This course provides information about snakes themselves, about the "bite," and how the venom affects organ systems and bodies as a whole. It also familiarizes the learner with the pit viper antivenoms available in the United States. From the field through follow-up after discharge from the hospital, step-by-step procedures encourage application of the standard of assessment and treatment, including separate procedures for patients arriving with no symptoms versus those who arrive with symptoms. This course is designed for use immediately at the bedside as well as for continuing education purposes.
This course is designed for physicians, nurses, and allied health professionals involved in the identification and care of patients who have been bitten by a snake.
This course is designed to provide emergency and intensive care nursing and physician staff with an accurate, concise, and practical clinical guide to the evaluation and management of snakebite victims who present with North American pit viper envenomation.
Upon completion of this course, you should be able to:
- Identify snakes that may be venomous and the characteristics of snakebite.
- Outline the local and systemic effects of pit viper envenomation.
- Describe the characteristics of available antivenoms.
- Evaluate general approaches to the assessment of patients with snakebite.
- Discuss the role of blood products and urinary catheterization in patients being treated for snakebite.
- Discuss the prehospital management of the patient who has suspected or known snakebite.
- Devise a strategy for the management of asymptomatic patients with suspected or known snakebite.
- Describe the approach to management of symptomatic patients with suspected or known snakebite.
- Outline the reconstitution, dosages, and application of antivenom, including management of potential untoward effects.
- Analyze the appropriate follow-up of patients who were treated for snakebite, including indications to re-treat.
Jon E. Soskis, BSN, RN, is a retired expert in pit viper snakebite assessment and treatment. He earned his BS in Biology from Stetson University in DeLand, Florida, and his BSN from Florida State University in Tallahassee, Florida, while completing his EMT training at Tallahassee Community College. Following a 30-year practice in emergency nursing at Tallahassee Memorial HealthCare, including numerous in-hospital snakebite consultations, he remains available to the local medical and nursing community for consultation during active treatment of pit viper envenomation, and has trained Florida State University College of Medicine surgical and internal medicine residents as well as Tallahassee Memorial HealthCare Family Practice residents in the subject. He has likewise presented at hospitals in Florida and North Carolina.
Mr. Soskis wishes to recognize the years-long contributions to this work by Rick Dart, MD, PhD, Director of Rocky Mountain Poison and Drug Center; Jeffrey Bernstein, MD, Medical Director of the South Florida Poison Information Center, Miami; and Ed Hall, MD, South Georgia Surgical Associates.
Contributing faculty, Jon E. Soskis, BSN, RN, has disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
John M. Leonard, MD
Mary Franks, MSN, APRN, FNP-C
The division planners have disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
Sarah Campbell
The Director of Development and Academic Affairs has disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
The purpose of NetCE is to provide challenging curricula to assist healthcare professionals to raise their levels of expertise while fulfilling their continuing education requirements, thereby improving the quality of healthcare.
Our contributing faculty members have taken care to ensure that the information and recommendations are accurate and compatible with the standards generally accepted at the time of publication. The publisher disclaims any liability, loss or damage incurred as a consequence, directly or indirectly, of the use and application of any of the contents. Participants are cautioned about the potential risk of using limited knowledge when integrating new techniques into practice.
It is the policy of NetCE not to accept commercial support. Furthermore, commercial interests are prohibited from distributing or providing access to this activity to learners.
Supported browsers for Windows include Microsoft Internet Explorer 9.0 and up, Mozilla Firefox 3.0 and up, Opera 9.0 and up, and Google Chrome. Supported browsers for Macintosh include Safari, Mozilla Firefox 3.0 and up, Opera 9.0 and up, and Google Chrome. Other operating systems and browsers that include complete implementations of ECMAScript edition 3 and CSS 2.0 may work, but are not supported. Supported browsers must utilize the TLS encryption protocol v1.1 or v1.2 in order to connect to pages that require a secured HTTPS connection. TLS v1.0 is not supported.
The role of implicit biases on healthcare outcomes has become a concern, as there is some evidence that implicit biases contribute to health disparities, professionals' attitudes toward and interactions with patients, quality of care, diagnoses, and treatment decisions. This may produce differences in help-seeking, diagnoses, and ultimately treatments and interventions. Implicit biases may also unwittingly produce professional behaviors, attitudes, and interactions that reduce patients' trust and comfort with their provider, leading to earlier termination of visits and/or reduced adherence and follow-up. Disadvantaged groups are marginalized in the healthcare system and vulnerable on multiple levels; health professionals' implicit biases can further exacerbate these existing disadvantages.
Interventions or strategies designed to reduce implicit bias may be categorized as change-based or control-based. Change-based interventions focus on reducing or changing cognitive associations underlying implicit biases. These interventions might include challenging stereotypes. Conversely, control-based interventions involve reducing the effects of the implicit bias on the individual's behaviors. These strategies include increasing awareness of biased thoughts and responses. The two types of interventions are not mutually exclusive and may be used synergistically.
#94112: Pit Viper Snakebite Assessment and Treatment
Many people have had rewarding experiences with snakes, while others have been deeply terrified. Common to both types of experiences seems to be that each is forever remembered, the latter perhaps better than the former. In my experience with treatment of snakebite victims and with being told many snake stories, the intensity of recollections and the ease with which they resurface seem to suggest that our brains are hypervigilant when it comes to this subject. This hypervigilance may help to avoid an impending snakebite; however, once bitten, it can also incite such fear that the "fight or flight" physiologic response promotes local spread and facilitates circulation of venom more quickly to other regions of the body. This heightened sensitivity and excitability may also affect first responders and caregivers, occasionally leading to excessively aggressive treatment of a patient.
The general population's underlying fear of snakes, perhaps including that held by medical staff, adds to the challenge of treating the patient who has been bitten. An important goal of this course is to put snakebite in perspective for the practitioner, allowing her or him to share that perspective with the patient so everyone involved can come to realize that, with treatment, snakebite is manageable and rarely fatal. Like anything that is less familiar, when one becomes educated about snakebite and has opportunities to practice what has been learned, confidence is gained. That has special significance with snakebite, because fear or lack of knowledge will likely be recognized by the patient and can affect his or her vital signs, course of treatment, and outcome. Controlling the emotions of everyone involved is important to the management of the snakebite victim for physiologic as well as human reasons. The perspective gained by knowing the important principles of snakebite assessment and treatment will allow healthcare professionals to contribute to a patient's healing by being a calming influence and a source of explanation and reassurance.
Although several thousand snakebites are reported in the United States each year, for any given practitioner there is typically only a rare opportunity to see and learn from a serious snakebite case. Most physicians and nurses have never cared for a snakebite victim. This lack of exposure to snakebite is a challenging circumstance when it comes to assuring an optimal outcome for these patients. Fortunately, the availability of consultants can mitigate the difficulty of having to immediately treat a complex poisoning for which one has had little or no practice and that requires considerable judgment. Consultants understand the limited opportunities that staff have to practice, as well as the deceptive complexity and risks inherent in treating a snakebite victim.
The smoothest hospital stays and best management outcomes result when a small (two- to three-member) physician group admits all snakebite patients to one intensive care unit (ICU). This limited number of physicians and nurses allows more practice per medical team member, thus providing patients with the experienced critical care that snakebite demands. Unfortunately, limiting admissions to a small group of the same physicians may not be practical, and while experienced consultants and toxicologists are typically available via telephone, the importance of clinical preparation, continuing education as to principles of care, and access to a manual of best practice management are fundamental to the safety of these patients.
This course will provide a brief primer on snakes, how to recognize the venomous variety, the mechanism and impact of the "bite," and how the venom affects organ systems and the body as a whole. Clinical management topics include a review of the indications and use of CroFab and Anavip; a look at the phospholipase A2 (sPLA2) inhibitor varespladib (antivenom), now in clinical trials; care of the bite victim from the field through follow-up in the office after discharge from the hospital; and step-by-step application of best practice principles for assessment and treatment, including separate procedures for patients arriving with no symptoms versus those who arrive with symptoms.
In snakebite assessment and treatment, getting "behind the curve" can occur quickly, increasing the risk of local tissue damage, bleeding complications, and vital organ malfunction, thereby putting the patient's life in jeopardy. Where possible, it makes good sense to include on the treatment team a "snakebite specialist" who is active throughout the hospital stay. (This is an unofficial designation, as there is no specific certification for snakebite.) The duty of the specialist is to monitor vital signs and the pace of local injury and swelling, while tracking serial laboratory values so as to provide timely, informed, consistent care that keeps the patient safe. This includes communicating with and teaching medical staff who are treating a patient in the emergency department or ICU and who may be unfamiliar with snakebite. The specialist may be a toxicologist who by training understands the critical need to very closely monitor the patient and serial laboratory values, or more likely may be an informed nurse who, if not already working in the ICU, has a unit manager who understands patient-centered care and the importance of allowing a visit to the ICU as necessary to assure consistency.
The point of having a specialist on the team is to provide continuity across the multiple physicians and nurses who come and go during the patient's stay. The specialist assures adherence to a standard of care that includes early recognition of clinical deterioration and timely administration of antivenom. If the specialist is a nurse, the hospital administration, physician group, and nurse should have a formal orphan agreement that describes their relationship so the imperative consistency is always present, and the hospital administration should ensure that as turnover occurs the resulting vacancy is filled as quickly as possible. The position of a specialist is particularly important if multiple physician groups admit such patients, as the risk of undertreating dramatically increases under that circumstance [1]. Consultation and the availability of the specialist who provides consistency count the most in safely assessing and treating snakebite victims.
The North American pit vipers include [9,13,15,18,21,47]:
Eastern diamondback rattlesnake(Crotalus adamanteus)
Western diamondback rattlesnake(Crotalus atrox)
Timber rattlesnake (Crotalus horridus)
Canebrake rattlesnake (Crotalus horridus atricaudatus)
Prairie rattlesnake(Crotalus viridis viridis)
Great Basin rattlesnake(Crotalus viridis lutosus)
Southern Pacific rattlesnake(Crotalus viridis helleri)
Northern Pacific rattlesnake(Crotalus viridis oreganus)
Mojave rattlesnake (Crotalus scutulatus)
Sidewinder rattlesnake (Crotalus cerastes)
Red diamond rattlesnake(Crotalus ruber ruber)
Northern black-tailed rattlesnake(Crotalus molossus)
Pygmy rattlesnake (Sistrurus miliarius)
Eastern massasauga (Sistrurus catenatus catenatus)
Western massasauga (Sistrurus catenatus tergemin)
Cottonmouth or water moccasin(Agkistrodon piscivorus)
Copperhead (Agkistrodon contortrix)
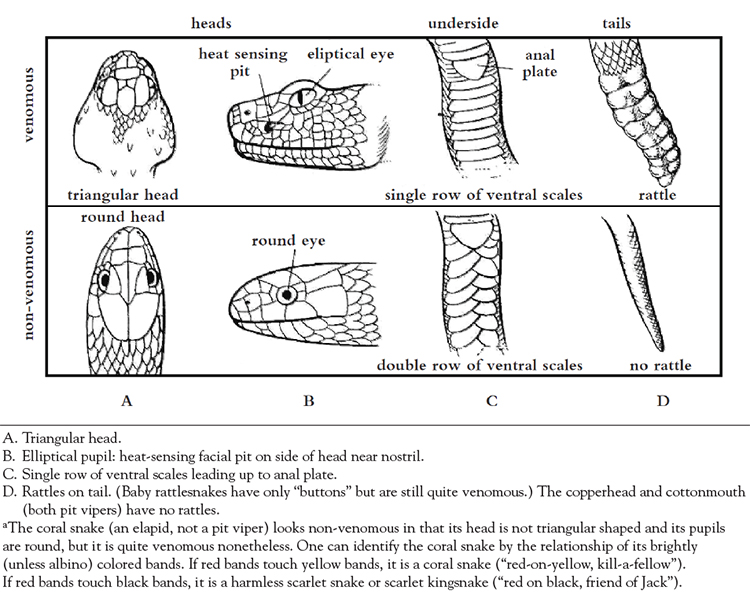
The rattlesnakes, which include the western massasauga and eastern massasauga, have rattles, while the cottonmouth and the copperhead do not. Pit vipers have relatively heavy bodies, vertically elliptical pupils, movable maxillary fangs, and "necks" that are noticeably narrower than their triangular shaped head (Figure 1). There is a single row of scales behind the anal plate as opposed to a double row seen in non-venomous snakes [27].
Pit vipers have poor vision and lack the heart anatomy (three chambers with a pulmonary shunt resulting from an incomplete ventricular septum) and function as well as lung capacity to execute a prolonged chase to gain a meal; however, making up for these limitations are a keen sense of smell and a highly complex and effective venom that is delivered with great precision[21]. This precision is in part due to the ability to sense infrared light emitted by warm-blooded prey. Through this adaptation the pit viper is able to measure extremely small differences in temperatures (as little as 0.003° C), thus allowing the detection of prey as well as recognition of its movement, even in total darkness. This ability to sense infrared light is used to guide a strike over the last few inches (up to 14 inches), and along with an advanced venom delivery mechanism makes successful injection of the venom very likely [21]. External evidence of the sensing mechanism is the easily seen (and namesake) "pit" that is located between the snake's eye and nostril on either side of the head.
Supporting all snakes' ability to gain a meal is a forked tongue that functions to sense volatile chemical odors and allows the snake to track prey (and potential mates) by differentiating between the strength of chemical odors detected by the right fork of the tongue and those detected by the left. The side with the stronger scent is followed [22].
While snakes have no external ear, which makes sense as burrowers, they do hear low frequency sounds. They do not "hear" through the ground, as some believe. Hearing is accomplished through a mechanism that serves as a substitute for the tympanic membrane. This mechanism consists of a thin plate of bone (the quadrate bone) that was once a part of the skull but that has become largely detached and is held loosely in place by ligaments. It lies beneath the surface of the face, covered by skin and muscle, and acts as a receiving surface for sound pressures. The columella, attached to the inner surface of the quadrate bone, conducts the received vibrations to its expanded inner end, which lies in the oval window of the cochlea. If the columella is severed, the sensitivity of the ear is significantly reduced [14].
Although the sensitivity of the snake ear varies with the species, it is appreciably sensitive only to tones in the low-frequency range, usually those in the region of 100–700 hertz. For this low range, the large mass of the conducting mechanism and the presence of tissues lying over the quadrate bone are not of any great consequence. Moreover, while the sensitivity of most snakes to the middle of the low-tone range is below that of most other types of ears, it is not seriously so. In a few snakes, however, the sensitivity is about as keen as in the majority of lizards with conventional types of ear openings and middle ears [14].
The ears of the snake can determine the direction of a sound in terms of its relative intensity in the two ears. Although snakes can perceive vibrations from the ground that are present at a sufficient intensity, this ability is not peculiar to them; all ears respond to vibrations transmitted to the head [14].
The following excerpt from Sullivan, Wingert, and Norris' chapter on North American venomous reptile bites further describes characteristics of snakes (From Auerbach PS (ed). Wilderness Medicine: Management of Wilderness and Environmental Emergencies. ©1995. Reproduced by permission of Taylor and Francis Group, LLC, a division of Informa) [94]:
Snakes lack internal means of regulating body temperature. Thus, environmental temperature variations influence their activity. Snakes become immobile at temperatures below 8 degrees centigrade and cannot survive for more than 12 minutes at temperatures above 42 degrees centigrade. Their optimal temperature range is 27 to 32 degrees centigrade, which occurs in evenings and at night in southern states and southwestern deserts. Therefore, snakes are nocturnal feeders and winter hibernators, the latter usually in rocky dens. Snakes mate in the spring, and in most cases the young are born between August and October. Influenced by temperature and food supply, their maximal growth rate occurs in the first 2 years of life. The rattlesnake's top speed of travel is 3 mph. The life span of some captive snakes has exceeded 30 years.
Venomous snakes are strict carnivores and capable of inflicting a fatal bite at birth. Their food consists largely of small nocturnal mammals, especially rodents, birds and bird eggs, frogs, lizards, and other snakes. Snakes secure prey by lying coiled and immobile beside animal trails or burrows. When the prey comes within striking range, which is usually a distance half the length of the snake, the snake opens its mouth wide, erects its fangs, and lunges forward, briefly burying its fangs into the prey to inject a lethal dose of venom. The amount of venom released is based on the snake's estimate of the victim's size. The speed of the strike is extremely rapid, approximately 8 feet per second. The strike is usually directed slightly downward, but rattlesnakes may strike horizontally or even upward at a 45-degree angle. Although the strike is usually single and from a coiled position, rattlesnakes may strike from almost any position and may strike several times. The strike is more an injection than a bite, so the fragile fangs and jaw articulation are less at risk. The potent venom is designed to immobilize the prey rapidly so that it cannot escape beyond the snake's area of sensory detection (about 20 feet), to prevent a retaliatory struggle that would injure the snake, and to begin digestion of the prey. After the victim is subdued, the snake swallows it head first. This is facilitated by a loose muscular (rather than connective tissue) articulation between the upper and lower jaws, which permits the ingestion of amazingly large prey. However, these articulations have a disadvantageous lack of rigidity and strength, so a large struggling rodent can readily break a rattlesnake's jaw.
Sullivan, Wingert, and Norris describe the mechanism that allows the pit viper to envenom as follows (From Auerbach PS (ed). Wilderness Medicine: Management of Wilderness and Environmental Emergencies. ©1995. Reprinted by permission of Taylor and Francis Group, LLC, a division of Informa) [94]:
The venom apparatus consists of a gland, duct, and one or more fangs on each side of the head. The venom glands are located at the outer edge of the upper jaw, immediately below the eye, and are analogous to the human parotid glands. Each secretory cell synthesizes all components, both toxic peptides and digestive enzymes. The gland is contracted by an external jaw muscle to discharge the venom. Because these muscles have an innervation separate from that of the biting mechanism, the snake can control the amount of venom to be discharged and injected. This is quantitated by the weight and size of the victim. Therefore, discharge is not an all-or-none phenomenon, and pit vipers rarely discharge the full contents at a single bite. High-speed sensory feedback in the heat-detecting pit enables the snake to make split-second adjustments in the force and direction of the strike and in the quantity of venom injected. Although the heat-detecting pit organ is believed to regulate the amount of venom discharge according to the size of the prey, these heat receptors seem to be confused by the large amount of heat radiated by a human. Consequently, on any given strike [directed at a human] the amount of venom released may vary from little or none to almost the entire content of the glands. Most rattlesnakes discharge between 25% and 75% of their venom when they bite a human. After discharge, venom is completely replenished in as little as 21 days. Lethal peptides are replenished first, probably as a defense mechanism.
The venom glands are connected by ducts to two elongated, hollow upper maxillary teeth or fangs that have a slit-like opening near the tip. The ducts empty into the fang sheath, which itself contains venom in a small pocket. When not in use, the fangs are folded against the upper jaw, along the roof of the mouth. During the strike the fangs rotate down and forward so that their base is at a right angle to the jaw. In adult snakes, the fangs vary from 8 mm to more than 20 mm in length. Fangs of large snakes may penetrate to the muscles of humans and may perforate rubber or even leather boots. Reserve fangs may move into position before the functional fang is shed. Therefore, a victim may demonstrate from one to four fang marks at the site of a single bite.
By far, most bites to humans occur in the spring and summer. Snakes are cold-blooded, so they seek warmth underground or among rocks in cold weather. In the spring they are quite active, so one may see relatively many bites as the ground warms. More bites occur in the evenings, as people are more active outside and as the snakes become more active in seeking food during the time of day that better meets their temperature requirements. Wingert notes that [37]:
Although snakes prefer nocturnal activity, a major proportion of the bites (approximately 45%) occur between 3 p.m. and 9 p.m. Males are bitten in a ratio of 4:1 over females. [Male humans suffer from a prefrontal cortex that matures later than that of the female, and many of this subset when bitten have recently consumed alcohol and/or other mind-altering drugs.] Fifty percent of bites occur in children or young adults younger than 20 years of age, with the highest proportion in the 16 to 24 year age group. Of all bites, 65% occur in the upper extremities (usually the hands or fingers) and 34% in the lower extremities. Approximately one-third of all bites are non-accidental; that is, the patient was purposefully handling or playing with a venomous snake.
Exposure to snakes occurs in a variety of places—gopher holes, open air conditioning ducting under buildings, on the roadside, commercial nurseries after the snakes are transported in potted plants, garages, gutter downspouts, corn fields, tall grass, woodpiles, and flowerbeds. According to experts at the Arizona Poison and Drug Information Center, 25% of rattlesnake bites are dry bites—no venom is injected. In another 25%, so little is injected that antivenom is not required [3]. Of an estimated 8,000 bites reported in the United States each year, only 8 to 15 result in death. Most of these deaths can be attributed to complications of the injury, errors in management, or other factors not directly related to snake venom [24].
Russell notes that, "Venomous snakes can leave ordinary tooth marks as well as fang marks, which can easily be confused with the bites of nonvenomous snakes" [12]. It is also important to remember that decapitated head reactions persist for 20 to 60 minutes, and the severed head of a snake can envenom a person [21].
Pit viper envenomation is the term used to describe an array of injurious effects and pathophysiologic changes that follow the release of venom into tissue at the site of a bite from a pit viper. Kitchens notes that [51]:
Pit viper venom is extremely complex, containing a broad range of proteolytic enzymes serving to digest protein, fat, connective tissue, nucleic acids, and other biologic material. Numerous small peptides contained within the venom may account for the autonomic symptoms observed, such as tachycardia, diaphoresis, diarrhea, and vomiting. There is tremendous interspecies variability in the nature of this complex poison. Likewise, variability may be encountered within the same specimen observed over time. This may very well account for the variability of signs and symptoms in pit viper envenomation.
Bearing in mind that the size or type of pit viper has little bearing on clinical assessment and treatment of envenomation (which rely on progression of signs, symptoms, and laboratory abnormalities), the following descriptions of what generally is known about the effects of bites by specific types of pit vipers are cautiously offered for educational purposes.
The venom of rattlesnakes is responsible for more deaths, more hospitalizations, and more injuries than the venom of any other animal in North America [24]. The eastern diamondback rattlesnake accounts for the majority of deaths from snakebite in the United States [51]. Unlike the less complex neurotoxic venom of elapids, pit viper venoms, particularly those of the rattlesnakes (crotalids), are composed of as many as 20 different components.
Eastern diamondback rattlesnakes (Crotalus adamanteus) are typically large snakes capable of delivering a large and quite toxic venom load. They are irritable and sometimes strike more than once, in some cases without warning. The venom is perfectly designed to knock prey down and to cause a disseminated intravascular clotting-like syndrome, which may aggravate bleeding induced by hemorrhagins or by invasive procedures, like a venous or especially arterial puncture, nasogastric tube insertion, or an incision [15].
The canebrake rattlesnake (Crotalus horridus atricaudatus) and its close cousin the timber rattlesnake (Crotalus horridus) are large snakes and should be considered dangerous, although deaths are uncommon. Venom is similar in its effects to that of the eastern diamondback but is less toxic. Still, the bite is a serious matter [15].
Bites by the pygmy rattlesnake (Sistrurus miliarius) may cause hemorrhage, as the venom has some fibrinolytic activity. It is not neurotoxic and does not produce intravascular clotting or hemolysis. It is highly necrogenic, producing much pain and a remarkable degree of swelling. Lethality is low, but a large specimen could be dangerous to a small child or the elderly [15].
While very lethal, patients bitten by the Mojave rattlesnake (Scutulatus scutulatus) may exhibit only moderate local swelling and symptoms of poisoning may be delayed, requiring careful and unhurried evaluation [40]. Neurotoxic symptoms may predominate, including ptosis, fasciculations, myokymia, weakness, and eventually paralysis. Respiratory failure is a risk [32,40].
Envenomation by the massasauga (Sistrurus catenatus) in general causes edema that is far less severe than with crotalid bites, although it may sometimes be marked in an untreated case. Ecchymosis is not unusual, and blebs may form. Necrosis is minimal. Patients do not complain of paresthesia, nor is fasciculation seen. In most cases, there is some immediate pain [11]. Nausea and vomiting may occur. Lethal toxicity is significant [13].
Venom of the cottonmouth moccasin (Agkistrodon piscivorus) is moderately toxic, highly proteolytic, and has both fibrinogenolytic and fibrinolytic activity. It also has bradykinin-releasing and high phospholipase A activity. Only fibrin peptide B is split off from fibrinogen by the venom, which is not sufficient to produce intravascular clotting, and defibrination syndrome does not occur [15]. While said by some to be aggressive and prone to chase humans, like other pit vipers it has poor vision and the "chase" may merely be a coincidence of traveling the same path as the human as each attempts to escape the other.
Copperhead (Agkistrodon contortrix) venom is moderately proteolytic, resembling that of the cottonmouth in its characteristics, but it is much less toxic. Few, if any, deaths are on record due to the bite of this snake. Color pattern somewhat resembles that of a juvenile cottonmouth and the banded water snake, with which it is often confused [15]. Death, permanent loss of function, systemic symptoms, and local necrosis are unusual occurrences [31]. However, in one study, clinically significant local effects (pain requiring parenteral analgesics, swelling, and ecchymosis over one-half of the bitten extremity) occurred in one-third of patients with copperhead snakebites [31]. Although speed of symptom progression is a useful measure for bites from other venomous snakes (e.g., rattlesnakes), results from this study suggest that this measure is less appropriately applied when evaluating copperhead snakebites. As such, copperhead snakebites should not be considered mild or of benign clinical significance [31].
The magnitude of injurious effect is directly related to the amount of venom (venom load), the location of the bite, and the ease with which venom reaches the general circulation. Central to the process are an individual snake's venom toxicity, local tissue capacity to resist a given load, vascular endothelial injury followed by leakage of intravascular fluid into the interstitial space, and interference with the coagulation cascade that may preclude clotting. Secondary effects such as bleeding and critical organ dysfunction can be lethal (which is, from the snake's perspective, precisely the desired effect on the prey).
The impact of envenomation may begin very soon after the bite and quickly affect mobility. As a result, prey may not be able to escape and thus are more likely to be ingested by the snake. Factors determining the prey's fate include size, general health and age, the amount of venom injected, and the location of the bite. While humans are not the snake's natural prey and cannot be ingested, these factors and others, combined with whether early and adequate amounts of antivenom are administered, greatly affect the prognosis.
The size of the victim is important. A mouse will succumb to a dose of venom that would likely pose a considerably smaller risk to a human. This is because the much larger mass of the human dilutes the concentration of venom, thereby weakening the impact of envenomation, in contrast to the limited resilience of the much smaller mouse. By analogy, this accounts for the greater degree of difficulty that a small child may have resisting a given venom load, compared with the level of difficulty that an adult would experience.
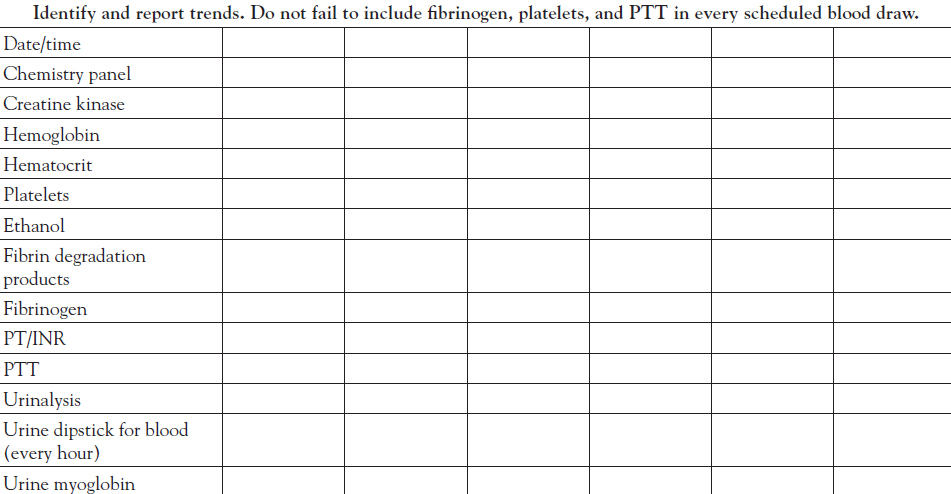
In order to safely and effectively manage the victim of a pit viper snakebite, one must understand the pathophysiology and clinical signs of envenomation, how the venom can impair the blood coagulation cascade, and how laboratory measures of coagulopathy (i.e., fibrinogen, fibrin and fibrinogen degradation products [FDP], platelets, prothrombin time [PT], and activated partial thromboplastin time [aPTT]) as well as urinalysis to identify urine myoglobin are used to assess the severity, progression, and resolution of envenomation.
Harrison provides a concise review of normal coagulation and explains the intricacies of pit viper venom's effects on clotting [53]:
In normal circumstances, the coagulation system can be activated via the extrinsic and/or intrinsic coagulation cascades. The extrinsic pathway is fast acting, responding to the release of procoagulants from injured tissue that quickly convert prothrombin to thrombin, which in turn converts liquid phase protein fibrinogen to the solid phase protein fibrin. The intrinsic pathway is initiated by platelets. The platelets are stimulated to activate in the presence of a variety of agonists, including collagen. The activating platelets adhere to the site of injury, releasing procoagulants that stimulate the slower acting intrinsic coagulation system. The intrinsic system, too, generates thrombin that converts fibrinogen to fibrin. The single-strand fibrin monomers become bundles of strands forming a fibrin polymer. The platelets at the site of injury send out tendrils or spicules that snare the fibrin polymer as it forms. The platelet spicules then retract, pulling the fibrin polymers together, forming a stable clot at the site of injury. Finally, the clot is dissolved by plasmin activity. Plasmin is the product of the plasminogen cascade.
The effect of pit viper venom on fibrinogen can be profound. For example, C. adamanteus, the eastern diamondback rattlesnake, delivers venom that forces the conversion of fibrinogen to fibrin with the thrombin-like enzyme crotalase while enhancing fibrinogen proteolysis through a 20-fold increase in plasminogen activator. The resulting effect causes both coagulation and clot lysis simultaneously. This rapid defibrination renders the blood incoagulable. There is a marked decrease in both fibrinogen and plasminogen levels. Fibrinogen assay is the more commonly performed in hospital laboratories and is an important and sensitive marker of the effect of pit viper venom on the coagulation system. For example, fibrinogen levels can be shown to increase to normal levels after administration of antivenom. Subsequent fibrinogen levels can monitor the decrease in fibrinogen levels as the antivenom's half-life is reached. Interestingly, C. adamanteus venom has little effect on the platelet count or levels of factor II (prothrombin), factor VIII, or anti-thrombin III. FDP can be either the product of primary fibrinogen proteolysis or the result of fibrin lysis mediated by the plasminogen cascade. In the C. adamanteus example, both products would be present. The FDP is a semiquantitative assay that will be increased if either product is present. A positive FDP is used to establish the diagnosis of disseminated intravascular coagulation (DIC). Once DIC is established with a positive FDP, subsequent FDP testing is not particularly useful.
D-dimer is a cross-linked multimer of the breakdown of fibrin. Venoms that force fibrinogen-to-fibrin conversion with thrombin-like enzymes will cause a measureable increase in D-dimer multimers that are inversely proportional to increasing or decreasing fibrinogen levels. This nuance, elegant as it is, does not typically add more insight with regard to the envenomation event than the fibrinogen assay. The D-dimer is more commonly used for the negative predictive value in ruling out pulmonary embolism and is not a suggested test for monitoring snakebite.
Snake venoms can have varying effects on platelets. The DIC resulting from envenomation in itself can consume platelets during the defibrination process. That said, some venoms have no direct effect on platelets, while others can cause platelet activation, leading to aggregation and a subsequent drop in platelet count. Some venom can inhibit platelet function, leaving the platelet count normal, albeit with nonfunctional platelets. Sequential hemograms are useful for monitoring the hemoglobin and hematocrit as well as the platelet count post-envenomation and during antivenom administration.
The PT and the aPTT are time-honored coagulation tests that are commonly requested and interpreted by clinicians. The PT is useful in the detection of coagulopathy in the extrinsic coagulation cascade and is the test of choice for the monitoring of warfarin therapy. The aPTT monitors the intrinsic coagulation cascade and is commonly used to assess the effect of heparin. Both PT and aPTT are typically affected by defibrination. Both the PT and the aPTT are clottable assays that depend on normal fibrinogen levels to detect the conversion of fibrinogen to fibrin for their respective test end-points. If fibrinogen is absent the PT and aPTT will be prolonged. Fibrinogen remains the more sensitive coagulation test for monitoring the effect of snake venom and assessing the half-life of antivenom.
The bite from a pit viper usually causes some degree of swelling around the bite area within 5 to 10 minutes. Nearly all bites cause pain, and pain that is initially mild usually increases in severity as edema progresses. The bite of a non-venomous snake may also be painful, but the pain results simply from the trauma of the bite and is of limited severity and duration, in contrast to the dull, and typically increasing, pain and tenderness caused by envenomation. As local tissue breaks down, there is progressive swelling and ecchymosis.
Local swelling resulting from a deposit of venom may be immediate, greatly delayed, or absent, depending on the venom load and the depth of the bite. Deeper bites that penetrate muscle are more likely than subcutaneous bites to swell less, especially early on, but the venom is likely to make a more rapid entry into the systemic circulation due to the greater vasculature of muscle. This increases the likelihood of rapid clinical deterioration and more severe clotting abnormalities.
Most bites penetrate subcutaneous tissue, and the early spread of venom is by lymphatic drainage rather than venous flow [10]. For this reason, swelling and tenderness of the regional lymph nodes is common, even if only a relatively minor degree of envenomation is present [15].
Local tissue reaction is the body's first line of defense, serving to contain and neutralize the venom to a greater or lesser degree. Depending on venom load, swelling can be very impressive, extending over a day or so in spite of what is thought to be reasonable doses of antivenom. Unfortunately, poor perfusion due to tissue dissolution accounts in large part for the difficulty getting intravenous (IV) antivenom into the injured region to bind effectively with the venom molecules. If early and adequate doses of properly administered antivenom are able to penetrate the local site of injury well, systemic envenomation is limited and any swelling generally resolves within 10 days.
Pit viper envenomation can rise to the level of a complex poisoning, affecting every organ system, resulting in coagulopathy and hemoconcentration (early) followed by a decrease in erythrocytes (anemia), thrombocytopenia, hematuria, proteinuria, hematemesis, melena, hemoptysis, epistaxis, and hypotension [19]. Hypofibrinogenemia and thrombocytopenia are common findings. Physiologic responses to envenomation, such as release of bradykinin, contribute to the severity of pain and, short of a shock state, may cause significant but transient hypotension [15]. Difficulty swallowing (dysphagia), blurred vision, and marked thirst may occur, and the patient may experience significant weakness after eastern and western diamondback envenomation [12]. The patient may feel faint, nauseated, and/or numb or tingly around the mouth, tongue, scalp, fingers, toes, or site of the bite [19]. Such paresthesias indicate that a significant envenomation exists. Fasciculations are very common in human victims, and when present after a bite by an unidentified snake should suggest the eastern diamondback, the Mojave, or the southern Pacific rattlesnake as the offending reptile. Fasciculations in severe cases may become generalized, especially with eastern diamondback rattlesnake bites [15]. Transient myosis is said to be a symptom of severe envenomation due to the bite of the eastern diamondback rattlesnake [15]. Convulsions are particularly common in children [12]. Prolongation of bleeding and clotting times may occur [19]. In addition to visible tissue destruction (including blebs, sloughing, and gangrene), changes in capillary permeability may occur, leading to great fluid loss and hypovolemic shock as well as pulmonary edema (both of which are frequent causes of death) and swelling of entire limbs. Anaphylaxis and Kounis syndrome are unlikely but possible results of snakebite and should be considered in the overall assessment of the patient.
According to Wingert, "large amounts of venom can result in…an immediate profound fall in systemic arterial pressure and a decrease in circulating blood volume believed due to pooling of blood in the major vessels and capillary beds in the lung. Hypovolemic shock and pulmonary edema appear to cause most fatalities. Hemoglobin decreases and bleeding occurs throughout the body, often as soon as six hours after envenomation" [37].
Russell describes the effects of serious envenomation as follows [11]:
In moderate or severe envenomations, the tingling or numbness may extend to the face and scalp within the first 30 minutes, with tingling of the fingertips and toes following later. Sometimes changes in taste and sensation occur even before swelling, so asking about them may help establish the diagnosis sooner. A metallic, rubbery, or minty taste in the mouth and tingling of the tongue and lips are virtually pathognomonic of rattlesnake envenomation, and may occur before or in the absence of swelling. Fasciculations may also appear very early in the course of some rattlesnake poisonings but are rarely seen after bites by copperheads. Fasciculations are most noticeable on the face and over the large muscles of the back and neck, as well as in the bitten extremity. Moderate-to-severe rattlesnake poisoning may also cause erythema and ecchymosis around the bite area within three to six hours. Ecchymosis may occur in any area whose skin rubs against itself, or may be precipitated by even slight trauma.
Pit viper venom has direct and indirect adverse effects on vital organs, including the kidneys and the lungs. When there is widespread tissue toxicity (to a large amount of venom), cellular debris, hemoglobin, and myoglobin released into the circulation may precipitate in the kidney tubules, resulting in renal shutdown [37]. This complication can be avoided if IV fluids are administered at a rate sufficient to provide a brisk urinary output and if antivenom administration is begun early and given in adequate amounts. Bush and Jansen advise that the risk of nephrotoxicity from rhabdomyolysis and myoglobinuria is increased at low urine pH and thus may be reduced by alkalinization [48].
While providing adequate hydration to support blood pressure and protect renal function is important, one should be mindful that venom-induced changes in capillary permeability within the pulmonary vascular tree render the lung more susceptible to pulmonary edema in response to fluid challenges. Wingert comments that, "Pulmonary edema occurs in severe cases due to injury of the epithelial lining of the pulmonary vasculature, with loss of serum proteins and plasma into the alveoli" [37]. Prevention and amelioration of pulmonary toxicity depends on early and adequate amounts of antivenom as well as careful monitoring for signs of fluid overload. Lung sounds should be closely monitored for signs of pulmonary edema. IV fluid administration rates should be carefully adjusted to prevent renal toxicity and to prevent fluid overload.
The heart can be directly affected as well. Some pit viper venoms contain a direct cardiac muscle depressant [37]. A single case of cardiac arrest in an otherwise healthy young adult male patient was ultimately believed to be a direct venom effect, as electrolytes were within normal limits, the patient was well-hydrated, and there was no reason to believe that antivenom would cause such an event. The patient recovered after a few moments of chest compressions. Antivenom administration was not stopped, and no further arrests occurred.
Snake venom is one of a number of environmental exposures known to trigger Kounis syndrome [96]. This should be considered along with the need of antihistamine administration, supplemental oxygen, and possibly other interventions for any patient with known or suspected snakebite who is experiencing chest pain or electrocardiographic changes.
Coagulopathy
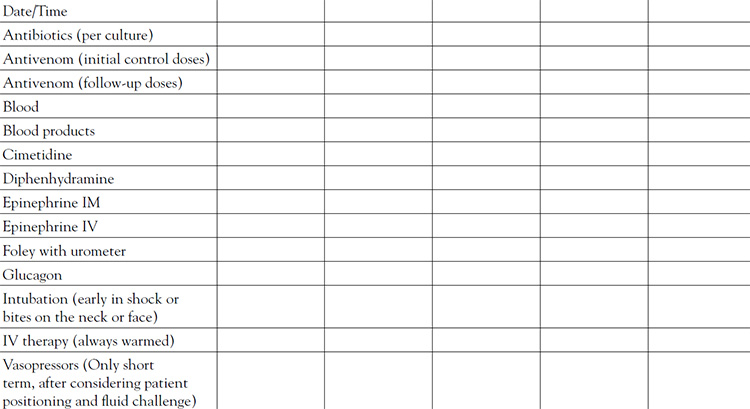
Activation of the coagulation cascade and multiple defects in blood clotting (coagulopathy) are common following pit viper envenomation, putting the patient at risk of a serious bleeding diathesis. Platelet counts and fibrinogen levels are key to assessment and treatment. Consultation should be considered in any case with confusing features and promptly sought for any patient whose clotting factors fail to improve after two 6- or 10-vial doses of CroFab or two 10- or 20-vial doses of Anavip. The platelet count is usually a sensitive indicator of systemic venom activity and an early marker of antivenom effectiveness, providing a quick laboratory measurement that is used (hourly in severe envenomations, along with hemoglobin, hematocrit, and fibrinogen) to assess progress in reversing the coagulopathy. The cause of thrombocytopenia in pit viper envenomation is not yet fully understood. There is no evidence of a defect in production of platelets, and the treatment approach to thrombocytopenia is administration of antivenom, not platelet transfusion [4]. Platelet counts usually respond rapidly to antivenom administration alone. Reversal of thrombocytopenia may be seen within one hour following treatment with an adequate dose of antivenom.
As important as it is to monitor platelets, it is at least as important to monitor fibrinogen, as this is a more sensitive marker of envenomation in the early hours after snakebite. At times, even in the face of a heavy venom load, it is possible to have a normal platelet count but low fibrinogen. The platelet count in such cases could remain normal in spite of potent venom activity because there is no fibrinogen available to convert to fibrin, and thus no fibrin available to be snared by platelets, leaving the platelets free to be counted.
Close monitoring of clinical status and laboratory parameters and, when indicated, early intervention with adequate doses of antivenom are required to assure a safe outcome. Initial laboratory studies to evaluate coagulopathy include fibrinogen, platelets, PT, PTT, and FDP. The unstable patient requires serial assessment of the PT, hematocrit, platelets, and fibrinogen at regular intervals, perhaps every hour, until stable. Laboratory studies are generally performed every six hours after initial control is achieved and the patient is stable. More severe cases may require selective laboratory monitoring every four hours after the patient is stabilized. Laboratory monitoring for coagulopathy is also performed one hour after each dose of antivenom has infused, including maintenance doses in the case of CroFab administration, to assist in assessing whether initial control has been attained or maintained.
While local swelling in the region of the snakebite may be the most visible sign used to identify progression or lack of progression of envenomation, it is important to bear in mind that an occasional patient will have scant or even no swelling and yet a severe coagulopathy demonstrated by a plummeting fibrinogen level and likely plummeting platelet count as well. The impact of the venom on the blood coagulation cascade, coupled with the possibility of hypovolemic shock resulting from leakage of intravascular fluid into the tissues, constitutes the greatest threat to the victim. Incipient coagulopathy following envenomation is, in a sense, a "stealth attack," because coagulopathy cannot be felt by the patient or seen by the clinician unless overt bleeding is present. This, along with the fact that an occasional coagulopathy develops only after a surprising number of hours, is the reason that all pit viper snakebite victims who have and continue to have no swelling or other signs or symptoms should be evaluated for coagulopathy and monitored clinically by an informed staff in an urgent care setting with the ability to perform all required laboratory studies for 8 to 12 hours before being discharged home. Typical urgent care units are unlikely to be able to perform timely fibrinogen or FDP testing and thus would be an inappropriate setting for pit viper snakebite evaluation. As such, patients typically initially present to a hospital-connected emergency center, and those who begin to demonstrate any signs or symptoms of envenomation should be admitted to an ICU for a minimum of 24 hours.
Derangement of the coagulation cascade, when pronounced, often results in overt signs of bleeding. This may present as renewed bleeding at the bite wound site or at venipuncture sites. New-onset hematuria may be a more common and early sign of bleeding diathesis. Microscopic hematuria often develops before gross hematuria is evident. Microscopic hematuria can be detected rapidly by dipstick urinalysis, a simple bedside test that can easily be included in regular clinical monitoring along with vital signs and laboratory tests. Importantly, a dipstick that is positive for blood may also indicate myoglobinuria (indicative of rhabdomyolysis), which should be confirmed by further testing. During clinical observation, the performance of hourly dipstick urinalyses at the bedside may lead to a significantly earlier treatment intervention than if one were to wait for the results of the next serial blood tests. Recalling that early and adequate doses of antivenom are the mainstay of snakebite treatment, the importance of this simple, inexpensive bedside test cannot be overemphasized.
Pit viper venom, if in a dose that precludes neutralization by local tissue, may attack the blood and blood coagulation cascade, breaking up fibrin, preventing the conversion of fibrinogen to fibrin, and/or causing thrombocytopenia, any of which puts the patient at risk of serious bleeding. In other clinical contexts (e.g., cancer chemotherapy), transient thrombocytopenia and hypofibrinogenemia are common findings that do not usually lead to overt bleeding; however, clotting abnormalities produced by envenomation tend to be progressive, sustained, and life-threating unless controlled by early and adequate doses of antivenom. At the same time, however, it is important to be alert to the patient's baseline health and medical history (e.g., anemia, other hematologic disorder, anticoagulant drug use) in interpreting initial laboratory data. In addition to pit viper envenomation, the following conditions are associated with hematologic and coagulation factor abnormalities [9]:
Cancer
Anemia
Infection
Collagen disease
Congestive heart failure
Diarrhea
Hepatic disorders
Hyperthyroidism
Poor nutritional state
Steatorrhea
Vitamin K deficiency
While alternate terms for antivenom include antivenin, antivenenum, antivenene, and antivenome, it is agreed that the snakes inject venom, so the logical name for an agent that counteracts the actions of venom is antivenom. This term is preferred in most discussions of the topic.
Antivenoms currently in use (CroFab and Anavip) that target pit viper venom are suspensions of venom-neutralizing antibodies prepared from the serum of sheep (CroFab) or horses (Anavip), both host animals having been hyperimmunized against specific venoms [33]. Protherics Inc., the manufacturer, describes Crotalidae Polyvalent Immune fragment antigen binding (Fab) (Ovine) (CroFab) as [9]:
…a sterile, nonpyrogenic, purified, lyophilized preparation of ovine Fab (monovalent) immunoglobulin fragments obtained from the blood of healthy sheep flocks immunized with one of the following North American snake venoms: Crotalus atrox (western diamondback rattlesnake), Crotalus adamanteus (eastern diamondback rattlesnake), Crotalus scutulatus (Mojave rattlesnake), and Agkistrodon piscivorus (cottonmouth or water moccasin). To obtain the final antivenin product, the four different monospecific antivenins are mixed. CroFab is a venom-specific "Fab" fragment of immunoglobulin G (IgG) that works by binding and neutralizing venom toxins, facilitating their redistribution away from target tissues and their elimination from the body.
Anavip, or Crotalidae Immune Fab 2 (Equine), is an equine-derived antivenom indicated for the management of adult and pediatric patients with North American pit viper envenomation [97]. It is a sterile, lyophilized, polyvalent preparation manufactured from the plasma of horses immunized with venom of Bothrops asper and Crotalus durissus. Anavip contains venom-specific Fab fragments of IgG that bind and neutralize venom toxins, facilitating redistribution away from target tissues and elimination from the body. The larger molecular mass of Anavip results in a longer elimination half-life than CroFab (mean: 133 hours vs. 30 hours), potentially resulting in fewer recurrent envenomation symptoms [97].
Anavip was approved for use in rattlesnake bites in 2018. In early 2021, usage was extended for use in patients envenomed by any North American pit viper. Ongoing research and further experience will help to define how the two types of antivenoms might best be considered in practice. The safety profiles of the two types of antivenoms appear similar. According to Rocky Mountain Poison and Drug Center, both antivenoms are safe and effective when adequate doses are given. Dosage recommendations for Anavip may shift as more is learned. Treatment with Anavip may require bigger doses than expected, and more vials of Anavip (compared with CroFab) are generally required to successfully treat a patient (in a ratio of 2:1). Anavip loading doses of 10 vials are recommended by the manufacturer until initial control is achieved, with doses of 4 vials administered as needed to suppress local and systemic envenomation effects that may follow initial control. Unlike with CroFab, there are no scheduled maintenance doses.
The CroFab and Anavip manufacturers do not address severe envenomation, though CroFab now allows a range of doses that can successfully address that circumstance. The 2:1 ratio is instructive regarding how one would use Anavip in severe envenomations. The Anavip package insert indicates a reconstitution time of less than 27 seconds, which is considerably quicker than CroFab. Until more is learned about Anavip dosage, the practitioner should remember that it is better to overtreat with antivenom than to undertreat, and that symptomatology and lab values determine when and how aggressively to treat the patient.
Both Anavip and CroFab are considered "polyvalent" because venoms from more than one species of snake are used to produce them, resulting in products that can protect against more than one type of snake venom [33]. The venoms of the pit vipers are varied but share common characteristics, allowing antivenom to work across species lines within the pit viper group of snakes. The CroFab title includes "Ovine" to denote its production using sheep serum, while the Anavip title includes "Equine" to indicate its production using horse serum.
In treating pit viper envenomation, since 2000, CroFab has proven to be much safer than the older product (Antivenin [Crotalidae] Polyvalent, Wyeth, no longer available) due to the method of purification used in its production. The older product was manufactured using methods from the early 1950s, which did not produce a pure antivenom. This impurity resulted in up to 10% of patients having mild-to-moderate allergic symptoms (e.g., hives or wheezing) and approximately 0.5% having major immediate reactions, including anaphylaxis [51]. Due to the higher purity of currently available antivenoms, there is a much lower risk of triggering a serious allergic response by its administration; therefore, there exists now a greater willingness to treat patients with antivenom and to treat in a more timely way, which is an important step forward in snakebite treatment.
As with the older product, CroFab and Anavip are employed essentially as a titration to stop the progression of evolving signs and symptoms, as well as restore balance to the blood coagulation cascade. Pit viper antivenoms do not reverse damage that is already present. Very importantly, due to the relatively small size of antigen binding fragments (Fab) and thus faster clearance by the kidneys compared to the older product, CroFab has an effective elimination half-life of approximately 12 to 30 hours, while the venom may continue to diffuse from the bite site and remain active for many days [1]. Consequently, periodic maintenance doses of CroFab are necessary following a titration of antivenom that leads to initial control. To date, the manufacturer of Anavip does not call for a routine maintenance dose after initial control is achieved due to the product's larger antigen binding fragments and slower renal clearance relative to CroFab, opting instead for controlling breakthrough progression with additional smaller doses of antivenom. However, it is important to remember that symptomology and laboratory testing guide treatment; achievement of initial control is not a guarantee of long-lasting control. Regardless of which antivenom is administered, titration should approximate as closely as possible the amount needed to slightly exceed the venom load, and as noted, one should be aware that venom may continue to reach the systemic circulation for a number of days following initial treatment in the ICU. Office follow-up is generally required following discharge from the hospital because of the possible presence of this protracted leakage, with additional laboratory studies performed depending on the values of inpatient laboratory reports and on any signs of recurrence of envenomation seen on re-exam.
It is noteworthy that in clinical trials involving CroFab, only patients with mild-to-moderate envenomation by a pit viper were enrolled in the study; however, it is possible that some patients with severe envenomation were enrolled because, importantly, a patient who initially appears to be minimally or moderately envenomed may actually be severely envenomed [9]. Such patients, by virtue of early treatment with antivenom, may not have developed clinical signs or laboratory evidence indicative of severe envenomation. The importance of this is that CroFab dosage recommendations do not include specific guidance for cases classified as "severe," though doses of 4 to 12 vials are allowed. (This author's experience with severe envenomation and those of South Georgia Surgical Associates—a group involved in CroFab clinical trials—led to a suggested titrated dosage that has worked well for this subset of patients.)
Important cautions are warranted concerning proper mixing of CroFab. The product should not be shaken, as it will form heavy foam comprised of needed protein that cannot be retrieved and administered to the patient. To avoid foaming, instill the diluent (18 mL 0.9% saline) slowly against the vial wall. After the diluent is instilled, in order to hasten mixing by neutralizing the pressure within the vial, insert a needle without a syringe into the vial and after a moment remove it. Next, mix by continuously and gently inverting the vial at a rate not to exceed two cycles per second.
Anavip is said to mix readily. It is diluted using 10 mL 0.9% saline per vial, followed by continuous gentle swirling.
Alternatively, either antivenom can be mixed by withdrawing from an IV bag of normal saline (sized to the amount of fluid the patient is to receive over the following hour) the amount appropriate to dilute the vials of antivenom, after which the vial contents are added back to the bag [4].
As mentioned, CroFab and Anavip are the two antivenoms currently in use in the United States to treat patients envenomed by North American pit vipers; however, on a U.S. Food and Drug Administration (FDA) fast-track is a synthetic, small molecule antivenom (varespladib) that is said to block secretory phospholipase A2 (sPLA2), the foundational, abundant, and active toxin found in most snake venoms worldwide, including snakes in the Viperidae, Elapidae, and Colubridae families. If positive experience thus far is replicated in human trials, this seems likely to result in a paradigm shift in the treatment of all or nearly all kinds of snakebites worldwide [102,103].
Notably, while current antivenoms can successfully rescue the patient if treatment is early and adequate, this prospective sPLA2 inhibitor varespladib (LY315920), which is administered intravenously, may be better able to preserve local tissue in addition to rescuing the patient. This is because the small size of the molecule allows it to penetrate tissue that the other antivenoms struggle (at best) to access.
Furthermore, oral varespladib (LY333013) is being tested in human trials in addition to the intravenous form [102,103]. Worldwide, many thousands of people die yearly from snakebite because they are inadequately treated, antivenom is not available, or the victim lives remotely and transportation is either unavailable or inadequate for the circumstance. The oral form of varespladib (LY333013) administered on scene and followed up with transport to a higher level clinic or hospital should save countless numbers of lives [102,103].
Varespladib, attached to venom, is cleared from the body a bit differently from CroFab and Anavip, likely primarily renal clearance and/or renal glomerular filtration followed by reabsorption and catabolism in the proximal tubule. sPLA2 is 12–14k Daltons, below the upper limit of glomerular filtration (50k Daltons). After glomerular filtration, proteins tend to be reabsorbed by proximal tubular cells and then degraded into free amino acids. How much venom sPLA2 is excreted in urine vs absorbed and broken down is not well characterized. After snakebite, venom sPLA2 distributes widely throughout the body and binds to target sites via proteins on cell membranes (e.g., neurons, muscle cells). When varespladib finds the PLA2, it binds with it and also disengages the binding of PLA2 to the cell. The venom PLA2 drifts off and can end up back in circulation, from which it can then be broken down and cleared by the kidneys.
Field experience should clarify whether varespladib alone is adequate to treat all or nearly all snakebites, or whether a mix of varespladib and traditional antivenoms would be more effective, as varespladib is not believed to target snake venom toxins other than sPLA2. According to the manufacturer [99,104,105,106,107,108,109]:
Varespladib is an experimental drug that is currently being developed for the treatment of snakebite envenoming. It is not yet approved for human use by the FDA or any other regulatory body. Varespladib is a small molecule that is a potent inhibitor of snake venom secretory phospholipase A2 (sPLA2). Varespladib methyl, its orally bioavailable methyl ester prodrug, is rapidly converted to the active compound after administration. Snake venom sPLA2 is one of the major toxin classes, found in the venom of more than 95% of venomous snake species, including all species native to the United States. Snake venom sPLA2 is implicated in a broad range of pathologies including neuromuscular and neurovascular effects, local tissue injury, and coagulopathy. Among U.S. pit vipers, all highly toxic venoms contain sPLA2 neurotoxins. In preclinical challenge-then-treat studies, varespladib prevents or delays the lethality of venoms from a wide range of viper and elapid snake species…including Crotalus atrox, C. scutulatus, Agkistrodon contortrix, and Sistrurus miliarius.
The first antivenom for use in patients envenomed by a pit viper was introduced in the United States in the early 1950s. A significant improvement in purity and safety came in 2000, and then, in 2018, an equally pure but larger molecule antivenom was introduced with the goal of providing longer lasting protection by way of an extended half-life. It has been about 70 years since antivenom was introduced, and 50 years passed before the first improvement in the product came along. Another 18 years passed before the next version arrived. Now, a true paradigm shift in snakebite treatment seems likely as the fast-tracked FDA evaluation of varespladib moves forward.
Clinicians who are faced with a case of pit viper venom poisoning would be wise to consider the patient as having a complex poisoning and to be prepared to institute treatment for cardiovascular dysfunction (including shock), renal failure, hemorrhage, respiratory distress or failure, and tissue destruction [11].
Because of the variable composition of pit viper venoms, the systemic manifestations of poisoning are variable in onset and progression; optimal management requires careful, frequently repeated clinical assessment over an extended period while anticipating the serious complications that may be associated with snake envenomation [39]. Hurlbut et al. make three fundamental and critical points about assessment of the snakebite victim [50]:
An unremarkable physical exam and laboratory profile at presentation does not reliably indicate that the degree of envenomation is insignificant.
The early clinical course does not necessarily indicate a benign outcome.
It is important that physicians observe patients who present with snakebite for more than eight hours.
For clinical purposes, the degree of envenomation has been classified by some in the literature as minimal, moderate, or severe, in an attempt to simplify the process of clinical analysis and aid in correlating severity with decisions to treat with antivenom. However, with the exception of severe envenomation, a level of severity constituting about 13% of victims treated with antivenom, this approach runs the risk of oversimplification and has been known to lead to complacency on the part of clinicians lacking experience with snakebite and to admitting these patients to areas other than an intensive care unit [88]. Clinical assessment of envenomation is a complex, fluid, and continuous process requiring experience with the treatment of snakebite (Figure 2). Consultation is strongly recommended if the practitioner is at all unsure of how to proceed. Documentation of consultation and of efforts to mitigate a language barrier that can complicate assessment is an important risk management step.
Severe envenomation is characterized by Russell as [12]:
Swelling, erythema, or ecchymosis may involve the entire extremity and be spreading rapidly. Systemic manifestations are pronounced and may include nausea and vomiting, changes in mental status, severe tachycardia or bradycardia, tachypnea, respiratory distress, and hypotension. Coagulation defects are usually present, and serious or spontaneous bleeding may occur. Platelets are often less than 20,000 per mcL and other laboratory values are abnormal.
Weakness, dizziness, cold and clammy skin, tachycardia, nausea, vomiting, and diarrhea are all nonspecific symptoms and may be manifestations of an emotional or anxiety reaction (Table 1), in which case they appear almost immediately after the bite. Such symptoms may also reflect systemic envenomation, in which case they generally occur after 30 to 60 minutes, or longer [15]. Importantly, "nausea and diaphoresis accompanied by dizziness and weakness are probably not primary neurologic signs but indications of early hypotension due to intravascular volume depletion," in which case treatment with antivenom and a normal saline bolus would be indicated [25].
CASE STUDY: EMOTIONAL REACTION
|
"Trivial" envenomation exists when swelling is localized 30 to 60 minutes after a bite and the patient ultimately exhibits no paresthesias, fasciculations, or ecchymoses; no symptoms other than minor pain; and no abnormal serial laboratory findings [12]. Antivenom is not indicated for apparent trivial envenomation. Instead, the affected part should be cleaned and immobilized and the patient should be immunized against tetanus and studied for at least 12 hours or overnight to ensure that delayed progression does not develop [12].
An observation (study) period that is limited to six hours from the time of the bite is likely to be inadequate. Coagulopathy can develop at 12 hours, with questionable scant other antecedent signs or symptoms. Certainly, the patient's level of function should be considered before being discharged at 6 hours, including whether a capable person is available to monitor and assist the patient for 24 hours at home. Additionally, the age and general health of the patient, distance from the hospital, and current use of anticoagulants should be considered before discharge. Laboratory studies should be repeated at least once before discharge from the emergency department in order to detect any deterioration of clotting ability and to ensure that the patient is reasonably safe for discharge. These repeat studies should be drawn six hours after the baseline labs were drawn, assuming no progression of signs and symptoms in the interim. By the time all results are available in most settings another hour or so would have passed, so time would begin to approach the more appropriate 8 to 12 hours of study in the emergency department. If the patient arrives six hours after the bite and is asymptomatic, one may consider obtaining only a single set of laboratory studies and a shorter period of observation. If this decision is made, the practitioner should be aware that by limiting the assessment to just baseline studies, only a snapshot of the patient's clinical course and blood coagulation status would have been observed, which would preclude identification of any trends. In such cases, there should be assurance of continued self-examination and easy return to medical care should clinical deterioration supervene. (It should be noted that a positive FDP test result can be a harbinger of things to come, and that such patients should be hospitalized and studied closely over time for evidence of deteriorating clotting factors.)
In general, it is advisable to elevate the extremity that sustained the bite; however, before elevating a swollen extremity, one should consider whether antivenom is available and whether the need for antivenom administration is already present, because the venom (and signs and symptoms) could spread more rapidly as a result of elevation. After the extremity is elevated, the leading edge tends to flatten or may disappear completely, and thus becomes less useful for assessing progression of injury. If the leading edge cannot be readily seen, gentle palpation may reveal it because the edge is where the venom is most active in dissolving tissue, resulting in a differential tenderness in the area. The leading edge of swelling is a more sensitive measure than circumference, though measures of circumference should be included for a complete assessment of progression of swelling and, importantly, of intravascular fluid loss that would necessitate replacement.
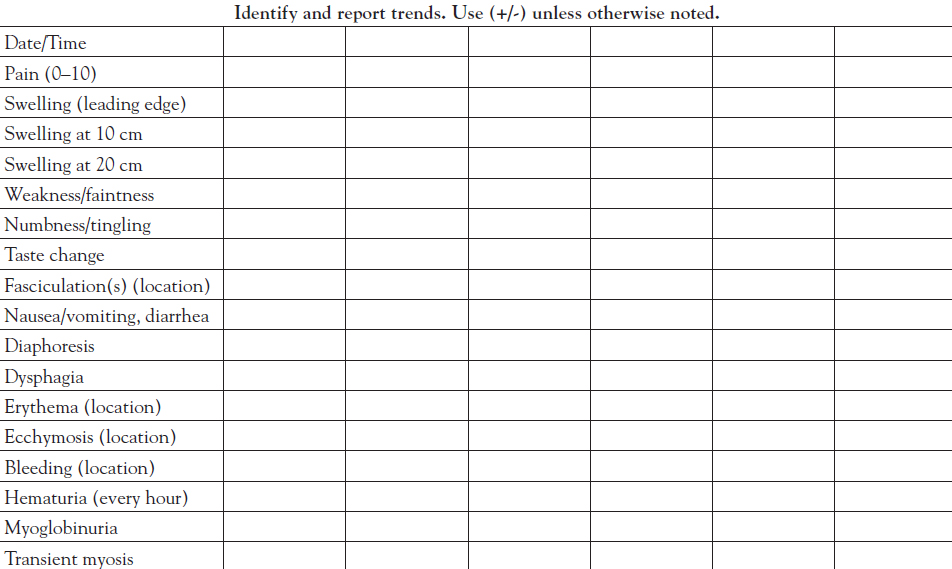
The need to treat with antivenom becomes clear when the assessment reveals obvious signs or symptoms from the time of the bite that continue to progress en route to care and during the course of emergency department observation. A myriad of clinical signs and symptoms may be seen, any one of which could indicate envenomation:
Pain
Swelling
Weakness or faintness
Numbness or tingling
Change in taste
Fasciculations
Nausea and/or vomiting
Diarrhea
Diaphoresis
Dysphagia
Erythema
Ecchymosis
Bleeding (including hematuria)
Myoglobinuria
Transient myosis
Persistent or episodic hypotension
Severe tachycardia or bradycardia
Respiratory distress
Abnormal blood studies
Changes in mental status
Pit viper venom does not cross the blood brain barrier and thus does not affect the brain directly. The presence of mental status changes suggests a comorbidity, prescription or recreational drug use, or the possibility of a cerebral bleed as a result of coagulopathy. Other complications of envenomation that may lead to mental status change include hypoxia secondary to pulmonary edema, cardiac failure, or hypovolemic shock. Aberrant behavior and seizure point to hypoxia, the etiology of which should be immediately explored.
Assessment can be especially challenging when shortly after arrival the patient has normal laboratory results and no obvious signs or symptoms other than minimal pain and swelling. This could mean that the venom load is relatively minimal and confined to tissue at the bite site, where it is being successfully neutralized. However, an alternate possibility is that a relatively large venom load has been temporarily sequestered by local tissue and has simply not yet definitively revealed the full potential for injury. Adding to the difficulty is the observation that progression of swelling can occur in an irregular, halting manner as enzymes in the venom disrupt different layers of tissue. Significant swelling is defined as that which progresses at a rate of at least 1 cm/hour at the leading edge or shows demonstrable change in circumference [4]. On occasion, a seemingly benign progression or an apparent halt to swelling may tempt the practitioner to conclude, prematurely, that the issue is resolving and that the prognosis is benign. Considerable judgment is required, and practitioners should take into consideration all signs and symptoms as well as the results of serial laboratory studies in determining whether the venom, if even present, has yet begun to have an effect and whether treatment is or is not indicated at the moment.
Ultimately, assessment of severity and need for treatment is a measured process requiring a series of clinical and laboratory evaluations over a number of hours, paying particular attention to signs of volume depletion and emerging abnormalities in the blood coagulation cascade. Even in the absence of progressing swelling, systemic spread of venom and early damage to organ systems may be in progress. Persistent vigilance is required, and intervention with appropriate doses of antivenom is important at the earliest indication of significant envenomation. One should not wait hours before treating, hoping that episodic progression ends. "Time is tissue," and getting behind the curve of coagulopathy can result in serious bleeding complications. Clearly, informed and continuous assessment is required to assure a positive outcome—continuous because the venom acts continuously, even when temporarily sequestered by tissue at or beyond the bite site. Continuous assessment in the ICU should occur until, applying the standard of care, there is certainty that the envenomation has largely run its course. Because snakebite is rarely seen in most clinical settings and because of the deceptive nature of envenomation and the importance of early and adequate doses of antivenom, it is strongly recommended that all patients remain under the care of ICU staff until they are ready to be discharged home.
While venom load cannot be calculated precisely, there are some clinical clues that can help determine whether the load may be small or significant. Pain, unless the patient has neuropathy affecting the area of the bite, may help to determine venom load. If pain is minimal and remains minimal in a healthy patient, equal perhaps to a thorn prick, that could be an encouraging sign; however, if pain is or becomes moderate or severe, then it is reasonable to believe the venom load may be significant. Progressive pain is an indication that the venom is spreading and adds to the list of reasons to consider treatment.
Time, as it relates to pain and swelling, can also be helpful in determining venom load. If it takes only moments after the bite to become very painful, and the pain is progressive, that likely speaks to a more significant venom load. Likewise, if swelling is dramatically increasing and does not slow as it spreads beyond the area of the bite, it is reasonable to believe that a significant amount of venom may have been injected.
Estimating venom load and accurately assessing the need for antivenom therapy also requires knowledge of the time that has elapsed from onset of the bite to when the practitioner first sees the patient. If it has been 30 minutes and there are continuing progressive signs and symptoms consistent with envenomation, then it is reasonable to assume that the patient is going to need to be treated, especially if the patient is experiencing one or more of the following:
Severe pain
Rapid swelling
Hypotension or hypotensive episodes
Heart rate greater than 130 beats per minute
Significant arrhythmia or bradycardia
Nausea
Fasciculations
Ecchymosis
Weakness
Faintness
Numbness
Tingling
Change of taste
Dysphasia
Coagulopathy
Hematuria
Overt bleeding
On the other hand, if the patient arrives one hour after the bite and has minimal pain, swelling localized to the bite site, and none of the preceding or other signs and symptoms, then one may consider that treatment with antivenom may ultimately not be necessary. However, continuous clinical observation in an ICU and serial laboratory studies are required for all symptomatic patients with pit viper bites to prove that the clotting mechanism is and remains intact. Immediate consultation should be sought if the practitioner feels at all unsure of whether to begin treatment, alter treatment, or back off the frequency of coagulation studies (maximum interval of six hours).
The goals of treatment of the patient with pit viper envenomation include limiting local tissue injury, preserving the integrity of the blood coagulation cascade, and protecting all organs from the effects of the venom. Both CroFab and Anavip antivenoms contain Fab fragments of antibodies with specific shapes that allow binding directly to specific pit viper venom protein sites, thus forming antigen-antibody complexes that inactivate the poison and permit excretion by the kidneys. Varespladib also binds to venom molecules, but it acts by binding to active sites within sPLA2 enzymes.
When clinically indicated, early administration of antivenom in adequate doses is essential for optimal clinical outcomes. Consultation with an experienced toxicologist or snakebite clinical specialist should be sought prior to the administration of antivenom, and discussion should include whether to administer one or the other available pit viper antivenoms unless the treating clinician is current in managing snakebite envenomation. (Access to a medical toxicologist is provided by the U.S. regional poison control center by calling 1-800-222-1222.)
The decisions of whether to treat and the initial dose (or doses) of antivenom to employ are determined by continuous clinical monitoring of any signs and symptoms and, very importantly, the results of laboratory studies. The goal of administering CroFab or Anavip is to bind, molecule for molecule, venom to antivenom [4]. For this reason, children require the same dose(s) of antivenom as adults; however, children may require antivenom earlier than adults due to their smaller volume and the resulting higher concentration of venom [8].
Russell suggests that the use of steroids may be contraindicated in the first six to eight hours after envenomation because they may suppress the patient's immune response, enhance the action of the venom, hinder the action of the antivenom, and possibly increase the likelihood of gastrointestinal hemorrhage [12].
Children are more likely than adults to present with hypovolemic shock from pit viper envenomation. Any patient presenting with shock from snakebite can challenge one's knowledge of snakebite and how to treat the patient, but shock resulting from envenomation is treated nearly exclusively with the administration of antivenom. In the case of CroFab, 10-vial increments are administered until the patient is stabilized, along with large amounts of warmed normal saline (at least 20 mL/kg) in both children and adults [6]. In the case of Anavip, 20-vial increments are administered in shock states until stabilized, along with warmed fluid resuscitation. It is antivenom that corrects the root cause of coagulopathy in pit viper envenomation (not administration of fresh frozen plasma, albumin, or platelets), and nothing more than saline in an appropriate amount should be necessary to manage hypotension resulting from the hypovolemia caused by envenomation [6]. If the practitioner is concerned that the patient's hypotension is not resolving by administering antivenom and saline alone, consultation with a critical care specialist and/or snakebite expert should be immediately sought.
Russell observes that [11]:
Hypovolemic shock may be further complicated by hemolysis, with a decrease in red cell mass, and renal and pulmonary failure. This may occur early or as late as three or four days after the envenomation. It should be kept in mind that blood fluid loss can be rapid and extensive; an increase of just over 2 centimeters in circumference of a thigh may represent the extravasation of half the total blood volume. In shock states, antivenom should always be given to prevent the shock from worsening and to inhibit those changes that contribute to shock.
As noted, IV fluid resuscitation with normal saline (at least 20 mL/kg in both children and adults) is usually adequate for the treatment of hypotension in patients with snakebite envenomation. It is advisable to give IV fluids cautiously, monitoring cardiac function and pulmonary signs so as not to exacerbate peripheral edema or precipitate pulmonary edema, especially in the treatment of severe envenomation. Vasopressors should be used only as short-term agents for the treatment of shock and only after hypovolemia as the likely cause of shock has been considered and addressed [12].
Acute renal failure can probably be prevented, and this should be a priority early in the treatment of snakebite. Given the importance of the kidneys and the threat that rhabdomyolysis poses to the health of snakebite victims, Roberts recommends that if the urine is discolored (by hemoglobin or myoglobin), attempts should be made to ensure a urine output of at least 1.5 mL/kg/hour in adults. In children older than 5 years of age, 30–50 mL/hour is suggested; for children 2 to 5 years of age, 20–30 mL/hour; and for children younger than 2 years of age, 10–20 mL/hour [10]. This urine output may be accomplished with IV fluids if the cardiovascular system can handle the fluid load, or with the use of osmotic diuretics such as mannitol.
Placement of a urinary catheter should always be accompanied by consideration of benefits versus risks. Risk of infection and the inability to fend off infection, especially in the elderly, is a well-known and increasingly appreciated concern. However, most patients treated for envenomation are young with healthy immune systems, and the catheter is needed only for a limited time.
Risk of significant bleeding as a result of urinary catheter insertion is not of great concern in patients envenomed by pit vipers. Hematuria due to coagulopathy does not preclude urinary catheter insertion. Some risk of bleeding obviously exists; however, for patients with severe envenomation and those who are unable to void every one to two hours, the benefits of catheterization include:
Continuous monitoring of fluid balance, which is required to maintain vital signs and to prevent fluid overload
Facilitation of fluid management of myoglobinuria and prevention of renal failure, by real-time monitoring of urinary output and color and the presence of blood and/or myoglobin
Timely detection of hematuria and/or myoglobinuria triggering immediate further laboratory analyses, which leads to early treatment with antivenom, a core component of proper management of pit viper envenomation
Van Mierop points out that adequate blood should be promptly obtained from victims of large pit viper bites for typing and cross-matching, as these procedures may be difficult or impossible to do later due to the effects of the venom [15].
With early or even delayed administration of antivenom given in adequate amounts, the need to administer blood products is unlikely to arise. There are certainly cases that require this level of treatment, but thorough understanding and management of pit viper envenomation should cause the numbers to be limited. Consultation with a snakebite expert should be sought if blood administration appears necessary.
According to Burgess and Dart (following section reprinted from Burgess JL, Dart RC. Snake venom coagulopathy: use and abuse of blood products in the treatment of pit viper envenomation. Ann Emerg Med. 1991;20(7):795-801, with permission from Elsevier) [42]:
Blood products are often given in an effort to reverse these coagulopathies, but in many cases they are given without a complete knowledge of the indications for transfusion or the risks of blood product transfusion. The hemolytic, non-hemolytic, and infectious risks of blood product transfusion reactions are significant. Antivenom has proven effective and reasonably safe in the treatment of rattlesnake bites; therefore, antivenom should be the initial treatment for snake venom coagulopathy. Blood products are indicated only if antivenom is not efficacious, for active bleeding, or for specific coagulation abnormalities.
Antivenom should be the primary treatment for snake venom coagulopathy unless the patient is unstable or actively bleeding. Minor bleeding, such as hematuria or oozing from gums, can initially be treated with antivenom alone. Antivenom has repeatedly been shown to reverse coagulopathies [often within an hour of an adequate dose of antivenom]. If the patient is unstable or actively bleeding or if antivenom is not effective, blood component transfusion is suggested. Because snake venoms vary widely in their effects on the coagulation system, blood product transfusion should be based on the specific coagulation abnormality.
The National Institutes of Health states that fresh frozen plasma transfusion is generally indicated for multiple coagulation protein deficiencies in the uncontrollably bleeding patient or as a replacement for isolated factor deficiencies [43]. Platelets are indicated for thrombocytopenia or abnormalities of platelet function with significant bleeding. Platelets are not indicated if thrombocytopenia is the sole abnormality and the platelet count is more than 20,000/mcL or for a bleeding time less than twice the upper limit of normal unless there are other abnormalities. Prophylactic platelet administration may be considered for counts less than 20,000/mcL, although there is no consensus on this topic. Note that platelets typically rebound in about one hour after an adequate dose of antivenom [44].
Uncomplicated hypofibrinogenemia should not be treated with blood products. Red blood cell transfusion is generally indicated for a patient with hemoglobin less than 7 mg/dL and should be considered in patients with a precipitous drop in hemoglobin. The routine transfusion of a stable patient for a hemoglobin level between 7 mg/dL and 10 mg/dL is not recommended [46]. Finally, Huestis and Bove recommend that, "hypofibrinogenemia should be treated with cryoprecipitate only for a level less than 100 mg/dL and clinically significant hemorrhage" [45].
Use of the following standard for treatment of snakebite by medical personnel presupposes that the staff is familiar with standard methods of treatment of emergency patients presenting with a wide variety of problems. Management of envenomation can require clinical skill equal to that required to treat a complex poisoning—it is not a simple process. While the following will greatly assist in the application of the standard, consultation with those who regularly treat snakebites is prudent when faced with a problem that is as complex and dangerous as envenomation.
In the prehospital setting, emergency responders should remain calm and reassure the patient continually. Death from snakebite is very rare. If the patient is stable and the hospital is close, avoid lights and sirens, as less excitement keeps capillary pressure (and thus the pressure gradient) lower, resulting in slower absorption of the venom. The patient should be instructed to avoid any unnecessary activity and movement.
If possible, the snake should be photographed and identified, but the animal should only be transported if its identity is in question and if it is safe to do so. Reflexes persist even after beheading, so all snakes should be treated as if alive.
Jewelry should be removed, but any constricting band or tourniquet should remain until removal is instructed by a physician's order. The extremity should be lightly splinted/immobilized after marking and timing the leading edge of swelling or tenderness. The extremity should be positioned slightly below heart level. If a suction pump device (e.g., the Sawyer Venom Extractor) is used, it should be limited to one minute to prevent further tissue damage.
If pain is severe, morphine sulfate should be administered (slowly, to avoid nausea), unless the patient has a history of allergy. One large-bore IV should be started away from the bitten extremity with warmed normal saline (as cold fluids can contribute to shock) administered and titrated to keep the patient's blood pressure within normal limits and pulse less than about 110–120 beats per minute in an adult; loss of vascular fluid volume should be considered. If circumstances allow for perfect sterile technique, start an IV lock away from the bitten extremity. Blood should be drawn using a large clot tube first, followed by tubes of all colors. All tubes should be fully labeled and signed, and a blood bank form labeled "Snakebite - Hold clot tube," signed by the phlebotomist, should be completed on arrival at the hospital and delivered to the blood bank along with the clot tube.
The time of the bite and the onset of symptoms should be recorded. The patient's vital signs should be documented, and the leading edge of swelling or tenderness should be marked (including the time) at least every 15 minutes. The patient should be observed for shock, pain, swelling, weakness/faintness, numbness/tingling, taste changes, fasciculations (potentially subtle and often found around the lips, face, upper back, or bite site), nausea, vomiting, diarrhea, diaphoresis, dysphagia, redness around the site of the bite, bruising, bleeding from the wound or elsewhere, hypotension, tachycardia (i.e., greater than 130 beats per minute), progression of signs and symptoms, and bloody-appearing urine, stool, or emesis. The need for intubation and the possibility of anaphylaxis should be considered if the patient is in shock.
For all patients with known or suspected snakebite, assessment is based on laboratory values and progression, if any, of signs and symptoms. It is important to remember that even asymptomatic patients should be studied for 8 to 12 hours from the time of the bite [13].
If the patient arrives from the field wearing a band or tourniquet applied to sequester venom, do not release the pressure all at once, but gradually, so as to prevent a bolus of venom from being released into the systemic circulation. When antivenom is available, apply a less constricting band proximal to the tourniquet, and after the IV solution has been infusing for five minutes, slowly release the original tourniquet, and in another three minutes remove the secondary band as well [11]. Consideration should be given to administering antivenom before the tourniquet or constricting band is removed, as a large (and possibly fatal) bolus of venom could be released [38]. Elevate the extremity after an IV is established and antivenom is available. Do not apply ice, heat, or tourniquets. Cleanse the puncture wounds and administer tetanus prophylaxis, if indicated.
If possible, determine whether the offending snake is venomous, non-venomous, or unknown. Notify a snakebite specialist and report to poison control. If likely to administer CroFab, note on the snakebite triage sheet (Figure 3) any allergy to sheep protein, papain, chymopapain, other papaya extracts, the pineapple enzyme bromelain, dust mites, latex, and any additional allergies [4]. If likely to administer Avavip, note any allergy to horse protein as well.
The patient should be kept NPO and on bed rest, with vital signs monitored every 15 minutes (including pulse oximetry). An IV lock should be placed in an unaffected extremity. Blood should be immediately collected and held for a possible type and crossmatch, followed by collection for a complete blood count (lavender top), coagulation studies (blue top), chemistry determinations (light green top), serology (red top), and FDP (another blue top or special top, depending on the laboratory) (Figure 4). A Foley catheter with urometer should be placed if the patient is unable to void every one to two hours or if menstruating.
A blood band should be applied, and the blood requisition is labeled "Snakebite: Hold clot tube." The signed blood sample and requisition are sent to the blood bank, but type and screen or crossmatch should be performed only if it becomes necessary. Differential, creatine kinase (CK), and ethanol (if indicated) tests should also be included in the initial laboratory assessment. The following tests should be performed immediately and at least every six hours thereafter:
Hemoglobin and hematocrit
Platelets
Fibrinogen
FDPs (Once positive, do not repeat unless ordered by physician.)
PT/international normalized ratio (INR)
PTT
Chemistry panel
Urinalysis
Urine myoglobin (Must be a fresh specimen. Once positive, do not repeat unless ordered by physician.)
All stools should be assessed for occult blood, and a fresh specimen of urine should be evaluated hourly for the presence of blood until the time of discharge from the hospital.
Patients with known or suspected snakebite having potential for envenomation should be observed every 15 minutes for:
Signs of shock
Pain (0–10 scale; may be 0 in envenomed patients with neuropathy)
Swelling/tenderness (Mark and time the leading edge plus measure and time the circumference at 10 cm and 20 cm proximal to the bite. May be absent in patients who have been severely envenomed.)
Weakness or faintness
Numbness or tingling (e.g., of the face, scalp, tongue, lips, fingers, toes, site of the bite)
Change in taste (e.g. a metallic, rubbery, or minty taste in the mouth indicative of rattlesnake envenomation)
Fasciculations (May be generalized or very subtle and most noticeable on the face, over the muscles of the back and neck, and in the bitten extremity)
Nausea/vomiting/diarrhea
Diaphoresis
Dysphagia
Erythema
Ecchymosis (where skin rubs against skin or where slight injury occurs)
Bleeding
Transient myosis
Any progression of signs or symptoms of envenomation or any abnormal laboratory values, including if the laboratory or blood bank reports that the patient's blood will not clot, should be immediately reported to the treating physician. All asymptomatic patients with known or suspected viper bites should be studied closely in the emergency department for 8 to 12 hours from the time of the bite before considering discharge [13].
Any patient with signs, symptoms, or abnormal laboratory values consistent with envenomation should be admitted to an ICU for at least 24 hours. Otherwise, after a minimum of eight hours of observation and repeated laboratory testing, patients who have had no paresthesias, fasciculations, erythema, ecchymosis, pain other than minor pain, swelling, or other signs or symptoms of envenomation, and no relevant laboratory abnormalities at baseline or six-hour follow-up, may be discharged to outpatient care [86]. Note: "Detection of fibrin split [degradation] products may be an early sign of a hematologic venom effect, and is a sensitive predictor of subsequent coagulopathy. In prospective studies, the presence of fibrin split products within the first 12 hours of observation or treatment predicted subsequent hypofibrinogenemia with 87% sensitivity and 69% specificity" [85]. At discharge, patient education should include information on reporting changes in signs/symptoms and avoiding anticoagulants until cleared by a physician (Figure 5). Patients with a prosthetic heart valve should have a plan (with their cardiologist) for restarting their anticoagulation regimen.
All patients with any sign or symptom of envenomation should be admitted to an ICU for at least 24 hours.
As part of the initial evaluation, steps should be taken to try to determine whether the offending snake is venomous or unknown. If the patient arrives wearing a band or tourniquet applied to diminish blood flow to and from the affected extremity, this should be managed/released in a step-wise fashion, as outlined previously, once antivenom is available. Consideration should be given to administering antivenom before the tourniquet or constricting band is removed entirely, as a large (and possibly fatal) bolus of venom could be released [38].
All jewelry should be removed. Bites to the face and neck are likely to produce a degree of soft tissue swelling that poses a threat to the patient's airway, and steps should be taken to assure that swift intubation can be performed if the need arises. If the bite is on an extremity, it should be elevated after an IV is established and antivenom is available. Do not apply ice, heat, electric shock, or tourniquets. Tetanus prophylaxis is administered at this point, if indicated. A snakebite specialist and poison control center should be notified [4].
If CroFab is used, note any allergy to sheep protein, papain, chymopapain, other papaya extracts, the pineapple enzyme bromelain, dust mites, or latex. If Anavip is used, document allergy to horse protein. Any additional allergies should also be noted on the triage sheet [4].
All patients with any signs/symptoms of envenomation should be admitted to an ICU. These patients are kept NPO and on strict bed rest initially, with vital signs taken every 15 minutes (including pulse oximetry); automated blood pressure measurements should be avoided. Morphine sulfate may be administered as needed for pain if the patient has no history of allergy. Infection is rarely an issue in the early management of snakebite, and prophylactic antimicrobials are not indicated. If there is uncertainty about the diagnosis or if objective signs (e.g., purulent discharge, fever) develop as a late complication of tissue breakdown, antimicrobial therapy may be indicated, guided by cultures [12].
It is helpful to have two IV lines available, away from the affected extremity—one IV lock for serial blood draws and one large-bore line for IV fluid and medication administration. The same blood draws should be conducted as those outlined for asymptomatic patients. Any swelling or tenderness should be marked and monitored for progression every 15 minutes. Intake and output should be monitored along with the presence of blood in urine or stools; a Foley catheter with urometer is typically placed in patients who are significantly envenomed to allow close monitoring of fluid balance and kidney function.
The affected extremity should be lightly immobilized in a position of function on a well-padded splint and elevated after antivenom is available. Any fang wounds should be cleaned daily with an antiseptic solution and covered with a dry, sterile dressing.
Blebs, vesicles, and superficial necrotic tissue should be aseptically debrided on the fourth or fifth day, per local protocol, if coagulation values are normal. Active and passive rehabilitation therapy should be initiated as soon as the patient is able to tolerate it to prevent contractures, and the patient with significant injury should be referred to rehabilitation therapy at discharge.
Serial electrocardiogram monitoring is recommended during the first 48 hours for all severe envenomations and for all patients older than 40 years of age or with a history of heart disease [40]. For patients with prosthetic heart valves, a cardiology specialist should be consulted.
A blood band should be applied, and the signed blood requisition is labeled "Snakebite: Hold clot tube." The blood and requisition are sent to the blood bank, but type and screen or cross should be ordered only if it becomes necessary. Differential, CK, and ethanol (if indicated) tests should be included in initial laboratory studies. CK may be repeated as necessary to assess muscle breakdown in the presence of generalized fasciculations. Hemoglobin/hematocrit with platelets and fibrinogen should be assessed every hour until the patient is stable. Urine dipstick for blood should be done every hour until discharge from the hospital. The following tests should be done as soon as possible and every six hours thereafter (or every four hours in severe cases) regardless of antivenom administration:
Complete blood count
Fibrinogen
Platelets
FDPs (once positive, do not repeat unless ordered by physician)
PT/INR
PTT
Chemistry panel
Urinalysis
Urine myoglobin (Must be a fresh specimen. Once positive, do not repeat unless ordered by physician.)
These tests should be coordinated with the following, which must be done one hour after each dose of antivenom has infused to track progression:
Hemoglobin/hematocrit
Platelets
Fibrinogen
FDPs (repeat not necessary after positive unless ordered by physician)
PT/INR
PTT
Urinalysis
Urine myoglobin (Must be a fresh specimen. Once positive, do not repeat unless ordered by physician.)
Patients with signs of envenomation should be observed every 15 minutes for:
Signs of shock
Pain (0–10 scale; may be 0 in envenomed patients with neuropathy)
Swelling/tenderness (Mark and time the leading edge of swelling/tenderness plus measure and time the circumference at 10 cm and 20 cm proximal to the bite. May be absent in patients who have been severely envenomed.)
Weakness or faintness
Numbness or tingling (e.g., of the face, scalp, tongue, lips, fingers, toes, site of the bite)
Change in taste (e.g., a metallic, rubbery, or minty taste in the mouth indicative of rattlesnake envenomation)
Fasciculations (May be generalized or very subtle and most noticeable on the face, over the muscles of the back and neck, and in the bitten extremity)
Nausea/vomiting/diarrhea
Diaphoresis
Dysphagia
Erythema
Ecchymosis (where skin rubs against skin or where slight injury occurs)
Bleeding
Transient myosis
Any progressive signs or symptoms of envenomation or any deteriorating laboratory values, including if the laboratory or blood bank reports that the patient's blood will not clot, should be immediately reported to the treating physician.
It is important not to wait hours before treating patients with antivenom, because local tissue is preserved only by early treatment (Figure 6). If one is unsure of whether to treat or re-treat, a snakebite specialist should be consulted.
Antivenom treatment should be initiated in the presence of any of the following signs or symptoms [86]:
Frank bleeding
Shock
Rapid swelling beyond the area of the bite
Significant swelling (i.e., progresses at a rate of 1 cm per hour, whether linearly or circumferentially) that fails to localize within 30 to 60 minutes from the time of the bite
Ecchymosis progressing beyond the bite site
Any degree of true neurotoxicity, including localized fasciculations or myokymia (with the possible exception of bites by the south-ern Pacific rattlesnake unless in combination with other significant signs or symptoms)
Single-component coagulopathy (one of the following):
Low fibrinogen (less than 50 mg/dL)
Fibrinogen trending significantly downward (consider history of familial afibrinogenemia)
Low platelets (less than 25,000/mcL)
Platelets trending significantly downward (consider history of taking anticoagulants or thrombocytopenia), INR greater than 3.0, or PTT greater than 50 seconds
PTT or INR trending significantly upward
Low hemoglobin/hematocrit or hemoglobin/hematocrit trending significantly downward (consider history of anemia)
Multicomponent coagulopathy (same as single-component coagulopathy except fibrinogen less than 75 mg/dL and platelet count less than 50,000/mcL)
Antivenom treatment should be strongly considered in the presence of the following signs or symptoms:
Definite change in taste
Paresthesias
Progressive pain
Envenomation of the finger or hand is treated more aggressively than most other areas of the body for the following reasons [3]:
The venom has less volume in which to immediately distribute, resulting in greater and earlier local tissue damage.
The structure of the hand and fingers is intricate and more subject to incomplete healing after snakebite if treatment is delayed or the dose of antivenom is inadequate.
To not have full function of the hand may keep a patient from adequately performing his or her chosen occupation.
Progression of circumferential or linear (leading edge) swelling measuring 1 cm/hour is considered to be significant, regardless of the site of the bite. Without early and adequate treatment, swelling and healing can take weeks to return to a level of maximum improvement. With early and adequate treatment, swelling can be expected to subside in approximately 10 days [4]. It is therefore important to not wait hours to see how far swelling progresses before beginning antivenom administration, because further and perhaps irreversible tissue destruction may well be occurring during that time. Swelling can occur haltingly as the venom dissolves different types and layers of tissue and can be thought to have stopped, only to become obvious again. One should not wait for multiple episodes of such a pattern to pass before beginning treatment.
The patient who has a hand or finger envenomation and whose life is not yet known to be in danger faces the decision of whether or not to accept early treatment. A clear, unhurried explanation to the patient by someone comfortable with the treatment of snakebite is essential if he or she is to make an appropriate decision. The basic conversation should include the following [4]:
The purpose of antivenom administration (i.e., to stop the progression of symptoms and signs)
The importance of early antivenom administration to best preserve tissue
The inability to predict the extent of damage without treatment
The estimated time to resolve swelling and to achieve healing (often weeks) in the absence of treatment
The possibility of antivenom, like any medication, precipitating a life-threatening allergic reaction
The limited option of a "wait and see" approach, with tissue and possibly organ damage and life-threatening internal bleeding being the cost of delayed treatment
The possibility of "serum sickness," which is preventable and treatable, occurring days or several weeks from the time of treatment
Early in the course of treatment, the patient should be evaluated for the need of forced diuresis (i.e., hematuria, positive myoglobin). The extremity should be elevated after an IV is established and antivenom is available. A crash cart should be available in the room, and the patient should not be left unattended. Nasogastric tubes, arterial punctures, anticoagulants (e.g., aspirin, aspirin-containing drugs, NSAIDs), ice or heat application, and tourniquets should be avoided. Exercise should be avoided initially. Prior to administering antivenom, the patient should be fully informed of potential risks and benefits and should complete a consent form specific to the type of antivenom(s) to be used. As noted, unless the treating physician has experience in the administration of any pit viper antivenom, consultation with a snakebite specialist or clinical toxicologist is advisable—and available through the regional poison control center (1-800-222-1222)—before administering antivenom.
Each vial of CroFab should be reconstituted with 18 mL of 0.9% saline drawn from an IV bag of normal saline containing the volume of fluid the patient is to receive over the next hour (minimum: 250 mL) [4]. The diluent should be instilled gently along the wall of each vial to avoid foaming. To hasten mixing, release pressure after all diluent is added. Each vial of CroFab should be mixed by continuous, gentle manual inversion at a rate no faster than two complete inversions per second, until no solid material is visible in the vial; to prevent foaming, it should not be shaken. The reconstituted antivenom is then returned to the IV bag.
Reconstitution of Anavip is similar to that of CroFab, except 10 mL of 0.9% saline drawn from the IV bag is added to each vial rather than 18 mL, and gentle swirling of each vial is employed rather than inversions.
With the physician at the bedside, pit viper antivenom should initially be infused slowly for 10 minutes (at a pump setting of 30 cc/hour with a limit of 5 cc), observing for frank anaphylactic shock, hypotension, urticaria, sneezing, dyspnea, apprehension, flushing, coughing, cyanosis, vomiting, wheals, pruritus, wheezing, and/or edema of the face, tongue, or throat.
Patients with more apparent signs and symptoms or more severe coagulopathy should receive antivenom over one hour if possible; others may benefit from a somewhat slower infusion, because the rate of infusion is directly linked to the likelihood of adverse reactions. Intravascular volume depletion, which can lead to hypotension and shock, is common in patients with a significant envenomation (especially children), and nearly all patients should receive generous resuscitation with warmed isotonic crystalloid fluids [89]. In volume-depleted children, the usual fluid bolus is 20 mL/kg (isotonic crystalloid), which can be repeated two times for a total of 60 mL/kg [90,91]. A 250 mL bolus of CroFab or Anavip, although not exclusively crystalloid, would be expected to be tolerated by most pediatric patients [4].
There are no studies examining the use of antivenom in volumes less than 250 mL. Because the rate of administration is tied to allergic responses, a more concentrated solution of antivenom should be run at a slower rate [4]. If the patient has known myocardial dysfunction or other reason for fluid restriction, the decision to reduce the total administration volume of antivenom should be left up to the treating physician [92]. After the test dose, regardless of dosage to be administered and in the absence of hypersensitivity symptoms, the rate of infusion should be doubled every three minutes to an end point that allows the total volume to infuse over one hour.
Severe Envenomation
Severe envenomation is characterized by swelling, erythema, or ecchymosis possibly involving the entire extremity and spreading rapidly; systemic manifestations are pronounced and may include nausea and vomiting, changes in mental status, severe tachycardia or bradycardia, tachypnea, respiratory distress, and hypotension. Coagulation defects are usually present; serious or spontaneous bleeding may occur. Platelet counts are often less than 20,000/mcL, and other laboratory values are abnormal [12]. Fasciculations, strong taste changes, weakness or faintness, transient myosis, and/or shock may be present. Generally (with the possible exception of bites by the copperhead), the less time between the bite and the onset of severe symptoms, the greater the venom load [31].
Fluid balance is a critical element in the treatment of severe envenomation. Andrews summarizes, "A severe snakebite should be treated and followed closely with fluid balance as in a very severe 50% third-degree burn, with careful attention to blood pressure, pulse, respiration, urine output, and hematocrit" [49].
Regardless of patient age, in severe envenomation CroFab is initially administered approximately each hour in increments of 10 vial doses to gain control (rather than the routine 6 vial doses discussed later) until initial control is achieved. Consultation should be sought early with severe envenomation, particularly if initial control is not achieved by the administration of two 10-vial doses of CroFab.
Rather than the routine CroFab maintenance dose of two vials every six hours for three doses, consider administering two- to four-vial doses every four to six hours for more than 24 hours [6]. Keep in mind this is a titration of antivenom to laboratory values and to signs and symptoms of envenomation, and it is safer to slightly overtreat with antivenom than to undertreat.
With severe envenomation, Anavip is administered approximately each hour in increments of 20 vials rather than the routine 10 until initial control is achieved. This dosage is used for all patients, regardless of age. After Consultation should be sought if initial control is not achieved by the administration of two 20-vial doses of Anavip.
For the patient with signs/symptoms of shock, infuse warmed normal saline (at least 20 mL/kg), and administer 10 vials of CroFab or 20 vials of Anavip. Consider intubation/oxygenation and patient positioning. Immediate consultation should be sought. Be prepared to administer additional doses (10 vials of Crofab or 20 vials of Anavip) before initial control is achieved. A vasopressor may be added, but not in lieu of an adequate amount of saline. Packed red cells may be added, if needed.
If the patient is hypotensive and fails to respond to resuscitative efforts, immediate consultation should be sought. Do not administer albumin or plasma expanders, as these can confuse the coagulation abnormality by creating one of their own [6].
Lesser Envenomation
The recommended initial antivenom dose for apparently lesser envenomations is 6 vials of CroFab or 10 vials of Anavip [1,97]. If initial control is not achieved by the first dose (approximately one-half of all cases), additional doses of 6 vials CroFab or 10 vials Anavip should be repeated approximately hourly until initial control of the envenomation syndrome has been achieved [87]. Consultation should be sought if initial control is not achieved by the administration of two 6-vial doses of CroFab or two 10-vial doses of Anavip [86]. Based on clinical trial data, after initial control has been established, additional two-vial doses of CroFab should be administered every 6 hours for 18 hours (three doses) to lessen the incidence of local and coagulopathy recurrence [9]. The manufacturer of Anavip does not recommend routine maintenance dosing but recommends administering four additional vials as needed to halt progression.
"Initial control" may be defined as complete arrest of local manifestations and return of coagulation tests and systemic signs toward normal in non-bleeding patients [6]. In general, one can judge achievement of initial control and adequacy of dosing on the basis of a combination of clinical and laboratory parameters, including a halt to progression of swelling; improvement in systemic symptoms, such as nausea, paresthesias, and fasciculations; restoration and stability of vital signs (e.g., blood pressure, pulse); and degree of reversal of coagulopathy. Laboratory studies performed one hour after infusion of each dose of antivenom are required and will help to define whether initial control has been achieved.
Further treatment following antivenom administration depends on the clinical course of the patient. Generally, two- to four-vial doses of CroFab or fourto eight-vial doses of Anavip are administered as deemed necessary based on laboratory values and symptomatology [9]. It is important to remember that the effective half-life of the antivenom is likely to be much less than that of the venom, which can be many days. The half-life of CroFab is 12 to 23 hours, and the half-life of Anavip is 133 hours, due to its larger molecular mass.
After swelling appears to have ceased and coagulation studies have normalized or are trending substantially toward normal, one should continue to measure and mark the zone of local swelling every 30 minutes to 2 hours for the next 48 hours. Importantly, recurrence or delayed-onset of one or more venom effects occurs in up to approximately half of patients treated with CroFab antivenom [87]. Recurrence can also occur after treatment with Anavip. Local tissue recurrence typically develops within 6 to 36 hours of initial control. The onset of recurrent or delayed-onset hematologic venom effects typically occurs 2 to 7 days after initial control, with some cases up to 10 days after initial control [88].
It is important to monitor all symptoms and signs on the same schedule as for the monitoring of swelling. The Foley catheter and regular urine dipstick testing for blood should continue until the patient is close to being discharged. It is important not to discontinue the catheter and dipsticks prematurely, as they are important visual monitoring tools that warn of recurrent coagulopathy (hematuria) and/or myoglobinuria. Serial laboratory studies every 6 to 12 hours should be continued until the patient is discharged, with close observation for downward trends.
Serum Sickness
Patients who receive antivenom therapy should be followed expectantly for serum sickness, a reaction that can occur 4 to 20 days after initial exposure to antivenom. If serum sickness develops in an adult patient, it can be treated with 10 mg of oral prednisone every six hours at the onset of pruritus and urticaria [18]. In children, prednisone may be administered at a dosage of 2 mg/kg/day in two divided doses [18,28]. For prophylaxis against serum sickness, consider prescribing a two-week supply of methylprednisolone at the time of discharge [29]. In children, the use of hydroxyzine 1 mg/kg every four to six hours, as needed, is helpful for sedation and control of pruritus [28].
The following section was written with significant contributions and guidance from Dr. Rand Malone, allergist and Associate Professor of Pediatrics at Florida State University College of Medicine.
CroFab and Anavip are prepared using sheep and horse serum, respectively, both of which include foreign protein and either of which could trigger an allergic response; however, the incidence of reactions is very low [23]. If possible, one should avoid using the potentially triggering antivenom in patients with known horse or sheep protein allergy. Untoward reactions, including wheezing, urticaria, and anaphylaxis, may be an indication of too rapid an infusion of antivenom. Treatment is not stopped for rash or itching but is held until the reaction subsides, usually about 30 minutes following adequate treatment [54]. If anaphylaxis occurs, the question becomes whether one can risk not treating with antivenom. Regardless, consultation should be sought under this circumstance.
If an allergic reaction occurs, hold the antivenom infusion and, if indicated, administer epinephrine, methylprednisolone (to prevent worsening or rebound of anaphylaxis over the next few hours), and diphenhydramine and cimetidine (for skin reactions or urticaria) [54].
Epinephrine is the drug of choice for anaphylaxis and should be administered as first-line therapy. Epinephrine may rarely be used prophylactically in patients known to be acutely sensitive to antivenom or its components and who have a life-threatening envenomation.
The actions of this agent address the pathophysiologic changes that occur in anaphylaxis better than any other single drug. Failure to administer epinephrine in the course of treatment has been repeatedly implicated in anaphylaxis fatalities [58,59,60,61,62,63,64]. The use of antihistamines is the most common reason reported for not using epinephrine [65]. The therapeutic actions of epinephrine include the following [66]:
Increased vasoconstriction, increased peripheral vascular resistance, and decreased mucosal edema via α1-adrenergic agonist receptor effects
Increased inotropy and increased chronotropy via β1-adrenergic receptor agonist effects
Bronchodilation and decreased release of mediators of inflammation from mast cells and basophils via β2-adrenergic receptor agonist effects
Epinephrine has a narrow toxic-therapeutic index (risk-to-benefit ratio). In therapeutic doses and by any route, epinephrine frequently causes mild transient adverse effects in individuals of all ages. These include anxiety, fear, restlessness, headache, dizziness, palpitations, pallor, and tremor. Rarely, epinephrine may lead to ventricular arrhythmias, angina, myocardial infarction, pulmonary edema, sudden sharp increase in blood pressure, and intracranial hemorrhage [66]. These severe adverse effects are more likely to occur when a dilution error has occurred in preparation of the dose to be administered, as for example, after an IV bolus injection of a 1:1,000 epinephrine solution instead of the proper 1:10,000 epinephrine solution [67].
Epinephrine has an onset of action within minutes, but it is rapidly metabolized. Therefore, the effect is often short-lived and repeated doses may be necessary [65,68,69]. Repeat doses should be administered within 5 to 15 minutes, if required.
Epinephrine can be delivered through a variety of routes, including intramuscular (IM), intravenous (IV), and endotracheal or intraosseous routes [70]. IM epinephrine is recommended over subcutaneous injection because it provides a more rapid increase in plasma and tissue concentrations of epinephrine [70,71,72]. The IM dose should be given in the anterolateral thigh, in the vastus lateralis muscle. The needle used should be of adequate length to reach the muscle beneath the subcutaneous adipose tissue. IM injection into the thigh may be impossible in overweight or obese individuals, especially those with thicker subcutaneous fat tissue in this area, in which case the deltoid muscle should be used[73,74].
Most patients suffering from anaphylaxis respond to IM epinephrine (possibly repeated doses) if promptly administered and if the injection site is well massaged. However, consideration should be given to the possibility of poor perfusion affecting the ability to absorb epinephrine administered intramuscularly.
IV epinephrine may be used for anaphylaxis, but this route should be reserved for severe, refractory cases characterized by imminent or actual airway obstruction or hypotension that is unresponsive to IM epinephrine (including repeated doses) and vigorous fluid resuscitation. The data regarding IV epinephrine for anaphylaxis are anecdotal, but poor outcomes from excessively vigorous IV epinephrine dosages have led to cautious recommendations. When epinephrine is infused slowly, beta-receptor effects predominate, while alpha effects predominate with rapid rates of infusion. In addition, beta-receptors respond to lower doses of epinephrine than alpha-receptors. Therefore, a low, slow dose of epinephrine favors beta effects while minimizing undesirable increases in systolic blood pressure. For patients with anaphylaxis who are in extremis, some experts recommend infusing 1 cc of 1:10,000 epinephrine (i.e., 1 cc of the "crash cart" 10-cc vial) further diluted in 10 cc of normal saline slowly over 5 to 10 minutes. An epinephrine drip can be administered as well, titrated to effect [34].
Although epinephrine is the first-line agent for anaphylaxis, there are relative contraindications to its use. When administered intravenously, epinephrine should be given with extreme caution to patients older than 35 years of age, those with underlying cardiovascular disease, pregnant women, and patients using beta-blockers. Malignant hypertension, myocardial infarction, and fetal demise have all been reported [34,35].
Generally speaking, pediatric patients tolerate IV epinephrine better than adults. In elderly patients or those with cardiovascular disease, consider a test dose of epinephrine of 0.10–0.15 cc of 1:1000 administered subcutaneously [36].
For airway symptoms (e.g., cough, wheezing, stridor), cardiovascular compromise or collapse, or severe skin reactions in adults, administer epinephrine 1:1,000 0.3–0.5 mL IM in the vastus lateralis immediately and massage the injection site well. This may be repeated at 5- to 10-minute intervals up to three to five doses. If a patient has poor perfusion (i.e., systolic blood pressure <30–40 mm Hg) that precludes absorption of IM doses, proceed to IV administration when access is available. The maximum IM dose for elderly patients is 0.3 mL [54].
If the response to IM doses is inadequate and IV access is delayed, keep giving the age-appropriate maximum IM dose and massage the injection site [54]. If the patient does not respond, epinephrine 1:10,000 3–5 mL IV should be administered (over five minutes) immediately when IV access becomes available (maximum dose for the elderly: 3 mL) [54]. Further dilution in 10 cc normal saline for IV use is recommended [36]. This may be repeated at five-minute intervals if there is no improvement, or every three minutes in severe reactions.
The approach to treating antivenom-related anaphylaxis in children is similar. IM epinephrine (1:1,000) is administered immediately in the thigh at a dose of 0.01 mL/kg (maximum: 0.3 mL), and the injection site is massaged well. If the calculated dose is not helpful, the maximum dose should be given [54]. The dose may be repeated every 5 to 10 minutes for three to five doses. Doses should be given IM unless the patient has signs of poor perfusion (e.g., systolic blood pressure less than 30–40 mm Hg), regardless of existing IV or endotracheal access. If hemodynamic improvement is not achieved after three to five IM doses, or if circulatory perfusion remains poor, a 0.1 mL/kg dose of IV epinephrine 1:10,000 should be given immediately over five minutes. This is repeated at five-minute intervals if no improvement is noted, or every three minutes in severe reactions. Further dilution in 10 cc normal saline is recommended [36].
The patient being treated for anaphylaxis should be placed in a recumbent position (when tolerated), with the lower extremities elevated to maximize perfusion of vital organs. This also helps prevent empty ventricle syndrome, in which severe hypotension leads to inadequate cardiac filling and electrical cardiac activity without a pulse [83].
Massive fluid shifts can occur rapidly in anaphylaxis due to increased vascular permeability, with transfer of up to 35% of the intravascular volume into the extravascular space within minutes [82]. Any patient who does not respond promptly and completely to injected epinephrine should be assumed to have intravascular volume depletion causing persistent hypotension despite maximum vasoconstriction. Large-volume warmed fluid resuscitation should be initiated immediately in patients who present with orthostasis, hypotension, or incomplete response to IM epinephrine [81]. Adults may require 1–2 L normal saline administered at a rate of 5–10 mL/kg in the first five minutes. Children should receive up to 30 mL/kg in the first hour [98].
Oxygen should be administered initially to all patients experiencing anaphylaxis, especially those with evidence of hypoxia or respiratory distress. Supplemental oxygen helps not only with optimization of oxygen delivery and organ perfusion, but also with bronchodilation [81]. Delivery of oxygen should be by a non-rebreather mask or simple cannula. (Simple masks without a reservoir bag attached can suffocate patients.)
Steroids do not relieve stridor, shortness of breath, wheezing, GI symptoms, or shock; therefore, they should be considered adjunctive therapy and should not be substituted for epinephrine [54,58,59,63,75,76,77,78]. Corticosteroids are not helpful in the treatment of acute anaphylaxis due to their slow onset of action (four to six hours). These agents are often given because of their anti-inflammatory properties, which counter allergic and inflammatory reactions, and also because they may help prevent biphasic or protracted reactions, which occur in up to 20% of individuals [60,79]. If used, methylprednisolone is the recommended agent.
For adults, the recommended dose of methylprednisolone in conjunction with epinephrine is 60–80 mg IV over at least one minute [54]. This may be repeated every 2 to 6 hours until stable, but it usually should not be continued beyond 48 to 72 hours [55]. In life-threatening shock, massive IV doses of methylprednisolone as the sodium succinate (e.g., 30 mg/kg initially and repeated every four to six hours if needed, or 100–250 mg initially and repeated at 2- to 6-hour intervals as required) have been recommended [55].
In children 12 years of age and younger, the recommended methylprednisolone dose is 30 mg IV over at least one minute. In children older than 12 years of age, adult dosing should be used. In either case, treatment may be repeated every 2 to 6 hours until stable, but usually should not be continued beyond 48 to 72 hours [55].
Albuterol administration via nebulizer is recommended for bronchospasm, as opposed to metered-dose inhalers with spacers. Albuterol does not relieve airway edema (e.g., laryngeal edema) and should not be substituted for IM epinephrine dosing in the treatment of anaphylaxis [67].
According to Russell, "IV diphenhydramine can be given before antivenom is administered to manage itching and urticaria and may be of particular value when the patient requires large amounts of antivenom in a short time" [12]. Cimetidine should be considered as well. Diphenhydramine (an H1-receptor antagonist) works synergistically with cimetidine (an H2-receptor antagonist). Therefore, if diphenhydramine is required, consideration should be given to also administering cimetidine to maximally protect from itching and urticaria.
Adults may be administered IV diphenhydramine every 6 hours at a dose of 50–100 mg and as needed during antivenom administration, up to a maximum of 400 mg in 24 hours. It should be administered undiluted at a rate not to exceed 25 mg/minute [101].
For pediatric patients, IV diphenhydramine is given at a dose of 1 mg/kg every 4 to 6 hours and as needed, up to a maximum of 300 mg in 24 hours. It is given undiluted at a rate not to exceed 25 mg/minute [7,101].
Adults may be administered IV cimetidine 300 mg every six hours during antivenom administration. It is diluted in 20 mL normal saline and administered over at least five minutes to avoid negative cardiac effects. Consideration should be given to using greater dilution and administering over a longer period, if appropriate.
For children younger than 2 years of age, IV cimetidine is given at a dose of 5–7 mg/kg, diluted with normal saline to a total volume of 20 mL and injected over at least five minutes. Neonates require cimetidine to be preservative-free. For children 2 through 16 years of age, IV cimetidine is given at a dose of 300 mg diluted in 50 mL D5W administered over 15 minutes. Those 17 years of age and older receive the same dose as adults [7].
In all patients, consideration should be given to extending administration of diphenhydramine and cimetidine for two to three days after resolution of the untoward reaction.
Consider intravenously administered atropine for patients with bradycardia [67].
No published prospective studies exist on the optimal management of refractory anaphylactic shock. Repeated use of epinephrine, as well as IV fluids, corticosteroids, and vasopressor agents, may be needed [81]. Patients who have persistent hypotension despite the administration of epinephrine and IV fluids should receive vasopressor medications titrated to the desired effect of restoring blood pressure [67,80]. No compelling evidence exists to support one vasopressor over another in this clinical scenario [67].
Glucagon should be considered in patients who do not respond to epinephrine. Adults are administered an initial dose of 50 mcg/kg IV over one to two minutes. Higher doses (up to 10 mg) may be needed if the initial dose is ineffective. This may be followed by an infusion of 2–5 mg/hour (to a maximum 10 mg/hour) [100].
In pediatric patients weighing less than 20 kg, glucagon is administered at an initial dose of 20–30 mcg/kg IV (maximum: 1 mg) given over five minutes and followed by an infusion of 5–15 mcg/minute, titrated to clinical response [98].
Glucagon is known to cause nausea and emesis and increases the risk of aspiration. Placement of the patient in the lateral recumbent position will assist in protecting the airway in those who are not intubated [98].
Epinephrine is the first-line drug for treatment of anaphylaxis for all patients, regardless of whether pregnancy exists. A risk of its use in pregnancy is uterine hypoxia. In spite of its lower efficacy, Entman and Moise suggest that "ephedrine (25–50 mg IV bolus) should be the first-line therapy of anaphylaxis during pregnancy to bypass the uterine hypoxia that may result from epinephrine use" [10]. However, by using ephedrine rather than epinephrine, and thus not as quickly reversing maternal hypotension (which causes fetal hypotension and hypoxia), one risks missing the roughly 5- to 10-minute window to most effectively reverse the maternal hypotension that quickly endangers both the mother and the fetus.
The American Society of Hospital Pharmacists states that it is not known whether ephedrine can cause harm to the fetus [2]. The question is whether with anaphylaxis in pregnancy one has the luxury of time to see if a less-efficacious drug will reverse the life-threatening effects of maternal, and fetal, hypotension. Because anaphylaxis places both the patient and fetus at risk, one must weigh the risk of epinephrine-induced uterine hypoxia against the risk of not using epinephrine as the first-line drug in anaphylaxis. The severity and rate of progression of a reaction helps delineate this conundrum.
One path to successful treatment of the pregnant patient with anaphylaxis may be to use a lower dose of epinephrine as a bridge to the use of ephedrine by administering epinephrine 1:1,000 0.15–0.2 mg IM while staff is readying IV ephedrine. One could administer a second dose of epinephrine 1:1,000 0.2–0.5 mg IM as needed, while steadily considering the degree of hypotension, the lower efficacy of ephedrine, the timing of ephedrine availability, and the point at which ephedrine rather than epinephrine might be administered, if at all. If the reaction is more prolonged but is controllable with ephedrine, then IV ephedrine could be titrated as required while antivenom is slowly administered. This method provides timely initial treatment of anaphylaxis (within 5 to 10 minutes of onset) that is important to maternal and fetal survival and may lessen the overall threat to the fetus [54].
Patients who are on beta-blockers are particularly susceptible to extreme blood pressure responses, as the alpha-receptor remains the only site available for epinephrine binding [36]. This class of drugs may cause patients to be resistant to treatment with epinephrine, and they can develop refractory hypotension and bradycardia. Glucagon should be administered in this setting, because it has inotropic and chronotropic effects that are not mediated throughβ-receptors [56].
Re-starting antivenom should wait until after the reaction has subsided. In acutely sensitive patients, it may be necessary to reduce the concentration of the antivenom to half that of the original solution. Re-treatment should be started at the initial test rate of 30 cc/hour. If there are no signs of another untoward reaction after 10 minutes, the rate of administration should be increased at a slower pace than before to no faster than the original rate less about 25%. Frequently repeated doses of diphenhydramine may be needed. In rare cases, a titration of epinephrine may be required.
The value of the rate of administration of the antivenom cannot be overstated. According to the Arizona Poison and Drug Information Center, slowing the rate of administration (without any further dilution of the original solution) will solve the problem in some cases [3]. Malone adds [54]:
Administering antivenom at a slow rate delivers a small enough amount of antivenom that individual antibody binding sites can attach without as much cross-linking occurring. Usually, some mild urticaria or pruritus will occur, but not full-blown anaphylaxis. Delivering medications (e.g., antivenom) this way transiently desensitizes the individual who is allergic and allows them to receive the medication on that occasion.
At the slower rate, perhaps in combination with the single dose of diphenhydramine and cimetidine, many patients do well and need no further treatment as antivenom is continued [3].
It is impossible to predict how long one will need to remain hospitalized after pit viper envenomation due to the large number of variables involved, particularly the unknown venom load. If one is hospitalized, it is for a minimum of 24 hours, but stays frequently extend to several days or even longer than a week. The hospitalized patient who is discharged should show no evidence of paresthesia, fasciculations, erythema, ecchymosis, pain other than minor pain, swelling, worsening clotting factors, or other signs or symptoms of envenomation.
After treatment of pit viper envenomation is "complete," venom effects can persist for days or even weeks because venom deposited in tissue is hard for the antivenoms currently in use to penetrate, leaving deposits to leach out over time. The hospital stay is intended to get the patient well enough to be safely discharged. In reality, due to the nature of envenomation, a number of patients will show evidence of local and coagulopathy recurrence after discharge, sometimes calling for additional coagulation studies and re-treatment with antivenom. For this reason, office follow-up is generally required. A likely exception to the need for follow-up is the case of envenomation by a copperhead. The toxicity of copperhead venom is measurably less than that of rattlesnakes, for example, but one should always bear in mind that symptomatology and laboratory values guide treatment, not the size or type of pit viper.
While patients should be encouraged to gradually exercise the affected extremity as soon as they can, bedrest is recommended until coagulation studies show a sustained trend toward normal. A reasonable level of activity (beyond bedrest) may be considered after clinical and laboratory parameters have improved significantly and are judged to be stable. However, physical activity can sometimes result in a significant amount of residual venom being released, necessitating re-treatment with antivenom. It is obviously better to discover this while the patient is hospitalized than to lose time and tissue by discharging prematurely and having to re-admit for further treatment. Repeat laboratory work is required after activity is resumed in order to evaluate its effects, if any, on the coagulation cascade.
The patient's laboratory values as they relate to envenomation should be at least near normal and not worsening prior to discharge. Detection of FDPs may be an early sign of a hematologic venom effect and is a sensitive predictor of subsequent coagulopathy. In prospective studies, the presence of fibrin split products within the first 12 hours of treatment predicted subsequent hypofibrinogenemia with 87% sensitivity and 69% specificity [85]. At discharge, a written plan (Figure 5) for follow-up and further laboratory testing should be clearly explained and agreed to by the patient, emphasizing that recurrence may occur. As noted, patients with a prosthetic heart valve should prior to discharge establish a plan with their cardiologist for restarting their anticoagulation regimen.
According to the Rocky Mountain Poison Center, many patients (40%) with at least one abnormal test result, including platelet count, protime, fibrinogen, or fibrin split products, experienced either prolonged or recurrent coagulopathy [23]. Many physicians, however, do not monitor abnormal coagulation parameters to resolution (61%) or monitor it for sufficient periods after envenomation (47%) to adequately determine whether these venom effects exist. Therefore, many cases of coagulopathy resulting from snake venom remain unresolved despite antivenom administration and could pose a health risk to the patient [23].
Boyer et al. recommend that patients who have received antivenom should be re-evaluated at least once within five days after treatment [5]. If the results of laboratory testing remain normal, recurrence is unlikely. However, patients at risk for recurrence (i.e., with abnormal coagulation during the first 36 hours) should be reassessed every 48 hours after the last antivenom dose, until coagulation values are clearly stable or improving for several days [5].
If coagulation values become significantly abnormal on follow-up, or if there is a definite downward trend, then laboratory test results should be monitored at least daily and consideration should be given to re-treatment with antivenom. Until more data are available, re-treatment with FabAV should be considered in patients with any of the following [5]:
Fibrinogen concentration less than 50 mg/dL
Platelet count less than 25,000/mcL
INR greater than 3.0
PTT greater than 50 seconds
Multicomponent coagulopathy with abnormal laboratory values of a lesser degree
A clear worsening trend at follow-up in patients who had a severe early coagulopathy
High-risk behavior or comorbid conditions
Frank bleeding should be treated with additional antivenom, regardless of whether the laboratory criteria have been exceeded. Antivenom therapy is recommended when asymptomatic patients develop multicomponent coagulopathy that reaches critical values (e.g., INR greater than 3.0, a PTT greater than 50 seconds, fibrinogen concentration less than 75 mg/dL, platelet count less than 50,000/mcL) [20].
Besides the often-overlooked consideration of the need to re-evaluate clotting ability following discharge from the ICU, until recently there were no specific techniques recommended by which to measure actual or patient-perceived improvement following snakebite treatment. In 2015, Lavonas and Gerardo published a study examining methods for measuring outcome following envenomation by a copperhead [95]. They suggest three techniques for re-evaluating discharged patients who were envenomed by any type of pit viper: grip strength, ability to walk 25 feet (7.62 meters) unassisted, and global function. In this study, global function was assessed with the Patient-Specific Functional Scale. On the initial assessment, the patient is asked to identify up to three important activities that they are unable to do or are having difficulty with as a result of the snakebite. The patient then provides a rating for each item, on an 11-point ordinal scale ranging from 0 ("unable to perform activity") to 10 ("able to perform activity at the same level as before the injury or problem"). During reassessments, the subject is prompted to re-rate the same three activities [95].
Management of envenomation by pit vipers is a dynamic process that requires practice in order for nurses and physicians to become proficient in assessing and treating; however, useful practice is precluded for most because there are few envenomed patients upon whom to practice. This fact calls for the practitioner to recognize the importance of consulting a toxicologist at Poison Control or an expert in snakebite treatment who will guide them safely through the management process. Use of the information contained herein will go a long way toward creating a deeper understanding of the subject and toward proper clinical application at the bedside. Use of Assessment and Laboratory Flow Sheets (Figures 2 and 4) will help assure continuity of care, particularly during shift changes, and that critically important trends are promptly recognized. Regardless, consultation should always be sought unless one is intimately familiar with the management of these patients.
Avoid snakebite.
| American Association of Poison Control Centers |
| Regional Poison Control Center Information |
| (800) 222-1222 |
| https://aapcc.org |
| BTG (CroFab) 24/7 Medical Information for Treatment of Snakebites and Toxicologist Access |
| (877) 377-3784 |
| Rare Disease Therapeutics, Inc. (Anavip) 24/7 Medical Information for Treatment of Snakebites and Toxicologist Access |
| (615) 399-0700 |
| Edward Hall, MD |
| South Georgia Surgical Associates Archbold Hospital–Thomasville, Georgia |
| (229) 226-8881 (Office) |
| (800) 287-2554 (Office) |
| (800) 341-1009 (Hospital) |
| Jon E. Soskis, RN |
| (850) 539-1915 Phone |
| (850) 539-7340 Fax |
| (850) 567-0984 Cell |
| rsoskis@gmail.com |
Tetanus prophylaxis?
Notify a snakebite specialist and poison control center?
Stay tight on laboratory testing (every 4 to 6 hours, sometimes extended to every 12 hours after prolonged stability is achieved). You cannot feel coagulopathy, and that is how the snake kills its prey. To prove no envenomation, one must repeat labs before discharge. Staff should be made aware that if laboratory reports indicate the patient's blood will not clot the physician should be contacted immediately.
Do not underestimate the meaning of an abnormal FDP, as it can be a harbinger of things to come.
Be cautious of attributing symptoms of envenomation (e.g., tingling, numbness) to anxiety.
Patients with diabetes may have little or no pain if bitten in an area affected by diabetic neuropathy.
Swelling is a great assessment tool, but the absence of swelling does not rule out the possibility of a severe envenomation.
Swelling comes from the venom destroying vessel walls, leading to hypovolemia, which leads to shock. Stay ahead with warmed normal saline and by administering antivenom. Elevated or increasing heart rate or periodic drops in blood pressure likely call for increasing the fluid rate and for re-evaluating the need for additional antivenom.
Did you consider the timing? It is probable that the sooner symptoms appear (especially if rapidly progressing), the greater the venom load.
Always admit to an ICU. Envenomation is a deceptive, complex poisoning, potentially affecting every organ system, requiring intensive observation and care.
Focusing on trends and progression? Trends are identified through serial laboratory studies and symptomatology.
Keep the Foley catheter in as a safety net. With the discovery of hematuria through hourly dipsticks for blood, blood studies are repeated before scheduled, potentially pre-venting lethal bleeding. Also, myoglobinuria or murky-appearing urine can be identified more quickly with a Foley in place, which is critical to protecting the kidneys. Evaluate the need for forced diuresis, but try not to overload with fluids, because the lungs are susceptible to damage from venom and pulmonary edema can result.
Remember that the elimination half-life of CroFab is approximately 12 to 30 hours, but venom can remain active for many days to weeks [1]. Be mindful of the potential for clinical relapse after discharge. (If coagulopathy occurred earlier, follow-up laboratory should be done about every 48 hours after the last antivenom dose, until coagulation values are clearly stable or improving for several days, otherwise re-check within five days [5].) More antivenom may be required.
Severe envenomation? The CroFab dosage is 10 vials, not 6, followed by 2 to 4 vials, as needed, and a more aggressive maintenance dose regimen [16]. The Anavip dosage is 20 vials, followed by 4 to 8 vials, as needed.
Plastic surgery consult instituted? Hands and fingers are especially important to one's future.
Early physical therapy instituted?
Does the patient know in detail what to watch for at home? Provide a discharge information sheet for snakebite victims.
Have you considered pretreating the highly allergic patient with diphenhydramine and cimetidine? Consider prophylactic use of epinephrine for acutely sensitive patients with a life-threatening envenomation.
Protecting local tissue requires early and adequate amounts of antivenom.
Pediatric? Dosages are the same; fluids may be less, resulting in a greater antivenom concentration, which would call for a slower rate of administration. Treatment may be required earlier than in adults.
Small pit vipers have to eat, too. Their venom has equal effects to that of adult snakes.
A decrease in platelets calls for antivenom, not platelet administration. Platelets typically rebound quickly once an adequate amount of antivenom is administered.
Study the asymptomatic patient for 8 to 12 hours before discharging (as significant symptoms can be quite delayed occasionally). Repeat laboratory studies before discharge. Hold minimally symptomatic patients for observation and repeated lab studies for at least 24 hours [3].
Consider the status of the patient and the wisdom of transferring from the emergency department if the ICU staff is unfamiliar with snakebite assessment and treatment.
Do not underestimate the potential severity of the bite based on initial evaluation. Frequently re-evaluate the clinical situation and the need for additional antivenom.
Avoid under-dosing and potential death of the patient. It is better to slightly overtreat than to undertreat [17].
Do not confuse CroFab and Anavip dosages. Anavip generally calls for a dosage twice that of CroFab.
Avoid fasciotomy unless a snakebite expert concurs with the need. Adequate antivenom given early avoids the need to consider fasciotomy.
Do not fail to administer antivenom in the case of life-threatening envenomation because of an allergic reaction to the antivenom. The question is: Can one take the risk of not giving antivenom [4]?
Pregnancy of the patient is no contraindication to serum therapy. Early and adequate treatment with antivenom may stop venom-induced contractions in cases of severe envenomation and may prevent abortion [15].
Watch closely for recurrence as the patient progresses to getting out of bed. A single occasion of scant exercise can trigger a surprising recurrence.
Do not spare the pain medicine—a necrotizing inflammatory reaction is very painful.
Consider snake venom poisoning in the case of an edematous, ecchymotic extremity in a child who was playing in an area where snakes may be found. The same is true for patients with diabetes.
Albumin and plasma expanders are simply not indicated and can confuse the coagulation abnormality by creating one of their own [6]. Support blood pressure with vasopressor drugs if necessary, only after adequate volume resuscitation.
Realize that there is no time limit to administering antivenom to reverse coagulopathy.
Consult, consult, consult.
Do not:
Insert nasogastric tubes.
Do arterial blood gas measurements.
Insert any non-critical lines.
Apply tourniquets, ice, heat, or shock.
Elevate the extremity until you are ready for further distribution of venom.
Allow any exercise until coagulation studies begin to trend toward normal.
Make incisions.
Wrap the extremity.
Release constricting bands or tourniquets without a physician's order.
Administer aspirin, aspirin-containing products, NSAIDs, or other anticoagulants.
Use an automatic blood pressure unit after envenomation is detected.
Most pit viper venoms mainly attack tissue and our ability to clot. They affect the nervous system to a lesser extent, with the exceptions of the Mojave and southern Pacific rattlesnakes. Even though you have been bitten by a venomous snake, the snake may not have injected any venom into you. We will watch you closely for a number of hours to be sure that no signs of injury appear. If signs of injury do appear, then we may need to treat you with antivenom. This would be infused into a vein through one of two intravenous (IV) lines. The amount of antivenom and how fast it is administered is determined by how much venom we believe you received. You could be given antivenom more than once, depending on laboratory tests, the progression of signs and symptoms (including swelling), and how you feel.
If you need antivenom, you should be comforted by knowing that the days of frequent and severe reactions to the medicine are happily behind us. We will use a modern product that has proven to be much safer and quite effective.
You can help us to properly care for you by reporting to us any change in the way you feel, now and during your treatment. Do not ignore any unusual feelings, regardless of whether you believe them to be related to the bite. We will be frequently monitoring your blood pressure, pulse, pain, and any swelling, and will question you frequently about how you feel. We will also need to perform blood tests at regular intervals, perhaps frequently at first. Do not be alarmed—this is standard procedure that you will tolerate well. As a member of your healthcare team, please ask questions and voice your concerns to the staff freely.
1. McEvoy GK. Crotalidae polyvalent immune fab (ovine). In: American Society of Health-System Pharmacists (ed). AHFS Drug Information 2007. Bethesda, MD: American Society of Health-System Pharmacists; 2007: 3283.
2. McEvoy GK. Ephedrine. In: American Society of Health-System Pharmacists (ed). AHFS Drug Information 2007. Bethesda, MD: American Society of Health-System Pharmacists; 2007: 1336.
3. Arizona Poison and Drug Information Center. Available at https://azpoison.com. Last accessed January 13, 2026.
4. Bernstein JN. Florida's Poison Control Centers, Miami. Available at https://floridapoisoncontrol.org/about-the-network/your-local-center. Last accessed January 13, 2026.
5. Boyer LV, Seifert SA, Cain JS. Recurrence phenomena after immunoglobulin therapy for snake envenomations: part 2. Guidelines for clinical management with crotaline fab antivenom. Ann Emerg Med. 2001;37(2):196-201.
6. Rocky Mountain Poison and Drug Safety. Available at https://www.rmpds.org. Last accessed January 13, 2026.
7. Gaskin H, Tallahassee Memorial Regional Medical Center, Department of Pharmacy. Conversation with J. Soskis, 1993.
8. McNally J, Assistant Director, Arizona Poison Control Systems. Conversation with J. Soskis, 1995.
9. Crotalidae Polyvalent Immune FAB (Ovine) Package Insert. Available at https://www.fda.gov/media/74683/download. Last accessed January 13, 2026.
10. Roberts JR. Diagnosis and treatment of snakebite. In: Schwartz GR, Cayten CG, Mangelsen MA, Mayer TA, Hanke BK (eds). Principles and Practice of Emergency Medicine. 3rd ed. New York, NY: Lea & Febiger; 1992.
13. Soskis JE. Snakebite Assessment and Treatment in the Eastern United States. Tallahassee, FL: Snakebite Publishing; 1995.
14. Snakes. In: The New Encyclopaedia Britannica Macropaedia. New York, NY: William and Helen Benton Publishers; 1974.
15. Van Mierop LH. Poisonous snakebite: a review 2. Symptomatology and treatment. J Fla Med Assoc. 1976;63(3):201-210.
18. Wingert WA. Snakebite: method of Willis A. Wingert. In: Conn's Current Therapy. 2nd ed. New York, NY: WB Saunders Co.; 1990.
19. Antivenom (Crotalidae) Polyvalent: Package Insert. Available at https://bi-animalhealth.com/pets/canine/products/therapeutics/antivenin. Last accessed January 13, 2026.
20. 20. Yip L. Rational use of crotalidae polyvalent immune Fab (ovine) in the management of crotaline bite. Ann of Emer Med. 2002;39(6):648-650.
21. Kanaan NC, Ray J, Stewart M, et al. Wilderness Medical Society practice guidelines for the treatment of pit viper envenomations in the United States and Canada. Wilderness Environ Med. 2015;26(4):472-487.
23. Bogdan GM, Dart RC. Prolonged and recurrent coagulopathy after North American pit viper envenomation. Ann Emerg Med. 1996;27(6):820.
29. Jurkovich G J, Luterman A, McCullar K, Ramenofsky M, Curreri PW. Complications of crotalidae antivenom therapy. J Trauma. 1988;28(7):1032.
31. Scharman E, Noffsinger V. Copperhead snakebites: clinical severity of local effects. Ann Emer Med. 2004;43(2):55.
32. Richardson W, Barry J, Tong T, Williams S, Clark R. Rattlesnake envenomation to the face of an infant. Pediatric Emergency Care. 2005;21(3):173.
34. Barach EM, Nowak RM, Tennyson GL. Epinephrine for treatment of anaphylactic shock. JAMA. 1984;251:2118.
35. Sullivan TJ. Cardiac disorders in penicillin-induced anaphylaxis: association with intravenous epinephrine therapy. JAMA. 1982;248:2161.
36. Heilpern K. The treacherous clinical spectrum of allergic emergencies: diagnosis, treatment, and prevention. Emer Med. 1994;15(22):211.
38. McCollough N, Gennaro J. Treatment of venomous snakebite in the United States. Clin Toxicol. 1970;3(3):483-500.
39. Norris RL, Ling J, Wang R. Snake venom poisoning in the United States: assessment and management. Emerg Med Reports. 1995;16:87-94.
40. Wingert WA, Wainschel J. Diagnosis and management of envenomation by poisonous snakes. South Med J. 1975;68(8):1015-1026.
42. Burgess JL, Dart RC. Snake venom coagulopathy: use and abuse of blood products in the treatment of pit viper envenomation. Ann Emerg Med. 1991;20(7):795-801.
43. Office of Medical Applications of Research. Fresh Frozen Plasma: Indications and Risks. Available at https://jamanetwork.com/journals/jama/article-abstract/396477. Last accessed January 13, 2026.
44. Office of Medical Applications of Research. Platelet Transfusion Therapy. Available at https://jamanetwork.com/journals/jama/article-abstract/365357. Last accessed January 13, 2026.
45. Huestis DW, Bove JR, Busch S. Practical Blood Transfusion. 4th ed. Boston, MA: Little, Brown and Co.; 1988.
46. Office of Medical Applications of Research. Perioperative Red Blood Cell Transfusion. Available at https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1349745/pdf/amjph00251-0070.pdf. Last accessed January 13, 2026.
47. Weidensaul S. The bellied viper: even hunters of timber rattlers now admit that these snakes are shy, placid—and very fragile. Smithsonian. 1997;28(9):96.
48. Bush SP, Jansen PW. Severe rattlesnake envenomation with anaphylaxis and rhabdomyolysis. Ann Emer Med. 1995;25(6):845-848.
49. Andrews CE. The treatment of diamondback rattlesnake bite (Crotalus adamanteus): vitamin K an adjunct. Arch Surg. 1960;81(5):699-705.
50. Hurlbut KM, Dart RC, Spaite D, McNally J. Reliability of clinical presentation for predicting significant pit viper envenomation. Ann Emer Med. 1988;17(4):438-439.
52. Soskis JE. Complete Guide to Snakebite Care: Specializing in Pit Viper Envenomation, Including Information about the Latest Antivenin. Unpublished Manuscript; 2008.
53. Harrison K. American Rattlesnake: Toxicology. Available at http://www.kingsnake.com/toxicology/old/snakes/American/rattlesnake.html. Last accessed May 21, 2019.
54. Malone RA IV, Allergist and Associate Professor of Pediatrics at Florida State University College of Medicine. Conversation with J. Soskis.
56. Webb LM, Lieberman P. Anaphylaxis a review of 601 cases. Ann Allergy Asthma Imunol. 2006;97(1):39-43.
57. Joint Task Force on Practice Parameters; American Academy of Allergy, Asthma and Immunology; American College of Allergy, Asthma and Immunology; Joint Council of Allergy, Asthma and Immunology. The diagnosis and management of anaphylaxis: an updated practice parameter. J Allergy Clin Immunol. 2005;115(3 Suppl 2):S483-S523.
58. Bock SA, Munoz-Furlong A, Sampson HA. Fatalities due to anaphylactic reactions to food. J Allergy Clin Immunol. 2001;107(1):191-193.
59. Sampson HA, Mendelson L, Rosen JP. Fatal and near-fatal anaphylactic reactions to food in children and adolescents. N Engl J Med. 1992;327(6):380-384.
60. Klein JS, Yocum MW. Underreporting of anaphylaxis in a community emergency room. J Allergy Clin Immunol. 1995;95(2):637-638.
61. Helbling A, Hurni T, Mueller UR, Pichler WJ. Incidence of anaphylaxis with circulatory symptoms: a study over a 3-year period comprising 940,000 inhabitants of the Swiss Canton Bern. Clin Exp Allergy. 2004;34(2):285-290.
62. Moneret-Vautrin DA, Morisset M, Flabbee J, Beaudouin E, Kanny G. Epidemiology of life-threatening and lethal anaphylaxis: a review. Allergy. 2005;60(4):443-451.
63. Bock SA, Munoz-Furlong A, Sampson HA. Further fatalities caused by anaphylactic reactions to food. J Allergy Clin Immunol. 2007;119(4):1016-1018.
64. Lieberman P. Anaphylactic reactions during surgical and medical procedures. J Allergy Clin Immunol. 2002;110(2Suppl):S64-S69.
65. Simons FE, Clark S, Camargo CA Jr. Anaphylaxis in the community: learning from the survivors. J Allergy Clin Immunol. 2009;124(2):301-306.
66. Simons FE. First-aid treatment of anaphylaxis to food: focus on epinephrine. J Allergy Clin Immunol. 2004;113(5):837-844.
67. Boyce JA, Assa'ad A, Burks AW, et al. Guidelines for the diagnosis and management of food allergy in the United States: report of the NIAID-sponsored expert panel. J Allergy Clin Immunol. 2010;126(6):S51-S58.
68. Jarvinen KM, Sicherer SH, Sampson HA, Nowak-Wegrzyn A. Use of multiple doses of epinephrine in food-induced anaphylaxis in children. J Allergy Clin Immunol. 2008;122(1):133-138.
69. Oren E, Banerji A, Clark S, Camargo CA Jr. Food-induced anaphylaxis and repeated epinephrine treatments. Ann Allergy Asthma Immunol. 2007;99(5):429-432.
70. Simons FE, Gu X, Simons KJ. Epinephrine absorption in adults: intramuscular versus subcutaneous injection. J Allergy Clin Immunol. 2001;108(5):871-873.
71. Sampson HA, Munoz-Furlong A, Bock SA, et al. Symposium on the definition and management of anaphylaxis: summary report. J Allergy Clin Immunol. 2005;115(3):584-591.
72. Pumphrey RS. Lessons for management of anaphylaxis from a study of fatal reactions. Clin Exp Allergy. 2000;30(8):1144-1150.
73. Song TT, Nelson MR, Chang JH, Engler RJ, Chowdhury BA. Adequacy of the epinephrine auto-injector needle length in delivering epinephrine to the intramuscular tissues. Ann Allergy Asthma Immunol. 2005;94(5):539-542.
74. Stecher D, Bulloch B, Sales J, Schaefer C, Keahey L. Epinephrine auto-injectors: is needle length adequate for delivery of epinephrine intramuscularly? Pediatrics. 2009;124(1):65-70.
75. Mehl A, Wahn U, Niggemann B. Anaphylactic reactions in children: a questionnaire-based survey in Germany. Allergy. 2005;60(11):1440-1445.
76. Pumphrey R. Anaphylaxis: can we tell who is at risk of a fatal reaction? Curr Opin Allergy Clin Immunol. 2004;4(4):285-290.
77. Sheikh A, ten Broek VM, Brown SG, Simons FE. H1-antihistamines for the treatment of anaphylaxis: Cochrane systematic review. Allergy. 2007;62(8):830-837.
80. Choong K, Bohn D, Fraser DD, et al. Vasopressin in pediatric vasodilatory shock: a multicenter randomized controlled trial. Am J Respir Crit Care Med. 2009;180(7):632-639.
81. Oswalt ML, Kemp SF. Anaphylaxis: office management and prevention. Immunol Allergy Clin North Am. 2007;27(2):177-191.
82. Brown SG, Blackman KE, Heddle RJ. Can serum mast cell tryptase help diagnose anaphylaxis? Emerg Med Australas. 2004;16(2):120-124.
84. 84. Dorland's Illustrated Medical Dictionary. 26th ed. Philadelphia, PA: W.B. Saunders Company; 1981.
85. Boyer LV, Seifert SA, Clark RF, et al. Recurrent and persistent coagulopathy following pit viper envenomation. Arch Intern Med. 1999;159(7):706-710.
86. Lavonas EJ, Ruha R-M, Banner W, et al. Unified treatment algorithm for the management of crotaline snakebite in the United States: results of an evidence-informed consensus workshop. BMC Emerg Med. 2011;11:2.
87. Dart RC, Seifert SA, Boyer LV, et al. A randomized multicenter trial of Crotalinae polyvalent immune Fab (ovine) antivenom for the treatment for crotaline snakebite in the United States. Arch Intern Med. 2001;161(16):2030-2036.
88. Lavonas, EJ, Kokko J, Schaeffer TH, Mlynarchek SL, Bogdan GM, Dart RC. Short-term outcomes following Fab antivenom therapy for severe crotaline snakebite. Ann Emerg Med. 2010;57(2):128-137.
89. Dart RC, Gold BS. Crotaline snakebite. In: Dart RC (ed). Medical Toxicology. Philadelphia, PA: Lippincott Williams and Wilkins; 2004: 1564.
90. Friedman AL. Pediatric hydration therapy: historical review and a new approach. Kidney International. 2005;67:380-388.
91. Carcillo JA, Tasker RC. Fluid resuscitation of hypovolemic shock: acute medicine's great triumph for children. Inten Care Med. 2006;32:958-961.
93. Schaeffer TH, Khatri V, Reifler LM, Lavonas EJ. Incidence of immediate hypersensitivity reaction and serum sickness following administration of Crotalidae polyvalent immune Fab antivenom: a meta-analysis. Acad Emerg Med. 2012;19(2):121-131.
94. Sullivan JB, Wingert WA, Norris RL. North American venomous reptile bites. In: Auerbach PS (ed). Wilderness Medicine: Management of Wilderness and Environmental Emergencies. 3rd ed. St. Louis, MO: Mosby; 1995: 680-709.
95. Lavonas EJ, Gerardo CJ, Copperhead Snakebite Recovery Outcome Group. Prospective study of recovery from copperhead snake envenomation: an observational study. BMC Emerg Med. 2015;15:9.
96. Kounis NG. Mast Cells and the Heart: Kounis Syndrome and Takotsubo Cardiomyopathy. Available at https://tmsforacure.org/expert-information/mast-cells-heart-kounis-syndrome-takotsubo-cardiomyopathy. Last accessed January 13, 2026.
97. U.S. Food and Drug Administration. Package Insert: Anavip. Available at https://www.fda.gov/media/92139/download. Last accessed January 13, 2026.
98. Joint Task Force on Practice Parameters; American Academy of Allergy, Asthma, and Immunology; American College of Allergy, Asthma, and Immunology; Joint Council of Allergy, Asthma, and Immunology. The diagnosis and management of anaphylaxis: an updated practice parameter. J Allergy Clin Immunol. 2005;115(3 Suppl 2):S483-S523.
99. Dawson CA, Marriott AE, Crittenden E, et al. A drug combination therapy consisting of toxin phospholipase A2 and metalloproteinase inhibitors provides preclinical protection against North American Crotalid snakebite envenoming. bioRxiv. 2025;1:2025-04.
102. Xiao H, Pan H, Liao K, Yang M, Huang C. Snake venom PLA2, a promising target for broad-spectrum antivenom drug development. Biomed Res Int. 2017;2017:6592820.
103. Gutiérrez JM, Lewin MR, Williams DJ, Lomonte B. Varespladib (LY315920) and methyl varespladib (LY333013) abrogate or delay lethality induced by presynaptically acting neurotoxic snake venoms. Toxins. 2020;12(2):131.
104. Lewin M, Samuel S, Merkel J, Bickler P. Varespladib (LY315920) appears to be a potent, broad-spectrum, inhibitor of snake venom phospholipase A2 and a possible pre-referral treatment for envenomation. Toxins. 2016;8(9):248.
105. Tasoulis T, Isbister GK. A review and database of snake venom proteomes. Toxins. 2017;9(9):290.
106. Gutiérrez JM, Ownby CL. Skeletal muscle degeneration induced by venom phospholipases A2: insights into the mechanisms of local and systemic myotoxicity. Toxicon. 2003;42(8):915-931.
107. Gutiérrez JM, Lomonte, B. Phospholipases A2: unveiling the secrets of a functionally versatile group of snake venom toxins. Toxicon. 2013;62:27-39.
Mention of commercial products does not indicate endorsement.