This course is designed for those in the medical profession with interests in perinatal and maternal-child health. It will assist the participant in supporting, protecting, and promoting breastfeeding for optimal infant growth and development. The historical, social, emotional, and political factors involved in breastfeeding will be reviewed. The hazards of feeding artificial baby milk, review of anatomy, physiology, and the uniqueness of human milk will be addressed within the course. Antepartum educational needs, antepartum assessment for lactational ability, and hospital routines that support lactation are considered. The effects of labor and delivery practices on the breastfeeding relationship are covered in detail. Special circumstances, nursing the premature infant, jaundice, and the use of breastfeeding devices will be explained.
- INTRODUCTION
- THE HISTORY OF INFANT FEEDING
- A PARADIGM SHIFT IN SUPPORT OF BREASTFEEDING
- ARTIFICIAL BABY MILK
- DECIDING TO BREASTFEED
- BREAST MILK
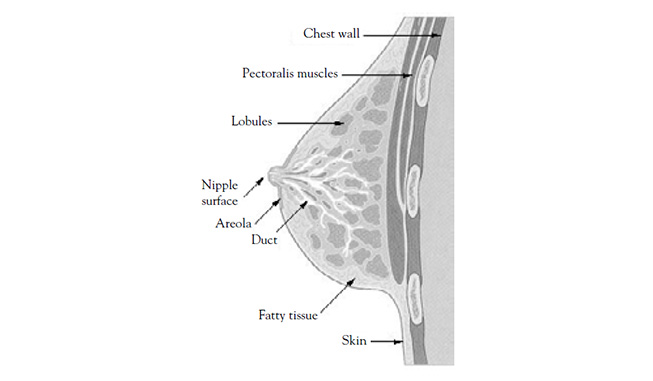
- BREAST ANATOMY AND DEVELOPMENT
- ANTEPARTUM EDUCATION
- ANTEPARTUM LACTATION ASSESSMENT
- HOSPITAL ROUTINES THAT SUPPORT LACTATION
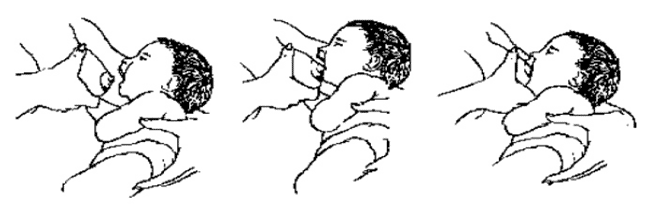
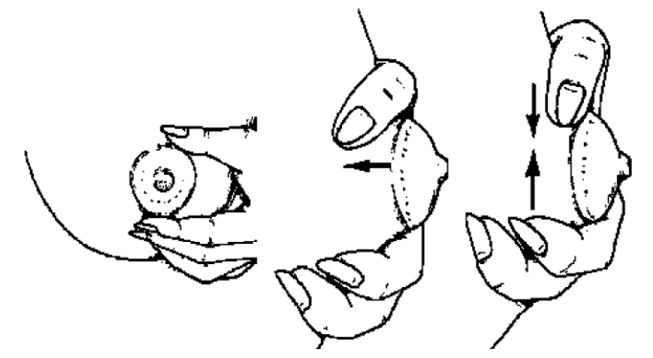
- THE LATCH-ON PROCESS
- COMPLICATIONS
- THE PREMATURE INFANT
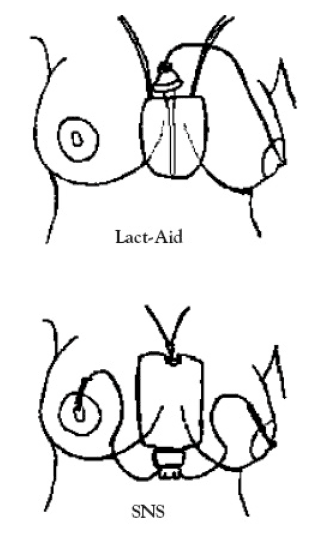
- BREASTFEEDING DEVICES
- BREASTFEEDING: THE EARLY MONTHS
- SUPPORT FOR THE BREASTFEEDING COUPLE
- CASE STUDIES
- FREQUENTLY ASKED QUESTIONS
- CONCLUSION
- RESOURCES
- GLOSSARY
- Works Cited
- Evidence-Based Practice Recommendations Citations
This course is designed for all nurses, especially those working in obstetrics, NICU, and pediatrics.
The purpose of this course is to provide the nurse with an in depth understanding of breastfeeding, including an intensive review of anatomy, physiology, emotional and social factors, and a review of problem solving protocols.
Upon completion of this course, you should be able to:
- Outline the history of infant feeding and breastfeeding.
- Discuss the American Academy of Pediatrics policy statement Breastfeeding and the Use of Human Milk and other signs of the paradigm shift in favor of breastfeeding.
- Review components of the Baby-Friendly Hospital Initiative.
- List the risk and disadvantages associated with artificial baby milk (ABM).
- Describe the components and types of breast milk.
- Discuss the anatomy and development of the breast and the physiology of breast milk production.
- Identify key points of antepartum education and lactation assessments.
- Outline the role of the doula.
- Review various hospital routines that affect breastfeeding, including the use of labor medications and the neonatal assessment.
- Describe the latch-on process.
- List potential maternal complications that may interfere with breastfeeding.
- List potential infant complications that may make breastfeeding difficult or impossible.
- Compare and contrast various devices available for the breastfeeding mother.
- Discuss issues that may arise during the early weeks and months of breastfeeding.
- Identify support available for the breastfeeding couple.
Trish Wierenga, FNP-BC, IBCLC, PMH-C, has been an International Board Certified Lactation Consultant (IBCLC) in central Illinois since 2011 and a board-certified Family Nurse Practitioner since 2021. She is also one of only a few local practitioners certified in Perinatal Mental Health. Trish is a member and the current Chairperson of the International Affiliation of Tongue Tie Professionals (IATP), a member of the Central Illinois Breastfeeding Task Force (CIBTF), and is a former member of the Board of the Illinois chapter of Postpartum Support International (PSI). She has spoken at conferences locally and internationally on the role of the IBCLC and in the assessment and management of tongue and lip tie issues in relation to breastfeeding challenges as well as the impact of feeding difficulties on perinatal mental health. She lives in central Illinois with her husband Brian and has four children.
Contributing faculty, Trish Wierenga, FNP-BC, IBCLC, PMH-C, has disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
Mary Franks, MSN, APRN, FNP-C
The division planner has disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
Sarah Campbell
The Director of Development and Academic Affairs has disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
The purpose of NetCE is to provide challenging curricula to assist healthcare professionals to raise their levels of expertise while fulfilling their continuing education requirements, thereby improving the quality of healthcare.
Our contributing faculty members have taken care to ensure that the information and recommendations are accurate and compatible with the standards generally accepted at the time of publication. The publisher disclaims any liability, loss or damage incurred as a consequence, directly or indirectly, of the use and application of any of the contents. Participants are cautioned about the potential risk of using limited knowledge when integrating new techniques into practice.
It is the policy of NetCE not to accept commercial support. Furthermore, commercial interests are prohibited from distributing or providing access to this activity to learners.
Supported browsers for Windows include Microsoft Internet Explorer 9.0 and up, Mozilla Firefox 3.0 and up, Opera 9.0 and up, and Google Chrome. Supported browsers for Macintosh include Safari, Mozilla Firefox 3.0 and up, Opera 9.0 and up, and Google Chrome. Other operating systems and browsers that include complete implementations of ECMAScript edition 3 and CSS 2.0 may work, but are not supported. Supported browsers must utilize the TLS encryption protocol v1.1 or v1.2 in order to connect to pages that require a secured HTTPS connection. TLS v1.0 is not supported.
The role of implicit biases on healthcare outcomes has become a concern, as there is some evidence that implicit biases contribute to health disparities, professionals' attitudes toward and interactions with patients, quality of care, diagnoses, and treatment decisions. This may produce differences in help-seeking, diagnoses, and ultimately treatments and interventions. Implicit biases may also unwittingly produce professional behaviors, attitudes, and interactions that reduce patients' trust and comfort with their provider, leading to earlier termination of visits and/or reduced adherence and follow-up. Disadvantaged groups are marginalized in the healthcare system and vulnerable on multiple levels; health professionals' implicit biases can further exacerbate these existing disadvantages.
Interventions or strategies designed to reduce implicit bias may be categorized as change-based or control-based. Change-based interventions focus on reducing or changing cognitive associations underlying implicit biases. These interventions might include challenging stereotypes. Conversely, control-based interventions involve reducing the effects of the implicit bias on the individual's behaviors. These strategies include increasing awareness of biased thoughts and responses. The two types of interventions are not mutually exclusive and may be used synergistically.
#33354: Breastfeeding
This course is designed for those in the medical profession with interests in perinatal and maternal-child health. It will assist the participant in supporting, protecting, and promoting breastfeeding for optimal infant growth and development. The historical, social, emotional, and political factors involved in breastfeeding will be reviewed. The risks of feeding artificial baby milk and a review of anatomy, physiology, and the uniqueness of human milk will be addressed. Antepartum educational needs, antepartum assessment for lactational ability, and hospital routines that support lactation are considered. The effects of labor and delivery practices on the breastfeeding relationship are covered in detail. Special circumstances, nursing the premature infant, jaundice, and the use of breastfeeding devices will be explained.
Taken alone, the term "breastfeeding" can describe numerous types of breastfeeding behaviors [1]. Therefore, when reviewing research on breastfeeding, it is important to note the author's definition of the term. For example, some researchers may apply study results to the breastfeeding group if any breastfeeding has occurred at any time. The conclusions drawn, therefore, can be incorrect. By identifying the type of feeding as a research variable (Table 1), the occasionally blurred differences between breastfed infants and infants fed artificial baby milk, especially in developed nations, will become abundantly clear.
BREASTFEEDING TERMINOLOGY
| Term | Definition |
|---|---|
| Breastfeeding Definitions | |
| Exclusive breastfeeding | Baby receives only breast milk. Milk can be directly from the breast, expressed, or from a wet-nurse. The infant does not receive solids, water, or other liquids. Exceptions made for oral rehydration solutions, vitamin drops, minerals, and medicines. |
| Almost exclusive breastfeeding | Baby receives all nourishment from the breast except for small amounts of supplements. |
| Breast milk feeding | The mother is separate from the feeding process. Infants are not fed at the breast but receive mother's milk or banked human milk. |
| Partial breastfeeding | Baby receives frequent or regular supplements. Partial breastfeeding can be listed as high (80% breastfeeding), medium (20% to 79% breastfeeding), or low (less than 20% breastfeeding). |
| Stages of Weaning | |
| Complimentary foods | When breast milk no longer provides complete nutrients. |
| Gradual replacement | Replacement of breast milk with other nutrient foods. |
| Total weaning | To stop all breastfeeding. |
| Methods of Total Weaning | |
| Mother-led weaning | Mother decides to wean the baby from the breast. |
| Forced weaning | Outside complication forces rapid weaning. |
| Infant-led weaning | When the baby naturally begins to taper off on nursing by himself/herself. Infant-led weaning is the least stressful for the baby. |
In the field of obstetrics and perinatal care, most research uses the terms "mother" and "father" and "breastfeeding." Throughout this course, the term "mother" and/or "birthing parent" will be used to refer to an individual who has given birth. However, persons of any gender identity and relationship to the infant may give birth and breastfeed or chestfeed. It is important to ask about preferred pronouns and titles and to respect the patient's identity. This is central to patient-centered care; it will help build rapport and improve education efforts. More research is needed in this field and in marginalized populations.
Writings from ancient times clearly spoke of the importance of breastfeeding. Attempts at artificial feeding were made throughout history, but without much success. Breastfeeding rates have historically dropped in periods of social dazzle and lowered moral standards. However, in times when society has been stable and hardworking, breastfeeding has thrived [1].
Before the advent of commercial formula, wet nurses flourished in times and places of sharp class distinctions [4]. The practice of wet nursing was deeply ingrained in ancient cultures. For example, contracts were made between parents and wet nurses in ancient Mesopotamia. In 1800 B.C.E., the Babylonian King, Hammurabi, wrote a set of laws concerning the wet nurse's care of the infant [5]. The practice of wet nursing continued among the aristocracy through the Greek and Roman empires and was probably introduced to Europe during the Roman occupation [5].
Among the wealthy upper class in European countries from 1500 to 1700, the hiring of wet nurses was the norm. When a member of royalty was expecting a child, several wet nurses were kept "on standby." The new heir may have had several wet nurses until weaning. For example, Louis XVI had four wet nurses before he was weaned at the age of 24 months [6].
The expectant mother was responsible for choosing a wet nurse of good temperament and morals. The wet nurse was under the strict supervision of the mother in her home. In return, wet nurses were well cared for and often lived in the same home for years. It was a tremendous employment opportunity for poor women who sought these jobs, and they often neglected, abandoned, or even deliberately suffocated their own infants so they could take a job as wet nurse [6]. While the upper class could pay for wet nurses, the general population resorted to group nursing (i.e., passing the baby around) if the mother was unable to nurse, if she was working in the fields, or if she died [7]. Wet nurses also had a vital function in foundling homes (orphanages), where it has been estimated that they breastfed as often as thirty times a day, producing up to five quarts of milk [6].
Live-in wet nurses went out of fashion for a time, and newborns were sent to live in the wet nurse's home until they were weaned. This "farming out" of newborns removed the child from his or her family and lacked the day-to-day supervisory aspect of the live-in wet nurse. French laws prohibited a wet nurse from breastfeeding more than two infants (besides her own). Separate beds had to be provided for each baby, and the wet nurse was prohibited from sleeping with the infants [1]. In the mid-18th century, it became fashionable again for wealthy Europeans to nurse their own infants. Babies were dying under the care of wet nurses, and there were legitimate concerns that untreatable diseases were being transmitted through breast milk.
Wet nursing was eventually exported to the New World. As early as 1711, colonial newspapers served as a means to locate a wet nurse [6]. In America's pre-Civil War South, privileged children were nursed and cared for by Black wet nurses [4]. The practice of "cross nursing," or wet nursing, the children of close friends or sisters continues in many developing countries when the mother is ill or has died.
The decline in breastfeeding has been traced to the social changes brought about by the industrial revolution [7]. In 1880, 95% of all infants were breastfed for two to four years, but as mothers began reporting breastfeeding difficulties, physicians blamed the stress of urban life and the "bad" human milk produced by urban women. These mothers, who were just learning about the germ theory of disease and anxious to protect their babies, began to gratefully rely on "scientific" food rather than that produced by their own bodies [5,8].
Cultural phenomena have also contributed to a decline in breastfeeding. These include: the loss of community/family knowledge, role models, and support of breastfeeding; the tendency to hand over responsibility for personal health to the medical profession; an increasingly more patronizing attitude toward patients by the medical professions; a greater reliance on institutions, along with their structured environments; and premature application of poorly conceived and poorly tested medical theory [5].
How a society views infants is also thought to affect whether women decide to breastfeed or bottle feed [6]. For example, at the beginning of the 20th century, psychologists and physicians were convinced that babies developed best if they were raised according to hard and fast rules [9]. This led medical and psychological communities to advise the mother that she must gain control over the infant and show the child that she is the master. By imposing arbitrary rules with little physiological basis, these scientific communities sought to regulate the process of breastfeeding [5]. By 1930, however, pediatricians were warning against rigid scheduling that might not correspond with the infant's natural hunger rhythm and could result in prolonged crying and feeding difficulties [5].
Historically, infant feeding has encompassed a wide range of recommendations from the psychology community as well [4,10]:
In 1896, Holt recommended feeding children by the clock.
Freud's views on human sexuality (1856–1939) led to breastfeeding being seen as a sexual, and therefore unhealthy, act.
Watson (1878–1958) believed that suppressing the normal impulses of childhood with rigid schedules would lead to better adult mental health. Infants were to eat, sleep, and even excrete at specified times.
Skinner's views (1904–1990) fostered a detached style of parenting. He recom-mended keeping children in a controlled environment and tried to prove his point by raising his own child in a padded box. (Bumper pads for cribs are thought to be leftovers from "Skinner's box.") Infants were removed from the family bed.
Erikson (1902–1994) felt that trust versus mistrust was the primary developmental task for the first year of life: to develop trust, an infant must learn that his needs are met; if they are not, mistrust develops.
During World War II, women entered the work force in great numbers. They wanted, and often needed, to work, but they worked in industrial settings that did not provide provisions for breastfeeding mothers and babies. The need for human milk substitutes became greater than ever, and companies that mass-produced artificial baby milk (ABM) were started.
As scientific methods improved, everything was reduced to "scientific" fact. Improvements in sterilization techniques made ABM safer. It was calculated to the last calorie, it was measurable, and its production was very much a scientific process. Breastfeeding, on the other hand, was neither scientific nor sterile. It also was not "measurable." ABM was considered "safe," nursing was rejected as unhealthy, and the push was on for "better" babies through "superior" ABMs.
During the 1960s, the ABM industry was in full swing, and ABM was proclaimed the "modern way" to feed a baby. The 1970s "flower children" spawned a back-to-nature movement and a return to breastfeeding, but by the 1980s, it still had not been accepted as the norm. By 1990, only 50% of infants were breastfed at birth, and three-fourths of these infants were weaned by 6 months of age [5].
More than half of the women giving birth today either were not breastfed themselves or were breastfed for only a few weeks or months, often with supplementation beginning at birth. For the last 80 to 90 years, women have been ABM feeding in such great numbers that much of the valuable information once possessed has been (temporarily) lost. During the lifetimes of our mothers and grandmothers—due mostly to profound social changes, but also to smart marketing—bottle feeding became the norm. It is important to work to re-establish the breastfed baby as the norm [11].
Every cultural group holds beliefs about the primary function of a woman's breasts [5]. It is important, therefore, to understand how culture influences the initiation and duration of breastfeeding.
Following World War II, when artificial feeding became a viable option, the cultural context of a woman's breasts began to shift from functional to purely sexual. Photographs in popular magazines began to depict women with large breasts as the ideal in beauty. "Sweater Girls" became popular, and by 1940, the ideal shape of the breast had changed to consist of two pointed cones attached at right angles to the female chest [9]. "Blond Bombshells" were noticeably heavy on top with pencil thin waistlines; movie actresses began to resemble an exaggerated caricature of femininity. The first issue of Playboy appeared in 1953, and its narrow definition of beauty quickly became a part of American culture, which has defined a small range of acceptable breast shapes and sizes [12].
Other western cultural assumptions about female breasts and breastfeeding include [13]:
Breasts are for sex.
Breastfeeding serves only a nutritional function.
Breastfeeding should be limited to very young infants.
Breastfeeding (like sexual activity) is appropriate only when done in private.
Many of the difficulties women experience in breastfeeding may be traced to these four assumptions. For many women, however, the biggest block to breastfeeding may be related to feelings of shame associated with breastfeeding as a result of the concept of the female breast solely as an object of sexual excitement. This shame, coupled with ignorance about women's breasts, has led to emotional pain, confusion, and, in some instances, serious health problems [1,14].
Women also have felt that there is a social stigma attached to nursing an infant beyond 6 months. This stigma has driven many well-educated, caring, dedicated mothers to closet (i.e., concealed) breastfeed, which may adversely affect other children in the family. Children learn about the breast and breastfeeding from their mothers. When a mother nurses away from her older children or when she forbids them to watch the new baby nurse, the children may associate shame or guilt with the act of breastfeeding and may develop shameful feelings about breasts [1].
To protect breastfeeding parents from harassment, states have passed laws that specifically address breastfeeding [15]. As of 2022, all 50 states have passed legislation specifically allowing women to breastfeed in any public or private location, and 31 states exempt breastfeeding from public indecency laws. Thirty states have passed laws related to breastfeeding in the workplace; 22 states exempt breastfeeding mothers from jury duty or allow jury service to be postponed; and 4 states (California, Illinois, Minnesota, Missouri) have implemented or encouraged the development of a breastfeeding awareness education campaign [15,16].
In addition to legislation at the state level, federal legislation related to breastfeeding also has been introduced and/or passed [15,17]:
The Providing Urgent Medical Protections (PUMP) for Nursing Mothers Act went into effect on December 29, 2022. This allows for improved break time protections of nursing parents in all sizes of workplace, and protections apply regardless of gender. This is improved from the Breaktime for Nursing Mothers law in multiple ways, including the ability for parents to file a lawsuit against employers that do not abide by the laws and clarifies that if an employee is "not completely relieved from their work duties" during their pump break, the pumping time counts in their time worked for calculating minimum wage and overtime hours.
The Fairness for Breastfeeding Mothers Act of 2019 was passed in July 2019. The law requires certain public buildings to provide a shielded, hygienic space (other than a bathroom) that contains a chair, working surface, and an electrical outlet for use by members of the public to express breast milk.
Section 4207 of the Patient Protection and Affordable Care Act of 2010 to amend the Fair Labor Standards Act (FLSA) of 1938; it requires employers to provide reasonable break time for a new mother to express breast milk for her nursing child.
Breastfeeding Promotion Act of 2009 to amend the Civil Rights Act of 1964 to protect breastfeeding by new mothers; to provide for a performance standard for breast pumps; and to provide tax incentives to encourage breastfeeding.
Exemplary Breastfeeding Support Act to amend the Child Nutrition Act of 1966 to promote and support breastfeeding.
Breastfeeding is perhaps the most misunderstood aspect of motherhood. A persistent lack of basic breastfeeding knowledge, the depth and strength of "old wives' tales," and an abundance of misinformation has contributed to this misunderstanding. Cultural, social, and medical interventions have modified childcare practices and made it difficult to see what nature intended [18]. Belief in the miracle of our innate physiology has been lost in western societies. By becoming so technical, we have lost respect and reverence for nature and natural processes [19].
The American Academy of Pediatrics (AAP) revised policy statement Breastfeeding and the Use of Human Milk has established principles to guide pediatricians and other healthcare professionals in assisting women and children in the initiation and maintenance of breastfeeding [20]. It also has recognized the breastfed infant as the standard against which all research and recommendations for infant feeding should be made. This is a paradigm shift in American scientific thinking. The AAP has recommended exclusive breastfeeding for the infant's first 6 months of life, followed by continued breastfeeding for two years or longer, in alignment with the WHO recommendations, as complementary foods are introduced. It also has recommended that [20,21,22,291]:
Parents be provided with complete, current information on the benefits and techniques of breastfeeding to ensure that decisions about feeding are fully informed
Peripartum policies and practices be developed to optimize the initiation and maintenance of breastfeeding
Healthy infants be placed in direct (skin-to-skin) contact with their mothers immediately after delivery and until the first feeding has been accomplished
Mother and infant sleep in proximity to one another to facilitate breastfeeding
Supplements (e.g., water, glucose water, formula, other fluids) not be given to breastfeeding infants unless medically indicated and ordered by a physician
Pacifiers not be used during the initiation of breastfeeding and then used only after breastfeeding is well established
Mothers be encouraged to have at least 8 to 12 feedings at the breast every 24 hours when the infant shows early signs of hunger (e.g., alertness, physical activity, mouthing, rooting)
Breastfeeding newborn infants be visited by a pediatrician or other healthcare professional by 3 to 5 days of age, and again at 2 to 3 weeks of age
All exclusively and partially breastfed infants receive 400 IU of oral vitamin D drops daily beginning at hospital discharge and continuing throughout breastfeeding. Alternatively, the breastfeeding parent can supplement themselves with 6,400 IU of vitamin D.
Supplementary fluoride not be provided during the first 6 months of life (or at all if local water contains fluoride)
Direct breastfeeding be maintained, if at all possible, should hospitalization of the mother or infant be necessary; if not possible, pumping and feeding expressed milk should be encouraged
Pasteurized human donor may be an option in the NICU and for at-risk infants
Changes in policy to addresses the impact of systemic racism, inequities, and structural barriers on breastfeeding outcomes
Support for skilled lactation support, workplace accommodations, and paid leave
The AAP policy statement has incorporated ways in which pediatricians can enthusiastically promote and protect breastfeeding in their individual practices, as well as in the hospital, medical school, community, and nation [20,21]. Promotion of breastfeeding by healthcare providers may help to: increase its support within families, by employers, and in society; halt the media's promotion of artificial feeding; and achieve stricter marketing regulations of ABM companies.
Infant mortality has been associated with a variety of factors, including maternal health, quality of and access to medical care, socioeconomic conditions, and public health practices. It is clearly an important indicator of the health of a nation. The infant mortality rate in the United States generally declined throughout the 20th century, from approximately 100 infant deaths per 1,000 live births in 1900, to 5.6 infant deaths per 1,000 live births in 2019 [23]. However, the U.S. infant mortality rate did not decline significantly between 2000 and 2018, which represented the first period of sustained lack of decline since the 1950s. The Healthy People 2030 initiative has set a target goal of 5.0 infant deaths per 1,000 live births [23]. In 2013, the United States ranked 51st internationally in infant mortality, ranking much higher than other countries of similar gross domestic product [24,25,26]. An international comparison compiled by the Organization for Economic Cooperation and Development (OECD) found that in 2023, the United States ranked 33rd out of 38 member countries with 5.4 deaths per 1,000 live births [27].
These statistics have generated concern among researchers and policy makers. All major medical societies in the United States, including the AAP, have recommended breastfeeding for as long as possible in infancy in order to improve childhood survival rates. More specifically, in 2022, the AAP increased its recommendation from at least one year of breastfeeding to two years or longer in accordance with the WHO [291]. The World Health Organization (WHO) and the United Nations Children's Fund (UNICEF) guidelines have recommended extending breastfeeding for a minimum of two years. This recommendation is supported by research, which has shown a 21% decrease in U.S. infant mortality in breastfed infants [21]. Exclusive breastfeeding, or human milk feeding for a minimum of six months, provides the greatest level of protection against infantile illnesses.
An objective of Healthy People 2030 is to increase the proportion of U.S. mothers who breastfeed their babies through 6 months of age and who continue to breastfeed their babies through 12 months of age. A review of Healthy People 2030 reported the following progress toward achieving this objective [28]:
24.9% of mothers exclusively breastfeed their babies through 6 months of age (target: 42.4%)
35.9% of mothers breastfeed their babies through 12 months of age (target: 54.1%)
The Healthy People 2030 breastfeeding objective also includes an increase in exclusive breastfeeding. To monitor progress toward achieving the objectives, the Centers for Disease Control and Prevention (CDC) analyzed data from the National Immunization Survey (NIS) for infants born from 2011 to 2018. According to the analysis, among infants born in 2018, the rates for exclusive breastfeeding through three months and six months were 46.3% and 25.8%, respectively [29]. Rates of exclusive breastfeeding are reported to be significantly lower among Black infants (compared with White infants), infants born to young (20 to 29 years of age) unmarried mothers (compared with married mothers), and infants born in rural areas, particularly infants living in the Southeast [29].
Since the Surgeon General's Workshop on Breastfeeding and Human Lactation in 1984, all maternity settings have been mandated to provide a supportive milieu for breastfeeding. At the nucleus of this mandated support is a strategy of breastfeeding education beginning in the antepartum setting and continuing through the pediatric care of the infant. Hospitals are to have a specially trained person available to help the breastfeeding dyad. By improving the perinatal setting, it was hoped that, by the year 2000, at least 75% of all women would be nursing their newborns at hospital discharge and 35% would continue to breastfeed for a minimum of six months [30,31]. The key elements of the Surgeon General's Workshop (Table 2) were incorporated into the Healthy People 2000, 2010, and 2020 and restated in Healthy People 2030.
KEY ELEMENTS FOR THE PROMOTION OF BREASTFEEDING
| Primary Care Settings |
|
Key elements for women of childbearing age include:
|
| Prenatal Care Settings |
|
Key elements for women of childbearing age include:
|
| In-Hospital Settings |
|
Key elements for women of childbearing age include:
|
| Postpartum Ambulatory Care Settings |
|
Key elements for women of childbearing age include:
|
The workshop represented a milestone in the effort to highlight breastfeeding as a public health priority. Since its occurrence, many professional health agencies and organizations (e.g., the Agency for Healthcare Research and Quality, the American Academy of Pediatrics, American Academy of Family Physicians, American College of Obstetricians and Gynecologists, American College of Nurse-Midwives, American Dietetic Association, and American Public Health Association) have reached a consensus on breastfeeding recommendations. For example, these agencies and organizations have recommended that most infants be breastfed for at least one to two years, or as long as the dyad desires. They also have recommended that for the first 6 months, infants be exclusively breastfed (i.e., they do not receive any foods or liquids other than breast milk). More than 50 health professional and educational organizations, as well as federal agencies, now participate in the U.S. Breastfeeding Committee, whose mission is "to improve the nation's health by working collaboratively to protect, promote, and support breastfeeding" [32].
Breastfeeding education for clinicians also has received national attention. In 2000, the Office of the Surgeon General published the Blueprint for Action on Breastfeeding, which represented the first comprehensive framework on breastfeeding for the nation. Among other things, the blueprint recommended action steps for healthcare systems, families, communities, researchers, and employers to promote breastfeeding [33,34]. From 2004 to 2006, the U.S. Department of Health and Human Services (HHS) sponsored a national campaign to increase awareness about the health advantages of breastfeeding. A 2007 national survey (the first of its kind) identified key opportunities for birth facilities and states to more effectively support new mothers in the initiation of breastfeeding. Evidence-based staff training on breastfeeding was one area of need identified that hospitals and birth centers could implement to improve their support of breastfeeding. Workplace support was another area of need identified; women who are employed full-time are less likely to initiate and continue breastfeeding after returning to work [32,35,36,37]. In 2011, the Office of the Surgeon General published The Surgeon General's Call to Action to Support Breastfeeding, with the goals of increasing public health impact, reducing inequities in health care quality for mothers and infants, and improving support for families in job and community settings. The Call to Action discusses the importance of breastfeeding, rates of breastfeeding in the United States, barriers to breastfeeding, and public health approaches to increase breastfeeding [38,39].
In 1991, the WHO and UNICEF launched the Baby-Friendly Hospital Initiative (BFHI), which was designed to create change in healthcare facilities. The aim of the BFHI was to foster an environment that supports, protects, and promotes breastfeeding, as indicated by the WHO's International Code of Marketing of Breast-milk Substitutes [40,41]. The process for obtaining certification as a Baby-Friendly Hospital has been established by UNICEF in collaboration with national governments worldwide. UNICEF's Ten Steps to Successful Breastfeeding has been the basis for evaluating hospitals and healthcare facilities for participation as a designated baby-friendly facility (Table 3). A 2000 statement by the United Nations (UN) regarding the "rights of the child" has been linked to the BFHI. The UN has stated that for hospitals with maternity units to be designated "child friendly" they must first fully implement the Ten Steps to Successful Breastfeeding and be accredited by WHO/UNICEF as baby friendly [42]. The Ten Steps to Successful Breastfeeding and the BFHI have been shown to be effective measures to increase breastfeeding initiation, duration, and exclusivity [43,44].
UNICEF'S TEN STEPS TO SUCCESSFUL BREASTFEEDING
|
More than 21,000 facilities, or 27.5% of maternity wards worldwide, have achieved baby-friendly status; in the United States, 551 hospitals and birthing centers in all 50 states have achieved this status [46,47]. Hospitals in the United States and elsewhere that have used the BFHI framework, in whole or in part, have demonstrated an extended positive impact on breastfeeding rates and duration [48,49,50,51,52].
The BFHI and the WHO's International Code of Marketing of Breast-milk Substitutes can and should be carried out in the United States, but it will take more time than initially anticipated. Although the baby-friendly evaluation clearly exposes the wide scope of hospital routines that have detrimental effects on successful breastfeeding, achieving change can be difficult. Some routines are so deeply ingrained that staff members may be unable to identify and correct problems. Ambiguous objectives, poorly defined strategies, and the use of a limited number of change agents and techniques additionally make implementing change difficult. What is needed is a structured approach that incorporates training, motivation, and specific practices for implementation [2]. A two- or three-day course that includes practical sessions and counseling skills has been shown to be effective in changing hospital practices, knowledge of healthcare workers, and breastfeeding rates [53]. Ongoing support and education should be provided by individuals with advanced knowledge and training, such as lactation consultants.
Studies have found that healthcare providers represent one of the major barriers to successful breastfeeding [54]. This finding is supported by mothers who have said that they would breastfeed for as long as their physicians said if the physicians stressed to them the importance of breastfeeding [1]. Many women have reported not receiving positive breastfeeding messages from health caregivers and hospital staff. This perceived "neutral attitude" has been associated with mothers not breastfeeding beyond 6 weeks [55]. The language healthcare professionals use to educate women about breastfeeding often is riddled with semantics that place artificial feeding and breastfeeding on the same plane, with only a slight difference in the method of delivery. To support the idea that breastfeeding is the norm, and that artificial feeding can result in adverse effects on infant health, healthcare providers should select and use language that supports breastfeeding [2].
Healthcare professionals should also recognize the negative impact on successful breastfeeding when they agree to display materials produced by ABM manufacturers. The conflict of interest presented when ABM companies "advise" women about breastfeeding should be obvious [56]. U.S. military hospitals do not accept free ABM or other perks because they consider such practices unethical [48]. Giving a new mother ABM samples at hospital discharge may undermine her confidence in her ability to breastfeed. Items such as discharge packs, diaper bags with ABM samples, videos, and discount coupons have been repeatedly shown to decrease the length of exclusive breastfeeding and are in direct violation of the WHO's International Code of Marketing of Breast-milk Substitutes, which was adopted by the World Health Assembly to reverse the decline in breastfeeding related to the production of manufactured breast milk substitutes [40,41]. In recognition of these effects on new mothers, Rhode Island (2011), Massachusetts (2012), Delaware (2015), Maryland (2015), New Hampshire (2016), and West Virginia (2018) banned the inclusion of infant formula in facility-provided gift bags [57,58,59]. Other cities and states are beginning to address this issue as well.
While national initiatives and recommendations from professional organizations may encourage more women to breastfeed, improvement in breastfeeding duration cannot be achieved if healthcare providers lack knowledge about breastfeeding [1]. Clinical lactation skills may be taught by an experienced clinician to perinatal providers in the clinical setting and subsequently to new mothers [56]. Nurses who work with families with children often are responsible for a majority of patient education. In this role, they may significantly affect a woman's decision to initiate and continue breastfeeding. Improving evidence-based practices in nursing programs has been shown to positively impact breastfeeding outcomes both in the hospital and in the community [60].
In addition to a lack of knowledge about breastfeeding among healthcare professionals, breastfeeding research has traditionally been a low priority for federal funding. For example, between 1994 and 1996, federal research funds totaled approximately $40.4 million, and of this amount, only $5.6 million (13.7%) was awarded to projects determined to have an impact on achieving federal goals for increasing the incidence and duration of breastfeeding. A total of 27 (7.5%) funded projects in this category, reflecting $4.1 million, had no relationship to breastfeeding per se; they involved the use of human milk composition and technologies to improve artificial milks and develop new pharmaceuticals and therapies [61]. However, the CDC has begun working with researchers to evaluate the impact of existing breastfeeding interventions across the United States. As of 2005, 10 universities had received funding to evaluate [62]:
Peer-counseling for breastfeeding education
In-home breastfeeding support
Telephone-based breastfeeding support
Hospital-based paraprofessional lactation clinic support
How breast pump loan programs affect breastfeeding duration among women who return to work after maternity leave
How "baby-friendly" hospital status affects breastfeeding initiation rates
How community-based breastfeeding policy and environmental interventions affect local breastfeeding rates
The cost-effectiveness of a variety of methods used to support the breastfeeding mother
In 2014, the U.S. Department of Agriculture released nearly $60 million in fiscal funds to its Special Supplemental Nutrition Program for Women, Infants, and Children (WIC). The WIC program supports 53% of infants born in the United States and promotes breastfeeding as the best form of nutrition for infants [63]. The National Institute of Child Health and Human Development supports and conducts research on breastfeeding and breast milk through several of its organizational units. The Institute also supports efforts to meet the public health goals outlined in the Healthy People 2030 initiative [64].
For a variety of reasons, women throughout history have sought alternatives to breastfeeding. Early attempts focused on feeding vessels and nutritional sources. The milk of other mammals was most commonly used; however, infant mortality was very high regardless of the nutrient source. Mothers who could not breastfeed and could not afford a wet nurse might prepare food for their babies for the purpose of "dry nursing." These foods typically included beer, gruel, and pastes made from bread and water [65]. Cows' milk became a popular substitute for breast milk in the early 19th century; however, because of its higher protein and lower carbohydrate content, it was more difficult for infants to digest [44]. Physicians then began recommending diluted cows' milk. Sugar or cereals were added to these diluted preparations when it was discovered that they lacked sufficient caloric density to support adequate growth. Infant mortality remained high, however, and by the end of the 19th century, a multitude of mathematical "formulas" had been developed to calculate an infant's caloric needs [65].
At the beginning of the 20th century, pasteurization and electric refrigeration led to a significant decline in infant mortality, and formula feeding became increasingly popular. Further advances to improve the digestibility of cow-milk protein and the direct marketing of new formulas to physicians influenced their popularity. With the arrival of evaporated canned milk, the need for fresh milk diminished. Evaporated milk formulas, which became popular during the 1930s and 1940s, were eventually replaced by modified milk formulas (to which water was added) and then by ready-to-feed formulas [65].
ABM has come a long way from the homemade recipes of generations ago. However, while scientists and chemists have continued to pursue the ingredients that will bring the composition of ABM closer to that of human milk, ABM still lacks a great deal. For example, there are 130 unique oligosaccharides (sugars) present in human milk that are not present in ABM. Even with manufacturers' addition of some oligosaccharides to ABM formulas, the majority are still missing. The latest additions to ABM have been various forms of essential fatty acids (EFAs), such as docosahexaenoic acid (DHA) and arachidonic acid (ARA). EFAs are considered essential for proper growth and development, especially during pregnancy and early childhood [2]. They are major components of cell membranes and are of special importance to brain tissue, retinal tissue, and blood vessels. They also are part of the substrate needed to make hormones. The omega-3 fatty acids are not prevalent in the adult food supply except in diets rich in fish oils. However, during pregnancy, EFAs are made in the mother's body and delivered through the placenta. After birth they present themselves in the mother's milk. Maternal diet can affect DHA levels, and pregnant and lactating women often take DHA supplements [66,67]. Studies have shown a link between higher DHA levels in breast milk and in utero and benefits to infant health [66]. Although the recommended dietary intake of EFAs for pregnant and lactating women has not yet been established in the United States, consensus guidelines recommend a minimum of 200 mg per day [68]. A woman can achieve this threshold by consuming one to two servings of seafood per week; however, studies indicate that pregnant and lactating women in the United States only consume an average of 60–80 mg per day [69,70,71,72].
Because EFAs such as DHA and ARA are naturally present in human breast milk, their addition to infant formulas has been thought to be beneficial. However, some infant formulas include DHA- and ARA-containing oils that are novel foods extracted from a variety of sources (e.g., genetically engineered algae and fungi) and are structurally different from the DHA and ARA found in human milk. Although ABM manufacturers in the United States have been fortifying their products with DHA and ARA since 2002 (with the U.S. Food and Drug Administration [FDA] approval) and marketing them as "closest to human milk," none of the studies conducted on the addition of DHA and ARA to infant formulas have reached a consensus regarding any benefit to infant development [73; 74]. The AAP Committee on Nutrition has urged caution regarding the addition of DHA and ARA to infant formulas [75]. Additionally, the National Alliance for Breastfeeding Advocacy and the Cornucopia Institute have filed a petition with the FDA requesting a warning label on infant formulas that contain DHA and ARA [2].
It is important to note that FDA approval does not constitute affirmation of the safety of new ingredients added to infant formulas. In the United States, a manufacturer may declare that an ingredient is generally recognized as safe (GRAS) if scientific consensus exists among qualified experts as to the ingredient's safety under the conditions of its intended use. The manufacturer then notifies the FDA, which issues a "letter of no objection" if it has no questions [76]. The FDA has expectations that formula manufacturers will independently pursue post-marketing scientific studies and surveillance of these additives [75].
Application of the GRAS process, however, does not address all concerns about the chemical characteristics, nutritional contributions, and pharmacologic and physiologic activities for the multitude of potential new ingredients in infant formulas. Consequently, the Institute of Medicine (IOM) Food and Nutrition Board has proposed additional safeguards, not required for other foods, for infant formulas to which new ingredients are added. The proposed rule would mandate that manufacturers demonstrate that the formula can sustain infant growth and development over 120 days (the period during which formula is likely to be the sole source of infant nutrition). The IOM also has recommended that data from growth/development studies be submitted as part of material demonstrating safety [76].
Research has indicated that women who were educated regarding the risks of not breastfeeding were more likely to commit to breastfeeding compared to women who were only given information regarding the benefits of breastfeeding [77]. Therefore, it is vital that healthcare professionals include information regarding the risks and disadvantages of ABM in addition to the benefits of breastfeeding when counseling mothers.
ABM carries inherent dangers (e.g., product contamination) that have not been associated with breast milk [78,79,80]. Many of these dangers, such as product recalls, may not be known to the women giving ABM to their infants [81]. Between 1982 and 1994, more than twenty ABM recalls occurred in the United States [82,83]. In February 2022, the FDA issued a recall for three brands of powdered infant formula manufactured at a facility in Michigan [84]. Five infants who received the formulas developed Cronobacter sakazakii and Salmonella Newport infections; two of the five infants died. Based on inspections conducted at the facility, the FDA found that the manufacturer did not have a system of controls for all stages of processing to ensure that the formulas would not become adulterated, and did not ensure that surfaces in contact with the formula were protected from contamination [84]. Worldwide, more than 10 million children die each year before their fifth birthday; approximately 50% of these deaths have been associated with nutrition problems. The use of infant formula tends to increase the illnesses and deaths that have been associated with a wide range of diseases. The risk of mortality has been shown to be about 27% higher among infants who have never been breastfed compared to breastfed infants [20].
Known risks associated with ABM include [1,5,20,82,85,86,87,88]:
Increased gastrointestinal illness (e.g., vomiting, diarrhea, dehydration)
Increased respiratory illnesses (e.g., pneumonia, asthma, respiratory syncytial virus)
Otitis media (four times more prevalent in the ABM-fed infant)
Increased likelihood of childhood obesity
Increased risk of tooth decay (nursing bottle caries)
Risk of contaminated formula (either at the factory or at home) and ingestion of "allowable" amounts of insect parts, rat hairs, droppings, iron filings, and accidental excesses of chlorine and aluminum
Severe illness resulting from improper dilution or home additives
Increased allergies ranging from skin rashes to asthma
Increased risk of immune system disorders, such as:
Accelerates the development of celiac disease
Risk factor in adult onset of Crohn disease, ulcerative colitis, and rheumatoid arthritis
Risk factor (2% to 26%) in childhood onset insulin-dependent diabetes mellitus
Five- to eightfold risk of developing lymphomas in children younger than 15 years of age
May impair effectiveness of vaccines
Twentyfold increase in necrotizing enterocolitis (NEC)
Increased risk of sudden infant death syndrome (SIDS)
Cost is a unique disadvantage of ABM. For example, the average family will spend approximately $150 every month for the first year if using ABM. If a specialized ABM is required, the cost may be as high as $300 per month, or as much as $3,900 for the first year [89,90]. Additional out-of-pocket expenses may include medical care due to the higher incidence of ear infections, gastrointestinal illness, and asthma in ABM-fed babies. One study found that if 90% of mothers complied with recommendations to breastfeed exclusively for six months, the U.S. economy could save $3.7 billion in direct and indirect pediatric health costs and $10.1 billion in premature death from pediatric disease [91,92]. Parents may lose wages if they miss work to care for a sick child. Other costs may not be as easily measured. For example, bottles must be bought and washed. ABM must be prepared and warmed before use. All these activities take time. Also, because the mother's fertility typically returns sooner if she bottle feeds, additional monies will be needed for birth control methods. A subsequent pregnancy that occurs sooner than planned may put additional financial burdens on the family.
ABM also is not a renewable resource. The cows that produce the milk for ABM must be fed. Growing food for cattle or soybeans for ABM uses large amounts of water and pollutes the environment with fertilizer and pesticide residues. Production uses fuel to power manufacturing. ABM also leaves solid waste products. For example, it has been estimated that 450 million tin cans are discarded each year for every 3 million bottle-fed babies [5].
When compared to the risks associated with ABM, the advantages to its use may seem insignificant. However, many mothers opt to bottle feed their infants. The underlying motives for the decision to bottle feed may include [9]:
A feeling that breastfeeding is repulsive or lacks appeal
A sense that breastfeeding is embarrassing
A busy lifestyle or feeling that breastfeeding will tie her down
A sense that she is too nervous to breastfeed
A perception that breastfeeding is complicated or restrictive
A partner who expresses jealousy
A concern that she will not be able to measure the baby's intake
Proper education may help to alleviate many of these concerns.
The ABM companies are powerful, and their marketing tactics can be subtle but effective. A history of collusion between physicians and the ABM industry has been documented, and the media has reported that federal health officials yielded to pressure from ABM industry officials by weakening an advertising campaign that was designed to promote breastfeeding [93]. ABM companies openly give hundreds of thousands of dollars to medical professionals, sponsor medical seminars and research, and provide massive amounts of free ABM to hospitals every year [83].
From 2012 to 2013, the global market for functional and fortified foods grew significantly, with infant formula topping the list. In 2013, nearly $5 billion in global sales of infant formulas were reported, with sales expected to grow at an annual rate of 11% through 2018 [94]. The revenue from this market is projected to increase to more than $319 billion in 2022 [95]. ABM marketers are specifically and aggressively targeting Asian, African, and Middle Eastern markets in order to compensate for the slower European and American infant-formula markets [94].
The U.S. Government Accountability Office (GAO) has reported that breastfeeding rates are particularly low among infants whose mothers participate in WIC, which provides free food and infant formula to low-income women, infants, and children. Approximately half of all infants born in the United States each year participate in the WIC program [96,97]. While ABM manufacturers generally agree that breastfeeding is best, they continue to market ABM as an "alternative" for mothers who do not exclusively breastfeed. Some ABM manufacturers have used the trademarked WIC acronym in their promotional materials, despite federal restrictions on this practice [96,98].
There are no biological disadvantages to breastfeeding, but there may be perceived barriers or challenges that can be mitigated with improved education, policies, and support.
Some of the advantages and benefits of breastfeeding include [2,77,92,99,100,101,102,103,104,105,106,107,108,109,291]:
Adjusts to the baby's needs
Contains appropriate nutrients; no need for supplemental vitamins besides vitamin D for all breast milk-fed infants and iron supplements for preterm infants
Provides nutritional factors for optimal growth and development
Provides immune factors
Protects against allergies in baby
Assists with normal jaw, tooth, and speech development
Always available, at the right temperature, and in the right amount
Provides closeness and enhances bonding
Produces milder odor to baby's stools, compared to the artificially fed infant
Protects against colic
Increases maternal postpartum weight loss, compared to artificial feeding
Improves maternal health by increasing the level of maternal circulating antibodies
Protects women who breastfeed against the development of cancer (breast and ovarian) and cardiovascular diseases later in life
Saves potentially millions in healthcare dollars. For example, the WIC program purchases $2.6 billion of ABM each year using taxpayer monies. Infant formula is the single most expensive food item for WIC. Twelve weeks of exclusive breastfeeding could result in a net healthcare savings of almost $3.9 billion annually in the United States.
Improves utilization of healthcare resources (Table 4)
Prevents multiple food allergies. As breastfeeding rates have declined, protein-related allergy, asthma, atrophic dermatitis, otitis media, and gastroesophageal reflux have become more prevalent in early childhood populations. Evidence has suggested that while the population allergic to cows' milk proteins remains steady at about 2% to 5%, there has been an increasing prevalence of multiple food allergies.
Economical for individual families, particularly when compared to the cost of ABM
THE COST OF NOT BREASTFEEDING
|
The potential healthcare cost savings from breastfeeding in four medical diagnoses were analyzed. Results showed the following potential savings in healthcare costs:
|
Breastfeeding has also been associated with a lower prevalence of hypertension, diabetes, hyperlipidemia, and cardiovascular disease among postmenopausal women.
As stated, infant formula is the single most expensive food item for WIC. To reduce costs, federal law requires that WIC state agencies enter into cost-containment contracts for the purchase of the infant formula used in the WIC program [109]. Cost containment is achieved in the form of rebates from formula manufacturers for each container of formula purchased by WIC. In exchange for the rebates, the WIC state agency agrees to issue the selected manufacturer's infant formula(s) as the "formula of first choice" to its infant participants [109].
Many medical professionals have been hesitant to take anything but a neutral position when discussing infant feeding options. However, parents should hear all the data, including the risk/benefit ratios of the various infant feeding methods, so they may make an informed decision. Healthcare professionals have an ethical responsibility to ensure that parents are aware of the health consequences of their infant feeding decisions [32]. Healthcare professionals should provide the information necessary for parents to make informed decisions and should support and respect their decisions.
The more that breast milk has been studied, the more apparent it has become that human milk is unique. Fresh breast milk is a living fluid composed of more than 200 known ingredients, including vitamins, minerals, trace elements, protein, fat, and carbohydrates. It contains 4,000 live cells (mostly leukocytes) per milliliter, which cannot be replaced by artificial feeding. Breast milk is a highly complex fluid with a balance of nutrients and an array of functional properties that promote a level of metabolic efficiency not attainable from formula based on cows' milk [110]. Breast milk also offers antibody protection (i.e., the infant will be protected through passive immunity from any diseases for which the mother has developed antibodies). This protection continues as long as the baby is breastfeeding.
Although breast milk is not uniform, variations in composition are functional, not random [10]. Its constituents also are multifunctional and interactive. Breast milk also contains "species specific" nutrients. When an infant suckles at the breast, an outpouring of 19 different gastrointestinal hormones occurs in both the mother and the infant. These hormones stimulate growth of intestinal villi, increasing the surface area and the absorption of calories with each feeding [111]. Breast milk also contains ingredients, which are not present in ABM, that provide for improved brain growth [112]. The optimum development of the brain and nervous system requires the presence of a precise balance of specific amino acids, fats, simple sugars, salts, and minerals [1]. A study of magnetic resonance imaging brain scans found that by 2 years of age, children who had been breastfed exclusively for at least three months had enhanced development in key parts of the brain (i.e., those associated with language, emotional function, cognition) compared to children who were fed formula exclusively or who were fed a combination of formula and breast milk [113].
Breast milk production requires very little maternal energy; the process is about 95% efficient [1]. As study methods have improved, it has become possible to look at the intricate components and variations of breast milk throughout the nursing experience.
Breast milk contains many non-nutritive, bioactive substances that directly affect infant physiology. Breast milk is not a uniform body fluid but a mammary gland secretion of changing composition [1]. No two samples of breast milk are the same, even when taken from the same mother. Because breastfeeding is an interactive process, the infant helps to determine the milk's composition. The major components of human milk include protein, fat (lipids), and lactose. Additional components include vitamins, certain minerals, hormones, enzymes, growth factors, and many types of protective agents (Table 5) [2].
COMPOSITION OF TERM AND PRETERM MILK DURING FIRST MONTH OF LACTATION
| Nutrients | 3 to 5 Days | 8 to 11 Days | 15 to 18 Days | 26 to 29 Days | ||||
|---|---|---|---|---|---|---|---|---|
| Full Term | Preterm | Full Term | Preterm | Full Term | Preterm | Full Term | Preterm | |
| Energy (kcal/dL) | 48 | 58 | 59 | 71 | 62 | 71 | 62 | 70 |
| Lipid (g/dL) | 1.85 | 3.00 | 2.9 | 4.14 | 3.06 | 4.33 | 3.05 | 4.09 |
| Protein (g/dL) | 1.87 | 2.10 | 1.7 | 1.86 | 1.52 | 1.71 | 1.29 | 1.41 |
| Lactose (g/dL) | 5.14 | 5.04 | 5.98 | 5.55 | 6.00 | 5.63 | 6.51 | 5.97 |
The proteins in human milk are specific to human mammary production and are not found elsewhere in nature. Protein synthesis is under the genetic control of ribonucleic acid [1]. Breast milk composition is relatively stable throughout the world [2]. Cows' milk proteins and proteins from other sources are different in structure, quantity, and quality and can cause allergic responses [10].
Mature breast milk is approximately 0.8% to 0.9% protein and provides the infant's protein requirements in a way that changes as the infant matures. Some human milk protein is not nutritionally available but serves immunologic needs. The protein content in colostrum is relatively high. The level declines as milk matures and stabilizes by the end of the third month. The protein levels in human milk are more than adequate for optimal growth and provide an appropriately low renal solute load for the baby [2,10].
Human milk contains casein and whey protein. These levels change to meet the infant's nutritional needs as lactation progresses. For example, the level of casein is lower in early lactation but increases rapidly. Whey proteins are highest in early lactation and then continually fall. The whey/casein ratio of human milk is about 90:10 in early lactation, 60:40 in mature milk, and about 50:50 in late lactation [2]. Whey proteins are acidified in the stomach and are quickly digested, supplying a continuous flow of nutrients to the infant. Caseins form a tougher, less digestible curd that requires a high expenditure of energy for an incomplete digestive process. Caseins are the primary protein in untreated bovine milk [2].
Lipids provide 50% of the energy content in human milk and are its most variable component. The fat content of mature milk is approximately 3.8%. Fat content varies from one mother to another, from early to late lactation, from feeding to feeding, and within individual feedings. The total fat content varies from 22 g/L to 62 g/L and is independent of the frequency of breastfeeding. Maternal diet affects the constituents of the lipids but not the total fat content. When a mother's caloric intake is poor, fat is mobilized from maternal fat stores (primarily in the hips and thighs) [2]. The cholesterol level of breast milk remains constant despite manipulation of the mother's cholesterol intake. The lipid fraction of human milk provides essential fatty acids, which are important to proper brain growth. Tissues of breastfed and ABM-fed infants have demonstrated distinctly different plasma fatty acid compositions. Levels of fatty acids in lactating women have been shown to be low, suggesting that their transfer to breast milk occurs at the expense of the maternal stores [2].
Lipase in human milk complements the low level of pancreatic lipase in infants, especially premature infants. When human milk is frozen or refrigerated, lipase is not affected, but heating severely reduces its activity [2]. Lipase activity is stable at a pH level of 3.5 at 37°C for one hour, which is just long enough for effective fat digestion at the level of the infant's small intestine [110].
Lactose is a sugar present only in milk. In human milk, the level of lactose is quite high. Other sugars are present, but lactose is the driver sugar in breast milk and provides approximately 50% of the caloric content. Lactose promotes the growth of Lactobacillus bifidus flora in the infant bowel, increasing intestinal activity and stemming the growth of pathogens [2]. L. bifidus limits colonization by other bacteria by occupying the limited number of binding sites along the intestinal wall.
Lactose enhances infant absorption of calcium from breast milk and readily metabolizes to galactose and glucose, which supply energy to the rapidly growing infant brain [2]. Alpha-lactalbumin is the highest concentration protein in human milk and is required for lactose synthesis. Lactose is responsible in part for milk volume; its concentration is affected by maternal diet [2]. Excessive use of sugar substitutes may affect maternal milk volume [1].
The enzyme lactase is present in the infant's intestinal mucosa from birth and is necessary to convert lactose into simple sugars that may be easily assimilated by the infant. There has been concern regarding lactose intolerance in infants, primarily fueled by a new lactose-free ABM. Lactose intolerance is common in many mammals as they age, primarily as a result of diminishing activity of intestinal lactase after weaning. Congenital or primary lactase deficiency is, however, exceedingly rare [2].
The mineral content of milk is also species specific. The type and amount of minerals present in milk reflect the growth rate and bone density of the offspring [1]. The mineral content of cow or elephant milk, therefore, is higher than in human milk because of the animal's larger bone mass. The total mineral content in human milk remains fairly constant, with the highest concentration of minerals in the first few days following birth. Mineral concentration then decreases slightly in a consistent pattern throughout lactation with little diurnal variation or variation within feedings [2].
Sodium levels in cows' milk-based ABM are more than three times that in human milk. The sodium level in breast milk is high in early colostrum but falls dramatically by the third day postpartum. Elevated sodium levels in breast milk occur during weaning, in women with mastitis, and during the first months of gestation. Even in infants, high sodium intakes can lead to hypertension. Persistently high sodium levels may indicate impaired lactation [2].
The iron in breast milk is 49% available, whereas only 4% of iron in iron-fortified ABM is absorbed. Breastfed infants are rarely at risk for iron deficiency anemia even though human milk contains a relatively small amount of iron. Breastfed infants generally are sustained by iron stores that are laid down in utero. The high lactose and vitamin C levels in human milk facilitate the infant's ability to absorb iron, which occurs approximately five times more efficiently than a similar amount of iron from cows' milk [2]. This means that while the amount of iron in breast milk is low, the natural iron in breast milk is highly bioavailable to the infant. Despite this efficiency, the iron stores have been found to be inadequate to meet the increased iron needs of infants 6 months of age and older. The AAP has therefore recommended that term breastfed infants receive 1 mg of iron per day for each kilogram of body weight, preferably from two servings of food (e.g., iron-fortified infant cereal and meat), beginning at 4 to 6 months of age. If the term breastfed infant is unable to consume sufficient iron from its diet after 6 months of age, a daily oral iron supplement of 1 mg per kilogram of body weight has been recommended [114]. However, these recommendations have raised concerns among some breastfeeding experts [115,116]. One meta-analysis found that iron interventions had no significant effect on iron deficiency or iron-deficiency anemia, serum ferritin level, or hemoglobin level. Iron supplementation was associated with slower growth during the exclusive breastfeeding period, but the long-term effect is unclear [117].
Like iron, calcium appears in small quantities in human milk. The calcium in breast milk is in a highly absorbable form suited to the human infant. Infants absorb 67% of calcium in human milk compared to only 25% of calcium in cows' milk. Hypocalcemia and tetany are more commonly seen in the neonate who is fed ABM. This is due to the higher concentration of phosphorus in cows' milk, which causes a decrease in calcium absorption and an increase in calcium excretion [2].
Zinc, phosphorus, magnesium, copper, and other trace elements also are present in breast milk. The mineral content of breast milk remains consistent despite changes in the maternal diet.
Although the amounts of vitamins and micronutrients in breast milk vary from mother to mother due to both dietary and genetic differences, breast milk will generally satisfy the requirements of a full-term, healthy infant. Both fat-soluble and water-soluble vitamins are present in breast milk. Generally, as lactation progresses, the level of water-soluble vitamins increases and the level of fat-soluble vitamins decreases. The fat-soluble vitamins (vitamins A, D, K, and E) may be drawn from stores in the mother's body and are therefore minimally influenced by recent maternal diet [2].
Vitamin A, which is necessary for vision and maintenance of epithelial structures, appears at its highest levels in the first week after birth and then gradually declines. Vitamin A deficiency is a serious problem for young children in many developing countries, and prolonged or partial breastfeeding provides an important source of vitamin A for this population [2].
Because breast milk contains very little fat-soluble vitamin D, breastfed infants may occasionally develop rickets. Infants at greatest risk include those who reside in inner-city areas, who receive inadequate exposure to sunlight, or whose mothers consume vegetarian diets that exclude meat, fish, and dairy products. Adequate sun exposure and adequate maternal consumption of nutrients generally precludes the need for routine supplementation of vitamin D. However, scattered reports of rickets have led the AAP to recommend vitamin D supplements for infants and children subject to certain conditions, beginning during the first few days of life [2,21,22].
Vitamin K is necessary for the synthesis of blood-clotting factors. It is present in breast milk only in small amounts, making neonates susceptible to vitamin K deficiency. A few days postpartum, infants normally produce sufficient quantities of vitamin K by enteric bacteria. However, until they are able to ingest large amounts of breast milk, thereby promoting gastrointestinal bacterial colonization, the mother may need to supplement her diet with vitamin K [2].
Human colostrum is rich in vitamin E, and mothers of both preterm and term infants have similar levels of vitamin E, which are higher than the levels in cows' milk. Premature infants are at greatest risk for hemolytic anemia, which is a result of vitamin E deficiency. The antioxidant properties of vitamin E provide protection for cell membranes in the retina and lungs against oxidant-induced injury [2].
Antibodies
Because the human immune system is not fully developed at birth, infants are at risk of infections and gastrointestinal illnesses. Breast milk has been found to both stimulate and supplement the infant's developing immune system. The secretory immune system, which involves and affects the body's surfaces (e.g., the breasts), acts locally and includes lymphocytes that differ from other lymphocytes. These lymphocytes are sensitized to antigens found in the gastrointestinal and respiratory tracts and travel through mucosal lymphoid tissues (e.g., breasts, intestines) where they secrete antibodies (immunoglobulins) that recognize and act on a particular antigen. For example, antigens to which a mother has been exposed help to sensitize lymphocytes that migrate to the breast where they secrete immunoglobulins into the milk [2].
The immunoglobulin found in highest concentration in human milk is IgA. The secretory form of IgA (sIgA) lines the gut and respiratory system in adults and is the major component conferring passive immunity to the breastfed infant. It is both synthesized and stored in the breast, reaching levels up to 5 mg/mL in colostrum, then decreasing to 1 mg/mL in mature milk [2]. sIgA is stable in breast milk and not degraded by either gastric acid or digestive enzymes. It provides local immunity by building a lining on the walls of the intestinal tract, the oral pharynx, and the urinary tract, protecting the infant from infection by preventing invasion of organisms through the mucosa. sIgA fights disease without causing inflammation. IgA protects the infant from invasion but does not fully line the gut until the infant is about 6 months of age. It may take months before the infant can manufacture IgA. Bottle-fed infants have few means for battling ingested pathogens until they begin making IgA on their own [2]. Protection through passive immunity continues for as long as the infant is breastfed. A child's immune response is not fully developed until 5 years of age.
sIgA has been reported to be active against Escherichia coli, Vibrio cholerae, Clostridioides difficile, Salmonella, Giardia lamblia, Entamoeba histolytica, Campylobacter, rotavirus, and poliovirus [2]. IgA prevents the absorption of protein macromolecules, protecting the infant from allergic responses. The specificity of IgA response is related to the mother's antigenic exposure [110]. By this mechanism, breast milk is not only species specific but infant specific and may be environmentally specific as well.
Nonantibodies
Nonantibody factors in breast milk also protect the infant against bacterial infection. For example, the potent iron-binding protein lactoferrin helps prevent diarrheal disease and inhibit the adhesion of pathogenic organisms (e.g., E. coli) to cells [2]. Oligosaccharides help block antigens (e.g., Pneumococcus) from adhering to the epithelium of the gastrointestinal tract. The dominant presence of gram-positive L. bifidus in the intestinal flora of breastfed infants promotes beneficial bacteria and inhibits enteropathogens (e.g., Salmonella, E. coli) [2].
Colostrum, commonly labeled as "liquid gold," is the first milk. It is a mixture of residual cells in the breast and newly formed milk. It is thick and yellow to orange in color with high ash content (i.e., the total mineral content after it is completely burned) and higher concentrations of sodium, potassium, chloride, protein, fat-soluble vitamins, and minerals than mature milk. Colostrum also is rich in antibodies that protect the newborn. It has an important laxative effect on the infant bowel that assists in the emptying of meconium. This is important because the retention of meconium may contribute to neonatal jaundice due to reabsorption of its bilirubin content [118].
The mother produces small amounts of colostrum (7–123 mL) in the first 24 hours postpartum. The newborn may take between 7 mL and 14 mL per feeding [2]. A gradual increase occurs during the first 36 hours. This is followed by a dramatic increase in milk output by the second day that continues through day four. At five days postpartum, milk production is approximately 500 mL/24 hours.
It is a common misconception that the baby gets nothing when nursing in the first day or two. Although the baby will get only a small amount of colostrum at each feeding, this amount matches what the baby's stomach (about the size of a shooter marble at birth) is able to hold. Still, it may be difficult to convince some mothers of the need to nurse early and often when they believe that their "milk has not come in." In addition, some cultures hold the belief that colostrum is "not good," causing some mothers to defer breastfeeding until the mature milk is in. Other women have the perception they need to be engorged before there is any milk for the baby. Likewise, if the mother is no longer engorged, she may falsely believe that her milk is gone. Expressing a drop or two of colostrum for the mother will give her a strong visual cue that her breasts are not empty.
Transitional milk is the creamy breast milk that immediately follows colostrum and may appear as early as 12 hours after delivery and continue for 7 to 14 days. Because the supply of transitional milk is greater than the supply of colostrum, the mother's breasts generally become larger and firmer during this stage. Transitional milk retains some of the yellow color of colostrum. The concentrations of immunoglobulins, total calories, and protein decrease while lactose and total fat increase [119].
Mature milk, seen as early as three days postpartum, is the final, more mature milk that is produced, and it becomes the predominant type by around day nine. Mature milk supplies everything the baby needs, including water; breast milk is 90% water. Even on the hottest days, breast milk provides sufficient water intake for the baby. The remaining 10% of mature milk is comprised of carbohydrates, proteins, and fats that are necessary for growth and energy [1,119].
There are two types of mature milk: foremilk and hindmilk. Foremilk is found at the beginning of the feeding; it contains water, vitamins, and protein. Hindmilk occurs after the initial release of milk and contains higher levels of fat, which are necessary for infant weight gain [119]. It is important to note that both types of breast milk are produced throughout a breastfeeding session; breast milk is never solely one type or the other.
A physiologic measure of milk maturation, the Maturation Index of Colostrum and Milk, has demonstrated that milk maturation occurs at rates that vary from mother to mother and that human milk matures more rapidly when the baby is exclusively breastfed and nursing well. When milk matures rapidly, infants are more content, they have less incidence of neonatal jaundice, and breastfeeding is sustained [120,121].
The process through which the mammary gland develops the capacity to secrete milk is called lactogenesis. It includes all the processes necessary to transform the gland from the undifferentiated state of early pregnancy to a fully differentiated state after pregnancy. It is this second state that allows full lactation (i.e., the process of milk production) [122].
Lactogenesis occurs in two stages. Stage I occurs by mid-pregnancy and includes increases in lactose, total protein, and immunoglobulins and decreases in sodium and chloride content along with the gathering of substrates for milk production [1]. The mammary gland is now sufficiently differentiated to secrete milk; however, high circulating levels of progesterone and estrogen hold secretion in check [122].
Stage II of lactogenesis begins with the sudden withdrawal of pregnancy hormones around the time of delivery; it may be defined as the time of copious milk secretion. Stage II is marked by increases in blood flow, oxygen, and glucose as well as sharply increased concentrations of citrate in the breast [122]. The breasts will begin to produce milk independent of infant suckling.
The breast is not merely a passive container of milk. It is an organ of active production. After Stage II lactogenesis has begun, continued milk production is governed by the infant [2]. When the infant suckles, a series of events takes place within the mother's body. Stimulation of the nipple and areola sends signals to the mammary gland, which are then relayed to the central nervous system. This cyclical process of milk synthesis and secretion is termed lactation. Factors that are necessary to maintain the milk supply include neuroendocrine (i.e., intact neurohormonal pathways and suckling) and autocrine (i.e., milk removal) [123].
The stages of lactation may be summarized to include [1,122]:
Mammogenesis: Breast growth occurs; the size and weight of the breast increases
Lactogenesis: Stages I and II
Galactopoiesis: From later than nine days after birth to the start of involution; secretion has been established and is maintained; the autocrine system is in control
Involution: The stage when milk production begins to decline. Milk secretion decreases due to the buildup of feedback inhibitors of lactation (FIL) and alveolar distension, which signal the mammary gland to reduce production.
The process of lactation occurs with the help of the hormones prolactin and oxytocin. Although they act independently on different cellular receptors, the combined action of prolactin and oxytocin are necessary for successful lactation to occur [122].
Prolactin, which is often called the "mothering" hormone, is secreted in the anterior pituitary. The very high levels of circulating estrogen that occur during pregnancy result in a parallel increase in the circulating levels of prolactin. This helps to complete lobular development in the breasts and prepares them for lactation. Prolactin levels rise from a nonpregnant baseline of 10–25 ng/mL to 100–400 ng/mL at term [1,2,124].
Progesterone antagonism from the placenta enables the prolactin level to rise without subsequent milk production. Progesterone interferes with prolactin's activity on the cell receptor sites in the alveoli of the breast. With the birth of the placenta and the sudden drop in the pregnancy hormones progesterone and estrogen, the elevated prolactin level, in addition to the presence of insulin and cortisol, increases the milk supply. Prolactin is released in pulses directly related to stimulation of the areola or breast. Prolactin levels decrease as lactation is established, but nursing stimulates prolactin release from the pituitary, which promotes continued milk production [1,2,124,125,126].
Frequent feeding in the early days increases the number of prolactin receptor sites within the breast. Research has indicated that breast milk output is largely determined by the number of prolactin receptors in the breast rather than by the level of serum prolactin. These prolactin receptors are laid down in the first three months postpartum and allow abundant milk production to continue when total baseline prolactin levels drop over the first three to four months. At near "normal" baseline levels, breast stimulation continues the doubling of the declining baseline prolactin levels into the second year [1,2].
Elevated prolactin levels in the early days of lactation help both milk and receptor site production. Each nursing produces a doubling in serum prolactin levels. Prolactin levels naturally rise in sleep states. Night nursing helps maintain an elevated baseline prolactin level. Thus, it is unwise for a mother to skip breastfeeding at night (e.g., having someone else give bottles) if she wants to develop a good milk supply. Frequent prolactin release inhibits follicle stimulating hormone (FSH) and luteinizing hormone (LH), causing lactational amenorrhea and preventing the return of fertility [1].
Prolactin is biologically potent for the infant. Milk prolactin levels are highest in colostrum and transitional milk. In mature milk, the highest prolactin concentration is in the foremilk. In the infant gut, prolactin affects fluid and electrolyte exchange, in particular, sodium, potassium, and calcium [1,2].
The prolactin receptor site theory has raised concern over progesterone-laden birth control methods when they are started within days of the birth. Medroxyprogesterone acetate (Depo-Provera) shots are sometimes given immediately postpartum, while the mother is still in the hospital. If progesterone is an antagonist to prolactin, logic dictates that progesterone shots, implants, and pills can inhibit early establishment of milk production [1]. Detectable amounts of Depo-Provera have been identified in the milk of mothers receiving the drug; however, it does not appear to adversely affect milk composition, quality, or amount [127]. Therefore, the Academy of Breastfeeding Medicine recommends that clinicians advise women that hormonal contraceptive methods may decrease milk supply, especially in the early postpartum period. Additionally, hormonal methods may be discouraged in some circumstances, such as [128]:
Existing low milk supply or history of lactation failure
History of breast surgery
Multiple birth (twins, triplets)
Preterm birth
Compromised health of mother and/or baby
While prolactin is essential for initiating and maintaining lactation, oxytocin is keyed more closely to milk ejection. Oxytocin receptor sites in the breast gradually increase by tenfold in pregnancy. Just before delivery, the number of oxytocin receptor sites in the uterus increases dramatically and then suddenly disappears after the birth. Oxytocin is taken up by the uterus first to facilitate delivery and prevent postpartum hemorrhage by uterine contraction, and then to cause milk ejection [1,129]. The nipple becomes more sensitive to tactile stimulation in the 24 hours following birth. Stretch receptors in the nipple stimulate the release of oxytocin from the posterior pituitary. Oxytocin causes the let-down reflex or milk ejection response [1,129].
In addition to prolactin and oxytocin, the manufacture of milk also requires several other hormones for milk synthesis at the alveolar level, including insulin, cortisol, thyroid, parathyroid, and growth hormone.
The surface tension in the breast is sustained so that milk does not freely move out of the breast. The milk ejection response, more commonly referred to as the "let-down reflex," is the action of oxytocin upon the smooth muscle of each alveolus at the microscopic level. Contraction of the alveoli actively pushes the milk into the ducts toward the nipple and finally to the infant. As the baby continues to nurse, additional milk ejection responses occur, although most mothers sense only the first milk ejection. In the early days of nursing, it may take five to eight minutes for the first let-down to occur [1].
Oxytocin, like prolactin, is released in pulses. It has a very short life span (about two to three minutes) in maternal serum. The first pulse begins before the baby is put to breast (e.g., triggered by mother thinking it is time to feed or by infant crying). Subsequent release is in response to nipple stretching. In addition to its action on the mammary gland, oxytocin causes uterine contractions, particularly in the term and postpartum uterus. In the early days of breastfeeding, these "afterbirth pains" or "uterine cramping" may be very uncomfortable, although the cramping is known to aid in uterine involution [2]. The higher the mother's parity, the more intense the afterbirth pains.
Mothers are very concerned when they cannot feel the let-down in the postpartum period. Although this lack of sensation is normal, interference with the let-down reflex may be caused by pain, stress, and anxiety. The mother can be taught to observe the infant for bursts of swallowing, sleepiness, and/or thirst or to be conscious of uterine cramping or dripping milk as cues that a let-down has occurred. Encouraging the mother to nurse in a comfortable position while relaxed and undisturbed may also help. After the first week or two, the mother will usually begin to feel the let-down. Most mothers state that they only feel the first let-down in a feeding. This may be due to the distention of the ducts with foremilk. A few women are aware of each let-down. The let-down reflex can be felt as a tingling sensation. Occasionally, women may describe the let-down as a burning or painful sensation. As a general rule, the more obvious it is to the mother that her milk is "letting down," the fuller her breasts are [130].
It is important to note that the breast never truly empties and that the rate of milk production depends on the rate of milk removal (i.e., the more milk removed by infant demand, the greater the milk production). Research has demonstrated that each breast makes milk slowly or quickly depending on how full or empty it is. A full breast produces milk slowly; an empty breast produces milk more quickly. Therefore, when the mother's breast seems emptiest, it is making milk the fastest. If baby is hungry and nurses vigorously, leaving the breast relatively empty, production speeds up. If baby does not take much milk while feeding, production slows down. Thus, baby's appetite controls mother's milk production. Assuring that baby has access to the breast when hungry allows the baby to regulate milk production [1,2].
Research has indicated that each breast has its own individualized maximum storage capacity, which is not related to breast size. However, mothers with larger breasts have greater storage capacity (the difference between maximum and minimum breast volumes during a 24-hour period) and, as a result, greater flexibility in feeding intervals. Women with smaller breasts can produce as much milk as women with larger breasts, but they must breastfeed more often [2]. This helps explain why cue feeding, rather than strict scheduling, is best for baby and for the mother's milk supply.
Engorgement is different from breast fullness. Breast fullness is the gradual accumulation of blood and milk in the breast a few days after birth and is a sign that the mother's milk is increasing. Breast fullness does not impair efficient breastfeeding because the breast tissues may be easily compressed by the baby's mouth [130].
Breast engorgement often occurs when the feedings are infrequent and/or of a limited duration. The best management of engorgement is prevention: nursing often and effectively (at least eight times or more in 24 hours); expressing milk manually or with a pump if baby is being fed away from the breast; and alternating taking warm showers and using cold compresses to relieve discomfort [130].
The process of painful engorgement is threefold: increased blood flow to the breasts causes tissue congestion, the ducts and alveoli become distended with milk, and edema develops secondary to swelling and obstruction of the lymphatic drainage system [1]. The nipples cannot protrude to allow the baby to latch on correctly, and nursing is difficult [130].
If the breasts are not signaled by a suckling infant or breast stimulation within the first 24 to 48 hours postpartum, the result is alveolar distention, tissue congestion, and destruction of alveolar tissue. Distended lactational tissue and tissue congestion may prevent the appropriate hormones from reaching the breast and producing the desired effect of milk synthesis and milk ejection. Some mothers develop a fever if excessive engorgement occurs. A fever as high as 100°F accompanying engorgement in the early postpartum period may be mistaken for postpartum infection. Unrelieved engorgement and over-distention of the alveoli may cause some alveoli to rupture, resulting in partial involution of the breast. Subsequently, complete involution of the breasts may occur in as little as six hours in unrelieved engorgement [131].
Early in fetal development, male and female breast tissue develop from an invagination of the ectoderm. During the fourth week, a raised area may be seen in the developing fetus. Ventral epidermal ridges, or milk lines, may be seen by the sixth week of fetal life. These lines arch down from the armpit to the groin on both sides. The breast develops along the ridge over the chest, and most of the remainder of the ridge disintegrates [14].
Between 18 to 19 weeks' gestation, specialized cells continue transforming into the smooth muscle of the nipple and areola. Mammary buds form and create milk ducts. Sex hormones in the placenta enter fetal circulation and control breast development until 32 weeks' gestation. From 32 to 40 weeks, milk ducts containing colostrum form within the nipple [122]. Near term, 15 to 25 ducts form the fetal breast. The withdrawal of maternal hormones may cause breast engorgement in the newborn. Shortly after birth, a neonate's breasts may produce a small amount of colostrum. Studies have shown that the components of neonatal milk resemble the components of mother's milk [132]. Milk production in the neonatal breast quickly subsides, and the glands become the mammary disk of childhood. The mammary glands are not fully developed at birth and will remain inactive until puberty [1].
Organogenesis begins just before the onset of puberty in the female (at 10 to 12 years of age). The internal breast structures begin to expand rapidly under the influence of estrogen. Typically, first menstruation will begin about one or two years after the breasts begin to grow [14]. With every menstrual cycle, a new phase of growth occurs, which includes extensive branching of the ductal system and organization of the internal structures. Fat deposits in the breast give it a more adult, rounded appearance. The greatest changes occur by 20 years of age, but the breast continues to develop until 35 years of age [2]. The breast is not considered fully mature until a person gives birth and begins to produce milk [14].
The breast is composed of glandular tissue, fibrous tissue, and adipose (fatty) tissue. The relative proportion of each type of tissue changes with a woman's age, menstrual cycle, pregnancy, and nutritional status. The significant structures of the adult female breast include (Figure 1) [2,14,122,133]:
Alveolus (acinus) cells: Where milk is produced, stored, and secreted
Myoepithelial cells: Surround the alveoli and contract so milk is forced into the ductules
Ductules: Filter out milk from the alveoli
Nipple (mammary papilla): Functions as a nozzle for delivery of milk; is the most sensitive to tactile stimulation and pain
Nipple openings
Areola: The darker portion behind the nipple; may vary widely in size and color
Montgomery glands or tubercles (areolar glands): Secrete oils that lubricate the nipple; may be antibacterial
Lobe: The branching network of the above structures; each breast contains approximately 20, ending in the nipple
It was previously thought that lactiferous sinuses stored milk and drained into 15 to 25 openings in the nipple, allowing milk to flow to the infant. However, based on extensive real-time ultrasonography, researchers have concluded that instead a lower number of milk ducts are much closer to the nipple than previously thought [134].
Pregnancy brings increased growth within the breast. The size of the breast varies greatly depending on the amount of fatty tissue present within the breast. By the time the baby is born, glandular tissue will have replaced most of the fatty tissue. Breast size bears no relationship to the amount of milk produced. The exception is a condition called insufficient glandular development of the breast. The darkening of the areola during pregnancy may serve to act as a visual target for the newborn. Secondary darkening or patchy pigmentation behind the areolar rim also may occur in pregnancy. The Montgomery glands may become enlarged and elevated, and the nipples may become more pronounced [2,9,135].
Important changes that occur during the first trimester include the expansion of the ductal system and its branches in response to estrogen; the expansion of the glandular tissues of the alveoli in response to hormones such as prolactin; and an increase in the size of the lobes and lobules in response to progesterone. Second trimester changes include continued development and enlargement of the duct system and additional lobule growth (continuing into the third trimester). Under the influence of a ten- to twentyfold increase in placental lactogen, colostrum appears near the end of the second trimester. The breast will produce colostrum if the fetus is born at 16 weeks. However, secretory milk-releasing alveolar cells do not develop until the third trimester [1].
Following lactation, the breasts involute (i.e., the mammary gland returns to its nonproductive state of milk secretion). If milk is not removed from the breasts, as during weaning, the glands become distended. This interferes with the blood supply to the breasts, and milk production ceases. There is also evidence that an enzyme produced by the unremoved milk decreases production. Milk remaining in the alveoli is gradually reabsorbed, and the alveoli collapse or rupture. Initially, after weaning, the breasts may appear smaller than their pre-pregnancy size. This is due to the lack of adipose tissue within the breast. The adipose tissue gradually increases, and the breast returns to its resting state. Some residual growth of the glandular tissue remains after lactation. Women often may express a drop or two of milk for up to 12 months following weaning [2,136].
Breastfeeding and nurturing are learned skills. In the few remaining traditional societies, women breastfeed openly with very few problems. The inverse is true in Western culture, where there are few open breastfeeding role models for young women. Some adult women have never watched another woman nurse. Expectant and new mothers may turn to an acquaintance who has nursed a baby for advice. Unfortunately, such advice may be tainted with tales of difficulties causing uneducated or frightened mothers to feel compelled to bottle feed. Home support may not be available because their mothers and grandmothers did not breastfeed. This leaves new mothers on their own to discover breastfeeding and mothering. Seventy-two percent of breastfeeding mothers have questions in the first two weeks and rely on books and the Internet, not healthcare professionals, as their most common source for breastfeeding information postpartum [137,138].
Successful breastfeeding is not automatic, as demonstrated by the failure rate [1]. Women must be educated about breastfeeding to succeed at breastfeeding. Furthermore, the definition of successful breastfeeding is the achievement of the mother's goal for breastfeeding, whatever that may be. For example, if her goal is to nurse for 12 months but she is only able to nurse for 12 days, then she has not achieved her goal.
As a healthcare professional, the most difficult task in breastfeeding education is addressing myths. Knowing the mother's culture and her expectations is an integral part of education. Intellectually, she may not be able to overcome her set of myths without enough knowledge. Common breastfeeding myths include [139]:
Myth: Frequent nursing leads to poor milk production, a weak let-down response, and unsuccessful nursing.
Fact: Milk supply is optimized when a healthy baby is allowed to nurse on demand.
Myth: A mother only needs to nurse four to six times per day to maintain good milk supply.
Fact: Research has shown that early and frequent breastfeeding (an average of 9.9 times a day in the first two weeks) encourages greater milk production, increased infant weight gain, and a longer duration of breastfeeding.
Myth: Infants get all the milk they need in the first 5 to 10 minutes of nursing.
Fact: While many older babies can take in the majority of their milk in the first 5 to 10 minutes, this cannot be generalized to all babies. Newborns, who are learning to nurse and are not always efficient at sucking, often need much longer to feed. It is best to allow the baby to suck until he shows signs of satiety.
Myth: A breastfeeding mother should space her feedings so that her breasts will have time to refill.
Fact: Every baby/mother dyad is unique. A lactating mother's body is always making milk. The emptier the breast, the faster the body makes milk to replace it; the fuller the breast, the more production of milk slows down. If a mother consistently waits until her breasts "fill up" before she nurses, her body may get the message that it is making too much and may reduce total production.
Myth: Babies need only six to eight feedings per day by 8 weeks of age, five to six feedings per day by 3 months of age, no more than four or five feedings per day by 6 months of age.
Fact: A breastfed baby's frequency of feeding will vary according to the mother's milk supply and storage capacity, as well as baby's developmental needs.
Myth: It is the quantity of milk that a baby takes in not the quality that determines how long a baby can go between feedings.
Fact: Breastfed babies have faster gastric emptying times than formula-fed babies. While intake quantity is one factor in determining feeding frequency, the type of milk is equally important.
Myth: Never wake a sleeping baby.
Fact: While most babies will indicate when they need to eat, babies in the newborn period may not wake often enough on their own and should be awakened if necessary to eat at least eight times a day.
Myth: Breastfeeding mothers must always use both breasts at each feeding.
Fact: It is more important to let baby finish the first breast first, even if that means that he or she does not take the second breast at the same feeding. In the early weeks, mothers should offer both breasts at each feeding to help establish a milk supply.
Myth: If a baby is not gaining well, it may be due to the low quality of the mother's milk.
Fact: Studies have shown that even malnourished women are able to produce milk of sufficient quality and quantity to support a growing infant. Most cases of low weight gain are related to breastfeeding management issues, insufficient milk intake, or an underlying health problem in the infant.
Myth: Poor milk supply is usually caused by stress, fatigue, and/or inadequate fluids and food intake.
Fact: The most common causes of milk supply problems are infrequent feedings and/or poor latch-on and positioning; both are usually due to inadequate information provided to the breastfeeding mother.
Myth: A mother must drink milk to make milk.
Fact: A healthy diet of vegetables, fruits, grains, and proteins is all that a mother needs to provide the proper nutrients to produce milk. Calcium can be obtained from a variety of nondairy foods such as dark green vegetables, seeds, nuts, and bony fish.
Myth: Non-nutritive sucking has no scientific basis.
Fact: Experienced breastfeeding mothers learn that the sucking patterns and needs of babies vary. While some infants' sucking needs are met primarily during feedings, other babies may need additional sucking at the breast soon after a feeding even though they are not hungry. Babies may also nurse when they are lonely, frightened, or in pain.
Myth: The mother should not be a pacifier for the baby.
Fact: Comforting and meeting sucking needs at the breast is nature's original design. Pacifiers are literally a substitute for the mother when she cannot be available. Sucking at the breast can result in superior oral-facial development, prolonged lactational amenorrhea, avoidance of nipple or flow preference, and stimulation of an adequate milk supply to ensure higher rates of breastfeeding success.
Myth: There is no such thing as nipple or flow preference.
Fact: Breast and bottle feeding require different oral-motor skills, and rubber nipples provide a type of "super stimulus" that babies may imprint upon instead of the softer breast. As a result, some babies develop suck confusion and apply inappropriate suckling techniques to the breast when they switch between breast and bottle.
Myth: Feeding on the baby's cue does not enhance maternal bonding behavior.
Fact: The responsive parenting of cue feeding brings the mother and the baby into synchronization, leading to enhanced bonding.
Myth: It is important that other family members get to feed the baby so they can bond, too.
Fact: Feeding is not the only method by which other family members can bond with the baby; holding, cuddling, bathing, and playing with infants are all important to their growth, development, and attachment to others.
Myth: Child-directed feeding (nursing on demand) has a negative impact on the husband/wife relationship.
Fact: Parents realize that a newborn's needs are very intense but also diminish over time. In fact, the teamwork of nurturing a newborn can bring a couple closer as they develop parenting skills together.
Myth: Some babies are allergic to their mother's milk.
Fact: Human milk is the most natural and physiologic substance that a baby can ingest. If a baby shows sensitivities related to feeding, it is usually a foreign protein that has entered into the mother's milk, and not the milk itself. This is carefully handled by removing the offending food from the mother's diet for a time.
Myth: Frequent nursing causes a child to be obese later in life.
Fact: Studies have shown that breastfed babies who control their own feeding patterns and intake tend to take just the right amount of milk for them. Formula feeding and early introduction of solids, not breastfeeding on demand, have been implicated in risk of obesity later in life.
Myth: Nursing a baby after 12 months is of little value.
Fact: The composition of human milk changes to meet the changing needs of baby as he or she matures. Even when a child is able to take solids, human milk is the primary source of nutrition during the first year and may become a supplement to solids during the second year. In addition, it takes between two and six years for a child's immune system to fully mature. Human milk continues to complement and boost the immune system for as long as it is offered.
Giving correct, consistent information will lead to success. Encourage all mothers to attend a breastfeeding class antepartum. A good class will contain most of the information shown in the sample outline (Table 6).
BREASTFEEDING CLASS OUTLINE
|
|
At the first prenatal appointment, the medical provider should ask the mother how she plans to feed her baby. If the mother is undecided or if she is positive about breastfeeding, her educational needs should be discussed. Support of the mother's decision should begin with careful history taking and physical assessment of the breasts.
Visual examination of the breast should include symmetry and general shape. The mother should be examined in both a sitting and supine position. Breast shape and size and variations of the nipple and areola are diverse. Large nipples with small areola, large areola with small nipples, or large areola with large nipples are variations of normal. The nipple is usually round, but it may appear keyhole shaped.
The areola should appear moist, not dry or scaly. If the areola is dry, itchy, and pink in color, a yeast infection of the areola and nipple should be suspected and treated appropriately. If the areola is dry, the mother should be questioned regarding the use of soap or other drying agents on the breast. Darker skin tones may be more prone to visible dryness or ashiness due to unique structural and biochemical differences in the skin, such as lower ceramide levels. However, skin hydration varies widely among individuals [292]. Commercial breast creams should be avoided. Creams can macerate the skin and plug the nipple pores and Montgomery glands [1].
The breasts should be palpated beginning at the edge of the areola outward, toward the axilla and sternum. The examination should be done in a circular manner with the fingertips, in a way that will encompass the entire breast. The breast tissue should be semi-firm in the primipara. Some laxity of the tissue is expected in the multipara. The breast should feel like a round mound of tissue and should not feel hollow or empty at any point within the breast proper. If the mother has a history of cystic breasts, the breasts may feel lumpy or nodular upon palpation. The breasts may also be very tender to touch early in pregnancy. Assure the mother that the tenderness will go away as pregnancy nears term.
The nipples should be examined for protractibility. Ask the mother if her nipples ever stand out when she is cold or sexually excited. There are several major nipple types: everted, flat, pseudo-inverted, and inverted. Everted nipples elongate outward from the juncture at the areola. Flat nipples are less distinct from the areola but the tissue everts slightly. Pseudo-inverted nipples appear inverted but when stimulated, they elongate to a flat or everted nipple. The truly inverted nipple is relatively rare, occurring in less than 3% of the population [2]. The inverted nipple appears as if it is drawn deep within the areola and does not evert. Often, flat or pseudo-inverted nipples are misdiagnosed as inverted. Use caution when telling a mother there is "something wrong" with her nipples or breasts. She may decide she cannot breastfeed because her nipples are inverted when, in fact, they are not. The words you use matter.
Inverted nipples are not a contraindication to breastfeeding [2]. Prenatal nipple preparation, such as nipple rolling, breast shells, and Hoffman exercises, have been shown to reduce the initiation rate of breastfeeding. None of these techniques are successful, and they may be dangerous [1].
Women may develop breast lumps at any time. Approximately 29% of cases of breast cancer occurs in women younger than 45 years of age [140]. The average patient is between 32 and 38 years of age [141]. Breast cancer is the most common cancer in pregnant and postpartum women, occurring in about 1 in 3,000 pregnant women; however, this percentage may increase as more women delay childbearing until their mid- to late-30s [141,142,143,144]. The breast changes that accompany pregnancy and lactation often make the mother aware of a lump that she has not felt before. Pregnant and lactating breasts are dense and lumpy. Women may delay treatment because they believe the lump is a disorder of lactation that will remedy itself. Inflammatory breast cancer, the most lethal of localized advanced cancers, often mimics mastitis; however, there are no systemic symptoms [143].
Monthly breast self-examination is no longer recommended, but being aware of the normal look and feel of the person's breast is important to allow for reporting of changes to a care provider. If an exam is needed, it is best for the lactating mother to examine her breasts after a feeding, when a lump might be more apparent [141,143]. It is important to note any new dimpling of the breast or nipple, or any lump that is painless, discrete, fixed, hard, or irregularly shaped. These cases should immediately be referred to a breast surgeon who is familiar with pregnant and lactating women. The most common cause of a breast lump in the lactating woman is a persistent plugged duct, or galactocele; other possible causes include mastitis and breast abscess. Lumps and/or mastitis that do not respond to treatment should be investigated [1]. Fine-needle biopsy may be done where necessary [141].
It is important to ask the mother if she has had breast surgery or surgery around the area of the breasts. Breast surgery may adversely affect a mother's ability to nurse by interfering with normal anatomy and neurohormonal pathways.
Procedures to remove breast lumps may affect the lobe where the lump was removed. Following lactogenesis, the mother may notice a hard, painful area in the quadrant of the breast near the incision. One or more of the ducts may have been severed during the surgery, especially if the lump was near the areola. The area will eventually reabsorb and involute with no effect upon total milk supply unless a large portion of the lactating tissue is involved. Because lumpectomy is unilateral in most cases, the mother should produce sufficient milk from the remaining tissue [2].
Of the more than 1.8 million breast augmentations performed worldwide in 2018, 17.3% were performed in the United States, which is a 48% increase since 2000 [116]. Many women are unaware of the impact of augmentation surgery on their future ability to lactate [145]. Following breast augmentation surgery, there is a chance that a woman will not be able to breastfeed due to insufficient milk production or surgical technique [146,147]. It is important to ask questions about the shape and size of her breast(s) before she had the surgery and her reasons for the implant surgery, as this may reveal an undiagnosed case of insufficient glandular development, especially if the augmentation was unilateral [148,149].
It is important to note the location of the surgical scars. If the implants were inserted from under the breast, the scar will follow the curvature of the underside of the breast. Ask the mother if she has experienced any permanent loss of nipple sensation or an increase in nipple sensitivity since having the surgery. If she has, the nerve supply to the nipple may have been interrupted and let-down may not occur. If the implants were inserted through the areola, the scar will appear as a half-moon shape on the lower edge of the areola. With this type of implant surgery, the nerves and the ducts may have been severed, trapping the milk within the breasts. This can cause severe engorgement [145,147].
Due to information in the media about implants that leak silicone, many women have expressed concern about the potential effects of such a leak on a breastfed infant or on breastfeeding success [150,151].
Breast reduction surgery may also adversely affect a woman's ability to nurse [146,152]. Small studies have indicated that women who have undergone reduction mammoplasty have a significantly shorter duration of breastfeeding [152]. However, due to advances in surgical techniques that allow for preservation of breast structures, breastfeeding may be possible for these women. For women who have had this surgery, higher percentages of successful breastfeeding have been associated with the receipt of information and encouragement from their healthcare providers [153,154,155].
Women with implants or those who have had reduction surgery should be referred to a lactation consultant before delivery to prevent problems. The baby should be followed closely for appropriate weight gain for several weeks after birth, and the mother should be instructed to immediately report any suspicion of low milk supply to her lactation consultant or medical provider. With proper assistance and follow-up, neither breast augmentation nor breast reduction surgery is a contraindication to breastfeeding. Primary care providers should assist their patients with decisions about breast surgery and communicate with the surgeon. Advocacy on behalf of their patient's future breastfeeding needs and breastfeeding conservation strategies also should be discussed [146].
Providers also should note the following during the lactation assessment [2]:
Breast changes during pregnancy (e.g., fullness, firmness, increased sensitivity). Women who report no pregnancy changes should be observed for Stage II lactogenesis postpartum. The lack of breast changes in pregnancy may indicate that the mother will not be able to fully lactate.
History of maternal thyroid difficulty, pituitary problems, diabetes, or other endocrine problems. Breastfeeding requires intact neuroendocrine pathways. The mother may require adjustments in insulin or thyroid medication postpartum.
Prior breastfeeding experience(s) in the multipara. This should include the length of nursing, any problems with nursing, what prompted weaning, and whether the mother felt she was successful at nursing. If the mother reports prior difficulty, a prenatal visit to a lactation consultant could help avoid additional problems.
A history of sexual abuse. Researchers have begun to look at the effects of childhood sexual abuse on a woman's ability and desire to breastfeed. Many survivors of childhood sexual abuse have reported distorted body images and low self-esteem. If the mother is not in a recovery program, she should be referred to one as soon as possible. If she chooses to pump her breast milk and bottle feed it to her baby, she should be encouraged to do so.
The risk/benefit ratio of breastfeeding should be determined by a team of knowledgeable primary care providers. This is typically the infant's pediatrician and the mother's obstetrician or primary care provider. Relative contraindications to breastfeeding include [1,2,28,156]:
HIV infection: In its updated guidance, the AAP recommends that breastfeeding parents with HIV who are receiving antiretroviral therapy (ART) and are experiencing viral load suppression have a less than 1% chance of transmitting the virus to their baby through their milk and can be allowed to breastfeed. Direct breastfeeding is not recommended for parents who are not receiving ART or are receiving ART but are without appropriate viral suppression [293]. These mothers could choose to pump their own milk and have it pasteurized at a local milk bank or obtain milk for their babies from a breast milk bank.
Lack of desire: The woman who has been fully informed of the risks associated with ABM use and who does not wish to breastfeed should not be compelled to nurse her infant.
Human T-cell leukemia virus type I (HTLV-I): This virus is endemic in the West Indies, Africa, and southwestern Japan. Women in the United States with HTLV-I disease should not breastfeed.
Suspected or confirmed Ebola virus disease (EVD): EVD is spread through direct contact with blood and other bodily fluids, including breast milk. EVD has been detected in samples of breast milk; however, no data exist about when in the course of the disease the virus appears in breast milk or when it is cleared. Those with confirmed EVD and those recently recovered from EVD should not breastfeed.
Breast cancer: The woman with active breast cancer requires treatment for herself. Women with a history of breast cancer should not be prohibited from breastfeeding. However, the infant should be monitored for appropriate growth, as radiation to the breast causes destruction of the milk-producing glands.
Active hepatitis B infection: Hepatitis B transmission through breastfeeding has not been reported. Infants born to hepatitis B-infected mothers should be immunized with hepatitis B immune globulin immediately after birth, plus given a first dose of human hepatitis B vaccine within 24 hours of birth. With appropriate follow-up immunizations, the baby may breastfeed.
Hepatitis C: Most patients with hepatitis C are asymptomatic with no traceable transmission route. The greatest risk is chronic active hepatitis C. Infection through breast milk appears to be infrequent.
Any life-threatening illness: The mother will need treatment and all additional energy stores for self-healing. This is a clinical judgment that should be made with all the facts presented.
Severe psychosis: When the mother's mental state poses risks to her life or her infant's, it may not be possible to breastfeed. Postpartum depression may be treated with compatible drugs and is not a contraindication to breastfeeding.
Although breast preparations have long been recommended for mothers who wish to breastfeed, studies have suggested that requiring nipple preparation may deter some women from breastfeeding [2]. Additionally, nipple preparation has not been shown to prevent postpartum nipple soreness.
The purpose of a nursing bra is to support the increased weight of the lactating breast. It is important that the bra is comfortable; however, it need not be worn 24 hours a day, unless it is more comfortable to do so. The bras should be purchased in the last trimester of pregnancy to help ensure proper fit. An ill-fitted bra may compress the breast tissue, resulting in plugged ducts.
When the mother is hospitalized in labor, support of her decision to breastfeed should begin on admission. Hospital staff can exert tremendous influence over this critical area of maternal-child health by promoting and modeling optimal breastfeeding practices, adopting the Baby-Friendly Hospital Initiative, and employing staff who are knowledgeable and well-trained in the principles of successful breastfeeding [1,2,56,157].
Hospitals can be frightening places for women in labor. Support in labor has been shown to have positive effects on birth, bonding, and breastfeeding success [111,158]. During labor and delivery, mothers should have continuous physical and emotional support from a knowledgeable, caring individual (e.g., a doula) in addition to support from her partner [159,160].
The word doula comes from ancient Greek, meaning "woman's servant" [119,161]. Throughout history, doulas have supported women through labor and birth and they continue to do so in much of the world today. Doulas provide continuous encouragement and physical assistance to the mother [7,161]. Doulas provide specific labor support skills, offer guidance and encouragement, assist mothers to cover gaps in their care, build team relationships, and encourage communication between the mother, nursing staff, and medical caregivers [162].
Birth doulas provide continuous physical, emotional, and informational support to the mother before, during, and just after childbirth. They provide a level of support that differs from that of a person who is intimately related to the woman in labor; however, they also play an important role in helping the mother's partner become involved in the birth to the extent that the partner is comfortable, without intruding on intimate moments between the couple [161,163]. Postpartum doulas do whatever the mother needs to best enjoy and care for her new baby. These needs often change from day to day. A large part of the role of postpartum doulas is education (e.g., sharing information about baby care, teaching siblings and partners to "mother the mother," assisting with breastfeeding education). They also ensure that the mother is comfortable, well fed, and properly hydrated. Postpartum doula support may last from one or two visits to more than three months. Doulas do not replace nurses or other medical staff. They do not perform clinical or medical tasks (e.g., taking blood pressure, monitoring fetal heart rate). Their role is to provide comfort and support to the mother and enhance communication between the mother and her medical caregivers [161].
Studies have shown that a doula's presence at birth tends to result in [161,163,164]:
Shorter labor with fewer complications
A reduction in the mother's negative feelings about the childbirth experience
A reduction in the need for pharmacologic pain management in labor
Decreased need for instrumented delivery and cesarean sections
Shorter hospital stays for the baby and fewer admissions to special care nurseries
Infants who breastfeed more easily
Improved breastfeeding outcomes both during the postpartum period and for several weeks following birth
Mothers who are more affectionate during the postpartum period
The level of support provided by doulas has been shown to have the greatest influence on successful outcomes (Table 7).
LABOR OUTCOMES USING A DOULA
| Outcome | With Doulaa | Without Doula | ||
|---|---|---|---|---|
| Length of labor (primiparous women) | 7.7 to 9 hours | 15.5 to 19 hours | ||
| NSVD (no medications used) | 55% | 12% | ||
| Epidural anesthesia | 8% | 26% | ||
| Oxytocin (augmentation/induction) | 2% to 17% | 44% | ||
| Forceps | 8% | 26% | ||
| Cesarean section delivery | 7% to 8% | 17% to 27% | ||
| ||||
Medications, certain procedures, and/or pain management techniques may mediate a woman's pain experience in labor and delivery. Despite the widespread availability of these interventions, however, most women, in all societies and cultures, associate childbirth with pain that exceeds expectations [165]. The amount of pain felt differs from woman to woman and may depend on factors such as the size and position of the baby and the strength of the woman's contractions [166].
Research literature has supported the notion that maternal comfort should be considered a priority and mothers should receive adequate information regarding any medication prior to receiving it [167]. The Academy of Breastfeeding Medicine has recommended that maternity care providers initiate informed consent discussions about pain management in labor during the mother's prenatal period before the onset of labor. The discussion should include what is known about the various modalities, including their risks and effects (on the newborn and on breastfeeding) [160].
Use of pharmacologic agents for pain relief in labor and the postpartum period may relieve suffering during labor and allow mothers to recover from birth, especially cesarean birth, with minimal interference. However, these agents also may affect the course of labor, the neurobehavioral state of the neonate, and the initiation of breastfeeding. Unmedicated birth followed by immediate skin-to-skin contact between mother and infant leads to the highest likelihood of baby-led breastfeeding initiation. Difficult, medicated labors may lower the rate of breastfeeding success due to the effects on the infant, including depressed or delayed suckling (Table 8) [167,168,169].
COMMONLY USED ANESTHETICS IN LABOR/POSTPARTUM AND THEIR IMPACT ON BREASTFEEDING
| Drug Name | Infant Impact |
|---|---|
| Bupivacaine | May produce central nervous system (CNS) depression with high maternal serum levels |
| Fentanyl (Sublimaze) | May cause respiratory depression at high levels |
| Halothane | Limited human data; animal data suggest low risk |
| Isoflurane | No human data |
| Ketamine (Ketalar) | Limited human data; animal data suggest low risk |
| Lidocaine | May produce CNS depression with high maternal serum levels |
| Midazolam (Versed) | Some evidence of relationship to "floppy infant syndrome" (e.g., mild sedation, hypotonia, reluctance to suck, cyanosis, decreased responses to cold stress) |
| Morphine (Duramorph) | Apnea and bradycardia with repeated maternal doses (dose-dependent) |
| Nitrous oxide | Unknown or no clinical effects |
| Propofol (Diprivan) | Limited human data; not recommended for obstetrics, including cesarean section deliveries; may be associated with neonatal CNS and respiratory depression |
| Remifentanil (Ultiva) | May produce respiratory depression and sedation |
| Rocuronium (Zemuron) | Unknown |
| Ropivacaine (Naropin) | May produce CNS or cardiovascular depression, bradycardia, jaundice, low Apgar scores |
| Sevoflurane | No human data |
| Succinylcholine | Unknown |
| Sufentanil (Sufenta) | Unknown |
| Thiopental | Limited data |
| Vecuronium | No human data |
Nonpharmacologic methods for pain management during labor (e.g., hypnosis, Lamaze exercises, intradermal or subcutaneous water injections for back pain) have no known adverse effects on the neonate; however, their effects on breastfeeding outcomes have not been adequately studied.
Table 9 contains a summary of recommendations for analgesia and anesthesia for breastfeeding mothers [160].
ANALGESIA AND ANESTHESIA FOR BREASTFEEDING MOTHERS
| Medication | Notes | |||||
|---|---|---|---|---|---|---|
| For Labor | ||||||
| Fentanyl | Short-acting; preferred | |||||
| Remifentanil | Potent; rapid onset/offset; maternal apnea, neonatal respiratory depression, sedation may occur; requires increased monitoring; minimal transfer in utero to fetus | |||||
| Meperidine/pethidine | Generally not recommended except in small doses, less than one hour prior to delivery due to increased incidence/duration of neonatal depression, cyanosis, bradycardia | |||||
| Nalbuphine, butorphanol | May be used for patients with certain opioid allergies or those at increased risk of difficult airway management or respiratory depression; may interfere with interpretation of fetal heart rate monitoring; severe fetal bradycardia has been reported following use of nalbuphine in labor/delivery | |||||
| All IV opioids | Multiple doses and timing of administration may cause greater neonatal effects. When mother has received IV narcotics for labor, mother/baby should be given more skin-to-skin time to encourage early breastfeeding | |||||
| Epidural analgesia | Select method that minimizes the dose and motor block. Avoid longer durations to minimize effect on labor outcomes and breastfeeding. Combined spinal-epidural/patient-controlled analgesia may be preferable. Labor medications may cause more infant weight loss during the first days postpartum. Ensure good breastfeeding support for the mother and follow-up care postpartum hospitalization. | |||||
| For Cesarean Section | ||||||
| Regional anesthesia | Regional anesthesia (e.g., epidural, intrathecal/spinal) preferred over general. Minimize separation of mother/baby and initiate breastfeeding as soon as possible; this includes in the operating room or recovery room, when feasible. Mother may breastfeed postoperatively as soon as she is alert enough to hold the baby. | |||||
| For Postpartum Period | ||||||
| Acetaminophen/ibuprofen | Safe, effective in postpartum mothers | |||||
| Parenteral ketorolac | Contraindicated in breastfeeding women (U.S. boxed warning) | |||||
| Diclofenac suppositories | Not available in the United States | |||||
| COX-2 inhibitors | Some theoretic advantages if maternal bleeding is a concern, but should be balanced with higher cost and possible cardiovascular risks; should be used minimally and short-term in healthy young women | |||||
| All analgesics | Nonopioid analgesics should generally be first choice for pain management in breastfeeding women due to lack of impact on maternal or infant alertness. Because both pain and opioid analgesia may negatively impact breastfeeding, mothers should be encouraged to control pain with lowest effective dose. Opioid analgesia postpartum may affect babies' alertness and suckling vigor. Encourage mothers to control pain, especially after cesarean birth or severe perineal trauma requiring repair. | |||||
| Meperidine (IV) | Avoid giving to breastfeeding mothers due to reports of neonatal sedation. Additional concerns with intrapartum administration include cyanosis, bradycardia, and risk of apnea. | |||||
| Morphine (IV or IM) | Moderate-to-low doses are preferred as the passage to milk and oral bioavailability in infants are lessened. | |||||
| Patient-controlled IV analgesia (PCA) | Morphine or fentanyl is preferred to meperidine. | |||||
| Nalbuphine (IV) | Enters breast milk; use caution | |||||
| Butorphanol (IV) | Enters breast milk; AAP indicates it is a reasonable choice | |||||
| Pentazocine (IV) | Enters breast milk; use caution | |||||
| Hydromorphone | Seven to 11 times as potent as morphine; low concentrations found in breast milk; nonopioid analgesics preferred | |||||
| Hydrocodone, codeine (oral) | Use lowest dose for shortest duration; observe infant for increased sleepiness, difficulty feeding or breathing, or limpness; higher doses (e.g., 10 mg hydrocodone) and frequent use may cause infant sedation. | |||||
| Epidural/spinal medications | Single-dose opioids (e.g., neuraxial morphine) minimal effects on breastfeeding due to negligible maternal plasma levels achieved; very low doses of morphine are effective. Continuous postcesarean epidural infusion may be effective pain relief that minimizes opioid exposure. | |||||
| For Surgery | ||||||
| ||||||
Epidural anesthesia has become an increasingly popular method of pain control during labor and is commonly administered to women who are planning to breastfeed. Evidence of an association between maternal analgesia and disorganized infant behavior was first reported in 1961 [171]. The theory that epidural anesthesia given intrapartum inhibits breastfeeding has not been conclusively supported by medical research or literature [160,167,172,173,174,175,176,177,178]. Some research has indicated that intrapartum epidural analgesia may negatively affect the establishment of breastfeeding, but the results are inconclusive [179,180]. A 2016 systematic review of the relationship between epidural anesthesia and its effects on breastfeeding included 23 studies. Of these studies, 12 showed negative associations between epidural analgesia and breastfeeding success, 10 showed no effect, and 1 showed a positive association [181]. A 2019 observational cohort study examined the effect of vaginal delivery with and without epidural analgesia on breastfeeding status six weeks postpartum [182]. A total of 1,204 women of mixed parity (398 nulliparous, 806 multiparous) who intended to breastfeed were included in the study. The nulliparous women received epidural analgesia at a higher rate than did the multiparous women (84.9% vs. 61.8%, respectively). Multiparous women were more likely to breastfeed at six weeks compared with nulliparous women (80.0% vs 70.6%, respectively). The overall breastfeeding rate at six weeks was 76.9%; it was lower among women delivering with epidural analgesia (74.0%) compared with women delivering without epidural analgesia (83.4%). Among a subset of multiparous women with previous breastfeeding experience, the breastfeeding rate did not differ between women delivering with and without epidural analgesia. The authors concluded that offering lactation support to women with no prior breastfeeding experience could improve breastfeeding success [182].
Reported complications to the mother from the administration of epidural analgesia include [168,183,184,185,186,187,188]:
Sudden drop in blood pressure. This may cause fetal distress and may require treatment of the mother with IV fluids, other medications, and oxygen.
Severe headache caused by a leakage of spinal fluid (<1%)
Slowed labor due to restricted movement
Shivering, ringing of the ears, backache, soreness at injection site, and nausea
Difficulty pushing, which may require additional interventions (e.g., oxytocin, forceps, vacuum extraction, or cesarean)
Numbness in the lower half of the body for a few hours after birth
Nerve damage (rare)
Some of these complications were addressed in a 2018 update to an existing Cochrane Review that assessed the efficacy and safety of all types of epidural analgesia, including combined spinal-epidural (CSE), when compared with nonepidural or no pain relief during labor [189]. Low-quality evidence supported epidural analgesia over nonepidural methods for reducing pain during labor and increasing maternal satisfaction with pain relief. Epidural analgesia had no impact on the risk of caesarean section or long-term backache and did not appear to have an immediate effect on neonatal status as determined by Apgar scores or in admissions to neonatal intensive care [189].
The CSE pairs the advantages of the popular epidural anesthesia with the additional advantages of spinal injection. CSE is believed to improve maternal mobility during labor and provide more rapid onset of analgesia than the epidural alone; however, research has indicated little basis for offering CSE over epidurals in labor [190,191]. CSE also may cause side effects, such as severe headache [192,193].
Many hospitals have established a period after delivery during which mother and child should not be disturbed. This is sometimes referred to as the "babymooning" period. The first two hours following birth are the optimal time for infants to initiate breastfeeding behaviors, such as rooting and sucking. During this sensitive period, infants are most responsive to tactile, thermal, and odor cues from their mothers. Sensitive periods in biologic phenomena are times when events can alter later behaviors [1,194]. A sensitive period occurs in infancy when success in establishing a skill (e.g., breastfeeding) may enhance survival and the ability to adapt [194]. For women, the first few hours after delivery are a sensitive period for bonding. If immediate contact is delayed (e.g., for general anesthesia, premature birth, illness), the bonding process will also be delayed until the mother's first contact with the infant. Delayed initial contact may result in bonding difficulties [1].
Immediately following birth, infants experience a surge of high levels of circulating catecholamines, which causes extreme sensitivity to odor cues. These cues guide the infant toward the mother's nipples. When skin-to-skin contact is initiated immediately after birth, an alert, active, unmedicated newborn may crawl unaided toward the mother's nipple and latch correctly within about 60 minutes [194,195,196,197]. The baby is most willing to nurse in the first two hours following birth because the suckling reflex is strongest during this period. When gratification of the suckling reflex is delayed, it may make it more difficult for the infant to learn to suckle later. After those first two hours have passed, the baby may be quite sleepy. Some infants may not nurse as vigorously as the mother expects following birth or may instead merely "nuzzle" or lick the breast. It is important to reassure the mother that this is normal positive behavior and not a "rejection" of her or her breast. The baby should still be offered the opportunity to feed at breast eight or more times in the first 24 hours, but the normal sleepiness that follows the initial feeding may necessitate alternate ways to stimulate the breast and provide supplementation, such as hand expression and spoon feeding [2].
Although a tradition of separating the mother and baby after birth persists in many parts of the world, including the United States, early mother-infant skin-to-skin contact has been recommended by the AAP and is associated with many benefits, including [49,194,198,199]:
Longer duration of breastfeeding
Positive influence on the mother-infant interaction for as long as 1 year
Reduction in infant crying, which in turn has been associated with increased infant heart and respiratory rates
Positive significant impact on infant health
Correct skin-to-skin contact involves placing the infant prone and skin-to-skin on the mother's chest or abdomen immediately following birth. The infant should be gently dried and covered loosely with a warmed blanket; a cap will prevent heat loss. Routine procedures (e.g., assigning Apgar scores, suctioning) may be done if indicated, and the initial physical assessment may be done at this time as well. Ideally, all other interventions (e.g., weighing, measuring, bathing, needle-sticks, eye prophylaxis) should be delayed until the first breastfeeding has been accomplished [21,194].
When a sick or premature infant is separated from its mother in a neonatal intensive care unit (NICU), the "mothering" duties will be assumed by other caregivers. Early discharge practices may further separate the mother from her infant, and she may feel that she is a stranger to her own baby. The ability to counsel the breastfeeding woman in this situation may significantly impact her success [200]. Skin-to-skin contact, peer support, simultaneous breast milk pumping, multidisciplinary staff training, and the baby-friendly accreditation of the hospital have been shown to be effective breastfeeding promotion activities for mothers whose infants are assigned to the NICU [201].
Breastfeeding can be unconsciously sabotaged by hospital routines. Newborn policies can have serious effects on lactation and may contribute to maternal engorgement and routine assignment of any cesarean section baby to the nursery for several hours after birth. In addition to ensuring skin-to-skin contact between mother and infant immediately following birth, nursery and postpartum routines that support lactation include [1,2,21,200,202,203,204,205,206,207,208]:
Support the mother's decision to breastfeed. When direct breastfeeding is not possible, expressed human milk should be provided. The newborn should have access to the breast around the clock. The baby will need at least 8 to 12 feedings per day during the first week of life. Both mother and baby will get off to the best possible start breastfeeding if the baby remains with the mother (rooming-in). Breastfeeding mothers should be advised to feed at night as well.
Avoid take-over behavior. The mother is the expert where her baby is concerned. Some hospitals have instituted a "hands-off technique" that emphasizes ways to teach mothers without doing tasks for them.
Minimize or modify the course of maternal medications that may alter the infant's alertness and feeding behavior.
Avoid procedures that may interfere with breastfeeding or traumatize the infant (e.g., unnecessary, excessive, over-vigorous suctioning).
Avoid giving supplements (e.g., water, glucose water, formula, other fluids) to breastfeeding infants unless physician ordered and medically indicated; normal, healthy newborns do not need supplementation. Additionally, some infants may have breastfeeding difficulties after being offered bottles for supplementation. The baby's initial recognition of the mother involves the distinctive features of the mother's nipple. If an infant who is learning to breastfeed receives supplementation, either from a bottle or a pacifier, the nipple-recognition signals become mixed, resulting in what has been referred to as "nipple confusion." Although the existence of nipple confusion has not been universally accepted, studies have shown that supplementation and the introduction of a foreign nipple are associated with decreased rates of continued breastfeeding.
Avoid pacifier use during initiation of breastfeeding and until after it has been well established.
Assess breastfeeding progress and success. Regularly observe the mother's breasts and nipples for any signs of lactation problems (e.g., pain, cracks, blisters, bruises). Also, regularly observe a feeding, paying attention to:
How the baby anticipates the feeding (e.g., crying, patient, impatient). The baby should flex toward the breast when anticipating a feeding.
How the baby roots to the breast. The baby should be relaxed, in a state of flexion.
The position of the baby in relation to the mother. Note the mother's body language and tension.
How the baby latches on to the breast.
How the mother supports her breast.
Observe for infant swallowing (short, faint sounding "ah" with the expiration of air between suckling sequences). Note any gulping, clicking, or popping sounds. The baby follows a pattern of suck-swallow-breathe. Note the ratio of sucks to swallows. Typically, colostrum averages three to four sucks per swallow. Mature milk averages one to two sucks per swallow. Note any choking, coughing, or sputtering that occurs during the feeding; note frequency and duration of pauses or if the baby pauses with periods of increased respiration after several sucking sequences.
Note how the nipple appears immediately after feeding. Nipple comes out of the infant's mouth round and of equal color with the areola. Note abnormal shape, blisters, and/or blanching.
Following a feeding, the baby should be calm, satiated, and relaxed. Note irritability and/or fussiness.
Assess the infant. A weight loss of 7% is average in the first week for the breastfed infant; a weight loss of 10% or greater requires a skilled assessment and follow up. If the baby loses 7% in the first 72 hours, breastfeeding should be observed for proper technique and milk transfer. If the baby is nursing well, appears otherwise healthy, and has appropriate stooling patterns, another weight check should be scheduled in two days.
Continuity of information is vital in the postpartum period. Physician instructions to the patient should not conflict with the nurse's teaching plan (Table 10). The hospital should have a written breastfeeding policy that is known to all staff members. Teaching plans and handouts for new mothers should be written at a 5th grade reading level. Verbal discharge instructions should be avoided. Discharge instructions should be written into the hospital policy and not left to chance.
KEY POINTS FOR BREASTFEEDING TEACHING
|
Instruct the mother to nurse the baby on cue, making sure the baby feeds at least eight or more times per 24 hours. Also instruct the mother to wake the baby by placing skin to skin if baby is not waking themselves for feedings. If baby is not waking themselves for feedings, parent can hand express and spoon feed expressed milk every two hours during the day and every four hours at night for the first 24 hours of life. . Assure the mother that weight loss is common and that the infant should regain their birth weight by 2 to3 weeks of age. At hospital discharge, the mother should be given simple, specific instructions about when to feed the baby, normal infant behaviors, how to tell if the baby is getting enough milk (i.e., number of wet diapers, stools), and community resources. A plan can be made for a follow-up visit with a lactation consultant to develop a feeding plan if breastfeeding is a challenge at hospital discharge. Babies should be seen by a primary care provider between 3 and 5 days of age. If all is well, the baby will have gained weight from the hospital discharge weight. Primary care providers should be knowledgeable of lactation consultants in their community for needed referrals for continued feeding challenges.
A key to successful breastfeeding is getting off to a good start. Some babies latch easily after birth, and some babies and mothers need a little help. The latch-on process takes only a few seconds, and if done correctly, the baby will learn how to obtain the most milk with the least amount of effort. The four main steps in the latch-on process are positioning, breast support, rooting/offering the breast, and latching on.
When the baby is well-aligned, the mother should be able to draw a straight line that connects the baby's ear, shoulder, and hip on either side of the baby's body. Once the mother and the baby are in proper position, the next step is for the mother to guide the infant to the breast so he or she can latch on properly and nurse. Unless the mother is reclining, it will be necessary for her to support her breast, at least in the early days of breastfeeding, so the baby can attach properly. This is especially true as milk production increases the size and weight of the mother's breast [209].
Mother should be taught to support her breast by cupping it in her hand, placing her thumb on top of the breast, and her fingers below the breast, well behind the areola. This is called the "C-hold" because the mother's hand makes the shape of the letter C. Practicing the breast hold in front of a mirror is often helpful. The weight of the breast should rest in her hand in a neutral position. Many women lift, pull, or twist the breast to reach the baby. The mother should be reminded to bring the baby to the breast, not take the breast to the baby [209].
Latching can be baby-led or parent-led. In parent-led latching, the rooting reflex is stimulated by the mother offering the breast and stroking the infant's lips with her nipple. The mother uses her hand in the C-hold to move her entire breast up and down, brushing the infant's lips with her nipple. Many mothers are tempted to use their index finger to stroke the lips or try to pry open the baby's mouth. However, this may interfere with the baby's attempt to latch on, and some infants may react to prying efforts with a tight clamp. The mother should continue tickling the lips until the infant opens his or her mouth very wide, as if giving a big yawn [209].
Today, however, there is an emphasis on allowing infants to initiate breastfeeding, sometimes called baby-led latching. Assist the mother into a comfortable position and allow the infant to make his or her way to the breast. The infant's reflexes will take over, and he or she will self-attach with little or no assistance.
When the baby opens wide, the mother should aim the nipple toward the baby's palate (roof of the mouth) and pull the infant in close (Figure 2). This motion should be done quickly, but gently, while the baby's mouth is wide open. The baby's head should never be held, pushed, or shoved into the breast. This may disturb the rooting reflex and startle the infant, and he or she may refuse to nurse [1,2,209]. The tip of the baby's nose should touch mother's breast. Mother should be able to look down and see the baby's nose. If she cannot see the baby's nose, she should either lift up the breast slightly or raise the infant's buttocks so the infant is level.
Mothers often worry about the baby's nose. It seems natural to use the thumb to pull the breast tissue away from the baby's nose. This action, however natural, will shift the nipple in the baby's mouth. This shift may partially pull the nipple out so that the baby is latched-on only to the nipple tip. Painful nipple cracking may result.
All human babies are born with turned up noses. The tip of the nose will touch the breast with plenty of room for air in the open spaces formed around the nostrils. Point out this air space to mother and reassure her. Breathing comes before eating. If a baby cannot breathe, he or she will not stay at the breast.
If the infant is correctly latched on to the breast, the mother will feel a tugging sensation. The first few suckles may be a bit tender as the infant pulls the nipple into the back of the mouth but should not last more than a short time. The nipple may remain sensitive for a few days after delivery. Most mothers report mild latch-on pain in the first days to weeks that disappears within the first 30 seconds of a feeding. Pain beyond the first minute or so of nursing can be a sign of improper latching and should be evaluated. If the pain persists past the first minute of feeding, she should detach the baby by inserting her finger in the corner of his or her mouth to break the latch and try again. Baby may need to go on and off the breast several times before the latch is correct [209].
If the baby is actively feeding, baby can remain on the breast. Active feeding signs include long sucking bursts, swallows, pauses between sucking bursts, and the baby's eyes may or may not be open during a portion of the feeding. In the early days, if the baby falls asleep easily, mother should use compression of her breast and switch breasts when suckling and swallowing slows. If the baby does not nurse at both breasts during a feeding, the mother should start the next feeding on the breast from which baby did not feed. Mother should watch the baby and the active feeding cues to determine when the feeding ends rather than the clock.
To take the baby off the breast, mother should press down on her breast, near the baby's mouth, press down on baby's chin, and insert a finger in the corner of the baby's mouth [131,210].
Parents should be instructed to watch for feeding cues, such as [2]:
Eye movement under closed lids
Increased alertness
Movements of arms and legs
Tossing, turning, or wiggling
Mouthing
Rooting
Changes in facial expression
Squeaking noises or light fussing
Crying (late sign of hunger)
As discussed, the baby should feed approximately 8 to 12 times or more in 24 hours. Instruct the mother that she should attempt to wake the baby for feedings if the baby is not on track to feed at least 8 to 12 or more times in 24 hours. If the baby is sleepy in the first 24 hours, the birthing parent can use hand expression to remove milk and feed to the baby an appropriate number of times in the first 24 hours. The baby should become more alert and eager to nurse after the first 24 hours [2].
Following the initial quiet alert stage after birth, baby goes into a deep sleep, recovering from the rigors of labor. Deep sleep is followed by increased wakefulness and increased nursing demands called "cluster feeding." Mothers often interpret cluster feeding as an indication that their baby is not getting enough milk [2]. This is the prime time during which mothers request supplemental bottles. Anticipatory guidance about these episodes can prevent the mother from assuming her milk is inadequate and giving supplemental bottles. Assure the mother that cluster feeding is normal and that it is actually a series of mini-feedings that are often followed by another period of deep sleep. Mother should be encouraged to catch up on her own sleep between cluster feedings [2].
Mothers should be encouraged to experiment with different breastfeeding positions prior to discharge from the hospital. There is nothing magical about a particular position, and she should be encouraged to use whatever position works best for her and her infant. The most frequently taught techniques are discussed below [2].
This position is ideal for early breastfeeding and for premature babies[131]. Mother should sit upright in a comfortable chair with armrests and should hold her baby crosswise in the crook of the arm opposite the breast she will feed from (i.e., the left arm for her right breast; the right arm for her left breast). Mother should support the baby's trunk and head with her forearm and palm and place her other hand beneath her breast in a C- or U-shaped hold. Baby is tucked around mother's body, so baby reaches up toward nipple, mother guides the baby's mouth to her breast. Mother should not bend over or lean forward but should cradle her baby close to her breast [210].
This is similar to the cross-cradle hold, but mother supports the baby with the arm on the same side as the nursing breast, rather than the opposite arm [210]. Mother should be comfortably sitting up, with her back supported by pillows. Her forearm can be supported with a pillow to keep the baby level with her breast. Baby's back or side should be lying along mother's forearm with his or her head nestled in the crook of her elbow. Baby's lower arm can be tucked under mother's arm or placed along the side of mother's body. Baby should entirely face mother in a straight line; the neck should not be twisted over his or her shoulder. Baby should be in a snug position with face, chest, and knees turned toward mother in a tummy-to-tummy position.
This position may help baby latch onto mother's breast in the early days of breastfeeding, especially if the mother has had a cesarean section. It may also be a good choice if mother is tired [131,210].
Mother should be well supported at a 45-degree angle to the mattress. She should not be lying completely on her side. Baby should be placed on his or her side, facing mother. Mother should cradle the baby's head with her lower arm resting on the bed. Baby's body should face the mother in a straight line. Mother should offer her breast in the same manner as when sitting, using the C-hold. As baby roots, mother can slightly roll him or her toward her, to ensure that baby is placed correctly at the breast.
The football hold is a good choice if mother is recovering from a cesarean section, has large breasts, or is nursing twins [11,210]. The position offers a clear view of the infant's mouth and gives the mother greater control over the infant's head. This can be of great assistance if the baby nurses in an overexcited manner (e.g., roots back and forth across the nipple without latching on).
Mother should be in a sitting position with a pillow placed beneath her forearm for support. Baby is placed next to mother with baby's body turned toward her side. Baby should rest in mother's forearm, with the base of his or her head in mother's hand and buttocks near the bend of mother's elbow. Mother's thumb and index finger should be spread around baby's head, just below or at the level of the ears, depending on the size of the mother's hand. The heel of mother's hand should rest below the baby's neck on the posterior aspect of the upper thorax. If the upper back is not supported, baby will arch away from the breast with his or her head and neck.
The latch-on process remains the same except that mother should lean forward slightly, toward the baby, to elicit the rooting reflex, and then lift the baby up to the breast with the latch-on. By rotating her wrist, mother can straighten baby's head so his or her cheeks are of an equal distance from the breast. Mother should then lean back with baby and relax.
In the early postpartum period, the mother of twins may find it easier to feed the babies individually at first. This will enable her to learn the latch-on process, one baby at a time. After she is comfortable with the latch-on process, she should be encouraged to attempt to get both infants to feed simultaneously. Feeding both babies on the same schedule will help the mother with better utilization of her time. An extra set of hands will be a great help with the latch-on. Several large pillows will help support the babies and bring them as close to the breast as possible before latch-on.
Thus far we have looked at normal, uncomplicated vaginal delivery. For most mothers, nursing immediately after delivery is not a problem. For the mother or infant with medical complications, some adjustments must be made.
If the mother has a cesarean section under epidural or spinal anesthesia, she can nurse the baby when the operative field is cleared and she is alert and oriented. Ask the mother how she wants to hold the infant. If she is comfortable holding the baby in the cradle position, offer pillows for support. Some mothers may be hesitant to hold the baby at all until they have been reassured that it will not increase their incisional pain. The football (clutch) hold might be useful for mothers with this concern. As the incisional pain decreases, the mother should be instructed and assisted in the use of other positions. By day two or three postpartum, the side-lying position generally is comfortable, if the mother is supported with pillows at her back and beneath her abdomen [2]. If baby cannot breastfeed, provide mother with a breast pump and assist her in pumping her breasts to total approximately eight times in 24 hours [2].
Unfortunately, many hospitals classify all cesarean section babies as "ill" for a period of hours following delivery, often assigning the infants to a special care nursery. Nursery personnel should be told that the mother intends to breastfeed, and they should not offer bottles unless medically necessary. Healthy babies should be brought to the mother either in the recovery room or as soon as she has been released from recovery to the postpartum unit. The baby should not be kept in the nursery at night for feedings. If the mother is well, it is unwise to offer feedings in the nursery as this increases the risk of nipple confusion, flow preference, neonatal jaundice, and maternal engorgement.
Cesarean mothers may worry about the pain medication they need and the effect it will have on breastfeeding. Some mothers "do without" pain medication because they believe it will harm the infant. Maternal pain medications given in the hospital after the delivery are generally considered safe for breastfeeding. However, it is important to note that if the mother is taking pain medication, someone should be at the bedside, such as the father, who is given the responsibility to keep the baby safe.
Occasionally, the mother is too ill to be concerned with breastfeeding. She may be in the intensive care unit for several hours to several days until her condition is stable. Primary lactogenesis will occur without breast stimulation. As soon as the mother is able, she should begin pumping her breasts or nursing the baby. Occasionally, in the case of a severe hemorrhage for example, milk production can be minimal for a few days, until the mother has recovered; her additional reserves are being used to heal her body. Until the crisis has passed, the breasts may or may not produce adequate volumes of milk.
Although primary failure of lactogenesis is rare, warning signs that require watching include [1,2]:
Acute illness in the mother
Severe toxemia of pregnancy (pregnancy-induced hypertension) and HELLP syndrome (hemolysis, elevated liver enzymes, and low platelet count)
Severe hemorrhage (hemoglobin level below 7) and/or anemia
Sheehan syndrome (infarct of the pituitary associated with severe postpartum hemorrhage or shock)
Any mechanical change in the breast (reduction or augmentation surgery)
Primary glandular insufficiency. Provisions should be made for the babies of these mothers to be followed closely for weight gain after hospital discharge
Maternal insulin increases glucose uptake by the mammary cells. The mother's glucose utilization may increase by 30% when lactation is established. Glucose is used for the production of lactose and is critical to the volume of milk produced [1]. The breast is a target organ for insulin, where rapid changes in insulin levels result in changes in the rate of lipogenesis and the utilization of glucose. Lactation has an insulin sparing effect [1]. Significant lactation problems arise for the diabetic mother when infants are kept in the nursery for observation and are bottle fed in the first few days.
Women with insulin-dependent type 1 diabetes and gestational diabetes should be encouraged to breastfeed [211,212]. Several studies have shown that breastfeeding reduces the risk of contracting childhood type 1 diabetes. The mother's blood glucose should be carefully monitored in the postpartum period, one day each week until her first postpartum visit, again in three to six months, and then annually thereafter [213]. The diabetic mother may be prone to hypoglycemia and require her insulin dose adjusted appropriately. The renal threshold for glucose is lowered in pregnancy. During lactation any lactose reabsorbed from the breasts is excreted in the urine. Lactosuria may be misdiagnosed as glucosuria, so the use of urine test strips is inappropriate [1].
Infants born to diabetic women are at higher risk for hypoglycemia. Early and frequent breastfeeding may facilitate glycemic stability in infants born to women with gestational diabetes [214,215].
Hypoglycemia is the most common metabolic problem in neonates. The overall incidence in newborns varies from 1.3 to 3 per 1,000 live births. Early feeding has been shown to decrease the incidence [216]. Hypoglycemia has been defined as a blood glucose level of less than 30 mg/dL in the first day of life and less than 45 mg/dL thereafter in the newborn [2,216]. Healthy infants experience transient hypoglycemia as a part of the normal adaption to extrauterine life, with a decline in blood glucose concentrations to values as low as 20–25 mg/dL in the first two hours of life [217]. However, blood glucose concentrations in healthy infants not at risk for hypoglycemia are not routinely measured; thus, it is difficult to define "normal" levels of blood glucose in the first 48 hours of life. For this reason and others, controversy exists over the quantitative definition of hypoglycemia in the newborn [217,218,219]. Hypoglycemia in neonates can cause apnea, irritability, lethargy, seizures, and brain damage, and prolonged or symptomatic hypoglycemia may correlate with long-term neurodevelopmental deficits. However, the long-term significance of early, asymptomatic, and transiently low glucose levels is not well established [217].
Infants born to diabetic women are at higher risk for hypoglycemia [217]. Early and frequent breastfeeding may facilitate glycemic stability in infants born to women with gestational diabetes [214,215,216]. Supplementation or "complimentary feeding" after breastfeeding is strongly discouraged. It can put both the mother and the infant on a downward spiral toward breastfeeding failure.
For cases in which the infant is at risk, supplemental feedings of expressed mother's milk or ABM may be required to maintain the infant's blood sugar. Supplementation after every breastfeeding, without documentation of continued low blood sugar, can greatly affect maternal milk production and the infant's willingness to breastfeed.
If breastfeeding is poorly tolerated and if hypoglycemia is continuing, intravenous glucose or ABM should be given, but not glucose water [217]. The use of glucose water to raise blood sugar is not recommended because it raises the blood sugar rapidly. The sharp increase in the blood glucose level stimulates the infant to produce insulin, and a rebound effect occurs. The increased insulin level lowers the blood sugar further [1,2].
Of all the topics connected with breastfeeding, jaundice remains the most confusing and misunderstood. Few issues in management of the newborn infant have generated as much confusion, uncertainty, and anxiety [17]. The aggressive management of jaundice in the past few decades, like so many things associated with breastfeeding, may be the result of faulty research. Poorly designated breastfeeding groups, definitions of hyperbilirubinemia, defining a "normal" serum bilirubin level that applies to all newborn populations, and measurement techniques have confounded the results of many studies [1,220,221].
Few evidence-based studies have linked uncomplicated neonatal hyperbilirubinemia with significant morbidity or mortality in term (38 to 42 weeks' gestation), healthy newborns [222]. If the breastfed infant's physiology is viewed as the norm, then the ABM-fed infant should be viewed as an artificially created physiologic example [223]. Perhaps the elevated bilirubin levels serve some valuable physiologic purpose that to date remains unclear.
Aggressive management styles and misinformation may cause some parents to believe that jaundice is a terrible illness. The diagnosis and treatment of jaundice have been associated with persistent maternal perceptions of infant vulnerability [2,224,225]. Death from physiologic neonatal jaundice is rare [221]. To mitigate parent worries and misperceptions, clinicians should provide parents with written and verbal information about newborn jaundice prior to discharge from the birth facility [221]. A novel detection tool—the two-color icterometer—appears to have the potential to facilitate parents' early detection of clinically significant jaundice and to help them decide when to seek medical assistance [221,226]. A smartphone application also has been developed to assess neonatal jaundice. The app analyzes data on color values from digital images to provide an estimate of total serum bilirubin [227].
Healthcare professionals should become familiar with all forms of neonatal jaundice, as well as current recommendations for diagnosis, testing, and treatment (Table 11) [222,228,229].
TYPES OF JAUNDICE
| Type | Causes | Onset | Peak | Feeding Behavior | Risks |
|---|---|---|---|---|---|
| Physiologic jaundice of the newborn | Normal reduction in fetal hemoglobin levels | Day 3 | Day 7 | Good | None if levels kept below 20 mg/dL |
| Improper breastfeeding jaundice |
Normal reduction in fetal levels plus:
| Day 3 | Day 7 | Poor | None if levels kept below 20 mg/dL |
| Breast milk jaundice | Unknown | Day 10–14 | Day 20–40 | Good | Self-limited and benign |
Jaundice (or icterus) is a condition marked by yellow staining of body tissues and fluids, resulting from excessive levels of bilirubin in the bloodstream. It is not usually visible until the total bilirubin level rises above 3 mg/dL. Jaundice is a symptom of an array of illnesses [230]. More than 50% of all newborns have some degree of visible jaundice. However, visual confirmation alone has been found to be unreliable and inaccurate [2,220,229]. All infants who appear jaundiced should be evaluated with a risk score or total serum bilirubin/transcutaneous bilirubin (TSB/TcB) measurement. A combination of universal screening and risk factor scoring seems to be the most effective method for identifying infants at risk for hyperbilirubinemia [229].
Jaundice in the newborn is the result of a normal elevation of unconjugated bilirubin and often referred to as "physiologic jaundice of the newborn" [2,221]. This is a common clinical condition that occurs in roughly two-thirds of all newborns. It disappears in about one week in the ABM-fed infant but may persist for several weeks in the breastfed infant. This pattern is known as "breast milk jaundice" [2,230].
Breast milk jaundice is not a disorder but rather a normal extension of neonatal jaundice that results from increased intestinal absorption of bilirubin, caused by the effect of an as yet unidentified substance in breast milk and/or delayed establishment of enteric flora [221]. The unidentified factor appears only with secretion of transitional and mature milk (not in colostrum) and coincides with observed identical serum bilirubin concentrations during the first five days of life in both the normal ABM-fed infant and the adequately breastfed infant. Higher values in breastfed infants typically occur only after the fifth day [221]. Discontinuation of breastfeeding, even for a day or two, is rarely necessary [2]. Infants with breast milk jaundice typically remain healthy and gain weight adequately [231].
Because breast milk jaundice is a prolongation of neonatal jaundice, it is typically identified after the infant has been discharged from the hospital. "Starvation jaundice," however, may be seen as early as the first few days after birth, although not before 24 hours. Starvation jaundice has been observed in infants who have not established adequate feedings. Whether due to maternal or neonatal factors, infants who have not established adequate breastfeeding should not be discharged. Breastfeeding evaluation, encouragement and training for the mother (e.g., positioning, latching), and continuation of breastfeeding to restore fluid and caloric intake should occur [2]. Poor caloric intake and/or dehydration associated with inadequate breastfeeding may contribute to the development of hyperbilirubinemia. Increasing the frequency of nursing decreases the likelihood of subsequent significant hyperbilirubinemia in breastfed infants [222].
Risk Factors
Identifying the few infants at risk of developing marked hyperbilirubinemia is a clinical challenge. The clinical, epidemiologic, and genetic risk factors that have been associated with severe hyperbilirubinemia include lower gestational age, exclusive breastfeeding, glucose-6-phosphate dehydrogenase deficiency, ABO hemolytic disease, Asian ethnicity, jaundice observed in the first 24 hours of life, cephalohematoma or significant bruising, and history of a previous sibling treated with phototherapy [221,228,229,232].
Kernicterus
Most jaundice is benign; however, because of the potential toxicity of bilirubin, newborn infants must be monitored to identify those who might develop severe hyperbilirubinemia and, in rare cases, acute bilirubin encephalopathy or kernicterus [222,229]. Bilirubin encephalopathy describes the clinical central nervous system findings caused by bilirubin toxicity to the basal ganglia and various brainstem nuclei. To avoid confusion and encourage greater consistency in the literature, the AAP Subcommittee on Hyperbilirubinemia recommends that in infants the term "acute bilirubin encephalopathy" be used to describe the acute manifestations of bilirubin toxicity seen in the first weeks after birth and that the term "kernicterus" be reserved for the chronic and permanent clinical sequelae of bilirubin toxicity [222]. Kernicterus has been associated with choreoathetoid cerebral palsy, sensorineural hearing loss, gaze paresis, and intellectual deficits [1,222,233,234]. Hyperbilirubinemia alone has not been found to be sufficient to account for these neurologic findings. For example, infants with extremely high levels of serum bilirubin but no apparent sequelae have been reported, and infants without documented high serum levels of bilirubin have been found to have kernicterus [234].
Excessive unbound bilirubin in the infant's system can also be deposited in various tissues. Bilirubin is toxic to the tissues, resulting in cell necrosis. Damage also may occur in the kidneys, intestines, and pancreas. Premature infants are particularly susceptible to bilirubin-related brain damage [1,222]. Kernicterus rarely occurs with serum bilirubin concentrations less than 20 mg/dL, yet controversy persists over what potential neurologic deficits may occur with levels less than 20 mg/dL [1,234,235].
Preventing Severe Neonatal Jaundice
To prevent the onset of neonatal jaundice severe enough to require treatment, the AAP has recommended the following [1,2,220,221,222,228,236]:
Promote and support successful breastfeeding and avoid routine supplementation.
Establish nursery protocols for the identification and evaluation of hyperbilirubinemia.
Measure the TSB/TcB level in infants jaundiced in the first 24 hours.
Recognize that visual estimation of the degree of jaundice can lead to errors, particularly in infants with darker skin tones.
Interpret all bilirubin levels according to the infant's age in hours.
Recognize that infants at less than 38 weeks' gestation, particularly those who are breastfed, are at higher risk of developing hyperbilirubinemia and require closer surveillance and monitoring.
Perform universal bilirubin screening using TSB or TcB measurements before discharge to assess risk of subsequent severe hyperbilirubinemia.
Provide parents with written and verbal information about newborn jaundice.
Provide appropriate follow-up based on the time of discharge, predischarge TSB/TcB, gestational age, and other risk factors.
Treat newborns, when indicated, with phototherapy or exchange transfusion.
Test all pregnant women for ABO and Rh(D) blood types and have a serum screen for unusual isoimmune antibodies. Erythroblastosis fetalis is a hemolytic disease of the newborn marked by anemia, jaundice, enlargement of the liver and spleen, and generalized edema [230]. It occurs when the mother's body has produced antibodies against the Rh factor of the fetus. The maternal antibodies cross the placenta and destroy the baby's red blood cells. The resulting hemolytic anemia and its sequela are life-threatening for the baby. Historically, Rh isoimmunization was an important cause of severe jaundice, often resulting in the development of kernicterus. Although this condition has become relatively rare in industrialized countries following the use of Rh prophylaxis in Rh-negative women, Rh isoimmunization remains common in developing countries.
Additional recommendations include [1,222,231,237]:
Encourage mothers to nurse at least 8 to 12 times every 24 hours. Colostrum has a laxative effect and assists in emptying the bowel of meconium. Human milk coats the gut, preventing the reabsorption of intestinal bilirubin and furnishing the natural intestinal flora and enzymes that enhance bilirubin transport.
Observe the baby for proper latch-on and suckling when put to breast. Swallowing initiates peristaltic movement that will aid in emptying the bowel.
Observe the baby's voiding and stooling frequency. Because stool and urine output are one of the measures of breastfeeding effectiveness, expect the infant to have appropriate output if feedings are going well.
Avoid the use of complementary ABM feedings and water supplementation. Both contribute to neonatal flow preference and maternal engorgement. Supplements will reduce the infant's suckling urge, decreasing colostrum and breast milk intake. Researchers have consistently noted an increase in bilirubin levels with the routine use of glucose water after nursing. There is also evidence that ABM solids coat the gut differently than human milk and can interfere with the normal processes of excretion and hydration.
Encourage the mother to nurse her infant at night. Prolactin levels rise with sleep, and stimulation of the breasts causes prolactin to rise further. Repeated elevations in prolactin level will encourage the early onset of an ample milk supply. As the milk matures, bilirubin levels drop. Night nursing is also important in prevention of engorgement, which often leads to cracked nipples, inhibited milk flow, and/or improper breastfeeding jaundice.
These simple steps should reduce the incidence of infants requiring phototherapy.
Treating Severe Neonatal Jaundice
Phototherapy is commonly used to treat hyperbilirubinemia, and severe hyperbilirubinemia may be treated with exchange blood transfusion [221]. Potential harms of phototherapy include weight loss, gastrointestinal problems, interruption of breastfeeding, disruption of the maternal-infant relationship, and possibly growth of melanocytic nevi. However, the U.S. Preventive Services Task Force has found inadequate evidence regarding the harms of phototherapy [234].
Exchange transfusion also may be used to treat extreme hyperbilirubinemia. Although death as a complication of exchange transfusion is rare, significant morbidity (e.g., apnea, bradycardia, cyanosis, vasospasm, thrombosis, necrotizing enterocolitis) has been shown to occur in as many as 5% of exchange transfusions, and the risks associated with the use of blood products should always be considered. Hypoxic-ischemic encephalopathy and acquired immune deficiency syndrome (AIDS) have occurred in otherwise healthy infants receiving exchange transfusions [234].
When an infant does require treatment, breastfeeding should be continued. The mother should be encouraged to nurse frequently and the appropriate support should be offered. The mother should have continuous access to her infant. Phototherapy and jaundice often make the baby sleepy and not interested in feeding. Mother may need additional help getting the baby awake for feedings. In no way should a mother ever be made to feel that it is her milk that is "bad" or that it is her milk causing the jaundice.
Since the late 1980s, more preterm infants of low gestational age and birth weight have been surviving. The percentage of preterm births in the United States rose 36% from 1984 to 2009 [238]. However, since 2010 the rate has been relatively stable, at 10.1% in 2021 [239,240]. Advances in technology have helped increase survival rates, and infant mortality rates for preterm infants are lower in the United States than in most European countries [238]. The milk from mothers of preterm infants is significantly different from that of mothers who deliver at term, and the composition of human milk changes in relation to how prematurely the infant was born. Discovery of the specialized nutritional and immunologic components in human milk have spawned enthusiasm for its use in the preterm infant.
Preterm infants fed human milk derive significant benefits, including host protection and improved developmental outcomes, when compared to formula-fed preterm infants. Studies have documented the following health benefits for preterm infants fed human milk [21,241,242]:
Decreased incidence and/or severity of a wide range of infectious diseases
Decreased postneonatal infant mortality rates
Decreased rates of SIDS in the first year of life
Reduced incidence of both type 1 and type 2 diabetes, lymphoma, leukemia, Hodgkin's disease, overweight and obesity, hypercholesterolemia, and asthma
Breastfeeding the preterm infant has also been associated with slightly enhanced performance on cognitive development and analgesic effect for infants undergoing painful procedures (e.g., screening heel-stick) [21]. For the mother whose infant is critically ill, human milk feeding becomes an important contribution to the infant's care. Additionally, it may encourage mother-infant attachment and provide maternal empowerment [241,243].
With preterm birth, the growth and development of the infant's gastrointestinal tract is interrupted and forced to continue under conditions that are less ideal than the environment in utero [244]. The fetus starts swallowing amniotic fluid at around 8 weeks' gestation in volumes of approximately 200–250 mL/kg/day [244]. At term, the baby may be swallowing as much as 500 mL/kg/day, through which it receives carbohydrates, proteins, peptides, electrolytes, lipids, enzymes, and hormones [1,244].
Human milk contains amino acids and fat blends that offer distinct advantages to preterm infants. The protein content of human milk, which is 30% casein and 70% whey, also is more suitable for preterm infants than ABM [243]. Human whey proteins contain lactoferrin, lysozyme, and secretory immunoglobulin A (sIgA), which help prevent nosocomial infections, sepsis, necrotizing enterocolitis, and bacterial and viral infections in the preterm infant [243]. The lipids in human milk also provide nutritional benefits. The fat globules in preterm milk are smaller, aiding in their absorption directly from the immature gut. Premature infants lack the enzymes and bile salts needed for digestion of fats. However, they absorb more than 90% of the fats in human milk [243]. The long chain fatty acids present in human milk have been associated with adequate neurologic growth. This unique protein structure, ability to promote fat absorption, and growth-promoting pattern of fatty acids makes human milk beneficial to preterm infants [243].
Despite the benefits derived from human milk, the incidence and duration of breastfeeding preterm infants remains less than that of term infants. This is primarily due to the challenges of breastfeeding that parents of preterm infants must face, including [19,241,243]:
Establishing and maintaining a milk supply
Transitioning from gavage feeding to breastfeeding
Providing adequate caloric and nutritional intake
While a good milk supply is critical to the success of breastfeeding, establishing and maintaining one can be challenging for a variety of reasons. For example, because of their immaturity and potential inability to suck effectively, preterm infants may be unable to breastfeed directly immediately after birth. This means that the mother must begin expressing milk immediately, sometimes for weeks or months, until her infant is physiologically stable and able to maintain nutritive sucking. Early expression is essential as delayed initiation may impair milk production and lead to possible breastfeeding challenges. Separation from her infant, due to the level of care the infant needs and/or prolonged hospitalization, can make maintaining milk supply uniquely challenging. If the infant's condition is precarious, the mother may be especially stressed and fatigued, which can further depress milk production [243,245].
If the baby is ill and feeding will be delayed for longer than 24 hours, mother should begin pumping her milk. It is crucial to have the mother begin pumping as soon as possible, at least within the first 24 to 48 hours after delivery to take advantage of the normal physiologic changes after parturition [2]. If the breasts are not stimulated, prolactin levels will fall by 50% within the first five days after giving birth and will drop to nonpregnant levels in seven days [2]. Very early pumping is for stimulation purposes. It is important to tell the mother that no one expects her to obtain large volumes of milk initially. Babies can get colostrum easily via negative pressure suction made while dropping the jaw during feeding , but colostrum does not pump out well [294]. Manual expression is recommended in the colostrum phase. What is important is the regular stimulation of the breasts to enhance the process of lactogenesis and to increase the milk supply. An abundant milk supply will assist later in the infant's transition to the breast. Counseling mothers of preterm infants has been shown to increase the incidence of breast milk feeding [246]. Praise the mother's expressing efforts. Assure her that all her milk will be used for her baby. Always use positive reinforcement. Be aware of subtle inferences that could be misconstrued by the mother that the amount of milk pumped is not enough for the baby.
The frequency and duration of milk expression directly correlates to the amount of milk produced [245]. The mother should pump every two or three hours to mimic a baby's natural feeding pattern. A tentative goal of 750–1,000 mL per day by 10 days postpartum will help her maintain her milk supply throughout lactation. This is a special concern in the mothers of preterm infants who often have milk supply difficulties [247]. Pumping should take full advantage of the increased vascularity in the breasts and the high prolactin level following birth. Pumping at least once during the nighttime is vital in the first few weeks to maintain milk supply. Good results may be obtained with a double electric pump system that allows for simultaneous pumping of both breasts. Continued use of a commercial electric pump appears to increase compliance with pumping and produces greater total volumes of milk [248]. Combining manual expression with electric pumping may increase milk production in the mothers of preterm infants [249].
Protocols for pumping and saving milk vary by hospital. Some hospitals require that all equipment is sterile at every pumping. Most authorities agree that human milk should be stored in sterile glass or polypropylene containers. Some breast pump manufacturers also sell 60-mL bottles specifically designed to avoid loss of components or leaching of plasticizers. Alternatively, 50-mL plastic centrifuge tubes have also been suggested for storage [1]. Plastic nurser bags are not appropriate because there is loss of immune components and a higher risk for contamination and breakage. Milk from several pumping sessions should not be mixed. If the milk is to be used within 24 hours, then the mother can refrigerate the milk and bring it to the hospital in a cooler. If it is to be used later than 24 hours after pumping, the milk should be frozen and kept frozen during transport [250]. When using pumped milk for infant feedings, a system of confirming proper identification should be used to prevent errors [251]. Even the smallest amounts of colostrum should be saved for the baby. Nurses should try to feed breast milk to the baby in the order that it was pumped to assist in the development of the infant gut.
Non-nutritive sucking, skin-to-skin contact, and knowledge about the benefits of transitioning infants to breastfeeding have been shown to help infants transition from gavage feeding to breastfeeding. Non-nutritive sucking facilitates the development of sucking behavior and improves digestion of enteral feedings. It also has been shown to decrease the length of stay in preterm infants receiving this intervention. Skin-to-skin contact has been associated with improved milk production and infant growth as well as maternal confidence and increased duration of breastfeeding [241,243,252].
Some mothers may be unaware of the advantages of transitioning their infant to breastfeeding. Factors that may contribute to a mother's inability or reluctance to transition her infant to breastfeeding include [241]:
Worry over an inadequate supply of breast milk
Lack of confidence or lack of commitment/desire to breastfeed prior to the birth
Concern about the infant's immature feeding behaviors
Belief that bottle feeding is more convenient and that pumping/storing breast milk is difficult
Lack of emotional support or embarrassment about breastfeeding
Concern that the father cannot participate in the feedings
By understanding these factors and providing mothers with knowledge and assistance, the healthcare team can better support mothers during the transition [222,252,253,254,255]. Support also includes realizing that mothers of preterm infants have multiple stressors (e.g., feelings of guilt, detachment, helplessness) [245].
Breastfeeding a premature infant can be both challenging and exhausting for the mother. An improved teaching protocol and breastfeeding ethos in the NICU and special care nursery may help support families of preterm infants [256]. Results of a national cohort study indicate that admitting mothers to the NICU with the infant and minimizing the use of a pacifier during breastfeeding transition were associated with 1.6 and 1.2 days, respectively, earlier establishment of exclusive breastfeeding [257].
The delivery of a preterm baby constitutes a nutrition emergency. Enteral nutrition is the preferred mode of support, and human milk is a key component of any strategy for enteral nutrition of all infants. Human milk is highly digestible and contains many anti-infective components, which confer a lower risk of infection [258]. However, the use of human milk as the sole source of nutrients for preterm infants is controversial because the nutrients of human milk alone are not sufficient to meet the needs of the premature infant. Safe, effective means to fortify human milk are essential to the care of premature infants [243,258,259,260]. Although this process remains controversial and requires further research, the AAP and the Canadian Pediatric Society have recommended the use of fortifiers containing protein, minerals, and vitamins to ensure that infants fed preterm human milk receive their estimated nutritional needs [243].
Commercial human milk fortifiers for preterm infants are available that contain different sources of macronutrients and a greater density of all nutrients than the formulas intended for term newborns. Preterm infants benefit from early enteral feedings. Slow, steady increases in feedings will help achieve full support [2,258].
The use of human milk fortifiers for the premature infant is considered a necessity to helping the infant reach appropriate nutritional levels. Be aware that mother may view human milk fortifiers as an indication that her milk is weak. The use of such fortifiers should be explained to the mother in detail [258].
A great deal of fuss has been made about bottles and pacifiers and ways to avoid them in term, healthy newborns. However, avoiding artificial nipples for the preterm infant often is inescapable and may not be as damaging as once believed. Because a major difficulty for mothers of preterm babies is the transition to feeding at the breast, nursery staff should try to help with this transition when the infant is stable and ready to begin oral feedings [241,261]. The transition from tube feeding to oral feeding can be accomplished in several ways [2,261,262]:
If the mother is consistently available for feeding, the baby may nurse at the breast as soon as he or she is stable; a nasogastric tube may be used when mother is not available.
Cup feeding: Although this method has become popular in many U.S. hospitals, it is almost exclusively used in underdeveloped nations. The process involves using a small flexible feeding cup, which is placed against the infant's lower lip and then tipped up just enough so that the milk touches the baby's mouth. The baby will then either "lap" up the milk or take small sips. Cup feeding requires considerable effort to keep the infant alert and considerable skill and concentration on the part of the feeder to maintain the feeding. Additionally, the volume of the mother's milk taken in by the infant is generally small, and a significant amount may end up on the infant. A review of cup feeding for preterm breastfed infants found that it resulted in more stable heart rate and oxygen saturation than bottle feeding, with similar weight gain, and that breastfeeding rates were higher at discharge with continued higher rates at 3 and 6 months post-discharge for cup-fed infants [262].
Finger feeding: This is popular among lactation consultants. With this technique, a soft feeding tube, or the tubes from a feeding tube device, rest on the feeder's gloved finger. The finger and the tubes are presented to the infant's lips and then gently inserted into the baby's mouth for feeding. This technique should only be done by trained individuals.
Bottle feeding: When bottle feeding the premature infant, nurses in the infant nursery should try to mimic an actual breast feeding. This may be accomplished by:
Tickling the baby's lower lip
Paced bottle feeding for 1–5 cc
Pausing the flow for a few seconds
Paced bottle feeding for another 1–5 cc
Pausing the flow again for a few seconds
Repeating these steps until the feeding has been completed
Imprinting with a bottle in this fashion mimics the multiple let-downs that naturally occur during breastfeeding. It also may help prevent flow preference, give the infant needed rest breaks, and reduce gastric distention from rapid feeding.
Remember that the baby's mouth is very sensitive. Multiple or forceful intrusions into the baby's mouth, often a part of survival for the preterm, may cause the infant to become "orally defensive." Gentleness and the utmost care are the rule.
Premature infants may be difficult to place at the breast in the cradle hold. A modified football (clutch) hold may help the mother latch the baby on, especially if the infant is attached to an IV line and monitor cables. The latch-on process remains the same, with the exception that the mother may wish to place her hand in the C-hold position with her fingers on the sides of the breast (i.e., the "dancer hold"). The premature infant may need jaw support. In the full-term infant, fat pads in the cheeks provide support for the lower jaw. The preterm infant normally has very little subcutaneous fat so the "suck pads" may not be fully developed. With the dancer hold, the baby's chin is supported by the fleshy portion of the mother's hand between the thumb and index finger. The baby's chin rests in the natural curve between thumb and index finger while the heel of the hand supports the mother's breast.
Try to stimulate the mother's let-down before latch-on. The mother who has been pumping long term must relearn to let-down to the baby instead of the pump when breastfeeding is initiated. Stimulation of the breast by the baby is different from that of the pump.
Studies have shown that breastfeeding produces less stress on the premature infant than bottle feeding. There is a decline in mean partial pressure of carbon dioxide and body temperature with bottle feeding. Body temperature and oxygen levels rise when the infant is breastfeeding [263].
Much controversy exists over test weighing before and after feedings as a measurement of breast milk intake (1 mL of breast milk weighs 1 gram). Electronic scales may give an accurate estimate of the infant's intake at the breast. The baby can then be complemented with additional expressed milk as needed [2].
The first feedings can be practice feedings rather than true feedings for nourishment. Non-nutritive breastfeeding sessions will decrease as the infant matures. During these initial sessions, the infant may not take a measurable amount of milk. Reassure the mother that this will improve as the baby matures. Recommend that the mother pump her breasts after each feeding until the infant suckle is stronger. The use of a feeding tube device may be needed to help strengthen the suckle. This plan is best managed by a trained lactation consultant.
The infant should not be discharged "breastfeeding" unless he or she is feeding very well and there is adequate follow-up [21]. If possible, contact a trained lactation consultant and arrange an appointment in the lactation clinic after hospital discharge.
Devices are available that can assist the nursing couple with breastfeeding difficulties. Breastfeeding devices may be purchased at many outlets throughout the United States. Parents should be warned of the dangers associated with the improper use of breastfeeding devices. A nursing couplet should not use a nipple shield or a feeding tube device without the appropriate referral to a lactation consultant for continued follow-up.
Breast shells are hard shells worn over the nipple between feedings and should not be confused with nipple shields. They can assist with inverted nipples, provide air circulation for healing sore nipples, and alleviate engorgement. Some manufacturers make the entire shell of rigid plastic; other shells have a soft silicone face that rests against the breast. Hobbit-type shells have large air holes for ventilation. Shells that provide ventilation are highly recommended. Breast pads that do not occlude air flow should be worn outside the shell, if needed.
Breast shells can be associated with problems. For example, they can cause pressure areas on the areola that result in plugged ducts. In clinical practice, cases have been seen in which mastitis either does not resolve after antibiotic treatment or recurs in a mother who is wearing breast shells. Solid shells may retain moisture against the breast, which can result in nipple soreness and/or thrush. The milk collected between feedings in the shells should not be kept to feed to the baby due to high bacterial contamination [2].
Breast pumps are medical devices regulated by the FDA. They are used to express breast milk. They also may be used to maintain or increase a mother's milk supply, feed multiple babies, relieve engorged breasts and plugged milk ducts, and pull out flat or inverted nipples so that a nursing baby may latch on to its mother's breast more easily [264].
All breast pumps consist of a few basic parts [264]:
A cone-shaped cup, called a breast shield or flange, that fits over the nipple and the areola
A pump to create the gentle vacuum that expresses milk. The pump may be attached to the breast shield/flange and have plastic tubing to connect the pump to the breast shield.
A detachable milk collection container that fits below the breast shield. The container is typically a disposable bag or a reusable bottle that can be used to store the milk or attached to a rubber nipple and used for feeding the baby.
The three main types of breast pumps include manual, battery-powered, and electric. They extract milk from the breasts by creating a vacuum around the nipple that pulls air into the breast shield and applies and releases suction. Each suction and release combination is called a "cycle" [264].
Many manual breast pumps use a system of two cylinders to create suction. After the breast shield is placed over the nipple and areola, a small cylinder-shaped tube is pumped in and out of a larger cylinder to create a vacuum that expresses milk and collects it in an attached container. Some manual pumps have a handle or lever that is squeezed to create suction and express milk from the breast. The breast milk is then collected in an attached container. Another type of manual pump is referred to as a bicycle horn pump and consists of a hollow rubber ball attached to a breast shield. One of the earliest breast pump designs, the bicycle horn pump got its nickname from the old-fashioned bicycle horns it resembles. To operate a bicycle horn pump, the breast shield is placed over the nipple and the rubber ball is squeezed and released to create a vacuum that expresses milk. Some experts discourage use of the bicycle horn pump because they may be difficult to clean and dry [264].
A powered breast pump uses an electricity source (batteries or an electrical outlet) to power a small motor that creates suction to extract milk from the breasts. One or more long plastic tubes connect the breast shield to the motor. The motor has a control panel with a dial or switch to control suction. Certain types of powered breast pumps may be adjusted to create different patterns of suction. Some manufacturers claim the adjustable suction allows the user to find a setting that closely mimics her nursing baby. It is important that women who use a powered breast pump are prepared for emergency situations when electricity or extra batteries may not be available. Having extra supplies for pumping and a back-up method, such as a manual breast pump, may help a woman maintain her breast pumping schedule during an emergency [264].
Success with pumps is highly individual; some brands or models work better for some mothers than others. The suction produced by the pump should be intermittent, not constant. It is the rhythm of the suction and compression of the areola inside the flange that empties the milk in combination with the suction. Suction alone will not empty the milk [2].
Numerous studies have shown that breast/nipple creams do not assist in the prevention or healing of sore nipples. Additionally, many ingredients in breast/nipple cream preparations may be hazardous to the newborn [265]. Breast and nipple creams generally are not recommended for the nursing mother by lactation consultants [2]. Table 12 is a brief list of some commonly used agents and comments on each.
NIPPLE CREAMS
| Cream | Necessary Information |
|---|---|
| A&D Ointment | There are no vitamins in A&D Ointment. Label reads: "For external use only, in case of ingestion contact poison control center." |
| Bag Balm | From the dairy industry. Contains fungistat and bactericide. Causes cancer in laboratory animals. |
| Eucerin | For external use only. |
| Mammol | Warning: Subnitrate may be changed in the infant's intestine to nitrate, which causes methemoglobinemia after absorption. |
| Masse | Contraindicated in mastitis and breast abscess. Glycerin is rapidly metabolized by the baby and can cause hyperglycemia. Cetyl alcohol is a laxative. Peanut oil aspiration can cause severe and fatal bronchitis in small children. |
| Vitamin E | Effect of increased serum concentrations on vitamin E is unknown. |
| Lanolin | Estimated to contain less than 1.5 ppm of combined impurities. No caution required for use with open wounds or where ingestion may occur. |
A nipple shield is an artificial nipple that is worn over the nipple and areola during feeding; the baby nurses through the shield. Nipple shields should not be confused with breast shells. Most nipple shields in use are made of silicone, although in the past rubber and latex versions were available. A study involving 34 preterm infants found that using ultra-thin silicone nipple shields significantly increased milk intake during breastfeeding sessions. The average milk transfer rose from 3.9 mL without the shield to 18.4 mL with it. Importantly, the use of nipple shields did not reduce the overall duration of breastfeeding, which averaged 169 days [295]. Studies of the rubber and latex designs have revealed reduced milk transfers of 58% and 22%, respectively [266,267]. Nipple shields have also been associated with infant malnutrition and with cases of failure to thrive [2,268,269]. If necessary, only the thin silicone shields should be used, and the need for the device should be revisited frequently.
The available evidence does not demonstrate that long-term use of nipple shields are safe for milk supply, infant weight gain, or duration of breastfeeding [267]. Nipple shields should be viewed as a "prescription" device requiring written documentation of need by clinical impression, a written treatment plan, and close follow-up. Lactation consultants maintain that the nipple shield is a device with a strong potential for misuse. Informed consent should be discussed by the mother, father, and the provider recommending the nipple shield.
Both the Supplemental Nutrition System (SNS) and the Lact-Aid Nursing Trainer System are feeding tube devices whereby the infant can nurse directly at the breast and receive supplemental breast milk and/or ABM (Figure 3). The presence of an increased flow organizes the infant suckle, making nursing more efficient. Feeding tube devices may be recommended and used in many situations (e.g., failure to thrive, twins, low milk supply, induced lactation, breast surgery) in which other measures have failed in order to prevent complications. The goal of feeding tube devices is to stabilize the infant and maternal milk supply as quickly as possible and wean the baby from the tubes. Because these devices are used only in special situations, close follow-up is imperative to ensure adequate milk intake by the infant and validate correct use by the mother [2]. Use of the tube system in a normal, healthy breastfeeding infant solely for ABM and/or water supplementation is considered inappropriate.
Many mothers have unrealistic expectations of the baby's nursing behavior in the first few days. She may expect that the baby will latch-on and nurse vigorously from the very start. If the first few feedings do not go as she anticipated, mother may become very discouraged. Remind her that she and the baby are still learning. If mother is home and feels overwhelmed or discouraged, she should be advised to seek assistance from a lactation consultant, support group (e.g., La Leche League), or supportive family member. She also should be advised to get plenty of rest, nap when the baby does, and avoid trying to do more than heal her body from the birth and support her milk supply in the early days postpartum.
The rapid changes in hormone levels after delivery can be expected to cause some minor problems for new mothers. Some mothers may lack enough essential fatty acids for hormone production in the postpartum period [270]. In addition, sleep deprivation can cause behavioral changes. An estimated one in five (20%) women and one in 10 (10%) men will experience a perinatal mood and anxiety disorder (PMAD) during the perinatal period [296,297]. Untreated maternal depression may negatively impact the mother and the mother's other children and, when it occurs during pregnancy, may lead to pre-eclampsia, preterm delivery, and low birth weight [271].
Unfortunately, many women fail to recognize the signs and symptoms of PMADs, and when they do, they fail to seek treatment [272]. Women with a history of prior depression, panic disorder, obsessive compulsive disorder, marital problems, premature delivery, premenstrual syndrome, or prior PMADs are at highest risk. Additional risk factors include lack of social support, lower income and education, single status, life stress, and unintended pregnancy [271]. The American Academy of Family Physicians has listed the following as warning signs of postpartum depression [273]:
Persistent feelings of sadness and crying
Having little desire to eat; significant weight gain or loss
Irritability, anxiety, restlessness, insomnia
Inability to find pleasure/interest in life (anhedonia); having little interest in the newborn
Feeling exhausted, lack of motivation
Suicidal ideation or thoughts of harming the baby (requires emergency attention)
The American College of Obstetricians and Gynecologists recommends that women be screened for depression during and after pregnancy and that the screening be coupled with appropriate follow-up and treatment when indicated [271].
As previously mentioned, the AAP has recommended exclusive breastfeeding for the first 6 months with a gradual introduction of iron-enriched solid foods around 6 months of age as a complement to breastfeeding [20,21]. The unique needs or feeding behaviors of some infants may indicate an earlier (i.e., around 4 months of age) introduction of complementary foods, whereas other infants may not be ready until around 8 months of age. Infants weaned before 12 months of age should not receive cows' milk [21]. Despite recommendations, some mothers introduce solid foods earlier or later than recommended. Education and information that target these mothers may help increase the number of infants being fed as recommended [274].
Mothers may agonize about when to start offering solid foods because they receive conflicting advice. Intense pressure to start solids often comes from well-meaning family members. Mothers also may believe that the introduction of solid foods is baby led and initiated by some physical characteristic or behavioral action of the infant. For example, studies have found that the main reason some mothers initiate solid feeding is a perception that weaning is "necessary" to satisfy the infant [275].
Certain developmental milestones can help her determine if her baby is ready for solids [2]:
Baby has lost the extrusion or tongue-thrust reflex (a protective action of the tongue that pushes solids out to prevent choking)
Baby can sit unassisted
Baby demonstrates purposeful movement of hands and fingers
Total weaning is the complete cessation of all breastfeeding. Prior to 1979, the average age of the baby at weaning worldwide was 4.2 years [1]. The average breastfeeding duration in the United States is well below one year; consequently, many physicians consider six months to be "extended breastfeeding" [13]. However, there is no deadline for weaning. The benefits of breastfeeding continue for both mother and baby for as long as they nurse. If possible, weaning should be done gradually so it is not stressful for mother or baby. Successful breastfeeding means attaining the mother's personal breastfeeding goal. Whether she chooses to breastfeed for six weeks, six months, one year, two years, or more, the decision is entirely hers and the baby's to make.
There are few drugs that require that mother stop breastfeeding. Questions that should be considered prior to prescribing drugs to a breastfeeding mother include [276]:
Is drug therapy necessary?
Which drug would be safest to use?
Will the drug present a risk to the infant?
What measures may be used to minimize drug exposure to the nursing infant?
The National Library of Medicine, a division of the National Institutes of Health, provides a searchable database of drugs (LactMed) to which a breastfeeding mother may be exposed. The database provides such information as maternal and infant levels of drugs, possible effects on breastfed infants and on lactation, and alternate drugs to consider (Resources). An AAP policy clinical report on the transfer of drugs and therapeutics into human milk focuses on the use of psychotropic medications, narcotics, galactagogues, and herbal products [276]. The AAP also recommends referring to LactMed for more detailed, up-to-date information.
Physicians who encounter adverse effects in infants who have received drug-contaminated human milk have been urged to document the effects and contact the FDA. Additionally, herbal remedies have become increasingly popular. Table 13 contains a list of commonly used herbs and their effect on the infant through mother's milk.
HERBS AND BREASTFEEDING COMPATABILITY
| Herb | Uses | Notes/Cautions | ||
|---|---|---|---|---|
| Blessed thistle leaf (tea, capsules) | Reportedly increases milk supply | Galactagogue, diaphoretic, strong emetic (at toxic levels) | ||
| Chamomile (tea, capsules) |
| In large amounts can cause contact dermatitis, vomiting, and vertigo | ||
| Comfrey (tea) | CONTRAINDICATED | Veno-occlusive disease, hepatic failure, possibly liver cancer | ||
| Echinacea (capsules, serum) | Reported immunostimulant | Repeated daily doses may depress immune system. No data available on transfer into preparations. | ||
| Ephedra (ma-haung) |
| Hypertension, tachycardia, toxic psychosis, and death may occur. | ||
| Fenugreek (tea, seeds, capsules) | Documented to increase milk production | Use caution if mother is diabetic; lowers blood glucose. Same family as peanuts and chick peas, use caution in allergic women. Some mothers may have severe diarrhea. Not recommended for pregnant women. | ||
| Ginkgo biloba (capsules) | Reportedly improves memory | Seeds are toxic and should not be consumed. No data available on transfer of ginkgo biloba extract into human milk. | ||
| Ginseng (tea, capsules) |
| Has estrogenic effects, causes breast pain, vaginal bleeding reported. One case of neonatal hirsutism reported. No data available on transfer into human milk. Caution is urged. | ||
| Goat's rue (tea) | Reportedly increases milk supply | No human trials have been completed. May cause drowsiness, poor suckling in infants. | ||
| Peppermint (tea, capsules) | Reported use for stomach upset—dries milk production | Candy and teas contain little or no peppermint extract. | ||
| Sage (tea, grated, capsules) | Reportedly dries milk production | Due to drying properties and pediatric hypersensitivity, use with caution in nursing mothers. | ||
| Shatavari (Asparagus racemosus) | Used in Ayurvedic traditions to improve female vitality | May result in delayed gastric emptying | ||
| St. John's wort (capsules, tea) | CONTRAINDICATED | May suppress prolactin release. Due to long half-life, should not be used in nursing mothers. Alternate compatible prescription antidepressants are recommended. | ||
| Tea tree oil (topical wound care) | Occasionally used for sore nipples | Toxic if ingested. As little as 10 mL orally can affect CNS. Caution is urged. |
New mothers frequently leave the hospital in as little as 24 to 48 hours after the birth. Preventable difficulties occur more frequently within the first week postpartum than at any other time during lactation. Half of the mothers who initiate breastfeeding prematurely wean within a few days or weeks due to preventable or treatable difficulties. Various states in the United States have passed laws requiring follow-up within 48 hours of discharge. Yet, the average mother-infant pair is not seen by a healthcare professional for two to six weeks after discharge [281,282].
Because of deep emotional connections, breastfeeding is an emotionally charged issue for healthcare providers. It has been noted that "physicians and nurses are notoriously eager to provide advice, even if their knowledge of, or experience with, the subject is limited" [5]. Most nurses have both positive and negative experiences with breastfeeding women or as mothers themselves. Nurses' own sets of cultural values and emotions dramatically affect patients' breastfeeding experiences. Research has indicated that physicians' attitudes about breastfeeding are influenced by how their own children were fed [1]. Healthcare providers should try not to let their personal feelings interfere in the way they approach the nursing couple.
La Leche League is a support group for breastfeeding women and their families that was founded in 1956 in response to the need for supportive breastfeeding information. It began as a small group and has since grown into an international organization with local chapters in many U.S. cities. Contact information for the League is available in the Resources section of this course.
A lactation consultant is a formally trained breastfeeding expert. An International Board Certified Lactation Consultant (IBCLC) is the only internationally certified healthcare professional who specializes in the clinical management of breastfeeding and human lactation. They are certified by the International Board of Lactation Consultant Examiners, under the direction of the U.S. National Commission for Certifying Agencies. IBCLCs work worldwide in a variety of healthcare settings [283].
Consultants come from a variety of backgrounds, and include nurses, educators, and individuals who have counseled breastfeeding women for many years. Hospitals often have a lactation consultant associated with their maternity unit. Training and approach to difficulties may vary slightly among consultants. The one thing that all lactation consultants have in common is the sincere desire to help women toward a successful breastfeeding experience.
Patient A and her son K, 6 days of age, were seen in the lactation clinic for sore nipples.
Baby K was initially ABM-fed in the nursery. First nursing was at 26 hours of age. Nipple pain began with the first breastfeeding. Patient A experienced moderate engorgement on postpartum day 4. Following engorgement, her nipples began to crack. Nipples now "seem to hurt all the time." Patient A reports that severe nipple pain occurs with latch-on. Nipple cracks began bleeding bilaterally yesterday. The infant vomited blood after this morning's feeding.
Patient A is 28 years of age, gravida 2 para 2. Pregnancy was uneventful except for a herpes outbreak at 36 weeks. Patient A reports breast changes accompanied pregnancy. The infant was born by elective cesarean section at 39 and 3/7 weeks for active genital herpes. The cesarean was done under epidural anesthesia. K's birth weight was 8 pounds, 7.5 ounces. Hospital discharge weight at 3 days of age was 7 pounds, 15 ounces. Bilirubin level was 10 mg/dL. Patient A has a daughter, 3 years of age, at home whom she attempted to nurse for three weeks but quit because of severely sore nipples. Family history is noncontributory except for a tendency toward allergies. Father is allergic to milk, wheat, and pollen. Patient A is allergic to codeine, penicillin, and sulfa. Three-year-old sibling has asthma. Patient A desires to nurse this child as long as possible to help prevent asthma and other allergies. Father was present during the consultation and appears very supportive. The couple did not attend breastfeeding classes.
During the last 24 hours, K has nursed every 2.5 hours; mother is switching breasts every 10 minutes. Feedings take 20 to 40 minutes. Patient A reports eight damp diapers and two semi-runny, yellow stools since this time yesterday.
Infant appears slightly jaundiced. Sclerae are white. Weight today is 8 pounds. Hydration status: good tissue turgor, level fontanelles, and mucous membranes slightly dry. Respirations easy. Has a lusty cry. Oral anatomy is within normal parameters. Soft and hard palate intact. Lingual frenulum visible, but tongue moves freely.
Maternal breast examination revealed moderately engorged breasts. Nipples are everted, approximately 1 cm in size with a linear fissure diagonally from 2 to 8 o'clock on the left side and 10 to 4 o'clock on the right. Areolae, approximately 3.5 cm, appear moist and intact.
Patient A positioned K on his back in cradle hold. Breast support technique was C-hold; however, mother pinched the nipple between the thumb and forefinger and placed the nipple into the baby's mouth before any evidence of a rooting response. K latched on to nipple only. The latch was immediately broken by the consultant.
Patient A was shown how to position the baby, proper breast support, and latch-on technique. K latched on with minimal assistance, wide-open mouth well back on the areola. Active suckling with a one-to-one suck-swallow ratio was observed. Milk ejection response was noted as dripping from the opposite breast. Patient A stated after slight initial nipple pain, the remainder of the feeding was pain free. K nursed actively before suckling slowed and he began to twist and squirm. Patient A was told that K's slower suckling rate and movements might be his signals that he needed to burp. Patient A inserted her finger into K's mouth to take him off the breast. The nipple was round and of normal color immediately after nursing. Following burping, mother latched K on to the opposite breast without assistance. He nursed for a shorter time before falling asleep and releasing the breast on his own. Patient A was instructed to start the next feeding at that breast.
Sore nipples due to knowledge deficit of latch-on technique.
Engorgement related to normal Stage II lactogenesis and inadequate feedings (breast emptying).
Poor weight gain due to inadequate intake related to nipple pinching.
Possibility for mastitis due to nipple fissures.
Remember the steps of latch-on.
Positioning the baby: Turn the baby entirely toward you. Place the baby's head in the crook of your arm. Support the baby and your arm with pillows.
Offering the breast: Thumb on top; all fingers below the areola. Keep fingers away from the areola. Bring the baby to the breast—not the breast to the baby.
Latching-on: Tickle the baby's lower lip with your nipple. Wait for the wide "ah" before latching on. Latch the baby on correctly at each feeding. If there is nipple pain, take a deep breath, let it out slowly; if the pain continues after the cleansing breath, break the latch and begin again.
Additional recommendations for this patient include:
Nurse on cue. Allow the baby to signal the timing of the feeding.
If the baby nurses minimally on the second breast, begin the next feeding on that breast.
Keep a written record of feedings and urine and stool output. Bring it to the next appointment.
Apply cool, moist compresses to breasts after nursing to help with engorgement.
Manual expression before latch-on if areola is too firm for proper latch-on.
Watch for signs and symptoms of mastitis.
Return to clinic in two days for weight check, or sooner if condition worsens or does not improve.
At his second visit, K weighed 8 pounds, 7 ounces, a gain of 7 ounces in 48 hours. Maternal nipples were healing. Breasts were soft. A nursing session was observed. K and his mother were not seen in the lactation clinic again. However, Patient A called the clinic for weaning information when K was 2 years of age. She stated they had no further breastfeeding problems.
Patient M and her daughter J, 16 days of age, were seen in the lactation clinic for low milk supply.
Due to the severity of her postpartum condition, Patient M was unable to breastfeed her infant. The mother was followed by the lactation team during her complicated hospital course.
She was started on a two-hour pumping schedule seven days postpartum using a commercial-grade electric pump with a double pumping set up. Milk yield was 0.5 to 1 ounce per session, and the milk remained colostral throughout her hospital stay. She never experienced any postpartum breast fullness. All expressed milk was fed to the infant in addition to ABM. M wants to be able to breastfeed this infant "forever, because nothing else went right."
She was discharged home on postpartum day 14 with instructions to continue pumping, provide lots of skin-to-skin contact, and patiently wait to see if J would latch-on by herself.
Patient M is 42 years of age, primigravida, with no history of fertility problems. M reports breast changes accompanied pregnancy, growing two full cup sizes. The pregnancy was difficult. During the first trimester she was hospitalized three times for dehydration resulting from hyperemesis gravidarum. M had an episode of pyelonephritis during the second trimester. Elevated blood pressure and proteinuria began at 31 weeks' gestation. The infant was born by cesarean section at 37 weeks due to pregnancy-induced hypertension and HELLP syndrome. Due to the low platelet count, a postpartum hemorrhage resulted. Her hemoglobin was 6.5 on postpartum day 3 and 8.2 at hospital discharge. M received transfusions of 2 units of packed cells and 4 units of platelets. She was diagnosed with a collapsed lung on postpartum day 5 and required chest tubes for decompression.
J's birth weight was 6 pounds, 12 ounces. Hospital discharge weight was 8 pounds, 2 ounces. J's hospital course was uneventful. The father remained in the hospital during the postpartum course and gave J most of her feedings.
Family history is noncontributory except that M and her husband are newlyweds (one year). This is the first marriage for both. The father was present during the consultation and appears very supportive. They were unable to attend any prenatal classes.
During the last 48 hours, J has been fed every three hours. She takes an average of 3 ounces of expressed mother's milk and ABM at each feeding. The baby latched-on spontaneously for three to four minutes during skin-to-skin contact twice in the last 24 hours.
Infant appears healthy and well nourished. Weight today is 8 pounds, 6 ounces. She is alert and active. Oral anatomy is within normal parameters. Soft and hard palate intact. Tongue has normal peristaltic motion on digital assessment.
Maternal breast examination reveals soft breasts. Nipples are everted and intact. Large areola appears dry. Milk will express in drops. Appears to be of transitional quality rather than colostral. Patient M reports that her milk yield with double pumping remained the same.
Mother positioned the baby in a clutch hold with pillows for support. J latched-on and suckled vigorously. However, there were infrequent swallows.
Patient M was shown how to fill and set up the SNS. The SNS was filled with 3 ounces of expressed milk. The medium tube was placed at the breast, and the baby latched-on and suckled with a one-to-one suck-swallow ratio. No milk ejection response was observed. The feeding was pain free. J nursed, taking 2.5 ounces from the SNS.
A dietician was consulted for diet suggestions to include high-protein, high-iron, and high-vitamin C foods for healing and anemia. A written list of appropriate foods was given to the mother.
Actual low milk supply probably related to poor maternal physical condition.
Potential for Sheehan syndrome due to postpartum hemorrhage.
Use the SNS with the medium tube at each nursing. Fill to 3-ounce level (adding more if needed). The bottom of the bottle should be level with the nipple line. Tape tubing in place as needed. Handout given for the care and use of the SNS.
Keep a written record of feedings, intake from SNS, and urine and stool output. Bring it to the next appointment.
Follow each feeding with a pumping session as per parent's goals.
Use breast massage during feedings and pumpings.
Rest as much as possible. (Mother was given permission to skip feedings as needed if she felt she was not getting enough rest.)
Add foods recommended by dietician to meals.
Continue vitamin supplements.
Return to clinic in two days.
At the second visit, J weighed 8 pounds, 7 ounces, a gain of 1 ounce in 48 hours. M had nursed the baby at the breast with the SNS every three hours. J averaged 2 ounces per feeding. Patient M stated her breasts felt fuller and firmer, which was confirmed by breast examination. A nursing session was observed. Patient M demonstrated proper use of the SNS and latch-on technique.
Continue the SNS at each nursing. Fill to 2-ounce level (adding more if needed). Lower the bottom of the bottle to one inch below the nipple line.
Keep a written record of feedings, intake from SNS, and urine and stool output. Bring it to the next appointment.
Continue previous plan of care.
Return to clinic in three days.
At the third visit, J weighed 8 pounds, 10 ounces, a gain of 3 ounces in 72 hours. M had nursed the baby at the breast with the SNS every three hours. J averaged 1 to 1.5 ounces per feeding. Urine and stool output were within normal limits. Mother stated her breasts were engorged this morning, which was confirmed by breast examination. A nursing session was observed with the SNS in place. J gulped at the breast after about one minute. Milk ejection response was observed as large drips from the opposite breast. The SNS tubing was clamped and J continued to nurse vigorously with a one-to-one suck-swallow ratio.
Low milk supply resolved.
Stage II lactogenesis evident as engorgement.
Sheehan's syndrome ruled out due to presence of milk ejection response and engorgement.
Start each feeding with the SNS; clamp tubing after one minute.
Keep a written record of feedings, intake from SNS, and urine and stool output. Bring it to the next appointment.
Continue previous plan of care.
Return to clinic in three days.
At the fourth visit, J weighed 8 pounds, 15 ounces, a gain of 5 ounces in 72 hours. M had breastfed the baby at the breast with the SNS for the first 24 hours only. Urine and stool output were within normal limits. A feeding session was observed without the SNS. J nursed vigorously with a one-to-one suck-swallow ratio.
Discontinue the SNS.
Nurse every three hours or on demand.
Keep a written record of feedings and urine and stool output. Bring it to the next appointment.
Return to clinic in three days.
M and J were followed for three weeks with weight checks every three days. J continued to gain well. Mother and infant were discharged from lactation clinic follow-up at 45 days postpartum. J's weight at the last visit was 12 pounds, 10 ounces. The couple was still nursing well at six months.
Patient B and her infant C, 7 days of age, were seen in the lactation clinic because her baby had not latched on since birth.
Patient B is 44 years of age, primigravida, with a history of fertility problems. In her words, this will most likely be her only biologic child. C was delivered by cesarean section at 32 weeks' gestation because of high blood pressure and HELLP syndrome. Cesarean section was done under epidural anesthesia. Baby C's birth weight was 4 pounds, 8 ounces. She was discharged from the hospital with her mother at 15 days of age. C is bottle fed with expressed mother's milk, taking approximately 90 cc every three hours. Patient B has been trying to maintain her milk supply using a small electric pump. In the last few days, B reports that she is pumping smaller and smaller amounts of milk at each session. Currently pumping 10 to 15 cc every two hours.
C is a healthy newborn with good color. Slightly hypertonic muscle toned is noted, probably related to crying state. Last feeding was 3.5 hours ago. Maternal breasts appear normal. Nipples are intact, and the breasts feel soft, not filling.
Unable to latch on for more than a few seconds without baby becoming visibly upset. Infant was bottle fed at this appointment.
Severe latch-on problem related to hunger state. Actual low maternal milk supply related to inefficient breast pump.
Increase milk supply. Decrease maternal stress level regarding latch-on.
Hospital-grade electric double pump every two hours.
Herbal supplements: fenugreek, blessed thistle, and brewer's yeast as instructed.
Do not attempt latch-on but do a lot of holding and skin-to-skin contact without asking baby to latch on.
Return to clinic in two days. Bring baby hungry but not starving.
Patient B has been pumping every two hours and noticed increase in milk supply yesterday. Enough milk pumped so she could have some extra expressed milk in the freezer. Baby C is calm with adequate two-day weight gain.
Physical exam is remarkable for hypertonia. When held in mother's arms, C's back arches. C has a stiff quality and is easily upset. C would not root to the breast; she arched, pushed the breast away with her fists, and screamed after only a few minutes of trying. She did not cuddle; C turned her whole body away from her mom when held. C's body looks like an arch, with only her head and bottom touching her mom across her mom's arm. When laid down she immediately flips to her right side and holds herself in a tight fetal position. Is extremely difficult to bottle feed, taking nearly an hour to finish a feeding of 45–60 cc.
Severe latch-on problem related to hypertonic muscle tone.
Low maternal milk supply resolving.
Continue present plan. Offer breast but do not insist. Stop if baby gets upset. Referral to neurodevelopmental therapist made. To follow-up in one week after appointment.
Patient B and C returned once a week for 16 weeks. C was gaining well and increasing her intake with mom now over-supplied.
At one visit, B confessed that she often worried that C hated her. She was assured that C's behavior was not directed at her, but the result of an immature nervous system. She was also warned that C might never latch on but help and support would be available for whatever she wanted to do. B said she had 12 weeks of maternity leave left, and she would continue to pump her milk for at least that long. "My husband and I have been talking about how my body couldn't allow her to finish growing inside so now she has to grow outside, and the best way is with my milk. I will pump for as long as I can if that is what we have to do."
B was doing a lot of skin-to-skin contact, co-bathing, and sleeping with C and attempting latch-on if C showed any interest. On the day C would have been 6 weeks of age if she was born on her due date, C began rooting toward the breast. So, mom leaned over, and C latched on and nursed for more than an hour. B stated she nursed like she had been doing it all along. She was still nursing at six months. Mother plans to nurse her until she self-weans.
Patient V was seen in urgent care five days ago. The diagnosis was mastitis of the right breast. She received a prescription for dicloxacillin 500 mg four times a day for 10 days. The patient was told at urgent care not to breastfeed on the affected side. She has been taking dicloxacillin "kind of hit and miss, but at least four per day" and is pumping "when the breast feels full." She is currently nursing on left side only, every two to three hours. Patient V has a history of nipple fissures with early engorgement. She states that she has not had a fever in the last two days; temperature is 98.7°F oral now. She feels "well" and denies headache or body aches. The infant, 22 days of age, is not present at this appointment.
Right breast severely engorged. Healing, linear crack noted horizontally across the right nipple face. Bright-red, wedge-shaped area in upper inner quadrant of right breast covering the 12 to 3 o'clock position. Area is exquisitely tender. Oval induration approximately 3–4 cm in size palpated central to inflamed area. There is a 1 cm area within the indurated region, slightly below surface, that is soft and mushy. Skin intact over the area.
Probable breast abscess related to inadequate breast emptying, delay in antibiotic treatment for apparent ascending mastitis, and inadequate use of antibiotics (Table 14).
COMPARISON OF PLUGGED DUCTS, MASTITIS, AND BREAST ABSCESS
| Condition | Symptoms | Treatment | |||||
|---|---|---|---|---|---|---|---|
| Plugged ducts | Afebrile with no systemic symptoms. Breast tender in the area of the plug. Plug will feel like a small knot. The breast should not appear inflamed or reddened. |
| |||||
| Mastitis | Fever, chills, body aches, and headache. Red, inflamed area on breast or red streaks on breast. Flu-like symptoms: Diarrhea, nausea, vomiting. |
| |||||
| Breast abscess | History of mastitis. Area may no longer be painful. Systemic symptoms may be absent. The abscessed area has risen to the surface of the breast tissue and is indurated in the center. | Surgical incision and drainage. Hospitalization for IV antibiotics as needed. Continue to nurse or pump on a regular schedule. Cover incision with a clean dressing at each feeding. Breast milk is not harmful to the incision, as the macrophages in the milk will assist in the destruction of the offending bacteria and aid in the healing process. |
To urgent care immediately.
Pump breast with hospital-grade electric pump or nurse every two hours.
Follow up in two days.
Call if weaning information is desired.
What can I expect in the first few days? Some infants nurse avidly right from the start. Others may take time to recover from labor and should pick up within 24 hours. The first feedings are a "get acquainted" time for both mother and baby.
Typically, the newborn will take one or two tries to latch-on, take four or five suckles, swallow, and then pause and begin suckling again. This is normal. Encourage the mother to keep the baby awake by talking, rubbing, and gently caressing him or her at the breast when pauses seem too long.
Is there anything in my breasts for my baby? My breasts feel empty. Many think of the breast as a container that is either full or empty. The breast does not work like a bottle. Breastfeeding requires a mother to confidently trust that her breasts will do what they are meant to do, even though she cannot see the milk being consumed. She can, however, easily see how many wet diapers and stools the baby is having every day.
Colostrum, the first milk, is produced even before the birth. It is very thick and yellow and is like sucking maple syrup through a straw; so, feedings will seem a little long at first. Colostrum is a super booster of antibodies and contains everything that baby needs. The baby will usually take small amounts of colostrum in the hours after birth, gradually increasing intake so the digestive system starts up slowly.
Are sore nipples normal? Mild tenderness is normal in the early days of nursing. The mother may feel some discomfort in the first minute of a feeding as baby stretches the nipple into his or her mouth. This nipple stretching pain should improve in a few days. When the baby is nursing correctly, the mother should feel a gentle tug, not a pinching sensation. If the latch is incorrect, break the latch and try latching on again until it is not painful. Limiting the time at breast will not prevent sore nipples. Frequent and effective feedings can help prevent engorgement, which is a contributor to sore nipples. If the mother develops blisters, scabs, marks, bleeding, or pain between feedings, she should contact a lactation consultant for assistance.
When will my milk come in? Mature milk increases in volume within three to five days after the baby is born. If the baby is having a good number of wet diapers, several stools per 24-hour period, is gaining weight, and wakes themselves to feed at least 8 to 12 times per 24 hours, feeding is likely effective. If there are symptoms of low breast milk intake, the mother should contact a lactation consultant for assistance.
How will I know if I am engorged? A mother's breasts will start to feel heavier and firmer as her milk increases in volume. This is due to increased circulation in the breast. Mild engorgement is a sign that the mature milk is increasing. Moderate-to-severe engorgement, during which the breasts become tender, firm, or lumpy, is not normal. Painful engorgement occurs from tissue swelling, trapped milk, or a combination of both. The breasts may not be "full of milk" as many believe. The tissue around the milk-producing glands may be swollen and painful. Sore nipples may follow engorgement because the areola becomes too firm for the baby to latch on properly.
Why can't I feel my let-down? In the early days of breastfeeding, many mothers do not feel the let-down or milk ejection reflex. It may take a week or two before she can recognize the milk ejection reflex. Occasionally, some mothers never feel the let-down, but they are aware of it because milk drips from the opposite breast during a feeding, she feels warm or thirsty, and/or the baby has bursts of rapid swallowing.
How much rest will I need? Immediately following the birth of the baby, it is important for the mother to get enough rest, whatever the feeding method. Many new mothers neglect their need for rest. The mother's sleep is interrupted by night feedings, so she should be encouraged to nap or, at the very least, rest with her feet up when the baby naps. For a week or two, the mother should not use the baby's slumber time as a time for getting household chores done. In the early weeks after giving birth, the mother's body needs to heal from labor. If she has had a cesarean birth, additional time may be needed to feel rested and healed from the birth.
What is most important in my diet? The mother should drink enough liquids to quench her thirst. For good health, a minimum of eight, 8-ounce glasses of fluid per day are recommended. A good habit for the mother to develop is to have a glass of something to drink every time she feeds her baby.
Many women worry needlessly about their diets while breastfeeding. Often, women feel deprived because someone told them that they cannot eat certain foods while nursing. Some common myths are that eating chocolate makes chocolate breast milk or that cabbage, beans, and other gas-producing foods will make the baby have gas. Onions, garlic, and other aromatic spices will flavor the milk, but babies rarely object to the difference in taste.
On occasion, some foods may affect the baby. The mother should make note of what the particular food was, eliminate it for about a month, and then reintroduce a little of the food. Usually, the reaction was merely a sensitivity and not a true allergy. The mother should avoid overdoing any one type of food or beverage. Everything in moderation is excellent advice.
The mother should eat a well-balanced diet consisting of all food groups. She will need three meals and at least two small snacks every day. As long as she is nursing, the mother should continue to take her prenatal vitamins to replenish her own body stores. If a mother does not drink milk or use dairy products, a non-lactose calcium supplement should be added to her diet.
The recommended caloric intake for the breastfeeding mother is 2,200–2,500 calories per day or about 500 calories more than her pre-pregnancy diet [284]. (The average U.S. adult woman's diet consists of about 3,000 calories per day.) The majority of the weight she gained in pregnancy should be lost by six to eight weeks postpartum. Most mothers will not gain weight while breastfeeding unless they are severely underweight at the end of pregnancy. Breastfeeding mothers will often retain an extra 5 or 10 pounds until several weeks after weaning. This additional body weight protects the mother's ability to produce milk in case of illness or a severe reduction in caloric intake.
Weight-loss diets are not recommended while breastfeeding. The mother who is gaining weight while breastfeeding should closely examine her diet for "empty calories" and activity level. Many women falsely believe that they need to drink large amounts of cows' milk to make breast milk. Cows' milk is an excellent calcium source, but the calcium is also present in low-fat and skim milk.
If a mother is more than 50 pounds overweight after six weeks, a reduction in calories to not less than 2,200 calories per day is recommended for weight loss while breastfeeding. Weight reduction diets should be started no sooner than six weeks postpartum. Liquid diets, powders, and other very low-calorie diets are not recommended. These diets are basically starvation diets. In the breastfeeding mother, body fat will be retained and lean muscle tissue used for the production of breast milk. When the mother returns to her previous diet, the body will then store more fat as a defense mechanism against future episodes of starvation.
Besides watching calories, an increase in activity is suggested, beginning with a walk outdoors with the baby at least once a day. The mother should gradually work up to an exercise program.
How will various drugs affect me? Just as when she was pregnant, most items the mother ingests can be passed on to the baby through her breast milk. This does not mean that the medication is contraindicated during breastfeeding. The mother should inform her physician or dentist that she is nursing a baby if a prescription medication is needed to treat an illness.
Alcohol in moderation is usually not harmful. However, overuse of alcohol reduces the milk supply, is harmful for both the baby and the mother, and is not advised.
Smoking should be avoided. Nicotine appears in quantity in breast milk, and the effects of secondhand smoke negate some of the benefits of breastfeeding. There are a variety of smoking cessation aids available if necessary.
If the mother wants to breastfeed, the fact that she smokes should not be used by healthcare professionals to prevent her from nursing. She should try to limit smoking to only when she "needs" a cigarette, smoke outside, and try not to smoke for at least 20 minutes before nursing the baby. She should never smoke while holding the baby. Other smokers in the home should be encouraged to smoke outside. When given realistic parameters, many women find it easy to comply and may even be able to quit after cutting down.
Caffeine can make a baby wakeful, restless, fussy, and/or gassy. However, many babies do not appear to be bothered by caffeine. The mother should be encouraged to limit her intake of caffeine to no more than three servings of caffeine-containing foods or beverages per day (e.g., coffee, tea, cola, chocolate).
The mother should always check with her healthcare provider or a lactation consultant before using any over-the-counter medications, including herbs and holistic remedies.
The maternal use or addiction to illegal drugs should be a cause of major concern to any healthcare professional. Drug use becomes a matter of child endangerment if the mother wishes to breastfeed. Mothers with a history of drug abuse are routinely given a drug screen upon admission in labor by some hospitals. A mother who has a positive drug screen in labor has used drugs recently. If the mother did not stop using during pregnancy, she is not likely to stop using because she wants to breastfeed. ABM is the feeding method of choice in these cases. The mother will need drug counseling, and the appropriate referrals to local agencies must be made.
How can I best care for my breasts? The breasts should be washed daily with water only during the mother's shower. The breast does not need to be washed before or after every feeding. The mother can apply a few drops of breast milk to the nipple and areola after each feeding or use a commercially available nipple balm, as desired. Remind her that she should wash her hands well before each feeding, after changing diapers, and after using the bathroom.
Leaking of milk from the breasts is common in the early weeks of breastfeeding. The mother can use breast pads to absorb any leakage of milk. (Leaking will usually stop in a few weeks.) The mother should not use pads with plastic liners. Washable pads can also have a moisture-proof barrier in them and can become rough after repeated washing. The mother can make the best reusable pads herself or she can use folded 100% cotton handkerchiefs or super-absorbent diapers.
Inconvenient let-downs may occur any time. If the mother is in a place where she needs to stop a let-down, she should place the heel of her hands against her nipples and press in, toward the chest wall. The pressure will stop the flow. It is important to tell her not to do this too often as repeatedly stopping the flow can lead to painful plugged ducts and mastitis.
What special clothing will I need? No special clothing is needed to breastfeed. The following is just a suggestion: The new mother should wear a good fitting nursing or supportive bra as much as possible, especially in the early weeks when the breasts are quite heavy. It is not necessary to tell the mother that she must wear a bra at night. Many mothers feel more comfortable if they wear a bra at night, but this may be uncomfortable for other mothers.
How can I nurse without exposing my breasts? Once she is comfortable nursing the baby, nursing around others will be easy. Nursing fashions with hidden openings are available online and at maternity shops. Wearing loose-fitting tops that pull up, rather than unbutton, are better for breastfeeding in public. The baby will hide the mother's bare midriff. A blanket or large scarf thrown over her shoulder works well, if desired. If a mother is at home and is concerned about visitors, she can just excuse herself and go into another room if needed. People are becoming more accustomed to seeing women breastfeed but will occasionally make comments. Tell the mother to handle comments as diplomatically as possible, but never make excuses to anyone for breastfeeding her baby. She is nursing because she feels that breastfeeding is best for her baby.
Are there women who do not make enough milk? Yes, there are women who are unable to achieve a full milk supply. But many women can partially or fully feed their babies using their own breast milk. Support from family and friends along with the skilled expertise of a lactation consultant or supportive healthcare provider can be instrumental in achieving their breastfeeding goals.
Do I have to wake the baby for feedings? Many babies are sleepy in the first few days of life. This is especially true if the mother had a long labor, received oxytocin or pain medications, or if there is a great deal of molding or bruising of the baby's head. It is important to wake the baby for feedings if baby is not waking on their own at least 8 to 12 times in 24 hours to feed. Repeatedly skipping breastfeeding sessions can interfere with the milk supply and cause engorgement.
Letting the baby sleep may contribute to weight loss and dehydration. To wake a sleepy baby, stimulate by bathing the baby, undress to provide skin-to-skin contact, talk to the baby, or rock and pat the baby. It may also be useful to lower the environmental temperature [136].She should seek medical assistance if the baby seems too sleepy, does not want to nurse as often as expected, or constantly falls asleep at the breast before a period of active sucking and swallowing.
Once mother's milk has increased, she can stop watching the clock. The baby should wake to feed at least 8 to 12 feedings in a 24-hour period. Of course, if the baby demands to be fed more often, she should feed him or her.
How do I know if my baby is getting enough? Breastfeeding mothers often ask this question. She can be assured that all is well if, at five days postpartum:
Her milk has increased, as evidenced by mild engorgement of the breasts
She hears consistent infant swallowing at the breast
The baby has at least six to eight very wet diapers per day
Infant stools have turned yellow and follow most feedings
The baby begins to self-wake for feedings
The baby has at least 8 to 12 feedings per day
Satiation occurs (i.e., baby relaxes at the breast after several minutes of nursing with active swallowing)
The infant has gained weight from hospital discharge weight
Mothers often need to be reminded that babies suck reflexively. Sucking does not always suggest that the baby is still hungry. Reflexive suckling means that the baby will suck on anything that is put in or near his or her mouth.
Should I give my baby supplemental bottles of water or ABM until my milk comes in? Supplemental bottles of water or ABM are not recommended for the healthy newborn. There is a danger of artificial nipple preference in these early days. There is also some evidence that the hard surface of the bottle nipple is a supernormal stimulus. The baby may not react to the soft nipple and areola with suckling when returned to the breast.
A baby must learn how to nurse. It is best to avoid the use of bottles or pacifiers until breastfeeding is well-established. By giving bottles, the baby may have difficulty learning to do both or may develop a preference for the fast, easy flow and/or hard surface of the bottle nipple. In addition, mother's body must be convinced that there is a baby to feed while the milk-making hormones are at their highest levels. Bottles of water or ABM can lower the milk supply or lead to painful engorgement.
What if my baby needs bottles after birth because of a medical problem? A baby that needs supplemental feedings should always be given the supplement according to the feeding plan. If the baby gets used to the bottle and is having difficulty latching on when breastfeeding resumes, the mother should try:
A variety of feeding positions; in some cases, using the football hold can be helpful for better control of the latch-on
Dripping expressed milk or ABM down the breast as the baby attempts to latch-on
Being patient. A baby should never be forced to the breast. She should stop trying if the baby becomes agitated or upset.
Seeking assistance from a lactation consultant if the problem persists
What if my baby will not latch-on at all? The number one rule is: feed the baby. If mother can express enough milk, she should give it to the baby in a bottle. If she is unable to express enough milk, then donor breast milk or ABM is necessary.
Remind the mother that if her baby is not latching on, she needs to pump her breasts at regular intervals (at least 8 times per 24 hours) to maintain her milk supply until she can get assistance from a lactation consultant with the latch-on problem.
Is it normal for my baby to have liquid stools? Normal breastfed stool is almost all liquid with small milk curds. It is yellow, resembling runny egg yolks, and has very little odor. The baby will most likely have a stool after every feeding. In some cases, the stools are rather explosive sounding. Diarrhea in a breastfed baby is rare. Diarrhea stools are frequent, green in color, full of mucus and/or blood flecks, and are rather foul smelling. If there is any doubt, the mother should contact the baby's physician immediately.
Is it normal if my baby sleeps all day? A baby can be overly sleepy in the newborn period. Some infants just stir around a little in their cradle and, if left alone, will fall back to sleep. It is important for the mother to observe the infant for sleep-wake cycles. She should attempt to wake the baby when he or she begins to stir.
To wake the baby, mother should pick him or her up, talk, play, and/or rub his or her feet. Any combination of these may be needed to wake the baby for feedings.
What if my baby is awake all night? For the first several weeks, babies may be more active at night than during the day. In utero, the mother's daily activities "rocked" the baby to sleep during the day. Remind her of what happened every time she got into bed at night: the baby would wake up and begin to kick. The baby should settle into a normal day/night routine soon.
Babies can start to sleep through the night (defined as sleeping a six-hour stretch) around 8 to 12 weeks of age. Some babies take longer. There are occasionally a few who do not sleep through the night until toddlerhood. Mothers frequently believe that if they give the baby ABM or start solid food, the baby will sleep better. Feeding method makes no difference. Giving the baby cereal will not necessarily make the baby sleep through the night. It can backfire and make the baby more wakeful or cause gastric distress.
What about parenting advice that recommends scheduling feedings or not allowing the baby to nurse on demand so he or she will learn a routine? Babies have no sense of time. Letting them cry it out, even for a few minutes, can be an eternity of abandonment for infants. Mothers should be told to listen to their instincts, not ill-placed advice. A baby's needs should come first, within reason. If the advice she is following feels wrong for her baby, she should stop. Severely restrictive schedules and unrealistic behavior expectations can lead to failure to thrive or child abuse.
Should my baby be in his or her own room? The AAP recommends room sharing on a separate sleep surface for 6 to 12 months. Additionally, the AAP recommends that, "the infant crib or bassinet be placed in the parents' bedroom, which, when placed close to their bed, will allow for more convenient breastfeeding and contact" [285]. There are special cribs that connect to the parents' bed to allow babies to be moved over to feed, then slid back safely into their own bed. Soft surfaces, bumper pads, waterbeds, blankets, sheets, pillows, heated mattress pads, and stuffed chairs and couches are all dangerous for the baby; both sleeping on the stomach and entrapment are significant causes of infant death. An infant should never share a bed with older children.
Is it safe to have my baby in my bed? Breastfeeding mothers often find it more convenient if the baby sleeps in the same bed for night feedings. Although the possibility of rolling over on the baby exists, most mothers cradle the baby in their arm while they are asleep. Because mothers become amazingly light sleepers, they are aware of the baby's every movement. If a mother were to roll over on her baby, the baby would struggle and mother should wake up. There is evidence both for and against co-sleeping to prevent SIDS.
In their 2022 poly, the AAP states, "room sharing without bed sharing decreases the risk of sudden infant death syndrome (SIDS) by as much as 50% and is safer than bed sharing or having the infant sleep in a separate room" [285,298]. Infants may be brought into bed for nursing or comforting but should be returned to their own crib or bassinet when the parent is ready to return to sleep. This, of course, applies only to normal sleep. The mother (or father) who is taking pain medication, sleeping pills, or drinks any amount of alcohol will sleep very deeply; the baby should not be left in the bed in these cases. Extra pillows can be a hazard if the baby is in the bed. Parents should be warned to never put the baby face down on a traditional waterbed. The waterbed mattress can fold up around the infant's face or trap the infant so that he or she cannot move to breathe. Also, the parent's bed should be in the center of the room, away from any walls on the sides and/or bottom. Headboards should not leave any space where the baby might fall in and become trapped. Parents often place a rolled towel at the head of the bed to help prevent trapping. Infants have died when they have become trapped between the mattress and the wall. A baby should never be left in an adult-sized bed alone. Babies are more mobile than parents realize. It is easy for the baby who is left alone to squirm or crawl into cracks and crevices or under pillows and blankets and possibly suffocate.
What if I get sick? The mother should continue to breastfeed her baby, even if she is ill. Once the signs of a viral infection begin, the baby has already been exposed to the virus for several days before her symptoms started. The antibodies that the mother's body is making against the viral illness will go into the breast milk and help the baby get over the illness faster. If the mother is too ill to nurse, instruct her to pump her breasts. Remind the breastfeeding mother to check with her doctor before using any over-the-counter remedies. Antihistamines will lower the milk supply if she does not keep well hydrated. The mother should drink plenty of fluids and get lots of rest.
What if my baby gets sick? In the event of a fever greater than 101°F, an exceptionally congested nose, vomiting, and/or diarrhea, an infant's medical provider should be contacted. A congested nose will make it difficult for a baby to feed. Babies with ear infections may refuse to nurse because suckling may worsen ear pain. The mother can feed breast milk from an eye dropper, sipper cup, or spoon. The baby may demand to feed more when sick. The medical provider may recommend that the baby receive only liquids for a while; breastfeeding is appropriate. If the baby must be hospitalized, make arrangements with the staff for the mother to stay with the baby while hospitalized. If this is not possible, the mother should pump her breasts on a regular basis to maintain a good milk supply.
What is colic? Colic is a catchall term used by parents for a fussy baby. Infants cry for a variety of reasons. Parents should be told that crying is normal. Lengthy bouts of infant fussiness are often labeled as "colic" by the parents. It is normal for every baby to have a daily, predictable, fussy period, usually between 4:00 p.m. and 11:00 p.m. The baby may be reacting to the increased level of activity in the household. The mother should prepare for the fussy time by getting any possible household duties out of the way, before the baby gets fussy. She should use calming measures such as a warm bath, rocking the baby, soft music, going for a walk outdoors, swaddling, or a baby carrier.
In the medical profession, colic is defined as inconsolable crying accompanied by apparent intestinal discomfort. If the baby cannot be comforted, the mother should seek assistance from the baby's doctor. Do not advise the parents to let the baby "cry it out." This is very dangerous. If the baby is crying, there is a reason. Colic is often defined as crying for more than three hours a day, three times per week, for more than three weeks [286].
Colic may be caused by a variety of things (e.g., an immature digestive or nervous system, excess torsion of the head and neck at birth, sensitivity to the environment). One possibility is that colic is the result of a reaction to foreign proteins, especially to ABM. If there are no identifiable allergens in the mother's diet (e.g., cows' milk, eggs, nuts), colic symptoms are often the result of an overactive let-down reflex or overfeeding syndrome. Infants of mothers with an overabundant milk supply will often gain up to one pound per week. A very small percentage (2% to 3%) of infants diagnosed with colic have true colic. The remaining percentage can be attributed to other causes.
If the infant is exceptionally gassy or is gulping and choking at the breast, the baby may be getting too much milk too fast or swallowing large amounts of air. The mother should be instructed to burp the baby more often. If burping more frequently does not help with the fussiness, the baby should be examined by a pediatrician for any signs of illness. If illness has been ruled out, the mother should be referred to a lactation consultant before the baby is placed on any medication for colic. In most cases, reaction to oversupply syndrome is quite simple to correct by changing the baby's feeding pattern. A baseline infant weight is critical before mother changes the feeding pattern. After a change is made, the baby should have several weight checks to determine if the baby is continuing to gain well.
What if my baby has a slow weight gain? Seek assistance from a lactation consultant at once. The consultant will try to identify if there is a problem with breastfeeding. Together with the baby's physician, a lactation consultant can suggest a method of treatment to assist with the lack of weight gain. The mother should always continue to see the baby's doctor for follow-up care and any testing that may be needed to determine or eliminate a medical cause while under the care of a lactation consultant.
Is my baby getting all the necessary vitamins? Breast milk contains everything necessary for the baby. Vitamins, including fat-soluble A, D, E, and K, are present in highly usable forms in breast milk. The AAP has recommended that all breastfed infants receive 400 IU of oral vitamin D drops daily. The AAP also has recommended that supplementary fluoride not be provided during the first 6 months of life. From 6 months to 3 years of age, the decision whether to provide fluoride supplementation should be made on the basis of the fluoride concentration in the water supply and in other food and fluid sources and toothpaste [21,287].
Should I give my baby extra water or juice? Breast milk is approximately 90% water. Baby gets all the water he or she needs from breast milk, even on the hottest days. To ensure an adequate supply of breast milk, the mother should drink to quench thirst.
I will be returning to work. How can I best introduce my baby to a bottle? If the baby will need to take a bottle later because the mother is returning work, the first bottle should be introduced when breastfeeding is well-established. An occasional bottle after three or four weeks usually will not interfere with the milk supply or cause "nipple confusion."
There is the occasional baby who will not take a bottle. Some laypersons may believe that the baby should be left hungry to "starve him into submission." This attitude defies logic and is dangerous. An eye dropper, spoon, or a small cup (shot glass) can be tried when the baby will not accept the bottle. Infants who refuse to take anything other than the breast will often choose to sleep while the mother is away and "stack up" or frequency feed when she is home. If the parent experiences challenges related to bottle feeding, they should seek the assistance of a lactation consultant trained in bottle refusal assessment and management.
What if I lose my milk? This is highly unlikely. For the first few weeks, the mother's body is not aware that she did not have twins. After about six weeks, the breasts become more efficient at making milk and no longer need to overproduce. Supply has finally caught up with demand. The firmness once felt as engorgement is expected to disappear. Production of milk is now entirely dependent on the infant nursing at regular intervals. When the baby suddenly seems hungrier than usual, one thing to consider is that he or she may be having a "growing day." The baby is trying to bring the mother's milk supply up to these new needs. These growth spurts usually occur around 3 weeks, 6 weeks, 3 months, and 6 months of age. The baby should be back to the normal schedule within 24 to 48 hours. The mother should be instructed to monitor the baby's output during these growth spurts.
What if I feel like my milk supply is low? The mother should be advised to improve her nutritional status with three meals and two small snacks per day (2,200–2,500 calories) of well-balanced foods. Other helpful hints include:
Drink to thirst.
Use both breasts at each feeding.
Nurse more frequently.
Lengthen feedings.
Avoid supplemental bottles of water, ABM, or juice. Avoid using a pacifier.
Mother should seek assistance from a lactation consultant if she suspects a low milk supply.
Will a breastfed baby become constipated? For the entirely breastfed infant, constipation is rare. Because ABM has more waste products, constipation is a common problem with ABM use. Constipation means the passage of dry, hard stools, not the frequency of stools. After 6 weeks of age, it is not uncommon for the baby to have a bowel movement every day to every other day. It is important to teach the mother that her baby will develop his or her own stooling pattern. If any ABM is used, the infant should have a stool at least once a day.
What if my baby suddenly refuses to nurse? An ear infection can make it painful for the baby to nurse. Nasal congestion can also make nursing difficult. If the baby is ill or has a fever, call the baby's physician.
When babies wean themselves from the breast, it is a gradual process. Provided there are no signs of illness, this may be a "nursing strike." Has nursing become stressful? Is the household overly busy? Has the baby been frightened at the breast? Has he or she had a mouth injury? Is he or she teething? Did the mother change bath soap, deodorant, or perfume? Has the baby been getting a bottle or pacifier frequently? Is the baby learning how to crawl? (Pre-crawling boredom can make a baby too restless to settle down to nurse.) There are several things to try. Nurse the baby when he or she is asleep or almost asleep, in a darkened room. Offer the breast frequently but do not insist. If the problem continues for longer than 24 hours, seek assistance first from the baby's physician to be sure that the baby is not ill, then refer to a lactation consultant.
What if my baby only takes one breast and will not take the other? Babies occasionally develop a preference for one breast over the other. Ask the mother whether there is any difference in the shape of the nipple or the entire breast. Have her offer the breast that the baby will not take between feedings or when the baby is almost asleep. Also, she can try to hold the baby in the same position as when nursing from the other breast. The problem should resolve itself in one or two days. If the baby will not take the other breast at all, the mother should pump the breast that the baby has not nursed on to prevent plugged ducts and mastitis and to help keep the milk supply even. If the problem does not resolve quickly, she should refer to a lactation consultant for assistance.
What can I do when my baby bites? Newborns do not bite; they clamp. Biting occurs more often in the baby who is 4 to 6 months of age and older. If the baby bites, the mother should immediately break the latch and gently tell the baby, "Don't bite." She should then put the baby down and walk away. This signals that the feeding is over. The baby will soon understand that biting means not finishing the feeding. The mother should take care not to frighten the baby or raise her voice, as this can induce a nursing strike. Secondly, tell the mother to begin to pay close attention to the baby's nursing pattern. Mother will be able to feel the tongue pull back just before the bite. She should be instructed to quickly insert a finger into baby's mouth and stop the feeding. She should always end the feeding with the same words: "Don't bite." Babies usually bite when they are teething, so mother should check the baby's mouth for swollen gums or the beginning tips of the teeth poking through the gum. The pressure feels good on the sore gums. Babies are most likely to bite at the beginning of a feeding or as they fall asleep.
What if my baby will not sleep unless he or she nurses? Most children younger than 3 years of age cannot go to sleep without assistance of some kind. Nursing helps them relax and doze peacefully off. In some cases, a baby will become so overly tired that it will take quite a bit of effort to settle down enough to sleep.
If the baby begins to wake again during the night, the mother may need to nurse him or her back to sleep. Night waking usually resumes during teething. The mother should try calming the baby first.
Is it appropriate to nurse in front of my other children? Yes. What better way is there of teaching them the value and normalcy of breastfeeding than to be an example to them? The bottle is considered so normal in this culture that children's stories depict animals using bottles. Baby dolls and stuffed toys come with bottles. Children can grow up believing babies are supposed to be bottle fed. Breastfed toddlers have been known to "nurse" their dolls, imitating the mothering skills they have seen. A child may ask her mother to nurse a doll or teddy bear. Have the mother play along with the game.
What should I do if one of my older children asks to nurse? First, do not panic. This is a normal question. The mother should try to discover the reason for the question. Most children are merely curious about the taste of breast milk, especially if they are told that they were nursed, or they may want to know why the baby likes it so much. The mother can express her breast milk into a cup and let the child taste it.
What if my husband feels like he has nothing to do for the baby? In the early weeks, the father's job is to "mother the mother." He can be in charge of making sure she rests, gets enough to eat, help with the household chores, protect her from too many visitors and people with negative attitudes about breastfeeding, and help her assume her new role as a mother. Taking care of a baby involves more than feeding. Dad can change diapers, take the baby for a walk, play, or cuddle. The options are endless.
Will breastfeeding interfere with my sex drive? A new baby is a full-time job. The mother may simply need to go to bed to sleep and nothing more. This will happen no matter how she chooses to feed the baby. Some women report an increase in sex drive while nursing. Others report that they do not have as much of a drive as they used to. Either extreme or a combination of these is quite normal. It is important for couples to remember to make time for intimacy.
What about birth control? Breastfeeding may help with natural family spacing. As long as a baby is exclusively breastfed, menstrual periods may not return for several months. While breastfeeding delays the return of ovulation, the mother should be informed that awaiting the onset of menstruation before beginning contraception is not advised and may put her at risk of unintended pregnancy [170]. There are many contraceptive regimens available for breastfeeding women, including hormone, barrier, and sterilization options. It is important to note that the lactational amenorrhea method (LAM) of birth control is only effective if the mother is exclusively or almost exclusively breastfeeding, baby is younger than 6 months of age, and menstruation has not returned.
What if my breasts leak during sex? If the breasts leak during intercourse, the mother should nurse the baby before intercourse and pad the bed with towels. The same hormone that causes the let-down reflex, oxytocin, is released with orgasm. In the early weeks, the breasts often respond to the mother's orgasm with a let-down. This is not harmful and is quite normal.
Can my partner fondle or kiss my breasts? Yes. Some women find that their breasts are more (or less) sensitive to touch while they are nursing. If they are too sensitive, she should tell her partner. Some men may falsely believe that they will harm the milk if they touch or kiss the breasts, and some men are fascinated by the whole process of lactation. If her partner has an active herpes outbreak, however, he should not kiss the breasts, as the virus could be transmitted to the baby.
Is it normal to have trouble with vaginal dryness? The hormones involved with breastfeeding can cause vaginal dryness, which makes intercourse uncomfortable. Recommend using a water-soluble lubricant especially made for this purpose.
Will I lose my figure if I breastfeed? Quite the contrary, as the body has been storing fat (especially in the stomach, hip, and thigh areas) for nine months in preparation for breastfeeding. Nursing helps metabolize the majority of this stored fat quickly.
Do women ever get sexual sensations while they are nursing? Because the hormones involved in lactation are the same as those involved in orgasm, it is believed that a woman could have an orgasm while breastfeeding. Because oxytocin causes the uterus to contract, some women may misinterpret the cramping sensations as orgasmic contractions [288]. The most common description of sensation has been of a "warm afterglow," which many women experience following orgasm. Women appear to have the innate ability to separate the breast's maternal function from sexuality while they are nursing. Prolactin, the mothering hormone, gives the mother a calm, warm, peaceful feeling after nursing. These sensations are quite normal.
Is there a particular type of pump to use? There are several types of breast pumps available on the market. How and why a mother intends to use the pump can assist you in recommending a pump that suits her needs.
Will insurance companies cover the cost of the pump rental? Few insurance companies will reimburse the parents for the cost of the rental for a sick baby. Companies may view the rental of a hospital-grade electric pump as a luxury when there are small hand pumps available at a lesser cost. If the pediatrician writes a prescription for the hospitalized baby for "breast milk," the parents should send a copy of the prescription and the pump rental receipt to the insurance carrier for reimbursement.
How do I keep the equipment clean? Always use clean equipment for pumping. Instructions for cleaning come with most pumps. Only the parts that contact the breast or milk must be cleaned after each use. The mother should rinse the parts immediately after each use in cold water to remove any residual milk. The parts should then be washed in hot, soapy water and rinsed well. Between uses, the pump kit should be stored in a clean area or clean plastic zip-type bag. Daily sterilization of the breast pump parts (not the tubing) is recommended only when the milk is collected for a hospitalized infant. Sterilization can be done by boiling the parts in a large pot at full boil for five minutes. If the mother notices hard water spots on the equipment, adding a little white vinegar to the water each time she sterilizes the parts should help. If the tubing has a filter, be sure that the filter does not get wet, as this will block the suction.
Will I pump as much as the baby takes? The pump is sometimes not as efficient as the baby. It also may take a while for the mother's let-down reflex to become trained to the pump. If no let-down reflex occurs, the mother can expect to obtain 0.5 to 1 ounce or less of milk from each breast. The mother who is pumping immediately after delivery for a sick baby may at best express only a drop or two of colostrum. The goal of early pumping is to establish a milk supply by repeated stimulation of the breasts.
While not all techniques work for all mothers, there are many things she can do to encourage the let-down reflex:
Relax. Try breathing techniques.
Apply warm packs or washcloths to the breast for a few minutes before pumping or take a warm shower.
Massage the breasts gently, with the heel of your hand, starting at the chest wall and moving toward the areola.
Bend over and let the breasts hang free for a moment or two.
Develop a pumping routine. Always follow the same steps each time you pump.
Turn the pump on for a minute before placing the flange against the breast.
Stroke your breasts in a feather light manner from the upper edge near the arm pit toward the nipple and areola while you are pumping.
Look at pictures of your baby.
Listen to a recording of your baby's cries.
Try smelling a piece of unwashed clothing that the baby has recently worn. Smells can be very powerful.
Do not worry about how much you obtain.
How is manual expression of breast milk done? Manual expression of breast milk is easy after a mother has learned the technique. The mother will need a clean, wide-mouthed jar, bowl, or funnel because it is difficult to aim the streams of milk into a small opening. With practice, some mothers feel that manual expression is easier and less time-consuming than a breast pump. The technique for manual expression involves the following steps:
Position the thumb and first two fingers at the edge of the areola (Figure 4).
Push in toward the chest wall, spreading the fingers apart slightly.
Let the thumb and fingers roll toward the nipple using light compression. (Do not squeeze.)
Rhythmically repeat compressions.
Rotate the thumb and fingers around the breast to ensure emptying of the ducts. Remember to avoid squeezing, pulling, or sliding on the breast or nipple.
How should I store my milk? Store milk in sealed, sterilized containers. Containers should be labeled with the date and time that it was expressed. Place the container in ice water to cool it quickly. If the expressed milk is to be used for the baby within 24 to 48 hours, keep it refrigerated until just before use. When freezing milk, containers of breast milk should be placed in the back of the freezer. Never store breast milk in the freezer door or near freezer walls. Breast milk will keep in the refrigerator for four days and in the refrigerator-freezer for about six months; in a chest freezer, or deep freeze, breast milk will keep for about one year at 0°F. However, because breast milk is constantly changing to meet the baby's needs at a given age, it is best to use breast milk within one or two weeks of expression. Breast milk should not be stored for long periods.
It is best to freeze milk in small amounts (2 oz.) to avoid waste and decrease defrost time. Baby bottles are not ideal for freezer storage because there can be a large air space at the top and the bottle may not be airtight. Special bags are available that protect the breast milk from freezer burn. Plastic nurser bags or bottle liners should not be used.
How should I defrost breast milk? If the milk will be used within the next 24 hours, defrost it in the refrigerator overnight. Frozen milk can be quickly defrosted by placing it in tepid (warm, not hot) tap water and gradually adding warm water. Never heat breast milk on a stove or in a microwave oven. High heat alters the proteins in the breast milk. Shake or rotate the container gently to mix the contents while it is defrosting. Be sure to discard any milk left in the bottle after a feeding. Reuse can cause serious illness.
Why does my milk have a yellow layer at the top? A yellow or white layer at the top of cooled or frozen breast milk is simply the cream layer of the milk that separates as the milk is cooled.
Can I mix my milk and ABM in the same bottle? ABM may inactivate some of the components of breast milk. It is best not to mix breast milk and ABM in the same bottle. Give the breast milk first and make up the rest of the feeding, if needed, with ABM added to the empty bottle.
What type of nipple should be used for bottle feedings? Actually, there is no single "best" type of bottle nipple, except the one the baby will take. The orthodontic has a small hole on the top side, rather than a large hole in the tip. The small hole keeps the flow from becoming too fast for the breastfed infant to control. A soft silicone nipple works well. The baby's mouth should be open wide around the base of the nipple, not closed around the narrower neck of the nipple. Short, stubby nipples that do not allow the baby to open his or her mouth wide can interfere with proper latch-on by encouraging the baby to feed at the tip. Standard bottle nipples should not have a large hole. (Test by holding the bottle upside down; milk should drip slowly from the top, not run out.)
Can I breastfeed and work? The primary problem in working and nursing is that the woman is the working mother of a young infant. The feeding method does not change some of the difficulties associated with working with an infant at home. However, in reality, thousands of mothers work and continue to breastfeed. Each mother has to decide what is best for her and her baby. There are several ways to manage working and breastfeeding. Here are a few suggestions:
Work at home or work part-time.
Pump milk while at work or have the baby brought to her for feedings.
Reverse the baby's nursing pattern so he or she nurses when mother is home and sleeps while she is gone. (Babies sometimes do this on their own.)
Partially wean the baby, giving ABM during the hours that she would be away at work and nursing only during the hours that she will be home. This eliminates the need for pumping in most mothers. It will take several days for the mother's milk supply to adjust. In many ways, this schedule is like nursing a toddler two or three times per day. For the child who must be in day care with other children, even partial breastfeeding has been shown to provide protection against illness.
How often will I need to pump? It will depend on the age of the baby when the mother returns to work and how long she will be away from the baby. A simple schedule would be to take the number of hours away from the baby (include drive time to and from work) and divide those hours by 3. The pumping parent should pump that number of times while away from the baby. If a mother can wait to return to work until the baby is 6 months of age, she may only need to pump once in the middle of her workday, or she may not need to pump at all.
What is the best breast pump for a working mother? I think the best pump for the mother who returns to work early and works full time (away from baby eight hours or more) is an electric breast pump with a double pumping kit. When the mother pumps both breasts simultaneously, pumping will take 10 to 15 minutes. Hand or single electric pumps require 30 minutes or longer to use. Double pumping also will help maintain her milk supply.
Is renting an electric pump expensive? The initial cash needed for renting a pump can be high because most rental stations require that the rental fee and a security deposit be paid in full at the time of rental. In most states, sales tax is added to the rental fee. Call around and get several price quotes. Pump rental averages $30 to $60 per month. Some rental stations offer a reduced rate for long-term rentals. Many women find this less expensive than ABM over the same span of time.
Are the large electric pumps available for purchase? The mother can purchase a large hospital-type electric pump from the manufacturers. Cost varies from $500 to $2,500. Most of these pumps have a life span of 30 years. Non-hospital-grade electric pumps are also available for $50 to $400. Under the Affordable Care Act, marketplace health insurance plans (except grandfathered plans) must cover the cost of a breast pump, whether rented or purchased [289]. Individual plan guidelines will vary.
On Monday I can pump several ounces, then less and less by Friday. Is this normal? The mother's body is adjusting to infrequent nursing at the breast. To increase the amount of breast milk, mother should increase her fluid intake, pump more frequently throughout the workday, rest, and nurse the baby as much as she can when she is home.
How can I store milk safely at work? Many women use a small cooler when a refrigerator is unavailable for storage. Cooler bags, which look like tote bags, are available through pump rental stations and baby shops. The cooler will usually hold several bottles of milk and may have a place for pumping equipment. Reusable cooling units are less messy than blocks or cubes of ice.
How can I tell if breast milk has gone bad (turned sour)? Unlike cows' milk, breast milk may not smell sour if it has spoiled. If the milk is curdled, severely separated, or has a bad or nasty smell, throw it out.
What if the baby will not eat while I am gone? Tell the sitter to keep trying but do not insist. If the baby will not take a bottle, an eye dropper or a spoon may work. Some mothers find that they have to experiment with several types of bottle nipples before they find one that the baby "likes." If the baby is older (i.e., 4 to 6 months), ask your pediatrician if solids may be safely introduced. The sitter may be able to give the baby solid foods instead of giving a bottle while the mother is away. Some babies prefer to "stack-up" on feedings while the mother is at home and not eat when she is gone. Other babies may nurse frequently when she arrives home.
How long is long enough to breastfeed? The choice is entirely the mother's to make. Authorities agree that all babies should be breastfed for as long as possible in infancy. There is never a time when breastfeeding no longer provides benefits for the baby and the mother. It is best to give the mother who asks this question general recommendations and let her decide when to discontinue breastfeeding. Major health agencies recommend exclusive breastfeeding for at least the first six months and continued breastfeeding during the introduction of solids. The Surgeon General, the WHO, and the AAP have recommended breastfeeding through the second year [21].
How is weaning usually done? Weaning begins when the baby starts taking solids at 6 months or is given ABM feedings to replace breastfeedings. The mother can begin by eliminating the least stressful feeding of the day and substituting solid food or ABM. Gradually, reduce the number of breastfeedings per day over a period of weeks. The last two feedings to be eliminated are first thing in the morning and at bedtime. The mother will need to decide which feeding is the most important for the baby and eliminate that one last.
What if my baby will not wean? Then the baby is not ready to wean. An older child may sense what the mother is doing and resist all efforts by wanting to nurse more. Tell the mother to try distraction. La Leche League International has suggested: "Don't offer. Don't refuse." When the child asks to nurse the mother should use a "not now, but later" routine and be sure that when later arrives, she does nurse as promised. This will help restore the child's trust that mother is not abandoning him or her.
Should I wean my baby from my breast to a cup or a bottle? Depending on the age of the baby, a cup may be the better choice. After four to six months, a baby can handle liquids from a training cup well. If the mother weans to a bottle, remind her that she will later have to wean the baby from the bottle.
When should I start my baby on solids? Solids should be introduced at6 months of age. The baby should have doubled his or her birth weight, lost the extrusion reflex (the reflex to push everything out of the mouth), and begun to show an interest in what others are eating. Some babies are not ready for solids until 6 to 8 months of age. Let the infant take the lead. The mother should hold the baby in her lap for the first few feedings and use infant-sized spoons. Cereal or other solids should never be put in a bottle or infant feeder. If a baby cannot take food from a spoon, he or she is not ready for solids. The pediatrician will give the mother a schedule of what foods to introduce and when. Usually a baby starts on cereal, vegetables, fruit, meat, and finally egg whites. The order may vary depending on the pediatrician. The most important thing to remember is to go slowly. Do not overfeed the baby. Breast milk or ABM should remain the major source of nutrition until 1 year of age.
Should I give my baby cows' milk? Children younger than 1 year of age should not receive cows' milk. It is irritating to the baby's intestine and can cause occult intestinal bleeding that results in anemia. If breastfeeding is discontinued before the baby is 1 year of age, the mother should use commercial ABM. Kitchen-prepared formulas are not nutritionally complete. After 1 year, cows' milk may be used. Whole milk, not skim or low-fat milk, is often recommended for young children. Babies need larger amounts of fat in their diets compared to adults to assist with brain and nervous system growth. Do not allow cows' milk to become the child's major source of calories. An infant needs a well-balanced diet to grow properly. Watch out for fruit juices as well. If a baby is constantly drinking fruit juice, the major source of calories will be from the sugar in the fruit juice. Hunger will be satisfied, and baby may not eat other foods or may nurse less often. Too much sugar can contribute to childhood obesity.
Can I put the baby to bed with a bottle? Putting the baby to bed with a bottle of ABM, milk, or juice can cause "nursing bottle" cavities in the baby's teeth. This type of dental decay is rare in infants who are nursed to sleep. Breast milk does not appear to have the same high sugar that leads to dental cavities. If a mother must put the baby to bed with a bottle, it should be plain water, and she should watch for choking. The baby should not suck on a bottle all night, or all day, for that matter. The bottle should be taken away after the baby is asleep.
Breastfeeding has numerous benefits for both mother and baby. However, societal pressure, common misconceptions, social stigma, and lack of education may prevent some women from starting or continuing to breastfeed. The role of the healthcare professional is to educate, support, and encourage breastfeeding while also remembering that the choice to breastfeed for any amount of time remains solely with the mother.
| Doulas of North America (DONA) International |
| https://www.dona.org |
| (888) 788-3662 |
| Human Milk Banking Association of North America |
| https://www.hmbana.org |
| (817) 810-9984 |
| Academy of Breastfeeding Medicine |
| http://www.bfmed.org |
| (800) 990-4226 |
| International Baby Food Action Network (IBFAN) |
| https://www.ibfan.org |
| International Board of Lactation Consultant Examiners (IBLCE) |
| https://www.iblce.org |
| (703) 560-7330 (North America) |
| International Childbirth Education Association (ICEA) |
| https://icea.org |
| (919) 674-4183 |
| International Lactation Consultant Association (ILCA) |
| https://www.ilca.org |
| (919) 861-5577 |
| La Leche League International (LLLI) |
| https://www.llli.org |
| (800) 525-3243 |
| Nursing Mothers Counsel |
| https://www.nursingmothers.org |
| San Francisco Bay Area: (650) 327-6455 |
| Santa Cruz County: (831) 688-3954 |
| Geddes Productions, LLC |
| https://www.geddesproduction.com |
| (323) 344-8045 |
| Bright Future Lactation Resource Centre Ltd. |
| https://bflrc.org |
| (937) 438-9458 |
| Baby-Friendly USA |
| https://www.babyfriendlyusa.org |
| (518) 621-7982 |
Includes a list of Baby-Friendly Hospitals in the United States as well as information for:
Hospitals and birth centers
Parents
Breastfeeding advocates and healthcare professionals
| Drugs and Lactation Database (LactMed) |
| https://www.ncbi.nlm.nih.gov/books/NBK501922 |
| U.S. Food and Drug Administration MedWatch |
| https://www.fda.gov/medwatch/index.html |
| (888) 463-6332 |
| World Health Organization (WHO) |
| https://www.who.int/health-topics/breastfeeding |
Abscess: A process whereby the body walls off or separates an area of infection. An abscess requires surgical incision and drainage.
Breastfeeding:
Exclusive: Baby receives all nourishment and fluids at the breast.
Almost exclusive: Baby receives all nourishment at the breast except for small amounts of culturally valued supplements.
Partial breastfeeding: Frequent or regular supplements.
Token breastfeeding: Minimal breastfeeding.
Breast milk feeding: Separates mother from the process, allows for bottles of pumped milk.
Colic: Muscle spasms of any tubular organ. Most commonly due to intestinal cramping in infants.
Dehydration: Loss of essential body fluids. Critical in infants younger than 1 year of age. Can be caused by poor nutritional intake, vomiting, diarrhea, or thermal losses.
Engorgement: Swelling of the breast, lactating, and surrounding tissue.
Failure to thrive: An infant who, despite adequate caloric intake, fails to grow.
Galactocele: Milk retention cyst in the breast due to the dilatation and obstruction of a milk duct. The cyst is usually filled with milk at first and later the fluid becomes thicker. The cyst can be aspirated but will fill again [136].
Galactopoiesis: Maintenance of lactation.
Newborn hypoglycemia: Low blood sugar, defined as less than 40 mg/100 mL in the full-term infant or 30 mg/100 mL in the premature infant during the first hours of life.
Induced lactation: Causing breast milk production in a woman who has not recently been pregnant.
Insufficient glandular development of the breast: Lack of breast tissue caused by inadequate or poor development of the lactating structure in the breast. Can be unilateral or bilateral or may affect only one portion of the breast.
Jaundice/hyperbilirubinemia: Presence of bilirubin in the bloodstream greater than 5 mg/dL.
Large for gestational age: Infant with birth weight greater than the 95th percentile for gestational age.
Mammalgia: Pain in the breast.
Mastectomy: Removal of all or part of the breast tissue. Can also include removal of underlying muscle and lymphatic tissue.
Mastitis: Infection of the breast.
Necrotizing enterocolitis (NEC): Condition more common in premature infants whereby area of the bowel becomes necrotic (or dies). Can be due to bacteria or lack of blood supply.
Relactation: Stimulating a milk supply in a woman who has recently given birth but did not breastfeed or recently weaned her baby.
Small for gestational age: Infant whose birth weight is less than the tenth percentile for gestational age. The infant is born with a nutritional deficit.
Sucking: Process where liquid is drawn into the mouth with negative pressure. Tongue action is primarily an up and down motion with very little jaw movement (bottle feeding).
Suckling: A method of sucking which involves an extension and retraction of the tongue. Liquid is obtained with a rhythmic licking motion of the tongue combined with jaw opening and closing (breastfeeding).
Thrush: Candida overgrowth in an infant's mouth or on mother's nipples. Candida (monilia) rashes can also be present in the diaper area.
1. Lawrence RA, Lawrence RM. Breastfeeding: A Guide for the Medical Profession. 8th ed. St Louis, MO: C.V. Mosby; 2015.
2. Wambach K, Riordan J. Breastfeeding and Human Lactation, Enhanced Fifth Edition. Sudbury, MA: Jones and Bartlett; 2015.
3. Greiner T. The concept of weaning: definitions and their implications. J Hum Lact. 1996;12(2):123-128.
4. Edwards M. Reclaiming Birth: History and Heroines of American Childbirth Reform. Trumansburg, NY: The Crossing Press; 1984.
5. Dettwyler KA, Stuart-Macadam P (eds). Breastfeeding: Biocultural Perspectives. New York, NY: Aldine De Gruyter; 1995.
6. Baumslag N, Michels DL. Milk, Money, and Madness: The Culture and Politics of Breastfeeding. Westport, CT: Bergin and Garvey; 1995.
8. Wolf JH. The social and medical construction of lactation pathology. Women Health. 2000;30(3):93-110.
9. Eiger MS, Wendkos Olds S, Marks L. The Complete Book of Breastfeeding. 4th ed. New York, NY: Workman Publishing; 2009.
12. Rodriguez-Garcia R, Frazier L. Cultural paradoxes relating to sexuality and breastfeeding. J Hum Lact. 1995;11(2):111-115.
15. National Conference of State Legislatures. Breastfeeding State Laws. Available at https://www.ncsl.org/health/breastfeeding-state-laws. Last accessed April 26, 2025.
16. State of Idaho. Housebill No. 448, 2018. ID law 181. Available at https://legislature.idaho.gov/sessioninfo/2018/legislation/H0448/. Last accessed April 26, 2025.
17. GovTrack.us. Advanced Search for Legislation: Breastfeeding. Available at https://www.govtrack.us/congress/bills/browse?q=breastfeeding#text=breastfeeding. Last accessed April 26, 2025.
18. Gartner LM. On the question of the relationship between breastfeeding and jaundice in the first 5 days of life. Semin Perinatol. 1994;18(6):502-509.
20. American Academy of Pediatrics. From the American Academy of Pediatrics policy statement: breastfeeding and the use of human milk. Pediatrics. 2012;129(3):e827-e841.
21. Gertner LM, Morton J, Lawrence RA, et al. Breastfeeding and the use of human milk. Pediatrics. 2005;115(2):496-506.
22. Centers for Disease Control and Prevention. Vitamin D and Breastfeeding. Available at https://www.cdc.gov/breastfeeding-special-circumstances/hcp/diet-micronutrients/vitamin-d.html. Last accessed April 26, 2025.
23. Centers for Disease Control and Prevention. Infant Mortality. Available at https://www.cdc.gov/maternal-infant-health/infant-mortality. Last accessed April 26, 2025.
25. MacDorman MF, Mathews TJ, Mohangoo AD, Zeitlin J. International comparisons of infant mortality and related factors: United States and Europe, 2010. Nat Vital Stat Rep. 2014;63(5):1-7.
26. Chen A, Oster E, Williams H. Why is infant mortality higher in the United States than in Europe? Am Econ J Econ Policy. 2016;8(2): 89-124.
27. United Health Foundation. America's Health Rankings. International Comparison: Infant Mortality. Available at https://www.americashealthrankings.org/learn/reports/2023-annual-report/international-comparison. Last accessed April 26, 2025.
28. U.S. Department of Health and Human Services. Healthy People 2030: Objectives and Data. Available at https://health.gov/healthypeople/objectives-and-data/browse-objectives. Last accessed April 26, 2025.
29. Centers for Disease Control and Prevention. About Breastfeeding Data. Available at https://www.cdc.gov/breastfeeding-data/about. Last accessed April 26, 2025.
30. Spisak S, Gross S (eds). Second Follow-Up Report: The Surgeon General's Workshop on Breastfeeding and Human Lactation. Available at https://profiles.nlm.nih.gov/spotlight/nn/catalog/nlm:nlmuid-101584932X750-doc. Last accessed April 26, 2025.
31. Waddell A (ed). The Reports of the Surgeon General: Surgeon General's Workshop on Breastfeeding and Human Lactation. Available at https://profiles.nlm.nih.gov/spotlight/nn/catalog/nlm:nlmuid-101584932X759-doc. Last accessed April 26, 2052.
32. Galson SK. Surgeon General's perspectives. The 25th anniversary of the Surgeon General's workshop on breastfeeding and human lactation: the status of breastfeeding today. Public Health Rep. 2009;124:356-358.
33. U.S. Surgeon General. HHS Blueprint for Action on Breastfeeding. J Perinat Educ. 2001;10(1):45-47.
34. Ip S, Chung M, Raman G, et al. Breastfeeding and maternal and infant health outcomes in developed countries. Evid Rep Technol Assess. 2007;(153):1-186.
35. DiGirolamo AM, Rollins School of Public Health, Emory University, et al. Breastfeeding-related maternity practices at hospitals and birth centers—United States, 2007. MMWR. 2008;57(23):621-625.
36. U.S. Department of Health and Human Services. Office on Women's Health. Business Case for Breastfeeding. Available at https://www.womenshealth.gov/breastfeeding/breastfeeding-home-work-and-public/breastfeeding-and-going-back-work/business-case. Last accessed April 26, 2025.
37. Fein SB, Mandal B, Roe BE. Success of strategies for combining employment and breastfeeding. Pediatrics. 2008;122:S56-S62.
38. U.S. Department of Health and Human Services Office of Disease Prevention and Health Promotion. Healthy People 2030. The Surgeon General's Call to Action to Support Breastfeeding. Available at https://odphp.health.gov/healthypeople/tools-action/browse-evidence-based-resources/surgeon-generals-call-action-support-breastfeeding. Last accessed April 26, 2025.
39. Office of the Surgeon General. The Surgeon General's Call to Action to Support Breastfeeding. Rockville, MD: Office of the Surgeon General; 2011.
40. World Health Organization. International Code of Marketing of Breast-Milk Substitutes. Geneva: World Health Organization; 1981.
41. Frank DA. Commercial discharge packs and breastfeeding counseling: summary of a study. J Hum Lact. 1989;5(1):7-10.
42. Southall DP, Burr S, Smith RD, et al. The Child-Friendly Healthcare Initiative (CFHI): healthcare provision in accordance with the UN convention on the rights of the child. Pediatrics. 2000;106(5):1054-1064.
43. Cramton R, Zain-Ul-Abideen M, Whalen B. Optimizing successful breastfeeding in the newborn. Curr Opin Pediatr. 2009;21(3):386-396.
44. DiGirolamo AM, Grummer-Strawn LM, Fein SB. Effect of maternity-care practices on breastfeeding. Pediatrics. 2008;122:S43-S49.
45. Baby-Friendly USA. The Ten Steps And International Code. Available at https://www.babyfriendlyusa.org/for-facilities/practice-guidelines/10-steps-and-international-code/. Last accessed April 26, 2025.
46. Labbok MH. Global baby-friendly hospital initiative monitoring data: update and discussion. Breastfeed Med. 2012;7:210-222.
47. Baby-Friendly USA. Find a Baby-Friendly Facility. Available at https://www.babyfriendlyusa.org/for-parents/find-a-baby-friendly-facility/. Last accessed April 26, 2025.
48. Philipp BL, Malone KL, Cimo S, Merewood MA. Sustained breastfeeding rates at a U.S. baby-friendly hospital. Pediatrics. 2003;112(3):e234-e236.
49. Forster DA, McLachlan HL. Breastfeeding initiation and birth setting practices: a review of the literature. J Midwifery Womens Health. 2007;52(3):273-280.
50. Broadfoot M, Britten J, Tappin DM, MacKenzie JM. The Baby Friendly Hospital Initiative and breast feeding rates in Scotland. Arch Dis Child Fetal Neonatal Ed. 2005;90(2):F114-F116.
51. Merten S, Dratva J, Ackermann-Liebrich U. Do baby-friendly hospitals influence breastfeeding duration on a national level? Pediatrics. 2005;116(5):e702-e708.
52. DiGirolamo AM, Grummer-Strawn LM, Fein S. Maternity care practices: implications for breastfeeding. Birth. 2001;28(2):94-100.
53. Cattaneo A, Buzzetti R. Effect on rates of breast feeding of training for the Baby Friendly Hospital Initiative. BMJ. 2001;323(7325):1358-1362.
54. Powers NG, Slusser W. Breastfeeding update 2: clinical lactation management. Pediatr Rev. 1997;18(5):147-161.
55. DiGirolamo AM, Grummer-Strawn LM, Fein SB. Do perceived attitudes of physicians and hospital staff affect breastfeeding decisions? Birth. 2003;30(2):94-100.
56. Powers NG, Naylor AJ, Wester RA. Hospital policies: crucial to breastfeeding success. Semin Perinatol. 1994;18(6):517-524.
57. Kotz D. All Massachusetts Maternity Hospitals Now Ban Infant Formula Gift Bags. Available at https://www.boston.com/uncategorized/noprimarytagmatch/2012/07/12/all-massachusetts-maternity-hospitals-now-ban-infant-formula-gift-bags/. Last accessed April 26, 2025.
58. Delaware Public Media. Delaware Bans Hospital Gift Bags of Baby Formula. Available at https://www.delawarepublic.org/science-health-tech/2015-07-23/delaware-bans-hospital-gift-bags-of-baby-formula. Last accessed April 26, 2025.
59. West Virginia Breastfeeding Alliance. West Virginia Has Banned the Bags! Available at https://www.wvbreastfeeding.org/ban-the-bags. Last accessed April 26, 2025.
60. Dodgson JE, Tarrant M. Outcomes of a breastfeeding educational intervention for baccalaureate nursing students. Nurse Educ Today. 2007;27(8):856-867.
61. Brown LP, Bair AH, Meier PP. Does federal funding for breastfeeding research target our national health objectives? Pediatrics. 2003;111(4):e360-e364.
62. Centers for Disease Control and Prevention. Breastfeeding. Available at https://www.cdc.gov/breastfeeding/php/about/index.html. Last accessed April 26, 2025.
63. United States Department of Agriculture Food and Nutrition Service. USDA Announces Grants to Support State Breastfeeding Programs. Available at https://www.fns.usda.gov/pressrelease/2014/fns-0001-14. Last accessed April 26, 2025.
64. National Institute of Child Health and Human Development. NICHD Breastfeeding and Breast Milk Research Information. Available at https://www.nichd.nih.gov/health/topics/breastfeeding/researchinfo. Last accessed April 26, 2025.
66. Carlson SE. Docosahexaenoic acid supplementation in pregnancy and lactation. Am J Clin Nutr. 2009;89:678S-684S.
67. van Goor SA, Dijck-Brouwer DAJ, Hadders-Algra M, et al. Human milk arachidonic acid and docosahexaenoic acid contents increase following supplementation during pregnancy and lactation. Prostaglandins Leukot Essent Fatty Acids. 2009;80(1):65-69.
68. Koletzko B, Lien E, Agostoni C, et al. The roles of long-chain polyunsaturated fatty acids in pregnancy, lactation and infancy: review of current knowledge and consensus recommendations. J Perinat Med. 2008;36:5-14.
69. Koletzko B, Cetin I, Brenna JT, for the Perinatal Lipid Intake Working Group. Consensus Statement: dietary fat intakes for pregnant and lactating women. Br J Nutr. 2007;98(5):873-877.
70. Benisek D, Shabert J, Skornik R. Dietary intake of polyunsaturated fatty acids by pregnant or lactating women in the United States. Obstet Gynecol. 2000;95:S77-S78.
71. Frithsen I, Goodnight W. Awareness and implications of fish consumption advisories in a women's health setting. J Reprod Med. 2009;54:267-272.
72. Bloomingdale A, Guthrie LB, Price S, et al. A qualitative study of fish consumption during pregnancy. Am J Clin Nutr. 2010;92:1234-1240.
73. Vallaeys C. Replacing Mother—Imitating Human Breast Milk in the Laboratory. Available at https://www.cornucopia.org/DHA/DHA_FullReport.pdf. Last accessed April 26, 2025.
74. U.S. Food and Drug Administration. Questions and Answers for Consumers Concerning Infant Formula. Available at https://www.fda.gov/food/people-risk-foodborne-illness/questions-answers-consumers-concerning-infant-formula. Last accessed April 26, 20255.
75. AAP Committee on Nutrition. New infant formula additives approved by FDA. AAP News. 2002;20(5):209-210.
76. Institute of Medicine of the National Academies, Committee on the Evaluation of the Addition of Ingredients New to Infant Formula, Food and Nutrition Board. Infant Formula: Evaluating the Safety of New Ingredients. Washington, DC: The National Academies Press; 2004.
77. Stuebe AM, Schwarz EB. The risks and benefits of infant feeding practices for women and their children. J Perinatol. 2010;30(3):155-162.
78. Estrada B. Infections Associated with Powdered Infant Formula. Available at https://novax.org/wp-content/uploads/2016/04/formulaNbacteria.pdf. Last accessed April 26, 2025.
79. Himelright I, Harris E, Lorch V, et al. Enterobacter sakazakii infections associated with the use of powdered infant formula—Tennessee, 2001. MMWR. 2002;51(14):298-300.
80. Bhalla V, Grimm PC, Chertow GM, Pao AC. Melamine nephrotoxicity: an emerging epidemic in an era of globalization. Kidney Int. 2009;75(8):774-779.
81. Blackwell AG, Salisbury L. Administrative petition to relieve the health hazards of promotion of infant formulas in the U.S. Birth. 2007;8(4):287-296.
84. U.S. Food and Drug Administration. FDA Investigation of Cronobacter Infections: Powdered Infant Formula (February 2022). Available at https://www.fda.gov/food/outbreaks-foodborne-illness/fda-investigation-cronobacter-infections-powdered-infant-formula-february-2022. Last accessed April 26, 2025.
86. Bernshaw NJ. Does breastfeeding protect against sudden infant death syndrome? J Hum Lact. 1991;7(2):73-79.
87. Gribble KD. Mechanisms behind breastmilk's protection again, and artificial baby milk's facilitation of, diarrhoeal illness. Breastfeed Rev. 2011;19(2):19-26.
88. Oddy WH. Breastfeeding protects against illness and infection in infants and children: a review of evidence. Breastfeed Rev. 2001;9(2):11-18.
89. Gigliotti E. When women decide not to breastfeed. MCN Am J Matern Child Nurs. 1995;20(6):315-321.
90. Ritz J. World's Fastest Growing Functional Food? Infant Formula. Available at https://thepaleomama.com/2014/01/17/cost-of-formula-feeding/. Last accessed April 26, 2025.
91. Bartick M, Reinhold A. The burden of suboptimal breastfeeding in the United States: a pediatric cost analysis. Pediatrics. 2010;125(5):e1048-e1056.
93. Greer FR, Apple RD. Physicians, formula companies, and advertising: a historical perspective. Am J Dis Child. 1991;145(3): 282-286.
94. Starling S. World's Fastest Growing Functional Food in 2013? Infant Formula (By a Mile). Available at https://www.dairyreporter.com/Article/2013/10/30/Infant-formula-is-world-s-fastest-growing-functional-food-in-2013. Last accessed April 26, 2025.
95. Statista. Revenue Generated By the Fortified/Functional Food Market Worldwide between 2013 and 2022 (in Billion U.S. Dollars). Available at https://www.statista.com/statistics/252803/global-functional-food-sales. Last accessed April 26, 2025.
96. U.S. Government Accountability Office. Report to Congressional Addressees. Breastfeeding. Some Strategies Used to Market Infant Formula May Discourage Breastfeeding; State Contracts Should Better Protect Against Misuse of WIC Name. Washington, DC: U.S. Government Accountability Office; 2006.
97. U.S. Department of Agriculture. Economic Research Service. WIC Program. Available at https://www.ers.usda.gov/topics/food-nutrition-assistance/wic-program. Last accessed April 26, 2025.
98. Arias D. Infant formula marketing can discourage U.S. breastfeeding: low-income moms are at higher risk. Nations Health. 2006;36(3).
100. Hetzner NM, Razza RA, Malone LM, Brooks-Gunn J. Associations among feeding behaviors during infancy and child illness at two years. Maternal Child Health J. 2009;13(6):795-805.
101. Hill DJ, Heine RG, Cameron DJ, et al. Role of food protein intolerance in infants with persistent distress attributed to reflux esophagitis. J Pediatr. 2000;136(5):641-647.
102. Kamer B, Chilarski A, Lange A, Piaseczna-Piotrowska A. Gastroesophageal reflux in infants with food allergy. Med Sci Monit. 2000;6(2):348-352.
103. Ravelli, AM, P. Tobanelli, S. Volpi, AG Ugazio. Vomiting and gastric motility in infants with cow's milk allergy. J Pediatr Gastroenterol Nutr. 2001;32(1):59-64.
104. Schach B, Haight M. Colic and food allergy in the breastfed infant: is it possible for an exclusively breastfed infant to suffer from food allergy? J Hum Lact. 2002;18(1):50-52.
106. Greer FR, Sicherer SH, Burks AW, American Academy of Pediatrics Committee on Nutrition, American Academy of Pediatrics Section on Allergy and Immunology. Effects of early nutritional interventions on the development of atopic disease in infants and children: the role of maternal dietary restriction, breastfeeding, timing of introduction of complementary foods, and hydrolyzed formulas. Pediatrics. 2008;121(1):183-191.
108. Oliveira V, Frazao E. THE WIC Program: Background, Trends, and Economic Issues, 2015 Edition, EIB-134. U.S. Department of Agriculture, Economic Research Service; 2015.
109. Oliveira V, Prell M, Cheng X. The Economic Impacts of Breastfeeding: A Focus on USDA's Special Supplemental Nutrition Program for Women, Infants, and Children (WIC), ERR-261. U.S. Department of Agriculture, Economic Research Service; 2019.
110. Garza C, Schanler RJ, Butte NF, Motil KJ. Special properties of human milk. Clin Perinatol. 1987;14(1):11-32.
111. Kennell J, Klaus M, McGrath S, Robertson S, Hinkley C. Continuous emotional support during labor in a U.S. hospital: a randomized controlled trial. JAMA. 1991;265(17):2197-2201.
112. Lucas A, Morley R, Cole TJ, Lister G, Leeson-Payne C. Breast milk and subsequent intelligence quotient in children born preterm. Lancet. 1992;339(8788):261-264.
113. Deoni SCL, Dean DC III, Piryatinsky I, et al. Breastfeeding and early white matter development: a cross-sectional study. Neurolimage. 2013;82:77-86.
114. Baker RD, Greer FR, ; American Academy of Pediatrics, Committee on Nutrition. Diagnosis and prevention of iron deficiency and iron-deficiency anemia in infants and young children (0–3 years of age). Pediatrics. 2010;126(5):1040–1050.
115. Schanler RJ, Feldman-Winter L, Landers S, Noble L, Szucs KA, Viehmann, on behalf of the AAP Section on Breastfeeding. Concerns with early universal iron supplementation of breastfeeding infants. Pediatrics. 2011;127(4):e1097.
116. Jalalabadi F, Doval AF, Neese V, Andrews E, Spiegel A. Breast implant utilization trends in USA versus Europe and the impact of BIA-ALCL publications. Plastic Reconstr Surg. 2021;9(3):e3449.
117. Cai C, Granger M, Eck P, Friel J. Effect of daily iron supplementation in healthy exclusively breastfed infants: a systematic review with meta-analysis. Breastfeed Med. 2017;12(10):597-603.
119. American Pregnancy Association. Breastfeeding Overview. Available at https://americanpregnancy.org/breastfeeding/breastfeeding-overview. Last accessed April 26, 2025.
120. Humenick SS. The clinical significance of breastmilk maturation rates. Birth. 1987;14(4):174-181.
121. Humenick SS, Mederios D, Wreschner TB, Walton MB, Hill PD. The Maturation Index of Colostrum and Milk (MICAM): a measurement of breast milk maturation. J Nurs Meas. 1994;2(2):169-186.
122. Tauber KA. Human Milk and Lactation. Available at https://emedicine.medscape.com/article/1835675-overview. Last accessed April 26, 2025.
123. Neville MC, Morton J, Umemura S. Lactogenesis: the transition from pregnancy to lactation. Pediatr Clin North Am. 2001;48(1):35-52.
124. Correa Marquez RR. Hypopituitarism (Panhypopituitarism). Available at https://emedicine.medscape.com/article/127650-overview. Last accessed April 26, 2026.
125. Pang WW, Hartmann PE. Initiation of human lactation: secretory differentiation and secretory activation. J Mammary Gland Biol Neoplasia. 2007;12(4):211-221.
126. Ostrom KM. A review of the hormone prolactin during lactation. Prog Food Nutr Sci. 1990;14(1):1-43.
127. U.S. Food and Drug Administration. Depo-Provera: Label Information. Available at https://www.accessdata.fda.gov/drugsatfda_docs/label/2010/020246s036lbl.pdf. Last accessed April 26, 2025.
128. The Academy of Breastfeeding Medicine. ABM Clinical Protocol #13: Contraception During Breastfeeding, Revised 2015. Available at https://abm.memberclicks.net/assets/DOCUMENTS/PROTOCOLS/13-contraception-and-breastfeeding-protocol-english.pdf. Last accessed April 26, 2025.
129. World Health Organization. Infant and Young Child Feeding: Model Chapter for Textbooks for Medical Students and Allied Health Professionals. Geneva; 2009.
130. MedlinePlus. Overcoming Breastfeeding Problems. Available at https://medlineplus.gov/ency/article/002452.htm. Last accessed April 26, 2025.
131. U.S. Department of Health and Human Services Office on Women's Health. Breastfeeding. Available at https://www.womenshealth.gov/breastfeeding. Last accessed April 26, 2025.
132. Pittard WB 3rd, Geddes KM, Pepkowitz SH, Carr R. The immunologic composition of neonatal milk: cellular components. Clin Immunol Immunopathol. 1988;46(2):294-298.
133. U.S. Department of Health and Human Services Office on Women's Health. Learning to Breastfeed. Available at https://www.womenshealth.gov/breastfeeding/learning-breastfeed. Last accessed April 26, 2025.
134. Furman L, Schanler RJ. Breastfeeding. In: Gleason CA, Juul SE (eds). Avery's Diseases of the Newborn. 10th ed. Philadelphia, PA: Elsevier; 2018.
135. Neifert MR, Seacat JM, Jobe WE. Lactation failure due to insufficient glandular development of the breast. Pediatrics. 1985;76(5):823-828.
136. Kumar Y, Chahal A, Garg M, Bhatia A, Mahajan NC, Ganju A. Delayed involution of lactation presenting as a non-resolving breast mass: a case report. J Med Case Rep. 2008;2:327.
138. Shaikh U, Scott BJ. Extent, accuracy, and credibility of breastfeeding information on the Internet. J Hum Lact. 2005;21(2):175-183.
140. National Cancer Institute. SEER Stat Fact Sheets: Female Breast Cancer. Available at https://seer.cancer.gov/statfacts/html/breast.html. Last accessed April 26, 2025.
141. National Cancer Institute. Breast Cancer Treatment During Pregnancy (PDQ). Available at https://www.cancer.gov/types/breast/hp/pregnancy-breast-treatment-pdq. Last accessed April 26, 2025.
142. Olsen CG, Gordon RE Jr. Breast disorders in nursing mothers. Am Fam Physician. 1990;41(5):1509-1516.
144. Andersson TM, Johansson AL, Hsieh CC, Cnattingius S, Lambe M. Increasing incidence of pregnancy-associated breast cancer in Sweden. Obstet Gynecol. 2009;114(3):568-572.
145. Michalopoulos K. The effects of breast augmentation surgery on future ability to lactate. Breast J. 2007;13(1):62-67.
146. American Academy of Family Physicians. Breastfeeding, Family Physicians Supporting (Position Paper). Available at https://www.aafp.org/about/policies/all/breastfeeding-position-paper.html. Last accessed April 26, 2025.
148. Bodley V, Powers D. Patient with insufficient glandular tissue experiences milk supply increase attributed to progesterone treatment for luteal phase effect. J Hum Lact. 1999;15(4):339-343.
149. Thorley V. Breast hypoplasia and breastfeeding: a case history. Breastfeed Rev. 2005;13(2):13-16.
150. Brown SL, Todd JF, Cope JU, Sachs HC. Breast implant surveillance reports to the U.S. Food and Drug Administration: maternal-child health problems. J Long Term Eff Med Implants. 2006;16(4):281-290.
151. Semple JL. Breast-feeding and silicone implants. Plast Reconstr Surg. 2007;120(7 Suppl 1):123S-128S.
152. Souto GC, Giugliani ERJ, Giugliani C, Schneider MA. The impact of breast reduction surgery on breastfeeding performance. J Hum Lact. 2003;19(1):43-49.
153. Witte PM, van der Lei B, van der Biezen JJ, Spronk CA. Successful breastfeeding after reduction mammaplasty. Ned Tijdschr Geneeskd. 2004;148(26):1291-1293.
154. Zimpelmann A, Kaufmann M. Breastfeeding nursing after breast surgery. Zentralbl Gynakol. 2002;124(11):525-528.
155. Kakagia D, Tripsiannis G, Tsoutsos D. Breastfeeding after reduction mammaplasty: a comparison of 3 techniques. Ann Plast Surg. 2005;55(4):343-345.
156. Centers for Disease Control and Prevention. Contraindications to Breastfeeding. Available at https://www.cdc.gov/breastfeeding-special-circumstances/hcp/contraindications. Last accessed April 26, 2025.
157. Zimmerman DR, Bernstein WR. Standing feeding orders in a well-baby nursery: "water, water everywhere..." J Hum Lact. 1996;12(3):189-192.
158. Langer A, Campero L, Garcia C, Reynoso S. Effects of psychosocial support during labor and childbirth on breastfeeding, medical interventions, and mothers' wellbeing in a Mexican public hospital: a randomized clinical trial. Br J Obstet Gynaecol. 1998;105(10):1056-1063.
159. Kennell JH, Klaus MH. Bonding: recent observations that alter perinatal care. Pediatr Rev. 1998;19(1):4-12.
160. Reece-Stremtan S, Campos M, Kokajko L, Academy of Breastfeeding Medicine. ABM clinical protocol #15: analgesia and anesthesia for the breastfeeding mother, revised 2017. Breastfeed Med. 2017;12(9):1-7.
161. DONA International. Available at https://www.dona.org. Last accessed April 26, 2025.
162. Gilliland AL. Beyond holding hands: the modern role of the professional doula. J Obstet Gynecol Neonatal Nurs. 2002;31(6):762-769.
163. Klaus MH, Kennell JH, Klaus PH. Mothering the Mother: How a Doula Can Help You Have a Shorter, Easier, and Healthier Birth. Reading, MA: Addison-Wesley; 1993.
164. Gordon NP, Walton D, McAdam E, Derman J, Gallitero G, Garrett L. Effects of providing hospital-based doulas in health maintenance organization hospitals. Obstet Gynecol. 1999;93(3):422-426.
165. Atherton MJ, Feeg VD, El-Adham AF. Race, ethnicity, and insurance as determinants of epidural use: analysis of a national sample survey. Nurs Econ. 2004;22(1):6-13.
166. American College of Obstetrics and Gynecology. Practice Bulletin Number 209: Obstetric Analgesia and Anesthesia. Available at https://www.acog.org/clinical/clinical-guidance/practice-bulletin/articles/2019/03/obstetric-analgesia-and-anesthesia. Last accessed April 6, 2022.
167. Howie WO, McMullen PC. Breastfeeding problems following anesthetic administration. J Perinat Educ. 2006;15(3):50-57.
168. Anim-Somuah M, Smyth RM, Jones L. Epidural versus non-epidural or no analgesia in labour. Cochrane Database Syst Rev. 2011;7(12):CD000331.
169. LexiDrug. Available at https://online.lexi.com. Last accessed April 26, 2025.
170. Faculty of Family Planning and Reproductive Health Care. Contraceptive choices for breastfeeding women. J Fam Plann Reprod Health Care. 2004;30(3):181-189.
171. Sinusas K, Gagliardi A. Initial management of breastfeeding. Am Fam Physician. 2001;64(6):981-988.
172. Chang ZM, Heaman MI. Epidural analgesia during labor and delivery: effects on the initiation and continuation of effective breastfeeding. J Hum Lact. 2005;21(3):305-314.
173. Torvaldsen S, Roberts CL, Simpson JM, Thompson JF, Ellwood DA. Intrapartum epidural analgesia and breastfeeding: a prospective cohort study. Int Breastfeeding J. 2006;1:24.
174. Wilson MJ, MacArthur C, Cooper GM, et al. Epidural analgesia and breastfeeding: a randomized controlled trial of epidural techniques with and without fentanyl and a non-epidural comparison group. Anaesthesia. 2009;65(2):145-153.
175. Jordan S, Emery S, Watkins A, Evans JD, Storey M, Morgan G. Associations of drugs routinely given in labour with breastfeeding at 48 hours: analysis of the Cardiff Births Survey. BJOG. 2009;116(12):1622-1632.
176. Baumgarder DJ, Muehl P, Fischer M, Pribbenow B. Effect of labor epidural anesthesia on breast-feeding of healthy full-term newborns delivered vaginally. J Am Board Fam Pract. 2003;16:7-13.
177. Leighton BL, Halpern SH. The effects of epidural analgesia on labor, maternal, and neonatal outcomes: a systematic review. Am J Obstet Gynecol. 2002;186(5 Suppl nature):S69-S77.
178. Devroe S, De Coster J, Van de Velde M. Breastfeeding and epidural analgesia during labor. Curr Opin Anaesthesiol. 2009;22(3):327-329.
179. Jordan S, Emery S, Bradshaw C, Watkins A, Friswell W. The impact of intrapartum analgesia on infant feeding. BJOG. 2005;112:927-934.
180. Wiklund I, Norman M, Uvnäs-Moberg K, Ransjö-Arvidson A-B, Andolf E. Epidural analgesia: breast-feeding success and related factors. Midwifery. 2009;25(2):e31-e38.
181. French CA, Cong X, Chung KS. Labor epidural analgesia and breastfeeding: a systematic review. J Hum Lact. 2016;32(3):507-520.
182. Orbach-Zinger S, Landau R, Davis A, et al. The effect of labor epidural analgesia on breastfeeding outcomes: a prospective observational cohort study in a mixed-parity cohort. Anesth Analg. 2019;129(3):784-791.
183. Alexander JM, Sharma SK, McIntire DD, Leveno KJ. Epidural analgesia lengthens the Friedman active phase of labor. Obstet Gynecol. 2002;100(1):46-50.
184. Lieberman E, Cohen A, Lang J, Frigoletto F, Goetzl L. Maternal intrapartum temperature elevation as a risk factor for cesarean delivery and assisted vaginal delivery. Am J Pub Health. 1999;89(4):506-510.
185. Lieberman E, Lang JM, Frigoletto F Jr, Richardson DK, Ringer SA, Cohen A. Epidural analgesia, intrapartum fever, and neonatal sepsis evaluation. Pediatrics. 1997;99(3):415-419.
186. Zhang J, Yancey MK, Klebanoff MA, Schwarz J, Schweitzer D. Does epidural analgesia prolong labor and increase risk of cesarean delivery? A natural experiment. Am J Obstet Gynecol. 2001;185(1):128-134.
187. Anim-Somuah M, Smyth R, Jones L. Epidural versus non-epidural or no analgesia in labour. Cochrane Database Syst Rev. 2005;(4):CD000331.
188. Lieberman E, Davidson K, Lee-Parritz A, Shearer E. Changes in fetal position during labor and their association with epidural analgesia. Obstet Gynecol. 2005;105(5 Pt 1):974-982.
189. Anim-Somuah M, Smyth R, Cyna AM, Cuthbert A. Epidural versus non-epidural or no analgesia for pain management in labour. Cochrane Database Syst Rev. 2018;5:CD000331.
190. Russell R. Combined spinal epidural analgesia is the preferred technique for labour analgesia. Acta Anaesthesiol Belg. 2002;53(4):331-334.
191. Simmons SW, Taqhizadeh N, Dennis AT, Hughes D, Cyna AM. Combined spinal-epidural versus epidural analgesia in labour.Cochrane Database Syst Rev. 2012;10:CD003401.
192. Hughes D, Simmons SW, Brown J, Cyna AM, Dennis AT. Combined spinal-epidural versus epidural analgesia in labour. Cochrane Database Syst Rev. 2007;(3):CD003401.
193. Aneiros F, Vazquez M, Valiño C, et al. Does epidural versus combined spinal-epidural analgesia prolong labor and increase the risk of instrumental and cesarean delivery in nulliparous women? J Clin Anesth. 2009;21(2):94-97.
194. Moore ER, Anderson GC. Randomized controlled trial of very early mother-infant skin-to-skin contact and breastfeeding status.J Midwifery Womens Health. 2007;52(2):116-125.
195. Vaglio S. Chemical communication and mother-infant recognition. Commun Integr Biol. 2009;2(3):279-281.
196. Vaglio S. Minicozzi P, Bonometti E, Mello G, Chiarelli B. Volatile signals during pregnancy: a possible chemical basis for mother-infant recognition. J Chem Ecol. 2009;35(1):131-139.
197. Righard L, Alade MO. Effect of delivery room routines on success of first breast-feed. Lancet. 1990;336(8723):1105-1107.
198. Moore ER, Bergman N, Anderson GC, Medley N. Early skin-to-skin contact for mothers and their healthy newborn infants. Cochrane Database Syst Rev. 2016;11:CD003519.
199. Bystrova K, Ivanova V, Edhborg M, et al. Early contact versus separation: effects on mother-infant interaction one year later. Birth. 2009;36(2):97-109.
200. Wagner CL. Counseling the Breastfeeding Mother. Available at https://emedicine.medscape.com/article/979458-overview. Last accessed April 26, 2025.
201. Renfrew MJ, Craig D, Dyson L, et al. Breastfeeding promotion for infants in neonatal units: a systematic review and economic analysis. Health Technol Assess. 2009;13(40):1-146.
202. Lauwers J, Swisher A. Counseling the Nursing Mother: A Lactation Consultant's Guide. 5th ed. Sudbury, MA: Jones and Barlett; 2010.
203. Mohrbacher N, Stock J. The Breastfeeding Answer Book. 3rd rev. ed. Chicago, IL: La Leche League International; 2003.
204. Newman J. Breastfeeding problems associated with the early introduction of bottles and pacifiers. J Hum Lact. 1990;6(2):59-63.
206. Walker M, Driscol JW. Sore nipples: the new mother's nemesis. MCN Am J Matern Child Nurs. 1989;14(4):260-265.
207. Napier D. Hands off technique has many benefits for breastfeeding mothers. BMJ. 2001;322(7291):929-930.
208. Fletcher D, Harris H. The implementation of the HOT program at the Royal Women's Hospital. Breastfeed Rev. 2000;8(1):19-23.
209. American Academy of Pediatrics. Ensuring Proper Latch On While Breastfeeding. Available at https://www.healthychildren.org/English/ages-stages/baby/breastfeeding/pages/Ensuring-Proper-Latch-On.aspx. Last accessed April 26, 2025.
210. Mayo Clinic. Slide Show: Breastfeeding Positions. Available at https://www.mayoclinic.org/healthy-lifestyle/infant-and-toddler-health/multimedia/breast-feeding/sls-20076017. Last accessed April 26, 2025.
211. Taylor JS, Kacmar JE, Nothnagle M, Lawrence RA. A systematic review of the literature associating breastfeeding with type 2 diabetes and gestational diabetes. J Am Coll Nutr. 2005;24(5):320-326.
212. Hummel S, Winkler C, Schoen S, et al. Breastfeeding habits in families with type 1 diabetes. Diabet Med. 2007;24(6):671-676.
213. Diabetes in pregnancy. In: Mazze RS, Strock E, Simonson GD, Bergenstal RM. Prevention, Detection and Treatment of Diabetes in Adults. 4th ed. Minneapolis, MN: International Diabetes Center; 2007.
214. Chertok IR, Raz I, Shoham I, Haddad H, Wiznitzer A. Effects of early breastfeeding on neonatal glucose levels of term infants born to women with gestational diabetes. J Hum Nutr Diet. 2009;22(2):166-169.
215. Voeten M, Gerrits GP, Voorhoeve PG. Treatment of neonatal hypoglycaemia: more frequent latching onto the breast versus supplementary feeding with formula: retrospective study of patient files. Ned Tijdschr Geneeskd. 2008;152(31):1732-1736.
216. Cranmer H. Neonatal Hypoglycemia. Available at https://emedicine.medscape.com/article/802334-overview. Last accessed April 26, 2025.
217. Abramowski A, Ward R, Hamdan AH. Neonatal Hypoglycemia. Treasure Island, FL: StatPearls Publishing; 2023.
218. Straussman S, Levitsky LL. Neonatal hypoglycemia. Curr Opin Endocrinol Diabetes Obes. 2010;17(1):20-24.
219. Cornblath M, Hawdon JM, Williams AF, et al. Controversies regarding definition of neonatal hypoglycemia: suggested operational thresholds. Pediatrics. 2000;105(5):1141-1145.
220. Maisels MJ. What's in a name? Physiologic and pathologic jaundice: the conundrum of defining normal bilirubin levels in the newborn. Pediatrics. 2006;118:805-807.
221. Hansen TWR. Neonatal Jaundice. Available at https://emedicine.medscape.com/article/974786-overview. Last accessed April 26, 2025.
222. American Academy of Pediatrics, Subcommittee on Hyperbilirubinemia. Management of hyperbilirubinemia in the newborn infant 35 or more weeks gestation. Pediatrics. 2004;114(1):297-316.
224. Brown LP, Arnold L, Allison D, Klein ME, Jacobsen B. Incidence and pattern of jaundice in healthy breast-fed infants during the first month of life. Nurs Res. 1993;42(2):106-110.
225. Hannon PR, Willis SK, Scrimshaw SC. Persistence of maternal concerns surrounding neonatal jaundice: an exploratory study. Arch Pediatr Adolesc Med. 2001;155(12):1357-1363.
226. Olusanya BO, Slusher TM, Imosemi DO, Emokpae AA. Maternal detection of neonatal jaundice during birth hospitalization using a novel two-color icterometer. PLoS One. 2017;12(8):e0183882.
227. Taylor JA, Stout JW, de Greef L, et al. Use of a smartphone app to assess neonatal jaundice. Pediatrics. 2017;140:3.
228. Maisels MJ, Bhutani VK, Bogen D, Newman TB, Stark AR, Watchko JF. Hyperbilirubinemia in the newborn infant ≥35 weeks' gestation: an update with clarifications. Pediatrics. 2009;124:1193.
229. Muchowski KE. Evaluation and treatment of neonatal hyperbilirubinemia. Am Fam Physician. 2014;89(11):873-878.
230. Taber's Online. Available at https://www.tabers.com/tabersonline. Last accessed April 26, 2025.
231. Auerbach KG, Gartner LM. Breastfeeding and human milk: their association with jaundice in the neonate. Clin Perinatol. 1987;14(1):89-107.
232. Watchko JF. Identification of neonates at risk for hazardous hyperbilirubinemia: emerging clinical insights. Pediatr Clin North Am. 2009;56(3):671-687.
233. Thilo E. Jaundice. In: Thureen PJ, Deacon J, Hernandez JA, Hall D (eds). Assessment and Care of the Well Newborn. 2nd ed. Philadelphia, PA: W. B. Saunders; 2004.
234. U.S. Preventive Services Task Force. Screening of Infants for Hyperbilirubinemia to Prevent Chronic Bilirubin Encephalopathy: U.S. Preventive Services Task Force Recommendation Statement. Rockville, MD: Agency for Healthcare Research and Quality; 2009.
235. Hansen TW. Kernicterus in term and near-term infants—the specter walks again. Acta Paediatr. 2000;89(10):1155-1157.
236. Dunn P, Bhutani V, Weiner S, Ludomirski A. Care of the neonate with erythroblastosis fetalis. J Obstet Gynecol Neonatal Nurs. 1988;17(6):382-386.
237. de Carvalho M, Hall M, Harvey D. Effects of water supplementation on physiologic jaundice in breast-fed babies. Arch Dis Child. 1981;56(7):568-569.
238. MacDorman MF, Mathews TJ. Behind international rankings of infant mortality: how the United States compares with Europe. NCHS Data Brief. 2009;(23):1-8.
239. March of Dimes. U.S. Preterm Birthrate on the Rise for Second Year in a Row. Available at https://www.marchofdimes.org/news/u-s-preterm-birth-rate-on-the-rise-for-second-year-in-a-row.aspx. Last accessed April 26, 2025.
240. Healthy Moms. Strong Babies. 2024 March of Dimes Report Card. Report Card for United States. Available at https://www.marchofdimes.org/report-card. Last accessed April 26, 2025.
241. Buckley KM, Charles GE. Benefits and challenges of transitioning preterm infants to at-breast feedings. Int Breastfeeding J. 2006;1:13.
242. Dougherty D, Luther M. Birth to breast: a feeding care map for the NICU: helping the extremely low birth weight infant navigate the course. Neonatal Netw. 2008;27(6):371-377.
243. Callen J, Pinelli J. A review of the literature examining the benefits and challenges, incidence and duration, and barriers to breastfeeding in preterm infants. Adv Neonatal Care. 2005;5(2):72-88.
244. Schurr P, Perkins EM. The relationship between feeding and necrotizing enterocolitis in very low birth weight infants. Neonatal Netw. 2008;27(6):397-407.
245. Schanler RJ, Hurst NM. Human milk for the hospitalized preterm infant. Semin Perinatol. 1994;18(6):476-484.
246. Sisk PM, Lovelady Ca, Dillard RG, Gruber KJ. Lactation counseling for mothers of very low birth weight infants: effect on maternal anxiety and infant intake of human milk. Pediatrics. 2006;117(1):e67-e75.
247. Hill PD, Aldag JC, Chatterton RT, Zinaman M. Comparison of milk output between mothers of preterm and term infants: the first 6 weeks after birth. J Hum Lact. 2005;21(1):22-30.
248. Becker GE, Smith HA, Cooney F. Methods of milk expression for lactating women. Cochrane Database Syst Rev. 2015;2:CD006170.
249. Morton J, Hall JY, Wong RJ, Thairu L, Benitz WE, Rhine WD. Combining hand techniques with electric pumping increases milk production in mothers of preterm infants. J Perinatol. 2009;29(11):757-764.
250. Linden DW, Paroli ET, Doron MW. Preemies: The Essential Guide for Parents of Premature Babies. 2nd ed. New York, NY: Pocket; 2010.
251. Drenckpohl D, Bowers L, Cooper H. Use of the six sigma methodology to reduce incidence of breast milk administration errors in the NICU. Neonatal Netw. 2007;26(3):161-166.
252. Pinelli J, Symington A. Non-nutritive sucking for promoting physiologic stability and nutrition in preterm infants. Cochrane Database Syst Rev. 2005;19(4):CD001071.
256. Lee TY, Lee TT, Kuo SC. The experiences of mothers in breastfeeding their very low birth weight infants. J Adv Nurs. 2009;65(12):2523-2531.
257. Maastrup R, Hansen BM, Kronborg H, et al. Breastfeeding progression in preterm infants is influenced by factors in infants, mothers and clinical practice: the results of a national cohort study with high breastfeeding initiation rates. PLoS One. 2014;9(9):e108208.
258. Groh-Wargo S, Sapsford A. Enteral nutrition support of the preterm infant in the neonatal intensive care unit. Nutr Clin Pract. 2009;24(3):363-376.
259. Ben XM. Nutritional management of newborn infants: practical guidelines. World J Gastroenterol. 2008;14(40):6133-6139.
260. Senterre T. Practice of enteral nutrition in very low birth weight and extremely low birth weight infants. World Rev Nutr Diet. 2014;110:201-214.
261. Dowling DA, Meier PP, DiFiore JM, Blatz M, Martin RJ. Cup-feeding for preterm infants: mechanics and safety. J Hum Lact. 2002;18(1):13-20.
262. Penny F, Judge M, Brownell E, McGrath JM. Cup feeding as a supplemental, alternative feeding method for preterm breastfed infants: an integrative review. Matern Child Health J. 2018;22(11):1568-1579.
263. Meier P, Anderson GC. Responses of small preterm infants to bottle- and breast-feeding. MCN Am J Matern Child Nurs. 1987;12(2):97-105.
264. U.S. Food and Drug Administration. Medical Devices: Breast Pumps. Available at https://www.fda.gov/medical-devices/consumer-products/breast-pumps. Last accessed April 26, 2025.
265. U.S. Food and Drug Administration. News Release: FDA Warns Consumers Against Using Mommy's Bliss Nipple Cream: Product Can Be Harmful to Nursing Infants [archive]. Available at https://www.reuters.com/article/business/healthcare-pharmaceuticals/fda-says-nipple-cream-could-harm-infants-idUSN23302729/ . Last accessed April 26, 2025.
267. McKechnie AC, Eglash A. Nipple shields: a review of the literature. Breastfeed Med. 2010;5(6):309-314.
268. Auerbach KG. The effect of nipple shields on maternal milk volume. J Obstet Gynecol Neonatal Nurs. 1990;19(5):419-427.
269. Woolridge MW, Baum JD, Drewett RF. Effect of a traditional and of a new nipple shield on sucking patterns and milk flow. Early Human Dev. 1980;4(4):357-364.
270. Llorente AM, Jensen CL, Voigt RG, Fraley JK, Berretta MC, Heird WC. Effect of maternal docosahexaenoic acid supplementation on postpartum depression and information processing. Am J Obstet Gynecol. 2003;188(5):1348-1353.
271. American College of Obstetricians and Gynecologists Committee on Obstetric Practice. Committee opinion number 757: screening for perinatal depression. Obstet Gynecol. 2018;132(5):e208-e212.
272. Righetti-Veltema M, Conne-Perréard E, Bousquet A, Manzano J. Risk factors and predictive signs of postpartum depression. J Affect Disord. 1998;49(3):167-180.
273. Kohnle D. Health Tip: Symptoms of Postpartum Depression. Available at https://abcnews.go.com/Health/Healthday/health-tip-symptoms-postpartum-depression/story?id=9042844. Last accessed April 26, 2025.
274. Coleman BL, Gutmanis I, Larsen LL, Leffley AC, McKillop JM, Rietdyk AE. Introduction of solid foods: do mothers follow recommendations? Can J Diet Pract Res. 2009;70(3):135-140.
275. Anderson AS, Guthrie CA, Alder EM, Forsyth S, Howie PW, Williams FL. Rattling the plate—reasons and rationales for early weaning. Health Educ Research. 2001;16(4):471-479.
276. Sachs HC. American Academy of Pediatrics Committee on Drugs. The transfer of drugs and therapeutics into human breast milk: an update on selected topics. Pediatrics. 2013;132(3):e796-e809.
277. Briggs GG, Freeman RK, Yaffe SJ. Drugs in Pregnancy and Lactation: A Reference Guide to Fetal and Neonatal Risk. 9th ed. Baltimore, MD: Lippincott Williams and Wilkins; 2011.
279. Academy of Breastfeeding Medicine Protocol Committee. Protocol 9: use of galactogogues in initiating or augmenting maternal milk supply, second revision 2018. Breastfeed Med. 2018;13(5):307-314.
280. Dalvi SS, Nadkarni PM, Gupta KC. Effect of Asparagus racemosus (Shatavari) on gastric emptying time in normal healthy volunteers.J Postgrad Med. 1990;36(2):91-94.
281. Madden JM, Soumerai SB, Lieu TA, et al. Effects of a law against early postpartum discharge on newborn follow-up, adverse events, and HMO expenditures. N Engl J Med. 2002;347(25):2031-2038.
282. Laugesen MJ, Paul RR, Luft HS, Aubry W, Ganiats TG. A comparative analysis of mandated benefit laws, 1949–2002. Health Serv Res. 2006;41(3 Pt 2):1081-1103.
283. International Lactation Consultant Association. Available at https://www.ilca.org. Last accessed April 26, 2025.
284. La Leche League International. Feeding the New Mother. Available at https://www.llli.org/feeding-the-new-mother/. Last accessed April 26, 2025.
285. American Academy of Pediatrics Task Force on Sudden Infant Death Syndrome. The changing concept of sudden infant death syndrome: diagnostic coding shifts, controversies regarding the sleeping environment, and new variables to consider in reducing risk. Pediatrics. 2005;116(5):1245-1255.
286. Mayo Clinic. Colic: Definition. Available at https://www.mayoclinic.org/diseases-conditions/colic/symptoms-causes/syc-20371074. Last accessed April 26, 2025.
287. Wagner CL, Greer FR. Prevention of rickets and vitamin D deficiency in infants, children, and adolescents. Pediatrics. 2008;122(5).
289. HealthCare.gov. Breastfeeding Benefits. Available at https://www.healthcare.gov/coverage/breast-feeding-benefits. Last accessed April 26, 2025.
290. World Health Organization. Exclusive Breastfeeding. Available at https://www.who.int/tools/elena/interventions/exclusive-breastfeeding. Last accessed April 25, 2025.
291. Meek JY, Noble L, Section on Breastfeeding. Policy statement: breastfeeding and the use of human milk. Pediatrics. 2022;150(1):e2022057988.
292. Alexis AF, Cook-Bolden F, Haley TT, Taylor SC. Racial/ethnic variations in skin barrier: Implications for skin care recommendations in skin of color. Journal of Drugs in Dermatology. 2021;20(9):932-938.
293. Kohn JR, Levison J, Swamy GK, AAP Committee on Pediatric AIDS. Infant feeding for persons living with and at risk for HIV in the United States: Clinical report. Pediatrics. 2024;153(6):e2024066843.
294. Geddes DT, Kent JC, Mitoulas LR, Hartmann PE. Tongue movement and intra-oral vacuum in breastfeeding infants. Early Human Development. 2008;84(7):471-477.
295. Meier PP, Brown LP, Hurst NM, et al. Nipple shields for preterm infants: effect on milk transfer and duration of breastfeeding. Journal of Human Lactation. 2000;16(2):106-114.
296. Postpartum Support International. About Perinatal Mental Health. Available at https://www.postpartum.net/perinatal-mental-health. Last accessed April 25, 2025.
297. Postpartum Support International. Help for Dads. Available at https://www.postpartum.net/get-help/help-for-dads/. Last accessed April 25, 2025.
1. Vanguri S, Rogers-McQuade H, Sriraman NK, Academy of Breastfeeding Medicine. ABM clinical protocol #14: breastfeeding-friendly physician's office: optimizing care for infants and children, revised 2021. Breastfeed Med. 2021;16(3):175-184. Available at https://www.bfmed.org/assets/DOCUMENTS/PROTOCOLS/Protocol%20%2314%20-%20English%20Translation.pdf. Last accessed April 28, 2025.
2. Holmes AV, Yerdon McLeod A, Bunik M. Peripartum breastfeeding management for the healthy mother and infant at term, revision 2013. Breastfeed Med. 2013;8(6):469-473. Available at https://www.bfmed.org/assets/DOCUMENTS/PROTOCOLS/5-peripartum-bf-management-protocol-english.pdf. Last accessed April 28, 2025.
3. Boies EG, Vauche YE, Academy of Breastfeeding Medicine. ABM clinical protocol #10: breastfeeding the late preterm (34–36 6/7 weeks of gestation) and early term infants (37–38 6/7 weeks of gestation), second revision 2016. Breastfeed Med. 2016;11(10):151-156. Available at https://abm.memberclicks.net/assets/DOCUMENTS/PROTOCOLS/10-breastfeeding-the-late-pre-term-infant-protocol-english.pdf. Last accessed April 28, 2025.
4. Academy of Breastfeeding Medicine Protocol Committee. ABM Clinical Protocol #9: Use of galactogoguesin initiating or augmenting maternal milk production, Second Revision 2018. Breastfeed Med. 2018;13(5):307-314. Available at https://www.bfmed.org/assets/DOCUMENTS/PROTOCOLS/9-galactogogues-protocol-english.pdf. Last accessed April 28, 2025.
Mention of commercial products does not indicate endorsement.