Polypharmacy in geriatrics is a serious problem that is expected to grow in scope as the population ages. Certain medications commonly used in the younger population may be considered inappropriate in older adults, due to pharmacodynamics, pharmacokinetics, and/or drug/disease interactions. Reduction in the number of medications that older patients take is associated with reduction in mortality rates, improved quality of life, and reduced costs. This program will educate clinicians on how to evaluate and reduce unnecessary polypharmacy.
- INTRODUCTION
- POLYPHARMACY DEFINED
- IDENTIFICATION OF PROBLEMATIC MEDICATIONS IN OLDER ADULT PATIENTS
- AGE-RELATED PHYSIOLOGIC CHANGES
- POTENTIAL PROBLEMS PRESCRIBING IN GERIATRICS
- ADVERSE DRUG REACTIONS
- PRESCRIBING CASCADE
- TRANSITIONS OF CARE
- MEDICATION RECONCILIATION
- CONCLUSION
- Works Cited
- Evidence-Based Practice Recommendations Citations
This course is designed for advanced practice nurses, nurses, pharmacists, pharmacy technicians, and allied healthcare professionals who work with the geriatric population.
The purpose of this course is to provide clinicians with the knowledge necessary to ensure that geriatric patients are effectively treated while reducing unnecessary polypharmacy.
Upon completion of this course, you should be able to:
- Define polypharmacy in the older adult patient.
- Identify guidelines to prevent the use of unnecessary medications in older adult patients.
- Describe physiologic changes of aging that cause differences in drug metabolism
- Discuss the potential problems polypharmacy may cause in older patients.
- Evaluate the impact of polypharmacy in specific conditions as well as approaches to avoid inappropriate prescribing.
- Describe a prescribing cascade and the importance of medication assessment and reconciliation.
Susan Waterbury, MSN, FNP-BC, ACHPN, entered the medical field in 1985 as a certified medical assistant and basic x-ray operator. She achieved her RN in 1990 and practiced in a variety of settings, including hospital, home health care, and hospice. Ms. Waterbury achieved her BSN in 1996 and her MSN as a Family Nurse Practitioner in 1999. She was board-certified as an FNP-BC in 2000 and has practiced in family practice, geriatrics, corporate leadership, hospice, and palliative care settings. She holds RN and NP licenses in Florida and Arizona.
In addition to her clinical roles, Ms. Waterbury continues to play an active role in educating and mentoring nurses and healthcare professionals. She has been a faculty member of the University of Phoenix since 2015, teaching in the nurse practitioner and MSN programs. She develops and presents educational programs for a variety of healthcare organizations and community groups.
Contributing faculty, Susan Waterbury, MSN, FNP-BC, ACHPN, has disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
Mary Franks, MSN, APRN, FNP-C
Randall L. Allen, PharmD
The division planners have disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
Sarah Campbell
The Director of Development and Academic Affairs has disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
The purpose of NetCE is to provide challenging curricula to assist healthcare professionals to raise their levels of expertise while fulfilling their continuing education requirements, thereby improving the quality of healthcare.
Our contributing faculty members have taken care to ensure that the information and recommendations are accurate and compatible with the standards generally accepted at the time of publication. The publisher disclaims any liability, loss or damage incurred as a consequence, directly or indirectly, of the use and application of any of the contents. Participants are cautioned about the potential risk of using limited knowledge when integrating new techniques into practice.
It is the policy of NetCE not to accept commercial support. Furthermore, commercial interests are prohibited from distributing or providing access to this activity to learners.
Supported browsers for Windows include Microsoft Internet Explorer 9.0 and up, Mozilla Firefox 3.0 and up, Opera 9.0 and up, and Google Chrome. Supported browsers for Macintosh include Safari, Mozilla Firefox 3.0 and up, Opera 9.0 and up, and Google Chrome. Other operating systems and browsers that include complete implementations of ECMAScript edition 3 and CSS 2.0 may work, but are not supported. Supported browsers must utilize the TLS encryption protocol v1.1 or v1.2 in order to connect to pages that require a secured HTTPS connection. TLS v1.0 is not supported.
The role of implicit biases on healthcare outcomes has become a concern, as there is some evidence that implicit biases contribute to health disparities, professionals' attitudes toward and interactions with patients, quality of care, diagnoses, and treatment decisions. This may produce differences in help-seeking, diagnoses, and ultimately treatments and interventions. Implicit biases may also unwittingly produce professional behaviors, attitudes, and interactions that reduce patients' trust and comfort with their provider, leading to earlier termination of visits and/or reduced adherence and follow-up. Disadvantaged groups are marginalized in the healthcare system and vulnerable on multiple levels; health professionals' implicit biases can further exacerbate these existing disadvantages.
Interventions or strategies designed to reduce implicit bias may be categorized as change-based or control-based. Change-based interventions focus on reducing or changing cognitive associations underlying implicit biases. These interventions might include challenging stereotypes. Conversely, control-based interventions involve reducing the effects of the implicit bias on the individual's behaviors. These strategies include increasing awareness of biased thoughts and responses. The two types of interventions are not mutually exclusive and may be used synergistically.
#99023: Geriatric Polypharmacy
The older adult population of the United States is projected to grow substantially over the coming decades, comprising 24.3% of the total population by 2060 [1]. The population of those 85 years of age and older (referred to as the oldest-old) is expected to increase from 7 million in 2025 to 17.5 million by 2060 [1]. As a result, the number of older adults with multiple chronic illnesses requiring drug therapy will also increase significantly. The presence of chronic illness is a predictor of greater utilization of healthcare services, including nursing facility residence, and greater number of medications. Older patients and residents of nursing facilities may have more than 20 daily medications prescribed, and polypharmacy in the older adult population is a serious problem that healthcare providers can evaluate and address to improve outcomes.
Hazards of polypharmacy include lack of adherence, overtreatment, adverse drug reactions, and incorrect dosage and administration regimens. Older patients may receive health care from a diverse set of providers, specialists, and consultants—often, one for each disease process. Each provider may prescribe additional medication(s), and as the number of medications increases, so does the risk of adverse drug reactions. In some cases, there is confusion regarding the proprietary (brand) and generic names of medications, with some patients taking a generic and brand version of the same drug. The increasing complexity of medication regimens leaves patients vulnerable to the hazards of polypharmacy and adverse drug reactions.
There is an increased demand for healthcare providers who are knowledgeable in geriatric prescribing practices. Specialized prescribing practices, geared toward managing multimorbidity in older patients, are mandated by best practices and standards for geriatric care. Confusion about medication regimens, duplicate medications, non-adherence, and errors in self-administration make older adults at high risk for problems. In many cases, the "brown bag" approach is used to assess all the prescription and non-prescription medications the patient takes. This involves the patient bringing all prescription and over-the-counter medications he or she takes to each office visit. The patient/caregiver can then describe how the medications are taken/given and any reminder system or assistive device used (such as a medicine box/planner) to ensure compliance. Patient education and communication with primary care providers are key components of comprehensive care. Patients should understand that over-the-counter drugs, supplements, and home remedies are not inherently "safe" and have the potential for serious adverse effects or interactions.
Prescribers should also be aware of financial constraints experienced by many older adults, some of whom may be faced with the decision to buy either food or medications. In an effort to cut costs, these patients may decide to take a daily medication every other day or cut pills in half. Some may choose to take one medication over another based on their personal priorities (e.g., stopping an antihypertensive but continuing a hypnotic because they place a higher priority on getting good sleep).
Geriatric practitioners should have a comprehensive understanding of the physiologic changes of aging that occur, particularly in the liver and kidneys. Physiologic changes of aging cause differences in the absorption, distribution, metabolism, and excretion of medications. This increases the risk for adverse drug reactions or interactions due to excess drug accumulation and/or reduced clearance.
Today, older healthcare consumers or their representatives are often informed and savvy and may request every treatment available. They may demand a treatment a neighbor or relative was prescribed, or one that was advertised on television. Some patients may fall prey to online scams to purchase fraudulent products or supplements.
A formal drug re-evaluation should be performed regularly for all older patients. Providers should continuously assess prescribed drug therapies for necessity and appropriateness according to the patient's goals of care and best evidence-based geriatric practices. Multiple medications may be necessary to treat comorbid conditions, but vigilance is required to monitor for adverse reactions or interactions.
Healthcare costs associated with the improper and unnecessary use of medications exceeds $200 billion annually, according to estimates from the IMS Institute for Healthcare Informatics [2]. These costs are related to adverse drug reactions or interactions, hospital admissions, emergency department visits, and outpatient care.
Reducing the number of medications older patients take is associated with reduction in mortality rates, improved quality of life, and reduced costs [3,47]. Medication regimen simplification is a priority to improve patient outcomes and reduce errors.
The term polypharmacy is often used but not well defined. There are varied definitions in medical literature, but in general, polypharmacy has been defined as a single patient taking more than 5 drugs every day, with excessive polypharmacy defined as the prescription of 10 or more daily medications [3,48]. Polypharmacy may be used to describe excessive or unnecessary medications, inappropriate prescribing, or excessive use, overuse, or duplication of medications. There has been a call to redefine polypharmacy beyond an arbitrary number of medications [14,48]. In some cases of multimorbidity and chronic conditions (e.g., hypertension), the use of multiple medications may be the best practice according to clinical guidelines; this may be referred to as "appropriate polypharmacy" [15]. However, even when the prescription of multiple medications is warranted, it raises the risks of drug interactions, compliance issues, and adverse effects. Generally, the term polypharmacy has a negative connotation and is associated with the co-prescribing of potentially inappropriate medications.
According to the American Geriatric Society (AGS), more than 90% of older people use at least one prescription, and more than 66% use three or more per month [4]. Care by a geriatrician and clinical pharmacist intervention have been found to decrease potentially inappropriate prescribing practices for these patients [4,6]. Several tools have also been developed to help prescribers make the best selection of agents and minimize the risks of problematic polypharmacy in older patients.
In older adults, certain drugs are considered inappropriate when the adverse pharmacodynamics, pharmacokinetics, and/or risk of drug interactions outweigh the potential benefits. In 1991, Mark H. Beers, MD, and his colleagues established a list of medications considered potentially inappropriate for patients 65 years of age or older. Known as the AGS Beers criteria (Table 1), the resource was designed to educate prescribers, improve prescribing practices, and enhance quality assurance. Prescribers should review the medication regimen and determine the medical necessity of each drug the patient takes. The Beers criteria list continues to be updated (most recently in 2023) and used to guide and evaluate prescribing practices in geriatric patients. Notable new inclusions to the 2023 update of the list are warfarin, rivaroxaban, and sulfonylureas (expanded to include all the sulfonylureas) [4].
EXAMPLES FROM THE 2023 AMERICAN GERIATRICS SOCIETY BEERS CRITERIA FOR POTENTIALLY INAPPROPRIATE MEDICATION USE IN OLDER ADULTS
| Therapeutic Category (Drugs) | Rationale | Recommendation | Quality of Evidence | Strength of Recommendation | ||
|---|---|---|---|---|---|---|
| First-generation antihistamines (brompheniramine, carbinoxamine, chlorpheniramine, clemastine, cyproheptadine, dexbrompheniramine, dexchlorpheniramine, dimenhydrinate, diphenhydramine [oral], doxylamine, hydroxyzine, meclizine, promethazine, pyrilamine, triprolidine) |
| Avoid | Moderate | Strong | ||
| Anti-infective (nitrofurantoin) | Potential for pulmonary toxicity, hepatoxicity, and peripheral neuropathy, especially with long-term use; safer alternatives available | Avoid in individuals with creatinine clearance <30 mL/min or for long-term suppression of bacteria | Low | Strong | ||
| Antiarrhythmic (digoxin) | Use in atrial fibrillation: Should not be used as a first-line agent in atrial fibrillation, because more effective alternatives exist and it may be associated with increased mortality | Avoid as first-line therapy for atrial fibrillation | Low | Strong | ||
| Use in heart failure: Questionable effects on risk of hospitalization and may be associated with increased mortality in older adults with heart failure; higher dosages not associated with additional benefit and may increase risk of toxicity | Avoid as first-line therapy for heart failure | Low | Strong | |||
| Decreased renal clearance of digoxin may lead to increased risk of toxic effects; further dose reduction may be necessary in patients with stage 4 or 5 chronic kidney disease | If used for atrial fibrillation or heart failure, avoid dosages >0.125 mg/day | Moderate | Strong | |||
| Antipsychotics (first- [conventional] and second- [atypical] generation) | Increased risk of cerebrovascular accident (stroke) and greater rate of cognitive decline and mortality in persons with dementia. Avoid antipsychotics for behavioral problems of dementia or delirium unless nonpharmacologic options (e.g., behavioral interventions) have failed or are not possible and the older adult is threatening substantial harm to self or others. | Avoid, except for schizophrenia, bipolar disorder, or short-term use as antiemetic during chemotherapy | Moderate | Strong | ||
| Antidepressants, alone or in combination (amitriptyline, amoxapine, clomipramine, desipramine, doxepin >6 mg/day, imipramine, nortriptyline, paroxetine, protriptyline, trimipramine) | Highly anticholinergic, sedating, and cause orthostatic hypotension; safety profile of low-dose doxepin (≤6 mg/day) comparable with that of placebo | Avoid | High | Strong | ||
| Insulin, sliding scale | Higher risk of hypoglycemia without improvement in hyperglycemia management regardless of care setting; refers to sole use of short- or rapid-acting insulins to manage or avoid hyperglycemia in absence of basal or long-acting insulin; does not apply to titration of basal insulin or use of additional short- or rapid-acting insulin in conjunction with scheduled insulin (i.e., correction insulin) | Avoid | Moderate | Strong | ||
| Proton-pump inhibitors | Risk of Clostridioides difficile infection and bone loss and fractures; risk of pneumonia and gastrointestinal malignancies | Avoid scheduled use for >8 weeks unless for high-risk patients (e.g., oral corticosteroids or chronic NSAID use), erosive esophagitis, Barrett esophagitis, pathologic hypersecretory condition, or demonstrated need for maintenance treatment (e.g., due to failure of drug discontinuation trial or H2 blockers) | High | Strong |
The Beers list separates potentially inappropriate medications into several categories according to the strength of the recommendation and the potential adverse event. The first category includes drugs that are "potentially inappropriate" due to a higher risk of adverse effects and/or reduced efficacy in older patients; the AGS recommends that prescribers consider avoiding these agents [4]. The second category is for medications used in the treatment of common health problems (e.g., heart failure, seizures) that may exacerbate comorbidities in older patients. The third category is for potentially inappropriate medications that should be used with caution (and perhaps increased monitoring) in older adult patients [4]. Finally, the AGS provides a list of medications with potentially clinically important non-anti-infective drug-drug interactions that should be avoided in older adults or should be decreased in dose in those with impaired kidney function. Careful consideration should be given when prescribing medications that are on the Beers list. The criteria are meant to support good clinical judgment [46]. If the listed medications are used, they should be prescribed at the lowest effective dose for the shortest duration possible.
The Screening Tool of Older Persons' potentially inappropriate Prescriptions (STOPP) criteria focus on the avoidance of potentially inappropriate prescribing in older adult patients experiencing acute illness [5]. This tool places a special focus on potential drug-drug interactions, duplicate drug class prescriptions, and techniques to minimize adverse drug reactions. Criteria are organized according to physiologic system, which may make the tool easier to use. STOPP is designed to be used in conjunction with the Screening Tool to Alert doctors to the Right Treatment (START) criteria, which provides guidance on the medications that are recommended for older patients with specific conditions/diseases (e.g., arthritis, depression) [18]. STOPP/START criteria were first published in 2008 and were updated in 2014, with a 31% increase in the number of criteria [49]. Several new categories were included in the 2014 update, including STOPP categories of antiplatelet/anticoagulant drugs; drugs affecting, or affected by, renal function; and drugs that increase anticholinergic burden. New START categories include urogenital system drugs, analgesics, and vaccines [49].
A 2016 study comparing the effectiveness of the original STOPP and the 2003 and 2012 Beers criteria in identifying potential adverse drug events (including hospitalizations and emergency department visits) found that STOPP was slightly more specific (though less sensitive) than either Beers criteria [41]. The authors of this study recommended using the Beers and STOPP tools together for the best predictive value.
Patient A is 82 years of age with a history of congestive heart failure, glaucoma, hypertension, and osteoarthritis. Her current medications are furosemide, potassium, lisinopril, metoprolol, aspirin, timolol maleate opthamic solution (Timoptic), acetaminophen (as needed), multivitamin, and a calcium/vitamin D supplement (800 IU daily). She has an appointment with a new orthopedic physician. During the appointment, the patient complains of persistent arthritic pain in her knee. The physician prescribes the nonsteroidal anti-inflammatory drug (NSAID) meloxicam (7.5 mg per day) for pain and inflammation.
Comments and Discussion : From the orthopedic standpoint, prescription of meloxicam is good practice, as it should help to ameliorate patient A's symptoms. However, from a cardiac standpoint, this is a risky approach due to the potential side effect of fluid retention and its effect on the heart. In general, NSAIDs can be dangerous for an individual of Patient A's age. NSAIDs (including meloxicam, but also over-the-counter options like ibuprofen) have been issued "black box" warnings by the U.S. Food and Drug Administration (FDA) for the increased risk of:
Serious and potentially fatal cardiovascular and thrombotic events, including myocardial infarction and stroke
Serious adverse gastrointestinal events such as bleeding, ulcer, and intestinal perforation (higher in older patients)
Patient A has a good working relationship with her primary care provider, who has instructed her to contact him regarding any changes in her medication regimen. She calls her physician prior to taking the medication, and he advises her not to take the NSAID. Instead, he devises a pain management plan that minimizes the potential risks. Previously, Patient A was taking acetaminophen as needed, averaging up to one dose daily. This is increased to twice daily extended-release acetaminophen (650 mg). For breakthrough pain, tramadol 25 mg every four hours (as needed) is prescribed. Another option considered was the topical anti-inflammatory diclofenac sodium 1% topical gel, which would have fewer side effects than systemic agents. Aside from pharmacotherapy, the patient is scheduled with a physical therapist to create a safe exercise plan, including strengthening and range-of-motion exercises.
Aging is a complex process with variable effects resulting from a combination of heredity, the environment, comorbidity, diet, exercise, and culture. Although the rate by which one ages is unique, there are universal changes that occur in the body as one gets older, and these changes can affect the pharmacokinetics and pharmacodynamics of prescribed and over-the-counter medications.
Pharmacokinetic processes altered in aging include absorption, first-pass metabolism, bioavailability, distribution, protein binding, and renal/hepatic clearance. Age-related physiologic factors that may affect the absorption of drugs include delayed emptying time of the stomach, altered pH of the stomach contents, and slowed gastrointestinal tract motility. Changes in these processes begin with alterations in the functioning of individual cells.
In older patients, cells become less efficient at performing necessary functions. This may be the result of atrophy, hypertrophy, hyperplasia, dysplasia, and neoplasia. Atrophy is the shrinkage of cells and is most commonly noted in the brain, heart, skeletal muscle, and reproductive organs. It may result in dementia or contracture. In some cases, cells enlarge due to an increase of proteins in cell structures; this is referred to as hypertrophy. It has been hypothesized that this change may be a compensatory mechanism in response to atrophied cells. Hypertrophic changes in older patients may result in cardiomegaly or benign prostatic hypertrophy. Similarly, older patients are at increased risk for hyperplasia, or the increased reproduction of cells. This overgrowth of cells may lead to the development of benign growths (e.g., focal nodular hyperplasia, sebaceous hyperplasia) or it may be a precursor to cancer. Dysplasia is also more common in older adults. This is characterized by mature cells becoming disorganized and abnormal in size and/or shape. This is often a first stage in the development of neoplasia, or the new growth of benign or cancerous tumors.
These changes at the cellular level can affect the overall systemic metabolism of medications and/or may result in changes in the organs involved in metabolizing and eliminating medications. Specifically, the liver, kidneys, and gastrointestinal tract may reflect cellular changes through loss of function.
The functioning of the liver plays a significant role in the metabolism of drugs. The liver is the major site of drug transformation and elimination, and drugs administered by the oral route must pass through the liver prior to reaching systemic circulation. The enzyme cytochrome P-450 (CYP-450) is the most important factor in the first phase of drug metabolism, and this enzyme is primarily expressed in the liver. However, the liver's capacity to metabolize medications (and toxins) with CYP-450 is reduced by at least 30% in older patients [19]. In addition, the liver undergoes structural and microscopic changes with aging (e.g., reduced blood flow), and liver damage is repaired more slowly. As such, the metabolism of substances by the liver decreases, causing reduced inactivation of medications. This places older adults at increased risk for side effects due to reduced clearance of medications, which can be potentiated by the presence of hepatic disease. In many cases, older patients require reduced doses to adjust for these changes.
Age- and illness-related declines in kidney function necessitate dosage adjustments and possibly avoidance of certain medications. Renal elimination is a common form of drug excretion, and the rate by which medications are cleared lengthens with age. This can extend the action of drugs that undergo renal elimination, including morphine, heparin, lisinopril, and furosemide. Even with kidney changes, decreased muscle mass and limited physical activity can maintain serum creatinine levels within normal limits [19]. This can be misinterpreted as a sign of normal kidney function despite deficits. As such, caution is required when prescribing to older adult patients even when kidney function appears normal.
Prescribing to geriatric patients can be complex, and there are a number of factors that should be considered every time a new medication is considered. The prescribing problems that arise in this patient population may be generally categorized as problems of selection, interaction, or dosage. Many of these issues can be avoided if extra care is taken to evaluate the patient and his or her medical and medication history and provide better patient/caregiver education on adherence to prescription medication regimens. Issues specific to geriatric patients that should be included in the decision to prescribe a medication include the presence of comorbidities, life expectancy, redundancies, and the likelihood of compliance.
Older adults often have multiple conditions, the treatment of which may call for several medications. This potentially necessary polypharmacy can quickly escalate to be a burden on patients and/or their caregivers. The use of multiple medications has been associated with decreased adherence due to cost, complexity of regimens, and burnout. If possible, steps should be taken to limit the number of medications to those most effective for the patient. If a single medication may be of benefit for more than one of a patient's conditions, it should be preferred over multiple agents. Selecting the appropriate medication(s) for geriatric patients is made even more complicated by the lack of clinical guidelines and completed research focusing on this population.
Due to age-related physiologic changes, older adult patients may be both more likely to experience adverse effects and less likely to derive a therapeutic benefit from a medication. This is compounded in patients with limited life expectancies. Life expectancy may be shorter than the time required to derive a benefit from a particular medication or treatment. The success of preventive treatment generally requires longer duration of treatment, and for patients with life-limiting conditions, preventive approaches may have little benefit. If the time to benefit from a certain drug is longer than the patient is expected to live, it should be avoided. Maintenance of quality of life and functional status become prime objectives.
Healthcare providers should perform a comprehensive review of patients' medication regimens, identifying any potential interactions or redundancies. Simplification is a priority to improve outcomes and reduce errors. If errors or inconsistencies are identified, further steps should be taken to avoid adverse reactions. Tapering or reducing medications may be indicated if a patient's condition has improved, stabilized, or resolved. The potential risks of pharmacotherapy may not be fully understood by patients and/or caregivers, so they may be reluctant to avoid or stop medications. Improved patient education is indicated in these cases.
With the development of new options for life-prolonging treatments and drugs comes a potential for an increased burden of treatment as well. Burdens of treatment include adverse drug reactions and/or interactions, overtreatment, non-adherence, medication errors, and cost. Focusing on quality-of-life issues, functional status, and preferences may be a better approach than aggressive attempts to reach target goals.
Evaluating and treating patients with polypharmacy requires a good working relationship and communication. Patients and caregivers require education about over-the-counter medications and supplements that may be dangerous and should be avoided. Some patients and their caregivers may require adaptive approaches to understand and adhere to the prescribed regimen. This includes ensuring that patients who use assistive devices (e.g., glasses, hearing aids) are using these tools. When appropriate, alternative communication approaches (e.g., large-print text, an interpreter) may be used. Education regarding medications should be provided in a quiet environment, free of distraction. Teaching may include repeated instructions in a stepwise fashion, with visual reinforcements and a return demonstration for functional tasks.
More than 3 million Americans receive care in skilled nursing facilities each year, and 1.3 million Americans reside in nursing homes [7]. The Centers for Medicare and Medicaid Services (CMS) work to promote nursing home quality improvement, address reimbursement issues, and ensure compliance with CMS-defined best practices [12,50]. Nursing facilities are required to comply with CMS regulatory requirements in order to receive payments under the Medicare or Medicaid programs.
CMS regulations state that nursing facility residents should only receive medications when the potential benefits outweigh the risks or burden of treatment [12,50]. There must be a clear clinical indication and diagnosis for any medication, and prescribed medications should be given for the proper duration at the correct dose. State surveyors review patients' medication regimens to assess for unnecessary medications. If a patient is found to have been prescribed an unnecessary drug, the facility may receive a citation (referred to as F-Tag 757 or F757) for violating the CMS requirement to avoid unnecessary medications. In 2014, CMS issued a memo indicating that surveyors have been trained to increase investigations for unnecessary drugs, particularly antipsychotic medications in the management of dementia [20]. In 2017, F-tag 758 (F758) was issued as a unique survey item to ensure the patient's drug regimen is free from unnecessary psychotropic medications [52]. These regulations are intended to help promote or maintain the patient's highest functional, emotional, and physical level of wellness. To comply with F757 and F758, each patient's drug regimen should be monitored on a regular basis, with goals of treatment identified. Only drugs that are medically necessary should be administered in the correct dosages and for only the clinically indicated duration. After a medication has been ordered, the patient should be monitored for therapeutic response, adverse reactions, interactions, and necessity of ongoing treatment. Any significant decline in functional or physical status should be immediately correlated with any new drugs or changes in drug dosages. Significant declines in status should be recognized and evaluated, with adjustment of the medication regimen if warranted.
Patient B is a man, 78 years of age, who resides in a nursing facility. One year ago, he fell and fractured his left hip and underwent surgical repair. He returned to the nursing facility, completed rehabilitation, and regained most of his prior function. After the surgery, Patient B was prescribed warfarin to prevent deep vein thrombosis (DVT) after surgery.
During a routine survey, a state surveyor discovers that Patient B is still being administered warfarin. After further investigation, it is discovered that the warfarin was never discontinued after the appropriate duration after the hip fracture repair. The surveyor considers warfarin an unnecessary drug, and a citation (F757) is issued. After contacting the attending physician, the warfarin is promptly discontinued.
Comments and Discussion : Patient B's case is an example of using the right drug but not using it for the correct duration. After orthopedic surgery, warfarin is usually indicated for approximately two to three months or until activity/ambulation has increased to a point that the risk of DVT is reduced. There is a substantial burden of treatment with warfarin, including weekly evaluations of prothrombin time/international normalized ratio (PT/INR), adverse reactions, interactions, and increased risk of bleeding and brain hemorrhage, especially for patients with a history of falls.
There is shared responsibility for this error between the prescriber/healthcare provider and the facility. The provider did not follow through and discontinue the medication when it was no longer needed, and the facility nursing staff should have realized that the drug was no longer necessary and approached the provider for an order to discontinue. The nursing facility could have called the orthopedic physician for orders and duration of warfarin treatment after surgery. When a medication is started, the stop date for that medication should be considered and established. The consultant pharmacist could have intervened as well.
Adverse drug reactions (ADRs) represent a significant economic burden to the healthcare system, causing nearly 600,000 emergency department visits annually for older adults [9]. Most hospitalizations among older adults are due to poor monitoring of anticoagulants, diabetes medications, anticonvulsants, and opioids [9].
An ADR is a symptom, consequence, or injury that occurs due to the administration of a medication, causing a noxious, unintended, and/or undesired effect. A reaction is categorized as an ADR if it occurs at normal human doses for prophylaxis, diagnosis, and/or treatment. It may be a secondary effect of a drug that is undesirable and different from the therapeutic effect or may cause a functional decline or impairment in mental or physical functioning.
A retrospective study analyzing the costs and incidence of emergency department visits related to ADRs in patients 65 years of age or older identified the following risk factors [10]:
Newly prescribed drugs
Use of multiple pharmacies
Multiple medications
Recent hospitalization/ER visit
Female gender
Comorbidity
Residence in a nursing facility
The subjects in the study had been prescribed an average of 12.9 medications in the year preceding emergency department admission [10].
A review of the National Mortality Statistics database for deaths due to adverse drug effects during clinical use found an increased incidence of death from ADRs in persons older than 55 years of age, with the highest risk occurring after 75 years of age [11]. Anticoagulants, opioids, and immunosuppressants were most commonly associated with an adverse effect resulting in death [11].
There are five major categories of ADRs: side effects, hypersensitivity, idiosyncratic response, toxic reactions, and adverse drug interactions. Side effects are secondary effects of a drug and may be dose related. Hypersensitivity to a drug is immunologically mediated. In severe cases, anaphylaxis may occur. An idiosyncratic response is an unusual or unexpected reaction. Toxic reactions are often related to the dose or duration of drug therapy. Less often, build-up of metabolites may precipitate a toxic reaction, as with digoxin or phenytoin toxicity.
The impact another drug or substance has on a medication is referred to as a drug-drug interaction. The interaction may alter drug metabolism, absorption, elimination, distribution, and/or pharmacokinetics. The intended effects of medications may be increased or decreased secondary to the interaction. In addition, undesired drug effects (e.g., exacerbation of a disease or condition) may develop in patients with certain disease states. For example, nonselective beta-blockers may induce bronchospasm in patients with asthma. These drug-disease interactions may be more common in older patients with multiple comorbidities.
Common ADRs include hypoglycemia, candidiasis, allergic reaction, gastrointestinal complications, hypotension, dysrhythmia, severe headache, dizziness, acute renal failure, and respiratory complications. Each patient has the potential for a unique reaction to a medication and may be genetically predisposed to either have an increased or decreased reaction to a drug.
Specific therapies are associated with the increased potential for ADRs in older patient, even with adherence to treatment guidelines for the general adult population. Improved management of several drug classes, including antithrombotic and antidiabetic drugs, has been recommended to prevent ADRs in older adults [9]. Heparin, aspirin, warfarin, and clopidogrel have all been associated with adverse bleeding events, including brain hemorrhage, hematuria, gastrointestinal bleeding, and hematoma [9]. Diuretics (e.g., furosemide, hydrochlorothiazide) may cause hyponatremia, azotemia, and falls in older patients. Decreased oral intake of food and fluids during illness may cause dehydration when combined with a diuretic. Urinary urgency and incontinence may cause older patients to engage in risky behavior to get to the bathroom, despite caregivers' instructions to call for assistance. During times of illness, these medications may need to be held or dosages adjusted to respond to the patient's change in condition.
Adverse reactions that lead to delirium or altered mental status most commonly occur after use of medications with sedation and antihistaminic effects. Confusion and delirium in older adult patients may occur secondary to opioid analgesics (e.g., morphine, oxycodone), anticonvulsants (e.g., phenytoin, valproic acid), sedatives (e.g., diazepam, alprazolam), hypnotics (e.g., temazepam, zolpidem), and antipsychotics (e.g., haloperidol, quetiapine).
As noted, adverse events associated with warfarin is one of the leading causes of emergency department visits and hospitalizations among older adults [9]. As such, improved management of anticoagulants has the potential to significantly reduce morbidity and mortality in this population. First used in the 1950s, warfarin is considered the criterion standard for anticoagulation and is widely used to prevent and treat thrombosis and thromboembolism. Other indications for warfarin include atrial fibrillation, artificial heart valve, and pulmonary embolism. Dosing of warfarin is individualized and closely monitored by the INR, which measures serum viscosity. In general, INR should be between 2 and 3 in patients taking warfarin. For high-risk patients or those with prosthetic valves, the desired range may be 2.5–3.5, and older adults who are at high risk for complications may be maintained at 1.8–2.
The FDA has issued a black box warning for warfarin regarding the risk of major or fatal bleeding. This risk is increased in patients older than 65 years of age with high-intensity coagulation (i.e., INR >4), variable INR, and/or other comorbidities [45]. Frequent INR monitoring is recommended along with careful dosage adjustment. Warfarin is contraindicated in patients with active bleeding, gastrointestinal bleeding, hemorrhagic stroke, blood dyscrasias, recent surgery, a high risk for non-compliance, and moderate-to-severe hepatic impairment. Caution is recommended in patients older than 65 years of age and in patients with a history of falls due to the risk of subdural hematoma and severe or fatal bleeding.
Aside from the risk of serious bleeding, common adverse reactions to warfarin include bruising, abdominal pain, nausea, vomiting, fatigue, headache, dizziness, taste changes, dermatitis, and fever. Over-the-counter medications can also interact with anticoagulants. The anticoagulant effects of warfarin and the antiplatelet effects of clopidogrel can be enhanced by the simultaneous use of ibuprofen, with the potential for internal bleeding. While the adverse interaction of warfarin with aspirin and NSAIDs is common knowledge, the significant potential interaction between warfarin and acetaminophen is less well known. Liver metabolism, drug and food interactions, and genetic factors also affect the efficacy of warfarin. Vitamin K-rich foods, such as green leafy vegetables and soy, decrease the effect of warfarin, reducing the PT/INR to subtherapeutic levels. Patients should be advised to eat a consistent daily amount of these foods in order to maintain a constant INR.
The consumption of alcohol may also affect the action of warfarin. Acute alcohol intake of more than a few drinks increases the anticoagulant effects of warfarin and can lead to increased bleeding [42]. Conversely, chronic alcohol use is associated with increased warfarin metabolism and impaired anticoagulation. In general, patients who are taking warfarin should abstain from alcohol consumption or limit their intake to one or two servings of alcohol occasionally.
Other agents used for anticoagulation include heparin, low-molecular-weight heparin, rivaroxaban, and dabigatran, all of which require a very cautious approach when treating older patients. Close monitoring and follow-up over time are essential to improved outcomes. As patients age, their health and functional status may change, and providers should reassess patients for contraindications to anticoagulant therapy, including falls, gastrointestinal bleeding, and hemorrhagic stroke. These complications are life-threatening, and their risk may outweigh the potential benefit of anticoagulation, requiring the cessation of therapy. Another important consideration is the patient's adherence to the prescribed therapy and regular blood testing. Patients should not be prescribed warfarin if they are not able to comply with monitoring requirements.
The management of diabetes often requires the prescription of multiple medications for optimum control, and this coupled with multiple medical comorbidities and a lack of research focusing on geriatric patients can make treatment complex. The main goal of treatment of diabetes in older adults is to decrease metabolic complications while maintaining functional status and quality of life. Individualized management is crucial.
Age-related changes in drug absorption, distribution, metabolism, and clearance should be considered in all older patients with diabetes. For frail patients, the risks of intensive glycemic control often outweigh the benefits. Older adult patients may have a variable dietary intake related to physical or mental illness. If a patient skips a meal because he or she feels ill but takes or is given the usual dosage of insulin (especially fast-acting insulin), hypoglycemia will occur. Even when adequate calories are consumed, the older adult's intestinal absorption of those calories is slowed. As a patient ages, the adrenergic response to low blood glucose diminishes or disappears. Additionally, the preliminary symptoms of hypoglycemia, including lack of motor skills and confusion, may be misdiagnosed or unrecognized [43]. This can result in hypoglycemia unawareness in older patients with diabetes, which can allow the condition to become more severe. Unawareness of hypoglycemia is associated with a six-fold and nine-fold increased risk of severe hypoglycemia in patients with type 1 and type 2 diabetes, respectively [44].
Risk factors for hypoglycemia include polypharmacy, chronic renal or hepatic impairment, poor nutrition, comorbidities, the use of insulin or sulfonylureas, and acute illness. The Beers criteria lists sliding-scale insulin as a potentially inappropriate medication for persons older than 65 years of age, with a higher risk of hypoglycemia without improvement in diabetic management regardless of care setting [4]. The preferred treatment for older patients who require insulin is a daily basal rate of long-acting insulin, such as insulin glargine or insulin detemir. As with other geriatric prescribing, it is important to start with a low dose and increase slowly. Weekly re-evaluation and close monitoring is required, especially when initiating insulin therapy in patients older than 60 years of age.
Oral hypoglycemics can have a rebound hypoglycemic effect in patients with decreased renal or hepatic function (including age-related changes), and sulfonylureas (e.g., glipizide, glyburide) should be used with caution in older adult patients or those who are debilitated or malnourished [45]. Metformin has a low risk of hypoglycemia, but its use in older patients may be limited by comorbid illnesses such as renal/hepatic dysfunction, congestive heart failure (CHF), or chronic obstructive pulmonary disease (COPD). Metformin begins to accumulate in the body when the glomerular filtration rate (GFR) is less than 50–60 mL/min/1.73 m2. Therefore, as a patient ages and renal function declines, it may be necessary to initiate more frequent monitoring of renal function and/or reduce or discontinue metformin use. For geriatric patients, especially those older than 80 years of age, the risks and potential adverse effects of metformin may preclude its use. If it is used, reduced dosage (250–500 mg twice daily) should be considered along with close monitoring of renal function and monthly metabolic panels.
Intense diabetes treatment and tight glycemic control can have serious consequences in older adult patients [21,22]. In the geriatric population, hypoglycemia is associated with an increased risk of myocardial infarction, functional decline, falls, and cognitive impairment. The AGS recommends a target glycated hemoglobin (HbA1c) of 7.5% to 8% in older patients with moderate comorbidities and life expectancy less than 10 years; the American Diabetes Association recommends a more relaxed goal of 8% to 8.5% for older patients with complex medical issues [23,51]. When diabetes is aggressively managed using guidelines more appropriate for younger adults, older patients are at risk for hypoglycemia and other complications. When deciding whether to offer or continue treatment of diabetes for patients older than 70 years of age, the benefit/risk ratio should factor in comorbidities, cognitive status, ability to self-manage, life expectancy, and vulnerability to hypoglycemia [24].
Most hospitalized patients with diabetes will require bolus insulin before meals and at bedtime based on blood glucose monitoring. These orders will be included on the discharge medication list if the patient is discharged to a skilled nursing facility. However, patients with stable type 2 diabetes should transition away from the daily blood glucose testing and on-demand insulin routinely ordered for hospitalized patients. One approach is to continue the bolus insulin at mealtimes for seven days while discontinuing bedtime doses to reduce the risk of nocturnal hypoglycemia. After seven days, the amount of insulin required daily may be used to establish a daily long-acting insulin regimen.
For example, if a patient required 72 units of regular insulin in one week, an average daily dose of about 10 units may be used as a starting point for long-lasting insulin (i.e., 10 units subcutaneous insulin each day). While in the process of adjusting insulin dosages, re-evaluation every week is prudent. Patients with glucose spikes may require the addition of bolus insulin before meals.
It is important to consider how often patients are willing to self-monitor blood glucose and give themselves insulin. While a small percentage of patients/caregivers would comply with testing and coverage four times per day, this is not a realistic expectation for most patients. Patients may complain about the pain of fingersticks and frequent injections of insulin, and episodes of hypoglycemia may make patients feel ill for days and require emergency intervention/hospitalization. Reducing any unnecessary medications, treatments, and intervention creates good rapport and allows more time to focus on improving the patient's quality of life.
Insomnia is a common complaint by older adult patients, causing many visits to primary care providers and psychiatrists. As a person ages, sleep patterns typically change. Increased sleep during the day causes a reduced need for sleep at night. There may be a perceived insomnia by the patient despite sleeping 7 hours or more in a 24-hour period. For patients with insomnia, providers should complete a comprehensive medical and psychologic assessment to diagnose underlying conditions that may cause a sleep disorder, including an acute stressor (e.g., grief), chronic pain, sleep apnea, anxiety, depression, or alcohol/substance misuse. Diagnosis and treatment of underlying comorbidities may make pharmacotherapy unnecessary.
A sleep hygiene regimen is a universally applicable prevention and treatment strategy that can improve sleep quality for those with and without a specific sleep disorder. Sound sleep hygiene practices should be discussed with patients and/or caregivers, and willingness to undertake these and other nonpharmacologic options should be assessed and encouraged [16]. Exercise history should also be obtained, and when levels are inadequate, exercise as treatment should be discussed. Increased physical activity has been shown to be as effective as benzodiazepines in improving sleep patterns in several studies [17]. Other nonpharmacologic treatment options include relaxation therapy and sleep restriction [17]. These are often first-line treatments due to the low cost, lack of side effects, and no risk of dependency. The use of over-the-counter sleep aids (especially those containing antihistamines) should be discouraged, as should the use of alcohol, due to marginal efficacy and reduction in sleep quality and because they may cause residual drowsiness and have the potential for dependency [17].
If medication is indicated to treat insomnia, the benzodiazepine temazepam (7.5 mg in older patients) is frequently used, as it is available in a generic form with a relatively low cost. According to the AGS Beers criteria, benzodiazepines are potentially inappropriate medications in the older adult population and should be avoided if possible. Safer alternatives to temazepam include sedative-hypnotics (e.g., suvorexant) and melatonin. If a hypnotic is selected, it should be prescribed at a lower dose than for younger patients. Melatonin may be of benefit to a subset of patients with delayed sleep phase syndrome (a disturbance of the circadian rhythm), but it does not appear to be helpful for most people who have insomnia. It is safe when used in modest dosage (0.2–0.3 mg per night) for short periods (three months or less) [39]. Melatonin is unregulated by the FDA, so formulations vary in strength, and higher doses can lead to adverse side effects (e.g., disrupted sleep, fatigue, headache).
Another frequently used sleep aid is diphenhydramine, with multiple over-the-counter formulations available. Despite its widespread use, diphenhydramine is included on the Beers criteria as a potentially inappropriate medication in older adults. Diphenhydramine is a first-generation antihistamine that is metabolized by the liver through the CYP-450 pathway. It non-selectively antagonizes central and peripheral histamine H1 receptors. It is a substrate/inhibitor, has a half-life of 3.4 to 9.2 hours, and is excreted in the urine. It has strong anticholinergic effects, which may cause constipation, dry mouth, impaired coordination, urinary retention, and hypotension. Drugs like this that have anticholinergic side effects may have a cumulative effect, leading to increased sedation or delirium. Drug-disease precautions include glaucoma, prostatic hypertrophy, asthma, COPD, and gastrointestinal obstruction. Tolerance may develop when used as a sleeping agent, causing reduced effect and resulting in patients increasing their dosages, with associated higher risks of adverse reactions. Caution is recommended when using this drug in older patients, especially those who take other central nervous system (CNS) depressants and/or anticholinergics. It should be avoided as a sleep aid in favor of better options.
The AGS also recommends that hypnotics (e.g., eszopiclone, zaleplon, zolpidem) should be avoided (without consideration of duration of use), particularly if any other CNS-active drugs are already prescribed. These drugs have adverse events in older adults similar to benzodiazepines [4]. Liver impairment and concomitant alcohol use are relative contraindications. Again, prescribers should weigh the potential of a positive benefit to the patient versus the risk of an adverse event. After prolonged use, tapering of these medications is required to prevent withdrawal symptoms.
As noted, CMS regulations for long-term care facilities require psychotropic/antipsychotic drugs to be used only when necessary to treat a specific condition, as diagnosed and documented in the medical record and based on a comprehensive assessment of the resident [12]. The dosage of these medications should be gradually reduced until the lowest effective dose is established, unless clinically contraindicated. Of particular concern is the chemical restraint of older long-term care facility residents, which has resulted in increased scrutiny of psychotropic medication prescribing by state and federal regulatory agencies.
Typical (e.g., haloperidol, thorazine) and atypical antipsychotics (e.g., olanzapine, risperidone) are considered dangerous when used in patients with dementia, and the FDA has issued a black box warning that older adult patients with dementia-related psychosis treated with antipsychotics are at an increased risk for death (mainly due to cardiovascular or infectious causes) compared with placebo [45]. In terms of liability, the burden of proof lays with providers and caregivers to provide the documentation that the medication is medically necessary.
Antipsychotic medications are used to treat psychosis associated with bipolar disorder, schizophrenia, agitated depression, delusions, dementia, and paranoia. However, before prescribing antipsychotic drugs, any medical or toxic causes of the behavior should be ruled out. A thorough patient history, physical examination, and laboratory studies (e.g., complete blood count, comprehensive metabolic panel, urinalysis, drug levels) are indicated for differential diagnosis.
For residents in long-term care facilities, nonpharmacologic methods of behavior control should be attempted and documented before antipsychotics are initiated. As with any treatment, a risk-benefit analysis should be performed, and the patient's/caregiver's agreement to use the drug should be documented (Table 2).
STEPS FOR ANTIPSYCHOTIC PRESCRIBING
|
1. History and physical exam of patient, with appropriate documentation 2. Metabolic/laboratory work-up, as indicated 3. Review of nonpharmacologic interventions 4. Discussion of potential risks and benefits of antipsychotics 5. Consider psychiatry evaluation 6. Review medication regimen and comorbidities for potential interactions/contraindications 7. Consult pharmacist, as necessary 8. Make a follow-up plan
|
| aPatients are most likely to experience adverse reactions in the first two weeks of taking an antipsychotic. |
Healthcare providers should work to promote and maintain the independence and mobility of patients with dementia without relying on antipsychotics. The first step for those in skilled nursing facilities is maintaining consistent staffing. Additional interventions should focus on maximizing independent activity and enhancing function while adapting and developing skills and may include:
Retaining a familiar environment
Minimizing relocations
Accommodating fluctuating abilities
Obtaining assessment and care-planning advice regarding activities of daily living, toileting skills, and skill training from an occupational therapist
Modifying the environment to aid independent functioning, including incorporating assistive technology, with advice from an occupational therapist and/or clinical psychologist
Encouraging physical exercise, with assessment from physical, occupational, and speech therapists (when indicated)
Specific therapeutic approaches (e.g., social worker/spiritual assessment, redirection, distraction, reality orientation, reminiscence therapy, art therapy, music therapy, bright-light therapy)
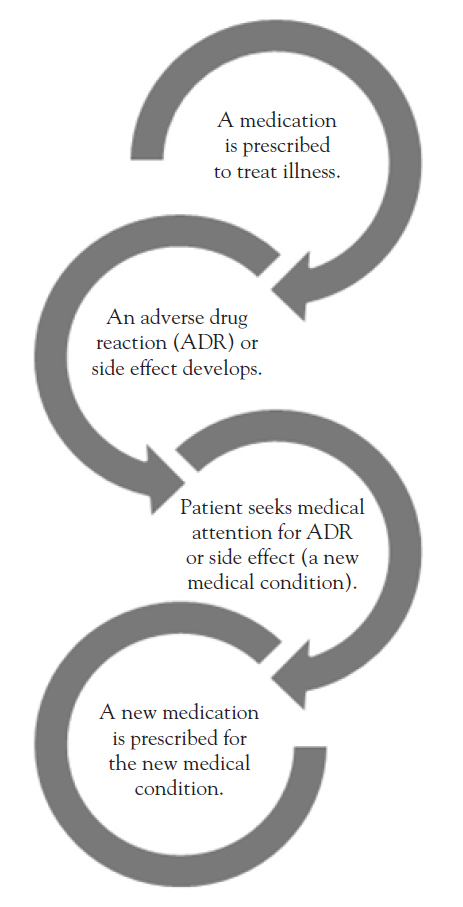
A prescribing cascade occurs when a patient has an ADR and additional drugs are prescribed to control the symptoms of this reaction (Figure 1). Adverse drug reactions should be vigilantly ruled out prior to diagnosing a new medical condition.
Prompt detection and correction of ADRs are essential to ensuring good outcomes. Changes in medications and dosages should serve as red flags when evaluating a potential new problem.
An example of a prescribing cascade involves the prescription of cholinesterase inhibitors (e.g., donepezil, rivastigmine, galantamine) for dementia and the secondary prescription of an anticholinergic (e.g., oxybutynin, tolterodine, flavoxate) for urinary incontinence. Cholinesterase inhibitors and anticholinergic drugs are considered to have opposing effects, and patients who receive a cholinesterase inhibitor have a significant increase in urinary incontinence, with the subsequent prescription of an anticholinergic agent for bladder control [8]. Even aside from potential drug-drug interactions, medications with anticholinergic side effects should be avoided in patients with dementia as they increase the risk for cognitive decline and delirium [12].
During a transition of care (i.e., when a patient moves from one site of health care to another) it is common for essential information not to be conveyed to the receiving care providers. It is vital for care transitions to be coordinated so each provider has a comprehensive view of the patient. Poor communication can result in duplication of services, waste of resources, and medication errors. Potentially and actually inappropriate medications for older patients are frequently prescribed in hospital and emergency department settings and continued after discharge [40]. Prescribers in these settings often fail to adhere to guidelines to modify prescribing practices for older patients [13].
When patients return home or to a care facility from a hospital, it is important to establish the indication(s), duration(s), and monitoring parameters of any new medications. If time is not taken to review new and existing orders and resolve any potential conflicts, the risk for unnecessary polypharmacy increases.
Medication reconciliation is the process of creating and updating a current medication list as compared with any previous lists. This should be conducted:
On admission
During routine and acute visits by providers
After transitions of care
During significant change in condition
When the goals of care change
Before prescribing new medications
When discontinuing any as-needed or routine orders
When considering the risks, benefits, and burden of any prescription
Attempts to reduce or discontinue medications should be done selectively, one medication at a time. Medications may have been initially prescribed many years previously for a chronic condition, and newer agents with improved side effect profiles may be available. In some patients, addiction and dependence issues may arise. Regular comprehensive assessments of the patient are crucial, with the appropriate and necessary referrals made to psychiatry, psychology, addiction specialists, pain management, and other specialists as necessary. Certain medications should not be abruptly stopped and should instead be slowly reduced with medical supervision. This includes benzodiazepines (due to risk of symptoms of drug withdrawal), beta-blockers (due to risk of rebound cardiac symptoms), and corticosteroids (due to risk of adrenal crisis).
When a patient is transferred to the hospital, certain routine medications (e.g., anticholinesterase inhibitors) may be stopped. Therefore, it is important to review the pre-hospital medication regimen and compare it with the medication list provided by the hospital. If important omissions are found, they may need to be reordered.
In long-term care facilities, residents are visited by their primary care provider every 30 to 60 days. The medication regimen should be carefully reviewed and reconciled at each of these visits. Psychotropic drug meetings are conducted monthly and include the medical director, nursing director, a social worker, and a psychiatrist. The continued necessity of antipsychotic, antidepressant, and psychoactive medications should be reviewed for medical necessity and possible gradual dosage reductions. If further dosage reductions are clinically contraindicated, this should be documented on the progress note.
Patient T is a man, 84 years of age, who resides in a long-term care facility. He has been diagnosed with congestive heart failure, hypertension, arthritis, and hyperlipidemia and has a history of two myocardial infarctions (eight and two years previously). He requires minimal assistance with his activities of daily living and remains ambulatory with a cane. His usual medications are:
Metoprolol ER: 50 mg daily
Aspirin: 325 mg daily
Omeprazole: 20 mg daily
Lisinopril: 10 mg daily
Furosemide: 40 mg every day
Potassium chloride: 20 mEq twice daily
Atorvastatin: 20 mg daily
Acetaminophen: 650 mg twice daily
Tramadol: 50 mg, as needed
Multivitamin
At baseline, he takes 10 medications/supplements.
Patient T is transferred to the emergency department for increased shortness of breath. He is diagnosed with bronchitis and spends 24 hours in the hospital for observation before being transferred back to the long-term care facility for ongoing care. At the care facility, the receiving practitioner reviews the medication list from the hospital:
Levofloxacin: 500 mg daily
Prednisone: 20 mg daily
Tiotropium bromide, inhalation: One puff daily
Levalbuterol tartrate, inhalation solution for nebulizer: As needed for shortness of breath
Promethazine: 25 mg every six hours as needed
Haloperidol: 1 mg every four hours as needed
Bisacodyl: 10 mg every day as needed
Including the as-needed medications, Patient T is currently prescribed 17 drugs. Physical assessment reveals a debilitated man who is in no acute distress (Table 3). He is alert and oriented and answers questions appropriately. His intake of food and fluids has been poor since his return from the hospital, and he is using oxygen per nasal cannula at 2 L/minute.
RESULTS OF PATIENT T'S PHYSICAL EXAM
| Parameter | Measurement |
|---|---|
| Blood pressure | 112/62 mm Hg |
| Temperature | 97.8° F |
| Heart rate | 92 beats per minute |
| Respiration rate | 22 breaths per minute |
| Height | 5 feet 9 inches (175 cm) |
| Weight | 65.3 kg (144 pounds) (usual: 154 pounds) |
| Heart sounds | S1, S2 with 2/6 systolic ejection murmur |
| Lung sounds | Few expiratory wheezes noted anteriorly |
| Extremities | No significant edema |
| Abdomen | Unremarkable |
As noted, Patient T's new diagnosis is bronchitis, and he does not appear to be having an exacerbation of his CHF. The first step in medication reconciliation is to discontinue any as-needed medications ordered in the hospital that are no longer necessary. Haloperidol is frequently used to treat delirium in geriatric patients in the hospital setting, but it is considered inappropriate for this use in long-term care facilities. Secondly, duration should be established for levofloxacin and prednisone. The receiving practitioner contacts the ordering physician and determines the levofloxacin should be continued for seven days and the prednisone continued for two weeks with a plan for tapering to discontinue. A pulmonary consultation follow-up is scheduled in two weeks, during which the pulmonologist will determine the duration of the inhalation drugs started during the hospitalization. The oxygen therapy was also acquired during the hospitalization, and serial oxygen saturation readings will be used to determine whether Patient T will require long-term oxygen therapy.
As discussed, the reduction or discontinuation of medications should be done cautiously, generally one medication at a time. The provider reviews the drugs the patient usually takes, evaluating for polypharmacy or any prescribing problems with the drugs ordered (Table 4).
REVIEW OF PATIENT T'S MEDICATIONS
| Medication | Indication/Diagnosis |
Potential Reduction | Considerations for Reduction or Discontinuation of Drugs |
|---|---|---|---|
| Metoprolol extended-release (50 mg daily) | Hypertension | No | If blood pressure or heart rate fall, or symptoms of orthostasis or hypotension occur, dosage reduction should be considered. |
| Aspirin (325 mg daily) | Heart disease | Yes | Lowest effective dose. Would 81 mg dose be as effective with less risk of gastrointestinal (GI) bleeding? |
| Omeprazole (20 mg daily) | Gastritis | Yes | Possibility that this may no longer be necessary if given for an acute episode. Consideration for GI prophylaxis related to steroids and/or aspirin. |
| Lisinopril (20 mg daily) | Congestive heart failure, hypertension | No | ACE inhibitor should be part of treatment plan for patients with congestive heart failure. Renal function must be monitored. |
| Furosemide (40 mg daily) | Congestive heart failure | Yes | Consideration of the lowest effective dose. During acute illness (except congestive heart failure), especially with dehydration, dose reduction or holding dose may be appropriate. |
| Potassium chloride (20 mEq [oral] daily) | Replacement | No | Potassium levels should be monitored, with adjustment of dose as required. |
| Atorvastatin (20 mg daily) | Hyperlipidemia | Yes | Limited evidence base for use of statins in patients older than 80 years of age. Comorbid heart disease is an important consideration. Monitoring of lipid levels and liver function levels guides treatment. |
| Acetaminophen (650 mg [oral] twice daily) | Arthritis | No | 1,300 mg of acetaminophen daily is well below the maximum recommended dose. Caution when used with alcohol or other drugs metabolized by the liver. |
| Tramadol (50 mg every four hours as needed) | Pain | Yes | Not used regularly. |
| Multivitamin (1 tablet daily) | Supplement | No | This was started because the patient had weight loss and poor intake of food and fluid. The nutritional support could potentially help. |
When reconciling the medication regimen, the first step is to identify the clinical indication for each medication. There are no obvious inappropriate prescribing practices in Patient T's record, and in a patient with multiple comorbidities, polypharmacy may become the standard. However, attempts at medication reduction and discontinuation should be attempted.
His primary care provider discusses the medication regimen with the patient and his daughter, who is his healthcare surrogate. They express interest in attempting to reduce the number of medications Patient T is taking.
The provider discontinues tramadol and writes orders to taper off omeprazole by giving every other day for 10 days, then stopping. The aspirin dose is reduced to 81 mg every day, and the atorvastatin is reduced to 10 mg daily with a plan for serial lipid measurements.
The ongoing dosages of furosemide and potassium are determined by the level of heart failure. When patients decline and lose weight, adjustments to diuretic dosages are required to prevent subsequent dehydration. Older patients are at increased risk of falls and complications due to orthostatic hypotension, and if the patient is dehydrated, the effects are more profound and potentially dangerous. Measurement of orthostatic vital signs will help to determine the appropriate dosage of metoprolol, lisinopril, and even furosemide. After the reconciliation, omeprazole and tramadol are discontinued and the dosages of several drugs are reduced (Table 5).
PATIENT T'S MEDICATION LIST AFTER RECONCILIATION
| Medication | Comments |
|---|---|
| Metoprolol extended-release (25 mg daily) | Patient was experiencing some orthostasis with position changes that placed him at risk for falls. The daily dose was reduced to 25 mg, which helped normalize the blood pressure readings while still providing the benefits of a beta-blocker. |
| Enteric-coated aspirin (81 mg daily) | Dosage was reduced due to risk of bleeding. |
| Lisinopril (10 mg daily) | Dose was reduced to the lowest effective dose, and blood pressure improved. |
| Furosemide (20 mg daily) | Dosage reduced as the patient's heart failure was stable. Chest x-ray clear, no edema. |
| Potassium chloride (20 mEq [oral] daily) | As the furosemide is reduced, the potassium dosage can be reduced, with follow-up testing ordered to monitor levels. |
| Atorvastatin (10 mg daily) | When tested, the cholesterol was 159 mg/dL, so the dosage was reduced. |
| Acetaminophen (650 mg twice daily) | This helped with his arthritis pain, so it was continued for pain management. |
| Multivitamin (1 tablet daily) | This was continued for nutritional support. |
Comments and Discussion : Mr. T's case shows an example of multiple comorbid conditions and polypharmacy. There are no obviously inappropriate drugs, and each medication has a clinical indication consistent with evidence-based medicine. This makes medication reduction and reconciliation difficult.
When medication regimens are adjusted, the patient should be monitored and re-evaluated regularly to detect any adverse reactions. In some cases, a trial dosage reduction or discontinuation of a medication, with close monitoring of the patient response, is necessary.
Medication reduction in older adults should be done slowly and conservatively to prevent rebound effects. Manufacturer and FDA recommendations should be followed for tapering to reduce or discontinue medications. The provider must refer to packaging inserts and information.
Individuals with life-limiting illnesses may not be candidates for curative or restorative therapy. These patients benefit from palliative care, defined as "processes of care designed to prevent and treat physical, emotional, and spiritual suffering in order to enhance quality of life for patients with chronic, progressive illnesses" [13]. Medications in palliative care provide comfort and address symptoms that impede function or negatively affect quality of life; many of these medications appear on the Beers List. In the case of terminal pain and suffering, medications known to cause side effects in the older adult population may be used if the benefits outweigh the drawbacks. Providing care and comfort to a patient at the end of life may only be possible with the use of drugs that would be potentially inappropriate in patients without life-limiting disease.
The goals of care usually change with age, as patients develop multiple illnesses, functional decline, and/or dependency. Goals should be determined by individual preferences and regularly reassessed by the treatment team. Although there is great diversity in individual preferences regarding interventions at the end of life, most patients do not want to suffer or experience pain during this stage.
Including multiple oral medications in palliative care plans at the end of life can be a burden to patients and caregivers. At the end of life, a patient's oral intake usually decreases due to dysphagia and a decline in the desire for food and fluids, and taking pills, tablets, or capsules may be difficult. Alternative routes of administration should be considered for these patients. Injectable medications are generally avoided due to the pain associated with administration, unless an intravenous line is available. As end-of-life care tends to take place in nursing facilities or at home, IV access is usually not available. However, transdermal, sublingual, sprays/tablets, buccal films, nasal sprays, suppositories, and lozenges may be appropriate. In addition, all medications not necessary to provide care and comfort to the patient should be discontinued.
As with any patient, informed consent should be obtained prior to the initiation of any new treatments or medications for patients receiving palliative care. This education should include potential benefits and risks of the proposed treatment. The responsible party should have the opportunity to ask questions, get a second opinion if desired, then accept or refuse any treatment or drug regimen.
Polypharmacy in geriatrics is a serious problem that is expected to grow in scope as the population ages. Certain medications commonly used in the younger population may be considered inappropriate in older adults, due to pharmacodynamics, pharmacokinetics, and/or drug-disease interactions. The Beers criteria may be used to identify potentially inappropriate prescribing practices for older patients.
Many older adult patients live in assisted living or nursing facilities, and these facilities are subject to regulatory scrutiny at the state and federal levels. Drugs prescribed in the presence of adverse reactions, in excessive duration, in excessive dose, duplication of therapy, and/or without clinical indications are considered unnecessary by regulatory bodies.
Healthcare professionals should conduct comprehensive assessments of their older patients. Social, financial, and/or functional issues may increase the risk for ADRs, non-adherence, and medication errors. Prescribing practices commonly used for adults may cause more harm than benefit in older individuals. It is important to create individualized treatment plans that effectively communicate the risk-benefit profile of any drug or treatment. A multidisciplinary approach is necessary to provide optimal care to older adult patients in all care settings.
1. U.S. Census Bureau. Population Projections. Available at https://www.census.gov/programs-surveys/popproj.html. Last accessed January 22, 2025.
2. IMS Institute for Healthcare Informatics. Avoidable Costs in U.S. Healthcare: The $200 Billion Opportunity from Using Medicines More Responsibly. Available at http://offers.premierinc.com/rs/381-NBB-525/images/Avoidable_Costs_in%20_US_Healthcare-IHII_AvoidableCosts_2013%5B1%5D.pdf. Last accessed January 22, 2025.
3. Jyrkkä J, Enlund H, Korhonen MJ, Sulkava R, Hartikainen S. Patterns of drug use and factors associated with polypharmacy and excessive polypharmacy in elderly persons: results of the Kuopio 75+ study: a cross-sectional analysis. Drugs Aging. 2009;26(6):493-503.
4. By the 2023 American Geriatrics Society Beers Criteria Update Expert Panel. American Geriatrics Society 2023 updated AGS Beers Criteria for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2023;71(7):2052-2081.
5. Gallagher P, Ryan C, Byrne S, Kennedy J. STOPP (Screening Tool of Older Person's Prescriptions) and START (Screening Tool to Alert doctors to Right Treatment): consensus validation. Int J Clin Pharmacol Ther. 2008;46(2):72-83.
6. Rothberg MB, Pekow PS, Liu F, et al. Potentially inappropriate medication use in hospitalized elders. J Hosp Med. 2008;3(2): 91-102.
7. Centers for Disease Control and Prevention. Health Care and Insurance. Available https://www.cdc.gov/nchs/fastats/health-care-and-insurance.htm. Last accessed January 22, 2025.
8. Gill S, Mamdani M, Naglie G, et al. A prescribing cascade involving cholinesterase inhibitors and anticholinergic drugs. Arch Intern Med. 2005;165(7):808-813.
9. Centers for Disease Control and Prevention. FastStats: Medication Safety Data. Available at https://www.cdc.gov/medication-safety/data-research/facts-stats. Last accessed January 22, 2025.
10. Wu C, Bell CM, Wodchis WP. Incidence and economic burden of adverse drug reactions among elderly patients in Ontario emergency departments: a retrospective study. Drug Saf. 2012:35(9):769-781.
11. Shepherd G, Mohorn P, Yacoub K, May DW. Adverse drug reaction deaths reported in United States vital statistics, 1999–2006. Ann Pharmacother. 2012;46(2):169-175.
12. Centers for Medicare and Medicaid Services. Advanced Copy: Dementia Care in Nursing Homes: Clarification to Appendix P State Operations Manual (SOM) and Appendix PP in the SOM for F309–Quality of Care and F329–Unnecessary Drugs. Available at https://www.cms.gov/medicare/provider-enrollment-and-certification/surveycertificationgeninfo/downloads/survey-and-cert-letter-13-35.pdf. Last accessed January 22, 2025.
13. Society for Post-Acute and Long-Term Care (AMDA). White Paper on Palliative Care and Hospice in Long-Term Care. Columbia, MD: Society for Post-Acute and Long-Term Care; 2007.
14. Hughes CM, Cooper JA, Ryan C. Going beyond the numbers: a call to redefine polypharmacy. Br J Clin Pharmacol. 2014;77(6): 915-916.
15. Cooper JA, Cadogan CA, Patterson SM, et al. Interventions to improve the appropriate use of polypharmacy in older people: a Cochrane systematic review. Cochrane Database Syst Rev. 2015;(5):e009235.
16. Hilty D, Young JS, Bourgeois JA, Klein S, Hardin KA. Algorithms for the assessment and management of insomnia in primary care. Patient Prefer Adherence. 2009;3:9-20.
17. Ramakrishnan K, Scheid DC. Treatment options for insomnia. Am Fam Physician. 2007;76(4):517-526.
18. Barry PJ, Gallagher P, Ryan C, O'Mahony D. START (Screening Tool to Alert doctors to the Right Treatment): an evidence-based screening tool to detect prescribing omissions in elderly patients. Age Ageing. 2007;36:632-638.
19. Le J. Drug Metabolism. Available at https://www.merckmanuals.com/professional/clinical-pharmacology/pharmacokinetics/drug-metabolism. Last accessed January 22, 2025.
20. U.S. Department of Health and Human Services. CMS Manual System. Pub. 100-07 State Operations Provider Certification. Available at https://www.cms.gov/Regulations-and-Guidance/Guidance/Transmittals/Downloads/R127SOMA.PDF. Last accessed January 22, 2025.
21. Thorpe CT, Gellad WF, Good CB, et al. Tight glycemic control and use of hypoglycemic medications in older veterans with type 2 diabetes and comorbid dementia. Diabetes Care. 2015;38(4):588-595.
22. Vijan S, Sussman JB, Yudkin JS, Hayward RA. Effect of patients' risks and preferences on health gains with plasma glucose level lowering in type 2 diabetes mellitus. JAMA Intern Med. 2014;174(8):1227-1234.
23. American Geriatrics Society Expert Panel on Care of Older Adults with Diabetes Mellitus, Moreno G, Mangione CM, Kimbro L, Vaisberg E. Guidelines abstracted from the American Geriatrics Society guidelines for improving the care of older adults with diabetes mellitus: 2013 update. J Am Geriatr Soc. 2013;61:2020-2026.
24. Sinclair AJ, Paolisso G, Castro M, et al. European Diabetes Working Party for Older People 2011 clinical guidelines for type 2 diabetes mellitus: executive summary. Diabetes Metab. 2011;37(Suppl 3):S27-S38.
25. Kapo J, Morrison LJ, Liao S. Palliative care for the older adult. J Palliat Med. 2007;10(1):185-209.
26. Bekelman DB, Hutt E, Masoudi FA, Kutner JS, Rumsfeld JS. Defining the role of palliative care in older adults with heart failure. Int J Cardiol. 2008;125(2):183-190.
27. Masoudi FA, Krumholz HM. Polypharmacy and comorbidity in heart failure. BMJ. 2003;327(7414):513-514.
28. Havranek E, Masoudi FA, Westfall KA, Wolfe P, Ordin DL, Krumholz HM. Spectrum of heart failure in older patients: results from the National Heart Failure project. Am Heart J. 2002;143(3):412-417.
29. Holmes HM, Sachs GA, Shega JW, Hougham GW, Cox Hayley D, Dale W. Integrating palliative medicine into the care of persons with advanced dementia: identifying appropriate medication use. J Am Geriatr Soc. 2008;56(7):1306-1311.
30. Stevenson J, Abernethy AP, Miller C, Currow DC. Managing comorbidities in patients at the end of life. BMJ. 2004;329(7471):909-912.
31. Helme RD, Gibson SJ. The epidemiology of pain in elderly people. Clin Geriatr Med. 2001;17:417-431.
32. Ferrell BA, Ferrell BR, Rivera L. Pain in cognitively impaired nursing home patients. J Pain Sympt Manage. 1995;10:591-598.
33. American Geriatrics Society Panel on Pharmacological Management of Persistent Pain in Older Persons. Pharmacological management of persistent pain in older persons. J Am Geriatr Soc. 2009;57:1331-1346.
34. Mitchell SL, Teno JM, Kiely DK, et al. The clinical course of advanced dementia. N Engl J Med. 2009;361:1529-1538.
35. Hanson LC, Eckert KJ, Dobbs D, et al. Symptom experience of dying long-term care residents. J Am Geriatr Soc. 2008;56:91-98.
36. Teno JM, Clarridge BR, Casey V, et al. Family perspectives on end-of-life care at the last place of care. JAMA. 2004;291:88-93.
38. Vandenberg EV, Tvrdik A, Keller BK. Use of the quality improvement process in assessing end-of-life care in the nursing home. J Am Med Dir Assoc. 2005;6(5):334-339.
39. Buscemi N, Vandermeer B, Hooton N, et al. The efficacy and safety of exogenous melatonin for primary sleep disorders: a meta-analysis. J Gen Intern Med. 2005;20(12):1151-1158.
40. Morandi A, Vasilevskis E, Pandharipande PP, et al. Inappropriate medication prescriptions in elderly adults surviving an intensive care unit hospitalization. J Am Geriatr Soc. 2013;61(7):1128-1134.
41. Brown JD, Hutchison LC, Li C, Painter JT, Martin BC. Predictive validity of the Beers and Screening Tool of Older Persons' Potentially Inappropriate Prescriptions (STOPP) criteria to detect adverse drug events, hospitalizations, and emergency department visits in the United States. J Am Geriatr Soc. 2016;64(1):22-30.
42. Weathermon R, Crabb DW. Alcohol and medication interactions. Alcohol Research and Health. 1999;23(1):40-54.
43. Vivian EM. Type 2 diabetes across the life span. In: Cornell S, Halstenson C, Miller DK (eds). The Art and Science of Diabetes Self-Management Education: Desk Reference. 4th ed. Chicago, IL: American Association of Diabetes Educators; 2017: 393-410.
44. Kalra S, Mukherjee JJ, Venkataraman S, et al. Hypoglycemia: the neglected complication. Indian J Endocrinol Metab. 2013;17(5):819-834.
45. LexiDrug. Available at https://online.lexi.com. Last accessed January 22, 2025.
46. Steinman MA, Beizer JL, DuBeau CE, Laird RD, Lundebjerg NE, Mulhausen P. How to use the American Geriatrics Society 2015 Beers Criteria: a guide for patients, clinicians, health systems, and payors. J Am Geriatr Soc. 2015;63(12):e1-e7.
47. Lavan AH, Gallagher PF, O'Mahony D. Methods to reduce prescribing errors in elderly patients with multimorbidity. Clin Interv Aging. 2016;11:857-866.
48. Masnoon N, Shakib S, Kalisch-Ellett L, Caughey GE. What is polypharmacy? A systematic review of definitions. BMC Geriatr. 2017;17(1):230.
49. O'Mahony D, O'Sullivan D, Byrne S, et al. STOPP/START criteria for potentially inappropriate prescribing in older people: version 2. Age Ageing. 2015;44(2):213-218.
50. U.S. Department of Health and Human Services, Centers for Medicare and Medicaid Services. Medicare and Medicaid Programs; Reform of Requirements for Long-Term Care Facilities. Available at https://www.govinfo.gov/content/pkg/FR-2016-10-04/pdf/2016-23503.pdf. Last accessed January 22, 2025.
51. American Diabetes Association. Position statement, older adults: standards of medical care in diabetes—2018. Diab Care. 2018;41(Supp 1):S119-S125.
52. U.S. Department of Health and Human Services. CMS State Operations Manual Appendix PP: Guidance to Surveyors for Long Term Care Facilities. Available at https://www.cms.gov/Medicare/Provider-Enrollment-and-Certification/GuidanceforLawsAndRegulations/Downloads/Appendix-PP-State-Operations-Manual.pdf. Last accessed January 22, 2025.
1. Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. KDIGO 2024 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Kidney Int. 2024 Apr;105(4S):S117-S314. Available at https://www.kidney-international.org/article/S0085-2538%2823%2900766-4/fulltext. Last accessed January 27, 2025.
2. Agency for Healthcare Quality and Research. Medications at Transitions and Clinical Handoffs (MATCH) Toolkit for Medication Reconciliation. Available at https://www.ahrq.gov/patient-safety/settings/hospital/match/index.html. Last accessed January 27, 2025.
3. Bergman-Evans B. Improving medication management for older adult clients residing in long-term care facilities. J Gerontol Nurs. 2013;39(11):11-17. Available at https://www.healio.com/nursing/journals/jgn/2013-11-39-11/{bef23293-68df-4c4e-8a87-89e254b4bef9}/. Last accessed January 27, 2025.
Mention of commercial products does not indicate endorsement.