Vaccinia is a live virus vaccine that was last routinely used in the United States in 1972. Since then, advances have been made in the safety of commonly used vaccines. Vaccinia's usual side effects are not expected in today's vaccines. In addition, the health profile of the population has changed, because there are more citizens with immune system problems. Therefore, this course will highlight the expected side effects, common reactions, and contraindications to the vaccine. Care of the vaccine site to protect the vaccinee and contacts will be stressed.
- INTRODUCTION
- OVERVIEW
- SMALLPOX
- MONKEYPOX
- ORTHOPOXVIRUS PREVENTION
- AVAILABLE VACCINES
- ACTION OF VACCINIA
- RECIPIENTS OF THE VACCINE
- HANDLING THE VACCINE
- ADMINISTRATION OF THE VACCINE
- CARE OF THE VACCINATION SITE
- EFFECTIVE HANDWASHING
- NORMAL REACTION TO VACCINIA
- EXPECTED REACTIONS
- COMPLICATIONS
- ADVERSE EVENTS
- CAREFUL SCREENING OF VACCINE CANDIDATES
- QUESTIONS REGARDING VACCINATION
- SUMMARY
- ACKNOWLEDGEMENTS
- WEBSITES
- GLOSSARY OF TERMS
- REPORTING
- Works Cited
This course is designed for members of the healthcare interdisciplinary team who must make informed decisions about participation in the voluntary pre-event vaccination program and to answer questions from patients, family, and friends about the vaccine.
The purpose of this course is to help members of the interdisciplinary team (IDT) make informed decisions about participation in the voluntary pre-event vaccination program and to answer questions of patients, family, and friends about the vaccine. In addition, the IDT will have necessary information about smallpox should a terrorist act result in the transmission of the smallpox virus.
Upon completion of this course, you should be able to:
- Relate the historical significance of the smallpox vaccine as well as the disease of smallpox, its symptoms, and its outcomes.
- Describe the technique of ring vaccination.
- Discuss the vaccine currently available, and describe the immunity developed by vaccinia.
- Identify four contraindications to receiving vaccinia in a pre-event scenario.
- List the steps and technique for administration of vaccinia, including the types of dressings recommended for the vaccine site and appropriate use of each.
- Enumerate the steps for effective handwashing.
- Describe the response and expected reactions to vaccinia.
- Identify complications of vaccinia and the appropriate treatments.
- List adverse events associated with vaccinia and available treatments.
- Discuss cultural factors that affect the informed consent process.
Elizabeth T. Murane, PHN, BSN, MA, received her Bachelor’s degree in nursing from the Frances Payne Bolton School of Nursing, Case Western Reserve University in Cleveland, Ohio and a Master of Arts in Nursing Education from Teachers College, Columbia University, New York, New York.
Her nursing experience includes hospital nursing on pediatric, medical, and surgical units. She lived for 15 years in a village in Eastern Papua New Guinea providing medical and linguistic/literacy services for the villagers. She was a public health nurse for a year with the Brooklyn, New York Health Department and 20 years with the Shasta County Public Health Department in Redding, California. As a public health nursing director, she developed response plans for environmental and health issue disasters for both Shasta County and adjacent Tehama County Public Health Departments.
Contributing faculty, Elizabeth T. Murane, PHN, BSN, MA, has disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
John M. Leonard, MD
Jane C. Norman, RN, MSN, CNE, PhD
Alice Yick Flanagan, PhD, MSW
Randall L. Allen, PharmD
James Trent, PhD
The division planners have disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
Sarah Campbell
The Director of Development and Academic Affairs has disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
The purpose of NetCE is to provide challenging curricula to assist healthcare professionals to raise their levels of expertise while fulfilling their continuing education requirements, thereby improving the quality of healthcare.
Our contributing faculty members have taken care to ensure that the information and recommendations are accurate and compatible with the standards generally accepted at the time of publication. The publisher disclaims any liability, loss or damage incurred as a consequence, directly or indirectly, of the use and application of any of the contents. Participants are cautioned about the potential risk of using limited knowledge when integrating new techniques into practice.
It is the policy of NetCE not to accept commercial support. Furthermore, commercial interests are prohibited from distributing or providing access to this activity to learners.
Supported browsers for Windows include Microsoft Internet Explorer 9.0 and up, Mozilla Firefox 3.0 and up, Opera 9.0 and up, and Google Chrome. Supported browsers for Macintosh include Safari, Mozilla Firefox 3.0 and up, Opera 9.0 and up, and Google Chrome. Other operating systems and browsers that include complete implementations of ECMAScript edition 3 and CSS 2.0 may work, but are not supported. Supported browsers must utilize the TLS encryption protocol v1.1 or v1.2 in order to connect to pages that require a secured HTTPS connection. TLS v1.0 is not supported.
The role of implicit biases on healthcare outcomes has become a concern, as there is some evidence that implicit biases contribute to health disparities, professionals' attitudes toward and interactions with patients, quality of care, diagnoses, and treatment decisions. This may produce differences in help-seeking, diagnoses, and ultimately treatments and interventions. Implicit biases may also unwittingly produce professional behaviors, attitudes, and interactions that reduce patients' trust and comfort with their provider, leading to earlier termination of visits and/or reduced adherence and follow-up. Disadvantaged groups are marginalized in the healthcare system and vulnerable on multiple levels; health professionals' implicit biases can further exacerbate these existing disadvantages.
Interventions or strategies designed to reduce implicit bias may be categorized as change-based or control-based. Change-based interventions focus on reducing or changing cognitive associations underlying implicit biases. These interventions might include challenging stereotypes. Conversely, control-based interventions involve reducing the effects of the implicit bias on the individual's behaviors. These strategies include increasing awareness of biased thoughts and responses. The two types of interventions are not mutually exclusive and may be used synergistically.
#98533: Smallpox Vaccination: An Update
Vaccinia was used for many years to protect people from smallpox. It was the organism in the vaccine that was used to globally eradicate "wild" smallpox. The last known natural case of smallpox in the United States occurred in 1949. Because no new cases of smallpox subsequently occurred and because the risk of adverse events, including death, from the vaccine were greater than the benefit of protection, routine vaccination of the general U.S. population was rescinded in 1971. Although routine smallpox vaccination of healthcare workers was discontinued in 1976, the 2015 Advisory Committee on Immunization Practices (ACIP) recommends routine vaccination against the disease for specific populations at high risk of occupational exposure to orthopoxviruses [1,2].
The last case of wild, or naturally occurring, smallpox occurred in Somalia in 1977. In 1980, the World Health Organization (WHO) certified that the world was free of naturally occurring smallpox and recommended worldwide cessation of vaccination against it. Various countries ceased vaccinating their military personnel in the 1980s and early 1990s. During the global program of smallpox eradication, which included vaccination, the WHO worked to limit the number of laboratories that retained variola (the virus that causes smallpox). By 1984, only the Research Institute of Viral Preparations, in Moscow, and the Centers for Disease Control and Prevention (CDC), in Atlanta, had retained variola virus isolates. In 1994, Russia unilaterally moved its virus stocks from Moscow to the State Research Center of Virology and Biotechnology VECTOR, a former biologic warfare research center in Koltsovo [3,4].
The smallpox vaccine does not contain smallpox (variola) virus. It contains vaccinia and protects against orthopox diseases, such as smallpox (variola), cowpox, monkeypox, mousepox, and rabbitpox. Dryvax and Wetvax, which were formerly the only smallpox vaccines available in the United States, have been superseded by ACAM2000, a "second-generation" vaccine derived from a clone of Dryvax, purified, and produced using modern cell culture technology, and JYNNEOS, a "third-generation," highly attenuated vaccine. The manufacturer of ACAM2000 has stated that it will provide the vaccine only for Strategic National Stockpile (SNS) use [4,5]. The SNS is a national repository of life-saving pharmaceuticals and medical material maintained by the CDC; it includes a post-event smallpox vaccine inventory. More modest supplies of the vaccine, reserved for pre-event vaccination efforts, are maintained by state and local health agencies, as recommended by the CDC. The SNS program works with government and nongovernment partners to upgrade the nation's public health capacity to respond to a national emergency. Critical to the success of this initiative is ensuring that capacity is developed at the federal, state, and local levels to receive, stage, and dispense SNS assets [6].
It is important to note that the vaccines discussed in the course are effective for pre-exposure prophylaxis against orthopoxvirus infection; their use is not limited to smallpox. JYNNEOS is FDA-approved for non-military immunization against smallpox and monkeypox.
The following underlying concepts should be kept in mind as this course progresses.
This course provides a description of the vaccination program before a case of smallpox has occurred. Should there be a case of smallpox diagnosed either in the United States or elsewhere in the world, different guidelines would apply. Since 2001, the CDC has recommended that each state and territory in the United States create and maintain at least one smallpox response team. The persons identified as part of the response team will also be part of the pre-event vaccination program. To enhance public health preparedness, the CDC has recommended that specific teams at the federal, state, and local levels be established to facilitate diagnostic evaluation of initial suspected cases of smallpox and to initiate control measures. Considerations for additional teams should include population and geographic concerns and be developed to comply with federal, state, and local terrorism-response plans [7].
The CDC also has recommended that each acute-care hospital identify vaccinated personnel who may act to provide medical care for potential smallpox patients. This team should provide care 24 hours per day for the first two or more days after patients with smallpox have been identified and until additional healthcare personnel have been vaccinated. When possible, those healthcare personnel who have already been vaccinated should be considered for inclusion as a means to decrease the incidence of possible adverse effects of the vaccination. Additionally, all persons administering or handling vaccinia in a pre-event vaccination program should be vaccinated [8]. Nonvaccinated workers should be restricted from entering the rooms of smallpox patients or, under emergency conditions, should wear personal protective equipment. The size and composition of the response teams will vary according to institutions and their patient populations. Ideally, each hospital should have enough vaccinated personnel from a spectrum of healthcare occupational categories to ensure continuity of care. As stated, ACIP recommends routine vaccination against the disease for specific populations at high risk of occupational exposure to the smallpox virus [1].
Mainly, the effects described are for primary vaccinees, that is, those who have never had a prior smallpox vaccination or do not know if they have had one. People born before 1971 and those who have had one or more vaccinations are considered secondary vaccinees, and the side effects are expected to be mitigated, providing contraindications are closely followed. It is important to note, however, that with ACAM2000, there is no longer the need to differentiate between a primary and a secondary vaccinee, although this distinction may be useful should an adverse event occur [4].
All medical personnel should think through their volunteer participation in a pre-event vaccination program. Because the risk of smallpox occurrence is an unknown factor, as is the possibility of its being used as a weapon of bioterror, the risk/benefit equation that one would normally weigh when considering whether to receive an immunization does not apply. The primary benefit from choosing to receive the vaccine would be the protection of the nation. Personal benefit would likely be realized only in the event of exposure to smallpox; therefore, each healthcare professional should weigh the likelihood of personal exposure. Personnel working in critical care, emergency response, or public health are more likely to be exposed than those on a surgical ward. Obviously, members of the public health response teams and care provider teams should be vaccinated if they have no contraindications. Vaccinators are assumed to have a certain level of risk; protection cannot be considered absolute [7].
Before 2006, the data on adverse events resulting from smallpox vaccination had been based on studies done in 1968. Between December 2002 and December 2017, the Department of Defense (DoD) vaccinated more than 2.6 million select military personnel in a national program of preparedness against the use of smallpox as a weapon of bioterror. This provided a useful sample size of more current data on the side effects of the vaccine [9,10].
Comprehensive training programs in vaccination technique, infection-control safeguards, screening and education methods, adverse event monitoring, and product storage and handling made immediate vaccinations possible. As a testimony to the effectiveness of good education, the DoD reported no cases of transmission of vaccinia from worker to patient among 27,700 smallpox-vaccinated healthcare workers. Most adverse events occurred at rates below historical rates, leading researchers to conclude that mass smallpox vaccinations could be conducted safely [9,10].
Much conflicting information has been circulated in both the popular media and medical literature. For example, many have written about the smallpox "shot." It is a vaccination, not a shot. Some older medical personnel may remember using acetone to clean the arm before applying the vaccine, while literature today mentions using alcohol before the vaccination is given. The current CDC recommendation is that the arm is cleaned with soap and water only if there is gross contamination [7,8]. Because so much has been written about the vaccine against smallpox, inconsistencies may occur, even from trustworthy sources.
It is important to differentiate between methods of respiratory transmission, such as "airborne" and "droplet." Diseases such as rubeola (hard measles, red measles, or 14-day measles), chickenpox, and influenza are airborne and infect the respiratory system via nasal and pharyngeal passages. Micro-organisms spread by airborne transmission are smaller than 5 microns in size and thus are in the air for longer periods of time. For example, rubeola remains in the air for 90 minutes after the contagious person has left the room and may infect those without immunity who enter the room during that time. Smallpox, meningitis, and many respiratory diseases are transmitted through droplets and are less contagious. These micro-organisms are larger than 5 microns in size and do not stay suspended in the air as long as airborne micro-organisms. Prolonged close contact is required to transmit the organism [11].
Through the centuries, smallpox has been a dreaded disease that has influenced the course of history and resulted in the death or disfigurement of individuals on virtually every continent. Today's population has less acquired immunity and less vaccine-induced immunity to the disease than in previous decades. In addition, the increase in immunocompromised individuals and mobility of people means that this feared disease has a potential for great morbidity and mortality [12,13,14].
Many experts believe that smallpox first appeared in northeastern Africa more than 12,000 years ago. Some evidence of lesions resembling smallpox has been found on the faces of mummies, including that of Ramses V. Egyptian merchants probably spread the disease to India. The first recorded smallpox epidemic occurred during the Egyptian-Hittite war and led to the decline of the Hittite empire. The first stages of the decline of the Roman Empire coincided with a large-scale epidemic. Several reigning monarchs died from smallpox. During the 18th century, the Hapsburg line of succession to European thrones changed four times in as many generations because heirs had succumbed to smallpox. During the same century in Europe, 400,000 people died each year of smallpox, and one-third of survivors were left blind [15].
Many countries have been affected historically by smallpox epidemics or pandemics. In addition, the use of smallpox as a biologic weapon has a long history. Contaminated blankets and a handkerchief were given to the American Indians by a British commander during the French and Indian War to help the British win a battle [16]. In 1520, the Aztecs captured one of Cortés' men, who was infected with smallpox, resulting in the decimation and defeat of the Aztecs [17]. The European pandemic of 1870–1874, which claimed the lives of more than 500,000 soldiers and civilians, was triggered by the Franco Prussian war, after smallpox-infected French soldiers were captured and imprisoned in Germany [18]. Benjamin Franklin, whose son was a victim of smallpox, lobbied for variolation (the preventive measure before vaccination) when the disease ravaged the American colonies in 1753 [15].
The potential for the use of smallpox as a weapon of war is still present. It is speculated that the smallpox virus may escape (or may have already escaped) Russian government control, and rogue groups may use it as a bioterrorism agent [19]. Another potential threat is the accidental unearthing of a live sample during construction, cleanup, or excavation. In 2014, several vials of live variola samples were found in an unused cold storage at a U.S. Food and Drug Administration (FDA) laboratory in Bethesda, Maryland [20].
The triumph over smallpox in the latter half of the 20th century was a major step for the health and well-being of all people. Smallpox, the world's most devastating plague, was declared eradicated in 1980 by the WHO [3]. This eradication required the cooperation of several countries, including their medical personnel and resources. The wide use of the vaccine—first developed by Edward Jenner in 1796 and improved over the years by scientists from various countries—resulted in virtual eradication of the disease. The last naturally occurring case in the United States was in 1949; the last case outside the United States was in Somalia in 1977 [3,14,21].
The WHO is collecting virus samples from all regions in which it is active, except the western Pacific. As of 2019, 120 strains have been collected, including 49 from Africa and 34 from Southeast Asia [60].
Variola virus is the smallpox causative organism. It is a double-stranded DNA virus and a member of the family Poxviridae, subfamily Chordopoxvirinae, and genus Orthopoxvirus. It replicates in the cytoplasm of the host cell, rather than in the nucleus, as do other viruses [22]. Electron micrographs show that variola organisms are shaped like bricks. This brick shape distinguishes variola from the varicella zoster virus (the virus that causes chickenpox) [3].
As previously mentioned, smallpox is transmitted mainly from person to person by the respiratory route through droplets. Close personal contact (normally within six feet of a patient) over a prolonged period, such as in a household, is required for transmission to occur. Smallpox is not as infectious as measles, chickenpox, or influenza. As a result, transmission in the workplace and school is less than in the household [23]. Data collected in the 1960s showed that an estimated 80% of the cases were contracted from an individual in the household [23]. In this course, "household contact" will be used in a broad sense and includes intimate and sexual contact. There is some indication in the medical literature that inanimate objects also may transmit the disease, but this is less common [9,24,25]. Unfortunately, the use of variola as a biologic weapon is a threat because of the aerosol infectivity of the virus and the relative ease with which it can be mass produced [26].
As noted, smallpox is transmitted from one person to another by droplets and close contact is required. Patients are most infectious during the first 7 to 10 days after rash onset, although infectivity may occur during the prodromal period, immediately before rash onset. Occasionally, transmission has been by direct contact or contact with fomites (e.g., clothes or bedding); airborne transmission has occurred rarely [7]. Droplets containing the variola virus may be transmitted through face-to-face contact while talking, singing, coughing, or sneezing. The disease also may be transmitted by saliva (through sharing food or drink) and by kissing on the mouth. In past centuries, most people did not share food or drink nor were children kissed on the mouth by adults. Today, people share food and drink and even kiss newborns on the mouth. These changes in lifestyle have contributed to a more vulnerable population than in the days before eradication.
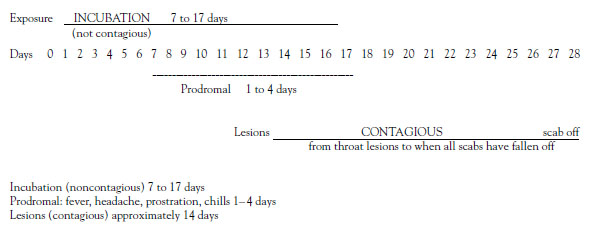
The virus is not shed during the incubation period, which can be from 7 to 17 days, but most commonly is 10 to 14 days. During the incubation period, the virus enters the respiratory tract, seeds the mucous membranes, passes quickly to the lymph nodes, and then multiplies in the reticuloendothelial system [3,27]. Probably only a few virus particles (virions) are sufficient to cause the infection (Figure 1) [28].
The prodromal phase, which follows the incubation period and lasts from two to four days, begins abruptly and is characterized by fever (usually 101–105°F) and at least one of the following [27,29,30]:
Prostration
Severe (splitting) headache (90%)
Backache (90%)
Chills (60%)
Vomiting (50%)
Delirium (15%)
Abdominal colic (13%)
Diarrhea (10%)
Convulsions (7%)
At the end of prodromal phase (about 24 hours before the skin rash erupts), minute red spots (the enanthem) appear on the tongue and soft palate. The patient may complain of a sore throat as lesions also may occur lower in the respiratory tract. When the lesions in the mouth and pharynx open and release the virus, the patient is contagious. Patients are most contagious for the first week but may still transmit the disease until all the epidermal scabs from the skin lesions have fallen off, usually in 21 to 28 days [27].
The smallpox rash erupts at the end of the prodrome. A few lesions usually appear first on the face, especially on the forehead. These are called the "herald spots." Occasionally, the rash is first seen on the forearms. Lesions tend to appear on the proximal portions of the extremities, on the trunk, and then on the distal portions of the extremities. However, the rash usually progresses so quickly that within 24 hours it is apparent on all parts of the body, and the patient may not notice how the rash progressed. Normally, more lesions appear over the next one or two days, possibly followed by a few fresh lesions later. Generally, the rash is distributed in a centrifugal pattern. The rash is most dense on the face and is denser on the extremities than on the trunk. It is more prominent on the distal regions than on the proximal, and on the extensor rather than on the flexor surfaces. There also may be lesions on the palms and soles [31].
The classic smallpox lesions are round, well-circumscribed vesicles progressing to pustules that are deep-seated and firm. They feel "shotty" (round and hard) when rolled between the thumb and forefinger. As they continue to develop, the lesions become umbilicated, having a central "naval-like" depression. The more confluent the lesions, the more grave the prognosis. One of the distinguishing features of smallpox rash is that the lesions on any specific area of the body are all in the same state of development, meaning that they are all vesicles, pustules, or umbilicated lesions. In contrast, the rash of chickenpox starts as a vesicle on top of erythema—the "dewdrop on rose petal." Chickenpox come in "crops," so in any one area of the body there will be vesicles, pustules, and crusts (scabs). The palms and soles are rarely involved, and patients are rarely toxic or moribund [30].
As previously stated, the smallpox rash usually appears on all parts of the body within 24 hours, and no new lesions generally appear after the second day. By the second day, all the lesions are vesicles with an opalescent fluid that becomes opaque and turbid in another 24 to 48 hours. By day 7, all are pustules. The maximum lesion size is reached by day 10, and resolution typically starts on day 11. The lesions flatten and umbilicate with scabs and crusts developing around day 14. In another 7 to 14 days, the scabs separate, leaving discolored areas and, in severe cases, pockmarks. Permanent facial pockmarks occur in 65% to 80% of survivors [32]. Scabs on the soles of the feet may take longer to separate and are sometimes artificially removed. After all scabs have separated, the patient is no longer contagious and has a life-long high degree of immunity to smallpox [27,31]. Fever usually diminishes on the second or third day, but rises again on day 7 or 8 and remains high until the scabs have formed over all the lesions.
There are many possible secondary complications in smallpox. Most are due to viral activity in an unusual site or to secondary bacterial infections. Smallpox may affect several systems. The skin lesions can become infected with bacteria, but broad-spectrum antibiotics and good hygiene will prevent many of these secondary infections. Mild conjunctivitis at the time of the skin eruptions is part of the disease; however, corneal ulceration and keratitis may occur, causing blindness. Approximately 5% to 9% of patients with smallpox develop ocular complications [33]. The joints may be involved, causing arthritis in approximately 2% of survivors [22]. The elbow is the most commonly affected joint. Respiratory complications may develop around day 8. Pulmonary edema is fairly common in hemorrhagic and flat-type smallpox. However, cough is a rare symptom in the disease. Encephalitis occurs in 1 in 500 cases, usually appearing between days 6 and 10. If the patient recovers, the recovery is slow but usually complete [35].
The sequelae in persons who recovered from smallpox, in order of frequency, were facial pockmarks, blindness (due to corneal scarring), and limb deformities (due to osteomyelitis and arthritis) [31].
There is no curative treatment for smallpox. Therefore, the development of a vaccine that can prevent or lessen the morbidity and reduce the mortality is significant. Because there have been no natural cases of smallpox for many decades, the antivirals currently available have officially never been tried on smallpox. A new agent, tecovirimat (TPOXX), was approved in 2018 for the treatment of smallpox based on animal trials [36]. Two million treatment courses of tecovirimat have been added to the SNS, with an additional 23,000 doses expected to be added to this number. The manufacturer also is developing an intravenous (IV) formulation of the drug [37]. Obviously, secondary bacterial infections would be treated with appropriate antibiotics.
Pregnant women and immunocompromised individuals may have more severe smallpox disease and/or atypical skin lesions (e.g., "flat" or "hemorrhagic"), leading to a very high mortality rate.
The overall fatality rate is 30% in unvaccinated populations and 3% in vaccinated populations [26]. However, different types of smallpox have different mortality rates. For example, flat and hemorrhagic smallpox (uncommon types) usually are fatal [4,27].
Monkeypox is caused by infection with monkeypox virus, a member of the Orthopoxvirus genus in the family Poxviridae. The Orthopoxvirus genus also includes variola virus, vaccinia virus, and cowpox virus [97].
Monkeypox was first discovered in 1958 when two outbreaks of a pox-like disease occurred in colonies of monkeys kept for research. The first human case of monkeypox was recorded in 1970 in the Democratic Republic of the Congo, and since then, it has been reported in people in several other central and western African countries. The majority of infections continue to be reported in the Democratic Republic of the Congo, but cases have been reported in other countries and continents, including the United States. In 2022, there was an outbreak of monkeypox in non-endemic countries, with nearly 2,000 cases reported in the United States as of July 18 [98].
Monkeypox is considered a zoonotic disease and can spread to people when they come into physical contact with an animal host (typically rodents or primates). However, person-to-person transmission is possible through close contact with lesions, body fluids, respiratory droplets and contaminated materials. It is unclear if the virus is present in semen or vaginal fluids or if asymptomatic individuals can transmit the disease [99]. Droplet and Contact Precautions are advised when caring for persons with monkeypox.
The first symptoms of monkeypox include fever, malaise, headache, sore throat, and cough. It generally has a similar clinical presentation and course as smallpox. However, a distinguishing feature of monkeypox is lymphadenopathy, which typically occurs with fever onset, one to two days before rash onset, or rarely with rash onset. Enlarged lymph nodes may be noted in the bilateral or unilater and can occur in submandibular, cervical, axillary, or inguinal areas [100].
After infection with monkeypox, there is an incubation period of approximately 7 to 14 days (range: 5 to 21 days), followed by the prodromal period, characterized by onset of initial symptoms. Following the prodrome, which typically lasts days, is the rash stage; the first lesions to develop are on the tongue and in the mouth (enanthem). Following the enanthem, a macular rash appears on the skin, starting on the face and spreading to the arms and legs and then to the hands and feet, including the palms and soles [100]. The rash typically spreads to all parts of the body within 24 hours, becoming most concentrated on the face, arms, and legs (as with smallpox). By the third day of rash, lesions have progressed from macular (flat) to papular (raised). By the fourth to fifth day, lesions have become vesicular, and by the sixth to seventh day, lesions have become pustular and show signs of umbilication. The pustules will remain for approximately five to seven days before beginning to crust. By the end of the second week, pustules have crusted and scabbed over. Scabs will remain for about one week before beginning to fall off. After all scabs have fallen off, the patient is no longer contagious [100].
The prognosis of monkeypox infection varies depending on the variant contracted. There are two clades of monkeypox virus: the West African clade and the Congo Basin (Central African) clade. Human infections with the West African clade appear to cause less severe disease compared with the Congo Basin clade; the case fatality rate is 3.6% for the West African clade and 10.6% for the Congo Basin clade [99]. The 2022 outbreak cases have all been confirmed as having been infected with the West African clade.
In 910, Rhazes, an Arab physician, wrote the first medical account of smallpox in which he attempted to explain why survivors did not contract the illness again. His was the earliest theory of immunity, and it started the search for a way to prevent the scourge of smallpox. Doctors and others then began to intentionally infect healthy persons with smallpox organisms, hoping that the resulting infection would be mild but sufficient to develop immunity [15,38].
In 1017, a hermit in China introduced the prime minister to "variolation," a type of inoculation using germs from smallpox survivors with mild cases. Various forms of smallpox material (e.g., vesicles, pus from pustules, or ground-up scabs) were administered to healthy adults through the nose or skin. These different methods were known as "variolation," which was spread by merchants traveling by caravan to distant lands. For example, Circassian traders introduced the practice in the Ottoman Empire. In 1717, Lady Mary Wortley Montagu accompanied her ambassador husband to Istanbul. In a letter to a friend in England, Lady Montagu described "ingrafting," which was her term for variolation. It was a procedure done by elderly women who made four or five scratches or slight punctures on the arm. Material from the pustules of patients with mild cases was introduced into the scratches or punctures. Lady Montagu had her son inoculated while in Istanbul and her daughter inoculated in the presence of the English court doctors. Variolation also was tried on prisoners, who were then exposed to smallpox; they did not develop the disease. After the two daughters of the Princess of Wales were treated in 1722, the procedure gained general acceptance [14,15].
Two to three percent of variolated persons died of smallpox, became the source of a new epidemic, or developed other illnesses, such as tuberculosis or syphilis. However, smallpox fatality rates were 10 times lower in those who had experienced variolation, which was widely practiced until Jenner's discovery [15].
In the rural areas of Europe, milkmaids who had previously had cowpox were noted to be immune to smallpox. Benjamin Jesty, an English farmer, vaccinated his wife and two sons with material taken from the udders of cows with cowpox. None of the Jestys developed smallpox during later epidemics nor did they have any reaction to variolation. However, Jesty's method was ignored because his wife nearly lost the arm in which she was vaccinated due to severe inflammation [15].
Edward Jenner was born in 1749 and, at 8 years of age, had the variolation procedure performed on him. Jenner developed an interest in medicine and later became a surgeon. He wrote several papers on other scientific studies; however, the connection between cowpox and smallpox continued to intrigue him throughout his medical career [15].
In 1796, a milkmaid named Sarah Nelmes developed cowpox. Jenner extracted fluid from a pustule on Sarah's hand and inserted it in two half-inch incisions on the arm of a healthy 8-year-old boy named James Phipps. Later, Jenner tried twice to inoculate James with smallpox samples; however, the boy did not develop the disease [14]. Jenner published a manuscript in which he described 10 cases of vaccination and 13 persons who had cowpox, in whom immunity was confirmed [14,15].
By 1800, most European countries were vaccinating for smallpox, mostly using pustule fluid. In 1801, U.S. President Thomas Jefferson had 18 family members, some neighbors, and the last of the Native American tribe of Mohicans vaccinated [15]. In 1805, Napoleon insisted that all his troops who had not had smallpox be vaccinated with the "Jennerian vaccine" [15,39].
This deliberate use of vaccination was the first scientific attempt to control an infectious illness. Technically, Jenner did not discover vaccination, but he was the first person to confer scientific status on the procedure. He named the procedure vaccinae after vaca, which is Latin for "cow." Jenner also stated that the universal use of the vaccine would lead to the eradication of smallpox. In 1977, less than 200 years after the vaccination of young James Phipps, a Somalian cook, Ali Maow Maalin, developed the characteristic smallpox rash. Maalin was the last person to develop natural smallpox as a result of contact with another person [15]. In honor of Jenner, all materials used to induce immunity are called "vaccines" [39].
Since the introduction of vaccination in 1796, controversy has existed as to the origin of Jenner's variolae vaccinae. In some cases, the vaccination was successful and it protected from smallpox; other times, there was either no "take" or an apparently successful vaccination did not protect the recipient from smallpox. Jenner suggested that other vaccinators had chosen material from cows with "spurious cowpox" rather than true cowpox. Jenner believed that his vaccine originated from an infection of the heels of horses, which he referred to as "grease," and that after passing through the cow, it was suitable for human use.
The debate has continued into the 21st century. The current vaccinia strains differ from cowpox and other poxviruses and also differ in various parts of the world. There is no natural host for vaccinia. Before vaccination programs were discontinued, the generally accepted view was that vaccinia was an artificially propagated virus that was modified in some way from the naturally occurring poxviruses. No one is sure of the origin of vaccinia [40,41].
It is important to remember that vaccinia has been tested in controlled experiments and proven to have no derivation from variola, the causative virus of smallpox. No one can develop smallpox from the vaccination as the organisms are different, even though both are orthopoxviruses [39].
In summary, vaccinia is the virus used to make the smallpox vaccine. Variola is the virus that causes smallpox; it is not used to make the vaccine. Vaccinia is spread only by direct contact and is not airborne. Variola also is spread by direct contact and by droplet aerosols in the air. Both viruses have more severe reactions or disease in immunocompromised individuals.
The underlying purpose in eradication of a disease is to give the causative organism no place to survive. This is very difficult when the organism has both a human host and an animal host. Thus, there are no plans to try to eradicate influenza, for example, in which the life cycle of the virus includes pigs, fowl, and humans. However, smallpox could be eradicated because only humans are susceptible to variola.
Total population vaccination (mass vaccination) is the theory behind the worldwide effort to eradicate polio. Realistically, one could never have every human protected against polio. The goal instead is to obtain "herd immunity," which means that enough people are immune so that the remaining few also are protected. In this theory, the organism fails to locate the few susceptible individuals and is thus unable to survive. The percentage of the world's population that would require vaccination to prevent smallpox through herd immunity is believed to be around 90% [42]. It is known that vaccinated populations have not approached this percentage in areas of Africa and Asia. This has led to the use of ring vaccination, commonly known as surveillance and containment [42].
The purpose of ring vaccination is to form a buffer of immune individuals around the organism to prevent the spread of the disease. It is incorrect to illustrate this concept by drawing a "ring" on a map, within which all individuals would be vaccinated. Ring vaccination is not a geographic designation. It is a social designation in which a "ring" is drawn around each smallpox victim, and it includes close household contacts who may have been exposed to the victim. (The victim must be isolated to limit the number of contacts.) There is a three- to four-day window in which a person exposed to smallpox may be vaccinated to prevent the disease, or at least make the symptoms milder. To help eradicate the disease, a second "ring" also is vaccinated. The second ring consists of those who have had contact with the individual(s) exposed to the smallpox victim and the members of their households [42]. These individuals also would need to be vaccinated because it is not possible to know exactly when a contact was exposed and whether the three- or four-day period had already lapsed. Contacts would be vaccinated up to seven days after exposure. They would still likely develop smallpox, but it would be less severe [17]. For anyone exposed to a smallpox patient, the pre-event contraindications will be modified, as the disease has a fatality rate of 30% [9]. This would mean 300,000 fatalities in one million cases of smallpox, compared to one to two fatalities in one million vaccinations.
For example, Mr. J has smallpox. The case investigation reveals that Mr. J lives with his wife, Mrs. J, and two children, JJ (4 years of age) and SJ (7 years of age). Mrs. J's niece, Miss L (7 years of age), has been staying with the family while her parents are on a trip and is in the household when Mr. J has the prodrome; she returns home the day he develops the smallpox rash. Mr. J is a farmer with no hired help. Mrs. J works for a guest home where she cares for six elderly residents. JJ attends preschool, and SJ is in the second grade. Miss L also is in the second grade in another city.
According to the ring vaccination protocol, who should be vaccinated? Obviously, Mrs. J, JJ, SJ, and Miss L should be vaccinated. Additional contacts in the "second ring" requiring vaccination would be the following: the six elderly residents exposed to Mrs. J; any family members who visited these elderly residents and the households of these family members; the cook at the guest home and the cook's household; the children and workers in JJ's preschool and their households; the children and teachers and their household contacts in both SJ's and Miss L's second grade classes; and Miss L's household contacts.
Please remember that this scenario describes the situation after a case of smallpox has been diagnosed. Vaccination is currently voluntary and done before a diagnosis of smallpox. Individuals with contraindications should not be vaccinated, nor is vaccination recommended for the general population. However, should an outbreak of smallpox occur, there is enough vaccine to vaccinate every person in the United States [43]. There are some individuals who, for religious, cultural or other reasons, will refuse to receive vaccinations; their wishes should be considered [44].
In 2007, prior to the removal of Dryvax from the SNS and other programs, the FDA licensed ACAM2000 (Sanofi Pasteur Biologics, formerly Acambis), a second-generation smallpox vaccine, for use in the United States. ACAM2000 is a live vaccinia virus derived from plaque purification cloning from Dryvax. Like Dryvax, ACAM2000 is contraindicated for immunocompromised individuals and has been associated with serious side effects, such as myocarditis, encephalitis, and ocular complications. Based on clinical studies, myocarditis and/or pericarditis have occurred in 1 in 175 adults who receive the vaccine for the first time [4]. Serious health problems, including those that are life-threatening, also may occur in unvaccinated individuals who are accidentally infected by someone who has recently been vaccinated [4]. The percentage of unvaccinated persons who develop a successful immunization reaction from ACAM2000 has been shown to be similar to that of Dryvax. ACAM2000 also has been found to be acceptable as a booster in those previously vaccinated for smallpox. It is important to note, however, that a 2008 study examining the continued immunity conferred by smallpox vaccination administered in childhood found that 97% of participants showed no decrease in vaccinia antibody titers (after a range of 13 to 88 years) [46]. As a result, there may be no need for booster vaccinations, and the available supplies of vaccinia may be better used in those who have not previously been vaccinated.
The CDC began distributing ACAM2000 to civilian laboratory personnel, the military, and state public health preparedness programs in 2008 [4,5]. Every person who receives the ACAM2000 vaccine must receive an FDA-approved medication guide, which contains information that may help patients avoid serious adverse events. These guides must be issued when the FDA has determined that [4]:
Certain information is necessary to prevent serious adverse events.
Patient decision making should be informed by information about a known serious side effect with a product.
Patient adherence to directions for the use of a product is essential to its effectiveness.
ACAM2000 is the first vaccine licensed with a medication guide. The guide answers questions about the vaccine, who should get it and why, possible side effects and adverse events, how to care for the vaccination site, and what to expect and avoid if vaccinated [2].
In 2007, the U.S. Department of Health and Human Services awarded a contract to Bavarian Nordic A/S to manufacture and deliver 20 million doses of Modified Vaccinia Ankara-Bavarian Nordic (MVA-BN), enough for 10 million individuals, to the SNS [47; 48]. This highly attenuated, third-generation vaccine cannot replicate in humans, and MVA-BN requires a booster at four weeks to achieve immune protection equivalent to ACAM2000 because of its low immunogenicity. However, this feature makes the vaccine appropriate for use in patients who have contraindications for ACAM2000, such as being a compromised immune system [2; 45]. There are approximately 10 million such individuals in the United States, and in the event of a possible smallpox or other orthopoxvirus outbreak, 20 million doses (a two-dose immunization schedule) of MVA-BN would be available to them [47,48].
Although MVA-BN was associated with dose-related local reactions, the events were self-limited and did not lead to the discontinuation of subjects from phase I studies. No vaccinia-associated cardiac complications occurred with MVA-BN in a phase II clinical trial (completed in 2015) in a study population of healthy young adults [49]. One serious adverse event (sarcoidosis) did occur, though the causation is uncertain [49]. MVA-BN was fast-tracked for approval by the FDA in 2008 [50].
In 2019, the FDA approved the MVA-BN vaccine JYNNEOS (Smallpox and Monkeypox Vaccine, Live, Non-Replicating) for the prevention of smallpox and monkeypox disease in adults 18 years of age and older determined to be at high risk for smallpox or monkeypox infection [51]. The clinical efficacy of JYNNEOS (also known as Imvamune or Imvanex outside the United States) to generate an immune response has been evaluated in six published clinical studies [52,53,54,55,56,57]. The MVA-BN vaccine has been shown to be highly effective against monkeypox in healthcare workers in Africa, but the efficacy against smallpox has not been tested in humans [34,60]. JYNNEOS is stockpiled in the SNS, with a total 28 million doses available (as of 2018) [2; 58]. Bavarian Nordic A/S is working on providing a freeze-dried formulation of MVA-BN to the SNS after clinical trials are completed and approval is granted (perhaps as early as 2022) [59].
For years, the only vaccines available for smallpox were the Aventis Pasteur Smallpox Vaccine (APSV), also known as Wetvax, (produced in 1956–1957) and Dryvax (produced in 1978). However, the manufacturer of Dryvax announced its intent to withdraw the license and asked that all remaining quantities of the vaccine held by civilian and military programs be quarantined by February 29, 2008 (when the remaining lots were due to expire) for the purpose of destruction. All programs holding supplies of Dryvax were instructed to provide the CDC with documentation of the vaccine's destruction no later than March 31, 2008 [5]. APSV (Wetvax) is currently stockpiled at the SNS for emergency use and is tested annually for potency; however, it will only be used when supplies of ACAM2000 are depleted, not readily available, or in a case-by-case basis when ACAM2000 is contraindicated and JYNNEOS is not available [2,45]. The CDC holds investigational new drug (IND) applications for APSV use at full strength and 1:5 dilution.
The National Institute of Allergy and Infectious Diseases is supporting studies of other smallpox vaccines under IND applications. The National Institutes of Health is supporting the study of new-generation smallpox vaccines that are made using highly attenuated forms of the smallpox vaccine virus (third-generation vaccines) or using only selected DNA segments of the virus (fourth-generation vaccines). Third-generation attenuated vaccines being tested for use in the United States are ACAM3000 and LC16m8 [58]. LC16m8, which was produced by passage of vaccinia through rabbit kidney cells, was licensed for use in 1975 in Japan, which has an 80 million dose per year manufacturing capability [34; 60]. Fourth-generation vaccines are attenuated through direct genetic engineering rather than extensive passage through alternative hosts; examples of these vaccines are VacDelta6 (VECTOR), VACVvD4-ZG, and VACVΔE3L, with some currently in clinical trials.
The purpose of the smallpox vaccination is to introduce the vaccinia virus into the malpighian layer of the epidermis. During the progress of the vaccination, from vesicle until the scab falls off, live vaccinia virus is present, may be transmitted to others, and may cause adverse health events in those vaccinated. However, once the scab has separated from the vaccination site, the individual is no longer contagious [61].
Like many vaccines, vaccinia produces both humoral and cell-mediated immunity [14]. The humoral response results in the production of short-lived IgM antibody, persistent IgG antibody, and other types of antibodies. A convenient memory clue when reading laboratory results for infectious diseases is that IgM could mean "I get medicine," which indicates the short-lived antibody and is a marker for acute infection. Of course, there may be no medicine or treatment available, but this person is probably contagious if they are still exhibiting symptoms. IgG could be thought of as "I get germs" and indicates a persistent antibody that confers some degree of immunity. The person with these laboratory results has had the infection in the past and would not be a candidate for any available treatment or medicine, nor would that person be contagious. This is especially significant in reading laboratory results for a patient with a suspected disease, such as hepatitis A.
In most diseases, the IgG antibody is specific for a disease, and most viral vaccines are now specific for a certain virus. An example is the annual influenza vaccine developed by the CDC and the FDA. Each year, the vaccine is produced to overcome four specific viral strains that are expected, usually two type A strains and two type B strains [62]. Persons who have received their annual influenza vaccine may still become ill from other viruses not covered by the immunization and against which they have no immunity.
As this course has stressed, the vaccinia vaccine does not contain smallpox virus. However, vaccinia does cause the vaccinated individual to develop antibodies that are specific for the viruses in the orthopoxvirus group. In this case, one could think of the antibody produced by vaccinia as a "master vaccination" that can protect against all of the orthopoxviruses. Therefore, the vaccination also is effective in protecting against smallpox [31,63].
In addition, vaccinia produces cell-mediated immunity, which may be even more significant in preventing smallpox [14]. In every person with a healthy immune system, humoral and cellular immunity always operate simultaneously. The immune response is a complicated process. In the following section, the complicated immune response is simplified in an attempt to make it understandable and useful for all healthcare professionals. Those who are familiar with the immune process may find this section to be a review.
Three types of cells are involved—macrophages and two types of lymphocytes. Certain macrophages present the antigen (variola virus, in the case of smallpox) to the lymphocytes. The lymphocytes are either B-cells (so named because they are mainly formed in the bone marrow) or T-cells (from the thymus; there are several subclasses).
The B-cells have antibody-like receptors. When the macrophage presents the antigen, the B-cell differentiates to form an antibody-synthesizing plasma cell and also divides to form a clone of identical cells. The plasma cell leads to immunoglobulins that neutralize viral infectivity and kill infected cells. The clone cells are memory B-cells that mount a secondary response if re-infection occurs [64].
T-cells also react specifically to different antigens, as they too have different kinds of antibody-like receptors on their surface or "overcoat." T-cells expand clonally and save a small portion of each clone as long-lived memory T-cells that are available if re-infection occurs. In addition, T-cells liberate substances called "lymphokines" that serve to activate macrophage cells, with the result that infected host cells are killed nonspecifically. Subclasses of T-cells are helper T-cells (Th) and suppressor T-cells (Ts). The helper and suppressor T-cells modulate the activity of the antibody-producing B-cells and other T-cells [64]. Another T-cell subclass is cytotoxic T-cells (Tc), which actively destroy infected host cells and produce lymphokines. Delayed hypersensitivity T-cells (Td) differentiate on contact with the specific antigen, release lymphokines, activate macrophages, and produce delayed hypersensitivity reactions. T-cells also produce gamma interferon [31,63,64]. Thus, vaccination helps protect the vaccinee through humoral and cellular immunity. Several factors influence the degree of protection [63]:
The time between vaccination and exposure (the longer the time, the less the protection because immunity wanes)
The ability of the vaccinee's immune system to respond to the vaccination
The viral load acquired and the amount of time between the exposure and the vaccination (if exposed)
All candidates for the vaccine in the pre-event program should receive culturally appropriate education on the contraindications of the vaccine in the language that is easiest for them to understand. Because vaccinia has possible severe adverse effects, it is extremely important to carefully screen each candidate for known contraindications, which will be discussed in detail later in this course. Persons with the identified contraindications could suffer significant complications and, in some cases, death if exposed to vaccinia, either through personal vaccination or contact with a vaccinated individual. No absolute contraindications exist to defer vaccination for persons with high-risk exposure to smallpox; persons at greatest risk for experiencing serious vaccination complications also are at greatest risk for death if they become infected with the smallpox virus [7].
In a pre-event program, smallpox vaccination has not been recommended for anyone younger than 18 years of age and is contraindicated for persons with any of the following [7,65,66]:
History or presence of eczema or atopic dermatitis
Other acute, chronic, or exfoliative skin conditions
Conditions associated with immunosuppression
Therapy with alkylating agents, antimetabolites, radiation, tumor necrosis factor inhibitors, and/or high-dose corticosteroids
Hematopoietic stem cell transplant recipients
Autoimmune disease (e.g., systemic lupus erythematosus)
Age younger than 1 year
Serious allergy to any component of the vaccine
Pregnant or breastfeeding
Heart disease or three or more risk factors for cardiac disease
Persons with inflammatory eye disease requiring steroid treatment risk inadvertent inoculation as a result of rubbing or touching the eye. The ACIP has recommended that these individuals defer vaccination until the condition resolves and the course of therapy is complete [7,65].
Any person who has ever been diagnosed with eczema or atopic dermatitis should not be vaccinated, even if the condition is currently not active. Because the majority of primary care providers do not distinguish between eczema and atopic dermatitis, including when describing chronic exfoliative skin conditions among infants, ACIP has recommended that smallpox vaccine not be administered to persons with a history of eczema or atopic dermatitis, irrespective of disease severity or activity [7]. These candidates are at high risk of developing eczema vaccinatum, a potentially severe and sometimes fatal complication.
If the potential vaccinee has other acute, chronic, or exfoliative skin conditions, such as burns, impetigo, chickenpox, contact dermatitis, shingles, herpes, severe acne, psoriasis, or an itchy, red, scaly rash that has lasted more than two weeks and tends to reoccur, he/she should not be vaccinated until the condition resolves. These conditions make the person at risk for inadvertent autoinoculation of the affected skin with vaccinia [7]. The vaccine site itches and may be unconsciously scratched, transferring the live vaccinia virus when the individual then scratches the skin lesions.
Additionally, persons with Darier disease, in which warty papules and plaques develop on the skin, may develop eczema vaccinatum and so should not be vaccinated [7].
There are more people with immune system problems today than in the years when vaccination against smallpox was routine. Any person with immunodeficiency or immunosuppression is at greater risk of progressive vaccinia, which results from unchecked replication of the virus [14]. Human immunodeficiency virus/acquired immunodeficiency syndrome (HIV/AIDS), solid organ or stem cell transplants, generalized malignancy, leukemia, lymphoma, humoral or cellular immunity disorders, and autoimmune diseases are all conditions that preclude vaccination.
Persons with HIV infection or AIDS might have an increased risk for severe adverse reactions resulting from live-virus vaccines. The HIV epidemic began after the cessation of routine smallpox vaccination, so data are limited regarding the risk from vaccination among HIV-infected persons. Therefore, risk assessment followed by counseling and testing is useful in identifying persons with HIV infection [7]. Because substantial numbers of HIV-infected persons might not recognize or acknowledge their risk during risk-assessment screening, candidates also should be educated to screen themselves if they have a lifestyle that would put them at risk for HIV [7].
There is no mandate for the agency providing the vaccination to also provide HIV testing; however, ACIP has recommended that testing be readily available [7]. Information on testing sites should be included in the educational materials provided to all potential vaccinees. In addition, the FDA has approved a rapid test of cell-mediated immunity, which is designed to quickly assess an individual's degree of immunosuppression [67].
Other primary immune deficiency diseases include cell-mediated immune deficiencies, such as severe combined immunodeficiency disease (SCID), Wiskott-Aldrich syndrome, DiGeorge syndrome, combined immune deficiency, and ataxia telangiectasia [68]. Antibody deficiency diseases that subject the patient to adverse events, if vaccinated, include X-linked agammaglobulinemia and common variable immune deficiency. It is important that candidates for vaccination be asked questions, in a language that is familiar to them, that would help to identify those who are undiagnosed with primary immune deficiencies [45].
Treatments such as radiation therapy, antimetabolites, alkylating agents, corticosteroids, chemotherapy agents, or organ transplant medication may suppress the immune system. People who are taking or have taken high-dose corticosteroids should not be vaccinated within one month of completing corticosteroid therapy, and persons treated with other immunosuppressive drugs within the previous three months should not be vaccinated [7]. High-dose corticosteroid therapy is defined as >2 mg/kg/day or prednisone at 20 mg/day for >14 days [7].
Generally, live virus vaccines are contraindicated during pregnancy. Pregnant women who receive the smallpox vaccine put the fetus at risk for fetal vaccinia. This is a rare, but serious, complication, usually resulting in stillbirth or death of the infant shortly after delivery [7]. Before vaccination, all women should be informed about this possible complication and screened for pregnancy or the intent to become pregnant within the next four weeks. Women who respond positively should not be vaccinated. Women also should be counseled to avoid becoming pregnant until at least four weeks after vaccination, and abstinence or highly effective contraceptive measures should be recommended to reduce the risk of pregnancy within four weeks after vaccination. Routine pregnancy testing of women of childbearing age has not been universally recommended but should be performed if desired by the woman or her medical provider [7].
Should a pregnant woman be inadvertently inoculated, she should be counseled regarding her concern for the baby. However, vaccination during pregnancy should not ordinarily be a reason to terminate the pregnancy. Because live vaccinia virus may be transmitted from a lactating mother to her infant, causing complications in the infant from inadvertent inoculation, a mother who is breastfeeding should not be vaccinated [7,9,66].
ACAM2000 contains neomycin and polymyxin B. Persons allergic to these components may be at higher risk for adverse events after vaccination [66].
A history of cardiac disease is a fairly recent addition to the list of contraindications to receiving vaccinia [9]. Ischemic cardiac events, including fatalities, have been reported following smallpox vaccination, although a relationship between these events and vaccination has not been established. Non-ischemic dilated cardiomyopathy has been reported following vaccination, although, again, a relationship to vaccination is unknown [66]. Candidates with known cardiac disease, including those diagnosed with previous myocardial infarction, angina, congestive heart failure, cardiomyopathy, chest pain or shortness of breath with activity, stroke or transient ischemic attack, or other cardiovascular conditions, may be at increased risk of adverse events from vaccination and should defer it. In addition, persons with three or more risk factors for coronary disease (i.e., high blood pressure, elevated blood cholesterol, diabetes mellitus or high blood glucose, first-degree relative with heart condition before 50 years of age, and cigarette smoking) also should defer vaccination [7,65,66].
Now that several years have passed since the implementation of smallpox vaccination for civilian healthcare workers in 2002–2004, research has determined that there is no causal relationship between vaccinia administration and myocardial infarction and other ischemic events. Of those with myocardial events, 96% recovered completely [19].
One death among DoD-vaccinated personnel may be attributed to the vaccine [19]. The individual, a woman 22 years of age, received vaccinia and four other vaccines at the same time, after which she developed a lupus-like condition. She died 33 days after receiving the vaccinations. In addition, the DoD reported that 10 of the initial 438,000 smallpox vaccinees had undiagnosed HIV. All 10 had a normal reaction to the vaccine including healing [19].
Because vaccinia is a live vaccine and will be shed from the vaccination site, household contacts should be considered at risk if they have any contraindications to the vaccine. When screening for a pre-event program, household contacts include persons with prolonged intimate contact (e.g., sexual contact) with the potential vaccinee as well as anyone who might have direct contact with the vaccination site. The contraindications for household contacts include [7]:
Skin conditions (as listed previously)
Any immunosuppression due to HIV/AIDS, generalized malignancy, leukemia, lymphoma, solid organ or stem cell transplantation, humoral or cellular immunity disorders
Autoimmune diseases
Pregnancy
Heart disease, such as coronary artery disease and congestive heart failure, or three or more risk factors for cardiac disease, such as diabetes, high cholesterol, and high blood pressure
Any candidate for vaccination who has a household contact with any contraindication should not be vaccinated. Because vaccination has been mandatory for select military personnel who have no contraindications, household contacts of vaccinated military personnel should be aware of the risks [10].
Contacts at most work sites and social events are not considered close contacts; therefore, their contraindication status does not need to be considered. However, when at work, in social situations, or when interacting with household members, all vaccinees should have the vaccination region covered with an appropriate dressing and a long sleeve until the scab falls off.
The presence of an adolescent or child (including an infant) in the household is not a contraindication to vaccination of adult members of the household. Nonetheless, programs might defer vaccination of household contacts of infants younger than 1 year of age because of data that has indicated a higher risk for adverse events among primary vaccinees in this age group, compared with that among older children. Neither the presence of a breastfeeding woman nor the presence of a person with a vaccine component allergy in the household are contraindications to vaccination of other household members [7].
Because all pre-event vaccinations are voluntary, it is important that each candidate thoroughly understand the vaccine's contraindications, actions, side effects, and possible adverse events and seek the advice of his/her medical provider, if needed. Developing knowledge of smallpox also is an effective way to counteract fear and cope with the threat of bioterrorism. Healthcare professionals should also evaluate personal hygiene habits, such as effective handwashing, to minimize any danger of exposing others.
The FDA has recommended that vaccinees defer donating blood for 21 days or until the scab has come off. Contacts of vaccinees who develop vaccinia should not donate blood for 14 days after full resolution of their complication [7].
As previously stated, the current primary smallpox (vaccinia) vaccine for pre-exposure and post-event use in healthy individuals is ACAM2000, which replaced Dryvax after 2008 [45]. The distribution of the ACAM2000 vaccine is controlled by the CDC and has been purchased (in quantity sufficient to vaccinate 300 million individuals) for inclusion within the SNS, which plans to distribute smallpox vaccines on the first day of a bioterrorism event to anyone who has been exposed. Following this initial shipment, the rest of the country will receive this vaccine or a suitable alternative (i.e., APSV or JYNNEOS/MVA-BN), as needed, over a five- to six-day period. The CDC will ship the vaccine in containers that have been designed to maintain the product at the proper temperature. State and local public health departments should immediately move the vaccine into refrigeration units maintained at those same temperatures and return the containers to the CDC as quickly as possible to facilitate additional shipments [4,69]. Each ACAM2000 unit includes [70]:
One 100-dose vial of smallpox vaccine (vaccinia)
One 100-dose vial of diluent
One bifurcated needle
One transfer syringe
One CD per box; the CD contains an FDA-approved package insert (prescribing information) and medication guide
The bifurcated needles used for Dryvax, APSV, and ACAM2000 are identical in design, production, delivery, and packaging. ACAM2000 bifurcated needles have an expiration date; Dryvax needles did not [71].
Smallpox vaccine must be kept at appropriate temperatures to retain effectiveness. The thermometers in cold storage unit monitors should be annually certified for accuracy. In the event of a power failure, a back-up generator should be available or another cold storage site should be identified for transfer of the vaccine if the power failure continues for more than four hours. The alternate site also should have a temperature monitor system and security protection [72].
ACAM2000 lyophilized should be stored frozen at -25°to -15°C (-13° to 5°F) until distributed for use. Upon distribution, the vaccine may be stored in a refrigerator with a temperature monitor that must be checked and recorded each morning and afternoon to assure that the vaccine is kept between 2° to 8°C (36° to 46°F) for up to 18 months. ACAM2000 reconstituted should be stored refrigerated at 2° to 8°C (36° to 46°F) and used within 30 days of the reconstitution date. The vaccine may be administered within six to eight hours (i.e., one clinic day) if kept at room temperature (20° to 25°C; 68° to 77°F); it should then be discarded as a biohazardous material. The reconstituted vaccine may be returned to refrigerated storage overnight for future use if it has been exposed to room temperature conditions for less than six to eight hours at a time. This cycle may be repeated as many times as is practical (within 30 days of reconstitution date). ACAM2000 has a shelf life of six years from the date of manufacture and should not be used after the expiration date. The average thaw time from frozen temperature to refrigerated temperature is 15 to 30 minutes; the average thaw time from refrigerated temperature to ambient temperature is approximately four hours. Unreconstituted vaccine should not be exposed to room temperature conditions for more than 48 hours [66,71].
Diluent for ACAM2000 should be stored at room temperature (15° to 30°C; 59° to 86°F). The diluent has a shelf life of five years [66,71].
The SNS has indicated the following regarding vaccine security [71]:
There are no mandated security requirements; the level of security should be determined by state or local governments where the vaccine will be used.
Formulation of smallpox vaccine security measures should be based upon risk assessments available to state and local governments.
CDC expects the states to keep the vaccine safe and secure (as with other vaccines) according to the Code of Federal Regulations.
Smallpox vaccine must be stored in fixed storage facilities with controlled access to both the facility and the storage system with surveillance and/or security staff provided. Armed guards on constant duty at the storage facility are not expected.
After the vaccine is distributed to the dispensing clinics, the same precautions for protecting any vaccine should be used.
ACAM2000 should only be reconstituted with 0.3 mL of the diluent provided. The vaccine vial should be removed from cold storage and brought to room temperature before reconstitution. Remove the flip-cap seals of the vaccine and diluent vials and wipe each rubber stopper with an isopropyl alcohol swab. Allow each stopper to dry thoroughly. Using aseptic technique and a sterile 1-mL syringe fitted with a 25-gauge, 5/8-inch needle (provided), draw up 0.3 mL of diluent, and transfer the entire contents of the syringe to the vaccine vial. Gently swirl to mix, but try not to get product on the rubber stopper. The reconstituted vaccine should be a clear to slightly hazy, colorless to straw-colored liquid that is free from extraneous matter. Reconstituted vaccine should be inspected visually for particulate matter and discoloration prior to administration. If particulate matter or discoloration is observed, the vaccine should not be used; the vial should be disposed of safely [66].
Personnel preparing and administering the vaccine should wear surgical or protective gloves and avoid contact of vaccine with skin, eyes, or mucous membranes. The vaccine vial, its stopper, the diluent syringe, the vented needle used for reconstitution, the bifurcated needle used for administration, and any gauze or cotton that came in contact with the vaccine should be discarded in leak-proof, puncture-proof biohazard containers. These containers should then be disposed of appropriately [66].
A package insert is included with each vaccine kit. ACIP recommendations are considered by the CDC and, if adopted as policy, published in the CDC's Morbidity and Mortality Weekly Reports. Although the bulk of recommendations related to smallpox vaccination are still valid, the CDC has encouraged providers to refer to the ACAM2000 package insert and follow the instructions regarding vaccine administration [8].
Only healthcare personnel who have been vaccinated should be vaccinators. Unvaccinated health workers should not handle the reconstituted vaccine, but they may handle vials of vaccine that have not had the diluent added.
ACAM2000 (and other smallpox vaccines) should be administered only by vaccine providers who have been trained to safely and effectively administer the vaccine. The manufacturer is responsible for ensuring that such training is available to all vaccine providers, as required by the manufacturer's Risk Minimization Action Plan (RiskMAP), a strategic safety program, designed to minimize the risk that may occur with administration of the vaccine [4,66]. The components of the RiskMAP include education, an adverse event reporting and collection system, and risk management evaluation [4].
Candidates for vaccination should be given an educational packet on the vaccine and its contraindications, at least a few days before the vaccination, to allow them time to carefully read and assimilate the material. This also will give the candidate time to seek the guidance of his/her personal medical provider. The staff onsite should review possible contraindications and the informed consent with the potential vaccinee and either answer any questions or refer the candidate to appropriate medical personnel for answers. Finally, the vaccinator should review the contraindications one last time with the potential vaccinee, as some people are screened-out just before the vaccine is administered. Latex gloves have been recommended for administration of the vaccine [66].
Because the CDC does not have enough information to determine the safety of administering the vaccine at other sites, the deltoid area of either arm is the preferred site for vaccination. This area offers ease of observation, ease of dressing changes, and is easier to keep dry while bathing or showering [8]. Some people may prefer that the nondominant arm be used. As previously stressed, alcohol should never be used as it inactivates the vaccine [7,8]. If there is visible dirt, the arm should be washed with soap and water and allowed to thoroughly dry to avoid inactivating the vaccine. Otherwise, no cleaning or site preparation is required unless the area is grossly contaminated [8].
Because vaccinating over a tattoo may change its appearance, the CDC has recommended that vaccinees be informed of this possibility and also that no data about what the use of an altered site means in terms of local adverse events or autoinoculation risk. Otherwise, the CDC has no standard policy concerning tattoos [8].
The vaccination needle is 6.5 cm in length and thin. The bifurcated prongs are approximately 2 mm in length. They are packaged for single-use and are sterile. The needle should be inserted into the vial just far enough to have the bifurcated tip touch the vaccine, allowing the needle to pick up a droplet of vaccine. If the needle is inserted too deeply, too much vaccine will be picked up because the vaccine is sticky. The needle should be inserted straight into the vial and then pulled straight out. The upper part of the needle (the part that has been in contact with fingers) should not be inserted into the vaccine vial [66]. The needle must not be reinserted into the vial after touching the patient's skin. Vaccinators who have presbyopia should wear their glasses, as the minute amount of vaccine is difficult to see.
The vaccinator's free hand should grasp the vaccinee's upper arm in a way to make the skin taut. Preferably, the arm should be held from underneath as the skin may be held taut effectively, and the vaccinator's hand may be protected from an accidental needle stick. The wrist of the hand holding the needle of the vaccinator should rest against the vaccinee's arm [66]. The vaccinator should rapidly make 15 jabs of the needle perpendicular to the skin through the vaccine droplet (approximately a 2.5-mcL dose) to puncture the skin, within a diameter of about 5 mm. The jabs should be vigorous enough to bring a drop of blood, after 15 to 20 seconds, at the vaccination site [66]. ACAM2000 uses 15 insertions for both primary (has not been previously vaccinated) and revaccination and has no provision for additional insertions if no trace of blood is visible after vaccination [8]. The needle should not be redipped into the vaccine after it has touched the vaccinee's skin. After use, the needle should be discarded in a suitable biohazard receptacle [66].
The vaccine site should be dabbed with a sterile gauze pad to remove any excess vaccine and blood; the gauze should then be discarded in a biohazard waste receptacle. Unless the vaccine vial will be immediately used to vaccinate another subject, the vial should be closed by reinserting the rubber cap; the vial should then either be returned to refrigerated storage or placed on ice [66].
The vaccination site should be covered loosely with a gauze bandage and secured with first aid adhesive tape to prevent spreading of the vaccinia virus. It is important to note that only persons working in healthcare settings should use semipermeable dressings. This is because additional coverings at the vaccination site may cause maceration and may increase and prolong irritation and itching, thereby increasing the potential for contamination of the hands. Dressings should be changed frequently to prevent the accumulation of exudates and consequent maceration [7]. Hospitals should designate staff to assess dressings for all vaccinated healthcare workers; when feasible, these staff should be vaccinated. All persons handling dressings should observe contact precautions [7].
After the site has been dressed, the vaccinator may remove the gloves and wash his/her hands with antimicrobial soap and warm water or with alcohol-based hand rubs (e.g., gels or foams). A new pair of gloves should be donned before the next person is vaccinated [8]. Good handwashing hygiene by the vaccinator is imperative to prevent any vaccine from being introduced inadvertently at any place other than the vaccine site [7,66].
Although "dry" hand wash preparations containing >60% alcohol are suitable for disinfecting the hands after touching one's own vaccine site, they should not be used in the vaccinated area because of the danger of accidentally inhibiting the vaccine [7].
In a post-event vaccination setting, the same administration dose (i.e., vaccine droplet of approximately 2.5-mcL dose) and procedure (i.e., scarification using 15 jabs of a stainless steel bifurcated needle) for ACAM2000 is also to be followed for the APSV formulation, if provided by CDC [45]. The administration of JYNNEOS/MVA-BV is one 0.5-mL subcutaneous injection at 0 and 4 weeks for primary vaccinees (i.e., two doses) and a single 0.5-mL dose for previously vaccinated individuals; these are supplied in single-dose vials. JYNNEOS/MVA-BV is indicated for healthy individuals and individuals with atopic dermatitis, other skin disorders, allergic rhinitis, and/or immunodeficiency [45].
Because of the danger of transferring the live vaccinia to another site on one's own body (autoinoculation) or to a close contact, care of the vaccination site should be carefully taught to each vaccinee and thoroughly understood by both the vaccinator and the vaccinee. There should be both verbal and written instructions in a language the vaccinee can easily understand. As previously stated, every person who receives the ACAM2000 vaccine must receive a medication guide [4]. Among other things, the guide answers questions about how to care for the vaccination site [66].
There are three possibilities for care of the vaccination site: no dressing, simple gauze, or a two-layer dressing of gauze covered with a semi-permeable dressing [74]. The important point is to ensure that the vaccination site is covered to prevent dissemination of the virus [50].
The vaccination must be allowed to dry as airing the site will speed healing; therefore, there will be times when the vaccine site may be left uncovered. It is important to note, however, that this should be limited to periods when the vaccinee will not have contact with anyone else. When around others, the vaccinee should always wear a garment with sleeves and/or an absorbent bandage to form a touch-resistant barrier [74].
Paper tape may be used to hold the dressing; however, microspore tape holds better. It is important that the dressing be held securely. To control any exudates, the "picture frame" method of taping is best. Only the outer edges of the gauze should be taped in order to ventilate the site. The gauze dressing should be covered by a washable long sleeve shirt. Patient history of tape sensitivity should be taken prior to dressing in this manner. Rolled or adhesive gauze also may be used to reinforce the gauze or to keep the dressing in place, especially if the vaccinee is having skin problems as a result of the tape.
This dressing is not appropriate for patient care water activities, such as bathing, although normal bathing may continue. The vaccinee should dry off carefully to ensure that the towel does not rub or spread the virus elsewhere [74]. All water activities that require immersion, such as swimming, should be avoided until the vaccine site scab has fallen off. Waterproof adhesive bandages may be worn if the vaccinee will exercise enough to cause sweating [74].
When working in a healthcare setting, vaccinees should cover the vaccine site with gauze and a semipermeable dressing placed over the gauze. The semipermeable dressing used must be one in which there are no added compounds, such as a microbicide. When placing the semipermeable dressing, make sure that all the edges adhere well to complete the seal. If not, reinforce the edges. Products that combine an absorbent base with an overlying semipermeable layer also may be used to cover the vaccination site. Healthcare workers do not need to be placed on leave after receiving a smallpox vaccination [66].
Any vaccinee in a setting where close personal contact is likely (e.g., parents of infants and young children) should cover the vaccination site with gauze or a similar absorbent material, wear a shirt or other clothing that covers the vaccination site, and practice good hand hygiene [66].
To avoid contact transmission of the virus, vaccinees should be cautioned to do the following [66]:
Do not rub or scratch the vaccination site.
Keep the site covered and change gauze-only dressings every one to two days, or if wet. Change semipermeable dressings at least every three to five days.
Keep the vaccination site dry; cover with a waterproof bandage while bathing.
Discard gauze carefully in plastic zip bags.
Set aside a laundry hamper for clothes, towels, sheets, and other items that may come into contact with the vaccination site.
Wash clothing or other materials that come into contact with the vaccination site in hot water with detergent and/or bleach. Wash hands afterward.
Wash hands thoroughly with soap and hot water or with alcohol-based hand rubs, such as gels or foams, after touching the vaccination site or bandages, clothing, towels, or sheets that have come into contact with the vaccination site.
When the scab falls off, throw it away in a plastic zip bag.
Report any problems immediately.
Return seven days after vaccination to determine whether the vaccination was successful.
Effective handwashing, using soap and water, results in the removal of the oils that are on the skin. Viruses and bacterial organisms are trapped in the oils produced by the skin. A quick pass under the faucet, even with some soap, may not be sufficient to remove the oils, and organisms may remain on the skin.
To remove the oils and organisms, soap must be used and the hands should be well lathered, vigorously rubbed, and then rinsed well under running water. A minimum of 20 seconds of lathering, rubbing, and rinsing are needed to assure that the oils and organisms are removed (imagine singing "Happy Birthday" twice through). Warm water, if available, is suggested for comfort, but the water used for hand washing would never be hot enough to kill the organisms. Rinse hands well under running water and then dry, using either a paper towel or an air dryer. Ideally, use the paper towel to turn off the faucet and then discard the towel appropriately; using an elbow to turn off the faucet is an alternative [75,76].
Soap and water are the preferred way to clean the hands, but in situations where soap and water are not available or are impractical, alcohol-based preparations may be used. It is important that these preparations have an alcohol content of >60% [75,77]. When using an alcohol-based hand sanitizer, apply the product to the palm of one hand, rub the hands together, and then rub the product over all surfaces of the hands and fingers until the hands are dry [75,76]. Never clean the vaccination site with a product containing alcohol and do not use an alcohol-based sanitizer near the site.
Usually, for the first two days after vaccination, the site appears unchanged and does not shed vaccinia (Figure 2). A papule will normally develop on either the third or fourth day. Whenever the site appears to have a "sore," vaccinia is shed and the vaccinee can transmit the virus to others or to other areas, especially the eyes. By the fifth to sixth day, the papule becomes a vesicle with erythema of various sizes around it. This vesicle will become depressed in the center. The vesicle becomes a well-formed pustule (mean diameter of 12 mm) by day eight or nine. After 12 or more days, the pustule crusts over and forms a scab. Normally, the scab falls off between the 17th and 21st days, revealing a scar. After the scab is gone, the vaccinee no longer has the potential to shed the virus and can no longer transmit vaccinia [78]. Images of these normal reactions may be seen on the CDC smallpox website.
The vaccine sites of both first-time vaccinees and revaccinees should be checked by a medical professional between days 6 and 8 [79]. Vaccine site evaluations on other days may not be reliable. For example, if the evaluation is done too early, certain "non-takes" may look reactive; if done too late, the vaccination "take" might be missed [79]. At the time the vaccine is administered, the vaccinee should be told where and when to have this check performed.
A"major reaction" or "take" indicates that the vaccination has been successful and the vaccinee should be developing immunity to orthopoxviruses. The take is characterized by a pustular lesion or an area of definite induration or congestion surrounding a central lesion, which may be either a scab or an ulcer. All other responses should be considered "equivocal" (a"non-take") [79].
In some cases, the site has a large area of erythema with possible induration that some inspectors may think is infected. However, this is a robust or exuberant reaction to the vaccination. The edema and induration are uniform, versus the irregularly raised surface and signs of impetigo present in a bacterial cellulitis. The robust reaction resolves within a few days; it is not infected and antibiotics are not required [80].
On the seventh day following vaccination, if the site does not have the vesicle, it is termed "equivocal" (a"non-take"). Equivocal reactions consolidate a variety of previously used terms, including accelerated, modified, vaccinoid, immediate, early, or immune reactions. Equivocal reactions have been defined as all responses other than "major reactions." There also may be no reaction at all. About 5% of vaccinations are equivocal [74]. There may also be an allergic reaction, which does not indicate immunity. An equivocal reaction means that there is no immunity and the vaccination must be repeated, preferably immediately after the evaluation. Usually, the second vaccination results in a take, but occasionally there is an equivocal reaction or no reaction even with the second vaccination. Equivocal readings may be caused by a number of factors, including improper vaccination technique, use of vaccine that has lost its potency, or residual immunity among previously vaccinated individuals. Persons with an equivocal reaction cannot be presumed to be immune to smallpox; revaccination is recommended [79].
Vaccinia will cause a local reaction at the vaccination site. This is desirable in order to develop immunity. Some vaccinees also may have systemic reactions. These are expected from a vaccination with vaccinia and will be briefly described in the following sections.
In addition to the reaction at the site that leads to immunity, there are other possible expected reactions. The most problematic of these is the robust or exuberant reaction or intense erythema and induration ringing the site [80]. As previously noted, practitioners unfamiliar with evaluating a vaccine site tend to incorrectly assume that a robust reaction is infected. Hopefully, as more practitioners become familiar with reading vaccinations, the diagnosis between a robust reaction and an infected vaccine site will be easier to make.
Other local reactions may include soreness at the vaccination site, reactions to the tape, and local lymphadenopathy [80,81]. Tape "burns" are distinguished by erythema corresponding to tape placement. Treatment consists of antihistamines and a change in type of tape or gauze wrap.
Systemic symptoms are expected and usually occur 8 to 10 days after vaccination when the vaccine site reaction reaches the peak of the inflammatory response. Occurrence of the following symptoms has been shown to vary considerably between primary vaccinees (higher rates) and revaccinees (lower rates) [78,81]:
Intense erythema ringing the vaccination site
Malaise
Myalgia, headache, chills, nausea, fatigue (0.3% to 37%)
Fever >37.7°C (2% to 16%)
Normal variants that are not considered adverse events include [78,81]:
Local satellite lesions (that are normal in appearance); the frequency of satellite lesions varies between 2.4% to 6.6%
Lymphangitis in the regional nodes with resulting lymphadenopathy
Considerable local edema
Intense erythema (viral cellulitis) at the site
None of these expected normal variants require treatment other than symptomatic relief [78].
Autoinoculation is the transfer of or appearance of vaccinia lesions elsewhere on the body. Because vaccinia is shed from the site after the vesicle appears and until the scab falls off, a vaccinee whose hand hygiene is inadequate may inadvertently inoculate other sites on his/her person. The most common sites are the face, eyelid, nose, mouth, genitalia, and anal region [80,81]. Infants and children are most susceptible because they tend to scratch the itching vaccination site [81]. Skin with disrupted integrity and the eyes are especially vulnerable as the infection may be extensive in skin lesions and a threat to eyesight. Data from the 1960s have revealed that accidental implantation of vaccinia is the most frequent complication of vaccination (i.e., 25.4 per million primary vaccinees, which accounted for approximately one-half of all the complications of primary vaccination and revaccination) [82]. This complication highlights the need for a proper dressing on the site and a sleeve that prohibits scratching, as well as attention by the vaccinee to good hand hygiene [80].
Lesions from autoinoculation will follow the same course as the vaccination site. Patients with cell-mediated immune dysfunction are at risk for lesions that will not heal and will expand. There is no treatment required if there are only a few lesions. Multiple lesions, especially if they are confluent and cover large portions of the body, should be treated with vaccinia immune globulin (VIG), which is discussed later in this course [81].
Vaccinia keratitis is a rare complication that results from the introduction of vaccinia from the vaccination site to the cornea. It is characterized by corneal lesions, abrasions, ulceration, and subsequent cloudiness [80,81]. The lesions are often indurated, edematous, and infiltrated. If the condition progresses untreated, it may lead to scarring and permanent impairment of vision. Individuals with pre-existing eye conditions, especially those that are inflammatory in nature, are more susceptible to implantation. Those who care for children who have received vaccinia are the most likely to develop vaccinia keratitis [80]. An ophthalmology consultation should be obtained for evaluation and management of keratitis and other eye complications resulting from inadvertent ocular implantation of vaccinia virus. The best method for diagnosis and monitoring of changes and response to treatment is slit-lamp evaluation. Treatment should include topical antiviral medications as advised by an experienced ophthalmologist. VIG is contraindicated and, if used, may increase the risk of scarring [83].
As with any skin lesion, other infectious organisms may be introduced, although this is an infrequent complication. Site manipulation increases the possibility of contamination, especially among children. Prolonged use of occlusive dressings may lead to maceration and increased risk of infection. The usual organisms are Staphylococcus aureus, group A streptococci, and group A beta hemolytic streptococci. Fecal contamination of skin is common in infants. Bacterial infection is common among the immunodeficient. There also may be anaerobic or mixed infections [80,81].
As discussed, the surface in a bacterial cellulitis infection will be irregularly raised, in contrast with an exuberant reaction in which the surface is of one level. After culture and sensitivities of the lesion, appropriate antibiotic treatment should be initiated [80,81].
Approximately 10 days after primary vaccination, one person in 1,000 will develop a toxic and/or hypersensitive rash [84]. The rash may be erythematous, vesicular/pustular, urticarial, or a typical "bull's-eye" lesion. All are grouped under the term "erythema multiforme." The vaccinee is usually afebrile, the rash does not progress, and it resolves spontaneously within two to four days. A more serious rash, bullous erythema multiforme (Stevens-Johnson syndrome), occurs, but rarely. Symptomatic treatment using antihistamines and other measures to counteract itching is usually sufficient. However, patients with Stevens-Johnson syndrome should be hospitalized for supportive care [81].
Generalized vaccinia (GV) is rare and probably caused by viremia. It consists of vesicles or pustules appearing on normal skin far from the vaccination site. The 1960s data has estimated that GV will occur in 242 per million primary vaccines [82]. Rates in the United States today may be higher because there may be more persons at risk from eczema or atopic dermatitis and immune suppression from cancer, cancer therapy, organ transplantation, and other illnesses (e.g., HIV/AIDS) [80]. The disease is usually mild, self-limited, and has little residual damage. Mild cases require no treatment, but more extensive lesions, especially in a vaccinee with an underlying immunosuppressed illness, may be toxic and treatment with VIG will be necessary [80,81,82].
In this course, adverse events are defined as life-threatening and include eczema vaccinatum, vaccinia keratitis, progressive vaccinia, post-vaccinial encephalitis, and fetal vaccinia. In the past, between 14 and 52 vaccinees per million vaccinated have had life-threatening reactions to vaccinia, and 1 to 2 died [82].
Either the healthcare professional or the patient should report an adverse event by using the Vaccine Adverse Events Reporting System (VAERS). Electronic reporting of all smallpox vaccine adverse events is strongly encouraged because it is the most timely and efficient method of providing information [85]. Adverse events that are serious or unexpected and require expert consultation or IND therapeutics (cidofovir) should be immediately reported by telephone to the local health department or the CDC and also should be reported to VAERS as soon as possible. A VAERS report should be submitted even if it is not certain that the event was caused by the smallpox vaccine [85].
Eczema vaccinatum is a localized or systemic spread of vaccinia on individuals who have eczema or atopic dermatitis, even if the skin condition is not active at the time of the vaccination [80]. There are an estimated 18 million adults and 9.6 million children younger than 18 years of age in the United States who have atopic dermatitis, many of whom would be susceptible to eczema vaccinatum if vaccinated or in contact with a vaccinee [86]. Data from the 1960s have indicated an occurrence of 10.6 cases of eczema vaccinatum per million among primary vaccinees 1 year of age or older [82].
The patient is infected through autoinoculation or from contact with a vaccinee who is shedding the virus. The virus is implanted into broken skin at one or multiple sites and it spreads, producing extensive lesions wherever there is abnormal skin. The patient should be hospitalized immediately and treated with VIG. Death has been prevented in patients with eczema vaccinatum by prompt and adequate treatment; however, even when there is a delay in recognition, prompt institution of VIG should be undertaken; scarring may be extensive [80,81,82].
In progressive vaccinia, also known as vaccinia necrosum, the vaccination site does not heal and continues to spread locally and by viremia to other parts of the body. There is no inflammatory response. If untreated, patients succumb to the viral effect or to secondary infections. A rare event in the past (i.e., 1 of every million primary vaccinations), progressive vaccinia may be a greater threat today given the larger proportion of susceptible persons in the population [82].
Progressive vaccinia occurs mainly in persons with T-cell deficient diseases (e.g., cancer, immunosuppressive therapy, HIV/AIDS). Viral and immunologic studies should be conducted after consultation with state or local health authorities, infectious disease or immunology experts with experience in complications of vaccination, and/or the CDC [80].
The treatment is VIG in massive doses [80,81,82,83]. (Up to 10 mL/kg of intramuscular VIG has been used.) Research is being conducted on VIG, and there are IND protocols that may result in new formulations of the drug. Both intravenous and intramuscular preparations are being produced. Immediate consultation with the CDC has been recommended to determine if any experimental antiviral drugs are available [80]. Future dosing will depend on the manufacturer's recommendations and experience [80,81,82].
Post-vaccinial encephalitis or meningoencephalitis is a rare event of primary vaccination, occurring in approximately 12.3 per million vaccinees [82]. Ten to fourteen days after vaccination, the patient will have initial symptoms of headache, vomiting, drowsiness, and fever. Approximately 15% to 25% of vaccinees with this complication die, and another 25% develop permanent neurologic sequelae [80]. There is no specific therapy. Supportive care, anticonvulsants, and intensive care may be required [81]. VIG is not effective and has not been recommended [82].
Fetal vaccinia is infection of the fetus in the last trimester with evidence of disease in the newborn infant. It is so rare that there are fewer than 50 known cases [82]. Most of these occurred in primary vaccinees who were vaccinated in early pregnancy. The result usually was stillbirth or death of the infant soon after birth. Vaccinia is not known to cause congenital malformations [82].
As discussed, vaccinia has complications that are mild to moderate and usually self-limited. Fortunately, serious adverse events are rare, but may be fatal; however, they require immediate evaluation for diagnosis and treatment. Currently, there are two available treatments: VIG, the first-line therapy, and cidofovir, the second-line therapy [82,83].
VIG has been used in the past and was felt (but not shown through controlled studies) to be effective. VIG was originally produced in the 1960s from plasma obtained from recently vaccinated donors. Like other globulins, VIG provides passive immunity. Because of the high proportion of aggregated protein, it was originally administered solely by the intramuscular route; it could not be given intravenously [82,83].
In 2005, the FDA approved CNJ-016, vaccinia immune globulin intravenous (human) (VIGIV), for use in the treatment of adverse events resulting from the administration of vaccinia [87]. VIGIV has a low level of aggregated protein, so it may be used both intravenously and intramuscularly [80,83]. Absolute contraindications for the use of VIGIV include hypersensitivity to immune globulin or any component of the formulation, isolated vaccinia keratitis, and selective IgA deficiency [70,87]. The dosing recommendation for CNJ-016 (the Cangene product) is 6,000 units/kg; however, 9,000 units/kg may be considered if the patient does not respond to the initial dose. The dosing recommendation for the DynPort product is generally 100 mg/kg; however, higher doses (200–500 mg/kg) may be administered if the patient does not respond to the initial recommended dose [70].
VIG has not been recommended for the following reactions [70,83]:
Autoinoculation with few lesions
Bacterial infections
Erythema multiforme
Generalized vaccinia that is mild and limited
Vaccinia keratitis
Post-vaccination encephalitis
VIG has been recommended for the following reactions [70,82,83]:
Autoinoculation with extensive lesions
Eczema vaccinatum
Generalized vaccinia that is severe or recurrent
Progressive vaccinia/vaccinia necrosum
Cidofovir is an antiviral drug with demonstrated activity against poxviruses in vitro and against cowpox and vaccinia viruses in mice. It has been approved for the treatment of cytomegalovirus (CMV) retinitis in patients with AIDS; it must be administered with probenecid [70]. Cidofovir must be used under an IND protocol in the treatment of vaccinia adverse events. Under the protocol, cidofovir may only be used when VIG is not efficacious [83]. Contraindications for use of cidofovir include hypersensitivity to cidofovir or a history of clinically severe hypersensitivity to probenecid or other sulfa-containing medications [70]. Nephrotoxicity is a known side effect [70,81,82,83]. In addition, in animal studies, cidofovir has not been effective after infection is established [19].
Preventing smallpox through vaccination is the current approach to fighting the disease. However, a new drug, Arestvyr (formerly known as ST-246), has been shown to provide protection against severe disease and death by reducing viral levels of vaccinia, variola, and monkeypox viruses [88]. The data show that Arestvyr limits the risk of vaccinia transmission after vaccination to susceptible contacts. It has also effectively reduced the disease burden and fatal outcomes in nonhuman primates against a variola challenge, and along with VIG, Arestvyr has been used to treat a severe case of eczema vaccinatum and progressive vaccinia in humans [88]. This novel smallpox antiviral agent is being stockpiled as an IND at the SNS.
When vaccination against smallpox was routine, the medical profile of the population was different than it is today. There was no HIV/AIDS, there were fewer cancer survivors, and organ transplants were rare; as a result, there was less immunosuppression. Most primary vaccinations were done on toddlers after their first birthday, thus pregnancy in the vaccinee was not an issue. Revaccinees were often individuals entering medical professions, military recruits, travelers over international borders, and people being treated for herpes simplex. Since then, there have been many successful efforts to improve all vaccines to reduce risks to the recipients. As a result, many in the public are not accustomed to an immunization with such serious adverse events. Reintroduction of vaccinia to a population with increased contraindications requires that medical professionals have a respect for both the vaccine and the necessity for any potential candidate to thoroughly understand the risks.
Healthcare professionals must understand the issues surrounding vaccination well enough to be able to explain the contraindications on a level that each potential candidate for vaccination can understand. Healthcare professionals also must examine their own feelings about the pre-event vaccination program to avoid influencing a candidate's decision about participation.
It is important to note that the ACIP has advised against non-emergency use of smallpox vaccine in anyone younger than 18 years of age [7]. Younger individuals should not receive the vaccine unless they have been exposed to smallpox. It is unlikely that healthcare response team members will be younger than 18 years of age, but they may be 65 years of age and older. Vaccination of persons 65 years of age and older has not been recommended in a non-emergency situation, unless they are smallpox response team members with no contraindications to receiving the vaccine [7]. Only immunocompromised individuals known to be exposed to smallpox should be vaccinated (with JYNNEOS/MVA-BV) post-event when antivirals are unavailable [45]. If it is suspected that an immunodeficient individual will not benefit from the vaccine (i.e., has not been/will likely not be exposed), it should not be given.
Candidates should be given information about the vaccine, especially the contraindications, through several means—videos, oral instruction, and written materials. Then, a few days should be provided to assimilate the materials. When the candidate returns for vaccination, a screener with a medical background should interview the candidate. Screening forms, completed by the candidate, should be reviewed at this time. Any unanswered questions that either the screener or the candidate has after this interaction should be referred to a physician available at the vaccination site [89]. It is difficult to address all possible questions about real or hypothetical conditions, situations, or combinations of factors that do or could exist. It is important to remember that there is currently no smallpox disease with which to contend. If individuals eligible for smallpox vaccination are unsure whether their particular set of circumstances places them at greater risk for an adverse reaction to vaccination, they should consult with their primary healthcare provider. If there are reservations or uncertainties, they should not be vaccinated. There is currently no reason to get vaccinated for smallpox if there are any questions or doubts about contraindications [65].
Finally, the vaccinator should discuss with the candidate the list of contraindications (e.g., skin conditions, immunosuppression from any cause, cardiac disease, pregnancy or planned pregnancy, breastfeeding, or allergy to any of the vaccine components) for themselves and also for any household contacts [7,65]. As previously mentioned, some candidates will be screened out at this last stage, and there will be some individuals who decide not to be vaccinated for religious, cultural, or other reasons.
Informed consent is now the backbone of Western bioethics; however, it was not an ethical mandate until 1957, when it was explicitly formalized in the Code of Ethics of the American Medical Association [90]. The Code of Ethics requires physicians and any helping professionals to communicate diagnoses, prognoses, courses of treatment or intervention, and alternative options in such a manner that is understood, so an informed decision regarding treatment may be made by the client/patient [90]. An individual's ability and prerogative to make one's own decision about treatment is now seen as a vital expression of autonomy and a prerequisite to participation in treatment or interventions [91].
The process of informed consent entails the explicit communication of information in order for the individual to make a decision. Western cultures value explicit information, which is centered on American consumerism, that is, the belief that having and exercising choice extends to healthcare purchases [92]. However, some cultures believe that language and information also shape reality [93]. In other words, explicit information, particularly if it is bad information, will affect the course of reality.
A signature is required on most Western informed consent forms to represent understanding and agreement on the part of the individual involved. Yet, this might be viewed as a violation of social etiquette in some cultures. For example, in Egypt, signatures are usually associated with major life events and legal matters. Therefore, requiring a signature outside these circumstances would imply a lack of trust, particularly when verbal consent has been given [94].
Consent forms also often contain technical and legal jargon that may be overwhelming to the native English-speaking individual, but can be much more daunting for immigrants who may not be proficient in English or familiar with various legal concepts. Asking for a signature on a consent form that contains foreign legal and technical terms may place some immigrants at risk for secondary traumatization, as some were persecuted, tortured, and forced to sign documents in their homelands [95].
Cultural dissonance can be a challenge to many general healthcare and mental health practitioners. Cultural experts may help mitigate this challenge by assisting with the interpretation and navigation of the complex web of cultural interactions. It is vital to keep these cultural concerns and possible barriers to understanding in mind when considering a vaccinee for participation in a pre-event vaccination program.
I was vaccinated as a child. Am I protected?
Smallpox vaccination provides high level immunity for three to five years and decreasing immunity thereafter. If a person is vaccinated again later, immunity lasts even longer. Historically, the vaccine has been effective in preventing smallpox infection in 95% of those vaccinated. In addition, the vaccine was proven to prevent or substantially lessen infection when given within a few days of exposure. It is important to note, however, that at the time when the smallpox vaccine was used to eradicate the disease, testing was not as advanced or precise as it is today [9].
I had a vaccination as a child and another when I went into the military. Am I protected?
Again, smallpox vaccination provides high level immunity for three to five years and decreasing immunity thereafter. If a person is vaccinated again later, immunity lasts even longer. If more than 10 years have passed since the last vaccination and the individual will be at risk through deployment, revaccination is recommended by the DoD [9].
What is the risk of smallpox being used in a terrorist attack?
The risk of a deliberate release of smallpox by terrorist is real; however, this risk is low, and the population at risk for such an exposure cannot be determined [96].
What would happen if there was a case of smallpox?
If there was a confirmed case of smallpox, post-event smallpox plans would be put into effect. Definitely, there would be careful case investigation to locate all the close contacts of the case, and contacts of those contacts, in order to vaccinate them. This would hopefully occur within two to four days of exposure. All contacts and their households would be vaccinated, according to the ring vaccination plan (surveillance and containment) [73].
What would happen if a contact to a smallpox case was HIV positive?
Because the risk of death from smallpox is much higher than the risk of death from the vaccine, the vaccination would be a lesser risk. There is not enough information to know whether VIG administered with the vaccine would protect a vaccinee with contraindications from an adverse event or a complication [80].
Have you been vaccinated?
There are many reasons why a physician, nurse, or healthcare professional may not wish to answer this question. One could answer that medical decisions are confidential. If the physician, nurse, or healthcare professional is asked this question by a potential vaccination candidate, it should not be answered. Any answer could influence a candidate in his/her decision about participation in the pre-event vaccination program.
Vaccinia is a live virus contained in the vaccination that protects against smallpox. Vaccinia does not contain the smallpox virus, but is an orthopoxvirus that is related to variola. Routine vaccination against smallpox was stopped in the United States in 1972 and around the world in 1980 because of the eradication of smallpox and because of the potential of fatalities from the vaccine. The last natural case of smallpox occurred in 1977 in Somalia. Before vaccination, smallpox was a dreaded disease that was fatal to 30% of the victims and left many others disfigured, blind, or deformed [26]. There is no known treatment.
The potential for smallpox to be used as a biologic weapon exists. Therefore, smallpox vaccination has been reinstituted for designated health and law enforcement personnel who may be exposed in the investigation or care of a case. At this time, vaccination for the general population is not recommended, although some military personnel have been vaccinated. ACIP has emphasized that it is critical for smallpox preparedness planning, within the context of broader terrorism and emergency response planning, to continue at the federal, state, and local levels [6].
Vaccinia produces expected reactions of fever, lymphadenopathy, malaise, and myalgia in many of the vaccinees [80,81]. Complications of autoinoculation, superimposed bacterial infections, rashes, and generalized vaccinia occur; however, these conditions either resolve spontaneously or respond to treatment. More serious life-threatening developments of eczema vaccinatum, progressive vaccinia, and post-vaccination encephalitis will have a fatal outcome in one or two persons in a million vaccinations with ACAM2000 or APSV [45,80,81].
Persons with contraindications (i.e., immunosuppression, skin conditions, allergy to components of the vaccine, cardiac disease, and pregnancy) have a higher risk of adverse events and should not be vaccinated when there is no case of smallpox. An individual who lives with or has intimate contact with anyone with these contraindications (except allergy to vaccine components) also should avoid vaccination [7].
The author wishes to thank E. Russell Alexander, MD, Professor Emeritus of epidemiology at the University of Washington School of Public Health, for his time and effort in reviewing this course. Sandra Jo Hammer, PHN, MSN, MPH, Nurse Consultant III at the Immunization Branch, California Department of Health Services, contributed immeasurably to the author's knowledge through personal contact, written materials, and train-the-trainer program for vaccinators. In addition, the staff and members of the Institute of Medicine Committee on Smallpox Vaccination Program Implementation provided many written resources and enriched the author's understanding of the implications of the vaccination program through meetings and discussions.
| Centers for Disease Control and Prevention |
| Smallpox: Bioterrorism Response Planning |
| https://www.cdc.gov/smallpox/bioterrorism-response-planning/index.html |
| Vaccine "Take" Evaluation |
| https://www.cdc.gov/smallpox/clinicians/vaccination-take-evaluation4.html |
| Vaccine Adverse Events |
| https://www.cdc.gov/smallpox/clinicians/vaccine-adverse-events5.html |
| U.S. Department of Health and Human Services |
| Vaccine Adverse Event Reporting System |
| https://vaers.hhs.gov |
| World Health Organization |
| Slide Set on the Diagnosis of Smallpox |
| http://www.who.int/entity/csr/disease/smallpox/Smallpox-English.ppt?ua=1 |
| Emergencies, Preparedness, and Response: Smallpox |
| http://www.who.int/csr/disease/smallpox/en/ |
| Emergencies, Preparedness, and Response: Preparedness in the Event of a Smallpox Outbreak |
| https://www.who.int/news-room/feature-stories/detail/preparedness-in-the-event-of-a-smallpox-outbreak |
| Global Vaccine Safety: Safety of Smallpox Vaccines |
| https://www.who.int/groups/global-advisory-committee-on-vaccine-safety/topics/smallpox-vaccines |
| Smallpox and Its Eradication (out of print) |
| http://whqlibdoc.who.int/smallpox/9241561106.pdf |
Airborne: any infectious organism that can stay suspended in air after it has been aerosolized through speaking, singing, coughing, or sneezing.
Contact, casual: contact that is sporadic and limited, as in the workplace and school.
Contact, close: contact that is continued, frequent, and involves face-to-face interchanges, as in a household or intimate situation.
Droplet: any infectious organism that is too large to be aerosolized and thus does not stay suspended in air for long periods after speaking, singing, coughing, or sneezing; less contagious than an airborne organism.
Equivocal: a vaccination reading between days 6 and 8 indicating a negative reaction (the process from vesicle to scar has not occurred); may also be called a "non-take." A person with an "equivocal" reading is not considered to have developed immunity.
Herald spots: first smallpox lesions that appear on the face, usually on the forehead.
Immunity, cell-mediated: immunity developed in the T-cells and the B-cells.
Immunity, humoral: immunity in which antibodies against a specific organism develop.
Primary vaccinee: an individual who has never been vaccinated against smallpox.
Revaccinee: an individual who has had one or more vaccinations against smallpox.
Ring vaccination: a protocol to create a double ring of immunity around any person with smallpox. The first ring would vaccinate all the household contacts of the case. The second ring would vaccinate any contacts of those vaccinated in the first ring and all their household contacts.
Take: a reading of the vaccination between days 6 and 8 indicating that the vaccination has developed the expected vesicle/pustule; indicates that immunity to smallpox has probably developed.
Umbilicate: a stage in the development of the reaction to vaccination or in the lesions of smallpox in which the pustule develops an indentation like an umbilicus.
Vaccinia: the virus used in the vaccination against smallpox. It cannot cause smallpox.
Variola: the virus that causes smallpox.
A suspected case of smallpox should be immediately reported to state and local health officials. If you have questions about your state's response plan, please call your local or state health department.
If you have questions about smallpox: Access CDC Emergency Preparedness and Response Program (https://emergency.cdc.gov)
If you have questions about smallpox vaccine:
| Contact the CDC-INFO Contact Center |
| https://wwwn.cdc.gov/dcs/ContactUs/Form |
| 800-232-4636 English and Spanish |
| 888-232-6348 TTY Users |
| National Center for Immunization and Respiratory Diseases |
| 1600 Clifton Road, Mailstop E-05 |
| Atlanta, GA 30333 |
| Email: ncirddvdinquiry@cdc.gov |
1. Frieden TR, Jaffe HW, Cono J, Richards CL, Iademarco MF. Clinical guidance for smallpox vaccine use in a postevent vaccination program. MMWR. 2015;64(RR2):1-32.
2. Centers for Disease Control and Prevention. Smallpox: Vaccines. Available at https://www.cdc.gov/smallpox/clinicians/vaccines.html. Last accessed November 19, 2021.
3. Brennan JG, Henderson DA. Diagnosis and management of smallpox. N Engl J Med. 2002;346(17):1300-1308.
4. U.S. Food and Drug Administration. ACAM2000 (Smallpox Vaccine): Questions and Answers. Available at https://www.fda.gov/vaccines-blood-biologics/vaccines/acam2000-smallpox-vaccine-questions-and-answers. Last accessed November 19, 2021.
5. Centers for Disease Control and Prevention. Notice to readers: newly licensed smallpox vaccine to replace old smallpox vaccine. MMWR. 2008;57(08):207-208.
6. U.S. Department of Health and Human Services. Strategic National Stockpile (SNS). Available at https://www.phe.gov/about/sns/Pages/default.aspx. Last accessed November 19, 2021.
7. Wharton M, Strikas RA, Harpaz R, et al. Recommendations for using smallpox vaccine in a pre-event vaccination program: supplemental recommendations of the Advisory Committee on Immunization Practices (ACIP) and the Healthcare Infection Control Practices Advisory Committee (HICPAC). MMWR. 2003;52(RR07):1-16.
8. Centers for Disease Control and Prevention. Smallpox: Who Should Get Vaccination. Available at https://www.cdc.gov/smallpox/vaccine-basics/who-gets-vaccination.html. Last accessed November 19, 2021.
9. Military Health System. Smallpox Vaccine Questions and Answers. Available athttps://www.health.mil/Military-Health-Topics/Health-Readiness/Immunization-Healthcare/Vaccine-Preventable-Diseases/Smallpox-ACAM2000/Smallpox-FAQs. Last accessed November 19, 2021.
10. Grabenstein JD, Winkenwerder W Jr. U.S. military smallpox vaccination program experience. JAMA. 2003;289(24):3278-3282.
11. Mount Sinai Hospital Department of Microbiology. Frequently Asked Questions. Available at https://eportal.mountsinai.ca/Microbiology//faq/transmission.shtml. Last accessed November 19, 2021.
12. Lane JM, Goldstein J. Evaluation of 21st-century risks of smallpox vaccination and policy options. Ann Intern Med. 2003;138(6):488-493.
13. Belongia EA, Naleway AL. Smallpox vaccine: the good, the bad, and the ugly. Clin Med Res. 2003;1(2):87-92.
14. Artenstein AW, Grabenstein JD. Smallpox vaccines for biodefense: need and feasibility. Expert Rev Vaccines. 2008;7(8):1225-1237.
15. Barquet N, Domingo P. Smallpox: the triumph over the most terrible of the ministers of death. Ann Intern Med. 1997;127(8):635-642.
16. Veenema TG, Toke J. Early detection and surveillance for biopreparedness and emerging infectious diseases. Online J Issues Nurs. 2006;11(1).
17. Riedel S. Edward Jenner and the history of smallpox and vaccination. Proc (Bayl Univ Med Cent). 2005;18(1):21-25.
18. Hays JN. Epidemics and Pandemics: Their Impacts on Human History. Santa Barbara, CA: ABC-CLIO; 2005.
19. Lucey DR, Breman JG, Henderson DA. Smallpox and bioterrorism. In: Lutwick LI, Lutwick SM (eds). Beyond Anthrax: The Weaponization of Infectious Diseases. Towata, NJ: Humana Press; 2009: 17-54.
20. Centers for Disease Control and Prevention. CDC Media Statement on Newly Discovered Smallpox Specimens. Available at https://www.cdc.gov/media/releases/2014/s0708-nih.html. Last accessed November 19, 2021.
21. Centers for Disease Control and Prevention. Vaccinia (smallpox) vaccine: recommendations of the Advisory Committee on Immunization Practices (ACIP), 2001. MMWR. 2001;50(RR10):1-25.
22. Hussain AN, Hussain F, Alam M, Cleri DJ. Smallpox. Available at https://emedicine.medscape.com/article/237229-overview. Last accessed November 19, 2021.
23. Eichner M, Dietz K. Transmission potential of smallpox: estimates based on detailed data from an outbreak. Am J Epidemiol. 2003;158(2):110-117.
24. Henderson DA, Inglesby TV, Bartlett JG, et al. Smallpox as a biological weapon: medical and public health management. JAMA. 1999;281(22):2127-2137.
25. Centers for Disease Control and Prevention. Smallpox: Transmission. Available at https://www.cdc.gov/smallpox/transmission/index.html. Last accessed November 19, 2021.
26. Dembek ZF Col, Alves DA LTC, Cieslak TJ COL, et al. USAMRIID'S Medical Management of Biological Casualties Handbook. 8th ed. Frederick, MD: U.S. Army Medical Research Institute of Infectious Diseases; 2014.
27. Centers for Disease Control and Prevention. Smallpox: Clinical Disease. Available at https://www.cdc.gov/smallpox/clinicians/clinical-disease.html. Last accessed November 19, 2021.
28. Alibek K, Handelman S. Biohazard: The Chilling True Story of the Largest Covert Biological Weapons Program in the World. New York, NY: Random House; 1999.
29. Centers for Disease Control and Prevention. Evaluating Patients for Smallpox. Available at https://www.cdc.gov/smallpox/pdfs/smallpox-diagnostic-algorithm-poster-2-pages.pdf. Last accessed November 19, 2021.
30. Centers for Disease Control and Prevention. Smallpox: Diagnosis and Evaluation. Acute, Generalized Vesicular or Pustular Rash Illness Testing Protocol in the United States. Available at https://www.cdc.gov/smallpox/clinicians/diagnosis-evaluation.html. Last accessed November 19, 2021.
31. Fenner F, Henderson DA, Arita I, Ježek Z, Ladnyi ID. Smallpox and Its Eradication. Geneva: World Health Organization; 1989.
32. Jezek Z, Hardjotanojo W, Rangaraj AG. Facial scarring after varicella: a comparison with variola major and variola minor. Am J Epidemiol. 1981;114(6):798-803.
33. Semba RD. The ocular complications of smallpox and smallpox immunization. Arch Ophthalmol. 2003;121(5):715-719.
34. Jacobs BL, Langland JO, Kibler KV, et al. Vaccinia virus vaccines: past, present and future. Antiviral Res. 2009;84(1):1-13.
35. Hamborsky J, Kroger A, Wolfe C (eds). Epidemiology and Prevention of Vaccine-Preventable Diseases (The Pink Book). 13th ed. Washington, DC: Public Health Foundation; 2015.
36. U.S. Food and Drug Administration. FDA Approves the First Drug with an Indication for Treatment of Smallpox. Available at https://www.fda.gov/news-events/press-announcements/fda-approves-first-drug-indication-treatment-smallpox. Last accessed November 19, 2021.
37. Public Health Emergency. HHS Sponsors New Formulation of World's First Approved Smallpox Drug. Available at https://www.phe.gov/Preparedness/news/Pages/tpoxx-25sept2018.aspx. Last accessed November 19, 2021.
41. Shchelkunov SN, Marennikova SS, Moyer RW. Orthopoxviruses Pathogenic for Humans. 1st ed. New York, NY: Springer Publishing Company; 2005.
42. Lau CY, Wahl B, Foo WK. Ring vaccination versus mass vaccination in event of a smallpox attack. Hawaii Med J. 2005;64(2):34-36, 53.
43. Centers for Disease Control and Prevention. Smallpox: Bioterrorism: Preparedness. Available at https://www.cdc.gov/smallpox/bioterrorism/public/preparedness.html. Last accessed November 19, 2021.
44. Bhattacharya S, Brimnes N. Simultaneously global and local: reassessing smallpox vaccination and its spread, 1789–1900. Bull Hist Med. 2009;83(1):1-16.
45. Petersen BW, Damon IK, Pertowski CA, et al. Clinical guidance for smallpox vaccine use in a postevent vaccination program. MMWR Recomm Rep. 2015;64(RR-02):1-26.
46. Taub DD, Ershler WB, Janowski M, et al. Immunity from smallpox vaccine persists for decades: a longitudinal study. Am J Med. 2008;121(12):1058-1064.
47. Gillis J. Safer Smallpox Vaccines in Works. Available at http://www.washingtonpost.com/wp-dyn/content/article/2005/11/13/AR2005111301006.html?noredirect=on. Last accessed November 19, 2021.
48. Michael LJ. Development of Newer Smallpox Vaccines. Available at http://austinpublishinggroup.com/infectious-diseases/fulltext/ajid-v3-id1025.php. Last accessed November 19, 2021.
49. Zitzmann-Roth EM, von Sonnenburg F, de la Motte S, et al. Cardiac safety of Modified Vaccinia Ankara for vaccination against smallpox in a young, healthy study population. PLoS One. 2015;10(4):e0122653.
51. U.S. Food and Drug Administration. FDA Approves First Live, Non-Replicating Vaccine to Prevent Smallpox and Monkeypox. Available at https://www.fda.gov/news-events/press-announcements/fda-approves-first-live-non-replicating-vaccine-prevent-smallpox-and-monkeypox. Last accessed November 19, 2021.
52. Frey SE, Newman FK, Kennedy JS, et al. Clinical and immunologic responses to multiple doses of IMVAMUNE (Modified Vaccinia Ankara) followed by Dryvax challenge. Vaccine. 2007;25:8562-8573.
53. Vollmar J, Arndtz N, Eckl KM, et al. Safety and immunogenicity of IMVAMUNE, a promising candidate as a third generation smallpox vaccine. Vaccine. 2006;24:2065-2070.
54. von Krempelhuber A, Vollmar J, Pokorny R, et al. A randomized, double-blind, dose-finding Phase II study to evaluate immunogenicity and safety of the third-generation smallpox vaccine candidate IMVAMUNE. Vaccine. 2010;28:1209-1216.
55. Frey SE, Winokur PL, Salata RA, et al. Safety and immunogenicity of IMVAMUNE smallpox vaccine using different strategies for a post event scenario. Vaccine. 2013;31:3025-3033.
56. Greenberg RN, Overton ET, Haas DW, et al. Safety, immunogenicity, and surrogate markers of clinical efficacy for modified vaccinia Ankara as a smallpox vaccine in HIV-infected subjects. J Infect Dis. 2013;207:749-758.
57. Walsh SR, Wilck MB, Dominguez DJ, et al. Safety and immunogenicity of modified vaccinia Ankara in hematopoietic stem cell transplant recipients: a randomized, controlled trial. J Infect Dis. 2013;207:1888-1897.
58. Center for Infectious Disease Research and Policy. Smallpox: Vaccination. Available at http://www.cidrap.umn.edu/infectious-disease-topics/smallpox#overview&1-7. Last accessed November 19, 2021.
59. Bavarian Nordic. MVA-BN Freeze-Dried. Available at https://www.bavarian-nordic.com/what-we-do/pipeline/smallpox.aspx. Last accessed November 19, 2021.
60. World Health Organization. WHO Advisory Committee on Variola Virus Research: Report of the Twenty-First Meeting. Available at https://apps.who.int/iris/bitstream/handle/10665/336343/9789240012998-eng.pdf. Last accessed November 19, 2021.
61. Centers for Disease Control and Prevention. Smallpox: Signs and Symptoms. Available at https://www.cdc.gov/smallpox/symptoms/index.html. Last accessed November 19, 2021.
62. Centers for Disease Control and Prevention. Influenza (Flu): Frequently Asked Flu Questions 2021–2022 Influenza Season. Available at https://www.cdc.gov/flu/season/faq-flu-season-2021-2022.htm. Last accessed November 19, 2021.
63. National Institute of Allergy and Infectious Diseases. Vaccine Types. Available at https://www.niaid.nih.gov/research/vaccine-types. Last accessed November 19, 2021.
64. Taber's Online. Cell-Mediated Immunity. Available at http://www.tabers.com/tabersonline. Last accessed November 19, 2021.
65. Centers for Disease Control and Prevention. Smallpox: Contraindications to Vaccination. Available at https://www.cdc.gov/smallpox/clinicians/vaccination-contraindications1.html. Last accessed November 19, 2021.
66. U.S. Food and Drug Administration. ACAM2000: Package Insert. Available at https://www.fda.gov/vaccines-blood-biologics/vaccines/acam2000. Last accessed November 19, 2021.
67. U.S. Food and Drug Administration. Cylex Inc. Immune Cell Function Assay. Available at https://www.accessdata.fda.gov/cdrh_docs/pdf10/K101911.pdf. Last accessed November 19, 2021.
68. Centers for Disease Control and Prevention. Vaccination of Persons with Primary and Secondary Immune Deficiencies. Available at https://www.cdc.gov/vaccines/pubs/pinkbook/downloads/appendices/A/immuno-table.pdf. Last accessed November 19, 2021.
69. U.S. Department of Health and Human Services. Centers for Disease Control and Prevention. Vaccine Storage and Handling Toolkit. Available at https://www.cdc.gov/vaccines/hcp/admin/storage/toolkit/storage-handling-toolkit.pdf. Last accessed November 19, 2021.
70. Lexi-Comp Online. Available at http://online.lexi.com. Last accessed November 19, 2021.
71. ACAM2000 Smallpox (Vaccinia) Vaccine, Live. Deployment Guidance: DoD. Available at https://docplayer.net/5420151-Acam2000-smallpox-vaccinia-vaccine-live-deployment-guidance-dod.html. Last accessed November 19, 2021.
72. Centers for Disease Control and Prevention. Smallpox: Packaging and Transporting Infectious Substances. Available at https://www.cdc.gov/smallpox/lab-personnel/specimen-collection/pack-transport.html. Last accessed November 19, 2021.
73. Centers for Disease Control and Prevention. Smallpox: Bioterrorism Response Planning. Available at https://www.cdc.gov/smallpox/bioterrorism-response-planning/index.html. Last accessed November 19, 2021.
74. Military Health System. Smallpox: DoD Smallpox Vaccination Program Resource Center. Available at https://www.health.mil/Military-Health-Topics/Health-Readiness/Immunization-Healthcare/Vaccine-Preventable-Diseases/Smallpox-ACAM2000. Last accessed November 19, 2021.
75. Centers for Disease Control and Prevention. Handwashing: Clean Hands Save Lives. Available at https://www.cdc.gov/handwashing. Last accessed November 19, 2021.
76. Centers for Disease Control and Prevention. When and How to Wash Your Hands. Available at https://www.cdc.gov/handwashing/when-how-handwashing.html. Last accessed November 19, 2021.
77. Centers for Disease Control and Prevention. Guideline for hand hygiene in health-care settings: recommendations of the Healthcare Infection Control Practices Advisory Committee and the HICPAC/SHEA/APIC/IDSA Hand Hygiene Task Force. MMWR. 2002;51(RR16):1-45.
78. Centers for Disease Control and Prevention. Smallpox: Expected Vaccine Reactions. Available at https://www.cdc.gov/smallpox/clinicians/vaccination-expected-reaction3.html. Last accessed November 19, 2021.
79. Centers for Disease Control and Prevention. Vaccine "Take" Evaluation. Available at https://www.cdc.gov/smallpox/clinicians/vaccination-take-evaluation4.html. Last accessed November 19, 2021.
80. Centers for Disease Control and Prevention. Smallpox: Vaccine Adverse Events. Available at https://www.cdc.gov/smallpox/clinicians/vaccine-adverse-events5.html. Last accessed November 19, 2021.
81. Fulginiti V, Henderson DA, Papier A. Smallpox Vaccination, Vaccination Method and Reactions. Version 2. Rochester, NY: Logical Images, Inc.; 2003.
82. Cono J, Casey CG, Bell DM. Smallpox vaccination and adverse reactions: guidance for clinicians. MMWR. 2003;52(RR4):1-28.
83. Centers for Disease Control and Prevention. Medical Management of Adverse Reactions. Available at https://www.cdc.gov/smallpox/clinicians/vaccine-medical-management6.html. Last accessed November 19, 2021.
84. Centers for Disease Control and Prevention. Smallpox: Side Effects of Smallpox Vaccination. Available at https://www.cdc.gov/smallpox/vaccine-basics/vaccination-effects.html. Last accessed November 19, 2021.
85. Vaccine Adverse Event Reporting System. Available at https://vaers.hhs.gov/index.html. Last accessed November 19, 2021.
86. National Eczema Association. Eczema Facts. Available at https://nationaleczema.org/research/eczema-facts. Last accessed November 19, 2021.
87. Centers for Disease Control and Prevention. Vaccinia Immune Globulin Intravenous (Human) (VIGIV). Available at https://www.fda.gov/downloads/BiologicsBloodVaccines/BloodBloodProducts/ApprovedProducts/LicensedProductsBLAs/FractionatedPlasmaProducts/UCM179514.pdf. Last accessed November 19, 2021.
88. Damon IK, Damaso CR, McFadden G. Are we there yet? The smallpox research agenda using variola virus. PLoS Pathog. 2014;10(5):e1004108.
89. Centers for Disease Control and Prevention. Vaccine Administration: Step-by-Step Instructions. Available at https://www.cdc.gov/smallpox/clinicians/vaccination-administration2.html. Last accessed November 19, 2021.
90. McLaughlin LA, Braun KL. Asian and Pacific Islander cultural values: considerations for health care decision making. Health Soc Work. 1998;23(2):116-126.
91. Ivashkov Y, Van Norman GA. Informed consent and the ethical management of the older patient. Anesthesiol Clin. 2009;27(3): 569-580.
93. Carrese JA, Rhodes LA. Western bioethics on the Navajo reservation: benefit or harm? JAMA. 1995;274(10):826-829.
94. Rashad AM. Obtaining informed consent in an Egyptian research study. Nurs Ethics. 2004;11(4):394-399.
95. Yick AG, Berthold SM. Conducting research on violence in Asian American communities: methodological issues. Violence Vict. 2005;20(6):661-677.
96. Centers for Disease Control and Prevention. CDC Interim Guidance for Revaccination of Eligible Persons who Participated in the U.S. Civilian Smallpox Preparedness and Response Program. Available at https://www.cdc.gov/smallpox/pdfs/revaccination-memo.pdf. Last accessed November 19, 2021.
97. Centers for Disease Control and Prevention. About Monkeypox. Available at https://www.cdc.gov/poxvirus/monkeypox/about.html. Last accessed June 2, 2022.
98. Centers for Disease Control and Prevention. U.S. Monkeypox 2022: Situation Summary. Available at https://www.cdc.gov/poxvirus/monkeypox/response/2022/index.html. Last accessed July 18, 2022.
99. World Health Organization. Multi-Country Monkeypox Outbreak in Non-Endemic Countries. Available at https://www.who.int/emergencies/disease-outbreak-news/item/2022-DON385. Last accessed June 2, 2022.
100. Centers for Disease Control and Prevention. Monkeypox: Clinical Recognition. Available at https://www.cdc.gov/poxvirus/monkeypox/clinicians/clinical-recognition.html. Last accessed June 2, 2022.
Mention of commercial products does not indicate endorsement.