they are still associated with significant morbidity and mortality related to bleeding events worldwide. Today, aspirin remains one of the most commonly used medications in the United States despite a slight decline in use over the past decade, and the DOACs have rapidly been adopted since their approvals, replacing warfarin as the oral anticoagulants of choice in most patients [3,4,5,6].
Although these agents continue to be prescribed often and provide significant benefit in many patients, experts indicate a need for improved use and monitoring of these medications. Recent studies have found that on one end of the spectrum, many older adults with disorders such as atrial fibrillation (AF) are undertreated with anticoagulants, while on the other end of the spectrum, many emergency department visits and hospitalizations are due to adverse reactions from these medications [7,8,9,10,11]. In a cross–sectional study by Dreijer et al., authors evaluated nearly 43,000 reported medical errors from 2012 to 2015 in both hospitals and primary care, and they found that anticoagulants were involved in over 8% of reported errors [12]. This recognition of the continued impact that antithrombotics (or the lack thereof) have on patient outcomes has sparked efforts to improve the use of these agents and has even prompted some hospitals to direct time and resources into developing multidisciplinary anticoagulation stewardship programs that specifically focus on appropriate antithrombotic use within their institutions [13,14]. With this in mind, it is obvious that despite the advancements in the overall safety of newer antithrombotic agents, improvement in the appropriate prescribing and monitoring of these agents should remain a top priority for prescribers and other healthcare professionals alike.
In order to identify the role of antithrombotics in the management of atherosclerotic and thromboembolic disease, it is first important to understand the pathophysiology related to hemostasis and thrombosis within the body. Hemostasis is the body's natural process of stopping blood from leaving a vessel after vascular injury by sealing the injured vessel and preventing further blood loss [15]. For hemostasis to occur, the body vasoconstricts the injured blood vessel in order to minimize blood loss and forms a platelet plug (a process known as primary hemostasis) that is further stabilized by a dense fibrin mesh (a process known as secondary hemostasis). Although it is common to think of these three phases in sequence, they actually occur simultaneously and potentiate one another during this process [16]. The formation of the platelet plug (primary hemostasis) involves three main steps, including platelet adhesion to the vessel wall, platelet activation, and platelet aggregation. Each of these three steps is initiated with the activation of various receptors on the platelet and can be a target for antiplatelet medications. The formation of a fibrin mesh (secondary hemostasis), on the other hand, involves activation of the coagulation cascade.
As opposed to hemostasis, thrombosis is the creation of a blood clot within a vessel through the activation of the coagulation cascade and is generally an unwanted result of coagulation within the body. When thrombosis occurs, a blood clot containing fibrin, platelet aggregates, and trapped red blood cells is created within a vessel, potentially resulting in partial or complete blockage of the vessel and preventing distal areas of the body from receiving oxygen and nutrients necessary for survival [16]. This can further lead to significant cell damage and death, making it an important focus for prevention in patients at greatest risk [15]. Although thrombosis may develop for a number of causes, thrombosis related to atherosclerotic disease is of particular relevance for this course, as it is a significant contributor to the development of disease states such as acute coronary syndrome (ACS) and acute ischemic stroke. In atherosclerosis, plaque made up of cholesterol, fatty substances, cellular waste products, calcium, and fibrin builds up in arteries, leading to a thickening of the blood vessel wall and a narrowing of the vessel opening. As the plaque continues to grow, it can partially or completely block blood flow through a vessel, or it can become unstable and rupture, sending pieces of this dislodged plaque buildup through the bloodstream until it becomes stuck within a narrowed vessel later in its path. Unstable plaques, which are at the greatest risk of rupture, typically have thin fibrous caps with large lipid cores, and, when ruptured, release thrombogenic material from within the plaque that leads to platelet adhesion, activation of additional platelets, and aggregation at the site until a thrombus is formed [17]. Since this platelet activation and aggregation can lead to a thrombus that blocks blood flow or embolizes to distal parts of the body, a primary goal of medical therapy for ACS, stroke, and other atherosclerotic diseases is to prevent the formation of these thrombi and the resultant death and disability that they may cause.
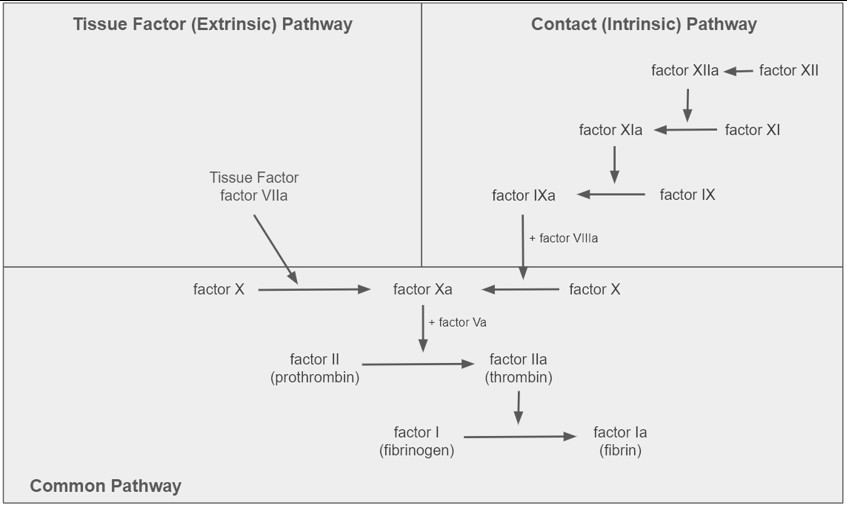
In order for either hemostasis or thrombosis to occur, the coagulation cascade must be activated by one of two separate pathways called the tissue factor pathway (also known as the "extrinsic" pathway) or the contact pathway (also known as the "intrinsic" pathway). Although the tissue factor (extrinsic) pathway is classically known as the pathway that is implicated in hemostasis, it likely also plays a role in many types of thrombosis as well. Alternatively, the contact (intrinsic) pathway does not affect normal hemostasis but rather is thought to contribute to thrombosis only [15]. The tissue factor (extrinsic) pathway becomes activated once blood is exposed to cells that express an extrinsic substance (i.e., tissue factor), leading to a complex of tissue factor and factor VIIa that further triggers coagulant activation. On the other hand, the contact (intrinsic) pathway is typically activated when blood comes into contact with exogenous material, resulting in an initial activation of factor XII to factor XIIa that leads to further activation of the cascade. Once either of these pathways are initiated, a series of proteolytic reactions occur that further activate various coagulation factors and substances within the body. Eventually, both pathways converge into the last few steps where factor X is converted to factor Xa by factor VIIIa, factor II (prothrombin) is converted to factor IIa (thrombin) by factor Va, and factor IIa (thrombin) converts factor I (fibrinogen) to factor Ia (fibrin) and activates platelets, leading to a clot [15,18]. Since both extrinsic and intrinsic pathways share the same final steps, this portion of the cascade is known as the "common pathway." As will be discussed in the next section, anticoagulant agents inhibit the activation of many of these coagulation factors (e.g., factor Xa inhibitors or direct thrombin inhibitors), preventing the development of thrombosis. A simplified version of the coagulation cascade can be seen in Figure 1.
Aspirin (also known as acetylsalicylic acid or ASA) has been used for well over a century and remains one of the most commonly prescribed medications to this day. It is currently approved for the prevention and treatment of many vascular diseases, including acute myocardial infarction (MI), unstable angina, stable ischemic heart disease, transient ischemic attack (TIA), and cerebral infarction as well as for prophylaxis of venous thromboembolism following total hip replacement and reduction of platelet adhesion following revascularization procedures such as carotid endarterectomy or coronary artery bypass grafting (CABG). Unrelated to its antiplatelet effects, aspirin is also approved for the treatment of pain, fever, and inflammation [19,20]. Aspirin works via irreversible inhibition of cyclooxygenase-1 and 2 (COX-1 and 2), which ultimately decreases the formation of prostaglandin precursors, leading to its therapeutic effects. Aspirin's antiplatelet effect, in particular, is due to its inhibition of the formation of thromboxane A2, a prostaglandin derivative, within the platelet. Thromboxane A2 is known to be responsible for the platelet aggregation step of clot formation, which can therefore be reduced with the use of aspirin [19,20]. Aspirin is currently available in both immediate and extended-release oral formulations as well as in the form of a rectal suppository, and no intravenous formulation is currently available. Aspirin doses vary by indication but are typically 81–325 mg orally daily for indications that require aspirin use for its antiplatelet effects, while doses for analgesia and anti-inflammatory effects are often much higher and given more frequently [19]. As with any antithrombotic, precautions should be used due to an increased risk of bleeding with this agent, and aspirin should be used with particular caution in patients at high risk of bleeding from gastrointestinal ulcers, in severe hepatic or renal failure, with heavy alcohol use, and in patients within their last trimester of pregnancy (except when being utilized at low doses for pregnancy-related conditions). Although no routine monitoring of aspirin levels or antiplatelet response are required, patients should be monitored for signs or symptoms of bleeding during use.
The P2Y12 inhibitors are a group of agents that provide their antiplatelet effects through a mechanism different from that of aspirin. These agents work by inhibiting the P2Y12 component of adenosine diphosphate (ADP) receptors on the platelet surface, resultantly preventing activation of the glycoprotein IIb/IIIA receptor complex and reducing platelet aggregation [21]. Currently, the P2Y12 inhibitors that are available in the United States include clopidogrel, prasugrel, ticagrelor, and the most recently approved intravenous agent, cangrelor. Although ticlopidine was historically included in this group, this agent is no longer available in the United States. Despite all of these medications having the same overall mechanism for platelet inhibition, important differences exist that must be recognized between agents in order to ensure safe and effective use.
Clopidogrel (Plavix) is a second generation thienopyridine that was first approved in the United States in 1997, making it the oldest of the currently available oral P2Y12 inhibitors. Similar to prasugrel, clopidogrel is a prodrug that requires activation by the cytochrome P450 system (primarily CYP2C19) to be converted into an active metabolite that provides its antiplatelet effect. In patients with normal metabolism, about 85% of the prodrug is hydrolyzed into an inactive metabolite, while only the remaining 15% is metabolized into the active form of the drug [21]. Patients who are poor metabolizers (clopidogrel nonresponders) or those concomitantly taking medications that inhibit CYP2C19, such as omeprazole, can experience reduced effectiveness of clopidogrel due to an inability to convert the parent drug into its active metabolite. These patients have been shown to have worse outcomes following ACS and percutaneous coronary intervention (PCI). In practice, patients may be tested to identify their CYP2C19 genotype or can receive platelet function testing to evaluate their response to clopidogrel in order to aid in treatment decisions. It is also important to note that clopidogrel provides irreversible platelet inhibition for the lifespan of the platelet, which can be for 7 to 10 days. If clopidogrel needs to be held prior to elective surgery, this should be done at least 5 days prior to the procedure due to clopidogrel's irreversible platelet inhibition and prolonged duration of effect [22]. Clopidogrel is currently indicated for prevention and treatment of ACS, acute ischemic stroke, TIA, and established peripheral artery disease (PAD). The most common adverse reaction associated with clopidogrel use includes bleeding, though other rare events such as thrombotic thrombocytopenic purpura (TTP) have been reported in the literature. Clopidogrel is only available as an oral tablet at this time and is typically dosed at 75–150 mg daily alone or in combination with aspirin or an anticoagulant [22]. A loading dose of 300–600 mg of clopidogrel may be used when it is imperative to achieve therapeutic concentrations rapidly, such as directly after an ischemic event or in patients who are receiving revascularization or stenting; however, the benefits of this are typically weighed against the risk of bleeding for each individual patient.
Prasugrel (Effient) is a third generation thienopyridine that was approved in the United States in 2009. Similar to clopidogrel, prasugrel is a prodrug that requires rapid metabolism in order to be converted into an active metabolite that provides its irreversible platelet inhibition; however, the formation of this active metabolite is much more efficient compared to clopidogrel [21]. Although the active metabolites of clopidogrel and prasugrel are approximately equipotent, the metabolism of prasugrel results in higher plasma concentrations of active drug compared to clopidogrel, leading to effects that are more potent, immediate, and predictable [21]. Currently, it is only approved in the management of ACS. Due to the risk of fatal and intracranial bleeding seen in initial studies, prasugrel is generally not recommended in patients 75 years of age and older, unless benefit is felt to exceed the risk in select scenarios. In addition, bleeding from prasugrel is noted to be increased in patients of low body weight (<60 kg), those who are already at an increased bleeding risk for other reasons, and those who are concomitantly taking medications that may also increase the risk of bleeding. It is also contraindicated in patients with a history of prior TIA or stroke. If temporary discontinuation is required due to surgery or invasive procedure, it is recommended that prasugrel be discontinued at least 7 days prior the procedure, and it is also recommended that prasugrel not be started in patients who are likely to undergo CABG [23,24]. The most common adverse reaction associated with prasugrel is bleeding, though other rare events such as TTP have been reported in the literature as well. Prasugrel is only available as an oral tablet at this time and is typically dosed as an initial 60-mg loading dose followed by 10 mg daily in combination with aspirin. A reduced dose of 5 mg daily may be utilized in patients at higher risk of bleeding to prasugrel, particularly those who weigh less than 60 kg [23,24]. Compared to other P2Y12 inhibitors, prasugrel's use has been limited in some populations due to safety concerns related to an increased risk of bleed and adverse effects.
Ticagrelor (Brilinta) is the most recently approved oral P2Y12 inhibitor with an initial United States approval in 2011. Unlike clopidogrel and prasugrel, ticagrelor is a direct-acting, reversible platelet inhibitor that does not require metabolism from prodrug to active metabolite in order to provide its antiplatelet effect. Ticagrelor and its metabolite both work in the inhibition of P2Y12 and are approximately equipotent [21,25]. Unlike other P2Y12 inhibitors, ticagrelor requires twice daily dosing, due to its reversible platelet binding and half-life of 6 to 12 hours. Currently, it is approved in the management of ACS, select patients with stable coronary artery disease (CAD), and minor stroke, and it has been shown to be superior to clopidogrel in the first 12 months following ACS [25,26]. Similar to other P2Y12 inhibitors, ticagrelor is a potent antiplatelet that can cause significant bleeding, and it is contraindicated in patients with a history of intracranial hemorrhage. In addition to this risk of bleeding, ticagrelor is also noted to cause dyspnea in approximately 14% of patients, and it has been reported to be associated with bradyarrhythmias, including atrioventricular block, as well [26,27]. Ticagrelor is only available as an oral tablet at this time and is typically dosed as an initial 180 mg loading dose followed by 90 mg twice daily for the first year following ACS in combination with aspirin 75–100 mg. Despite some conflicting reports, aspirin doses greater than 100 mg are currently thought to reduce the effectiveness of ticagrelor and should be avoided, based on a subgroup analysis of the PLATO trial [26,28]. If prolonged use beyond one year is required, ticagrelor dose should be reduced to 60 mg orally twice daily. If temporary interruption in therapy is required prior to surgery due to the risk of bleeding, ticagrelor should be held five days prior to the procedure when possible [25].
Cangrelor (Kengreal) is currently the only available intravenous P2Y12 inhibitor in the United States and was first approved in 2015. Similar to ticagrelor, cangrelor is a direct-acting P2Y12 platelet inhibitor that binds selectively and reversibly to the platelet receptor in order to prevent platelet activation in a dose-dependent manner [21]. It is currently approved as an adjunct to PCI for reducing peri-procedural MI, repeat revascularization, and stent thrombosis in patients not yet treated with another P2Y12 inhibitor or glycoprotein IIb/IIIa inhibitor (e.g., eptifibatide), and it is also used off-label as bridging therapy prior to cardiac surgery in patients at high risk of thrombotic complications when oral antiplatelet therapy is held. Cangrelor's main advantages include its quick onset of action (approximately 2 minutes) and extremely short half-life of 3 to 6 minutes. These properties, paired with its reversible platelet inhibition, allow for platelet function to return to normal within one hour of cangrelor discontinuation [21]. Cangrelor is typically dosed as a 30-mcg/kg intravenous bolus followed by a 4-mcg/kg/min infusion and continued for at least two hours or the duration of the procedure, whichever is longer, when used for its labeled indication. Off-label use of cangrelor as bridging therapy requires a lower dosing regimen, most commonly cited as 0.75 mcg/kg/min, based on the BRIDGE trial [29]. Reported adverse effects related to cangrelor are limited and are primarily related to its increased risk of bleeding.
Monitoring of P2Y12 inhibitors can be done through the use of platelet function testing, such as with the VerifyNow assay. VerifyNow is a P2Y12 point-of-care, optical aggregometry assay that evaluates the level of platelet inhibition in terms of platelet reactivity units or P2Y12 reaction units (PRU) based on the rate and extent of platelet aggregation in a blood sample [30,31]. Although not routinely performed in all patients, this test can be particularly useful in select clinical scenarios, such as when evaluating a patient's ability to metabolize clopidogrel into its active metabolite and receive its therapeutic effect. By assessing PRU in a patient taking clopidogrel, this test can assist in determining if the patient is a poor metabolizer (clopidogrel nonresponder) that may be at an increased risk of ischemic events. In patients without appropriate response, providers can then assess if another agent, such as one of the newer P2Y12 inhibitors, may be more appropriate for treatment. When evaluating the results of the VerifyNow assay, certain cutoffs have been associated with ischemic events (PRU >208) as well as with bleeding events (PRU <85) [31]. Although this test has certain limitations and may not be as accurate in select populations, such as patients from East Asia, it provides additional information to make informed decisions regarding antithrombotic therapy.
With the widespread use of P2Y12 inhibitors, it may be common to encounter the clinical scenario of switching between agents during therapy. Although this change may be necessary, it also presents potential concerns regarding the safety of making this switch appropriately. When switching agents, patients may be at risk for inadequate platelet inhibition (and an increased risk of thrombotic complications related to gaps in therapy) as well as at risk of excessing platelet inhibition (and an increased risk of bleeding related to overlapping therapy). By being mindful of drug-specific pharmacological properties such as half-life, site of action, and speed of onset, these risks can be mitigated. Due to these concerns, Angiolillo et al. published a white paper detailing the international expert consensus on switching platelet P2Y12 receptor-inhibiting therapies [21]. This document reviews the relevant pharmacology and timing considerations when switching between oral P2Y12 inhibitors and between an intravenous P2Y12 inhibitor (i.e., cangrelor) and oral P2Y12 inhibitors. A brief summary of these recommendations, based on time from the index event, is provided inTable 1 and Table 2. Although these general consensus recommendations provide a starting point, they may not be applicable in all clinical scenarios and should be evaluated in the context of personalized patient care.
SWITCHING ORAL P2Y12 INHIBITORS DURING ACUTE/EARLY PHASE (WITHIN 30 DAYS OF INDEX EVENT)
| Clopidogrel Transitioned to: | |
| Prasugrel | Begin prasugrel 60-mg loading dose, regardless of timing and dosing of clopidogrel |
| Ticagrelor | Begin ticagrelor 180-mg loading dose, regardless of timing and dosing of clopidogrel |
| Prasugrel Transitioned to: | |
| Clopidogrel | Begin clopidogrel 600-mg loading dose, administered 24 hours after last prasugrel dosea |
| Ticagrelor | Begin ticagrelor 180-mg loading dose, administered 24 hours after last prasugrel dose |
| Ticagrelor Transitioned to: | |
| Clopidogrel | Begin clopidogrel 600-mg loading dose, administered 24 hours after last ticagrelor dosea |
| Prasugrel | Begin prasugrel 60-mg loading dose, administered 24 hours after last ticagrelor dose |
| aIf a patient is being de-escalated to clopidogrel for bleeding concerns, consider omitting loading dose and initiating clopidogrel 75-mg maintenance dose 24 hours after last prasugrel or ticagrelor dose. | |
SWITCHING ORAL P2Y12 INHIBITORS DURING LATE/VERY LATE PHASE (GREATER THAN 30 DAYS AFTER INDEX EVENT)
| Clopidogrel Transitioned to: | |
| Prasugrel | Begin prasugrel 10-mg maintenance dose, beginning 24 hours after last clopidogrel dose |
| Ticagrelor | Begin ticagrelor 90-mg BID maintenance dose, beginning 24 hours after last clopidogrel dose |
| Prasugrel Transitioned to: | |
| Clopidogrel | Begin clopidogrel 75-mg maintenance dose, beginning 24 hours after last prasugrel dose |
| Ticagrelor | Begin ticagrelor 90-mg BID maintenance dose, beginning 24 hours after last prasugrel dose |
| Ticagrelor Transitioned to: | |
| Clopidogrel | Begin clopidogrel 600-mg loading dose, administered 24 hours after last ticagrelor dosea |
| Prasugrel | Begin prasugrel 60-mg loading dose, administered 24 hours after last ticagrelor dose |
| aIf a patient is being de-escalated to clopidogrel for bleeding concerns, consider omitting loading dose and initiating clopidogrel 75-mg maintenance dose 24 hours after last prasugrel or ticagrelor dose. | |
When transitioning from oral P2Y12 inhibitors to intravenous P2Y12 inhibitors in the setting of bridging for surgery, clopidogrel and ticagrelor should typically be discontinued 5 days before the procedure, while prasugrel is recommended to be discontinued for 7 days prior to procedure. Following this, it is reasonable to begin cangrelor up to 3 to 4 days after prasugrel discontinuation and 2 to 3 days of clopidogrel and ticagrelor discontinuation. When transitioning patients from intravenous P2Y12 inhibitor to an oral P2Y12 inhibitor, it is important to note that the loading doses of clopidogrel or prasugrel should not be administered until immediately after discontinuation of cangrelor. Ticagrelor, on the other hand, may be administered anytime during or immediately following cangrelor infusion [21].
Unfractionated heparin (UFH) is another antithrombotic with a long-standing, rich history in the field of medicine. Heparin was initially approved in the United States in 1939 and remains one of the most commonly used injectable anticoagulants to this day. Heparin provides its anticoagulant effect by binding to antithrombin III (ATIII) in order to create a complex that inhibits the activation of factor IIa (thrombin) as well as factors IXa, Xa, XIa, and XIIa, in addition to other mechanisms [32]. Heparin also helps prevent the formation of a fibrin clot by inhibiting activation of the fibrin stabilizing factor. Contrary to some belief, heparin does not break down an existing clot like thrombolytics, but rather, solely acts to prevent the continued propagation and further stabilization of an active clot. Heparin is typically administered as a continuous intravenous infusion or as a subcutaneous injection, depending on indication and availability of intravenous access. Due to its extensive history and favorable pharmacokinetic profile, heparin remains a drug of choice in numerous disease states and clinical scenarios. It is currently approved for prophylaxis and treatment of venous thromboembolism (VTE) including deep vein thrombosis (DVT) and pulmonary embolism (PE), peripheral arterial embolism, AF with embolization, acute and chronic consumptive coagulopathies (e.g., disseminated intravascular coagulation), prevention of clotting in arterial and cardiac surgery, and as an anticoagulant for use in blood transfusions, extracorporeal circulation, and dialysis procedures [33,34]. The dosing of heparin can vary significantly by indication. A few of the most common dosing regimens that may be seen in practice are listed in Table 3; however, this list is not all inclusive and may also vary slightly by institution.
COMMON HEPARIN DOSING REGIMENS
| Indication | Heparin Dosing Regimena | ||
|---|---|---|---|
| VTE prophylaxis | 5,000 units subcutaneously every 8 or 12 hoursb | ||
| VTE treatment | Initial: 80 units/kg IV bolus followed by 18 units/kg/hour IV infusion | ||
| AF | Initial: 60–80 units/kg (maximum 5,000 units) IV bolus followed by 12–18 units/kg/hour (maximum 1,000 units/hour) IV infusion | ||
| ACS, NSTEMI (off-label) | Initial: 60 units/kg (maximum 5,000 units) IV bolus followed by 12 units/kg/hour (maximum 1,000 units/hour) IV infusion | ||
| |||
In terms of cautions and contraindications, heparin is known to increase the risk of bleeding, and it is also known to cause thrombocytopenia, elevations in serum aminotransferases, and heparin-induced thrombocytopenia (HIT) with or without thrombosis in some patients. Of note, not all patients who experience heparin-related thrombocytopenia have HIT, and appropriate assessment of thrombocytopenia using risk scores (e.g., 4T score) and confirmatory testing (e.g., heparin antibody and serotonin release assay) should be performed if HIT is suspected. Heparin is contraindicated in patients with HIT as well as those with hypersensitivity to heparin or pork since it is derived from animal tissue [33]. In patients who require temporary interruption of a heparin infusion due to surgery, the heparin infusion should be held at least 4 to 6 hours before the procedure due to its variable half-life of 0.5–2 hours [35].
After a heparin infusion has been initiated, monitoring of its response and titration to a therapeutic goal is imperative in order to ensure both safety and efficacy. For decades, the standard method of monitoring heparin infusions in hospitalized patients had been through assessment of the aPTT, with a target range of 1.5 to 2.5 times the control. Due to the high degree of variability in aPTT readings between laboratories and reagents, guiding organizations made recommendations in the 1990s that institution-specific therapeutic goal ranges should be determined according to a corresponding heparin level, with heparin concentrations of 0.2–0.4 unit/mL by protamine titration or 0.3–0.7 unit/mL by antifactor Xa assay being used for reference of therapeutic range [36]. Monitoring of heparin by anti-factor Xa assay has been shown to have certain benefits over traditional aPTT monitoring, including a faster time to therapeutic levels of heparin, a longer time in goal range, and fewer adjustments in dosing and repeated testing [37,38]. This has prompted some institutions to change from using aPTT to antifactor Xa assay as the standard for heparin monitoring and titration. Regardless of institution-specific monitoring strategy, heparin should be monitored at standard intervals (e.g., every 6 hours) until consistently resulting in the therapeutic range before further extending monitoring frequency. In patients with subtherapeutic heparin monitoring despite receiving more than 35,000 units of heparin per day after titration, heparin resistance should be suspected [39]. Heparin resistance may occur in patients due to various causes, including deficiency of endogenous ATIII, the necessary cofactor required for heparin's anticoagulant effect. Although current practice varies in the management of patients with suspected heparin resistance, potential mitigation strategies that have been employed include continuing to increase the dose of heparin until reaching response, providing supplementation of exogenous ATIII if available, and changing to a different anticoagulant that does not require ATIII for activity, such as argatroban or bivalirudin [39].
Transitioning from heparin to other anticoagulants is a clinical scenario that occurs often in the hospital setting and can be associated with a significant risk of medication errors and adverse effects if not handled appropriately. Based on the pharmacokinetic properties of each anticoagulant, currently recommended transitioning strategies are provided in Table 4 [40].
HEPARIN (INTRAVENOUS INFUSION) TRANSITION TO OTHER ANTICOAGULANTS
| Transitioned to | Transition Strategy |
|---|---|
| Enoxaparin (SUBQ) | Begin enoxaparin within one hour of stopping heparin infusion. |
| Warfarin (oral) | Begin warfarin and continue heparin until INR is within therapeutic range (based on indication) for at least 2 measurements about 24 hours apart. |
| DOAC (oral)a | Begin DOAC when the heparin infusion is stopped. |
| aIncluding apixaban, dabigatran, rivaroxaban, and edoxaban | |
The low-molecular-weight heparins (LMWHs) are a group of anticoagulants that provide anticoagulant effects with a similar mechanism to heparin except with an increased inhibition of factor Xa over factor IIa (thrombin) [32]. Multiple agents are included within this class including enoxaparin, dalteparin, nadroparin, and tinzaparin; however, enoxaparin is currently the most widely used agent within the United States and will be the focus of this section.
Enoxaparin (Lovenox) was first approved in the United States in 1993 and remains one of the most commonly used subcutaneous anticoagulants to this day. Enoxaparin is currently approved for prophylaxis and treatment of VTE in a variety of patient populations, prophylaxis of ischemic complications of unstable angina and non-Q-wave MI, and treatment of ST-segment elevation MI. Since enoxaparin is similar in composition to heparin, it is also contraindicated in patients with a history of HIT within the past 100 days (or with circulating antibodies) as well as in patients with a hypersensitivity to heparin or pork products. Enoxaparin is currently available as a subcutaneous injection, though it can also be administered intravenously in the management of ACS, and dosing varies based on indication. Of note, enoxaparin exposure is significantly increased in those with renal impairment and dosage or frequency adjustments are required, particularly in those with a creatinine clearance <30 mL/min [41,42]. Although not all inclusive, a list of common enoxaparin dosing regimens are provided in Table 5.
COMMON ENOXAPARIN DOSING REGIMENS
| Indication | Enoxaparin Dosing Regimen |
|---|---|
| VTE prophylaxis | 40 mg subcutaneously every 24 hours (Select patient populations may require alternate dosing or frequencya) |
| VTE treatment | 1 mg/kg subcutaneously every 12 hours or 1.5 mg/kg every 24 hours |
| ACS (NSTEMI) or AF (periprocedural management) | 1 mg/kg subcutaneously every 12 hours |
| aTrauma, morbid obesity, low body weight, or reduced renal function | |
Adverse effects associated with enoxaparin include bleeding, thrombocytopenia, increases in serum aminotransferases, and injection site pain. Additionally, enoxaparin has been associated with epidural and spinal hematomas in patients undergoing neuraxial anesthesia or spinal puncture, resulting in significant long-term or even permanent paralysis. Factors increasing the risk of bleeding and the timing of the last enoxaparin dose should be closely evaluated in patients receiving these procedures [41]. In patients undergoing surgery, the last dose of therapeutic enoxaparin should be given no closer than 24 hours prior to surgery [35].
Therapeutic drug monitoring is not routinely performed in most patients receiving enoxaparin; however, antifactor Xa monitoring can be useful in the management of special populations or those with a high risk of altered pharmacokinetics (e.g., morbid obesity, low body weight, pregnancy, or those with reduced kidney function in whom other anticoagulants are not appropriate) [32]. Due to enoxaparin's greater impact on factor Xa than factor IIa (thrombin), antifactor Xa monitoring is preferred over other monitoring parameters, such as aPTT. Peak antifactor Xa activity is generally measured 4-6 hours after enoxaparin dosing once steady state is reached after the third or fourth dose of enoxaparin. Although slight differences may exist between institutions, an antifactor Xa goal range of 0.2-0.5 units/mL is generally acceptable for VTE prophylaxis, and a range of 0.6-1 unit/mL is acceptable for therapeutic dosing when enoxaparin is administered twice daily. If therapeutic enoxaparin is being dosed once daily (e.g., 1.5 mg/kg every 24 hours), an antifactor Xa range of 1-2 units/mL is typically used [32]. When transitioning from enoxaparin to a different anticoagulant, particular care should be utilized to ensure efficacy and safety. Current recommendations for transitioning between agents are provided in Table 6 [40].
ENOXAPARIN (SUBCUTANEOUS) TRANSITION TO OTHER AGENTS
| Transitioned to | Transition Strategy |
|---|---|
| Heparin (intravenous) | Begin intravenous heparin without a loading dose (rate based on indication) 1 to 2 hours before the next dose of enoxaparin would be due. |
| Warfarin (oral) | Begin warfarin and continue enoxaparin until INR is within therapeutic range (based on indication) for at least 2 measurements about 24 hours apart. |
| DOAC (oral)a | Begin DOAC within 2 hours prior to when the next scheduled dose of enoxaparin would be due. |
| aIncluding apixaban, dabigatran, rivaroxaban, and edoxaban | |
Warfarin (Coumadin) is an oral vitamin K antagonist that was initially approved in the United States in 1954. Until recently, warfarin had been the most widely prescribed oral anticoagulant in the management of a variety of thromboembolic and cardiovascular disease states, only to be replaced with the DOACs over the past decade. Warfarin provides its anticoagulant effect by inhibiting the synthesis of activated vitamin K-dependent clotting factors, including factors II, VII, IX, and X, and also the production of the body's natural anticoagulants, proteins C and S [43]. Warfarin is indicated in the prophylaxis and treatment of VTE, prophylaxis and treatment of thromboembolic complications from AF and cardiac valve replacement, and reduction in the risk of death, recurrent MI, or thromboembolic events after MI. Initial dosing of warfarin should be individualized based on patient factors including age, weight, and any relevant drug-drug interactions and adjusted based on INR response. Although variation exists based on patient factors, an initial starting warfarin dose in most patients is 5 mg once daily. Adverse reactions related to warfarin include the risk of bleeding, necrosis of skin and other tissues due to its effect on proteins C and S, and atheroemboli and cholesterol microemboli, among others. A few of the most relevant contraindications to warfarin include pregnancy (except in women with mechanical heart valves), recent surgery in critical areas, underlying bleeding tendencies or blood dyscrasias, and unsupervised use in patients with high potential for noncompliance [44,45]. In patients requiring treatment interruption before surgery, warfarin should be held at least 5 days prior to the procedure in order to allow time for the INR to normalize. In select patients at high risk of thromboembolic events, a shorter-acting anticoagulant such as heparin infusion may be utilized as a bridge during the perioperative period once INR becomes subtherapeutic and held closer to surgery, if required [35,44].
Monitoring and titration of warfarin dosing is achieved with the use of the INR. Target INR varies by indication, with the most common therapeutic range being 2–3 for indications such as the treatment of VTE and prevention of stroke in AF; however, some patients may require other target ranges based on risk of bleeding or indication (e.g., target range of 2.5–3.5 for management of mechanical mitral valve). Although some anticoagulant effect occurs within 24 hours of initiating warfarin, it is important to recognize that peak effect of warfarin may not be reached for 72 to 96 hours following administration, due to the variation in duration of half-lives of the inhibited coagulation factors. This complicates dosing of warfarin as a change in warfarin dose may not be fully reflected in the INR for multiple days [43,44].
In addition to warfarin's effect on INR, dosing titration can further be complicated by the impact of other endogenous and exogenous factors on INR as well as significant drug-drug interactions between warfarin and many other medications. Since INR reflects vitamin K-dependent clotting factors, patients with poor nutritional intake, hepatic disorders, or other reasons for vitamin K deficiency may have an increased INR, regardless of warfarin. It is, therefore, important to educate patients to have a consistent intake of foods that are rich in vitamin K, such as leafy greens. Additionally, drug-drug interactions can have a significant impact on warfarin's effect on INR and should be accounted for when initiating or adjusting warfarin doses. Warfarin can be impacted by both inhibitors and inducers of CYP2C9, CYP1A2, and CYP3A4 as well as antibiotics, antifungals, and herbal products. A few of the most notable CYP450 inhibitors that may increase warfarin concentrations include amiodarone, fluconazole, cotrimoxazole, ciprofloxacin, oral contraceptives, and voriconazole. Notable CYP450 inducers that may reduce concentrations of warfarin include carbamazepine, phenobarbital, rifampin, and phenytoin [43,44]. It is recommended to evaluate drug-drug interactions whenever medications are being changed in patients receiving warfarin. When transitioning from warfarin to a different anticoagulant, particular care should be utilized to ensure efficacy and safety. Current recommendations for transitioning between agents are provided in Table 7 [40].
WARFARIN (ORAL) TRANSITION TO OTHER AGENTS
| Transitioned to | Transition Strategy |
|---|---|
| Heparin (intravenous) | Discontinue warfarin and begin heparin once INR is ≤2 or lower INR limit of therapeutic range |
| Enoxaparin (subcutaneous) | Discontinue warfarin and begin enoxaparin once INR is ≤2 or lower INR limit of therapeutic range |
| DOAC (oral)a | Discontinue warfarin and begin DOAC once INR is <2 or lower INR limit of therapeutic range |
| aIncluding apixaban, dabigatran, rivaroxaban, or edoxaban | |
The direct thrombin inhibitors (DTIs) are a group of anticoagulants that provide their effects through direct inhibition of factor IIa (thrombin), as their name suggests. Through their reduction of thrombin generation, these agents ultimately prevent the conversion of fibrinogen into fibrin and the formation of a fibrin-bound clot. Currently, the only available oral DTI is dabigatran; however, intravenous agents, such as argatroban and bivalirudin, are also available for use as continuous infusions. Although argatroban and bivalirudin are important agents in the management of HIT and cardiac intervention as an alternative to heparin, this review will focus on the use of dabigatran.
Dabigatran etexilate (Pradaxa) was the first of the currently available direct oral anticoagulants to come to market with an original approval date in the United States in 2010. Currently, it is approved for reducing the risk of stroke and systemic embolism in patients with non-valvular AF, prevention and treatment of VTE, and VTE prophylaxis in total hip arthroplasty. Dabigatran is only available in oral formulations at this time, and dosing for common indications is listed below in Table 8. Due to dabigatran's renal elimination, use should be avoided in patients on hemodialysis or with a creatinine clearance < 30 mL/min, with the exception of a recommended renal dosage adjustment in patients with severe renal impairment (CrCl 15–30 mL/min) during the management of nonvalvular AF, specifically [46].
COMMON DABIGATRAN DOSING REGIMENS
| Indication | Dabigatran Dosing Regimen |
|---|---|
| AF (nonvalvular) | 150 mg orally every 12 hours |
| VTE treatment | 150 mg orally every 12 hours, after at least 5 days of initial therapy with a parenteral anticoagulant |
| VTE prophylaxis in total hip arthroplasty | 220 mg orally daily, after an initial dose of 110 mg or 220 mg based on timing from surgery and hemostasis |
Adverse reactions and precautions related to the use of dabigatran include an increased risk of bleeding as well as gastritis-like symptoms reported in greater than 15% of patients. Since dabigatran is known to be influenced by P-glycoprotein (P-gp), caution should be used when dabigatran is concomitantly administered with P-gp inducers, such as rifampin, or P-gp inhibitors, such as dronedarone. If dabigatran therapy interruption is required for surgery, it should be held 1 to 2 days prior to the procedure in patients without renal dysfunction (CrCl ≥50 mL/min) and for 3 to 5 days in patients with renal dysfunction (CrCl <50 mL/min). Although routine monitoring of dabigatran is not recommended, bleeding risk with dabigatran in select scenarios can best be assessed with use of the ecarin clotting time (ECT), if available, or aPTT if not [43,46]. When transitioning from dabigatran to a different anticoagulant, particular care should be utilized to ensure efficacy and safety. Current recommendations for transitioning between agents are provided in Table 9 [40].
DABIGATRAN (ORAL) TRANSITION TO OTHER AGENTS
| Transitioned to | Transition Strategy | ||
|---|---|---|---|
| Heparin (intravenous) |
| ||
| Enoxaparin (subcutaneous) |
| ||
| Warfarin (oral) | Dabigatran can elevate the INR and complicate warfarin dosing. Consider stopping dabigatran, beginning warfarin the same day, and bridging with a parenteral anticoagulant until INR is within therapeutic range. | ||
| Another DOAC (oral)a | Begin the new DOAC when the next dose of dabigatran would be due. | ||
| aIncluding apixaban, rivaroxaban, or edoxaban | |||
The factor Xa inhibitors are a group of anticoagulants that provide their effects through direct inhibition of factor Xa, as their name suggests. Through factor Xa inhibition, these agents ultimately decrease thrombin generation and prevent the formation of a fibrin-bound clot. Currently, the available oral factor Xa inhibitors include apixaban, rivaroxaban, and edoxaban, while fondaparinux is the only available agent for injection. Although betrixaban had previously been included within this class as well, it was removed from the market in the United States over the past few years. Despite these agents having the same mechanism for providing anticoagulation, important differences exist between these medications that must be recognized.
Rivaroxaban (Xarelto) was the second of the currently approved direct oral anticoagulants to be approved for use in the United States and was originally approved in 2011. Rivaroxaban currently has an extensive list of approved indications including nonvalvular AF, stable CAD, treatment and prevention of VTE, stable peripheral artery disease (PAD), and VTE prophylaxis following total hip or knee arthroplasty. Of note, rivaroxaban should not be used in the setting of prosthetic heart valves and valvular AF or in the setting of patients with triple positive antiphospholipid syndrome [47]. Most common adverse effects related to rivaroxaban include the risk of bleeding. Similar to enoxaparin, rivaroxaban carries a warning for the occurrence of epidural or spinal hematomas in patients treated with the drug and receiving neuraxial anesthesia or a spinal procedure. In patients requiring temporary discontinuation of therapy due to surgery or an invasive procedure, rivaroxaban should be held at least 24 hours prior to the procedure to reduce the risk of bleeding. Rivaroxaban dosing varies by indication, and due to some renal elimination of rivaroxaban, dosing alterations are often required in patients with renal dysfunction. Rivaroxaban should typically be avoided in patients on hemodialysis and those with CrCl <15 mL/min. Examples of common rivaroxaban dosing regimens are provided inTable 10 [47,48].
COMMON RIVAROXABAN DOSING REGIMENS
| Indication | Rivaroxaban Dosing Regimen |
|---|---|
| AF (nonvalvular) | 20 mg orally once daily with evening meal |
| VTE treatment | 15 mg orally twice daily with food for 21 days, followed by 20 mg once daily with food |
| CAD or stable PAD | 2.5 mg orally twice daily, in combination with low dose aspirin |
As with the other DOACs, routine coagulation monitoring of rivaroxaban is not required or recommended. In clinical situations where assessment of the anticoagulant effect is useful, prothrombin time (PT) may be useful to determine presence of rivaroxaban, though normal values cannot rule out use. In select institutions, antifactor Xa calibrated for rivaroxaban may be used if available, or antifactor Xa calibrated for LMWH can detect presence of drug concentrations but not accurately quantify the level of anticoagulation. Although drug interactions are not as common as with other agents such as warfarin, use with combined CYP3A4 and P-gp inducers or inhibitors should be avoided [43,47]. When transitioning from rivaroxaban to a different anticoagulant, particular care should be utilized to ensure efficacy and safety. Current recommendations for transitioning between agents are provided in Table 11 [40].
RIVAROXABAN (ORAL) TRANSITION TO OTHER AGENTS
| Transitioned to | Transition Strategy |
|---|---|
| Heparin (intravenous) | Begin heparin when the next dose of rivaroxaban would be due. |
| Enoxaparin (subcutaneous) | Begin enoxaparin when the next dose of rivaroxaban would be due. |
| Warfarin (oral) | Rivaroxaban can elevate the INR and complicate warfarin dosing. Consider stopping rivaroxaban, beginning warfarin the same day, and bridging with a parenteral anticoagulant until INR is within therapeutic range. |
| Another DOAC (oral)a | Begin new DOAC when the next dose of rivaroxaban would be due. |
| aIncluding apixaban, dabigatran, or edoxaban | |
Apixaban (Eliquis) is another direct oral anticoagulant that was initially approved for use in the United States in 2012. As of 2021, it is the most prescribed oral anticoagulant in the United States and has surpassed the use of other direct oral anticoagulants and warfarin, due to its enhanced safety profile [4]. Apixaban is currently approved for use in the management of AF, VTE, and postoperative VTE prophylaxis following hip or knee replacement surgery. Of note, apixaban is not currently approved for use in the management of prosthetic heart valves or valvular AF. The most common adverse reactions to apixaban are related to minor or major bleeding, though it has shown to be comparable, if not superior, to other agents in terms of bleeding risk. In patients requiring elective surgery or invasive procedures, apixaban should be discontinued at least 48 hours prior to procedures with a high-risk of bleeding and at least 24 hours prior to procedures with a low risk of bleeding or with procedures in a non-critical location where bleeding can be easily controlled [49]. Like other agents, apixaban carries a warning for the occurrence of epidural or spinal hematomas in patients treated with the drug and receiving neuraxial anesthesia or a spinal procedure. Apixaban is most often dosed at 5 mg orally twice daily; however, certain dosing nuances exist based on indication and patient factors [49]. Example dosing regimens for some of the most common indications are listed in Table12.
Similar to the other direct oral anticoagulants, routine coagulation monitoring of apixaban is not required or recommended. If assessment of apixaban's anticoagulant effect is required in select scenarios, PT or anti-factor Xa calibrated for LMWH can be used to determine the presence of relevant apixaban drug concentrations; however, this method does not provide accurate quantification of how much anticoagulant effect is present. Although drug interactions are not as common as with other agents such as warfarin, dose reduction or avoidance of use may be required in patients concomitantly receiving inhibitors or inducers of both CYP3A4 and P-gp [49,50]. When transitioning from apixaban to a different anticoagulant, particular care should be utilized to ensure efficacy and safety. Current recommendations for transitioning between agents are provided in Table 13 [40].
APIXABAN (ORAL) TRANSITION TO OTHER AGENTS
| Transitioned to | Transition Strategy |
|---|---|
| Heparin (intravenous) | Begin heparin when the next dose of apixaban would be due. |
| Enoxaparin (subcutaneous) | Begin enoxaparin when the next dose of apixaban would be due. |
| Warfarin (oral) | Apixaban can elevate the INR and complicate warfarin dosing. Consider stopping apixaban, beginning warfarin the same day, and bridging with a parenteral anticoagulant until INR is within therapeutic range. |
| Another DOAC (oral)a | Begin new DOAC when the next dose of apixaban would be due. |
| aIncluding rivaroxaban, dabigatran, or edoxaban | |
Edoxaban (Savaysa) was the next of the currently approved DOACs approved for use in the United States with an initial approval in 2015. Although this agent has some potential advantages over warfarin in select scenarios, edoxaban use has fallen out of favor, particularly due to its reduced efficacy in the treatment of nonvalvular AF in patients with creatinine clearance greater than 95 mL/min. Currently, edoxaban is approved for use in the management of nonvalvular AF and treatment of VTE, though certain limitations to its use exist. In addition to reduced efficacy in patients with nonvalvular AF with CrCl >95 mL/min, edoxaban is currently only approved for treatment of VTE following 5 to 10 days of initial parenteral anticoagulation, further limiting its use as an initial treatment option. Edoxaban should not be utilized in patients with mechanical heart valves, moderate-to-severe mitral stenosis, or those with triple positive antiphospholipid syndrome [51]. The most common adverse effects reported with edoxaban use include bleeding, as well as anemia, rash, and abnormal liver function tests. Edoxaban also carries the same warning for spinal and epidural hematoma in patients receiving neuraxial anesthesia or spinal puncture as other DOACs and enoxaparin. Concomitant use of rifampin or other agents that also increase risk of bleeding should be avoided, and dose reduction is required in patients who use certain P-gp inhibitors. The most common dosing regimen for edoxaban is 60 mg once daily in patients with a CrCl 51–95 mL/min, and a dosage reduction to 30 mg once daily is required for patients with a CrCl 15–50 mL/min or in patients with low body weight (≤60 kg) when being treated for VTE, specifically. In patients requiring temporary discontinuation of therapy due to surgery or an invasive procedure, edoxaban should be held at least 24 hours prior to the procedure to reduce the risk of bleeding [51].
Similar to the other DOACs, routine coagulation monitoring of edoxaban is not required or recommended in patients receiving this medication. In clinical situations where assessment of the anticoagulant effect is useful, PT may be useful to determine presence of edoxaban, though normal values cannot rule out use. In select institutions, antifactor Xa calibrated for edoxaban may be used if available, or antifactor Xa calibrated for LMWH can detect presence of drug concentrations but not accurately quantify the level of anticoagulation [52]. When transitioning from edoxaban to a different anticoagulant, particular care should be utilized to ensure efficacy and safety. Current recommendations for transitioning between agents are provided in Table 14.
EDOXABAN (ORAL) TRANSITION TO OTHER AGENTS
| Transitioned to | Transition Strategy |
|---|---|
| Heparin (intravenous) | Begin heparin when the next dose of edoxaban would be due. |
| Enoxaparin (subcutaneous) | Begin enoxaparin when the next dose of edoxaban would be due. |
| Warfarin (oral) | Edoxaban can elevate the INR and complicate warfarin dosing. Consider stopping edoxaban, beginning warfarin the same day, and bridging with a parenteral anticoagulant until INR is within therapeutic range. |
| Another DOAC (oral)a | Begin new DOAC when the next dose of edoxaban would be due. |
| aIncluding apixaban, dabigatran, or rivaroxaban | |
As a whole, atherosclerotic cardiovascular disease (ASCVD) is the leading cause of morbidity and mortality worldwide [53]. ASCVD-related conditions are caused by the buildup of plaque in arterial walls, which can then lead to a variety of disease processes including acute coronary syndromes, ischemic stroke and TIA, PAD, and coronary or carotid arterial stenosis. Although each disease process has its own unique management, the use of antithrombotics remains a standard of practice in the prevention and treatment of each of these ASCVD-related disease states. One of the most significant changes in the management of ASCVD-related disease states over the past few years has been the change in recommendations for providing aspirin as primary prevention of ASCVD. Historically, aspirin has been widely administered for ASCVD prevention in many patient populations. As of the 2019 ACC/AHA Guideline on the Primary Prevention of Cardiovascular Disease, aspirin is no longer recommended for widespread use but rather limited to patients who may receive the greatest benefit with the least risk of harm. The guideline provides a Class 2b recommendation to consider utilizing low-dose aspirin (75–100 mg orally daily) for primary prevention of ASCVD in patients aged 40 to 70 years who are at a higher risk of ASCVD but no increased risk of bleeding [53]. Additionally, these guidelines go on to recommend against the use of low-dose aspirin routinely for primary prevention of ASCVD in patients older than 70 years of age (Class 3, potential harm) or in patients of any age who are at an increased risk of bleeding (Class 3, potential harm). Examples of increased bleeding risk include those patients with a history of peptic ulcer disease or gastrointestinal bleeding, patients with thrombocytopenia or coagulopathy, or patients concomitantly taking other medications that also increase the risk of bleeding such as nonsteroidal anti-inflammatory drugs, steroids, and other anticoagulants. In patients being considered for primary prevention of ASCVD, the guidelines recommend utilizing ASCVD risk estimators such as the pooled cohort equations or evaluating patient-specific ASCVD risk factors to assess which patients may benefit from the use of aspirin in appropriate age ranges.
Chronic coronary disease (CCD) is an encompassing term that includes obstructive and nonobstructive CAD, ischemic heart disease, and chronic angina symptoms. Within the United States, approximately 20.1 million people live with CCD, and despite significant advancements in management, it remains the leading cause of death both in the United States and worldwide [54]. In patients with CAD, an atherosclerotic narrowing of the coronary arteries occurs, potentially limiting the flow of blood to vital areas of the heart. Although many patients with CAD may remain asymptomatic, this disease can progress to the point of causing symptoms or complications that must be managed via medications or intervention. Antiplatelets and anticoagulants can be used in the management of CCD to prevent atherosclerotic plaque from forming blockages within the coronary vasculature or other cardiovascular complications leading to comorbidity or death. For simplicity, antithrombotic recommendations will be discussed in the context of stable CCD not requiring revascularization and CCD requiring revascularization (e.g., PCI or CABG), while acute coronary syndrome will be discussed separately.
As per the 2023 AHA/ACC/ACCP/ASPC/NLA/PCNA guidelines for patients with CCD, low-dose aspirin 81 mg monotherapy is recommended in order to reduce atherosclerotic events in patients with stable CCD and no indication for anticoagulant therapy (Class 1). In patients with CCD requiring revascularization, guideline recommendations become a bit more nuanced, based on intervention. In patients with CCD treated with PCI, dual antiplatelet therapy (DAPT) with aspirin and clopidogrel for 6 months post-PCI followed by single antiplatelet therapy (SAPT) is recommended (Class 1). In patients with CCD treated with PCI and drug-eluting stent (DES) who completed 1-3 months of DAPT, monotherapy with a P2Y12 inhibitor for at least 12 months is reasonable for management (Class 2a). Additional recommendations of a lower class include that it may be reasonable for patients with CCD and history of MI to have their duration of DAPT be extended from 12 months to up to 3 years (Class 2b) and that the use of DAPT after CABG may be useful to reduce the incidence of saphenous vein graft occlusion (Class 2b) [54].
In patients with CCD who have electively been treated with PCI and require a DOAC for another indication, DAPT should be administered for 1 to 4 weeks followed by SAPT with clopidogrel for 6 months in addition to the DOAC (Class 1). If it is determined that a patient in this category has a high thrombotic risk and low bleeding risk, DAPT with aspirin and clopidogrel can be extended for up to 1 month in addition to the DOAC (Class 2a). In patients with CCD who require a DOAC and receive PCI, discontinuation of aspirin therapy may be considered after one year from PCI with continuation of DOAC alone, if low atherothrombotic risk exists (Class 2b). Additionally, DOAC monotherapy may be considered over aspirin in patients with CCD who already require DOAC use and have no acute indication for concomitant antiplatelet therapy (Class 2b). Lastly, a select population may benefit from low-dose aspirin 81 mg daily plus low-dose rivaroxaban 2.5 mg twice daily in patients with CCD who do not have an indication for therapeutic DOAC or DAPT, are high risk of recurrent ischemic events, and have a low-to-moderate bleeding risk (Class 2a) [54].
Acute coronary syndrome (ACS) is a common yet devastating event that occurs in nearly 1 million patients each year in the United States, with a mortality rate of up to 14% of these events [17]. ACS is an encompassing term that includes STEMI, NSTEMI, and unstable angina. Most commonly, ACS occurs due to atherosclerotic plaque rupture leading to thrombus formation and coronary artery occlusion; however, additional mechanisms such as coronary dissection, vasospasm, embolism, or arteritis may lead to this event as well. In the occurrence of disrupted plaques, exposure of thrombogenic material within the plaque leads to platelet adhesion, further platelet activation via P2Y12 receptors and glycoprotein IIb/IIIa receptors, and ultimately platelet aggregation and thrombus formation. With this progression in mind, it's easy to see the rationale for utilizing antiplatelet medications to prevent thrombus formation in ACS, and the importance of these medications is even greater in patients requiring emergent PCI, where stretching of the arterial wall can further disrupt the epithelium and cause additional platelet activation, potentially leading to stent thrombosis and in-stent restenosis in select patients [17].
In the initial treatment for ACS, current guidelines recommend combination of DAPT and anticoagulant therapy regardless of the choice of invasive (e.g. PCI) or conservative (e.g. medical) management strategies. As soon as ACS is suspected, aspirin 162–325 mg should be administered (chewed) and continued indefinitely unless contraindications exist. Parenteral anticoagulants including unfractionated heparin, enoxaparin, bivalirudin, or fondaparinux should be utilized in the initial period for up to 48 hours or until PCI is performed in patients with NSTEMI (Class 1), and parenteral anticoagulation using unfractionated heparin (with or without an intravenous glycoprotein IIb/IIIa inhibitor) or bivalirudin should be used to assist in emergent revascularization (e.g., PCI) in patients with STEMI (Class 1) [55,56,57]. Generally speaking, anticoagulation does not need to be continued long-term in these patients unless a patient has an additional indication for anticoagulation, such as AF, or in the select use of low-dose anticoagulation plus antiplatelet agent(s).
From an antiplatelet standpoint, DAPT is currently the cornerstone of management and typically includes aspirin (begun as soon as possible when ACS is suspected) plus a P2Y12 inhibitor such as clopidogrel, prasugrel, or ticagrelor (begun as soon as possible if conservative management is chosen or at the time of intervention with PCI, in applicable patients). DAPT has been proven to reduce the risk of recurrent ischemic events in those with ACS. Despite current guidelines favoring the use of the newer, more potent P2Y12 inhibitors such as prasugrel or ticagrelor, clopidogrel remains one of the most commonly used agents in the United States [58]. In the 2014 ACC/AHA guidelines for the management of NSTEMI, clopidogrel or ticagrelor is recommended, with a preference for ticagrelor; however, prasugrel is also recommended in patients undergoing PCI who do not have a high risk for bleeding. Since the release of the guidelines, additional studies have continued to evaluate the use of these newer P2Y12 inhibitors in patients with ACS. Results from comparisons of these medications have led to changes in the most recent European guidelines for ACS, which now recommend considering prasugrel over ticagrelor in patients with ACS who are proceeding to PCI (Class 2a) [59]. Caution should still be utilized when using prasugrel in patients 75 years of age or older due to potential increase in bleeding, and this agent remains contraindicated in patients with a history of stroke.
An important decision in the management of ACS includes the duration of antiplatelet therapy following the event. Within the first 30 days after ACS, the risk of thrombosis tends to be greater than the risk of bleeding, making this a time frame when patients are particularly vulnerable to recurrent events; however, the risk of thrombosis significantly declines after those initial 30 days and the risk of bleeding may be held in higher regard. For these reasons, a balanced risk-assessment that includes both the risk of thrombosis and the risk of bleeding should be incorporated. According to the 2016 ACC/AHA guidelines, both aspirin and a P2Y12 inhibitor should be given for at least 12 months in patients with ACS requiring DAPT (Class 1). In select patients who do not have a high risk of bleeding, DAPT may be continued beyond 12 months if deemed beneficial (Class 2b); alternatively, in select patients requiring DAPT who develop significant bleeding or have a high risk of bleeding, aspirin monotherapy can be considered after 6 months of DAPT, with discontinuation of P2Y12 inhibitor therapy (Class 2b) [58]. In patients with ACS undergoing CABG, the P2Y12 inhibitor may be held or discontinued prior to surgery; however, it should be resumed following CABG and continued as part of DAPT to complete one year of treatment (Class 1) [58].
Atrial fibrillation (AF) is the most common arrhythmia in the world and is associated with significant morbidity and mortality. AF was estimated to be present in 5.2 million people in the United States in 2010, and due to an aging population, increased detection, and other factors, it is expected to rise to over 12 million people by 2030. AF is associated with a 1.5- to 2-fold increased risk of death and carries a significant burden of cost and resource utilization in the healthcare system [61]. AF is commonly described as an "irregularly irregular" rhythm, in which chaotic rapid atrial beating leads to the potential for complications and poor outcomes in patients. Management of AF is multifaceted and includes lifestyle and risk factor modification, AF burden mitigation with rate or rhythm control, and reduction in the risk of stroke and systemic embolism with the use of anticoagulants. All patients with AF should be evaluated for their thromboembolic risk using validated clinical risk scores such as the CHA2DS2-VASc score, which can provide an annual risk assessment of thromboembolic events. In patients with an intermediate annual risk of thromboembolic events (e.g., CHA2DS2-VASc score of 1 in men or 2 in women), anticoagulation should be considered along with an overall assessment of potential risks and benefits (Class 2a), and in patients with AF and an estimated annual thromboembolic risk of ≥2% (e.g., CHA2DS2-VASc score ≥2 in men or ≥3 in women), anticoagulation is recommended, in order to prevent stroke and systemic embolism (Class 1) [61].
In terms of anticoagulant choice for AF, current guidelines recommend the use of DOACs over warfarin, except in the setting of mitral stenosis or mechanical heart valve (Class 1) and recommend against the use of aspirin either alone or in combination with clopidogrel for the management of AF (Class 3, harm/no benefit). The superiority of DOACs over warfarin in patients with AF has been consistent in the DOAC trials, including Re-LY, ROCKET AF, ARISTOTLE, and ENGAGE AF-TIMI 48. In patients who are required to receive warfarin, a target INR range 2–3 is recommended, along with routine INR monitoring, a diet with consistent vitamin K intake, and regular drug-drug interaction evaluations (Class 1). In patients with AF who require temporary interruption of oral anticoagulation therapy for an invasive procedure or surgery, bridging with an intravenous anticoagulant (e.g., heparin) is generally not required, except in the setting of patients with high thromboembolic risk, such as those with mechanical heart valve [61].
Although guidelines for patients with isolated AF are relatively straightforward, AF often complicates other disease states that also require antithrombotic therapy, posing new risks and considerations. In patients with AF complicated by ACS or those undergoing PCI, indications for DAPT plus anticoagulation therapy (i.e., triple therapy) may exist; however, triple therapy has been shown to significantly increase the risk of bleeding and should be minimized whenever possible. In the current AF guidelines, it is recommended in patients with AF who undergo PCI; DOACs along with antiplatelet therapy are preferred over warfarin and antiplatelet therapy. In most of these patients, triple therapy with DAPT and a DOAC may be required initially; however, early discontinuation of aspirin after 1-4 weeks and continuation of a P2Y12 inhibitor plus DOAC is preferred over continued use of triple therapy, in order to reduce the risk of bleeding (Class 1). Additionally, patients with CCD may also have AF, and although patients with CCD typically require aspirin, current guidelines recommend that patients with both CCD and AF utilize monotherapy with an oral anticoagulant alone in order to decrease the risk of major bleeding (Class 1). Although a lower class of recommendation, a similar recommendation exists for patients with stable PAD and concomitant AF. In this patient population, monotherapy with an oral anticoagulant is deemed reasonable over therapy with an anticoagulant plus an antiplatelet agent (Class 2a) [61].
JP is a 76-year-old male with a past medical history significant for coronary artery disease, asthma, hypertension, and hyperlipidemia who presents to the hospital due to complaints of feeling extremely fatigued and having a very fast pounding feeling in his chest. After initial assessment and electrocardiogram, JP is diagnosed with new onset atrial fibrillation and initiated on metoprolol 25 mg orally BID for rate control. After calculating his CHA2DS2-VASc score, the primary team determines that JP is at greater than 2% annual risk of ischemic stroke and requires anticoagulation as well. Prior to admission, JP was taking aspirin 81 mg orally daily, lisinopril 20 mg orally daily, atorvastatin 20 mg orally daily, and an albuterol inhaler as needed.
Study Questions
Based on current guidelines, what anticoagulant(s) should JP be started on?
What other components of his antithrombotic regimen may need to change?
Does JP require any anticoagulant-specific monitoring based on his new medication regimen?
Discussion
According to the 2023 ACC/AHA/ACCP/HRS guidelines for the diagnosis and management of atrial fibrillation, it is recommended that JP be initiated on anticoagulation for prevention of stroke and systemic embolism from atrial fibrillation based on his CHA2DS2-VASc score. As this is nonvalvular atrial fibrillation, first-line therapy includes any of the DOACs (e.g., apixaban, rivaroxaban, dabigatran, or edoxaban), based on their enhanced safety profile over vitamin K antagonists (e.g., warfarin). When selecting a specific agent, edoxaban should be avoided if JP's CrCl is greater than 95 mL/min; however, the remainder of the decision may be based on insurance coverage, patient preference (e.g., once-daily dosing versus twice-daily dosing), drug-interactions, or availability.
Since JP was taking an aspirin for his CAD prior to admission, this must also be addressed. According to the same AF guidelines, monotherapy with an oral anticoagulant alone should be used in patients with CCD and AF in order to decrease the risk of major bleeding. For this reason, JP's aspirin may be discontinued now that he is being started on an oral anticoagulant.
As for specific monitoring, JP will not require routine monitoring with any of the DOACs, another benefit of utilizing these agents over older agents such as warfarin.
Acute ischemic stroke (AIS) and transient ischemic attack (TIA) are related neurologic events with significant disability but common occurrence worldwide. Acute ischemic stroke and TIA can both be described as a blockage that prevents blood flow from reaching various areas of the brain, though in TIA this blockage is brief and does not cause permanent damage. In the United States alone, nearly 700,000 people experience an ischemic stroke, while 240,000 people experience a TIA each year [62,63]. The risk of recurrent stroke or TIA is high, with estimates of approximately 1 in 25 people having a recurrent event within 30 days; however, this risk can be mitigated with appropriate secondary prevention. In the initial acute management of stroke or TIA, the focus is to ensure that cerebrovascular blood flow is restored to all affected areas if possible. This is typically done through the use of thrombolytics (e.g., alteplase or tenecteplase) that break down the clot as well as the use of endovascular therapies in select patient populations. Following initial management, secondary prevention should be initiated after 24 hours in patients who received thrombolytic therapy or immediately if no thrombolytic therapy was administered. The choice of antithrombotic regimen and duration can be impacted significantly by the etiology as well as severity. In patients with stroke or TIA caused by non-valvular AF, anticoagulation with a DOAC is recommended over warfarin (except in the setting of severe mitral stenosis or mechanical heart valve) or antiplatelet therapy (Class 1). In patients with stroke or TIA and valvular AF, warfarin is recommended to reduce the risk of recurrent stroke or TIA (Class 1). In patients with stroke or TIA caused by significant (50% to 99%) stenosis of a major intracranial artery, aspirin 325 mg daily is recommended (Class 1), though the addition of clopidogrel 75 mg per day may be considered for up to 90 days in patients with severe stenosis (70% to 99%) of a major intracranial artery (Class 2a). Patients with stroke or TIA and left ventricular thrombus (LV) thrombus should receive anticoagulation with warfarin for at least 3 months (Class 1), though other guidance documents have discussed the potential use of DOACs in select patients based on more recent data [64]. In patients with a noncardioembolic ischemic stroke or TIA, antiplatelet therapy is preferred over anticoagulant therapy and should be dependent on the severity of disease. In patients with noncardioembolic minor stroke (NIHSS score ≤3) or high-risk TIA (ABCD2 score ≥4), DAPT with aspirin plus clopidogrel should be initiated and continued for 21 to 90 days, followed by single antiplatelet therapy (Class 1). In patients with non-cardioembolic stroke or TIA who do not meet the aforementioned criteria, single antiplatelet therapy should be used [63].
Valvular heart disease (VHD) occurs when any valve in the heart is damaged or diseased, and it is a term that includes conditions such as regurgitation or stenosis of various valves. VHD can have numerous etiologies including congenital conditions (e.g., malformations of the valves), infection (e.g., infective endocarditis), rheumatic disease, or conditions linked to other types of heart disease (e.g., heart failure, atherosclerosis, aortic aneurysm, or MI). In the United States, approximately 2.5% of people have VHD, with higher representation in older adults, and almost 30,000 deaths can be traced back to the disease annually [67]. Although many patients may be managed conservatively, intervention with valve repair or replacement is often required in more severe disease. Following prosthetic valve implantation with either a bioprosthetic or mechanical heart valve, antithrombotic therapy is a cornerstone of management in order to prevent valve or leaflet thrombosis and reduce the incidence of thromboembolic complications.
When considering the most appropriate antithrombotic regimen for patients with valve replacement, the most important point to evaluate is the type of prosthetic valve that was placed (e.g., mechanical or bioprosthetic). In patients with a mechanical prosthetic valve, current guidelines recommend therapeutic anticoagulation with a vitamin K antagonist, such as warfarin (Class 1). Of note, DOACs are contraindicated in patients with a mechanical valve prosthesis and should be avoided (Class 3, harm). Target INR goal for patients with a mechanical valve varies based on location of the valve and the presence of other risk factors for thromboembolism, such as AF, previous thromboembolism, LV dysfunction, hypercoagulable state, or older generation of prosthesis such as ball-in-cage valve. In patients with a mechanical aortic valve replacement (AVR) and no other risk factors for thromboembolism, a vitamin K antagonist with a target INR range of 2–3 is recommended (Class 1). In patients with a mechanical mitral valve alone or those with a mechanical AVR and risk factors for thromboembolism, a vitamin K antagonist with a target INR range of 2.5–3.5 is recommended (Class 1). In any patient with a mechanical valve receiving warfarin, the addition of aspirin 75–100 mg daily may be considered if additional antiplatelet therapy is indicated and risk of bleeding is low (Class 2b) [68].
In patients who receive a bioprosthetic valve, the risk of thromboembolism is comparatively lower than those with a mechanical valve and can often be managed by antiplatelets alone or after a short course of anticoagulation following valve replacement in those who are at a low risk of bleeding. In all patients with a bioprosthetic AVR or MVR, aspirin 75–100 mg daily as lifelong therapy is reasonable in the absence of other indications for anticoagulation, including surgical AVR or MVR and transcatheter aortic valve implantation (TAVI) (Class 2a). In patients with a surgical bioprosthetic AVR or MVR who are at a low risk of bleeding, anticoagulation with a vitamin K antagonist is reasonable for the first 3 to 6 months with a goal INR range of 2–3 (Class 2a). In patients receiving TAVI who are at a low risk of bleeding, anticoagulation with a vitamin K antagonist with a goal INR range of 2–3 or DAPT with aspirin 75–100 mg orally daily plus clopidogrel 75 mg orally daily (Class 2b) are reasonable options for the first three to six months (Class 2b) [68].
The selection of antithrombotic therapy in patients with VHD may also be further complicated by the presence of AF when determining whether to use a vitamin K antagonist or DOAC. In general, patients who have established AF plus rheumatic mitral stenosis, mechanical AVR, or mechanical MVR should receive long-term anticoagulation with a vitamin K antagonist (Class 1), and in those with new onset AF within three months of bioprosthetic valve implantation, anticoagulation with a vitamin K antagonist is reasonable (Class 2a). In patients with AF and VHD who have not received a prosthetic valve replacement (native valve disease that is not rheumatic mitral stenosis) or in those with a bioprosthetic valve who have AF occurring greater than three months after replacement may receive DOAC, if anticoagulation is indicated based on CHA2DS2-VASc score (Class 1) [68].
Venous thromboembolism (VTE) is the overarching term that includes both DVT and PE. In the United States, approximately 300,000 to 600,000 VTE events occur annually, causing significant morbidity for patients and increased resource utilization in healthcare settings [69]. The occurrence of VTE is often multicausal and is likely triggered by interactions between multiple risk factors. VTE development is often traced back to Virchow's triad, which is the combination of stasis, vascular wall damage or dysfunction, and hypercoagulability [70]. Blood stasis in valve pockets of large vessels is thought to predispose the formation of venous thrombi, especially during times of immobilization. Vascular damage or dysfunction in the area further leads to an increased expression of surface adhesion molecules and ultimately platelet activation and adhesion. Through the increased inflammation and activation of leukocytes, the extrinsic pathway of the coagulation cascade is thought to be activated, resulting in the formation of a fibrin-bound clot. Once this thrombus forms, it may lead to blockages within the vessel or embolize to other areas of the body, such as in the case of PE [70].
The cornerstone of therapy for VTE centers around anticoagulation. Currently, guidelines from both the American Society of Hematology and the American College of Chest Physicians recommend DOACs over vitamin K antagonists for the treatment of DVT and PE, due to their enhanced safety and efficacy profiles, and any of the DOACs can be used (strong recommendation) [71]. In terms of duration of therapy, anticoagulation with an oral anticoagulant should be continued for at least three months, followed by an assessment for the need to continue therapy (strong recommendation). In patients with VTE who are unable to receive a DOAC or those who have antiphospholipid syndrome, vitamin K antagonists with a target INR range of 2–3 may be used (weak recommendation). In select patients with a chronic risk factor or unprovoked VTE, extended duration of therapy beyond three to six months may be required. One such population includes patients who develop acute VTE in the setting of cancer, where anticoagulation may be continued beyond standard therapy and often indefinitely. Although LMWH had previously been the anticoagulant of choice in patients with cancer-associated thrombosis, guidelines now recommend that DOACs, including apixaban, edoxaban, or rivaroxaban, be used over LMWH for the initiation and treatment of VTE in these patients, with the exception of patients with luminal gastrointestinal malignancies (strong recommendation) [71]. In these patients, apixaban or LMWH is preferred, due to increased risk of gastrointestinal bleeding with edoxaban and rivaroxaban.
Peripheral arterial disease (PAD) of the lower extremity refers to the atherosclerotic obstruction of vessels within the region from the aortoiliac segments to the pedal arteries. Recent estimates report that PAD is the third leading cause of atherosclerotic morbidity after coronary heart disease and stroke and is present in approximately 7% of adults 40 years of age or older in the United States [72]. PAD can range in severity from asymptomatic disease to critical limb ischemia and require significant intervention if left untreated. Despite strong recommendations with high levels of evidence in most recent guidelines, patients with PAD remain undertreated and adherence to antiplatelet use in symptomatic patients at hospital discharge is reported to be as low as 57.7% to 67.3% in the literature [72].
In most recent guidelines, antiplatelet therapy is recommended in symptomatic patients in order to reduce the risk of MI, stroke, or vascular death (Class 1) and should be considered in asymptomatic patients as well (Class 2a). Currently, this can include monotherapy with aspirin 75–325 mg orally daily or clopidogrel 75 mg orally daily (Class 1). DAPT may also be beneficial in select patients with PAD, including those with symptomatic PAD after lower extremity revascularization (Class 2b). In patients progressing to acute limb ischemia, systemic anticoagulation with heparin should be provided to all patients unless contraindicated (Class 1) and revascularization with either surgery or intra-arterial thrombolysis should be performed [73,74].
The use of antithrombotics, including both antiplatelets and anticoagulants, can be complex; however, healthcare providers can improve the use of these agents in various healthcare settings by understanding the underlying mechanisms, drug properties, monitoring strategies, and indications for use of these agents. By applying this knowledge, healthcare providers are able to improve patient outcomes and avoid adverse effects that lead to patient harm. As with all areas of medicine, the indications for antiplatelets and anticoagulants continue to change, and it is important to be mindful of updates and advancements as treatment options change in the future.
1. Montinari MR, Minelli S, De Caterina R. The first 3500 years of aspirin history from its roots—a concise summary. Vascul Pharmacol. 2019;113:1-8.
2. Pirmohamed M. Warfarin: almost 60 years old and still causing problems. Br J Clin Pharmacol. 2006;62(5):509-511.
3. Boakye E, Uddin SMI, Obisesan OH, et al. Aspirin for cardiovascular disease prevention among adults in the United States: trends, prevalence, and participant characteristics associated with use. Am J Prev Cardiol. 2021;8:100256.
4. ClinCalc.com. ClinCalc DrugStats Database: The Top 200 Drugs of 2021. Available at https://clincalc.com/DrugStats/. Last accessed June 10, 2024.
5. Barnes GD, Lucas E, Alexander GC, Goldberger ZD. National trends in ambulatory oral anticoagulant use. Am J Med. 2015;128(12):1300-1305.e2.
6. Navar AM, Kolkailah AA, Overton R, et al. Trends in oral anticoagulant use among 436 864 patients with atrial fibrillation in community practice, 2011 to 2020. J Am Heart Assoc. 2022;11(22):e026723.
7. Budnitz DS, Shehab N, Lovegrove MC, Geller AI, Lind JN, Pollock DA. US emergency department visits attributed to medication harms, 2017–2019. JAMA. 2021;326(13):1299-1309.
8. Davies EC, Green CF, Taylor S, Williamson PR, Mottram DR, Pirmohamed M. Adverse drug reactions in hospital in-patients: a prospective analysis of 3695 patient-episodes. PLoS One. 2009;4(2):e4439.
9. Geller AI, Shehab N, Lovegrove MC, et al. Emergency visits for oral anticoagulant bleeding. J Gen Int Med. 2020;35(1):371-373.
10. Geller AI, Shehab N, Lovegrove MC, Weidle NJ, Budnitz DS. Bleeding related to oral anticoagulants: trends in US emergency department visits, 2016–2020. Thromb Res. 2023;225:110-115.
11. Pirmohamed M, James S, Meakin S, et al. Adverse drug reactions as cause of admission to hospital: prospective analysis of 18 820 patients. BMJ. 2004;329:15-19.
12. Dreijer AR, Diepstraten J, Bukkems VE, et al. Anticoagulant medication errors in hospitals and primary care: a cross-sectional study. Int J Qual Health Care. 2019;31(5):346-352.
13. Burnett AE, Barnes GD. A call to action for anticoagulation stewardship. Res Pract Thromb Haemost. 2022;6(5):e12757.
14. Koolian M, Wiseman D, Mantzanis H, Kampouris N, Kerzner RS, Kahn SR. Anticoagulation stewardship: descriptive analysis of a novel approach to appropriate anticoagulant prescription. Res Pract Thromb Haemost. 2022;6(6):e12758.
15. Smith SA, Travers RJ, Morrissey JH. How it all starts: initiation of the clotting cascade. Crit Rev Biochem Mol Biol. 2015;50(4):326-336.
16. Scridon A. Platelets and their role in hemostasis and thrombosis—from physiology to pathophysiology and therapeutic implications.Int J Mol Sci. 2022;23(21):12772.
17. Kamran H, Jneid H, Kayani WT, et al. Oral antiplatelet therapy after acute coronary syndrome: a review. JAMA. 2021;325(15):1545-1555.
20. LexiDrug. Aspirin. Available at http://online.lexi.com. Last accessed June 10, 2024.
21. Angiolillo DJ, Rollini F, Storey RF, et al. International expert consensus on switching platelet p2y12 receptor-inhibiting therapies. Circulation. 2017;136(20):1955-1975.
24. LexiDrug. Prasugrel. Available at http://online.lexi.com. Last accessed June 10, 2024.
26. Wallentin L, Becker RC, Budaj A, et al. Ticagrelor versus clopidogrel in patients with acute coronary syndromes. N Engl J Med. 2009;361(11):1045-1057.
27. Bonaca MP, Bhatt DL, Cohen M, et al. Long-term use of ticagrelor in patients with prior myocardial infarction. N Engl J Med. 2015;372(19):1791-1800.
28. Mahaffey KW, Wojdyla DM, Carroll K, et al. Ticagrelor compared with clopidogrel by geographic region in the platelet inhibition and patient outcomes (PLATO) trial. Circulation. 2011;124(5):544-554.
29. Angiolillo DJ, Firstenberg MS, Price MJ, et al. Bridging antiplatelet therapy with cangrelor in patients undergoing cardiac surgery: a randomized controlled trial. JAMA. 2012;307(3):265-274.
30. Angiolillo DJ, Been L, Rubinstein M, Martin M, Rollini F, Franchi F. Use of the VerifyNow point of care assay to assess the pharmacodynamic effects of loading and maintenance dose regimens of prasugrel and ticagrelor. J Thromb Thrombolysis. 2021;51(3):741-747.
31. Mason GA, Rabbolini DJ. The current role of platelet function testing in clinical practice. Semin Thromb Hemost. 2021;47(07):843-854.
32. Garcia DA, Baglin TP, Weitz JI, Samama MM. Parenteral anticoagulants: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 2012;141(2 Suppl):e24S-e43S.
34. LexiDrug. Heparin. Available at http://online.lexi.com. Last accessed June 10, 2024.
35. Douketis JD, Spyropoulos AC, Spencer FA, et al. Perioperative management of antithrombotic therapy: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 2012;141(2 Suppl):e326S-e350S.
36. Vandiver JW, Vondracek TG. Antifactor Xa levels versus activated partial thromboplastin time for monitoring unfractionated heparin. Pharmacother: J Human Pharmacol Drug Ther. 2012;32(6):546-558.
37. Rosborough TK. Monitoring unfractionated heparin therapy with antifactor Xa activity results in fewer monitoring tests and dosage changes than monitoring with the activated partial thromboplastin time. Pharmacother: J Human Pharmacol Drug Ther. 1999;19(6):760-766.
38. Guervil DJ, Rosenberg AF, Winterstein AG, Harris NS, Johns TE, Zumberg MS. Activated partial thromboplastin time versus antifactor Xa heparin assay in monitoring unfractionated heparin by continuous intravenous infusion. Ann Pharmacother. 2011;45 (7-8):861-868.
39. Levy JH, Sniecinski RM, Rocca B, et al. Defining heparin resistance: communication from the ISTH SSC subcommittee of perioperative and critical care thrombosis and hemostasis. J Thromb Haemost. 2023;21(12):P3649-P3657.
40. Chen A, Stecker E, Warden BA. Direct oral anticoagulant use: a practical guide to common clinical challenges. J Am Heart Assoc. 2020;9(13):e017559.
42. LexiDrug. Enoxaparin. Available at http://online.lexi.com. Last accessed June 10, 2024.
43. Ageno W, Gallus AS, Wittkowsky A, Crowther M, Hylek EM, Palareti G. Oral anticoagulant therapy. Chest. 2012;141(2 Suppl):e44S-e88S.
45. LexiDrug. Warfarin. Available at http://online.lexi.com. Last accessed June 10, 2024.
48. LexiDrug. Rivaroxaban. Available at http://online.lexi.com. Last accessed June 10, 2024.
50. LexiDrug. Apixaban. Available at http://online.lexi.com. Last accessed June 10, 2024.
52. LexiDrug. Edoxaban. Available at http://online.lexi.com. Last accessed June 10, 2024.
53. Arnett DK, Blumenthal RS, Albert MA, et al. 2019 ACC/AHA guideline on the primary prevention of cardiovascular disease: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2019;140(11):e596-e646.
54. Virani SS, Newby LK, Arnold SV, et al. 2023 AHA/ACC/ACCP/ASPC/NLA/PCNA guideline for the management of patients with chronic coronary disease: a report of the American Heart Association/American College of Cardiology Joint Committee on Clinical Practice Guidelines. Circulation. 2023;148(9):e9-e119.
55. Rodriguez F, Harrington RA. Management of antithrombotic therapy after acute coronary syndromes. N Engl J Med. 2021;384(5):452-460.
56. Amsterdam EA, Wenger NK, Brindis RG, et al. 2014 AHA/ACC guideline for the management of patients with non–ST-elevation acute coronary syndromes: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation. 2014;130(25):2354-2394.
57. O'Gara PT, Kushner FG, Ascheim DD, et al. 2013 ACCF/AHA guideline for the management of ST-elevation myocardial infarction: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Circulation. 2013;127(4):e362-e425.
58. Levine GN, Bates ER, Bittl JA, et al. 2016 ACC/AHA guideline focused update on duration of dual antiplatelet therapy in patients with coronary artery disease: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2016;68(10):1082-1115.
59. Byrne RA, Rossello X, Coughlan JJ, et al. 2023 ESC guidelines for the management of acute coronary syndromes. Eur Heart J. 2023;44(38):3720-3826.
60. Schüpke S, Neumann FJ, Menichelli M, et al. Ticagrelor or prasugrel in patients with acute coronary syndromes. N Engl J Med. 2019;381(16):1524-1534.
61. Joglar JA, Chung MK, Armbruster AL, et al. 2023 ACC/AHA/ACCP/HRS guideline for the diagnosis and management of atrial fibrillation: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2024;149(1):e1-e156.
62. Mendelson SJ, Prabhakaran S. Diagnosis and management of transient ischemic attack and acute ischemic stroke: a review. JAMA. 2021;325(11):1088-1098.
63. Kleindorfer DO, Towfighi A, Chaturvedi S, et al. 2021 guideline for the prevention of stroke in patients with stroke and transient ischemic attack: a guideline from the American Heart Association/American Stroke Association. Stroke. 2021;52(7):e364-e467.
64. Levine GN, McEvoy JW, Fang JC, et al. Management of patients at risk for and with left ventricular thrombus: a scientific statement from the American Heart Association. Circulation. 2022;146(15):e205-e223.
65. Wang Y, Wang Y, Zhao X, et al. Clopidogrel with aspirin in acute minor stroke or transient ischemic attack. N Engl J Med. 2013;369(1):11-19.
66. Johnston SC, Easton JD, Farrant M, et al. Clopidogrel and aspirin in acute ischemic stroke and high-risk TIA. N Engl J Med. 2018;379(3):215-225.
67. Centers for Disease Control and Prevention. About Valvular Heart Disease. Available at https://www.cdc.gov/heartdisease/valvular_disease.htm. Last accessed June 10, 2024.
68. Otto CM, Nishimura RA, Bonow RO, et al. 2020 ACC/AHA guideline for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2021;143(5):e72-e227.
69. Ortel TL, Neumann I, Ageno W, et al. American Society of Hematology 2020 guidelines for management of venous thromboembolism: treatment of deep vein thrombosis and pulmonary embolism. Blood Adv. 2020;4(19):4693-4738.
70. Khan F, Tritschler T, Kahn SR, Rodger MA. Venous thromboembolism. Lancet. 2021;398(10294):P64-P77.
71. Stevens SM, Woller SC, Kreuziger LB, et al. Antithrombotic therapy for VTE disease: second update of the CHEST guideline and expert panel report. Chest. 2021;160(6):e545-e608.
72. Criqui MH, Matsushita K, Aboyans V, et al. Lower extremity peripheral artery disease: contemporary epidemiology, management gaps, and future directions: a scientific statement from the American Heart Association. Circulation. 2021;144(9):e171-e191.
73. Gerhard-Herman MD, Gornik HL, Barrett C, et al. 2016 AHA/ACC guideline on the management of patients with lower extremity peripheral artery disease: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2017;135(12):e726-e779.
1. O'Gara PT, Kushner FG, Ascheim DD, et al. 2013 ACCF/AHA guideline for the management of ST-elevation myocardial infarction: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2013;61(4):e78-e140. Available at https://www.ahajournals.org/doi/10.1161/CIR.0b013e3182742c84. Last accessed February 17, 2026.
2. Ortel TL, Neumann I, Ageno W, et al. American Society of Hematology 2020 guidelines for management of venous thromboembolism: treatment of deep vein thrombosis and pulmonary embolism. Blood Adv. 2020;4(19):4693-4738. Available at https://ashpublications.org//bloodadvances/article/4/19/4693/463998. Last accessed February 17, 2026.
Mention of commercial products does not indicate endorsement.