During the past decades, advances in the prevention, diagnosis, and treatment of cerebrovascular disorders have led to decreases in the prevalence of stroke (primarily ischemic stroke), but the high rates of morbidity and mortality, despite a number of modifiable risk factors, point to the need for enhanced education for both patients and physicians. Enhancing healthcare professionals' knowledge about evidence-based guidelines, as well as about advances in diagnostic tools, is crucial for improving the outcomes for individuals who are at risk for stroke or in whom a stroke has occurred. A better understanding is needed about the options for patients at risk for stroke, as well as for treatment and rehabilitation. This course explores the roles of diagnosis and screening, evaluation of individuals with suspected stroke, immediate treatment of stroke, and the elements of effective rehabilitation programs. Heightened awareness of variations in the prevalence of stroke according to age, sex, and racial/ethnic groups will help clinicians and healthcare educators target individuals in need of enhanced education about the risks of stroke, especially those that are modifiable.
This course is designed for physicians, nurses, and physician assistants in the primary care setting. Neurologists and other healthcare practitioners will also benefit from this course.
The early identification and management of the risk factors for ischemic stroke can lead to substantial health benefits and reductions in cost. However, research has documented gaps between healthcare professionals' knowledge and practice with respect to prevention, demonstrating that adherence to evidence-based or guideline-endorsed recommendations pertaining to all interventions for primary and secondary prevention are underutilized or ineffective. The purpose of this course is to provide needed information about the roles of diagnosis and screening, timely evaluation of individuals with suspected stroke, immediate treatment of stroke, and the elements of effective rehabilitation programs so that healthcare professionals may implement the necessary interventions appropriately.
Upon completion of this course, you should be able to:
- Describe the primary types of cerebrovascular disorders and their causes.
- Discuss differences in prevalence, morbidity, and mortality according to age, sex, and race/ethnicity.
- Identify the nonmodifiable and modifiable risk factors for ischemic stroke.
- Implement primary prevention strategies according to evidence-based guidelines.
- Discuss the need for education at the community and patient levels.
- Apply models of predicting risk of ischemic stroke.
- Select the appropriate tools for screening, diagnosis, and early management of ischemic stroke.
- Describe the elements of stroke systems of care and a comprehensive stroke center.
- Discuss evidence-based treatment options for ischemic stroke.
- Describe the benefits and components of a specialized stroke rehabilitation team.
- Outline the aspects of patient assessment for stroke rehabilitation.
- Discuss evidence-based recommendations for secondary prevention of ischemic stroke.
Lori L. Alexander, MTPW, ELS, MWC, is President of Editorial Rx, Inc., which provides medical writing and editing services on a wide variety of clinical topics and in a range of media. A medical writer and editor for more than 30 years, Ms. Alexander has written for both professional and lay audiences, with a focus on continuing education materials, medical meeting coverage, and educational resources for patients. She is the Editor Emeritus of the American Medical Writers Association (AMWA) Journal, the peer-review journal representing the largest association of medical communicators in the United States. Ms. Alexander earned a Master’s degree in technical and professional writing, with a concentration in medical writing, at Northeastern University, Boston. She has also earned certification as a life sciences editor and as a medical writer.
John M. Leonard, MD, Professor of Medicine Emeritus, Vanderbilt University School of Medicine, completed his post-graduate clinical training at the Yale and Vanderbilt University Medical Centers before joining the Vanderbilt faculty in 1974. He is a clinician-educator and for many years served as director of residency training and student educational programs for the Vanderbilt University Department of Medicine. Over a career span of 40 years, Dr. Leonard conducted an active practice of general internal medicine and an inpatient consulting practice of infectious diseases.
Contributing faculty, Lori L. Alexander, MTPW, ELS, MWC, has disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
Contributing faculty, John M. Leonard, MD, has disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
John V. Jurica, MD, MPH
Margo A. Halm, RN, PhD, NEA-BC, FAAN
The division planners have disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
Sarah Campbell
The Director of Development and Academic Affairs has disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
The purpose of NetCE is to provide challenging curricula to assist healthcare professionals to raise their levels of expertise while fulfilling their continuing education requirements, thereby improving the quality of healthcare.
Our contributing faculty members have taken care to ensure that the information and recommendations are accurate and compatible with the standards generally accepted at the time of publication. The publisher disclaims any liability, loss or damage incurred as a consequence, directly or indirectly, of the use and application of any of the contents. Participants are cautioned about the potential risk of using limited knowledge when integrating new techniques into practice.
It is the policy of NetCE not to accept commercial support. Furthermore, commercial interests are prohibited from distributing or providing access to this activity to learners.
Supported browsers for Windows include Microsoft Internet Explorer 9.0 and up, Mozilla Firefox 3.0 and up, Opera 9.0 and up, and Google Chrome. Supported browsers for Macintosh include Safari, Mozilla Firefox 3.0 and up, Opera 9.0 and up, and Google Chrome. Other operating systems and browsers that include complete implementations of ECMAScript edition 3 and CSS 2.0 may work, but are not supported. Supported browsers must utilize the TLS encryption protocol v1.1 or v1.2 in order to connect to pages that require a secured HTTPS connection. TLS v1.0 is not supported.
The role of implicit biases on healthcare outcomes has become a concern, as there is some evidence that implicit biases contribute to health disparities, professionals' attitudes toward and interactions with patients, quality of care, diagnoses, and treatment decisions. This may produce differences in help-seeking, diagnoses, and ultimately treatments and interventions. Implicit biases may also unwittingly produce professional behaviors, attitudes, and interactions that reduce patients' trust and comfort with their provider, leading to earlier termination of visits and/or reduced adherence and follow-up. Disadvantaged groups are marginalized in the healthcare system and vulnerable on multiple levels; health professionals' implicit biases can further exacerbate these existing disadvantages.
Interventions or strategies designed to reduce implicit bias may be categorized as change-based or control-based. Change-based interventions focus on reducing or changing cognitive associations underlying implicit biases. These interventions might include challenging stereotypes. Conversely, control-based interventions involve reducing the effects of the implicit bias on the individual's behaviors. These strategies include increasing awareness of biased thoughts and responses. The two types of interventions are not mutually exclusive and may be used synergistically.
#90284: Ischemic Stroke
Cerebrovascular disease is associated with significant morbidity and mortality in the United States. Stroke occurs in approximately 795,000 individuals each year, of which 610,000 are first-time strokes and 185,000 are recurrent [1]. National surveillance data for the period 2017 to 2021 found that the prevalence rate (per 100,000 population) of stroke was 7.7 (men) and 8.8 (women) in the 60- to 79-year age group, and 13.9/14.0 among all those older than 80 years of age [1]. Stroke is the leading cause of long-term disability in adults; 65% of stroke survivors have some degree of impairment [1,2]. Approximately 3% of men and 2% of women reported that they were disabled because of stroke [3]. The effect of stroke on mortality is illustrated by the fact that cerebrovascular disease is the third leading cause of death in the United States, with an age-adjusted mortality rate of 37.3 per 100,000 population as of 2016 [1,3]. This represents a 16.7% decrease in the mortality rate and a 3.7% increase in actual deaths over the previous decade. It is a substantial healthcare issue, as one of every three deaths can be attributed to stroke [3]. Despite the considerable progress being made in the areas of prevention, management, and rehabilitation, it appears that stroke will increasingly cause death and disability in the coming decades as the population ages [4]. By 2030, an additional 3.4 million U.S. adults (3.9% of the population) will have had a stroke, which is a 20.5% increase in prevalence from 2012.
In addition, the financial cost associated with cerebrovascular disease poses a substantial economic burden, with an estimated direct and indirect cost of $351.2 billion between 2014 and 2015 [3]. In 2018 to 2019, the direct and indirect cost of stroke in the United States was $56.6 billion [1]. The estimated direct medical cost of stroke totaled $36.5 billion, including hospital outpatient or office-based professional visit expense, hospital inpatient stays, ED visits, medication costs, and home health care. Additionally, the total direct medical stroke-related costs are projected to more than double by 2035 ($94 billion), with much of the projected increase arising from those older than 80 years of age [1].
The two primary types of acute stroke are ischemic and hemorrhagic. Of all strokes, 87% are ischemic, 10% are intracerebral hemorrhages (ICHs), and 3% are subarachnoid hemorrhages (SAHs) [1]. There are important modifiable risk factors for ischemic stroke, and predicting risk is an important element in prevention. In predicting risk, consideration should be given not only to comorbidities but also to age, sex, and race/ethnicity, as disparities in stroke prevalence, morbidity, and mortality have been attributed to these patient characteristics [5,6,7,8]. Evidence-based guidelines for primary and secondary prevention have been developed and should be implemented [9,10].
Transient ischemic attack (TIA) is one of the most important risk factors for ischemic stroke. Approximately 240,000 persons experience a TIA each year, and 12% of ischemic strokes are preceded by a TIA [1]. The risk of sustaining a stroke following TIA increases from 1.2% within the first 48 hours to 7.4% at three months [1,11]. Improved understanding of TIAs among both clinicians and patients is needed. A survey of 200 primary care physicians showed that 88% could not correctly identify the typical symptoms and duration of a TIA, and studies have indicated that half of individuals who have a TIA do not report the event to their primary care clinician [1,12].
The early identification and management of the risk factors for ischemic stroke can lead to substantial improvement in health and reductions in cost [13]. For example, the incidence of stroke was reduced by 30% to 40% following improved blood pressure control and the use of antiplatelet therapies [10]. Yet, research has documented gaps between physicians' knowledge and practice with respect to prevention, with data on adherence to evidence-based or guideline-endorsed recommendations demonstrating underuse or ineffective use of all interventions for primary and secondary prevention [11,14,15]. Evidence-based guidelines have also been developed for the early management of stroke and for rehabilitation after stroke and should be followed to provide optimum care [16,17,18].
The focus of this course is ischemic stroke, due to its overwhelming prevalence. Advances have been made in tools for the screening and diagnosis of ischemic stroke, and a better understanding of the options for patients at risk is needed. This course explores the role of the physical examination and history, laboratory studies, and imaging techniques in screening and diagnosis. Also discussed are evidence-based guidelines for the prevention and early management of ischemic stroke, as well as emerging treatment options. Because data have shown that outcome is improved by care provided in comprehensive stroke centers and by early rehabilitation, these topics are addressed as well [17,19,20,21]. The importance of a multidisciplinary rehabilitation team, appropriate patient assessment, and an exercise program is emphasized.
Although "cerebrovascular disease" is often used interchangeably with the term "stroke," the disease encompasses any neurovascular disorder that exists in the presence or absence of an ictus (e.g., carotid artery stenosis, arteriovenous malformations). Despite advances in understanding the pathophysiology of cerebrovascular diseases, the term "stroke" (also known as cerebrovascular accident or brain attack) is inconsistently defined. Stroke has been classically characterized as an injury to the central nervous system (CNS) by a vascular cause. Because this definition is mainly clinical and not inclusive of advances in science and technology, the American Heart Association (AHA)/American Stroke Association (ASA) convened a writing group to develop an updated definition of stroke. The AHA/ASA recommend that the term "stroke" be broadly used to include a variety of episodic brain injury events, whether symptomatic or asymptomatic (Table 1). The cerebrovascular anatomy at risk for stroke consists of the anterior circulation (cervical and internal carotid artery system) and posterior circulation (vertebrobasilar artery system).
AHA/ASA DEFINITION OF STROKE
| Injury/Episode | Definition |
|---|---|
| CNS infarction |
Brain, spinal cord, or retinal cell death attributable to ischemia, based on:
|
| Ischemic stroke | Episode of neurologic dysfunction caused by focal cerebral, spinal, or retinal infarction |
| Silent CNS infarction | Imaging or neuropathologic evidence of CNS infarction, without history of acute neurologic dysfunction attributable to the lesion |
| Intracerebral hemorrhage | Focal collection of blood within the brain parenchyma or ventricular system, not caused by trauma |
| Stroke caused by intracerebral hemorrhage | Rapidly developing clinical signs of neurologic dysfunction attributable to focal collection of blood within brain parenchyma or ventricular system, not caused by trauma |
| Silent cerebral hemorrhage | Focal collection of chronic blood products within the brain parenchyma, subarachnoid space, or ventricular system on neuroimaging or neuropathologic examination, not caused by trauma and without history of acute neurologic dysfunction attributable to the lesion |
| Subarachnoid hemorrhage | Bleeding into subarachnoid space |
| Stroke caused by subarachnoid hemorrhage | Rapidly developing signs of neurologic dysfunction and/or headache because of bleeding into the subarachnoid space, not caused by trauma |
| Stroke caused by cerebral venous thrombosis | Infarction or hemorrhage in the brain, spinal cord, or retina because of thrombosis of a cerebral venous structure. Symptoms or signs caused by reversible edema without infarction or hemorrhage do not qualify as stroke. |
| Stroke, not otherwise specified | Episode of acute neurologic dysfunction presumed to be caused by ischemia or hemorrhage, persisting ≥24 hours or until death, but without sufficient evidence to be classified as one of the above |
| CNS = central nervous system. | |
The multiple sources, pathophysiologic mechanisms, and sequelae of stroke are reflected in the diverse types of cerebrovascular disorders. The World Health Organization classifies cerebrovascular diseases under "Diseases of the nervous system" in the 11th revision of the International Statistical Classification of Diseases and Related Health Problems (ICD-11), the international standard diagnostic classification for all general epidemiologic purposes and many health management purposes (Table 2) [22]. TIAs and traumatic intracranial hemorrhage are also included in the nervous system disease category in the ICD-11; vascular dementia is not. Its exclusion illustrates the heterogeneity of stroke and its sequelae. Vascular dementia, listed as dementia due to cerebrovascular disease, is categorized under "Mental, behavioral, or neurodevelopmental disorders: Neurocognitive disorders."
SELECTED CEREBROVASCULAR DISEASES AS CLASSIFIED BY THE INTERNATIONAL STATISTICAL CLASSIFICATION OF DISEASES AND RELATED HEALTH PROBLEMS, 11TH REVISION (ICD-11)
| ICD Code | Disease |
|---|---|
| 8B00 | Intracerebral hemorrhage |
| 8B01 | Subarachnoid hemorrhage |
| 8B02 | Nontraumatic subdural hemorrhage |
| 8B03 | Nontraumatic epidural hemorrhage |
| 8B0Z | Intracranial hemorrhage, unspecified |
| 8B10 | Transient ischemic attack |
| 8B11 | Cerebral ischemic stroke |
| 8B1Y | Other specified cerebral ischemia |
| 8B1Z | Cerebral ischemia, unspecified |
| 8B20 | Stroke, not known if ischemic or hemorrhagic |
As noted, the two primary types of stroke are ischemic and hemorrhagic. In the United States, approximately 87% of strokes are ischemic [1]. An ischemic stroke (impending infarction) denotes interruption of tissue perfusion because of critical vascular occlusion resulting from stenosis, embolism, or sustained spasm of a regional artery [23]. Hemorrhagic stroke occurs from aneurysm rupture or a spontaneous hemorrhage below the arachnoid location of the brain's arteriolar network; the resultant expansion of blood within a confined space results in secondary ischemic and pressure injury to brain tissue. Although ischemic stroke is the main focus of this course, a brief overview of hemorrhagic strokes will help to provide context and comparison of the clinical features of both types of stroke. In addition, TIAs are discussed here, as they are often a precursor to ischemic stroke.
Hemorrhagic stroke is associated with a higher risk of fatality than ischemic stroke, and roughly one-third of patients die within 30 days after the event [1,24]. Hemorrhagic strokes are categorized by the location of the hemorrhage, either intracerebral or subarachnoid, with the former being more common.
The majority of hemorrhagic strokes are caused by ICH, and because of this, discussion of hemorrhagic stroke often refers to ICH [1,25]. ICH is defined as bleeding directly into the brain parenchyma [25,26]. Intraventricular hemorrhage refers to extension of bleeding into the brain ventricles [26,27]. Nontraumatic ICH is categorized as primary (unrelated to congenital or acquired lesions), secondary (caused by a congenital or acquired condition), or spontaneous (unrelated to trauma or surgery) [26]. Hypertension, congenital arterial aneurysm, thrombocytopenia (platelet counts <20,000), and depletion of intravascular clotting factors (e.g., hypothrombinemia) are conditions that elevate the risk of sustaining a hemorrhagic stroke, important because risk can be anticipated and treated.
The clinical features of ICH include sudden, intense headache, vomiting, seizures, depressed consciousness, meningeal irritation, and blood-tainted cerebrospinal fluid. Onset of symptoms may occur within seconds to minutes after the start of an ICH. Individuals with this type of stroke often feel more ill than those with an ischemic stroke.
ICH is the least treatable type of stroke [28]. Functional independence is regained within 6 months in approximately 20% of survivors [29]. The morbidity and mortality depend on the volume and location of the hematoma. The 1-year mortality rate varies according to anatomic location, with the highest mortality rate (65%) associated with ICH in the brain stem; the rate is 57% for lobar hemorrhage, 51% for deep hemorrhage, and 42% for cerebellar hemorrhage [30]. Overall, 46% of patients with ICH survive one year and 29% survive five years [31].
As many as 80% of primary ICHs occur after small vessels are compromised by chronic hypertension [32]. Hypertension is associated with ICH originating in the periventricular deep white matter, deep subcortical structures, pons, and cerebellum [33]. In individuals older than 70 years of age, cerebral amyloid angiopathy, a condition that leads to amyloid protein infiltration into the cortical arterioles, is responsible for approximately 20% of ICHs [34]. Other causes of ICH include anticoagulant and antiplatelet use, drug use (e.g., cocaine, phenylpropanolamine), and other bleeding diathesis [28,35]. Fewer than 15% of all cases of ICH are secondary to congenital vascular abnormalities and malignant brain lesions [26].
Subarachnoid hemorrhages occur less frequently than ICHs. The hallmark of subarachnoid hemorrhage is the immediate onset of a severe headache with signs of meningeal irritation [36]. Individuals may describe this headache as their "worst ever." Nausea, vomiting, neck pain, and photophobia are also classic symptoms, although they are not always present [36]. Neurologic deficits may be acute or may manifest hours to days after the onset of bleeding.
Nontraumatic subarachnoid hemorrhages are subcategorized as aneurysmal or non-aneurysmal [37]. Aneurysmal subarachnoid hemorrhage is associated with higher rates of morbidity and mortality than non-aneurysmal hemorrhage. Among patients who live 3 months after the event, the risk of death is 8.7% within 5 years and 17.9% within 10 years [38]. In contrast, non-aneurysmal subarachnoid hemorrhages are associated with better outcomes and are less likely to cause death [39].
Most nontraumatic subarachnoid hemorrhages involve rupture of an intracranial aneurysm or cerebral arteriovenous malformation. Congenital arteriovenous anomalies are more likely to cause stroke in adolescents and young adults [40]. The incidence of perimesencephalic subarachnoid hemorrhage, a non-aneurysmal type, is increasing. Although the cause remains unknown, increased use of antithrombotic medications may be a factor [41,42].
Guidelines for the management of spontaneous ICH have been published by AHA/ASA [28]. Rapid diagnosis and assessment are crucial, as early deterioration is common in the hours after onset. Prehospital management objectives are maintenance of the airway, cardiovascular support, and rapid transport to the nearest facility prepared to care for patients with acute stroke. Advanced notice to the ED of impending arrival is key to expediting assessment and shortening the time to neuroimaging. Any combination of severe headache, vomiting, systolic blood pressure >220 mm Hg, decreased level of consciousness, and rapid symptom progression suggests ICH, although none are specific and neuroimaging is required to distinguish ICH from ischemic stroke. Computed tomography (CT) is sensitive and usually readily available. Key components of initial evaluation include time of symptom onset, progression of symptoms, vascular risk factors, medications (e.g., anticoagulant medication, illicit drug use), and a focused neurologic examination to assess severity. Laboratory studies should include complete blood count, prothrombin time, partial thromboplastin time, and toxicology screen to detect cocaine and other sympathomimetic drugs associated with ICH. Medical management and follow-up imaging is predicated on initial findings and the underlying vascular abnormalities identified [28].
TIAs are sometimes referred to as "ministrokes" because, like ischemic strokes, they are caused by inadequate cerebral blood flow. TIAs are also called warning strokes, as they often precede an ischemic stroke [43]. The superseded definition of a TIA was "a sudden, focal neurologic deficit that lasts for less than 24 hours, is presumed to be of vascular origin, and is confined to an area of the brain or eye perfused by a specific artery" [44]. The 24-hour time limit was an arbitrary remnant of the time interval used in prospective surveys in the early 1970s [45]. Magnetic resonance imaging (MRI) and computed tomography (CT) have demonstrated that one-third of TIAs, including those that last only minutes, cause infarcts [46].
Because TIA and ischemic stroke are less distinct from one another than once believed, a new TIA definition was proposed, revised, and endorsed in 2009. The proposed definition states that TIA is "a brief episode of neurologic dysfunction caused by focal brain or retinal ischemia, with clinical symptoms typically lasting less than 1 hour, and without evidence of acute infarction" [44]. This definition was thought to better reflect the ischemic pathogenesis of TIA, promote its early management, and support the use of diagnostic imaging techniques to ensure that the patient does not have infarction [44]. The definition was endorsed by the 2009 AHA/ASA guideline, with the omission of "typically less than 1 hour" (as infarction is not necessarily bound by a set period of time) and reads, "Transient ischemic attack (TIA): a transient episode of neurologic dysfunction caused by focal brain, spinal-cord, or retinal ischemia, without acute infarction" [10,47]. A 2023 AHA scientific statement on diagnosis and management of TIA stipulates no evidence of tissue infarction determined by diffusion-weighted (DW) MRI. Resolution of symptoms combined with MRI demonstrating acute infarction should be diagnosed as an ischemic stroke [402].
TIAs are caused by similar conditions as those leading to ischemic stroke [10]. Among the common causes are embolic platelet thrombi shed from atherosclerotic plaque within large vessels and cardioembolism from mural thrombi related to atrial fibrillation (AF) or valvular heart disease. Other conditions complicated by TIAs include hypercoagulable states, arterial dissection, sympathomimetic drugs (e.g., cocaine), and arteritis (caused by noninfectious necrotizing vasculitis, drugs, irradiation, or local trauma) [53].
Research shows that TIA should be considered a dire event, a herald sign of sustained risk for adverse cardiovascular events in subsequent years. Prompt intervention is necessary for prevention of more potent ischemic stroke. The risk of stroke is 1.2% at 2 days and increases to 7.4% within 90 days after TIA [1]. A large multicenter TIA registry study found that the risk of stroke within one year was 5.1% and the five-year risk 9.5%; the combined risk of stroke, acute coronary syndrome, or death attributable to cardiovascular disease was 6.2% at one year and 12.9% at five years (398). Approximately 15% of ischemic strokes are preceded by a TIA [3]. However, there are several challenges to immediate treatment of TIAs [10]:
A wide majority of the general population and many healthcare professionals either fail to recognize TIA or underestimate the significance.
Individuals experiencing a TIA often believe they can postpone or forego professional treatment because clinical symptoms usually resolve quickly and without care.
Due to the 24-hour arbitrary time limit in the previously accepted definition, healthcare professionals often choose to monitor a patient with a TIA rather than provide immediate treatment.
As with any stroke, the symptoms of TIA depend on the affected vascular territory. For instance, involvement of the carotid artery causes disturbances in the ipsilateral eye or brain [49]. Although the most common focal neurologic signs of TIA are sudden-onset unilateral weakness and numbness or tingling in a limb, a TIA can cause any of the following symptoms [49,50]:
Numbness of the face, hand, or leg, with or without weakness
Paralysis
Slurred speech
Dizziness
Double vision
Hemianopia
Transient monocular blindness
Imbalance
Aphasia
Confusion
Head pain
Transient graying or blurring of vision is also common. Occasionally, the line of sight will be shaded. Vertebrobasilar TIAs reflect vestibulocerebellar symptoms such as ataxia, dizziness, vertigo, dysarthria, vision abnormalities (e.g., double vision, hemianopia, bilateral vision loss), and unilateral or bilateral motor and sensory dysfunctions [10]. Clinicians should bear in mind that patients may appear asymptomatic on initial evaluation. This is because TIAs often resolve within 5 minutes [51]. The possibility of TIA should be considered if the patient says, "I don't know why I'm here. Whatever it was, it is all better now" [52].
The risk factors for TIA are similar to those for ischemic stroke and include many modifiable factors, such as hyperhomocysteinemia, hyperlipidemia, smoking, obesity, and diabetes [10]. Risk is substantially reduced by treatment of vascular disorders such as hypertension and AF, two conditions commonly associated with older age. Younger individuals (18 to 45 years of age) who have a TIA or ischemic stroke often have no detectable vascular risk factors [54].
As noted, the risk of ischemic stroke is high in the period following a TIA. A meta-analysis found that approximately 5% of patients who have a TIA will have an ischemic stroke within the next 7 days [11,43]. The risk of stroke within three months after a TIA is approximately 10%; the cumulative risk approaches 25% over the ensuing 5 years [11]. Prompt initiation of secondary prevention strategies for TIA and minor stroke with existing therapies has been shown to reduce the risk of early recurrent stroke by 80% [48]. AHA/ASA guidelines support the concept that, with few exceptions, secondary prevention in patients who present with TIA is the same as that for those with ischemic stroke [10]. A 2020 clinical practice review and 2023 AHA published statement on diagnosis and management of TIA provide updated clinical guidance [399,402]. Selected key principles are as follows:
Suspected TIA should be evaluated urgently in a TIA clinic or ED where appropriate specialist expertise and imaging are available.
An initial noncontrast CT of the head, which is part of many stroke/TIA protocols, is useful to evaluate for hemorrhage, mass lesions, and subacute ischemia; however, CT offers limited diagnostic utility for evaluating the patient whose symptoms have resolved. Diffusion-weighted imaging of the head is the preferred imaging modality for patients with suspected TIA and should be performed immediately.
TIAs typically last for minutes and the likelihood of stroke (infarction) increases with increasing duration of symptoms. DW-MRI demonstrates infarct lesions in about 40% of patients with TIA symptoms, and DW-MRI positivity is associated with a more than sixfold increased risk of recurrent stroke within the next year [402]. If a DW-positive lesion is identified, a diagnosis of ischemic stroke is typically made, followed by hospital admission.
If possible, immediately after onset of symptoms, the patient should take aspirin 300 mg, followed by 75–100 mg daily; clopidogrel should be added to aspirin during the first 21 days after TIA (at a 300 mg loading dose, followed by 75–100 mg per day).
Acute phase vascular imaging studies to screen for high-grade cervical carotid and intracranial stenosis, especially in patients older than 50 years of age, is important because it has immediate implications for treatment and stroke prevention.
Long-term prevention of subsequent stroke includes antiplatelet or anticoagulant treatment (depending on etiologic findings), attention to modifiable risk factors (e.g., blood pressure, glycemic control, lipid lowering, smoking cessation, lifestyle and diet counseling), and carotid endarterectomy, if appropriate.
Within minutes of the onset of ischemic stroke, the core of an infarct can begin to form at the least-perfused site. This site is encircled by an area partially altered metabolically and ionically by cytotoxic edema [55]. This area, the ischemic penumbra, is structurally intact and generally salvageable if reperfusion is achieved promptly. Because cerebral function deficits develop rapidly (within minutes to hours) as an ischemic stroke progresses, these brain attacks are a medical emergency. Each minute that passes results in an average loss of 1.9 million neurons and 14 billion synapses; an ischemic brain ages 3.6 years for every hour that passes after the onset of stroke [56]. For this reason, stroke specialists use the mantra, "time is brain." Although irreversible damage occurs, most individuals with stroke have recoverable penumbral tissue for at least three hours following the onset of symptoms [16].
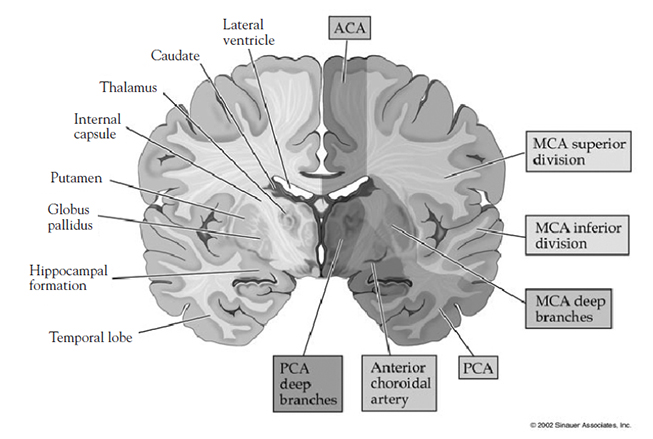
The physical signs, symptoms, and sequelae of ischemic stroke are usually unilateral because of the circulatory anatomy of the brain (Table 3). Anterior circulation is composed of the paired internal carotid arteries and vessels that supply blood to the cerebral hemispheres. Each common carotid artery bifurcates into the internal and external carotid arteries. The ophthalmic artery, posterior communicating artery, and anterior choroidal artery are supplied by the internal carotid artery (Figure 1). Most importantly, the internal carotid artery provides blood to the middle cerebral artery, the largest intracerebral vessel. The middle cerebral artery provides oxygen to the lateral, frontal, parietal, and temporal lobes and the basal ganglia. It also supplies the anterior cerebral artery, which is responsible for the medial part of the frontal and parietal lobes, most of the corpus callosum, the frontobasal cerebral cortex, deep structures, and the anterior diencephalon. The anterior choroidal artery supplies a portion of the thalamus and the posterior limb of the internal capsule.
GENERAL REGIONS OF ISCHEMIC STROKE AND CORRESPONDING NEUROLOGIC DEFICITS
| Affected Region | Common Signs and Potential Sequelae | |||||
|---|---|---|---|---|---|---|
| Left anterior hemisphere |
| |||||
| Right anterior hemisphere |
| |||||
| Left posterior cerebral artery |
| |||||
| Right posterior cerebral artery |
| |||||
| Vertebrobasilar territory (posterior circulation) |
| |||||
| Caudate nucleus, thalamus, frontal lobe (anterior circulation) | Sudden abnormal behavior | |||||
| Thalamus (posterior circulation) | Numbness, decreased sensation on face, arm, leg on same side |
Posterior circulation is primarily composed of the vertebrobasilar artery, the posterior cerebral artery, which it supplies, and other branching vessels. The posterior cerebral artery provides blood to the occipital and medial temporal lobes, as well as regions of the midbrain, subthalamic nucleus, basal nucleus, thalamus, mesial inferior temporal lobe, and occipitoparietal cortices. The two main segments of the posterior cerebral artery (P1 and P2) are connected by the posterior communicating artery. The Circle of Willis links the anterior and posterior circulation at the base of the brain.
In general, ischemic strokes are categorized according to etiology: thrombotic and embolic [61]. In addition, they are classified into five subtypes according to a system developed by the Trial of ORG 10172 in Acute Stroke Treatment (TOAST) [62].
A thrombotic stroke results from progressive atherosclerosis that impairs cerebral arterial blood flow. The stroke event takes two forms: atherosclerotic plaque formation leads to critical stenosis of an artery supplying blood to a region of the brain, or when a fragment of unstable plaque (embolus) breaks loose and lodges in a branch point downstream. The stenosed or occluded artery may be a large vessel (e.g., carotid artery systems, vertebral arteries, the Circle of Willis) or a small vessel (e.g., branches of the Circle of Willis, the posterior circulation). Approximately 21% to 27% of ischemic strokes arise from atherosclerotic disease of the large vessels [63,64]. The cerebral artery branch points, especially those of the internal carotid artery, are the most vulnerable [65]. Small-vessel disease is associated with 21% to 25% of ischemic strokes [63,64]. Thrombotic strokes caused by small-vessel disease are traditionally associated with lacunar infarcts, small, deep, subcortical lesions of 15 mm or less in diameter resulting from occlusion of a single penetrating artery [62,66]. As many as 20% of older individuals who are otherwise healthy have asymptomatic lacunar infarcts unrelated to an ictal event [67]. These silent infarctions were previously believed to be benign with a good long-term prognosis. However, they now have been linked to increased risks of stroke and death and can lead to debilitating cognitive impairments such as vascular dementia [67,68].
Independent risk factors for lacunar infarcts include hypertension, gender, age, diabetes, smoking, and a history of TIA [67]. Although hypertension is strongly associated with the development of small-vessel occlusion, lacunar infarcts also occur in individuals without hypertension. However, normotensive individuals with lacunar infarcts are typically 85 years of age or older, suggesting that hypertension accelerates the arteriopathy underpinning small-vessel disease. Diabetes is an independent risk factor for lacunar infarcts [69].
An embolic stroke occurs when an embolus (i.e., any circulating clot or particle originating from a distal point) lodges in a cerebral arterial vessel, interrupting blood supply to the brain. Stroke registries indicate that 14% to 30% of ischemic strokes are embolic [70,71,72,73,74]. Arterial emboli may originate from the heart (chamber or diseased valves), from the carotid or intracranial arteries (atheromatous plaque), aortic arch, or peripheral venous system by way of a right-to-left shunt (paradoxical embolus). Emboli may be composed of thrombus, fatty deposits, atherosclerotic plaque fragments, or cellular debris emanating from intravascular infections (e.g., endocarditis) or tumors (e.g., atrial myxoma). Clinical symptoms of the resulting infarct correspond to the location of the embolus, not its type. The region of the middle cerebral artery is most frequently blocked by emboli [75].
AF is the most common cardiogenic cause of embolic stroke, and the proportion of strokes caused by AF increases with increasing patient age. The presence of AF increases the risk of embolic stroke fivefold and doubles the risk of death [76,77]. Atrial fibrillation is associated with blood stasis and thrombus formation within the left atrial appendage. Other cardiogenic sources of brain embolism include valvular thrombi (e.g., rheumatic disease, endocarditis, prosthetic valves), mural thrombi from myocardial infarction (MI) or severe heart failure, and paradoxical embolization across a patent foramen ovale [78,79]. MI is associated with a 2% to 3% incidence of embolic stroke, 70% of which occur in the first week after the event [77,79].
The subtype of ischemic stroke influences treatment decisions, prognosis, and risk of recurrent stroke. The TOAST system was designed to facilitate decision making and standardize ischemic stroke research (Table 4) [62]. Most strokes are subclassified as a large-artery atherosclerosis (caused by either a thrombus or an embolus), a cardioembolism, or an occlusion of a small vessel (lacuna) [62]. Approximately 4% of individuals with stroke have coexisting large-vessel and small-vessel disease [61,80]. Other determined causes are rare (approximately 3%), and registries have classified up to 33% of ischemic strokes as being of "undetermined etiology" [81].
FIVE SUBTYPES OF ISCHEMIC STROKE AS CLASSIFIED BY THE TRIAL OF ORG 10172 IN ACUTE STROKE TREATMENT (TOAST)
| Classification | Major Characteristics | ||||
|---|---|---|---|---|---|
| Large-artery atherosclerosis (may be an embolus or thrombus) |
| ||||
| Cardioembolism (may be high or medium risk based on evidence of embolism) |
| ||||
| Small-vessel (lacuna) occlusion |
| ||||
| Other determined cause |
| ||||
| Undetermined etiology |
| ||||
| aThe five classic lacunar syndromes are pure motor hemiparesis, pure sensory stroke, sensorimotor stroke, ataxic hemiparesis, and clumsy-hand dysarthria. | |||||
Approximately 16% of men and 14% of women have a stroke by 85 years of age, and stroke is the fifth leading cause of death in the United States, accounting for more than 150,000 deaths in 2019 [1,3,84]. Morbidity associated with stroke is also high, with at least 65% of stroke survivors having some sort of impairment [85]. At three months after a stroke, approximately 20% of survivors depend on long-term care. Between 15% and 30% of stroke survivors are permanently disabled [86]. A six-month follow-up of ischemic stroke survivors (65 years of age and older) demonstrated that [87]:
50% had some degree of hemiparesis
35% had depressive symptoms
30% were unable to walk without some assistance
26% were dependent in activities of daily living
26% were in a nursing home
19% had aphasia
Fortunately, stroke mortality has been declining over the past 50 years. In the United States, stroke has fallen from the third to the fifth leading cause of death, representing a true mortality decline rather than a repositioning of causes of death [3,84]. This decline is a result of reduced incidence of stroke and lower case-fatality rates, concurrent with cardiovascular risk factor control interventions (e.g., hypertension control). Other efforts (e.g., diabetes control, smoking cessation programs) likely have also contributed to the decline in stroke mortality. The effects of telemedicine and stroke systems of care require additional study but appear to be significant. The decline has occurred in both women and men, for all racial/ethnic and age groups, and represents a major improvement in public health and a reduction in years of potential life lost [88].
Age, gender, and race/ethnicity play major roles in the prevalence of stroke and its associated mortality.
Age
Prolonged damage of the aging cardiovascular system by various risk factors for stroke doubles the risk of ischemic stroke for each decade of life after 55 years of age [9]. Thus, clinicians should be sensitive to their patients' modifiable risk factors, most notably hypertension, starting at an early age [1]. As many as 70% of strokes occur in individuals older than 65 years of age, and the average age at the time of ischemic stroke is 71 years in men and 75 years in women [1]. Stroke patients 85 years of age and older comprise 17% of all stroke patients [1]. Clinicians should be particularly aware of silent cerebral infarctions in older individuals, as these infarctions occur more commonly in this population [89].
Although stroke risk increases with age, strokes can occur at any age; hospital surveillance data from 2014 found that 38% of people hospitalized for stroke were younger than 65 years of age [25]. Approximately 10% to 15% of all strokes occur in adults 18 to 50 years of age, and the underlying pathogenesis and risk factors are more diverse [1,401]. The common types of ischemic stroke in older persons are large vessel atherosclerosis and small vessel occlusions; these two subtypes account for only 10% to 20% of stroke in younger adults. Studies of subtypes have reported a higher prevalence (20% to 47%) of cardioembolism among younger patients with stroke [1,401].
Gender
Women have a higher lifetime risk of stroke than men [90,91,92]. According to estimates from the Framingham Heart Study, among people 55 to 75 years of age, the risk of stroke is 1 in 5 for women and 1 in 6 for men [93]. About 55,000 more women than men have a stroke each year; stroke is the cause of death for about 80,000 women each year [3]. Age-specific stroke incidence rates are substantially lower among women than men, except in those 80 years of age and older, in which the incidence rate in women is approximately equal to or higher than the rate in men [90,91,94,95,96]. Risk factors unique to women or more common in young women include estrogen-containing oral contraceptives, pregnancy, and migraine with aura. The combination of migraine with aura, oral contraceptive use, and cigarette smoking increases stroke ninefold compared with women who do not have any of these three risk factors [401]. The underlying conditions associated with stroke in women with these risk factors include cardioembolism, patent foreman ovale, and prothrombotic states. Pregnancy itself is a prothrombotic state, which in addition to hypertensive disorders of pregnancy increases the risk for ischemic stroke, ICH, and cerebral venous thrombosis. The approximate incidence of stroke is 30 per 100,000 pregnancies, which is three times higher than the incidence of stroke in nonpregnant young adults of comparable age [401]. The complication may occur during the antepartum, peripartum, or postpartum periods.
Among individuals 65 years of age and older, the median survival time after a first stroke is typically longer for women than for men. For both men and women, the median survival decreases with age (Table 5) [97]. However, the possibility of gender disparities in how health care is provided to individuals who present with stroke symptoms is also being evaluated.
Studies suggest that, compared with men, women are evaluated less frequently following a stroke and that any evaluation is more likely to be delayed [98,99,100]. This pattern results from women's presentation with nontraditional symptoms or without traditional symptoms and inappropriate worry by both clinician and patient about treatment-related risks (Table 6) [98,99,101,102,103]. Although studies to evaluate differences in strokes between men and women are in early stages, preliminary results indicate that emergency medical service (EMS) personnel and clinicians need an accurate understanding of symptom presentation patterns for men and women. The prevalence of nontraditional symptoms is higher among women than men; nontraditional stroke symptoms include headache, face and limb pain, nausea, and hiccups as well as symptoms typically believed to be unrelated to neurologic deficits (e.g., chest pain, shortness of breath, palpitations) [102].
Race/Ethnicity
The decline in stroke mortality has reduced, but not eliminated, the racial/ethnic gap in stroke mortality [88]. Racial/ethnic disparities in the incidence of stroke and its related mortality are substantial, and the factors contributing to the disparities are complex and poorly understood [1,5,6,7,8,104]. The risk of first-time stroke among Black individuals is 1.5 times higher than that for White individuals in the United States, and the incidence of first-ever stroke is increasing among Hispanic Americans compared to White Americans [1]. The Northern Manhattan Study (NOMAS) showed that the age-adjusted incidence of first ischemic stroke per 100,000 individuals was 191 in the Black population, 149 in the Hispanic population, and 88 in the White population [1,104]. Another study showed that the prevalence of stroke among these three groups also varied according to age [6]. In the Black population, the prevalence was 4.8% for individuals 45 to 64 years of age and 10% for individuals 65 years of age and older; the corresponding prevalences were 2.3% and 10% for Hispanic individuals and 2% and 9% for White individuals [6]. Intracranial atherosclerotic strokes were the most common type of strokes among the Black and Hispanic populations [1,104]. Higher rates of hypertension, obesity, and diabetes among Black Americans might account for some of this disparity [400].
According to the AHA 2019 update on statistics for heart disease and stroke, the prevalence of stroke is highest for American Indian/Alaskan Natives and lowest for Asian/Pacific Islanders (Table 7) [1]. Data indicate variation among stroke-related mortality rates as well and show that stroke deaths have increased in minority populations while decreasing in the White population. Projections indicate a 20.5% increase in stroke by 2030, with the greatest increase (29%) in Hispanic men [1].
Data from 2016 demonstrated an overall rate of 37.3 deaths per 100,000, with a rate of 51.9 for the Black (non-Hispanic) population, 36.1 for the White (non-Hispanic) population, 32.1 for the Hispanic population, 31.0 for the Asian/Pacific Islander population, and 30.7 for the American Indian/Alaskan Native population [1]. Risk factors, such as hypertension, diabetes, and obesity, vary among these populations as well, but increased risks alone cannot completely explain increased prevalence or mortality [9].
Several modifiable risk factors for stroke have been well-documented in the literature, and some have been less well-documented (Table 8). The well-documented risk factors include many associated with cardiovascular disease as well, and proper management of these factors can reduce both the risk of a first-time stroke and the development of a cardiovascular condition [7,9]. Clinicians should discuss the potential for stroke associated with risk factors specific to patients and offer strategies to reduce or eliminate them [9]. Four lifestyle factors warrant brief review because of the substantial role the patient has in helping to manage risk: smoking, diet and nutrition, physical inactivity, and obesity and body fat distribution.
MODIFIABLE RISK FACTORS FOR STROKE
| Well-Documented Factorsa | Less-Documented Factorsb | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
| ||||||||||||||||||||
| |||||||||||||||||||||
Consumption of fruits and vegetables reduces the risk of stroke in a dose-dependent manner. Risk is reduced 6% for each serving of fruits and vegetables per day [105]. Reducing sodium intake and increasing antioxidants, potassium, and calcium also mitigates the risk of stroke [9,106,107]. The AHA/ASA recommend that individuals with stroke residing in long-term care facilities be evaluated for calcium and vitamin D supplementation [21]. The Dietary Approaches to Stop Hypertension (DASH) diet, which is rich in fruits, vegetables, and low-fat dairy products and which limits saturated and total fat intake, has been shown to lower blood pressure and likely reduce the risk of stroke [106,107].
Overwhelming evidence shows an undeniable association between cigarette smoking and stroke [9,108]. Smoking doubles the risk for ischemic stroke and increases the risk of hemorrhagic stroke 2 to 4 times [109]. The stroke risk for Black Americans who smoke is more than double that for nonsmokers in the same racial/ethnic group. A cohort study found stroke risk was 2.5 times higher for current smokers compared with never smokers [400]. The stroke risk was dose-dependent, according to smoking intensity; the risk increased 2.3 times for individuals who smoked 1 to 19 cigarettes per day and 2.8 times for those who smoked more than 20 cigarettes per day. Recurring exposure to environmental ("secondhand") smoke almost doubles the risk of stroke [110]. Secondhand smoke exposure is a contributing factor in more than 8,000 stroke deaths each year [111]. Using data from the National Health Interview Survey and death certificate data for 2000 through 2004, the Centers for Disease Control and Prevention estimated that smoking resulted in an annual average of 61,616 stroke deaths among men and 97,681 stroke deaths among women [112].
Individuals who smoke should be prescribed smoking cessation medications and be informed about cessation programs, counseling, and nicotine-replacement products. Epidemiologic studies show that, following smoking cessation, stroke risk decreases over time [9]. Individuals who do not smoke should be encouraged not to start.
One study has found that in the body mass index (BMI) range of 25 to 50 kg/m2, each 5 kg/m2 increase in BMI was associated with a 40% increased risk of stroke mortality; in the BMI range of 15 to 25 kg/m2, there was no relationship between BMI and stroke mortality [113]. Another large epidemiologic study showed that the risk almost doubled for overweight and obese men [114]. A meta-analysis of data from 25 studies involving more than 2.2 million people showed an increased risk of ischemic stroke of 22% in overweight individuals and 64% in obese individuals [115]. Studies have not yet addressed if losing weight reduces this risk [9]. However, it is well-documented that weight loss lowers blood pressure and cholesterol and positively affects other risk factors for stroke. Clinicians should encourage overweight patients to begin a weight-loss program that includes a healthful diet and exercise and provide patients with safe weight-loss strategies, such as those recommended by the AHA [9].
Body fat distribution has proven to be a fairly reliable indicator of stroke risk [116,117]. While some studies support abdominal adiposity as a significant risk factor for stroke only in men, other research indicates that a high concentration of abdominal fat is a significant risk factor in both men and women, independent of BMI [9]. On the other hand, gynoid adiposity in women appears to be associated with a lower risk for stroke, even for women with a higher BMI [117]. A high hip-to-waist ratio may be a more important risk factor for vascular disease, including stroke, than BMI score alone [118].
A large and generally consistent body of evidence indicates that routine physical activity prevents stroke [9]. A habitually sedentary lifestyle increases an individual's chance of stroke. Physically active men and women generally have a 25% to 30% lower risk of stroke or death than the least active people [119]. Individuals should be strongly encouraged to engage in aerobic exercise at moderate intensity for 150 minutes or more weekly, at vigorous intensity for 75 minutes or more per week, or a combination of both that fulfills these requirements [9]. It should be noted that only moderate-to-vigorous intensity exercise has been found to reduce the incidence of stroke [104].
To decrease the incidence of first-time stroke in the United States, primary prevention should focus on individuals at high risk with modifiable risk factors. In areas of the United Kingdom, the incidence of major stroke has been reduced 40% through a reduction in the incidence of risk factors [120]. The AHA has established evidence-based recommendations for primary prevention of stroke and has developed public campaigns and educational materials to help raise awareness of stroke.
For most individuals with nonmodifiable stroke risks, the probability of stroke can be decreased substantially with rigorous preventive measures and the treatment of modifiable risks [9,16]. The AHA recommends addressing lifestyle risk factors, as discussed, and medically managing several risk factors, including hypertension, diabetes, AF, other cardiac conditions, dyslipidemia, and asymptomatic carotid stenosis [9].
Hypertension
Hypertension is perhaps the most significant risk factor for stroke, and risk increases as blood pressure increases [9]. Sustained elevation in blood pressure has adverse effects on small and large arteries throughout the cerebrovascular system, increasing risk for multiple stroke types (ICH, aneurysmal subarachnoid hemorrhage, and ischemic stroke). Fortunately, the prevalence of hypertension has plateaued over the past decade and remained stable, at 29%, between 2008 and 2017 [121,122]. Control of hypertension (defined as blood pressure less than 140/90 mm Hg) also has improved, with rates of control increasing from 27.3% in 1988–1994 to 50.1% in 2007–2008 [9]. These improvements are likely attributable to heightened awareness and treatment, with awareness among U.S. residents increasing from 69% in 1988–1994 to 81% in 2007–2008 [9]. Still, more than two-thirds of people 65 years of age and older are hypertensive [123].
Treatment of hypertension is effective for primary and secondary prevention of stroke. The AHA and other professional organizations recommend that all adults 18 years of age and older be screened for high blood pressure [124]. The U.S. Preventive Services Task Force (USPSTF) recommends screening for all adults 18 years of age and older and annual screening for adults 40 years of age and older who are at increased risk for high blood pressure. Increased risk is defined as [124]:
High-normal blood pressure (130–139/85–89 mm Hg)
Overweight or obese
Black race
Individuals 18 to 39 years of age with normal blood pressure (<130/85 mm Hg) who do not have other risk factors should be screened every 3 to 5 years. The USPSTF additionally recommends rescreening with properly measured office blood pressure. If the blood pressure is elevated, the diagnosis should be confirmed with ambulatory blood pressure monitoring [124].
The 2017 Guideline for High Blood Pressure in Adults recommends screening every 2 years for adults with a blood pressure less than 120/80 mm Hg and screening every 3 to 6 months for people with systolic blood pressure of 120 to 129 mm Hg or with diastolic blood pressure greater than 80 mm Hg [125]. The AHA/ASA recommends that women be screened for high blood pressure before taking birth control pills, as the combination increases the risk of stroke [126].
Appropriate management of hypertension may also include dietary changes, other lifestyle modifications, and pharmacologic therapy. There is no evidence that any particular class of antihypertensives offers special protection against stroke in all patients; the desired result is more important than the choice of treatment. Treatment should be individualized on the basis of other clinical characteristics and medication tolerance. Among patients with other cardiovascular risk factors, more intensive control (systolic blood pressure <130 mm Hg) reduces risk of stroke more than usual control (<140 mm Hg) (9). Studies have shown that management is possible for the majority of patients, but most will require treatment that includes two or more drugs [9].
Diabetes
Diabetes, if untreated, leads to enhanced atherogenesis and microvascular disease within small arterioles. The risk for stroke is 2 times higher among individuals with diabetes. In 2017, after adjusting for population age differences, hospitalization rates for stroke were more than three times higher among adults 20 years of age and older with diagnosed diabetes compared with those without diagnosed diabetes [127]. Type 2 diabetes is associated with an increased prevalence of atherogenic risk factors, such as hypertension, obesity, and dyslipidemia. The combination of hyperglycemia and hypertension is thought to increase the risk of stroke [9]. The AHA recommends that the target blood pressure for individuals with diabetes be less than 140/90 mm Hg [9]. Pharmacologic therapy with angiotensin-converting enzyme (ACE) inhibitors or angiotensin receptor blockers (ARBs) has been shown to be safe and effective in this population [9]. In addition, lipid-lowering statins reduce the risk of first-time strokes in patients with diabetes, irrespective of the baseline lipid levels, pre-existing vascular conditions, and glycemic control [128].
Atrial Fibrillation
AF is associated with a fourfold to fivefold increased risk of ischemic stroke, and approximately 60,000 strokes occur among the 2.3 million individuals with AF annually [9]. According to the AHA/ASA, women older than 75 years of age should be screened for AF risk due to its link to greater stroke risk [126]. All patients with AF should be considered for anticoagulant therapy. Whether to use warfarin (a vitamin K antagonist) or a non-vitamin K-dependent, direct-acting anticoagulant depends on underlying predisposition, age, patient comorbidities and concomitant medications, bleeding risk, and stroke risk. High-risk factors for stroke include previous TIA/stroke, mitral stenosis, prosthetic heart valves, and systemic embolism. Moderate-risk factors include age older than 75 years, heart failure, hypertension, and diabetes. AHA guidelines recommend that eligible patients with nonvalvular AF be treated with warfarin or one of the direct non-vitamin-K oral anticoagulants [195]. Patients with AF and mechanical heart valves or mitral stenosis should only receive treatment with warfarin, as the direct non-vitamin K oral agents are contraindicated. The individual's preferences and access to high-quality anticoagulation monitoring should be considered. Warfarin therapy to maintain an international normalized ratio (INR) of 2.0 to 3.0 (target: 2.5) is strongly recommended for high-risk individuals (those with more than a 4% annual risk of stroke) with AF and no significant contraindications to oral anticoagulants [9,129]. Despite the effectiveness of such treatments, anticoagulant therapy continues to remain underprescribed due to overestimation of the associated risks of warfarin (e.g., intracranial, extracranial hemorrhage) and underestimation of stroke risk [129]. Some physicians err on the side of caution and aim for an INR greater than the recommended target, with a corresponding reduction in therapeutic effectiveness.
Other Cardiac Conditions
The management of valvular heart disease, unstable angina, chronic stable angina, acute MI, and other cardiac conditions is a critical factor in stroke prevention. Strategies to prevent postoperative neurologic injury and stroke in patients undergoing surgical revascularization for atherosclerotic heart disease can be found in the American College of Cardiology coronary artery bypass graft surgery guidelines, which are approved by the AHA [9,130]. The AHA states that it is "reasonable" to use warfarin for patients who have had ST-elevation MI and left ventricular dysfunction with extensive regional wall-motion abnormalities [9].
Dyslipidemia
The AHA recommends that statin therapy for individuals with known coronary heart disease or high-risk hypertension (e.g., patients with diabetes), including those with normal low-density lipoprotein (LDL) cholesterol levels [9]. In addition, these individuals should be encouraged at every interaction to exercise, eat a healthful diet, and quit smoking. Niacin, bile acid sequestrants, ezetimibe, or fibric acid derivatives may also be considered for individuals with known coronary heart disease and low levels of high-density lipoprotein cholesterol, such as people in whom target cholesterol levels cannot be achieved with statins or people who cannot tolerate statin therapy; however, their effectiveness in decreasing stroke risk has not been established [9].
Asymptomatic Carotid Stenosis
Individuals with asymptomatic carotid artery stenosis should be screened for other modifiable risk factors, and any risk factors identified should be controlled as soon as possible. Antiplatelet therapy with aspirin is recommended, unless contraindicated [9]. Carotid revascularization (endarterectomy or vascular stent) for stroke prevention is an option for asymptomatic patients with >70% stenosis of the internal carotid artery if the risk of perioperative stroke and other adverse outcomes is low (<3%). However, the effectiveness compared with best medical management is not well established [9,10]. Comorbidities and life expectancy should be considered when determining if surgery is appropriate. In addition, thorough discussion with the patient and his or her family/caregivers is a necessity. Topics should include the possibility of surgery-related death, the risks and benefits associated with the procedure, and the patient's preferences. For patients with a high surgical risk, carotid angioplasty/stenting may be considered. However, the periprocedural and overall 1-year event rates in some studies have dampened the AHA's enthusiasm for the stenting option [9,10].
Depressive Symptoms
After publication of the AHA guidelines, the findings of a large study showed that depressive symptoms are an independent risk factor for stroke, especially for patients younger than 65 years of age [131]. Clinicians may consider managing depressive symptoms and mood disorders as aggressively as hypertension or diabetes, because mood disorders appear to increase risk for all types of stroke [132].
Although public knowledge regarding the warning signs and risks of stroke has improved, the majority of the general public is still unaware that early treatment can prevent severe disability and death [133,134]. Estimates vary widely, however; the International Stroke Trial found that only 4% of patients with acute ischemic stroke arrive at the emergency department (ED) within 3 hours after the onset of symptoms, and a separate study found that 21% to 25% of individuals with acute ischemic stroke arrive at an ED within the same timeframe [135,136]. Of these individuals, 2% to 4% receive thrombolytic treatment [137,138]. It has been estimated that if all individuals called for emergency help at the onset of symptoms, as many as 29% could realistically receive treatment within 3 hours [137]. In addition, if all patients arrived at the ED within 1 hour after known symptom onset and received optimal treatment, the projected rate of thrombolysis would be 57%.
To improve the rate of early arrival in the ED, public education campaigns designed to help individuals recognize a stroke and seek early treatment often use the "five sudden warning signs" devised by the Brain Attack Coalition, or "FAST," a mnemonic device created by study investigators on the basis of the Cincinnati Prehospital Stroke Scale [139,140]. The AHA and the National Stroke Association and other organizations use FAST as a component of public education campaigns and stroke risk assessment promotion [141]. The mnemonic addresses the symptoms most common to an evolving stroke:
Face drooping
Arm weakness
Speech difficulty
Time to call 911
Other symptoms that comprise the five sudden warning signs include [140,141]:
Numbness or weakness of the face, arm, or leg (especially on one side)
Trouble seeing from one or both eyes
Severe headache with no known cause
Dizziness, difficulties with walking, and loss of balance and coordination
Confusion and trouble speaking or understanding
FAST was designed to focus on fewer common signs of stroke onset (face numbness, arm numbness, and slurred speech) and to include an action component (time) for lay persons who may have trouble recalling the warning signs and the appropriate action. A retrospective study exploring the capacity of the FAST campaign to facilitate the recognition of stroke suggests that it leads to the identification of approximately 89% of individuals who have a stroke or TIA [140]. The most common stroke symptoms were related to the face, arm, and speech/language. The same study found that a modified version of FAST (with removal of the word "numbness") decreased the number of TIAs identified and targeted ischemic stroke more readily than hemorrhagic stroke. Ultimately, it is unknown whether the general public is more likely to remember FAST or the five sudden warning signs.
In 1989, the United States Department of Health and Human Services Public Affairs launched a National Health Observance to help stimulate awareness of the risk factors, prevention, and early treatment of stroke. For more than 25 years, May has been recognized as National Stroke Awareness Month, with special campaigns to heighten awareness of stroke among the general public. Resources to aid community campaigns can be obtained from the American Stroke Association at https://www.stroke.org/en/about-stroke/stroke-symptoms.
Patient education should be presented in several forms and focus on modifiable risk factors, patients' needs, lifestyle, and life stage. Healthcare professionals can be most effective in reducing the risk of stroke when they demonstrate an interest in a patient's lifestyle and psychologic status. For instance, if a patient depends on his or her spouse or companion for meals, optimum benefits will result from educating the spouse/companion about healthy diet practices. Clinicians should also consider cultural needs when addressing prevention strategies.
When a patient/caretaker does not speak the same language as the clinician, a professional interpreter should be consulted to ensure accurate communication. A systematic review of the literature has shown that the use of professional interpreters provides better clinical care than the use of informal interpreters, with the former improving the quality of care for patients with limited English language skills to a level equal to that for patients with no language barriers [142]. Use of professional interpreters has been associated with improvements in communication (errors and comprehension), utilization, clinical outcomes, and satisfaction with care [142]. Individuals with limited English language skills have indicated a preference for professional interpreters rather than family members [143].
Whether the education involves stroke prevention, stroke recognition, care after stroke, coping with the effects of stroke, or palliative care, written materials are as important as verbal communication. Several organizations supply general or specialized educational resources, and many also provide patients and family/caregivers with psychosocial, financial, and assisted-living information or aid (Table 9). Clinicians should attempt to obtain materials written in languages appropriate for their patient population and, if appropriate, that target patients in high-risk racial/ethnic populations, especially American Indian/Alaskan native and Black individuals.
ORGANIZATIONS PROVIDING PATIENT EDUCATION RESOURCES ON STROKE
|
When determining the appropriate degree of risk management, information regarding an individual's risk of first stroke is valuable. The goals of risk assessment tools include [9]:
Identifying patients who are unaware of their elevated risk
Assessing the total risk of multiple factors
Discerning the utility of modifications and treatments
Projecting a quantitative risk in order to select appropriate treatments or stratification in clinical trials
Guiding appropriate use of diagnostic tests
The numerous nonmodifiable and modifiable factors that contribute to the risk of stroke have been discussed. Although many of these are independent risk factors, their interactions can affect predictions and management decisions in unexpected ways. No simple, validated stroke risk-assessment tool is currently available [9]. Although risk-assessment tools may have some utility, it is unknown if they improve primary prevention, especially when applied across subgroups according to age, gender, and race/ethnicity [9,144].
Because TIA is a substantial risk factor for a subsequent stroke, clinicians in many EDs are stratifying such patients by degree of risk with use of the ABCD or ABCD2 assessments [145,146]. The ABCD clinical tool is designed to predict 7-day risk of stroke through assessment of age (1 point for patients 60 years of age or older), blood pressure (1 point for a blood pressure greater than 140/90 mm Hg), clinical features (2 points for unilateral weakness with or without speech impairment or 1 point for speech impairment without weakness), and duration (1 point for 10 to 59 minutes, 2 points for greater than 59 minutes) [147]. The "2" designation in ABCD2 was added to represent the presence or absence of diabetes. The effectiveness of these screening tools is lessened by the fact that some individuals do not seek emergency care for a TIA or do not report a TIA to their clinician. However, the ABCD2 assessment has been shown to identify 21% of individuals with a high 2-day risk of having an ischemic stroke [145]. Individuals with high-risk TIA require the same intensity of evaluation and stroke prevention as individuals with ischemic stroke. Scores that predict future stroke risk should be used in conjunction with other diagnostic studies (e.g., imaging) and laboratory tests. The use of ED diagnostic protocols and observation units can reduce length of stay while improving patient treatment and reducing stroke rate [148,149].
Because the temporal window for effective stroke treatment is short, it is imperative that evaluation and diagnosis are performed promptly and accurately. Use of EMS by persons with stroke is associated with earlier ED arrival, faster ED evaluation, more rapid treatment, and more eligible patients receiving treatment. However, only 60% of all patients with stroke use EMS. In particular, male, Black, and Hispanic patients are less likely to use EMS [150]. The AHA/ASA has established several evidence-based recommendations for the diagnosis and early management of adult-onset ischemic stroke [16]. These recommendations address the evaluation of the individual before he or she arrives at an ED, diagnosis in the ED, the history and physical examination, laboratory tests, carotid ultrasonography, cerebral angiography, and imaging studies.
The single most important factor influencing the treatment of stroke within 3 hours after the onset of symptoms is the rapid triage and transportation provided by EMS [151]. Public health leaders and medical professionals should design and implement education programs that emphasize the need to rapidly seek emergency care when a stroke is suspected. These programs should be racially/ethnically, age, and sex/gender diverse and should target the public, healthcare providers, and hospital and EMS personnel to increase use of the EMS system, decrease stroke onset to ED arrival times, and increase the timely use of appropriate treatment [16].
Stroke assessment should begin with the EMS dispatcher [16]. When stroke or TIA is suspected, the dispatcher should notify the appropriate EMS provider and coordinate with an appropriate acute stroke treatment facility (Table 10). Regardless of the degree of the neurologic deficits, an individual with suspected stroke or TIA should be dispatched and triaged as if he or she were a serious trauma patient [16]. The goal is rapid transportation to the closest facility that can capably administer intravenous (IV) thrombolysis with alteplase, a recombinant tissue plasminogen activator (rt-PA); if possible, the individual should be taken to a designated stroke center [16,19]. The benefits of IV rt-PA are time-dependent, and treatment should be initiated as quickly as possible, within three hours of stroke onset and after brain imaging (e.g., noncontrast head CT) to screen for hemorrhagic stroke, embolic brain infarction, and tumor [16].
"STROKE CHAIN OF SURVIVAL" FROM THE 2013 AMERICAN HEART ASSOCIATION GUIDELINES
|
Initial EMS evaluation should assess breathing respiration, blood pressure, and positioning the patient to assure maintenance of the airway, Small clinical series shown that the lying-flat position may improve cerebral perfusion in patients with acute ischemic stroke when the intervention is initiated early after stroke onset [16]. EMS personnel should then initiate a prehospital evaluation [16,153]. If the individual exhibits common signs of stroke and/or a stroke is indicated by a validated prehospital examination tool (e.g., Los Angeles Prehospital Stroke Screen, Cincinnati Prehospital Stroke Scale), EMS providers should notify the ED that an individual with suspected stroke is in transport [16,153,154]. Any information about coexisting conditions and, most importantly, time of symptom onset should be provided in advance. A blood glucose level should also be determined, as symptoms of hypoglycemia may mimic those of a stroke [16].
Prehospital evaluation expedites the physician's evaluation of the patient on arrival in the ED. If possible, a witness (preferably a close family member or companion) should be transported with the patient to assist with patient history, symptom onset, and contact information. If no witness was present, a family member should be contacted to go to the hospital and should be informed that he or she may need to provide consent for the patient's treatment. History obtained by EMS providers should include [16]:
Information about recent events (e.g., stroke, MI, trauma, surgery, bleeding)
Comorbid diseases (e.g., hypertension, diabetes)
Use of medications (e.g., anticoagulants, insulin, antihypertensives)
The patient's medication containers should be transported to the ED as well, especially if medications include anticoagulant, antiplatelet, or antihypertensive drugs. Because 60% of individuals with suspected stroke do not use EMS for initial medical care access, ED staff should be alert to signs of stroke among individuals waiting to be seen in the ED [16,150].
Organization of the ED's stroke team and assessment protocol is paramount to maximize the likelihood of early and successful management [16]. The acute stroke team should include physicians, nurses, and laboratory/radiology personnel. After the patient has been triaged and stabilized, the inaugural ED evaluation (history and physical examination), laboratory studies, and CT imaging should be performed concurrently. The multimodal approach has three goals [16]:
Rapid and careful identification of ischemic stroke for treatment purposes
Determination of the underlying cause of the stroke for secondary prevention
Detection of stroke-mimicking conditions that may require immediate care
Within 1 hour of the patient's arrival, the evaluation (including a neurologic examination) and treatment decision should be completed. The AHA recommends that all patients receive a standardized battery of tests and procedures, with alternative tests performed only if a particular condition is suspected or the patient's history is incomplete (Table 11) [16]. Generally, diagnostic tests should be limited to save time. In addition, all diagnostic tests for stroke should be available 24 hours a day, 7 days a week.
IMMEDIATE DIAGNOSTIC STUDIES TO EVALUATE PATIENTS WITH SUSPECTED ISCHEMIC STROKE, AS RECOMMENDED BY THE AMERICAN HEART ASSOCIATION
| Population | Diagnostic Test |
|---|---|
| All patients | Cerebral computed tomography or magnetic resonance imaging (without contrast medium) |
| Blood glucose | |
| Serum electrolytes/renal function tests | |
| Markers of cardiac ischemia | |
| Complete blood count, including platelet count | |
| Prothrombin time/international normalized ratio (INR) | |
| Activated partial thromboplastin time | |
| Oxygen saturation | |
| Electrocardiography | |
| Selected patients | Hepatic function tests |
| Toxicology screen | |
| Blood alcohol level | |
| Pregnancy test | |
| Arterial blood gas tests (if hypoxia is suspected) | |
| Chest radiography (if lung disease is suspected) | |
| Lumbar puncture (only if stroke is suspected to be secondary to an infectious disease) | |
| Electroencephalography (if seizures are suspected) |
In addition to time of onset, other crucial historical data that should be obtained include [16]:
Information about atherosclerotic and cardiac disease risk factors
Prior and current drug abuse and history of migraine, seizure, infection, trauma, or pregnancy
Eligibility for treatment of ischemic stroke
If a patient presents with classic signs of stroke and has one or more cardiovascular risk factors, the diagnosis of stroke can be straightforward. However, identifying more unusual cases may be a challenge. If fever and a cardiac murmur are present, the cause of the stroke may be infective endocarditis [156,157]. Giant cell arteritis may be the cause if the patient is 50 years of age or older, has a prominent history of headache, and a strikingly elevated erythrocyte sedimentation rate. The presence of ptosis and miosis contralateral to the deficit may suggest carotid artery dissection [156,157]. If the symptoms were maximal at their onset, a subarachnoid hemorrhage or embolic stroke should be suspected [158]. In up to 40% of patients with subarachnoid hemorrhage, a severe headache (sometimes called a thunderclap or sentinel headache) that may abate within minutes or hours is the only symptom [158]. Head CT should be immediately performed in any patient with a suspected subarachnoid hemorrhage [36].
Following a structured neurologic examination, the general physical examination should include assessment of the head, neck, heart, lungs, abdomen, extremities, and skin [16]. Pulse oximetry and body temperature are also important evaluations. When performing the head and neck examination, the clinician should look for physical signs of trauma or seizure activity (e.g., contusions, tongue lacerations) or heart failure (jugular venous distention). Thrombolytic treatment is inadvisable for a seizure in the absence of acute ischemia detected by imaging [21].
When examining the patient's neck, carotid bruits may be heard. Although a bruit may be indicative of carotid atherosclerosis, confirmatory diagnosis and degree of carotid stenosis requires imaging with Doppler or duplex ultrasound [159]. Because the prevalence of carotid bruit increases with age, its specificity as a clinical marker of stroke risk in asymptomatic individuals 65 years of age or older has been questioned [160]. In a symptomatic patient with acute TIA/stroke event, a focal carotid bruit increases the likelihood of significant (>70%) carotid stenosis, which has implications for secondary stroke prevention. However, the absence of bruit is not reliable for concluding the carotid vessels are normal.
The cardiac examination may demonstrate contributing factors or comorbidities, such as myocardial ischemia, valvular conditions, arrhythmias, or aortic dissection (a rare cause of ischemic stroke). The diagnostic evaluation should include electrocardiography (ECG), because cardiovascular conditions such as AF and MI are prevalent among individuals with stroke [16,155]. The findings of studies have suggested that the value of chest x-ray as part of the diagnostic workup is debatable, and the 2019 AHA/ASA guideline recommends a chest x-ray only if lung disease is suspected [16]. Although cardiac monitoring of patients with stroke has not been evaluated, the AHA/ASA recommend its use for the first 24 hours to monitor for AF and MI, either of which can lead to stroke or be a dangerous coexisting condition [16].
Severe elevation in blood pressure (systolic >210 mm Hg and/or diastolic >120 mm Hg) may signal hypertensive encephalopathy and the possibility of cerebral hemorrhage [123,161]. Examination of the lungs and abdomen may reveal additional clues to pathogenesis of the stroke. The skin and extremities should be examined for signs of systemic conditions, including coagulopathies, platelet disorders, or liver dysfunction [16].
If only historical information and/or physical examination are relied on, up to 19% of stroke mimics are mistaken for stroke. Yet, a patient's history and examination may also identify a condition masquerading as a stroke (Table 12) [16,162]. The use of MRI or CT with laboratory studies enhances the accuracy of diagnosis, but detecting mimics with imaging techniques is a challenge if the patient has a history of stroke [16].
COMMON NONCEREBROVASCULAR CONDITIONS THAT MIMIC STROKE
| Stroke Mimic | Differential Symptoms | ||||||
|---|---|---|---|---|---|---|---|
| Brain tumor | Gradual progression of symptoms | ||||||
| Drug overdose | Altered mental status without focal findings | ||||||
| Conversion disorder |
| ||||||
| Hypertensive encephalopathy |
| ||||||
| Hypoglycemia |
| ||||||
| Migraine with aura (complicated migraine) |
| ||||||
| Seizures/postictal paresis |
| ||||||
| Drug toxicity | History of lithium, phenytoin, or carbamazepine use |
Another important component of the physical examination is assessment with the National Institutes of Health Stroke Scale (NIHSS). Although initially devised as a research tool to quantify neurologic deficits, this tool is now widely used to measure the severity of a stroke, devise an effective treatment plan that establishes a priority for patient safety, identify the affected vascular region, and predict patient outcome [16,164]. The NIHSS is standardized, reliable, and fast and facilitates communication among healthcare professionals. Any trained healthcare professional can use the scale at the bedside in 5 to 8 minutes. In addition, results are based only on patient evaluation; a history or information from others is not necessary. It is recommended that the scale be used to assess the patient during the ED evaluation and during treatment with recombinant tissue plasminogen activator (rt-PA) or other therapies and that it be repeated often in the first 24 hours. Further information about the NIHSS and a copy of the scale can be obtained from the National Institute of Neurological Disorders and Stroke [165].
For all patients with suspected stroke, the battery of diagnostic tests should be carried out concurrently with laboratory tests, including blood glucose level (to rule out hypoglycemia as a stroke mimic), complete blood count (with platelet count), serum electrolyte levels, renal function studies, and cardiac ischemia biomarkers [16]. Prothrombin time and partial thromboplastin time determinations should be included when considering thrombolysis. Generally, waiting for the results of a diagnostic test should not be a reason to delay thrombolytic therapy [16]. The risk of increased neurologic damage and death caused by ischemic stroke outweighs that of a secondary hemorrhage except when a bleeding or blood disorder is suspected, the patient was given warfarin or heparin, or the patient takes anticoagulant drugs.
Electrocardiography and telemetry monitoring are warranted in all patients with ischemic stroke and TIA to screen for AF and silent MI. Studies show that initial assessment detects AF in as many as 7% of patients in this clinical setting; longer cardiac monitoring yields higher detection rates, especially among patients with heart disease [402].
All patients with suspected acute stroke should receive emergency brain imaging evaluation on first arrival to a hospital before initiating any specific therapy to treat ischemic stroke. The purpose is to exclude ICH in patients who are potentially eligible for IV fibrinolysis (tPA) or mechanical thrombectomy or both [16]. High diagnostic accuracy of stroke and treatment decisions is optimized by the use of imaging tools such as CT and MRI. Collectively, these neuroimaging options provide detailed information that influences stroke treatment decisions, including infarct age, severity, and distribution; intracranial vascular status; cerebral hemodynamics; estimated reversibility of ischemic damage; and hemorrhage type and distribution [16]. For instance, a widespread distribution of early infarction or edema increases hemorrhagic transformation risk following thrombolytic therapy. CT without contrast medium is readily sensitive to these conditions [16].
For prompt and accurate diagnosis, the use of noncontrast CT (NCCT) or DW-MRI is recommended for initial imaging in a patient with suspected stroke [16,173]. Although NCCT is less expensive, faster, and more widely available, DW-MRI more readily detects small cortical, subcortical, and posterior fossa infarcts and distinguishes chronic ischemia from acute ischemia. MRI is also sensitive to subclinical satellite ischemic lesions that characterize the pathophysiology of the stroke [152,180]. As stated, DW-MRI can detect acute ischemic changes within minutes after the onset of stroke; CT may not detect a zone of ischemic change for some hours after the event [16]. For patients with acute ischemic stroke who are candidates for thrombolysis (0- to 4.5-hour time window), NCCT or MRI of the head is recommended to exclude ICH and determine extent of ischemic changes, while ensuring that patients receive timely IV rt-PA [16]. The AHA recommends that CT imaging be obtained within 25 minutes after the patient's arrival in the ED [152]. If a patient is eligible for rt-PA, treatment should be initiated as quickly as possible without waiting for further imaging [16].
Once a decision regarding thrombolytic therapy has been reached, multimodal CT and MRI are useful for refining the etiologic diagnosis, confirming location and extent of ischemic injury, and selecting patients who may benefit from endovascular therapy within a 6- to 24-hour window. Multimodal brain MRI is the preferred method to evaluate for acute ischemic infarct, should be obtained within 24 hours of symptom onset, and, in most centers, will follow NCCT. In centers with rapid access to MRI in the ED, DW-MRI might be substituted for NCCT as initial imaging procedure in stable patients. The distribution of DW lesions helps identify the stroke pathogenesis (e.g., a single lesion in a deep structure suggests small vessel disease; scattered lesions in multiple territories points to cardioembolism) [402].
Computed tomographic angiography (CTA), magnetic resonance angiography (MRA), and duplex ultrasound or transcranial Doppler are noninvasive imaging modalities that, in selected patients with TIA/stroke, are used for identification of high-grade carotid stenosis and intracranial vascular occlusions amenable to endovascular therapy. Stenosis/occlusion of major vessels carries a high risk for recurrent stroke. CTA has high sensitivity and specificity for detecting intracranial arterial stenoses and embolic occlusions. MRA has the advantage of three-dimensional views, detects vascular patency and can be extended to assess the intracranial vasculature. Imaging strategies for selecting patients under consideration for endovascular therapy fall into three categories [173]. The first is NCCT, followed immediately by digital subtraction angiography (DSA), which enables assessment of vascular patency, demonstrates the collateral pattern, may indicate infarct volume, and has the advantage of minimizing door-to-recanalization time. The second strategy is to obtain a CT angiogram to assess vascular patency, with or without perfusion imaging, in order to better characterize the size of occlusion and zone of ischemic tissue before deciding whether to proceed with an endovascular intervention. The third option is MRI and MRA, possibly with diffusion- and perfusion-weighted imaging where it can be performed quickly and on a constant basis [173]. In selecting candidates for mechanical thrombectomy within 6 to 24 hours after last known well time, AHA/ASA guidelines recommend using either CTA with computed tomographic perfusion (CTP) or MRA with DW-MRI [16].
Conventional catheter angiography is invasive and requires a nephrotoxic contrast medium [169]. It is particularly valuable for the detection of vasculopathies associated with cerebral complications, including aneurysm, vasculitis, arteriovenous malformation, atherosclerosis, and arterial dissection [10,16]. Digital subtraction angiography (DSA), a type of catheter angiography, remains the standard against which newer technologies for detection of many types of cerebrovascular lesions and diseases are measured [170,171,172,173,174]. Its sensitivity and specificity equal or exceed those of noninvasive techniques; however, because of time considerations and procedural difficulties, it is not recommended in initial evaluation of stroke patients [16].
Unlike catheter angiography, MRA creates a map of blood flow rather than a true image of the vasculature anatomy. Noninvasive time-of-flight MRA is performed without contrast medium, and like phase-contrast (gadolinium) MRA is minimally invasive (catheter insertion required). Although MRA provides a high-resolution image of many vessels, it cannot reliably resolve distal or branch occlusions of the intracranial vasculature [16]. MRA also tends to overestimate the degree and length of high-grade stenosis (compared with DSA) by signaling loss of flow when blood flow is turbulent, in-plane, or slow [175]. MRA is helpful for detecting uncommon causes of ischemic stroke or TIAs (e.g., arterial dissection, venous thrombosis) [172]. The 2019 AHA/ASA guideline recommends the use of MRA with DW-MRI when selecting candidates for mechanical thrombectomy between 6 and 24 hours after last known well time [16]. DW-MRI is sensitive to acute cellular injury in cerebral ischemia and can be used to assess ischemic lesions in the first few hours [177].
CTA also permits noninvasive, rapid screening of intracranial and extracranial vasculature for stenosis and occlusion. Preliminary data suggest that CTA effectively detects large-vessel intracranial occlusions when compared with ultrasound and DSA; however, because it provides a static image of vascular anatomy, it is inferior to DSA for demonstrating flow rates and direction [16,178]. Although CTA is fast and can be adapted to conventional CT, contrast medium is required, and patients are exposed to additional radiation [16]. The 2019 AHA/ASA guideline recommends the use of CTA with CTP or MRA/DW-MRI for selecting candidates for mechanical thrombectomy between 6 and 24 hours after last known well time [16].
Doppler ultrasonography is a noninvasive imaging technique used to measure arterial blood flow and determine the site and degree of stenosis/occlusion of the external carotid and cerebral vasculature. A common use of transcranial Doppler ultrasonography is for primary and secondary stroke prevention in patients presenting with TIA and stroke. It can be used to monitor progress of vascular recovery during the early post-acute thrombolysis treatment phase of stroke care [10,16,166]. Additional uses of transcranial Doppler ultrasonography in patients with stroke include [16,166,167]:
Detection of intracranial and extracranial vascular disorders
Assessment of recurrent stroke risk (e.g., microemboli detection)
Identification of candidates for intensive prophylactic interventions (e.g., carotid endarterectomy, angioplasty/stenting)
Intraoperative monitoring of carotid endarterectomy
Detection of right-to-left shunts
Identification of subclavian steal syndrome
Measurement of a post-subarachnoid hemorrhage vasospasm
Assessment of recanalization (spontaneous or thrombolytically induced)
Prognosis of patients with stroke (performed during post-acute phase)
Despite the usefulness of this technique, its performance is highly operator-dependent [166]. Considerable anatomic and physiologic knowledge of the cerebral vasculature is required, as vessel images are not produced, and skill and experience are prerequisites for data interpretation. Data acquisition and interpretation tend to be time-consuming and may delay treatment [166,168]. While these reasons suggest that vascular imaging is not practical for the diagnosis of ischemic stroke in most cases, it may be useful in some. A recommendation in the 2019 AHA/ASA guidelines states that noninvasive imaging of the cervical carotid arteries should be routinely performed within 24 hours of admission in patients with nondisabling acute ischemic stroke in the carotid territory who are candidates for carotid endarterectomy [16]. Additionally, intracranial vessel imaging is recommended to guide selection of appropriate secondary stroke prevention treatments [16].
Transcranial Doppler (TCD) ultrasonography has been used to detect intracranial vessel abnormalities and to evaluate occlusions and stenoses in intracranial vessels. It is less accurate than CTA or MRA for steno-occlusive disease but can detect microembolic signals that indicate extracranial or cardiac sources of embolism [152]. TCD has been shown to predict and enhance outcomes with IV rt-PA [179].
Because the time between the onset of stroke and treatment should be limited, catheter angiography is not recommended by the AHA for the diagnosis of a suspected stroke [152]. In the case of a stroke, the utility of the imaging tool increases after a diagnosis has been made. As with transcranial Doppler ultrasonography, cerebral angiography is particularly useful for confirming the diagnosis, monitoring the progression of thrombolytic therapy, and assisting with the prognosis, particularly during the first 72 hours after a stroke [152].
Because strokes are heterogeneous, are associated with multiple medical complications, and are time-critical, their effective management depends on organized and comprehensive care. Such treatment is optimally provided in comprehensive stroke centers and stroke systems of care. In 2007, the AHA/ASA published evidence-based guidelines to provide recommendations for treatment. These guidelines were updated in 2013 and again in 2019 [16,152].
Comprehensive stroke centers are designed to accommodate the needs of patients with complicated forms of stroke, intracranial hemorrhages, and subarachnoid hemorrhages, as well as patients in need of aggressive intervention measures and intensive care [19]. In general, primary and comprehensive stroke centers have been shown to be associated with better adherence to evidence-based guidelines and with an increased use of IV rt-PA [181]. Nationally, only 2% to 3% of individuals with stroke are treated with rt-PA, but the rate is typically greater than 10% at primary and comprehensive stroke care centers [19,138]. Overall care may also be improved at comprehensive stroke centers [181,182]. A formal certification process for comprehensive stroke centers has been established by the Joint Commission and the ASA. The Joint Commission has been certifying primary stroke centers since 2004, and it began providing certification for comprehensive stroke centers in 2014 [16]. Because patients are more likely to receive thrombolytic therapy at primary and comprehensive stroke centers, many states have enacted policies mandating the direct routing of individuals with suspected stroke (with onset of symptoms less than 3 hours previously) to either of these types of facilities. As of 2015, 1,505 of the 4,640 general hospitals and emergency rooms in the United States have been certified as primary stroke centers [183]. Seventy-four percent of the primary stroke centers have been certified by the Joint Commission and the AHA/ASA, 20% have been certified by state organizations, and 6% have been certified by other organizations. The highest proportion of primary stroke center certifications has occurred in the Northeastern United States [183]. Telemedicine for stroke (also called telestroke) and air transport are being increasingly used to serve individuals in rural areas that lack local stroke expertise [16,182].
Stroke centers are commonly part of a comprehensive support network known as a stroke system of care [16]. The system seeks ways to coordinate the highest quality of stroke prevention, community education, EMS, acute care, and post-acute care. Without a system of care, these components often operate less effectively and in isolation. Implementation of a stroke system of care in underserved regions could substantially improve treatment statistics statewide or nationwide; for example, one study showed that the additional resources at as few as six target hospitals in the western part of North Carolina would increase patient access to stroke care by 61.5% throughout the state [184].
Guidelines for establishing a stroke system of care were developed by an ASA Task Force [20]. Because of the multidisciplinary aspects of a stroke system, Task Force members were experts in areas of stroke prevention, EMS, acute stroke care, rehabilitation, and healthcare policy. Overall, the recommendations promote the communication and collaboration of patients, clinicians, facilities, and agencies. General ASA recommendations for stroke systems of care are [20]:
Support local/regional educational initiatives designed to increase stroke awareness among the general population, with enriched targeting of populations at increased risk of stroke and poor post-stroke outcomes.
Design/implement innovative behavioral interventions that address barriers to healthy behaviors and prevention adherence.
Design/implement public education programs that are repetitive, designed to reach diverse populations, and focused on stroke systems and the need to urgently seek emergency care (by calling 911).
Develop triage protocols that ensure all patients with known or suspected stroke are rapidly identified and assessed with validated stroke screening instrument.
All stroke center certification systems should meet or exceed the standards set by nationally certified stroke centers and should work regionally in an integrated way to provide and share best practices.
Adopt approaches to secondary prevention that address all major modifiable risk factors and are consistent with national guidelines for patients with a history or suspected history of stroke or TIA.
Standardize postacute care and discharge. Trained stroke nurses, nurse practitioners, social workers, community health workers, and others play a critical role in this process.
Advance the use of technology and patient-reported outcomes to facilitate improvements in stroke care transitions.
Enact federal-level policies that standardize the organization of stroke care throughout the continuum.
Several important considerations apply to the management of all patients with stroke in any acute clinical care venue. In the early period after sustaining a stroke, patients are often unable to communicate effectively or move voluntarily to change position in bed. Steps should be taken to assure maintenance of the airway, and attention should be paid to proper positioning (e.g., raising head of the bed) to reduce the risk of aspiration. If there are signs of hypovolemia, IV fluid should be administered to assure normal blood pressure and circulating blood volume, which are necessary to optimize circulation to the region of ischemic injury. Blood pressure is frequently elevated at presentation and for some days afterward. A certain degree of restraint is required for managing hypertension safely in this setting. This is because cerebrovascular autoregulation is impaired following occlusion of a major cerebral artery, and normalizing the blood pressure too quickly may result in reduced blood flow around the infarct penumbra, making the ischemic deficit worse. While it may be prudent to treat hypertension, the goal is gradual improvement without normalization. For safe administration of rt-PA, blood pressure should be <180/<105 mm Hg and maintained so for at least 24 hours [16].
Hypertensive encephalopathy ("malignant hypertension") does require vigorous treatment and is potentially confusing, as patients present with acute neurologic symptoms and severe hypertension. The pathogenesis and clinical signs differ from stroke, though the syndrome may be complicated by ICH. Severe hypertension with mean arterial pressure in excess of 110–120 mm Hg leads to vasogenic cerebral edema. Patients present with headache, global signs of encephalopathy (e.g., vomiting, lethargy, confusion), blurred vision, and papilledema. Blood pressure is usually >220 mm Hg systolic and >120 mm Hg diastolic. Hypertensive encephalopathy is a medical emergency treated with IV antihypertensive agents to prevent further deterioration (e.g., rostro-caudal brain herniation) and death. In those with prior history of hypertension, the goal is to restore blood pressure to previous control levels.
Thrombolytic therapy with rt-PA is the only treatment approved by the U.S. Food and Drug Administration (FDA) for ischemic stroke [16]. Anticoagulant and antiplatelet agents are also used, but their appropriateness is a source of debate and ongoing research. Intra-arterial rt-PA may be beneficial for select patients; however, the drug is not FDA approved for this use [18]. Mechanical thrombectomy is a consideration as both a primary reperfusion strategy and in conjunction with pharmacologic fibrinolysis [16].
The AHA/ASA recommendations for the treatment of ischemic stroke are based on review of published studies and emphasize the importance of early management [16,18]. Since publication of the 2013 guidelines, substantial new high-quality evidence on the clinical efficacy of endovascular treatments has become available. This new evidence is the basis of the AHA/ASA 2015 focused update to the 2013 guidelines and is included, where appropriate, in the recommendations that follow [18].
The IV administration of alteplase (rt-PA) has been FDA-approved for the treatment of stroke since 1996. Rapid administration of rtPA to appropriate patients remains the mainstay of early treatment of acute ischemic stroke [18]. Treatment with rt-PA is highly effective if administered within 3 hours. The earlier treatment is initiated, the higher the probability of a full recovery. Treatment within 90 minutes has been associated with a higher rate of favorable outcome at 3 months compared with treatment administered within 180 minutes [185]. For patients who meet national and international eligibility guidelines, IV rtPA improves functional outcomes at 3 to 6 months when given within 4.5 hours after ischemic stroke onset and should be administered [18]. However, the therapeutic window may extend to 6 hours [186,187]. Studies to determine the threshold of rt-PA benefits are ongoing. The AHA/ASA recommend that health systems set a goal of increasing their percentage of stroke patients treated within 60 minutes of presenting to hospital (i.e., door-to-needle time of 60 minutes) to at least 80% [152]. Tenectaplase is a newer tissue plasminogen activator investigated for treatment of ischemic stroke; it appears to be as effective as alteplase and may have a better safety profile [16].
The AHA/ASA have changed some of their recommendations regarding rt-PA treatment since their 2013 guidelines (Table 13) [16]. Administration of rt-PA is not recommended for patients who have a systolic blood pressure greater than 185 mm Hg or a diastolic blood pressure greater than 110 mm Hg [16].
AMERICAN HEART ASSOCIATION/AMERICAN STROKE ASSOCIATION RECOMMENDATIONS FOR THROMBOLYTIC THERAPY
| Class I Recommendations | |||||||||||
| |||||||||||
| Class II Recommendations | |||||||||||
| |||||||||||
| Class III Recommendations | |||||||||||
| |||||||||||
| MCA=middle cerebral artery; CMB=cerebral microbleed; MRI=magnetic resonance imaging; sICH=symptomatic intracerebral hemorrhage. |
Between 31% and 50% of patients treated with rt-PA have a 4-point or greater improvement on the NIHSS by 3 months after the stroke [152]. These clinical improvements do not recede for at least 1 year after the stroke. In general, the best response to rt-PA has been found for patients who are younger than 75 years of age, with a baseline NIHSS score of less than 20, and no history of diabetes or pre-existing disability [152].
The most common serious medical complication of rt-PA is secondary brain hemorrhage, which occurs in 6% of patients [188]. Yet, the risk does not outweigh the benefits of rt-PA. Three months following rt-PA therapy, approximately 30% of patients are neurologically normal or near normal; 30% have mild-to-moderate neurologic deficits; 20% have moderate-to-severe deficits; and 20% have died [189]. Other dangerous complications of rt-PA, although rare, are angioedema, anaphylaxis, systemic hemorrhage, and, if rt-PA is administered soon after an acute MI, myocardial rupture [16,190].
The AHA/ASA Task Force reviewed and discussed several studies addressing the use of heparin or low-molecular-weight heparin (LMWH) as an adjunct to a thrombolytic agent in the treatment of stroke [16]. In general, the Task Force concluded that early administration of heparin or LMWH is inadvisable partly due to the increased risk of bleeding complications, especially the hemorrhagic transformation of ischemic strokes. Additionally, early administration has not been shown to prevent recurrent stroke, lessen the risk of neurologic worsening, or improve patient outcome [16].
Although no new data regarding antiplatelet treatment have emerged since the 2003 version of the AHA/ASA guideline for ischemic stroke management, the AHA/ASA recommendations for antiplatelet therapy have changed (Table 14) [16]. Data combined from two large clinical trials suggest that administration of aspirin (325 mg) within 24 to 48 hours after the onset of stroke slightly reduces mortality and morbidity by preventing early recurrent stroke in some patients [191,192]. The findings of these trials do not suggest the use of aspirin within 24 hours of thrombolytic administration or as a substitute for thrombolytic therapy. A 2014 Cochrane review confirmed the results of these trials [193]. A review that summarized the results of nine randomized controlled trials also confirmed these findings and demonstrated increased odds of complete recovery [194]. In patients presenting with minor (NIHSS ≤3) noncardioembolic stroke who are not treated with rt-PA, dual antiplatelet therapy (aspirin and clopidogrel) started within 24 hours after symptom onset and continued for 21 days is effective in reducing risk of recurrent stroke for a period up to 90 days [16].
AMERICAN HEART ASSOCIATION/AMERICAN STROKE ASSOCIATION RECOMMENDATIONS REGARDING ANTIPLATELET TREATMENT FOR ISCHEMIC STROKE
| Class I Recommendations | |||
| |||
| Class II Recommendations | |||
| The efficacy of intravenous tirofiban and eptifibatide is not well established (Class IIb, Level of Evidence B). Further trials are necessary to clarify the safety and efficacy of this intervention. | |||
| Class III Recommendations | |||
|
Ticagrelor is a reversible, short-acting P2Y12 agonist. The Acute Stroke or Transient Ischemic Attack Treated with Aspirin or Ticagrelor and Patient Outcomes (SOCRATES) trial tested the efficacy of ticagrelor (18-mg loading dose, then 90 mg twice daily) to aspirin in 13,199 patients with acute ischemic stroke or high-risk TIA [196]. The primary outcome (i.e., time to stroke occurrence, MI, or death within 90 days) occurred in 6.7% of patients treated with ticagrelor versus 7.5% treated with aspirin. Ischemic stroke occurred in 5.8% in the ticagrelor arm versus 6.7% in the aspirin arm. There were no differences in major bleeding, intracranial hemorrhage, or fatal bleeding. A subgroup analysis of the SOCRATES trial found that ticagrelor was effective at preventing the primary outcome in patients with a background history of aspirin use [197]. Ticagrelor versus placebo was the subject of the THALES (Acute Stroke or Transient Ischemic Attack Treated with Ticagrelor and ASA for Prevention of Stroke and Death) trial, which was completed in December 2019. As of 2020, the results have not yet been published [198].
Several trials have examined the efficacy of clopidogrel plus low-dose aspirin [199,200,201,202,203,204]. While the data from two of these trials suggest a benefit from dual antiplatelet therapy (i.e., clopidogrel/aspirin), the results of the other trials suggest a lack of benefit and possibly harm in the longer term. The combination dipyridamole plus aspirin for ischemic stroke management has been studied in several trials involving more than 17,000 patients [205,206,207,208,209,210]. The combination was found to be an acceptable antiplatelet therapy for patients with ischemic stroke or TIA and probably superior to aspirin alone. However, twice-daily dosing and headache cause many patients to discontinue the regimen [207,208,209,210].
The efficacy of IV glycoprotein IIb/IIIa receptor blockers (GP IIb/IIIa inhibitors), such as abciximab, in combination with other interventions or alone is under investigation. Preliminary results from the Abciximab in Emergent Stroke Treatment Trial (AbESTT) indicate that IV GP IIb/IIIa inhibitors may accelerate spontaneous recanalization, improve microvascular patency, and offer an adequate safety profile [211,212]. However, a systematic review of GP IIb/IIIa inhibitors as well as results of the AbESTT-II (a phase III trial of abciximab) did not demonstrate either safety or efficacy of the drug for treatment of acute ischemic stroke and found an increased rate of symptomatic or fatal intracranial hemorrhage, with no reduction in death or disability in survivors [213,214]. The 2019 AHA/ASA guideline contains both revised and new recommendations regarding the use of IV GP IIb/IIIa inhibitors, including abciximab (Table 14) [16].
Although emergent angioplasty and stenting are high-risk procedures, progressing strokes, which occur when patients' moderate neurologic deficits deteriorate significantly within 72 hours after onset, are associated with very poor outcomes and high mortality rates [215]. Therefore, some case studies suggest that emergency angioplasty followed by immediate or delayed stenting is appropriate for patients with a progressing stroke caused by carotid artery occlusion or stenosis, respectively [216,217]. However, the safety and efficacy of emergency carotid artery stenting are not established, and the procedure remains controversial [218,219].
Angioplasty and stenting may be appropriate for patients with acute stroke secondary to carotid artery dissection [220,221]. In one study, emergency angioplasty and stenting of the internal carotid artery performed in conjunction with intra-arterial thrombolysis was associated with more favorable outcomes than pharmacologic treatment alone in patients with acute carotid artery occlusion and secondary artery-to-artery embolism to the middle cerebral artery [222]. In a larger study by the same group of investigators, treatment with urokinase followed by angioplasty and stenting increased recanalization [223]. Another study found that angioplasty and stenting of the proximal occlusion and stent-based thrombectomy of the intracranial occlusion may be feasible, effective, and safe in selected patients with acute internal carotid artery occlusion and concomitant major vessel embolic stroke [221].
The AHA/ASA assert that the use of angioplasty and intra-arterial thrombolytics in the emergency management of stroke should be limited to comprehensive stroke centers, which have the resources and physician expertise to perform them safely, and in the setting of clinical trials [18].
In addition to use in emergent angioplasty and stenting, mechanical thrombectomy is both a primary reperfusion strategy and an adjunct to pharmacologic fibrinolysis for achieving recanalization in patients with acute ischemic stroke [16]. Mechanical treatments include the use of catheters during angiography to directly deliver either a clot-disrupting or retrieval device to an artery-occluding thromboembolus [224].
The primary advantage of mechanical devices is their ability to remove a clot in a matter of minutes, compared with pharmacologic thrombolytics (even those delivered intra-arterially) that may take as long as two hours to dissolve the clot [225,226]. A second advantage is that newer devices (e.g., retrievable stents) have shown higher recanalization rates and better outcomes than those seen with older devices (e.g., the Merci Retriever) [224]. The primary disadvantage of endovascular therapy is the delay in initiation of treatment because of the time required to mobilize the interventional team and, in many cases, the need to transfer the patient to another hospital [227,228]. In the absence of sufficient trial data, it had been uncertain whether endovascular therapy, with or without the previous use of IV rt-PA, would be more effective than IV rt-PA alone [229]. However, newer trials (i.e., MR CLEAN, EXTEND-IA, ESCAPE, and SWIFT-PRIME) have demonstrated the efficacy of endovascular therapy using the newer, retrievable stents [230,231,232].
In addition to reviewing the results of these four trials, the AHA/ASA also reviewed the results of the REVASCAT trial [18,233]. Of the five stent retriever trials, MR CLEAN, ESCAPE, and SWIFT-PRIME permitted use of salvage intra-arterial fibrinolytic drugs, whereas EXTEND-IA and REVASCAT did not [18]. Every or nearly every patient in the trials first received IV rt-PA. All five studies enrolled participants 18 years of age and older. Four of the trials used NIHSS scores (>2, >5, and 8–29) as eligibility criteria, and the fifth trial enrolled patients with a similar distribution of NIHSS scores. Four of the five trials used a prestroke function eligibility criterion. All five trials required baseline nonenhanced CT or MRI and used different strategies of an imaging-based selection criterion in addition to nonenhanced CT or MRI. A large majority of patients in the trials had ICA or proximal MCA (M1) occlusion. All five trials allowed the inclusion of patients with proximal cervical carotid stenosis, and all but one trial (SWIFT-PRIME) allowed the inclusion of patients with complete atherosclerotic cervical carotid occlusion. General anesthesia and conscious sedation were the two most frequently used anesthetic approaches for patients with acute ischemic stroke receiving endovascular therapy. None of the trials established the usefulness of mechanical thrombectomy devices other than stent retrievers [18].
The AHA/ASA analysis of and conclusions about these five stent retriever trials form the basis of their 2015 focused update to the guidelines on the management of patients with acute ischemic stroke (Table 15) [18].
AMERICAN HEART ASSOCIATION/AMERICAN STROKE ASSOCIATION RECOMMENDATIONS REGARDING ENDOVASCULAR INTERVENTIONS FOR ISCHEMIC STROKE
| Class I Recommendations | |||||||||||
|
Eligible patients should receive intravenous rt-PA even if endovascular treatments are being considered (Class I, Level of Evidence A). Patients should receive endovascular therapy with a stent receiver if they meet all the following criteria (Class I, Level of Evidence A):
| |||||||||||
| Class II Recommendations | |||||||||||
| |||||||||||
| Class III Recommendations | |||||||||||
| Observing patients after intravenous rt-PA to assess for clinical response before pursuing endovascular therapy is not required to achieve beneficial outcomes and is not recommended (Class III, Level of Evidence B). |
RECOMMENDATIONS RATING SCHEME
| Class/Level | Definition |
|---|---|
| Classification of Interventions | |
| I | Conditions for which there is evidence for and/or general agreement that the procedure or treatment is useful, effective, and beneficial |
| IIa | Conditions for which there is evidence for and/or general agreement that the procedure or treatment may be useful, effective, and beneficial |
| IIb | Usefulness/efficacy is less well established by evidence or opinion |
| III | Conditions for which there is evidence and/or general agreement that the procedure or treatment is not useful/effective and in some cases may be harmful |
| Level of Evidence | |
| A | Data derived from high-quality multiple randomized clinical trials or meta-analyses of high-quality randomized clinical trials |
| B | Data derived from a single randomized trial or nonrandomized studies |
| C | Randomized or nonrandomized observational or registry studies of limited design or execution or meta-analyses of such studies |
| E | Consensus opinion of experts |
In the setting of acute ischemic stroke, justification for emergent (within the first 24 hours) or early revascularization with carotid endarterectomy (CEA) is based on reports of increased risk of recurrent stroke in patients undergoing medical therapy while awaiting revascularization. Some studies have found that CEA is most beneficial when performed within 2 weeks of the last cerebrovascular symptom and that the benefits decline rapidly after 3 weeks [194,234]. However, the risk associated with emergency CEA is believed to be high, for several reasons, particularly in patients with an unstable neurologic status [235]. First, embolic and hemodynamic injuries can occur [236]. Second, detection of an arterial lesion and mobilization of an operating room staff is time-intensive. Lastly, hyperperfusion, which occurs in 0.3% to 1.2% of patients who have CEA, can lead to brain edema and hemorrhagic transformation [236]. Other complications may also develop.
For some patients, however, the benefit of CEA may outweigh the risk. For instance, patients with acute ischemic stroke who have already had previous CEA may be successfully treated with surgical revascularization. Some studies indicate that early CEA may also be appropriate for patients with small, nondisabling stroke in whom the goal is to reduce ongoing thromboembolism or flow-limiting ischemia [235,237,238,239,240,241]. Results of other small studies have suggested that administration of an anticoagulant and delaying surgery until after the patient is stabilized is a better option [242]. Due to the limited and conflicting data, high perceived risk, and unestablished usefulness, the AHA/ASA have a Class II, Level of Evidence B recommendation regarding the use of carotid endarterectomy for treatment of acute ischemic stroke [16].
More than two-thirds of patients who survive stroke receive rehabilitation services after hospitalization, yet only a minority receive thrombolytic therapy and many have residual functional deficits, despite the development of designated stroke centers and improvements in stroke recognition and care delivery [21]. Effective stroke rehabilitation is likely to continue to be an essential component of the continuum of stroke care for the foreseeable future. Thus, in 2016 the AHA/ASA published a guideline for stroke rehabilitation and recovery [21]. This guideline spans the entire course of rehabilitation, from the early actions taken in the acute care hospital through the patient's reintegration into the community. The guideline uses the framework (rating scheme) established by the AHA concerning classes and levels of evidence [21].
The likelihood of functional independence and survival is enhanced by organized multidisciplinary rehabilitation; five more patients for every 100 treated are able to live independently following structured rehabilitation [243]. Early initiation of rehabilitation is a particularly strong predictor of improved outcome [21,244].
Rehabilitation in an inpatient stroke unit has been associated with better outcomes than rehabilitation in a general healthcare facility, with improved quality of life, survival, and functional status at five years [17,21,245,246,247,248,249]. Yet the decision to refer a stroke patient to a particular setting after discharge involves a complex set of demographic, clinical, and nonclinical factors that are also inevitably related to patient outcomes [21]. Variations in inpatient stroke rehabilitation outcomes have been found among racial/ethnic populations [21,250,251,252,253,254]. Black individuals have less functional improvement at discharge compared with White individuals and are more likely to be discharged to home despite worse functional independence measure (FIM) scores [255]. Asian individuals have functional improvements similar to those for White individuals but have less improvement at 3 months after discharge [255]. These disparities point to the need for focused attention on appropriate rehabilitation services for minority populations.
Dedicated, multidisciplinary stroke centers are more likely to provide specific interventions for stroke rehabilitation that promote functional recovery and reduce disability. Arm-robot and mirror therapy have been shown to reduce motor defects and enhance arm function; the use of electro-mechanical gait training increases the number of patients able to regain the ability to walk; treadmill training with partial body-weight support improve walking speed and walking endurance in ambulatory patients [57].
Because the effects of stroke are multifaceted and unique to each patient, multidisciplinary and organized services play an important role in patient recovery [21,246]. Post-acute stroke care settings include specialized inpatient rehabilitation hospitals, stroke rehabilitation units in acute care hospitals, outpatient therapy clinics, long-term care facilities, and patients' homes. The findings of a systematic review showed that, of these settings, an inpatient specialized stroke unit is best for providing care due to the presence of skilled nursing services, physician care, and variety of therapies [256]. However, the rehabilitation needs of some patients with mild or no disabilities may be addressed effectively in an outpatient facility (e.g., all-day care at a hospital) or in their homes [21,257]. For select patients, early discharge to a community setting for ongoing rehabilitation may provide outcomes similar to those achieved in an inpatient rehabilitation unit. This "early supported discharge" model links inpatient care with community-based rehabilitation services and allows some patients to return home sooner [21]. In some cases, recovery may occur without the need for rehabilitation services.
For inpatient and outpatient rehabilitative intervention, the multidisciplinary teams typically consist of several or all of the following: physicians, physical therapists, occupational therapists, kinesiotherapists, speech and language pathologists, social workers, recreational therapists, and nurses [21]. Nursing care for patients in the post-acute phase is particularly intensive. Patients who are triaged to inpatient facilities receive great benefit from 24-hour care by nurses who specialize in stroke care [257,258].
Depending on the patient's and family's/caregivers' specific needs, a clinical psychologist, psychiatrist, dietitian, and other healthcare professionals may join a patient's stroke rehabilitation team [21,258]. However, a team's exact composition is less important in maximizing a patient's outcome than is early intervention and the use of a coordinated, interdisciplinary approach. Without communication and coordination, isolated efforts to rehabilitate the stroke survivor are unlikely to achieve their full potential [21].
The AHA/ASA recommendations for the organization of post-stroke rehabilitation care and interventions specific to the inpatient hospital setting are summarized in Table 16 and Table 17 [21].
AMERICAN HEART ASSOCIATION/AMERICAN STROKE ASSOCIATION RECOMMENDATIONS FOR THE ORGANIZATION OF POST-STROKE REHABILITATION CARE (LEVELS OF CARE)
| Class I Recommendations | |||
| |||
| Class II Recommendations | |||
| Early supported discharge services may be reasonable for people with mild-to-moderate disability (Class IIb, Level of Evidence B). |
AMERICAN HEART ASSOCIATION/AMERICAN STROKE ASSOCIATION RECOMMENDATIONS FOR REHABILITATION IN THE INPATIENT HOSPITAL SETTING
| Class I Recommendations | ||
| ||
| Class III Recommendations | ||
| High-dose, very early mobilization within 24 hours of stroke onset can reduce the odds of a favorable outcome at 3 months and is not recommended (Class III, Level of Evidence A). |
Stroke is an acute and harrowing event, and the emotions and deficits that follow are usually overwhelming to the patient and the patient's family. The multidisciplinary rehabilitation team should therefore develop a treatment strategy to help individual patients based on a consensus model that incorporates family members and caregivers. Securing the family's/caregivers' active involvement early in the rehabilitation process optimizes the patient's chances for recovery and community reintegration [21,259]. After the rehabilitation team has formulated a plan of action for the patient, a team liaison should present its recommendations to the patient and family/caregivers through open discussions [260]. Providing patients and families/caregivers with both interactive and written materials is equally important [260]. Information to be presented should include:
Preferred setting and environment based on the patient's projected recovery
Treatment options, including suggested rehabilitation programs, estimated length of stay, frequency of therapy, and discharge criteria
Information regarding the patient's prognosis and the anticipated recovery process
Once rehabilitation has commenced, involving the patient's family/caregivers in the rehabilitation sessions and training them to assist the patient with functional activities may aid patient recuperation [21,261]. During rehabilitation, at least one or two informal meetings per week should be held with family/caregivers to reassess their concerns about the process [262].
As stated, the stroke survivor's family members and caregivers are integral to the post-stroke treatment plan [21]. However, 12% to 55% of caregivers suffer from emotional distress, most commonly depression [21,263,264]. Untreated depression is associated with a lower quality of life for both the caregiver and the stroke survivor [21,265]. A growing body of research is focused on treatment strategies to benefit both the caregiver and the stroke survivor and on educational programs that target issues such as supportive problem solving, physical care needs, financial and domestic assistance, respite, reassurance, and counseling [21,266,267,268,269,270].
For individuals who have had a stroke and are medically stable, rehabilitation assessment, prevention of medical complications, and secondary prevention become the focal points [10,16,21]. To begin, the rehabilitation team's systematic evaluation of the patient addresses various issues, including the need for rehabilitation services; the risk of complications; physical functioning, cognition, and communication; and psychosocial conditions [21]. Next, the team works with the patient and family to implement a rehabilitation plan that includes a detailed exercise program and general as well as tailored strategies for secondary prevention [10]. Throughout, the team should strive to foster a climate of familial support [21].
When a patient is medically stabilized, a rehabilitation physician is consulted to assess the patient's rehabilitative needs and recommend the proper rehabilitation setting (Table 18) [17]. Additionally, the patient's complete medical history is provided to the rehabilitation physician. If indicated, other rehabilitation specialists may perform specialized or more intensive assessments. Overall, rehabilitation assessments should be [21]:
Multidisciplinary, to account for the heterogeneous effects of stroke.
Well-coordinated and prompt, so patients can begin a rehabilitation program as soon as possible.
Well-documented, to provide the patient's rehabilitation team with accurate and detailed information.
Conducted using formal standardized, validated measures.
AREAS OF SPECIAL INTEREST FOR PURPOSES OF REHABILITATION ASSESSMENT
|
The NIHSS is a strong prognosticator of functional outcome, rendering it a valuable tool for determining rehabilitation needs [271,272]. It is the most widely used global assessment of impairment in the United States. It is a good predictor of short- and long-term morbidity and mortality and has been found to be sensitive to change in numerous studies [21]. Multiple assessments with the scale provide the rehabilitation team with a sense of the patient's recovery trajectory as he or she enters the rehabilitation stage.
The NIHSS provides detailed information in addition to an overall stroke severity score. A score of less than 5 indicates minor or no functional disability, whereas a score of more than 16 suggests severe disability and an increased probability of death [273]. With the advent of stroke treatment, patients with a score of 5 to 15 are likely to benefit from rehabilitation without the need for a nursing facility [274]. Patients with a score of more than 15 may improve with rehabilitation; however, these patients will likely require long-term care. More than one-half of survivors with a baseline score greater than 20 are initially sent to some form of rehabilitation facility rather than directly discharged to a nursing facility or home [274].
Because the type of stroke affects the usefulness of the NIHSS, its results should be qualified by medical history, examination, and brain imaging data [275]. Specifically, the scale is better suited for predicting functional outcomes at 3 and 6 months after stroke for patients with subcortical lesions than for patients with cortical lesions [21,276,277]. Additionally, the reliability of the scale in predicting depressive symptoms and cognitive dysfunctions depends on the hemisphere affected [21,277]. Because the NIHSS does not include evaluation of weakness of the distal part of the upper extremity, which is common in patients with stroke, a finger extension evaluation should be conducted in addition to the scale [278,279]. The presence of finger extension or partial shoulder abduction within 72 hours after stroke onset can help predict the patient's functional recovery at six months [280,281].
Although new measurement tools are being developed, they are difficult to evaluate with the traditional criteria (e.g., validity, reliability) normally used in evidence-based reviews [21]. The AHA/ASA recommendations for the assessment of rehabilitation needs (Table 19) are based on traditional measurement models, such as the FIM [21].
AMERICAN HEART ASSOCIATION/AMERICAN STROKE ASSOCIATION RECOMMENDATIONS FOR DISABILITY ASSESSMENT AND REHABILITATION NEEDS
| Class I Recommendations | ||||
| ||||
| Class II Recommendations | ||||
| ||||
| ADLs = activities of daily living, IADLs = instrumental activities of daily living (tasks involving more complex domestic, community, and leisure activities), SNF = skilled nursing facility. |
Soon after patient assessment, family/caregivers should be educated about and referred to community resources [21,282]. If it is recommended that the patient be discharged from an acute care facility to a nursing facility or the patient's home, relevant contact and background information for nursing home facilities, assisted-living services, social support groups, and stroke-related organizations should be provided to facilitate familial decision-making. Viable options should be presented to the family, especially information about long-term care placement if a severely disabled patient is not a candidate for rehabilitation [21]. If the family/caregivers will be taking care of the patient at a private residence (with or without professional assistance), it is essential to foster discussions about the needs of the patient, challenges the patient and family/caregivers may face, and the benefits of social support programs for the patient and family/caregivers. Whenever possible, written materials should be provided, and they should be in the primary language of the family. Among the important topics to discuss and provide education about include [21]:
Secondary prevention and medication administration specifics
Nutrition and hydration
Symptoms of complications
Specifics regarding assistance with activities of daily living (e.g., transfers, positioning, bathing, toileting, dressing, and grooming)
Swallowing difficulties
Feeding tube use
Bladder catheter care
Signs of mood disorders
Strategies to improve cognitive skills and communication
Exercises (range of motion)
Education about and referral to appropriate community resources can help to support the needs and priorities of the patient and the family or caregiver [21]. A systematic review and meta-analysis demonstrated that functional outcomes (i.e., motor, cognitive, and psychosocial function) can be improved or, at a minimum, maintained in stroke survivors when community interventions are available [21,283,284].
The patient's psychosocial status will influence his or her willingness and approach to participating in a rehabilitation program. A psychosocial assessment enables the rehabilitation team to incorporate family/caregivers more effectively into the rehabilitation process [285]. In addition, how the team manages the patient's care may be contingent on the patient's life circumstances and personality profile. Some patients and their caregivers fail to discuss psychosocial issues with their providers [21,264]. Cultural differences may also play a part in a patient's willingness to discuss these issues [21]. Areas of emphasis for the psychosocial assessment should include:
Medical history
Coping style
Therapeutic style and recovery expectations
Demographic information
Response to treatment
Substance use and abuse
Psychiatric/psychologic evaluation
Emotional and mental status and history
Education and employment
Spiritual and cultural beliefs
Family/caregiver relationship
Preferred activities
When designing and implementing the patient's treatment plan, the rehabilitation team should also take into account the residual effects of any difficulties the patient may have had before the stroke, such as drug or alcohol addiction; stress from recent life events, such as divorce, a loved one's death, or retirement; or clinical depression [21]. Individual patients will vary in how well they respond to challenging and demanding therapeutic approaches. After a patient's unique needs and circumstances are determined, relevant specialists will be incorporated into the team. All members of the rehabilitation team should be sensitive to the patient's psychosocial needs.
Medical complications related to illness, being bedridden, or lack of proper care/attention can prolong hospitalization, impede rehabilitation, increase disability, or result in death. Living in an inappropriate post-stroke environment also substantially increases a patient's risk for complications. Complications may develop in as many as 85% of hospitalized patients who have had a stroke [286]. Thus, medical examinations before and during a patient's rehabilitation program should assess the most common risks of complications: skin breakdown, deep vein thrombosis (DVT), swallowing dysfunction, bowel and bladder incontinence, falls, and pain [21].
Skin Breakdown and Contractures
Pressure ulcers are a commonly encountered complication in hospital and long-term care facilities, occurring in approximately 10% and 25% of patients in those settings, respectively [286]. According to the National Pressure Ulcer Advisory Panel, a pressure ulcer is a "localized injury to the skin and/or underlying tissue usually over a bony prominence, as a result of pressure, or pressure in combination with shear" [287,288]. Although ulcers typically occur proximal to osseous prominences (e.g., the sacrum, hips, ankles), skin breakdown at the ears is also common in certain settings [289,290]. Most pressure ulcers are associated with deep tissue injury [291]. Regular assessment of skin and the use of objective risk scales (e.g., the Braden scale) may help prevent skin injury and should be followed by regular skin inspection with documentation [21,292]. Risk factors for pressure ulcers include [286]:
Immobility
Poor hygiene
Urinary incontinence and other sources of moisture
Diabetes and other causes of poor circulation
Peripheral vascular disease
Lower body mass index
Localized infection or trauma
Edema
Poor hydration and/or nutrition
Conditions secondary to pressure ulcers include pain, localized infection, cellulitis, osteomyelitis, sepsis, and even death [293,294]. However, not all pressure ulcers, even those that are severe, will elicit a pain sensation [295].
Ulcers can be prevented or minimized with several measures, including proper positioning, turning, and transferring techniques; good hygiene; proper nutrition and hydration; and the use of barrier sprays, special mattresses, and protective dressings [21,292]. Good pain management may be necessary to perform correct positioning and frequent turning. Patients should be monitored daily for skin breakdown [21]. Any sign of a pressure ulcer warrants daily treatment [21].
Following stroke with hemiparesis, contracture on the affected side will develop in approximately 60% of patients within the first year, with wrist contractures occurring most commonly in patients who do not recover functional hand use [21,296,297]. Contractures are painful and can make self-care difficult. Many clinicians recommend daily stretching of the affected limb(s); families and patients should be taught proper stretching technique [21]. The effectiveness of resting hand splints is not well established [21]. Contractures of the ankle/foot can affect gait quality and patient safety. Ankle-foot orthosis and the nighttime use of a resting ankle splint may be beneficial in preventing ankle contracture [21].
Deep Vein Thrombosis
DVT affects only 2% of patients with stroke; yet, its prevention is critical [286]. The American Academy of Family Physicians recommends that DVT risk be calculated with use of the Well's DVT prediction model, and the results of this model will dictate subsequent DVT testing (Table 20) [298,299]. For patients with symptoms in both legs, the more symptomatic leg should be tested. A score of 3 or more on the Well's test indicates a high clinical probability of DVT; the probability is intermediate for a score of 1 or 2.
WELL'S PREDICTION RULE FOR DIAGNOSING DEEP VENOUS THROMBOSIS: CLINICAL EVALUATION TABLE FOR PREDICTING PRETEST PROBABILITY OF DEEP VEIN THROMBOSIS
| Clinical Characteristic | Score |
|---|---|
| Active cancer (treatment ongoing, within previous 6 months, or palliative) | 1 |
| Paralysis, paresis, or recent plaster immobilization of the lower extremities | 1 |
| Recently bedridden >3 days or major surgery in previous 12 weeks requiring general or regional anesthesia | 1 |
| Localized tenderness along the distribution of the deep venous system | 1 |
| Entire leg swollen | 1 |
| Calf swelling 3 cm larger than asymptomatic side (measured 10 cm below tibial tuberosity) | 1 |
| Pitting edema confined to the symptomatic leg | 1 |
| Collateral superficial veins (nonvaricose) | 1 |
| Alternative diagnosis at least as likely as venous thrombosis | -2 |
The D-dimer test alone is not recommended to rule out DVT in patients who have had a stroke. Patients with intermediate-to-high risk for DVT should be screened with use of ultrasonography. This imaging modality may not detect DVT in the calf, however, and repeat ultrasonography or venography should be used when DVT in the calf is suspected. Contrast venography was previously the most definitive test for the diagnosis of DVT, but today, Doppler ultrasound is the diagnostic study of choice [299].
Measures such as early mobilization and anticoagulation therapy are recommended to decrease the incidence of DVT after stroke. A patient's risk can be substantially reduced by 50-foot walks daily (with assistance, if necessary), administration of subcutaneous, low-dose unfractionated heparin (5,000 units twice a day, unless contraindicated), and the use of graduated compression stockings (GCS) as an adjunct to medication [300]. GCS and intermittent pneumatic compression may be considered for prevention of post-stroke DVT, but their routine prophylactic use requires further study. The benefit of treatment should be weighed against the increased risk of skin complications [21,301,302].
Swallowing Dysfunction
Based on instrumental testing results, dysphagia may develop in 42% to 78% of patients within three days after stroke [21,303]. This disorder is strongly linked to the development of malnutrition and pneumonia if not identified early and managed properly [21,303,304]. The AHA/ASA guidelines include a Class I recommendation for early dysphagia screening in patients with acute stroke [21]. Patients with dysphagia often have problems with aspiration, which can cause serious consequences if the stroke has suppressed cough sensations [21]. Although cerebral and cortical strokes can cause dysphagia, swallowing is most severely compromised by brain stem strokes [303].
The speech and language pathologist on the rehabilitation team should perform a brief swallow assessment on all patients with stroke before oral intake of food and fluids [21,305]. A dysfunctional swallow should be further examined using a complete bedside swallow examination. If bedside swallow screenings indicate an affected swallow, or if the patient has a high risk for aspiration and/or dysphagia, performing a videofluoroscopy swallowing study or fiberoptic endoscopic examination is recommended [21,305,306]. The grade of dysphagia correlates with dysarthria, aphasia, low functional independence, and level of cognitive functioning [306]. The speech and language pathologist may best identify the specific physiologic problem and recommend the necessary management and interventions for treatment. A Cochrane review assessing the effectiveness of a variety of interventions (e.g., acupuncture, neuromuscular electrical stimulation, physical stimulation) on functional outcome found that behavioral interventions and acupuncture reduced dysphagia, and pharyngeal electrical stimulation reduced pharyngeal transit time. However, the authors concluded that data are insufficient to determine the effect of these and other interventions (e.g., nutritional/fluid supplementation) on functional outcome and death. [304]. Authors of another review found that acupuncture may be effective for treatment of post-stroke dysphagia, but concluded that the reported benefits should be verified with further studies [307].
Bladder and Bowel Dysfunction
Upon admission to community-based facilities, approximately 50% of stroke survivors have urinary incontinence and 30% have fecal incontinence [308,309]. Almost all patients with fecal incontinence (98%) suffer from urinary incontinence. Urinary and fecal incontinence can lead to patient discomfort, skin breakdown, and sepsis. Fecal incontinence, in particular, reduces patient and family morale.
Large infarcts, aphasia, cognitive impairment, functional disability, lesions in the frontal cortex or frontoparietal lobes, and advanced age are associated with post-stroke urinary dysfunction [310]. Medications such as diuretics, alpha-adrenoreceptor blockers, and anticholinergic drugs can cause or exacerbate this complication [311,312]. Hyper-reflexia and hyporeflexia are the most common mechanisms of urinary incontinence in stroke survivors [313]. Detrusor sphincter dyssynergia, a cause of incomplete bladder voiding, is uncommon because its pathogenesis involves lesions between the brain stem and spine [312]. When assessing bladder function in patients with acute stroke, it is important to evaluate urinary retention with use of a bladder scanner or an in-and-out catheterization; urinary frequency, volume, and control; and the presence of dysuria. Patients who have urinary incontinence may benefit from bladder-training regimens and scheduled voiding [311,314].
Fecal incontinence can be due to neurogenic impairments or leakage around a fecal impaction (overflow incontinence) [315]. If the underlying cause of fecal incontinence is neurogenic, the signs and symptoms would likely include reduced rectal sensation and tone, inability to voluntarily contract the rectal sphincter, and stool in the rectal vault [315]. A diagnosis of constipation with overflow incontinence is more likely if the patient has rectal sensation and tone.
Risk factors for impaction and constipation include immobility, inactivity, dehydration, some medications, mood disorders, and cognitive deficits [316,317]. Multivariate analysis has shown that advanced age and diabetes are risk factors for fecal incontinence [317]. Patients with persistent constipation or fecal incontinence may benefit from bowel-management programs and psychosocial support [318]. Because of the risk of skin breakdown, the social stigma, and the burden of care associated with bowel and bladder incontinence, management is an essential component of the rehabilitation process [21].
Falls
Within 12 weeks after a stroke, approximately 25% of patients will fall [286]. Up to 70% of individuals with a stroke fall during the first six months after discharge from the hospital or rehabilitation facility [21,319]. Individuals with stroke are also at risk of repeated falls that include injury [21,320]. One study found that most falls occur at home in the first 3 months following post-stroke risk assessment [320]. Falls are a common complication for several reasons, including [315,321,322]:
Unfamiliar environment and physical state
Pain, fatigue, poor balance, and muscle weakness
Incontinence
Frequent positioning, turning, and transferring, especially in rehabilitative settings
Cognitive impairments, mood disorders (including depressive symptoms), visual impairments, spatial neglect, and any other condition that can decrease a patient's safety awareness
The Berg Balance Scale may be the most appropriate screen for patients who are likely to fall [21,323,324]. This scale tests 14 specific functional movements of daily living of increasing difficulty [325]. The 56-point maximum score indicates adequate balance and low risk of a fall. A score of less than 45 is associated with a proclivity for falling [323,325]. The score at 2 months post-stroke is useful for informing a patient's risk of falls, but it does not account for the multifactorial nature of the problem and should not preclude risk management provided in conjunction with exercise interventions, such as rehabilitation that targets gait coordination, to improve mobility [320,326]. If the patient is able to walk, the Stops Walking When Talking test may further help to identify the risk for a fall [323]. With this test, the examiner initiates a conversation with the patient while walking; if the patient stops walking to respond, the risk of a fall is increased [327]. St. Thomas' Risk Assessment Tool in Falling Elderly Inpatients (known as STRATIFY), a tool used commonly in the rehabilitation setting, has been shown to be a poor predictor of the risk for fall when screening patients with stroke [328].
In addition to the physical consequences associated with falls, there are also psychologic and social consequences. Impairments in balance, gait, motor control, perception, and vision contribute to a heightened fear of falling in the stroke survivor, with 30% to 80% reporting various levels of fear associated with falling and mobility [21]. This fear can cascade into reduced levels of physical activity and deconditioning, resulting in greater physical decline, loss of ability to perform activities of daily living, loss of independence, social isolation, and depression. Education in fall prevention and balance training are essential components of the rehabilitation process [21].
Pain
Pain is one of the most frequently experienced complications. Almost one-half of all stroke survivors experience chronic pain, 65% of whom have shoulder pain [286]. Whether chronic or periodic, pain can delay functional recovery by masking motor function improvement, diminishing a patient's motivation or willingness to perform rehabilitative tasks, or limiting the patient's movement or requiring the use of a cane or wheelchair for ambulation [17]. Pain most often results from joint immobilization and the fixation of tendons and ligaments in one position [85]. In some patients, however, stroke-induced sensorimotor pathway damage leads to the sensation of pain in an affected extremity or side of the body. The most common pain syndrome of this type is central post-stroke pain, which affects 8% of patients, or at least 56,000 stroke patients in the United States each year [21,329,330]. Four percent of patients with central post-stroke pain experience it as shoulder pain. Central post-stroke pain can be difficult to manage, even with medications. Only amitriptyline and lamotrigine have been shown to be effective in placebo-controlled studies [331].
The AHA/ASA recommend patient and family education (i.e., range of motion, positioning) about shoulder pain and care following stroke [21]. A clinical assessment of the pain that includes musculoskeletal evaluation, evaluation of spasticity, identification of any subluxation, and testing for regional sensory changes is also recommended. Ultrasound may be considered for diagnosis of shoulder soft tissue injury [21]. Botulinum toxin injection or a trial or neuromodulating pain medications may be useful to reduce severe hypertonicity in hemiplegic shoulder muscles. Positioning and the use of supportive devices may help reduce pain [21].
Use of diagnostic criteria for central post-stroke pain can be helpful [21]. Additionally, initial medical examinations should thoroughly document suspected etiology of any pain, its location and characteristics (e.g., burning, tingling, stabbing, dull), its duration and intensity, and what aggravates or relieves the pain. Any pain that interferes with the rehabilitation process should be identified and treated accordingly. There is limited evidence on the efficacy of proposed treatments for central post-stroke pain. Combined pharmacotherapy (e.g., amitriptyline, lamotrigine) and therapeutic exercise may be reasonable. Few nonpharmacologic options exist [21].
Approximately 45% of stroke survivors have residual neurologic deficits that impair mobility, which is one of the most devastating sequelae of stroke [21,87]. At 6 months, about half of ischemic stroke survivors who are 65 years of age or older have hemiparesis, nearly one-third require assistance with walking, and more than one-quarter need assistance with activities of daily living [87]. Although functional outcome primarily depends on the patient's post-stroke neurologic damage and compensatory capacity, the multidisciplinary rehabilitation team plays a major role in recovery [246]. The team's coordinated and customized efforts can help many stroke survivors adopt an active and social lifestyle. To tailor services to a patient's needs, the team should assess his or her functional abilities during the immediate post-acute stroke phase before hospital discharge. Assessment relies on a physical examination and a systematic battery of tests that measure a patient's ability to complete activities of daily living and that screens for cognitive/communication skills as well as visual/spatial neglect disorders [21]. Knowledge of the patient's preferred activities is also helpful.
Several functions/activities are typically measured to assess comprehensive functional status initially and during the rehabilitation process (Table 21). Although many measurement tools can be used to objectively record a patient's comprehensive functional acuity, the most widely used and trusted instrument in the stroke rehabilitation setting is the FIM [332,333]. Throughout the rehabilitation process, FIM-supported systematic screening can help the rehabilitation team to [21,332]:
Identify functional, cognitive, and visual/spatial deficits not previously detected
Set realistic functional goals and document progression toward these goals
Deduce discharge or extended care plans
Ensure patients' safety as they perform functional tasks and teach proper mechanics to reduce their risk of injury with continued performance
ACTIVITIES MEASURED TO ASSESS COMPREHENSIVE FUNCTIONAL STATUS INITIALLY AND DURING THE REHABILITATION PROCESS
|
As stated, loss of mobility is a devastating post-stroke outcome and restoration of gait-related activities (e.g., rising to stand, sitting down, climbing stairs) is often one of the primary goals of rehabilitation. Additionally, many patients will require assistive devices, adaptive equipment, and other items to maximize independent functioning after stroke [21].
In general, major rehabilitation goals are for patients to regain safe ambulation in the home and community and to regain the ability to perform activities of daily living with minimal or no assistance. Thus, patients should be reassessed for daily tasks that are appropriate to their expected level of dependency [21]. If a return to independent community or home living is possible, domestic functioning should be evaluated [17,334]. Skills needed to stay home alone include preparing a meal, using safety precautions, properly taking medications, and obtaining emergency services. Patients who wish to resume driving should be assessed thoroughly for driving-related physical, cognitive, and behavioral functions [21]. Driving is an instrumental activity of daily living for many individuals due to its impact on their ability to participate in activities outside the home. Between one- and two-thirds of post-stroke survivors resume driving after one year, but because it is a highly complex activity requiring skills in cognition, perception, and emotional and motor control, the ability to drive is often affected by stroke. The AHA/ASA recommend that [21]:
Individuals who prove ready to return to driving, as demonstrated by successful performance on fitness-to-drive tests, should have an on-the-road test administered by an authorized person and should be referred to a driver rehabilitation program for training if the test is failed.
It is reasonable to assess individuals for cognitive, perception, physical, and motor abilities to determine readiness to return to driving. This may be achieved with a driving simulation.
Healthy cognition and communication are essential to an individual's sense of well-being. Unfortunately, stroke has an adverse effect on cognitive and communicative abilities.
Cognition
Calculation, executive functioning (the integration of multiple and complex processes), and visual perception/construction are the cognitive arenas most often affected during the first several weeks after a stroke [335,336]. Up to 88% of patients with a cerebellar stroke have cognitive deficits, such as impairments in abstract thought, attention, control, memory, planning, and speech [337]. In many cases, patients with stroke-associated right brain damage have anosognosia, a condition in which patients are rendered unaware of their contralateral sensory and motor neurologic deficits (hemiplegia, hemianesthesia, and hemianopia) [338]. Although many survivors regain some or all cognitive skills soon following a stroke, up to 38% remain cognitively impaired at 3 months [339]. Recovery rates may be as high as 80% within 6 months for stroke survivors, with visual perception and visual memory showing the most improvement and language and abstract reasoning showing the least [340,341]. At 1 and 3 years after a stroke, cognitive impairment is one of the factors most strongly linked with poor physical and mental health status [342]. Cognitive status is an important determinant of post-stroke success. The AHA/ASA recommend that all stroke patients be screened for cognitive deficits before being discharged to home [21].
Stroke-induced damage to the brain's cognition centers is second only to Alzheimer disease as the leading cause of dementia. Stroke-associated dementia manifests with the following symptoms [343]:
Memory loss, especially short-term memory
Attention deficits and difficulty following instructions
Difficulty planning/organizing tasks or solving problems
Confusion
Poor judgment
Behavioral changes, including inappropriate emotions and extreme mood fluctuations
Mood disturbances and depression
Areas of cognitive and arousal ability that should be assessed before and during rehabilitation are learning and memory, attention, visual/spatial neglect and impairments, executive functioning, and apraxia (loss of the ability to execute skilled movements despite having the desire and the physical ability to perform them) [21]. In addition to its use in assessing functional ability, the FIM is effective as an initial screen of cognitive and functional communication deficits [344]. Because stroke-related cognition deficits are independently associated with left hemispheric stroke, visual field defect, and urinary incontinence, the presence of any of these conditions should heighten awareness of the possibility of the presence of the others [339].
Visual and spatial dysfunctions may be particularly difficult to identify during the initial post-stroke examination because multiple neuroanatomic systems can be affected to varying degrees. When a standard medical examination does not include the brief, systematic screening for visual and spatial neglects, more than 60% of these dysfunctions remain undiagnosed. However, the use of the FIM may increase their early identification [344]. Patients with neglect deficits are unknowingly inattentive to specific body parts and/or spaces in the external environment. For instance, patients may brush half of their teeth or only eat food on half of their tray. Unilateral neglect is present in almost 50% of patients with right hemispheric stroke [345]. Patients with unilateral neglect are also unaware of limbs contralateral to the site of the brain lesion(s). Neglect disorders are strongly associated with poor functional outcomes and safety issues. Patients with neglect are prone to falls and injuries as well as burns to the affected limbs [322]. Addressing visual and spatial deficits as early as possible in the rehabilitation process using multiple functional adaptation techniques (e.g., visual scanning, external cues) and patient/caregiver education may decrease a patient's risk for injury [21].
Communication
As with cognitive difficulties, communication problems strain relationships between stroke survivors and their social system, impede rehabilitation, and lead to poor quality of life. Common communication-related stroke sequelae are aphasia, dysarthria, and apraxia (a speech disorder in which there is incoordination of muscles required for speech). Patients with communication deficits may also have dysphagia. How these disorders are manifested as well as their severity depends on the location and degree of the stroke. Interventions for apraxia of speech should be individually tailored and may include behavioral techniques and strategies that target [21]:
Physiologic support for speech, including respiration, phonation, articulation, and resonance
Global aspects of speech production (e.g., loudness, rate, prosody)
Augmentative and alternative communication devices and modalities should be used to supplement speech [21].
Aphasia affects one-third of stroke survivors and is one of the most common stroke-associated communication deficits [21,346].
The three types of aphasia are expressive, receptive, and global (Table 22) [347]. Patients with expressive aphasia have difficulties using words and sentences, whereas patients with receptive aphasia struggle to understand what others are communicating to them. Global aphasia is a combination of these two types. Aphasia is typically related to lesions on the left side of the brain, as the language center is located within this hemisphere in most individuals [348]. In many cases, aphasia (mild aphasia in particular) can be an elusive diagnosis because patients may [347]:
Be able to carry on normal conversations in many settings
Have trouble understanding only when sentences are long or complex
Have trouble finding the words to express an idea or may say, "the word is right on the tip of my tongue"
CHARACTERISTICS OF EXPRESSIVE AND RECEPTIVE APHASIA
|
A variety of treatment approaches for aphasia have been developed, but no conclusions can yet be made about the effectiveness of one treatment over another [21].
An additional challenge in assessment is that members of the rehabilitation team typically do not have a clear sense of the patient's communication skills before the stroke. Lastly, reading and writing skills are usually more affected than oral communication.
Well-trained, organized rehabilitation teams can use alternative methods of communicating to mitigate the effects of cognitive and communication disorders. Ideally, these problems should be recognized and managed early. However, arriving at a diagnosis can be challenging. The speech and language pathologist on the rehabilitation team is best suited to evaluate the patient for cognitive/communication disorders. In some cases, problems are initially undetected or develop after the evaluation. Training the rehabilitation team to recognize symptoms of cognitive and communication deficits early (especially those that are subtle) and report findings to the speech and language pathologist can serve as a "safety net" for patients [21]. For proper diagnosis, the speech and language pathologist should also seek the help of the patient's family to gain an understanding of the patient's cognition and communication history.
Through interviews, conversation, structured observations and other formal tests, the speech and language pathologist can evaluate the individual's cognition and communication skills, including speech, expression, social communication, and reading/writing (Table 23) [21,349].
MEASURES EVALUATED TO ASSESS COGNITION AND COMMUNICATION SKILLS
| Category | Skills to Evaluate | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Speech |
| ||||||||
| Expression |
| ||||||||
| Social communication |
| ||||||||
| Reading and writing | Reading and writing of letters, words, phrases, sentences, and paragraphs | ||||||||
| Other |
|
If necessary, the speech and language pathologist can formulate remediation strategies to accelerate recovery of the affected communication skills, development of compensatory techniques, or use of residual skills [21,349]. In many cases, patients with stroke-induced attention deficits, visual neglect, memory deficits, executive function deficits, and problem-solving difficulties can be retrained or taught compensation techniques [350]. Strategies to enhance communication with the patient should be taught to the rehabilitation team as well as the family/caregivers [21]. Any interventions should be individually tailored and designed to target the overt communication deficit as well as any deficits that accompany or underlie the communication deficit, including attention, memory, and executive functions. The use of drugs to improve cognitive impairments is not well established [21].
Following a stroke, it is understandable that patients and their families experience intense emotions. In many cases, the staff's kindness and helpfulness, familial support, and the passage of time allow patients and their families to deal with the grief and other feelings precipitated by the stroke without medication or psychologic therapy. However, approximately 33% of patients experience post-stroke depression, and other mood disorders also manifest in stroke survivors [1,351,352]. In general, psychologic conditions can have a significant impact on the success of rehabilitation. Thus, all patients should be thoroughly evaluated for psychologic disorders as early as possible and on an ongoing basis [21].
Detecting post-stroke depression can be particularly challenging, as symptoms often appear to be typical post-stroke symptoms or are subtle. Patients may experience fatigue, sleeping difficulties, loss of appetite, tearfulness, and feelings of hopelessness. They may refuse to participate in therapy [21]. Additionally, cognitive deficits may prevent the patient from recognizing or having the ability to communicate depressive symptoms. Patients with an acquired flat affect may "sound sad" or indifferent to their situation without having post-stroke depression. Although several screening tools for depression in the older population are available (Table 24), a single, universally accepted evaluation tool for post-stroke depression has not been developed. Because little research in this area is available and the condition is underdiagnosed by nonpsychiatric physicians, the diagnosis of post-stroke depression should be based on information from multiple sources, including medical evaluation, patient self-report, observation of patient behavior, patient history, and staff reports of changes in behavior and motivation.
SCREENING TOOLS FOR DEPRESSION
| Evaluation Instrument | Time Required | Benefits | Disadvantages | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Beck Depression Inventory (BDI) | 10 minutes |
|
| |||||||
| Center for Epidemiologic Studies Depression (CES-D) Scale | <15 minutes |
| Not appropriate for patients with aphasia | |||||||
| Geriatric Depression Scale (GDS) | 10 minutes | Easy to use with older or cognitively impaired individuals, as well as with individuals with visual or physical problems or low motivation | High rates of false-negative results in minor depression | |||||||
| Hamilton Depression Scale | <30 minutes |
| Multiple versions compromise interobserver reliability | |||||||
| Folstein Mini-Mental State Examination | 10 minutes | Widely used for screening |
| |||||||
| Patient Health Questionnaire-2 (PHQ-2) | 15 minutes |
| Poor specificity in detecting major depression |
The accompaniment of post-stroke depression with other psychologic disorders is not uncommon [21]. Therefore, the medical evaluation should also screen for other categories of psychiatric symptoms [21]. Generalized anxiety disorder, which affects 20% of survivors, often coexists with post-stroke depression [356]. Generalized anxiety disorder delays the recovery of the ability to carry out activities of daily living and negatively affects social functioning [21]. Additionally, up to 15% of stroke survivors have pseudobulbar affect, characterized by uncontrollable laughing/crying [21].
Both post-stroke depression and pseudobulbar affect respond well to selective serotonin reuptake inhibitors [357]. Although these drugs carry some risk, they are safe in most patients who have had a stroke. However, these medications should not be administered prophylactically [358,359]. Although studies are limited, the use of cognitive-behavioral therapy techniques and brief supportive therapy in conjunction with medication may be beneficial to those with post-stroke depression and other neuropsychiatric sequelae of stroke [21].
Physical inactivity that typically occurs following a stroke can exacerbate muscle weakness (through atrophy and changes in muscle fibers), fatigue, cardiovascular and metabolic deconditioning, and poor balance [360,361]. These complications have been shown to slow physical and social recovery and hinder brain activation over time [362]. Comprehensive fitness training may offset these effects (Table 25) [360,363,364]. Moreover, exercise programs can benefit a stroke survivor by reducing recurrent stroke and cardiovascular risks; reducing the risk and severity of post-stroke osteoporosis, preventing injuries and falls; increasing fitness, strength, flexibility, and functional activities; and promoting socialization [21,360,365]. However, exercise is not without risks. Training programs should be tailored to the patient's capabilities and conditions to promote safety and reduce musculoskeletal injuries [366,367]. For some patients, stroke severity and coexisting conditions may render exercise inadvisable. For instance, silent coronary artery disease, especially in sedentary patients, increases the chance of exercise-induced cardiac death. Because up to 75% of stroke survivors have cardiac comorbidities, the AHA suggests that the medical evaluation for an exercise program should include a graded exercise test with ECG monitoring [360]. Thorough screening, customized exercise program design, monitoring, and patient education should be performed during rehabilitation to maximize benefits and safety.
SUMMARY OF EXERCISE PROGRAM RECOMMENDATIONS FOR STROKE SURVIVORS
| Mode of Exercise | Major Goals | Intensity, Frequency, Durationa | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hospitalization and early convalescence | ||||||||||||||
|
|
| ||||||||||||
| Inpatient/outpatient exercise therapy or "rehabilitation" | ||||||||||||||
| Large-muscle activities (e.g., walking, graded walking, stationary cycle/arm ergometry, functional activities seated exercises), if appropriate |
|
| ||||||||||||
| Strength/endurance (e.g., circuit training, weight machines, free weights, isometric exercise) |
|
| ||||||||||||
| Flexibility/stretching |
| 2 to 3 days/week (Before or after aerobic or strength training) Hold each stretch for 10 to 30 seconds | ||||||||||||
| Neuromuscular (coordination and balance activities) |
|
| ||||||||||||
| ||||||||||||||
As with any rehabilitation program, the degree of a patient's cognitive and communicative deficits may impact an exercise program's success. The Neurobehavioral Cognitive Status Examination is a brief screening tool that provides a rapid and sensitive measure of the patient's cognitive function. However, this and other brief mental status scales cannot adequately assess executive skills and other higher-level cognitive functions [21]. Personalized, tailored counseling interventions have demonstrated mixed results in improving adherence to an exercise program, whereas physical activity counseling has resulted in greater physical activity at 9 and 52 weeks post-stroke [360]. The crucial elements of a successful physical activity counseling intervention have not been identified definitively [360]. Barriers that may need to be addressed include lack of familial support, depression, fatigue, social integration, and cultural issues [360].
The consequences of inactivity may be most noticeable in patients with hemiparesis or other gait deficits. More than 50% of stroke survivors require rehabilitation to regain a functional level of ambulation [368]. Effects of neural damage underlying gait impairment, spasticity, and poor muscle performance are significantly compounded by muscle weakness, a lack of fatigue resistance, and the increased energy demands of rehabilitation [360]. Patients with mild-to-moderate conditions can benefit from treadmill training with partial body weight support [360]. As the patient walks on a treadmill, his or her body weight is supported by harnesses to facilitate walking at a comfortable speed. This training augments conventional gait rehabilitation therapies by increasing gait speed, muscle performance, and fatigue resistance; however, its effect on long-term walking outcomes requires further study [368,369].
Incorporation of progressive resistance training increases the generalizability of the fitness program and may improve the ability to carry out activities of daily living [370]. Although there are no accepted guidelines, the AHA suggests more repetitions with reduced loads (10 to 15 repetitions rather than 8 to 12), similar to programs recommended for patients recovering from MI [360]. Additionally, at least one set of at least 8 to 10 exercises should involve the major muscle groups (arms, shoulders, chest, abdomen, back, hips, and legs).
As technology continues to advance, rehabilitation programs may become enhanced with new ways to engage stroke survivors in exercise [21]. A study of virtual reality training sessions was shown to improve arm and hand movement skills in two patients with chronic hemiparesis [371]. The approach was also able to provide individualized, progressive practice based on the patient's level of movement ability and rate of improvement [371]. Other studies have found virtual reality rehabilitation to be an equally effective and potentially motivating alternative and/or supplement to conventional training in post-stroke care [372,373].
Nerve stimulation therapy has also been proposed as a means to improve motor deficits and physical activity capacity. In 2021, the FDA approved a vagus nerve stimulation system intended to treat moderate-to-severe upper extremity motor deficits associated with chronic ischemic stroke [397]. The device may be incorporated into a comprehensive rehabilitation program for these patients.
The transition from inpatient care to home after a stroke can be difficult for both patients and caregivers. Ongoing rehabilitation, profound or permanent disability, or discharge to a long-term care facility affects not only the transition in care but also the continuity of that care. To ensure medical and rehabilitation continuity for the patient through the rehabilitation process and into the home or community, the AHA/ASA recommend individualized discharge planning in the transition from hospital to home that includes comprehensive assessment of activities of daily living, instrumental activities of daily living, and mobility assessments and discussion of sexual issues (e.g., safety, changes in libido), recreational and leisure activities, and return to work ability and timeline, where appropriate [21]. Patients for whom the discharge living setting is evaluated should be considered candidates for community- or home-based rehabilitation when feasible. Providers should consider alternative methods of communication and support (e.g., telephone, visits, telehealth, online support), particularly for patients who reside in rural settings [21].
Rehabilitation services provided in the community can improve cardiovascular health, decrease the risk of cardiovascular events, and increase short-term survival rates for stroke survivors [21]. Among the benefits associated with community- and home-based rehabilitation programs are reduced costs, decreased length of stay in hospitals or institutional settings, increased opportunities for involvement by patient and family/caregiver, and less stress for family/caregiver. Patient satisfaction also is generally higher.
In the United States, approximately 23% of stroke incidences are recurrent [1]. Due to the high risk of recurrent stroke and its consequences, secondary ischemic stroke prevention tends to follow a risk-stratified model of disease management [374]. Treatment of at-risk patients' conditions is typically aggressive, as inadequate management can have serious implications.
A patient's risk for a recurrent stroke is highest during the first year; 14% of survivors have a recurrent stroke within one year after the initial cerebrovascular event, suggesting that secondary prevention is time-critical and should be initiated during the rehabilitation process [375]. After the first year, the chance of recurrent stroke decreases to 4% per year [374]. Because TIA is a precursor to stroke, secondary prevention applies to this category as well. AHA/ASA guidelines, updated 2021, are for prevention of stroke in patients with stroke and TIA [10]. The guidelines focus on four areas: diagnostic evaluation for secondary prevention, risk factor management, treatment in relation to etiology, and systems of care for secondary ischemic stroke prevention. Key take-home points and selected guideline recommendations are discussed in the following sections.
A major component of secondary prevention is the treatment of modifiable risk factors and, in select patients, correcting the underlying etiology of the stroke. Most strokes can be prevented by controlling blood pressure, following a healthy diet, regular physical activity, and smoking cessation. Global population-based studies have found that five factors—blood pressure, diet, physical activity, smoking, and abdominal obesity—account for 82% to 90% of risk for ischemic and hemorrhagic stroke worldwide [10]. Management of vascular risk factors also requires effective treatment of hypertension, diabetes, hyperlipidemia, and substance misuse. Patient education regarding healthy diet, regular physical activity, and avoidance of sedentary behavior is important. Multidisciplinary teams and goals tailored to the individual patient are often required for optimal results [10].
The results of meta-analyses have indicated that lowering blood pressure reduces the risk of stroke 30% to 40% [376,377]. The findings of longitudinal studies suggest that treatment with antihypertensive medications in hypertensive and normotensive patients reduces the incidence of recurrent strokes, MI, and other vascular events [378]. In particular, diuretics or diuretics combined with ACE inhibitors (e.g., ramipril, perindopril) most significantly reduce the risk of recurrent ischemic stroke [10,378]. However, lifestyle modifications that include weight loss; increased intake of fruits, vegetables, and low-fat dairy products; habitual aerobic physical activity; and limited alcohol consumption are crucial components of controlling blood pressure [123]. Although most studies address the prevention of additional ischemic strokes, hypertension management has also been shown to reduce the risk of recurrent hemorrhagic stroke and is included in guideline recommendations published by the AHA/ASA [10,378]. In the context of secondary stroke prevention, the target blood pressure is <130 mm Hg systolic, <80 mm Hg diastolic.
AHA Recommendations
Initiation of antihypertensive medication can be beneficial in patients with no history of hypertension who experience ischemic stroke or TIA and have an average office blood pressure of >130/80 mm Hg, reducing risk of recurrent stroke, ICH, and other vascular events.
Resumption of blood pressure therapy is indicated for previously treated patients with known hypertension for both prevention of recurrent stroke and prevention of other vascular events in those who have had an ischemic stroke or TIA and are beyond the first several days. An office blood pressure goal of <130/80 mm Hg is recommended for most patients to reduce the risk of recurrent stroke and vascular events.
The choice of specific antihypertensive agent should be individualized on the basis of pharmacologic properties, mechanism of action, and consideration of the patient's comorbidities (e.g., extracranial cerebrovascular occlusive disease, renal impairment, cardiac disease, diabetes).
Lifestyle modifications are an integral part of a patient's antihypertensive therapy.
Diabetes is a well-documented independent risk factor for recurrent stroke [379,380]. Aggressive control of hypertension in diabetic patients, with a lower target of 130/80 mm Hg, has been shown to reduce the risk of stroke as well as other cardiovascular events [123]. Including ACE inhibitors in the treatment regimen of patients with diabetes effectively lowers blood pressure, slows the progression of renal disease, and reduces albuminuria [10,381].
AHA Recommendations
After a TIA or ischemic stroke, all patients should probably be screened for diabetes with testing of fasting plasma glucose, glycated hemoglobin (HbA1c), or an oral glucose tolerance test. Choice of test and timing should be guided by clinical judgment and recognition that acute illness may temporarily perturb measures of plasma glucose. In general, HbA1c may be more accurate than other screening tests in the immediate postevent period.
Use of existing guidelines for glycemic control and blood pressure targets for patients with diabetes is recommended for patients who have had a stroke or TIA.
Smoking doubles the risk of stroke [382]. Its cessation eliminates the elevated risk after 5 years and reduces the overall risk of stroke-related hospitalization [10,382,383]. Exposure to secondhand smoke also appears to increase the risk of stroke [10,384]. The most effective combination of therapies for smoking cessation is nicotine replacement, social support, and counseling [10,385].
AHA Recommendations
All patients with TIA or stroke who smoke should be strongly urged to quit and to avoid passive smoke. Counseling with or without nicotine replacement/drug therapy is recommended to assist in smoking cessation.
In patients with ischemic stroke or TIA, avoidance of environmental (passive) tobacco smoke is recommended.
The results of a meta-analysis suggest that the risk of stroke is increased 69% for individuals who have more than five drinks (with one drink defined as 12 ounces of beer, 4 ounces of wine, or 1.5 ounces of liquor) per day compared with nondrinkers [386].
AHA Recommendations
Patients with ischemic stroke or TIA who drink more than two alcoholic drinks per day for men or more than one drink per day for women should be counseled to eliminate or reduce their alcohol consumption to lessen risk factors that increase the likelihood of recurrent stroke.
In patients who have substance use disorder, specialized services are recommended to help manage this dependency.
Habitual exercise clearly prevents stroke [365]. In addition, a sedentary lifestyle impedes functional recovery and places patients at a higher risk for stroke recurrence.
AHA Recommendations
After successful screening and completion of formal stroke rehabilitation, an individu-ally tailored exercise program is indicated to enhance cardiorespiratory fitness and reduce risk of stroke recurrence.
In patients with stroke or TIA who are capable of physical activity, engaging in at least moderate-intensity aerobic physical exercise for 10 to 30 minutes three or four times per week or vigorous-intensity exercise for at least 20 minutes two times per week is recommended to lower the risk of recurrent stroke. Moderate-intensity exercise is sufficient to break a sweat or noticeably raise heart rate (e.g., walking briskly); vigorous-intensity exercise includes activities such as jogging.
Patients who are able and willing to initiate increased physical activity should be encouraged to engage in an exercise class that includes counseling to change physical activity behavior.
Supervision by a healthcare professional (e.g., physical therapist) on initiation of an exercise regimen is beneficial for individuals with disability after ischemic stroke.
Pharmacologic antithrombotic therapy with either an antiplatelet or anticoagulant agent is recommended for nearly all patients who do not have contraindications. Dual antiplatelet therapy is recommended only for short-term use in specific patients (early-arrival minor stroke and high-risk TIA, or severe symptomatic intracranial stenosis) [10].
For patients who have large-artery atherosclerosis, the specific underlying condition should be managed. Similarly, identification and treatment of cardioembolic stroke source (e.g., AF, cardiomyopathy and left ventricular thrombus, valvular heart disease, patent foramen ovale) is recommended. Patients with ischemic stroke and a high-risk source of cardioembolism generally benefit from anticoagulant therapy.
In patients with paradoxical embolus and patent foramen ovale, clinical studies support percutaneous closure of the defect for secondary stroke prevention in patients meeting the following criteria: age 18 to 60 years, nonlacunar stroke, no other identifiable cause, and high-risk patent foramen ovale features [10].
Extracranial carotid stenosis is an important and treatable cause of stroke. Patients with severe stenosis ipsilateral to a disabling stroke or TIA who are candidates for intervention should be treated early after presentation. The choice between carotid endarterectomy and carotid artery stenting should be determined by specific patient comorbidities and vascular anatomy [10].
AHA Recommendations
Carotid endarterectomy (CEA) is recommended for all patients who had a recent TIA or an ischemic stroke within the past 6 months and ipsilateral severe (70% to 99%) carotid artery stenosis if the morbidity/mortality risk is less than 6%.
CEA is recommended for certain patients (based on age, gender, and comorbidities) who had a recent TIA or an ischemic stroke within the past 6 months and ipsilateral severe (50% to 69%) carotid artery stenosis if the morbidity/mortality risk is less than 6%. CEA is not recommended when the degree of stenosis is <50%.
When CEA is indicated for patients with TIA or stroke, surgery within 2 weeks is reasonable rather than delaying surgery if there are no contraindications to early revascularization.
Carotid artery stenting (CAS) is indicated as an alternative to CEA for symptomatic patients at average or low risk of complications associated with endovascular intervention when the diameter of the lumen of the internal carotid artery is found to be reduced by more than 70% on noninvasive imaging or more than 50% on catheter angiography or noninvasive imaging with corroboration and the anticipated rate of periprocedural stroke or death is less than 6%.
It is reasonable to consider patient age in choosing between CAS and CEA. For patients older than 70 years of age, CEA may be associated with improved outcome compared with CAS, particularly when arterial anatomy is unfavorable for endovascular intervention. For younger patients, CAS is equivalent to CEA in terms of risk for periprocedural complications (e.g., stroke, MI, death) and long-term risk for ipsilateral stroke.
CAS may be considered for patients with symptomatic severe stenosis (greater than 70%) if the stenosis is difficult to access surgically, medical conditions are present that greatly increase the risk for surgery, or other specific circumstances exist (such as radiation-induced stenosis or restenosis after CEA). In this setting, CAS is reasonable when performed by operators with established periprocedural stroke and mortality rates of less than 6%, similar to those observed in trials of CEA and CAS.
For patients with recurrent or progressive ischemic symptoms ipsilateral to a stenosis or occlusion of a distal carotid artery, or occlusion of a midcervical carotid artery after institution of optimal medical therapy, the usefulness of extracranial to intracranial bypass is considered investigational.
Optimal medical therapy, which should include antiplatelet therapy, statin therapy, and risk factor modification, is recommended for all patients with carotid artery stenosis and a TIA or stroke.
The rate of stroke recurrence for patients with symptomatic intracranial atherosclerosis is approximately 9% [10]. The findings of retrospective studies have suggested that the greatest rate of recurrence is found among patients with this condition who do not have a response to antithrombotic therapy [387]. Although the results of some studies have indicated that angioplasty or stenting should be considered for such patients, the usefulness of these surgical interventions is unknown and considered investigational [10]. AHA/ASA recommends that patients with severe intracranial stenosis in the territory of stroke or TIA should not receive angioplasty and stenting as the first-line therapy for stroke prevention; aggressive medical management and short-term dual antiplatelet therapy are preferred [10].
AHA Recommendations
With regard to patients with stroke or TIA due to 50% to 99% stenosis of a major intracranial artery:
Aspirin 325 mg per day is recommended in preference to warfarin.
The addition of clopidogrel 75 mg per day (or ticagrelor 90 mg twice per day) to aspirin for up to 90 days is reasonable to further reduce recurrent stroke risk.
Long-term maintenance of blood pressure <140 mm Hg systolic, high-intensity statin therapy, and at least moderate-intensity physical activity are recommended.
Angioplasty and/or stent placement is not recommended, given the low rate of stroke with medical management and the inherent periprocedural risk of endovascular treatment.
Extracranial-intracranial bypass surgery is not recommended.
With regard to patients with stroke or TIA due to 70% to 99% stenosis of a major intracranial artery, the addition of clopidogrel 75 mg/day to aspirin for 90 days might be reasonable.
The anticoagulant warfarin has a narrow therapeutic margin and numerous food and drug interactions, and these factors (which necessitate frequent INR testing and dose adjustment), combined with the associated significant bleeding risks, have led to the underutilization of this drug despite having been shown to prevent recurrent stroke substantially in patients with ischemic stroke or TIA and AF and despite the adoption of performance measures and guidelines advocating its use in these patients [10,388,389]. Easy-to-use alternative therapies are required and include dabigatran, rivaroxaban, and apixaban [390,391,392]. Significant adverse effects (i.e., serious, sometimes fatal bleeding, acute coronary events) have been associated with some of these agents, making their use inappropriate for some patients [393,394]. However, a 2011 focused update on dabigatran published by the American College of Cardiology, the AHA, and the Heart Rhythm Society recommends this agent as a useful alternative to warfarin for select patients [395].
Between 35% to 45% of patients with stroke and AF have coexisting conditions that may have caused the stroke [396]. In many cases, both the AF and the other condition (usually stenosis) will require treatment [10].
AHA Recommendations
For patients who have experienced an acute ischemic stroke or TIA with no other apparent cause, prolonged rhythm monitoring (for approximately 30 days) for AF is reasonable within 6 months of the index event.
For patients with ischemic stroke or TIA and nonvalvular paroxysmal (intermittent), persistent, or permanent AF, an oral anticoagulant (e.g., apixaban, dabigatran, edoxaban, rivaroxaban, or warfarin) is recommended to reduce the risk of stroke.
In patients who do not have severe mitral stenosis or a mechanical heart valve, apixaban, dabigatran, edoxaban, or rivaroxaban is recommended in preference to warfarin.
The selection of an antithrombotic agent should be individualized based on risk factors, cost, tolerability, patient preference, potential for drug interactions, and other clinical characteristics, including renal function and time in INR therapeutic range if the patient has been taking warfarin.
The combination of oral anticoagulation (i.e., warfarin or one of the newer agents) with antiplatelet therapy is not recommended for all patients after ischemic stroke or TIA but is reasonable in patients with clinically apparent CAD, particularly an acute coronary syndrome or stent placement.
For patients unable to take oral anticoagulants, aspirin alone (75 mg to 100 mg per day) is recommended. The combination of clopidogrel plus aspirin, compared with aspirin therapy alone, might be reasonable.
Following stroke, for patients at low risk for hemorrhagic conversion in the setting of AF, it is reasonable to initiate oral anticoagulation within 2 to 14 days after the onset of the index event. In the presence of high risk for hemorrhagic conversion (i.e., large infarct, hemorrhagic transformation, uncontrolled hypertension, hemorrhage tendency), it is reasonable to delay initiation of oral anticoagulation beyond 14 days.
For patients with AF at high risk for stroke (i.e., stroke or TIA within 3 months; Cardiac failure, Hypertension, Age, Diabetes, Stroke system [CHADS2] score of 5 or 6; mechanical or rheumatic valve disease) who require temporary interruption of oral anticoagulation, bridging therapy with a LMWH administered subcutaneously is reasonable.
The use of certain antiplatelet therapies rather than oral anticoagulation for noncardioembolic ischemic strokes and TIAs has been shown to reduce the overall risk of recurrent stroke and decrease the incidence of fatal recurrent strokes [10]. Clopidogrel is appropriate for patients who are allergic to aspirin or for patients in whom dipyridamole-associated headaches occur.
AHA Recommendation
Aspirin (50 mg to 325 mg per day) monotherapy, the combination of aspirin 25 mg and extended-release dipyridamole 200 mg twice daily, or clopidogrel 75 mg monotherapy are all acceptable options for initial therapy. The selection of an antiplatelet agent should be individualized on the basis of patient risk factor profiles, cost, tolerance, and other clinical characteristics.
Patient M is an active woman, 70 years of age, who lost consciousness and collapsed at home. Her daughter, who was visiting her at the time, did not witness the collapse but found her mother on the floor, awake, confused, and slightly short of breath. The daughter estimated that she called EMS within 5 minutes after the collapse, and EMS responded within 15 minutes. EMS evaluated Patient M, drew blood for a glucose level, and determined that she may have had a stroke. They notified the nearest designated comprehensive stroke center that they would be arriving with the patient in approximately 20 minutes. Patient M's daughter accompanied her.
The triage and transportation of an individual with suspected stroke should be similar to that for an individual with serious trauma, and treatment is recommended within 3 hours after the onset of stroke. Because of the limited time available for assessment and diagnosis before optimal treatment, the EMS dispatcher should notify EMS personnel immediately and coordinate transport of the individual to the closest emergency facility, preferably one that is a designated primary (or comprehensive) stroke care center.
On presentation in the emergency department, Patient M is immediately triaged. Because Patient M is still somewhat confused, her daughter is asked to provide information on the patient's history. The daughter reports that her mother had had an episode of sudden-onset numbness and tingling in the right limb, with slight confusion and slurred speech, 3 days previously. The episode lasted only 5 minutes, and Patient M had not called her primary care physician. Additional information provided by the daughter indicates that Patient M has been treated for hypertension for 10 years but notes that she is often not compliant with her antihypertensive medicine, a diuretic. The patient has never smoked, drinks occasionally, and is of normal weight.
Patient M has two significant risk factors for stroke; one is a long history of hypertension. More than two-thirds of individuals older than 65 years of age are hypertensive, and it is important for individuals with hypertension to have regular blood pressure screening and to maintain a blood pressure of less than 140/90 mm Hg. Antihypertension therapy has been found to reduce the incidence of stroke by 30% to 40%. Patient M's noncompliance with her antihypertension medicine likely includes her among the 65% of known hypertensive individuals in whom blood pressure is not controlled.
Patient M's previous episode of numbness, confusion, and slurred speech appears to be evidence of a TIA, another substantial risk factor for stroke. Research has shown that approximately 5% of patients will have an ischemic stroke within 7 days after a TIA. In addition, the risk of stroke within 7 days is doubled for patients with TIAs who did not seek treatment. As is the case for many individuals who have a TIA, Patient M did not seek medical attention because the clinical symptoms resolved quickly. However, research findings indicate that urgent treatment should be provided for TIAs, as early treatment for TIA and minor stroke has been shown to reduce the risk of early recurrent stroke by 80%.
On physical examination, Patient M's blood pressure is 150/95 mm Hg. She has pain in her left arm and a slight headache. There is a focal carotid bruit on the right. She is assessed with use of the NIHSS and found to have 1/5 weakness in the left upper and lower extremities and left visual/spatial neglect. The results of laboratory tests, including a complete blood count, prothrombin time, serum electrolyte levels, cardiac biomarkers, and renal function studies, are all within normal limits. CT of the head obtained about 45 minutes into her ED evaluation (1.5 to 2 hours since last well) indicates an occlusion in a branch of the right internal carotid artery with 50% narrowing due to atherosclerosis. An area of ischemia/infarction is visible in the right anterior cerebral hemisphere. There is no evidence of subarachnoid hemorrhage. Approximately 2.5 to 3 hours after Patient M collapsed at home, she is treated with IV rt-PA at a dose of 0.9 mg/kg. Twenty-four hours later, aspirin antiplatelet therapy is started at an initial dose of 325 mg, and a maintenance dose of 75 mg per day.
Many of the patient's symptoms, including her loss of consciousness, shortness of breath, pain, and headache, are nontraditional symptoms of stroke. Studies have demonstrated that nontraditional symptoms are more prevalent among women, often leading to a delay in the evaluation for stroke. EMS personnel and clinicians should be aware of the potential for nontraditional symptoms in women and carry out a diagnostic evaluation addressing a suspicion of stroke.
Patient M is eligible for thrombolytic therapy with rt-PA according to evidence-based guidelines developed by the AHA/ASA: her blood pressure is lower than 185/110 mm Hg, the onset of symptoms is less than 3 hours prior to the start of treatment, and the laboratory values are within normal limits. Antiplatelet therapy with aspirin 325 mg daily (versus anticoagulant therapy with warfarin) is recommended for treatment of patients with stroke or TIA due to intracranial atherosclerosis with 50% to 99% occlusion. Antiplatelet therapy is not recommended as an adjunctive therapy within 24 hours of thrombolytic therapy.
When Patient M's condition is stabilized, her primary care physician and consultant neurologist provide a referral for stroke rehabilitation, and a multidisciplinary rehabilitation team is formed to assess her rehabilitative needs, recommend the proper rehabilitation setting, and develop a treatment strategy tailored to her specific needs that includes daily antiplatelet therapy. Patient M is again assessed with the NIHSS, and the score is 12. The patient's cognitive and communication skills are intact on evaluation with the FIM, with the exception of the previously documented left visual/spatial neglect. The assessment also includes evaluation of the patient's risk for complications. Because of her spatial neglect, she is screened with the Berg Balance Scale and the Stops Walking When Talking test. The score on the Berg Balance Scale is 43, and Patient M does stop walking to engage in conversation. Psychosocial assessment includes screening with the Center for Epidemiologic Studies Depression (CES-D) Scale, as well as review of the medical history and conversations with the patient and her children; no signs of depression are present.
Patient M's score of 12 on the NIHSS falls within the range (6 to 15) that indicates she is likely to benefit from rehabilitation. Evaluating a stroke survivor's risk of complications is an important component of the overall assessment, and among the most common complications are falls, deep vein thrombosis, pressure ulcers, swallowing dysfunction, bladder and bowel dysfunction, and depressive symptoms. In assessing the risk of complications, the Berg Balance Scale appears to be the most appropriate screen for patients who are likely to fall, and a score of less than 45 is associated with a likelihood of falling. The risk of a fall is also increased when a patient stops walking to talk, as Patient M did, during the Stops Talking When Walking test.
Screening for signs of depression is also essential, as depression affects approximately 33% of stroke survivors. Signs of depression are subtle and may be vague. Several screening tools are available, but there is no universally accepted tool for use in the post-stroke setting. The CES-D was chosen in this case because it is easy to administer, is useful in older individuals, and has been found to be effective for screening in the stroke population, except for individuals who have aphasia. The diagnosis of depression in stroke survivors should be based on sources in addition to a formal screening tool, such as a medical evaluation, patient self-report, observation of patient behavior, patient history, and staff reports of changes in behavior and motivation.
The rehabilitation team discusses the results of the assessment with Patient M's daughter and son, both of whom live about 45 minutes away from the patient. Together, the team and the family members explore options to determine the best approach to rehabilitation. A decision is made to transfer Patient M to an inpatient stroke unit, and a rehabilitation program is developed. The nurse on the team discusses the program with Patient M and her children and explains the course of rehabilitation and the expectations. Rehabilitation will focus on an exercise program consisting of aerobic exercise, strength training, stretching, and coordination and balance activities.
Early initiation of rehabilitation is a particularly strong predictor of improved outcome, and rehabilitation in a stroke unit has been associated with improved quality of life, survival, and functional status at 5 years compared with a general healthcare facility. No studies have demonstrated the superiority of one rehabilitation setting over another, and the inpatient setting was chosen primarily to ensure consistent care, given how far away Patient M's children live, and the limited support she otherwise has for healthcare needs. Decisions about the setting and program for rehabilitation should be shared with family members, and family and other caregivers should be provided with educational resources about the rehabilitation process.
The exercise program developed for Patient M is designed to help her regain the ability to independently carry out activities of daily living safely and to regain a functional level of ambulation. The benefits of an exercise program include increasing fitness, strength, and flexibility; improving function; preventing injuries and falls; and reducing the risk of recurrent stroke.
Patient M gradually resumes the ability to function independently, and after more than 2 weeks in the stroke rehabilitation unit, the score on the NIHSS has improved to 5. Before she is discharged to her home, the rehabilitation team provides instructions for exercises to continue at home and recommends moderate physical activity as a secondary prevention measure. The team also educates Patient M about the importance of maintaining a normal blood pressure through use of her antihypertension medication and lifestyle modifications. At a follow-up visit with her primary care clinician at 3 months, Patient M's blood pressure is 135/80 mm Hg, and she reports that she has been compliant with her antihypertension medicine and antiplatelet therapy and is functioning well at home.
Ischemic stroke remains a significant contributor to morbidity and mortality in the United States. Stroke is associated with several modifiable risk factors including hypertension, smoking, physical inactivity, and obesity. Clinicians should encourage their patients to adopt healthy lifestyle habits, especially those at highest risk. In addition, primary prevention of stroke involves effective control of many medical conditions, such as hypertension, diabetes, dyslipidemia, and AF.
Early diagnosis and appropriate immediate treatment are key to recovery of neurologic function and survival after stroke, making it imperative for both healthcare professionals and the general public to recognize the symptoms of stroke and TIAs, a substantial risk factor for stroke. Clinicians, as well as EMS staff and ED staff, should be skilled in identifying possible stroke-related symptoms, especially noting that women are more apt to have nontraditional symptoms of both TIAs and stroke. Efforts to educate the public about the importance of seeking medical care for TIAs and about the warning signs of stroke are also essential.
Evidence-based guidelines are available for the primary and secondary prevention of ischemic stroke, as well as for early management and rehabilitation. Adherence to these guidelines has been shown to reduce the incidence of stroke and to decrease morbidity and mortality resulting from stroke. Thrombolytic therapy with rt-PA is the cornerstone of treatment for ischemic stroke, and the AHA now recommends beginning antiplatelet therapy with aspirin 24 to 48 hours after the onset of stroke. Other antiplatelet agents, as well as anticoagulant therapy, are not recommended on the basis of the evidence to date.
Outcomes after stroke are improved when care is provided in a comprehensive stroke center and when rehabilitative care is provided by a multidisciplinary team. Effective rehabilitation begins with a systematic evaluation of the patient to determine the need for specific rehabilitation services, the risk of complications, the patient's physical functioning and level of cognition and communication, and the patient's psychosocial status. This evaluation allows for the development of a rehabilitation program that addresses an individual patient's specific needs, with tailored strategies for secondary prevention. When all available resources are utilized, prevention and appropriate treatment of stroke will result.
1. Tsao CW, Aday AW, Almarzooq ZI, et al. Heart disease and stroke statistics—2023 update: a report from the American Heart Association. Circulation. 2023;147:e93-e621
2. National Institute of Neurological Disorders and Stroke. Stroke. Available at https://www.ninds.nih.gov/health-information/disorders/stroke. Last accessed March 9, 2023.
3. American Heart Association/American Stroke Association. 2023 Heart Disease and Stroke Statistics Update Fact Sheet At-A-Glance. Available at https://professional.heart.org/-/media/PHD-Files-2/Science-News/2/2023-Heart-and-Stroke-Stat-Update/2023-Statistics-At-A-Glance-final_1_17_23.pdf. Last accessed March 7, 2023.
4. Elkins J, Johnston SC. Thirty-year projections for deaths from ischemic stroke in the United States. Stroke. 2003;34(9):2109-2112.
5. Ruland S, Richardson D, Hung E, et al. Predictors of recurrent stroke in African Americans. Neurology. 2006;67(4):567-571.
6. Brown DL, Boden-Albala B, Langa KM, et al. Projected costs of ischemic stroke in the United States. Neurology. 2006;67(8):1390-1395.
7. Kurth T, Moore SC, Gaziano JM, et al. Healthy lifestyle and the risk of stroke in women. Arch Intern Med. 2006;166(13):1403-1409.
8. Ovbiagele B, Levine SR. Addressing racial and ethnic disparities in stroke: the time is now. Neurology. 2006;67(8):1328-1329.
9. Meschia JF, Bushnell C, Boden-Albala B, et al. Guidelines for the primary prevention of stroke: a statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2014;45(12):3754-3832.
10. Kleindorfer DO, Towfighi A, Chaturvedi S, et al. 2021 guideline for the prevention of stroke in patients with stroke and transient ischemic attack: a guideline for healthcare professionals from the American Heart Association/American Stroke Association.Stroke. 2021;52:e354-e467.
11. Gladstone DJ. Toward an emergency response to transient ischemic attacks. Postgrad Med J. 2005;117(1):9-14.
12. Nguyen-Huynh MN, Fayad P, Gorelick PB, Johnston SC. Knowledge and management of transient ischemic attacks among U.S. primary care physicians. Neurology. 2003;61(10):1455-1456.
13. Holloway R, Benesch C, Rush SR. Stroke prevention: narrowing the evidence-practice gap. Neurology. 2000;54:1899-1906.
14. Castaldo J, Nester J, Wasser T, et al. Physician attitudes regarding cardiovascular risk reduction: the gaps between clinical importance, knowledge, and effectiveness. Dis Manag. 2005;8(2):93-105.
15. Christian AH, Mills T, Simpson SL, Mosca L. Quality of cardiovascular disease preventive care and physician/practice characteristics.J Gen Intern Med. 2006;21(3):231-237.
16. Powers WJ, Rabinstein AA, Ackerson T, et al. Guidelines for the early management of patients with acute ischemic stroke: 2019 update to the 2018 guidelines for the early management of acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2019;50(12):e344-e418.
17. Duncan PW, Zorowitz R, Bates B, et al. Management of adult stroke rehabilitation care: a clinical practice guideline. Stroke. 2005;36(9):e100-e143.
18. Powers WJ, Derdeyn CP, Biller J, et al. 2015 American Heart Association/American Stroke Association focused update of the 2013 guidelines for the early management of patients with acute ischemic stroke regarding endovascular treatment: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2015;46(10):3020-3035.
19. Alberts MJ, Latchaw RE, Jagoda A, et al. Special report: revised and updated recommendations for the establishment of primary stroke centers: a summary statement from the Brain Attack Coalition. Stroke. 2011;42:2651-2665.
20. Adeoye O, Nystrom KV, Yavagal DR, et al. ASA policy statement: recommendations for the establishment of stroke systems of care: a 2019 update. Stroke. 2019;50:e187-e210.
21. Winstein CJ, Stein J, Arena R, et al. AHA/ASA guidelines for adult stroke rehabilitation and recovery. A guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2016;47:000-000.
22. World Health Organization. International Statistical Classification of Diseases and Related Health Problems, 11th Revision. Available at https://icd.who.int/browse11/l-m/en. Last accessed March 9, 2023.
23. Feigin VL, Lawes CM, Bennett DA, Anderson CS. Stroke epidemiology: a review of population-based studies of incidence, prevalence, and case-fatality in the late 20th century. Lancet Neurol. 2003;2(1):43-53.
24. National Heart, Lung, and Blood Institute. Morbidity and Mortality: 2012 Chart Book on Cardiovascular, Lung, and Blood Diseases. Available at https://www.nhlbi.nih.gov/files/docs/research/2012_ChartBook_508.pdf. Last accessed March 9, 2023.
25. Centers for Disease Control and Prevention. Stroke Resources for Health Professionals. Available at: https://www.cdc.gov/stroke/educational_materials.htm. Last accessed March 7, 2023.
26. Manno EM, Atkinson JL, Fulgham JR, Wijdicks EF. Emerging medical and surgical management strategies in the evaluation and treatment of intracerebral hemorrhage. Mayo Clin Proc. 2005;80(3):420-433.
27. Shah QA, Ezzeddine MA, Qureshi AI. Acute hypertension in intracerebral hemorrhage: pathophysiology and treatment. J Neurol Sci. 2007;261(1-2):74-79.
28. Hemphill JC 3rd, Greenberg SM, Anderson C, et al. Guidelines for the management of spontaneous intracerebral hemorrhage: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2015;46(7):2032-2060.
29. Counsell C, Boonyakarnkul S, Dennis M, et al. Primary intracerebral hemorrhage in the Oxfordshire Community Stroke Project: Prognosis. Cerebrovasc Dis. 1995;5:26-34.
30. Flaherty ML, Haverbusch M, Sekar PK, et al. Long-term mortality after intracerebral hemorrhage. Neurology. 2006;66(8):1182-1186.
31. Poon MT, Fonville AF, Al-Shahi Salman R. Long-term prognosis after intracerebral hemorrhage: systematic review and meta-analysis.J Neurol Neurosurg Psychiatry. 2014;85(6):660-667.
33. Qureshi AI, Tuhrim S, Broderick JP, Batjer H, Hondo H, Hanley DF. Spontaneous intracerebral hemorrhage. N Engl J Med. 2001;344:1450-1460.
34. Ritter MA, Droste DW, Hegedüs, et al. Role of cerebral amyloid angiopathy in intracerebral hemorrhage in hypertensive patients. Neurology. 2005;64(7):1233-1237.
35. Kernan WN, Viscoli CM, Brass LM, et al. Phenylpropanolamine and the risk of hemorrhagic stroke. N Engl J Med. 2000;343:1826-1832.
36. Suarez JI, Tarr RW, Selman WR. Aneurysmal subarachnoid hemorrhage. N Engl J Med. 2006;354(4):387-396.
37. van Gijn J, Rinkel GJ. Subarachnoid haemorrhage: diagnosis, causes and management. Brain. 2001;124(Pt 2):249-278.
38. Niewwkamp DJ, de Wilde A, Wermer MJ, Algra A, Rinkel GJ. Long-term outcome after aneurysmal subarachnoid hemorrhage—risks of vascular events, death from cancer and all-cause death. J Neurol. 2014;261(2):309-315.
39. Mayer SA, Kreiter KT, Copeland D, et al. Global and domain-specific cognitive impairment and outcome after subarachnoid hemorrhage. Neurology. 2002;59(11):1750-1758.
40. MedlinePlus. Cerebral Arteriovenous Malformation. Available at https://medlineplus.gov/ency/article/000779.htm. Last accessed March 9, 2023.
41. Flaherty ML, Haverbusch M, Kissela B, et al. Perimesencephalic subarachnoid hemorrhage: incidence, risk factors, and outcome.J Stroke Cerebrovasc Dis. 2005;14(6):267-271.
42. Konczalla J, Kashefiolasl S, Brawanski N, Senft C, Seifert V, Platz J. Increasing numbers of nonaneurysmal subarachnoid hemorrhage in the last 15 years: antithrombotic medication as reason and prognostic factor? J Neurosurg. 2016;124(6):1731-1731.
43. Giles MF, Rothwell PM. Risk of stroke early after transient ischemic attack: a systematic review and meta-analysis. Lancet Neurol. 2007;6(12):1063-1072.
44. Albers GW, Caplan LR, Easton JD, et al. Transient ischemic attack-proposal for a new definition. N Engl J Med. 2002;347(21):1713-1716.
45. Dyken ML, Conneally, M. The Study Group on TIA Criteria and Detection. XI. Transient focal cerebral ischemia: epidemiological and clinical aspects. Stroke. 1974;5:277-284.
46. Ovbiagele B, Kidwell CS, Saver JL. Epidemiological impact in the United States of a tissue-based definition of transient ischemic attack. Stroke. 2003;34(4):919-924.
47. Easton JD, Saver JL, Albers GW, et al. Definition and evaluation of transient ischemic attack: a scientific statement for healthcare professionals from the American Heart Association/American Stroke Association Stroke Council; Council on Cardiovascular Surgery and Anesthesia; Council on Cardiovascular Radiology and Intervention; Council on Cardiovascular Nursing; and the Interdisciplinary Council on Peripheral Vascular Disease. Stroke. 2009;40(6):2276-2293.
48. Rothwell PM, Giles MF, Chandratheva A, et al. Effect of urgent treatment of transient ischaemic attack and minor stroke on early recurrent stroke (EXPRESS study): a prospective population-based sequential comparison. Lancet. 2007;370(9596):1432-1442.
50. Johnston SC, Gress DR, Browner WS, Sidney S. Short-term prognosis after emergency department diagnosis of TIA. JAMA. 2000;284(22):2901-2906.
52. Henry GL, Johnston SC, Pinkowish MD. Acute neurologic symptoms—TIA, migraine, or something else? Patient Care. 2004;38(3):50-56.
53. Nanda A. Transient Ischemic Attack. Available at https://emedicine.medscape.com/article/1910519-overview. Last accessed March 9, 2023.
54. Bos MJ, van Goor ML, Koudstaal PJ, Dippel DW. Plasma homocysteine is a risk factor for recurrent vascular events in young patients with an ischaemic stroke or TIA. J Neurol. 2005;252(3):332-337.
55. Muir KW, Buchan A, von Kummer R, Rother J, Baron JC. Imaging of acute stroke. Lancet Neurol. 2006;5(9):755-768.
57. Platz T. Evidenced-based guidelines and clinical pathways in stroke rehabilitation: an international perspective. Front Neurol. 2019;10:200.
58. Hewko C. Acute ischemic stroke-swift assessment and quick action produce optimal outcomes. JAAPA. 2004;17(7):19-25.
60. Blumenfeld H. Neuroanatomy Through Clinical Cases. 2nd ed. Boston, MA: Sinauer Associates, Inc.; 2013.
61. Jauch EC. Ischemic Stroke. Available at https://emedicine.medscape.com/article/1916852-overview. Last accessed March 7, 2023.
62. Adams HP Jr, Bendixen BH, Kappelle LJ, et al. Classification of subtype of acute ischemic stroke. Definitions for use in a multicenter clinical trial. TOAST: Trial of Org 10172 in Acute Stroke Treatment. Stroke. 1993;24(1):35-41.
63. Petty GW, Brown RD Jr, Whisnant JP, Sicks JD, O'Fallon WM, Wiebers DO. Ischemic stroke subtypes: a population-based study of incidence and risk factors. Stroke. 1999;30(12):2513-2516.
64. Grau AJ, Weimar C, Buggle F, et al. Risk factors, outcome, and treatment in subtypes of ischemic stroke: the German stroke data bank. Stroke. 2001;32(11):2559-2566.
65. Frishman WH, Ismail AA. Role of infection in atherosclerosis and coronary artery disease: a new therapeutic target? Cardiol Rev. 2002;10(4):199-210.
66. Donnan G, Norrving B, Bamford J, Bogousslavsky J. Classification of subcortical infarcts. In: Donnan G, Norrving B, Bamford J, Bogousslavsky J (eds). Subcortical Stroke. 2nd ed. Oxford: Oxford Medical Publications; 2002: 27-34.
67. Vermeer SE, Longstreth WT Jr, Koudstaal PJ. Silent brain infarcts: a systematic review. Lancet Neurol. 2007;6(7):611-619.
68. Bokura H, Kobayashi S, Yamaguchi S, et al. Silent brain infarction and subcortical white matter lesions increase the risk of stroke and mortality: a prospective cohort study. J Stroke Cerebrovasc Dis. 2006;15(2):57-63.
69. Tuttolomondo A, Pinto A, Salemi G, et al. Diabetic and non-diabetic subjects with ischemic stroke: differences, subtype distribution and outcome. Nutr Metab Cardiovasc Dis. 2008;18(2):152-157.
73. MacDougall NJJ, Amarasinghe S, Muir KW. Secondary prevention of stroke. Expert Rev Neurother. 2009;7:1103-1115.
74. Khoo CW, Lip GYH. Clinical outcomes of acute stroke patients with atrial fibrillation. Expert Rev Neurother. 2009;7:371-374.
75. Olsen TS. Regional cerebral blood flow after occlusion of the middle cerebral artery. Acta Neurol Scand. 1986;73(4):321-337.
76. Kannel WB, Benjamin EJ. Status of the epidemiology of atrial fibrillation. Med Clin North Am. 2008;92(1):17-40, ix.
77. Arboix A, Alió J. Cardioembolic stroke: clinical features, specific cardiac disorders and prognosis. Curr Cardiol Rev. 2010;6(3):150-161.
78. Gardner C, Lee D, Lee K. Embolic stroke: classification, risk factors, and clinical pearls. Appl Neurol. 2006;2(7):27-33.
79. Komrad MS, Coffey CE, Coffey KS, et al. Myocardial infarction and stroke. Neurology. 1984;34(11):1403-1409.
80. Moncayo J, Devuyst G, Van Melle G, Bogousslavsky J. Coexisting causes of ischemic stroke. Arch Neurol. 2000;57(8):1139-1144.
81. Li L, Yin GS, Geraghty OC, et al. Incidence, outcome, risk factors, and long-term prognosis of cryptogenic transient ischaemic attack and ischaemic stroke: a population-based study. Lancet Neurol. 2015;14(9):903-913.
83. Papamitsakis NIH. Lacunar Syndrome: Lacunar Syndromes. Available at https://emedicine.medscape.com/article/1163029-overview#a5. Last accessed March 9, 2023.
84. Xu J, Murphy SL, Kochanek KD, Arias E. Deaths: final data for 2019. Natl Vital Stat Rep. 2021;70 (8):1-87.
85. National Institute of Neurological Disorders and Stroke. Post-Stroke Rehabilitation. Available at https://www.stroke.nih.gov/materials/rehabilitation.htm. Last accessed March 9, 2023.
86. National Institute of Neurological Disorders and Stroke. Stroke: Challenges, Progress, and Promise. Available at https://www.stroke.nih.gov/documents/NINDS_StrokeChallenge_Brochure_508C.pdf. Last accessed March 9, 2023.
87. Kelly-Hayes M, Beiser A, Kase CS, Scaramucci A, D'Agostino RB, Wolf PA. The influence of gender and age on disability following ischemic stroke: the Framingham study. J Stroke Cerebrovasc Dis. 2003;12(3):119-126.
88. Lackland DT, Roccella EJ, Deutsch AF, et al. Factors influencing the decline in stroke mortality: a statement from the American Heart Association/American Stroke Association. Stroke. 2014;45:315-353.
89. Sacco RL, Kasner SE, Broderick JP, et al. An updated definition of stroke for the 21st century: a statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2013;44(7):2064-2089.
90. Reeves MJ, Bushnell CD, Howard G, et al. Sex differences in stroke: epidemiology, clinical presentation, medical care, and outcomes. Lancet Neurol. 2008;7:915-926.
91. Lewsey JD, Gillies M, Jhund PS, et al. Sex differences in incidence, mortality, and survival in individuals with stroke in Scotland, 1986 to 2005. Stroke. 2009;40:1038-1043.
92. Reeves MJ, Lisabeth LD. The confounding issue of sex and stroke. Neurology. 2010;74(12):947-948.
93. Seshadri S, Beiser A, Kelly-Hayes M, et al. The lifetime risk of stroke: estimates from the Framingham Study. Stroke. 2006;37:345-350.
94. Rothwell PM, Coull AJ, Silver LE, et al. Population-based study of event-rate, incidence, case fatality, and mortality for all acute vascular events in all arterial territories (Oxford Vascular Study). Lancet. 2005;366:1773-1783.
95. Vega T, Zurriaga O, Ramos JM, et al. Stroke in Spain: epidemiologic incidence and patterns: a health sentinel network study. J Stroke Cerebrovasc Dis. 2009;18:11-16.
96. Sealy-Jefferson S, Wing JJ, Sanchez BN, et al. Age- and ethnic-specific sex differences in stroke risk. Gend Med. 2012;9:121-128.
97. Mozaffarian D, Benjamin EJ, Go AS, et al. Heart disease and stroke statistics–2016 update: a report from the American Heart Association. Circulation. 2016;133(4):e38-e360.
98. Smith MA, Lisabeth LD, Brown DL, Morgenstern LB. Gender comparisons of diagnostic evaluation for ischemic stroke patients. Neurology. 2005;65(6):855-858.
100. Gail SL, Donnan G, Dewey HM, et al. Sex differences in presentation, severity, and management of stroke in a population-based study. Neurology. 2010;74(12):975-981.
101. Towfighi A, Saver JL, Engelhardt R, Ovbiagele B. A midlife stroke surge among women in the United States. Neurology. 2007;69(20):1898-1904.
102. Labiche LA, Chan W, Saldin KR, Morgenstern LB. Sex and acute stroke presentation. Ann Emerg Med. 2002;40(5):453-460.
103. Towfighi A, Zheng L, Ovbiagele B. Sex-specific trends in midlife coronary heart disease risk and prevalence. Arch Intern Med. 2009;169(19):1762-1766.
104. White H, Boden-Albala B, Wang C, et al. Ischemic stroke subtype incidence among whites, blacks, and Hispanics: the Northern Manhattan Study. Circulation. 2005;111:1327-1331.
105. Joshipura KJ, Ascherio A, Manson JE, et al. Fruit and vegetable intake in relation to risk of ischemic stroke. JAMA. 1999;282(13):1233-1239.
106. Mehdi Foroughi M, Akhavanzanjani M, Maghsoudi Z, Ghiasvand R, Khorvash F, Askari G. Stroke and nutrition: a review of studies. Int J Prev Med. 2013;4(Suppl 2):A165-S179.
107. Larsson SC, Wallin A, Wolk A. Dietary Approaches to Stop Hypertension diet and incidence of stroke: results from two prospective cohorts. Stroke. 2016;47(4):986-990.
108. Rodriguez BL, D'Agostino R, Abbott RD, et al. Risk of hospitalized stroke in men enrolled in the Honolulu Heart Program and the Framingham Study: a comparison of incidence and risk factor effects. Stroke. 2002;33(1):230-236.
109. Broderick JP, Viscoli CM, Brott T, et al. Major risk factors for aneurysmal subarachnoid hemorrhage in the young are modifiable. Stroke. 2003;34:1375-1381.
110. Bonita R, Duncan J, Truelsen T, Jackson RT, Beaglehole R. Passive smoking as well as active smoking increases the risk of acute stroke. Tob Control. 1999;8(2):156-160.
111. Centers for Disease Control and Prevention. Smoking and Heart Disease and Stroke. Available at https://www.cdc.gov/tobacco/campaign/tips/diseases/heart-disease-stroke.html. Last accessed March 9, 2023.
112. Centers for Disease Control and Prevention. Smoking-attributable mortality, years of potential life lost, and productivity losses— United States, 2000–2004. MMWR. 2008;57:1226-1228.
113. Prospective Studies Collaboration, Whitlock G, Lewington S, Sherliker P, et al. Body-mass index and cause-specific mortality in 900,000 adults: collaborative analyses of 57 prospective studies. Lancet. 2009;373:1083-1096.
114. Song YM, Sung J, Davey Smith G, Ebrahim S. Body mass index and ischemic and hemorrhagic stroke: a prospective study in Korean men. Stroke. 2004;35(4):831-836.
115. Strazzullo P, D'Elia L, Cairella G, Garbagnati F, Cappuccio FP, Scalfi L. Excess body weight and incidence of stroke: meta-analysis of prospective studies with 2 million participants. Stroke. 2010;41:e418-e426.
116. Hu G, Tuomilehto J, Silventoinen K, Sarti C, Männistö S, Jousilahti P. Body mass index, waist circumference, and waist-hip ratio on the risk of total and type-specific stroke. Arch Intern Med. 2007;167(13):1420-1427.
117. Toss F, Wiklund P, Franks PW, et al. Abdominal and gynoid adiposity and the risk of stroke. Int J Obes (Lond). 2011;35(11):1427-1432.
118. Hu G, Jousilahti P, Antikainen R, Katzmarzyk PT, Tuomilehto J. Joint effects of physical activity, body mass index, waist circumference, and waist-to-hip ratio on the risk of heart failure. Circulation. 2010;121(2):237-244.
119. Office of Disease Prevention and Health Promotion. Physical Activity Guidelines for Americans. Available at https://health.gov/sites/default/files/2019-09/Physical_Activity_Guidelines_2nd_edition.pdf. Last accessed March 9, 2023.
120. Rothwell PM, Coull AJ, Giles MF, et al. Change in stroke incidence, mortality, case-fatality, severity, and risk factors in Oxfordshire, UK from 1981 to 2004 (Oxford Vascular Study). Lancet. 2004;363(9425):1925-1933.
121. Fields LE, Burt VL, Cutler JA, Hughes J, Roccella EJ, Sorlie P. The burden of adult hypertension in the United States 1999 to a rising tide. Hypertension. 2004;44:398-404.
122. Egan BM, Zhao Y, Axon RN. U.S. trends in prevalence, awareness, treatment, and control of hypertension, 1988–2008. JAMA. 2010;303:2043-2050.
123. Chobanian AV, Bakris GL, Black HR, et al. The seventh report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure: the JNC 7 report. JAMA. 2003;289(19):2560-2572.
124. U.S. Preventive Services Task Force. Screening for hypertension in adults: U.S. Preventive Services Task Force reaffirmation recommendation statement. JAMA. 2021;325(16):1650-1656.
125. Whelton PK, Carey RM, Aranow WS, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APHA/ASH/ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2018;71(6):e13-e115.
126. Bushnell C, McCullough LD, Awad IA, et al. Guidelines for the prevention of stroke in women: a statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2014;45(5):1545-1588.
127. Centers for Disease Control and Prevention. National Diabetes Statistics Report, 2020: Estimates of Diabetes and Its Burden in the United States. Available at https://www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf. Last accessed March 9, 2023.
128. Collins R, Armitage J, Parish S, Sleigh P, Peto R, Heart Protection Study Collaborative Group. MRC/BHF Heart Protection Study of cholesterol-lowering with simvastatin in 5963 people with diabetes: a randomised placebo-controlled trial. Lancet. 2003;361(9374):2005-2016.
129. Lane DA, Lip GY. Anticoagulation intensity for elderly atrial fibrillation patients: should we use a conventional INR target (2.0 to 3.0) or a lower range? Thromb Haemost. 2010;103(2):254-256.
130. Hillis LD, Smith PK, Anderson JL, et al. 2011 ACCF/AHA guideline for coronary artery bypass graft surgery: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Circulation. 2011;124(23):e652-e735.
131. Salaycik KJ, Kelly-Hayes M, Beiser A, et al. Depressive symptoms and risk of stroke: the Framingham Study. Stroke. 2007;38(1):16-21.
133. Müller-Nordhorn J, Nolte CH, Rossnagel K, et al. Knowledge about risk factors for stroke: a population-based survey with 28,090 participants. Stroke. 2006;37(4):946-950.
134. Schneider AT, Pancioli AM, Khoury JC, et al. Trends in community knowledge of the warning signs and risk factors for stroke. JAMA. 2003;289(3):343-346.
135. Rosamond WD, Gorton RA, Hinn AR, Hohenhaus SM, Morris DL. Rapid response to stroke symptoms: the Delay in Accessing Stroke Healthcare (DASH) study. Acad Emerg Med. 1998;5(1):45-51.
136. International Stroke Trial Collaborative Group. The International Stroke Trial (IST): a randomised trial of aspirin, subcutaneous heparin, both, or neither among 19,435 patients with acute ischaemic stroke. Lancet. 1997;349(9065):1569-1581.
137. California Acute Stroke Pilot Registry (CASPR) Investigators. Prioritizing interventions to improve rates of thrombolysis for ischemic stroke. Neurology. 2005;64(4):654-659.
138. Wattigney WA, Croft JB, Mensah GA, et al. Establishing data elements for the Paul Coverdell National Acute Stroke Registry. Part 1: proceedings of an expert panel. Stroke. 2003;34(1):151-156.
139. Kothari RU, Pancioli A, Liu T, Brott T, Broderick J. Cincinnati Prehospital Stroke Scale: reproducibility and validity. Ann Emerg Med. 1999;33(4):373-378.
140. Kleindorfer DO, Miller R, Moomaw CJ, et al. Designing a message for public education regarding stroke: does FAST capture enough stroke? Stroke. 2007;38(10):2864-2868.
141. American Heart Association. Stroke Quiz English. Available at: https://www.stroke.org/en/about-stroke/stroke-risk-factors/stroke-quiz-english. Last accessed March 7, 2023.
142. Karliner L, Jacobs EA, Chen AH, Mutha S. Do professional interpreters improve clinical care for patients with limited English proficiency? A systematic review of the literature. Health Serv Res. 2007;42(2):727-754.
143. Ngo-Metzger Q, Massagli MP, Clarridge BR, et al. Linguistic and cultural barriers to care: perspectives of Chinese and Vietnamese immigrants. J Gen Intern Med. 2003;18(1):44-52.
144. Wolf PA, D'Agostino RB, Belanger AJ, Kannel WB. Probability of stroke: a risk profile from the Framingham Study. Stroke. 1991;22:312-318.
145. Johnston SC, Rothwell PM, Nguyen-Huynh MN, et al. Validation and refinement of scores to predict very early stroke risk after transient ischaemic attack. Lancet. 2007;369(9558):283-292.
146. Koton S, Rothwell PM. Performance of the ABCD and ABCD2 scores in TIA patients with carotid stenosis and atrial fibrillation. Cerebrovasc Dis. 2007;24(2-3):231-235.
147. Rothwell PM, Giles MF, Flossmann E, et al. A simple score (ABCD) to identify individuals at high early risk of stroke after transient ischaemic attack. Lancet. 2005;366(9479):29-36.
148. Long B, Koyfman A. Best clinical practice: controversies in transient ischemic attack evaluation and disposition in the emergency department. J Emerg Med. 2017;52(3):299-310.
149. Chang BP, Rostanski S, Willey J, et al. Safety and feasibility of a rapid outpatient management strategy for transient ischemic attack and minor stroke: the Rapid Access Vascular Evaluation-Neurology (RAVEN) approach. Ann Emerg Med. 2019;74(4):562-571.
150. Mochari-Greenberger H, Xian Y, Hellkamp AS, et al. Racial/ethnic and sex differences in emergency medical services transport among hospitalized US stroke patients: analysis of the national Get With The Guidelines-Stroke Registry. J Am Heart Assoc. 2015;4:e002099.
151. Centers for Disease Control and Prevention. Prehospital and hospital delays after stroke onset—United States, 2005–2006. MMWR. 2007;56(19):474-478.
152. Jauch EC, Saver JL, Adams HP Jr, et al. Guidelines for the early management of patients with acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. American Heart Association Stroke Council, Council on Cardiovascular Nursing, Council on Peripheral Vascular Disease, Council on Clinical Cardiology. Stroke. 2013;44:870-947.
153. Acker JE 3rd, Pancioli AM, Crocco TJ, et al. Implementation strategies for emergency medical services within stroke systems of care: a policy statement from the American Heart Association/American Stroke Association Expert Panel on Emergency Medical Services Systems and the Stroke Council. Stroke. 2007;38(11):3097-3115.
154. Kidwell CS, Starkman S, Eckstein M, Weems K, Saver JL. Identifying stroke in the field. Prospective validation of the Los Angeles prehospital stroke screen (LAPSS). Stroke. 2000;31(1):71-76.
155. Christensen H, Fogh Christensen A, Boysen G. Abnormalities on ECG and telemetry predict stroke outcome at 3 months. J Neurol Sci. 2005;234(1-2):99-103.
156. Eberhardt RT, Dhadly M. Giant cell arteritis: diagnosis, management, and cardiovascular implications. Cardiol Rev. 2007;15(2): 55-61.
157. Cervera A, Amaro S, Obach V, Chamorro A. Prevention of ischemic stroke: antithrombotic therapy in cardiac embolism. Curr Drug Targets. 2007;8(7):824-831.
158. Polmear A. Sentinel headaches in aneurysmal subarachnoid hemorrhage: what is the true incidence? A systematic review. Cephalalgia. 2003;23(10):935-941.
159. van der Worp HB, van Gijn J. Clinical practice: acute ischemic stroke. N Engl J Med. 2007;357(6):572-579.
160. Aronson L, Landefeld CS. Examining older people for carotid bruits: listen to your patient, not her neck. J Gen Intern Med. 1998;13(2):140-141.
161. Alexander MR. Hypertension Treatment and Management. Available at https://emedicine.medscape.com/article/241381-treatment#a1156. Last accessed March 9, 2023.
163. Jauch EC. Ischemic Stroke Differential Diagnoses. Available at https://emedicine.medscape.com/article/1916852-differential. Last accessed March 9, 2023.
164. Frankel MR, Morgenstern LB, Kwiatkowski T, et al. Predicting prognosis after stroke: a placebo group analysis from the National Institute of Neurological Disorders and Stroke rt-PA Stroke Trial. Neurology. 2000;55(7):952-959.
165. National Institute of Neurological Disorders and Stroke. NIH Stroke Scale. Available at https://stroke.nih.gov/resources/scale.htm. Last accessed March 9, 2023.
166. Sarkar S, Ghosh S, Ghosh SK, Collier A. Role of transcranial Doppler ultrasonography in stroke. Postgrad Med J. 2007;83(985):683-689.
167. Demchuk AM, Burgin WS, Christou I, et al. Thrombolysis in brain ischemia (TIBI) transcranial Doppler flow grades predict clinical severity, early recovery, and mortality in patients treated with intravenous tissue plasminogen activator. Stroke. 2001;32(1):89-93.
168. Adams H, Adams R, Del Zoppo G, Goldstein LB. Guidelines for the early management of patients with ischemic stroke: 2005 guidelines update a scientific statement from the Stroke Council of the American Heart Association/American Stroke Association. Stroke. 2005;36:916-923.
169. Essig M, Nikolaou K, Meaney JF. Magnetic resonance angiography of the head and neck vessels. Eur Radiol. 2007;17(Suppl 2):B30-B37.
170. Citron SJ, Wallace RC, Lewis CA, et al. Quality improvement guidelines for adult diagnostic neuroangiography: cooperative study between ASITN, ASNR, and SIR. J Vasc Interv Radiol. 2003;14(9 Pt 2):S257-S262.
171. Culebras A, Kase CS, Masdeu JC, et al. Practice guidelines for the use of imaging in transient ischemic attacks and acute stroke: a report of the Stroke Council, American Heart Association. Stroke. 1997;28:1480-1497.
172. Okumura A, Araki Y, Nishimura Y, et al. The clinical utility of contrast-enhanced 3D MR angiography for cerebrovascular disease. Neurol Res. 2001;23:767-771.
173. Wintermark M, Sanelli PC, Albers GW, et al. A Joint Statement By The American Society of Neurology, The American College of Radiology, and The Society of Neurointerventional Surgery. Imaging Recommendations For Acute Stroke and Transient Ischemic Attack Patients. Charlottesville, VA: ANJR; 2013.
175. Jäger HR, Moore EA, Bynevelt M, et al. Contrast-enhanced MR angiography in patients with carotid artery stenosis: comparison of two different techniques with an unenhanced 2D time-of-flight sequence. Neuroradiology. 2000;42(4):240-248.
176. Meaney JF, Goyen M. Recent advances in contrast-enhanced magnetic resonance angiography. Eur Radiol. 2007;17(Suppl 2):B2-B6.
177. Zhang XH, Liang HM. Systematic review with network meta-analysis. Medicine (Baltimore). 2019;98(30):e16360.
178. Verro P, Tanenbaum LN, Borden NM, Sen S, Eshkar N. CT angiography in acute ischemic stroke: preliminary results. Stroke. 2002;33(1):276-278.
179. Alexandrov AV, Molina CA, Grotta JC, et al. Ultrasound-enhanced systemic thrombolysis for acute ischemic stroke. N Engl J Med. 2004;351:2170-2178.
180. Mullins ME, Schaefer PW, Sorensen AG, et al. CT and conventional and diffusion-weighted MR imaging in acute stroke: study in 691 patients at presentation to the emergency department. Radiology. 2002;224(2):353-360.
181. Rymer MM, Thurtchley D, Summers D, America Brain and Stroke Institute Stroke Team. Expanded modes of tissue plasminogen activator delivery in a comprehensive stroke center increases regional acute stroke interventions. Stroke. 2003;34(6):e58-e60.
182. Frey JL, Jahnke HK, Goslar PW, Partovi S, Flaster MS. tPA by telephone: extending the benefits of a comprehensive stroke center. Neurology. 2005;64(1):154-156.
183. American Heart Association. State Stroke Legislation Increases U.S. Primary Stroke Centers. Available at https://news.heart.org/state-stroke-legislation-increases-u-s-primary-stroke-centers. Last accessed March 9, 2023.
184. Goldstein LB, Hey LA, Laney R. North Carolina stroke prevention and treatment facilities survey. Statewide availability of programs and services. Stroke. 2000;31(1):66-70.
185. Marler JR, Tilley BC, Lu M, et al. Early stroke treatment associated with better outcome: the NINDS rt-PA stroke study. Neurology. 2000;55(11):1649-1655.
186. Hacke W, Donnan G, Fieschi C, et al. Association of outcome with early stroke treatment: pooled analysis of ATLANTIS, ECASS, and NINDS rt-PA stroke trials. Lancet. 2004;363(9411):768-774.
187. Clark WM, Albers GW, Madden KP, Hamilton S, Thrombolytic Therapy in Acute Ischemic Stroke Study Investigators. The rtPA (alteplase) 0-to 6-hour acute stroke trial, part A (A0276g): results of a double-blind, placebo-controlled, multicenter study. Stroke. 2000;31:811-816.
188. Balami JS, Sutherland BA, Buchan AM. Complications associated with recombinant tissue plasminogen activator therapy for acute ischaemic stroke. CNS Neurol Disord Drug Targets. 2013;12(2):155-169.
189. Saver JL. Thrombolytic Therapy in Stroke. Available at https://emedicine.medscape.com/article/1160840-overview. Last accessed March 9, 2023.
191. Chinese Acute Stroke Trial (CAST) Collaborative Group. CAST: randomised placebo-controlled trial of early aspirin use in 20,000 patients with acute ischaemic stroke. Lancet. 1997;349(9066):1641-1649.
192. International Stroke Trial Collaborative Group. The International Stroke Trial (IST): a randomised trial of aspirin, subcutaneous heparin, both, or neither among 19435 patients with acute ischaemic stroke. Lancet. 1997;349(9065):1569-1581.
193. Sandercock PA, Counsell C, Tseng MC, Cecconi E. Oral antiplatelet therapy for acute ischaemic stroke. Cochrane Database Syst Rev. 2014;3:CD000029.
194. Fairhead JF, Mehta Z, Rothwell PM. Population-based study of delays in carotid imaging and surgery and the risk of recurrent stroke. Neurology. 2005;65:371-375.
195. January CT, Wann LS, Calkins H, et al. 2019 AHA/ACC/HRS focused update of the 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: a report of the American College of Cardiology/American Heart Association TASK Force on Clinical Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2019;74:104-132.
196. Johnston SC, Amarenco P, Albers GW, et al. Ticagrelor versus aspirin in acute stroke or transient ischemic attack. N Engl J Med. 2016;375:35-43.
197. Wong KSL, Amarenco P, Albers GW, et al. Efficacy and safety of ticagrelor in relation to aspirin use within the week before randomization in the SOCRATES trial. Stroke. 2018;49:1678-1685.
198. U.S. National Library of Medicine. ClinicalTrials.gov. THALES – Acute Stroke or Transient IscHaemic Attack Treated With TicAgreLor and ASA for PrEvention of Stroke and Death. Available at https://clinicaltrials.gov/ct2/show/NCT03354429. Last accessed March 9, 2023.
199. Johnston SC, Easton JD, Farrant M, Barsan W, Conwit RA, Elm JJ, et al; Clinical Research Collaboration, Neurological Emergencies Treatment Trials Network, and the POINT Investigators. Clopidogrel and aspirin in acute ischemic stroke and high-risk TIA.N Engl J Med. 2018;379:215-225.
200. Wang Y, Wang Y, Zhao X, Liu L, Wang D, Wang C, et al; CHANCE Investigators. Clopidogrel with aspirin in acute minor stroke or transient ischemic attack. N Engl J Med. 2013;369:11-19.
201. Diener HC, Bogousslavsky J, Brass LM, Cimminiello C, Csiba L, Kaste M, et al; MATCH Investigators. Aspirin and clopidogrel compared with clopidogrel alone after recent ischaemic stroke or transient ischaemic attack in high-risk patients (MATCH): randomised, double-blind, placebo-controlled trial. Lancet. 2004;364:331-337.
202. Benavente OR, Hart RG, McClure LA, Szychowski JM, Coffey CS, Pearce LA; SPS3 Investigators. Effects of clopidogrel added to aspirin in patients with recent lacunar stroke. N Engl J Med. 2012;367:817-825.
203. Hankey GJ, Hacke W, Easton JD, Johnston SC, Mas JL, Brennan DM, et al; CHARISMA Trial Investigators. Effect of clopidogrel on the rate and functional severity of stroke among high vascular risk patients: a prespecified substudy of the Clopidogrel for High Atherothrombotic Risk and Ischemic Stabilization, Management and Avoidance (CHARISMA) trial. Stroke. 2010;41:1679-1683.
204. Sacco RL, Diener HC, Yusuf S, Cotton D, Ounpuu S, Lawton WA, et al; PRoFESS Study Group. Aspirin and extended-release dipyridamole versus clopidogrel for recurrent stroke. N Engl J Med. 2008;359:1238-1251.
205. Halkes PH, Gray LJ, Bath PM, Diener HC, Guiraud-Chaumeil B, Yatsu FM, et al. Dipyridamole plus aspirin versus aspirin alone in secondary prevention after TIA or stroke: a meta-analysis by risk. J Neurol Neurosurg Psychiatry. 2008;79:1218-1223.
206. Halkes PH, van Gijn J, Kappelle LJ, Koudstaal PJ, Algra A; ESPRIT Study Group. Aspirin plus dipyridamole versus aspirin alone after cerebral ischaemia of arterial origin (ESPRIT): randomised controlled trial. Lancet. 2006;367:1665-1673.
207. de Vos-Koppelaar NC, Kerkhoff H, de Vogel EM, Zock E, Dieleman HG. The effect of a slower than standard dose escalation scheme for dipyridamole on headaches in secondary prevention therapy of strokes: a randomized, open-label trial (DOSE). Cerebrovasc Dis. 2014;37:285-289.
208. Douen AG, Medic S, Sabih M, Pageau N, Shuaib A. Titrated initiation of acetylsalicylic acid-dipyridamole therapy reduces adverse effects and improves tolerance in patients with stroke. J Stroke Cerebrovasc Dis. 2008;17:356-359.
209. Lipton RB, Bigal ME, Kolodner KB, Gorelick PB, Wilks K, Schoebelock M, et al. Acetaminophen in the treatment of headaches associated with dipyridamole-aspirin combination. Neurology. 2004;63:1099-1101.
210. Sacco RL, Diener HC, Yusuf S, Cotton D, Ounpuu S, Lawton WA, et al; PRoFESS Study Group. Aspirin and extended-release dipyridamole versus clopidogrel for recurrent stroke. N Engl J Med. 2008;359:1238-1251.
211. Lapchak PA, Araujo DM. Therapeutic potential of platelet glycoprotein IIb/IIIa receptor antagonists in the management of ischemic stroke. Am J Cardiovasc Drugs. 2003;3(2):87-94.
212. Abciximab Emergent Stroke Treatment Trial (AbESTT) Investigators. Emergency administration of abciximab for treatment of patients with acute ischemic stroke: results of a randomized phase 2 trial. Stroke. 2005;36(4):880-890.
213. Adams HP Jr, Effron MB, Torner J, et al. Emergency administration of abciximab for treatment of patients with acute ischemic stroke: results of an international phase III trial: Abciximab in Emergency Treatment of Stroke Trial (AbESTT-II). Stroke. 2008;39(1):87-99.
214. Ciccone A, Motto C, Abraha I, Cozzolino F, Santilli I. Glycoprotein IIb-IIIa inhibitors for acute ischemic stroke. Cochrane Database Syst Rev. 2014;8(3):CD005208.
215. Birschel P, Ellul J, Barer D. Progressing stroke: towards an internationally agreed definition. Cerebrovasc Dis. 2004;17(2-3):242-252.
216. Hayashi K, Kitagawa N, Takahata H, et al. Endovascular treatment for cervical carotid artery stenosis presenting with progressing stroke: three case reports. Surg Neurol. 2002;58(2):148-154.
217. Du Mesnil de Rochemont R, Sitzer M, Neumann-Haefelin T, Harmjanz A, Berkefeld J. Endovascular recanalization of acute atherothrombotic carotid artery occlusion holds up progressive stroke. Neuroradiology. 2004;46(7):583-586.
218. Adachi K, Sadato A, hayakawa M, Maeda S, Hirose Y. Acute carotid artery stenting in symptomatic high-grade cervical carotid artery stenosis. Neurosurg Rev. 2017;40(1):45-51.
219. Mpotsaris A, Kabbasch C, Borggrefe J, Gontu V, Soderman M. Stenting of the cervical internal carotid artery in acute stroke management: the Karolinska experience. Interv Neuroradiol. 2017;23(2):159-165.
220. Cohen JE, Leker RR, Gotkine M, Gomori M, Ben-Hur T. Emergent stenting to treat patients with carotid artery dissection: clinically and radiologically directed therapeutic decision making. Stroke. 2003;34(12):e254-e257.
221. Cohen JE, Gomori M, Rajz G, et al. Emergent stent-assisted angioplasty of extracranial internal carotid artery and intracranial stent-based thrombectomy in acute tandem occlusive disease: technical considerations. J Neurointerv Surg. 2013;5(5):440-446.
222. Nedeltchev K, Brekenfeld C, Remonda L, et al. Internal carotid artery stent implantation in 25 patients with acute stroke: preliminary results. Radiology. 2005;237(3):1029-1037.
223. Brekenfeld C, Remonda L, Nedeltchev K, et al. Endovascular neuroradiological treatment of acute ischemic stroke: techniques and results in 350 patients. Neurol Res. 2005;27(Supp l):S29-S35.
224. Lutsep HL. Mechanical Thrombolysis in Acute Stroke. Available at https://emedicine.medscape.com/article/1163240-overview. Last accessed March 9, 2023.
225. Berlis A, Lutsep H, Barnwell S, et al. Mechanical thrombolysis in acute ischemic stroke with endovascular photoacoustic recanalization. Stroke. 2004;35(5):1112-1116.
226. Furlan A, Higashida R, Wechsler L, et al. Intra-arterial prourokinase for acute ischemic stroke. The PROACT II study; a randomized controlled trial. JAMA. 1999;282(21):2003-2011.
227. Tomsick TA, Khatri P, Jovin T, et al. Equipoise among recanalization strategies. Neurology. 2010;74:1069-1076.
228. Tonarelli SB, Tibbs M, Vazquez G, Lakshminarayan K, Rodriguez GJ, Qureshi AI. Accuracy of the new ICD-9-CM code for "drip-and-ship" thrombolytic treatment in patients with ischemic stroke. J Stroke Cerebrovasc Dis. 2012;21:121-123.
229. Broderick JP, Palesch YY, Demchuk AM, et al. Endovascular therapy after intravenous t-PA versus t-PA alone for stroke. N Engl J Med. 2013;368:893-903.
230. Berkhemer OA, Fransen PS, Beumer D, et al. A randomized trial of intraarterial treatment for acute ischemic stroke. N Engl J Med. 2015;372(1):11-20.
231. Campbell BC, Mitchell PJ, Kleinig TJ, Dewey HM, Churilov L, Yassi N, et al. Endovascular therapy for ischemic stroke with perfusion-imaging selection. N Engl J Med. 2015;372:1009-1018.
232. Goyal M, Demchuk AM, Menon BK, Eesa M, Rempel JL, Thornton J, et al. Randomized assessment of rapid endovascular treatment of ischemic stroke. N Engl J Med. 2015;372:1019-1030.
233. Jovin TG, Chamorro A, Cobo E, et al. Thrombectomy within 8 hours after symptom onset in ischemic stroke. N Engl J Med. 2015;372:2296-2306.
234. Johansson EP, Wester P. Delay from symptoms to carotid endarterectomy. J Intern Med. 2008;263:404-411.
235. Rerkasem K, Rothwell PM. Systematic review of the operative risks of carotid endarterectomy for recently symptomatic stenosis in relation to the timing of surgery. Stroke. 2009;40:e564-e572.
236. Macdonald S. Brain injury secondary to carotid intervention. J Endovasc Ther. 2007;14(2):219-231.
237. Sbarigia E, Toni D, Speziale F, Acconcia MC, Fiorani P. Early carotid endarterectomy after ischemic stroke: the results of a prospective multicenter Italian study. Eur J Vasc Endovasc Surg. 2006;32:229-235.
238. Ballotta E, Meneghetti G, Da Giau G, Manara R, Saladini M, Baracchini C. Carotid endarterectomy within 2 weeks of minor ischemic stroke: a prospective study. J Vasc Surg. 2008;48:595-600.
239. Huber R, Müller BT, Seitz RJ, Siebler M, Mödder U, Sandmann W. Carotid surgery in acute symptomatic patients. Eur J Vasc Endovasc Surg. 2003;25:60-67.
240. Welsh S, Mead G, Chant H, Picton A, O'Neill PA, McCollum CN. Early carotid surgery in acute stroke: a multicentre randomised pilot study. Cerebrovasc Dis. 2004;18:200-205.
241. Paty PS, Darling RC 3rd, Feustel PJ, et al. Early carotid endarterectomy after acute stroke. J Vasc Surg. 2004;39:148-154.
242. Vellimana AK, Kadkhodayan Y, Rich KM, et al. Symptomatic patients with intraluminal carotid artery thrombus: outcome with a strategy of initial anticoagulation. J Neurosurg. 2013;118(1):34-41.
243. Langhorne P, Duncan P. Does the organization of postacute stroke care really matter? Stroke. 2001;32(1):268-274.
244. Miller EL, Murray L, Richards L, et al. Comprehensive overview of nursing and interdisciplinary rehabilitation care of the stroke patient: a scientific statement from the American Heart Association. Stroke. 2010;41:2402-2448.
245. Teasell R, Foley N, Bhogal SK, Speechley M. Outpatient Stroke Rehabilitation. Available at http://www.ebrsr.com/evidence-review/7-outpatient-stroke-rehabilitation. Last accessed March 9, 2023.
246. Kalra L, Langhorne P. Facilitating recovery: evidence for organized stroke care. J Rehabil Med. 2007;39(2):97-102.
247. Deutsch A, Granger CV, Heinemann AW, et al. Poststroke rehabilitation: outcomes and reimbursement of inpatient rehabilitation facilities and subacute rehabilitation programs. Stroke. 2006;37:1477-1482.
248. Maulden SA, Gassaway J, Horn SD, Smout RJ, DeJong G. Timing of initiation of rehabilitation after stroke. Arch Phys Med Rehabil. 2005;86(Suppl 2):S34-S40.
249. Prvu Bettger JA, Stineman MG. Effectiveness of multidisciplinary rehabilitation services in postacute care: state-of-the-science: a review. Arch Phys Med Rehabil. 2007;88:1523-1534.
250. Buntin MB, Garten AD, Paddock S, Saliba D, Totten M, Escarce JJ. How much is postacute care use affected by its availability? Health Serv Res. 2005;40:413-434.
251. 251. Report to the Congress: Variation and Innovation in Medicare. Washington, DC: Medicare Payment Advisory Commission (MedPAC); 2003.
252. 252. Report to the Congress: Medicare Payment Policy. Washington, DC: Medicare Payment Advisory Commission (MedPAC); 2003.
253. Ellis C, Breland HL, Egede LE. Racial/ethnic differences in utilization of post-stroke rehabilitation services: a systematic review.Ethn Dis. 2008;18:365-372.
254. Finlayson M. Changes predicting long-term care use among the oldest-old. Gerontologist. 2002;42:443-453.
255. Bhandari VK, Kushel M, Price L, Schillinger D. Racial disparities in outcomes of inpatient stroke rehabilitation. Arch Phys Med Rehabil. 2005;86(11):2081-2086.
256. Stroke Unit Trialists' Collaboration. Organised inpatient (stroke unit) care for stroke. Cochrane Database Syst Rev. 2013;(9):CD000197.
257. White JH, Alston MK, Marquez JL, et al. Community-dwelling stroke survivors: function is not the whole story with quality of life. Arch Phys Med Rehabil. 2007;88(9):1140-1146.
258. Langhorne P, Cadilhac D, Feigin V, Grieve R, Liu M. How should stroke services be organised? Lancet Neurol. 2002;1(1):62-68.
259. Glass TA, Matchar DB, Belyea M, Feussner JR. Impact of social support on outcome in first stroke. Stroke. 1993;24(1):64-70.
260. Forster A, Brown L, Smith J, et al. Information provision for stroke patients and their caregivers. Cochrane Database Syst Rev. 2012;11:CD0011919.
261. Tsouna-Hadjis E, Vemmos KN, Zakopoulos N, Stamatelopoulos S. First-stroke recovery process: the role of family social support.Arch Phys Med Rehabil. 2000;81(7):881-887.
262. Scottish Intercollegiate Guidelines Network. Management of Patients with Stroke: Rehabilitation, Prevention and Management of Complications, and Discharge Planning. A National Clinical Guideline. Edinburgh: Scottish Intercollegiate Guidelines Network; 2010.
263. Chen Y, Lu J, Wong KS, Mok VC, Ungvari GS, Tang WK. Health-related quality of life in the family caregivers of stroke survivors.Int J Rehabil Res. 2010;33:232-237.
264. Bergersen H, Froslie KF, Stibrant Sunnerhagen K, Schanke AK. Anxiety, depression, and psychological well-being 2 to 5 years poststroke. J Stroke Cerebrovasc Dis. 2010;19:364-369.
265. Oupra R, Griffiths R, Pryor J, Mott S. Effectiveness of Supportive Educative Learning programme on the level of strain experienced by caregivers of stroke patients in Thailand. Health Soc Care Comm. 2010;18:10-20.
266. Choi-Kwon S, Mitchell PH, Veith R, et al. Comparing perceived burden for Korean and American informal caregivers of stroke survivors. Rehabil Nurs. 2009;34:141-150.
267. Smith SD, Gignac MA, Richardson D, Cameron JI. Differences in the experiences and support needs of family caregivers to stroke survivors: does age matter? Top Stroke Rehabil. 2008;15:593-601.
268. Perrin PB, Johnston A, Vogel B, et al. A culturally sensitive Transition Assistance Program for stroke caregivers: examining caregiver mental health and stroke rehabilitation. J Rehabil Res Dev. 2010;47:605-617.
269. Lurbe-Puerto K, Leandro ME, Baumann M. Experiences of caregiving, satisfaction of life, and social repercussions among family caregivers, two years post-stroke. Soc Work Health Care. 2012;51:725-742.
270. Bucki B, Spitz E, Baumann M. Emotional and social repercussions of stroke on patient-family caregiver dyads: analysis of diverging attitudes and profiles of the differing dyads. PLoS One. 2019;14(4):e0215425.
271. Adams HP Jr, Davis PH, Leira EC, et al. Baseline NIH Stroke Scale score strongly predicts outcome after stroke: a report of the Trial of Org 10172 in Acute Stroke Treatment (TOAST). Neurology. 1999;53(1):126-131.
272. Schlegel D, Kolb SJ, Luciano JM, et al. Utility of the NIH Stroke Scale as a predictor of hospital disposition. Stroke. 2003;34(1):134-137.
273. Hage V. The NIH stroke scale: a window into neurological status. Nurs Spectrum. 2011;24(15):44-49.
274. Schlegel DJ, Tanne D, Demchuk AM, Levine SR, Kasner SE, the Multicenter rt-PA Stroke Survey Group. Prediction of hospital disposition after thrombolysis for acute ischemic stroke using the National Institutes of Health Stroke Scale. Arch Neurol. 2004;61(7):1061-1064.
275. Yoo AJ, Barak ER, Copen WA, et al. Combining acute diffusion-weighted imaging and mean transmit time lesion volumes with national institutes of Health Stroke Scale Score improves the prediction of acute stroke outcome. Stroke. 2010;41(8):1728-1735.
276. Glymour MM, Berkman LF, Ertel KA, Fay ME, Glass TA, Furie KL. Lesion characteristics, NIH stroke scale, and functional recovery after stroke. Am J Phys Med Rehabil. 2007;86(9):725-733.
277. Peters HT, White SE, Page SJ. The National Institutes of Health Stroke Scale lacks validity in chronic hemiparetic stroke. J Stroke Cerebrovasc Dis. 2015;24(10):2207-2212.
278. Planton M, Peiffer S, Albucher J, et al. Neuropsychological outcome after a first symptomatic ischaemic stroke with 'good recovery'.Eur J Neurol. 2012;19(2):212-219.
279. Van Zandvoort MJ, Kappelle LJ, Algra A, De Haan EH. Decreased capacity for mental effort after single supratentorial lacunar infarct may affect performance in everyday life. J Neurol Neurosurg Psychiatry. 1998;65(5):697-702.
280. Nijland RH, van Wegen EE, Harmeling-van der Wel BC, Kwakkel G, EPOS Investigators. Presence of finger extension and shoulder abduction within 72 hours after stroke predicts functional recovery: early prediction of functional outcome after stroke: the EPOS cohort study. Stroke. 2010;41(4):745-750.
281. Ghaziani E, Couppe C, Siersma V, et al. Easily conducted tests during the first week post-stroke can aid the prediction of arm functioning at 6 months. Front Neurol. 2020;10:1371.
282. Kalra L, Evans A, Perez I, et al. Training care givers of stroke patients: randomised controlled trial. BMJ. 2004;328(7448):1099.
283. Teasell R, Mehta S, Pereira S, McIntyre A, Janzen S, Allen L, Lobo L, Viana R. Time to rethink long-term rehabilitation management of stroke patients. Top Stroke Rehabil. 2012;19:457-462.
284. Ferrarello F, Baccini M, Rinaldi LA, et al. Efficacy of physiotherapy interventions late after stroke: a meta-analysis. J Neurol Neurosurg Psychiatry. 2011;82:136-143.
285. Visser-Meily A, Post M, Gorter JW, Berlekom SB, Van Den Bos T, Lindeman E. Rehabilitation of stroke patients needs a family-centered approach. Disabil Rehabil. 2006;28(24):1557-1561.
286. Langhorne P, Stott DJ, Robertson L, et al. Medical complications after stroke: a multicenter study. Stroke. 2000;31(6):1223-1229.
287. National Pressure Ulcer Advisory Panel. Pressure ulcer prevalence, cost, and risk assessment: consensus development conference statement. Decubitus. 1989;2(2):24-28.
288. National Pressure Ulcer Advisory Panel. Pressure Injury Stages: NPUAP Pressure Injury Stages. Available at https://npiap.com/page/PressureInjuryStages. Last accessed March 9, 2023.
290. Groeneveld A, Anderson M, Allen S, et al. The prevalence of pressure ulcers in a tertiary care pediatric and adult hospital. J Wound Ostomy Continence Nurs. 2004;31(3):108-120.
291. Berlowitz DR, Brienza DM. Are all pressure ulcers the result of deep tissue injury? A review of the literature. Ostomy Wound Manage. 2007;53(10):34-38.
292. Institute for Clinical Systems Improvement. Pressure Ulcer Prevention and Treatment Protocol: Health Care Protocol. Bloomington, MN; Institute for Clinical Systems Improvement (ICSI); 2012.
293. Edlich RF, Winters KL, Woodard CR, et al. Pressure ulcer prevention. J Long Term Eff Med Implants. 2004;14(4):285-304.
294. Redelings MD, Lee NE, Sorvillo F. Pressure ulcers: more lethal than we thought? Adv Skin Wound Care. 2005;18(7):367-372.
295. Edsberg LE, Black JM, Goldberg M, McNichol L, Moore L, Sieggreen M. Revised National Pressure Ulcer Advisory Panel pressure injury staging system: revised pressure injury staging system. J Wound Ostomy Continence Nurs. 2016;43(6):585-597.
296. Malhotra S, Pandyan AD, Rosewilliam S, Roffe C, Hermens H. Spasticity and contractures at the wrist after stroke: time course of development and their association with functional recovery of the upper limb. Clin Rehabil. 2011;25:184-191.
297. Sackley C, Brittle N, Patel S, et al. The prevalence of joint contractures, pressure sores, painful shoulder, other pain, falls, and depression in the year after a severely disabling stroke. Stroke. 2008;39:3329-3334.
298. Wells PS, Anderson DR, Bormanis J, et al. Value of assessment of pretest probability of deep-vein thrombosis in clinical management. Lancet. 1997;350(9094):1795-1798.
299. Qaseem A, Snow V, Barry P, et al. Current diagnosis of venous thromboembolism in primary care: a clinical practice guideline from the American Academy of Family Physicians and the American College of Physicians. Ann Fam Med. 2007;5(1):57-62.
300. Muir KW, Watt A, Baxter G, Grosset DG, Lees KR. Randomized trial of graded compression stockings for prevention of deep-vein thrombosis after acute stroke. QJM. 2000;93(6):359-364.
301. Naccarato M, Chiodo Grandi F, Dennis M, Sandercock PA. Physical methods for preventing deep vein thrombosis in stroke. Cochrane Database Syst Rev. 2010;8:CD001922.
302. Gould MK, Garcia DA, Wren SM, et al. Prevention of VTE in nonorthopedic surgical patients: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(Suppl 1):e-227S-e277S.
303. Martino R, Foley N, Bhogal S, Diamant N, Speechley M, Teasell R. Dysphagia after stroke: incidence, diagnosis, and pulmonary complications. Stroke. 2005;36(12):2756-2763.
304. Bath PM, Lee HS, Everton LF. Swallowing therapy for dysphagia in acute and subacute stroke. Cochrane Database Syst Rev. 2018;10:CD000323.
305. Perry L, Love CP. Screening for dysphagia and aspiration in acute stroke: a systematic review. Dysphagia. 2001;16(1):7-18.
306. Falsetti P, Acciai C, Palilla R, et al. Oropharyngeal dysphagia after stroke: incidence, diagnosis, and clinical predictors in patients admitted to a neurorehabilitation unit. J Stroke Cerebrovasc Dis. 2009;18(5):329-335.
307. Zhang JH, Wang D, Liu M. Overview of systematic reviews and meta-analyses of acupuncture for stroke. Neuroepidemiology. 2014;42(1):50-58.
308. Kolominsky-Rabas PL, Hilz MJ, Neundoerfer B, Heuschmann PU. Impact of urinary incontinence after stroke: results from a prospective population-based stroke register. Neurourol Urodyn. 2003;22(4):322-327.
309. Thomas LH, Coupe J, Cross LD, Tan AL, Watkins CL. Interventions for treating urinary incontinence after stroke in adults.Cochrane Database Syst Rev. 2019;2:CD004462.
310. Pettersen R, Haig Y, Nakstad PH, Wyller TB. Subtypes of urinary incontinence after stroke: relation to size and location of cerebrovascular damage. Age Ageing. 2008;37(3):324-327.
311. Harari D, Coshall C, Rudd AG, Wolfe CDA. New-onset fecal incontinence after stroke: prevalence, natural history, risk factors, and impact. Stroke. 2003;34(1):144-150.
312. Vasavada SP. Urinary Incontinence. Available at http://emedicine.medscape.com/article/452289-overview. Last accessed March 9, 2023.
313. Stroke Foundation. Incontinence after Stroke Fact Sheet. Available at https://strokefoundation.org.au/what-we-do/for-survivors-and-carers/after-stroke-factsheets/incontinence-after-stroke-fact-sheet. Last accessed March 9, 2023.
314. Eustice S, Roe B, Paterson J. Prompted voiding for the management of urinary incontinence in adults. Cochrane Database Syst Rev. 2000;(2):CD002113.
315. Heart and Stroke Foundation of Ontario, Registered Nurses' Association of Ontario. Stroke Assessment across the Continuum of Care. Toronto: Heart and Stroke Foundation of Ontario and Registered Nurses' Association of Ontario; 2005.
316. Robain G, Chennevelle JM, Petit F, Piera JB. Incidence of constipation after recent vascular hemiplegia: a prospective cohort of 152 patients. Rev Neurol (Paris). 2002;158:589-592.
317. Winge K, Rasmussen D, Werdelin LM. Constipation in neurological diseases. J Neurol Neurosurg Psychiatry. 2003;74:13-19.
318. Chatoor DR, Taylor SJ, Cohen CR, Emmanuel AV. Faecal incontinence. Br J Surg. 2007;94(2):134-144.
319. Batchelor F, Hill K, Mackintosh S, Said C. What works in falls prevention after stroke? A systematic review and meta-analysis. Stroke. 2010;41:1715-1722.
320. Tilson JK, Wu SS, Cen SY, et al. Characterizing and identifying risk for falls in the LEAPS study: a randomized clinical trial of interventions to improve walking poststroke. Stroke. 2012;43(2):446-452.
321. Jørgensen L, Engstad T, Jacobsen BK. Higher incidence of falls in long-term stroke survivors than in population controls: depressive symptoms predict falls after stroke. Stroke. 2002;33(2):542-547.
322. Czernuszenko A. Risk factors for falls in post-stroke patients treated in a neurorehabilitation ward. Neurol Neurochir Pol. 2007;41(1):28-35.
323. Andersson AG, Kamwendo K, Seiger A, Appelros P. How to identify potential fallers in a stroke unit: validity indexes of 4 test methods. J Rehabil Med. 2006;38(3):186-191.
324. Teasell R, McRae M, Foley N, Bhardwaj A. The incidence and consequences of falls in stroke patients during inpatient rehabilitation: factors associated with high risk. Arch Phys Med Rehabil. 2002;83(3):329-333.
325. Berg KO, Wood-Dauphinee SL, Williams JI, Maki B. Measuring balance in the elderly: validation of an instrument. Can J Public Health. 1992;83(Suppl 2):S7-S11.
326. Hollands KL, Pelton TA, Tyson SF, Hollands MA, van Vliet PM. Interventions for coordination of walking following stroke: systematic review. Gait Posture. 2012;35(3):349-359.
327. Beauchet O, Annweiler C, Dubost V, et al. Stops walking when talking: a predictor of falls in older adults? Eur J Neurol. 2009;16(7):786-795.
328. Smith J, Forster A, Young J. Use of the "STRATIFY" falls risk assessment in patients recovering from acute stroke. Age Ageing. 2006;35(2):138-143.
329. Andersen G, Vestergaard K, Ingeman-Nielsen M, Jensen TS. Incidence of central post-stroke pain. Pain. 1995;61(2):187-193.
330. Thom T, Haase N, Rosamond W, et al. Heart disease and stroke statistics—2006 update: a report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation. 2006;113(6):e85-e151.
331. Frese A, Husstedt IW, Ringelstein EB, Evers S. Pharmacologic treatment of central post-stroke pain. Clin J Pain. 2006;22(3):252-260.
332. The Center for Outcome Measurement in Brain Injury. Introduction to the FIM. Available at http://www.tbims.org/combi/FIM. Last accessed March 9, 2023.
333. Ottenbacher KJ, Hsu Y, Granger CV, Fiedler RC. The reliability of the functional independence measure: a quantitative review. Arch Phys Med Rehabil. 1996;77(12):1226-1232.
334. Nourhashémi F, Andrieu S, Gillette-Guyonnet S, Vellas B, Albarède JL, Grandjean H. Instrumental activities of daily living as a potential marker of frailty: a study of 7364 community-dwelling elderly women (the EPIDOS study). J Gerontol A Biol Sci Med Sci. 2001;56(7):M448-M453.
335. Nys GM, Van Zandvoort MJ, De Kort PL, et al. Domain-specific cognitive recovery after first-ever stroke: a follow-up study of 111 cases. J Int Neuropsychol Soc. 2005;11(7):795-806.
336. Chung CS, Pollock A, Campbell T, Durward BR, Hagen S. Cognitive rehabilitation for executive dysfunction in adults with stroke or other adult non-progressive acquired brain damage. Cochrane Database Syst Rev. 2013;4:CD008391.
337. Kalashnikova LA, Zueva YV, Pugacheva OV, Korsakova NK. Cognitive impairments in cerebellar infarcts. Neurosci Behav Physiol. 2005;35(8):773-779.
338. Vallar G, Ronchi R. Anosognosia for motor and sensory deficits after unilateral brain damage: a review. Restor Neurol Neurosci. 2006;24(4-6):247-257.
339. Patel MD, Coshall C, Rudd AG, Wolfe CD. Cognitive impairment after stroke: clinical determinants and its associations with long-term stroke outcomes. J Am Geriatr Soc. 2002;50(4):700-706.
340. Rasquin SM, Verhey FR, Lousberg R, Lodder J. Cognitive performance after first ever stroke related to progression of vascular brain damage: a 2 year follow up CT scan study. J Neurol Neurosurg Psychiatry. 2005;76(8):1075-1079.
341. de Haan EH, Nys GM, Van Zandvoort MJ. Cognitive function following stroke and vascular cognitive impairment. Curr Opin Neurol. 2006;19(6):559-564.
342. Patel MD, McKevitt C, Lawrence E, Rudd AG, Wolfe CD. Clinical determinants of long-term quality of life after stroke. Age Ageing. 2007;36(3):316-322.
343. Erkinjuntti T. Vascular cognitive deterioration and stroke. Cerebrovasc Dis. 2007;24(Suppl 1):189-194.
344. Edwards DF, Hahn MG, Baum CM, Perlmutter MS, Sheedy C, Dromerick AW. Screening patients with stroke for rehabilitation needs: validation of the post-stroke rehabilitation guidelines. Neurorehabil Neural Repair. 2006;20(1):42-48.
345. Buxbaum LJ, Ferraro MK, Veramonti T, et al. Hemispatial neglect: subtypes, neuroanatomy, and disability. Neurology. 2004;62(5):749-756.
346. Townend E, Brady M, McLaughlan K. A systematic evaluation of the adaptation of depression diagnostic methods for stroke survivors who have aphasia. Stroke. 2007;38(11):3076-3083.
347. American Speech-Language-Hearing Association. Aphasia. Available at https://www.asha.org/public/speech/disorders/aphasia. Last accessed March 9, 2023.
348. Crinion JT, Leff AP. Recovery and treatment of aphasia after stroke: functional imaging studies. Curr Opin Neurol. 2007;20(6):667-673.
349. American Speech-Language-Hearing Association. Preferred Practice Patterns for the Profession of Speech-Language Pathology. Available at https://www.asha.org/policy/pp2004-00191. Last accessed March 9, 2023.
350. Cicerone KD, Dahlberg C, Malec JF, et al. Evidence-based cognitive rehabilitation: updated review of the literature from 1998 through 2002. Arch Phys Med Rehabil. 2005;86(8):1681-1692.
351. Paolucci S. Epidemiology and treatment of post-stroke depression. Neuropsychiatr Dis Treat. 2008;4(1):145-154.
352. Barker-Collo SL. Depression and anxiety 3 months post stroke: prevalence and correlates. Arch Clin Neuropsychol. 2007;22(4):519-531.
353. Bienenfeld D, Screening Tests for Depression. Available at https://emedicine.medscape.com/article/1859039-overview. Last accessed March 9, 2023.
354. Mini-Mental State Examination (MMSE). Available at https://cgatoolkit.ca/Uploads/ContentDocuments/MMSE.pdf. Last accessed March 9, 2023.
355. Arroll B, Goodyear-Smith F, Crengle S, et al. Validation of PHQ-s and PHQ-0 to screen for major depression in the primary care population. Ann Fam Med. 2010;8(4):348-353.
356. Leppävuori A, Pohjasvaara T, Vataja R, Kaste M, Erkinjuntti T. Generalized anxiety disorders three to four months after ischemic stroke. Cerebrovasc Dis. 2003;16(3):257-264.
357. Rosen HJ, Cummings J. A real reason for patients with pseudobulbar affect to smile. Ann Neurol. 2007;61(2):92-96.
358. Ramasubbu R. Cerebrovascular effects of selective serotonin reuptake inhibitors: a systematic review. J Clin Psychiatry. 2004;65(12):1642-1653.
359. Legg LA, Tilney R, Hsieh CF, et al. Selective serotonin reuptake inhibitors (SSRIs) for stroke recovery. Cochrane Database Syst Rev. 2019;11:CD009286.
360. Billinger SA, Arena R, Bernhardt J, et al. Physical activity and exercise recommendations for stroke survivors: a statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2014;45(8):2532-2553.
361. Michael KM, Allen JK, Macko RF. Reduced ambulatory activity after stroke: the role of balance, gait, and cardiovascular fitness. Arch Phys Med Rehabil. 2005;86(8):1552-1556.
362. Wittenberg GF, Chen R, Ishii K, et al. Constraint-induced therapy in stroke: magnetic-stimulation motor maps and cerebral activation. Neurorehabil Neural Repair. 2003;17(1):48-57.
363. American Association of Cardiovascular and Pulmonary Rehabilitation. Guidelines for Cardiac Rehabilitation and Secondary Prevention Programs. 5th ed. Champaign, IL: Human Kinetics Publishers; 2013.
364. American College of Sports Medicine, Chodzko-Zajiko WJ, Proctor DN, et al. American College of Sports Medicine position stand: exercise and physical activity for older adults. Med Sci Sports Exerc. 2009;41(7):1510-1530.
365. Thompson PD, Buchner D, Piña IL, et al. Exercise and physical activity in the prevention and treatment of atherosclerotic cardiovascular disease: a statement from the Council on Clinical Cardiology (Subcommittee on Exercise, Rehabilitation, and Prevention) and the Council on Nutrition, Physical Activity, and Metabolism (Subcommittee on Physical Activity). Circulation. 2003;107(24):3109-3116.
366. Morris JH, Macgillivray S, Macfarlane S. Interventions to promote long-term participation in physical activity after stroke: a systematic review of the literature. Arch Phys Med Rehabil. 2014;95(5):956-967.
367. Coupar F, Pollock A, Legg LA, Sackley C, van Vliet P. Home-based therapy programmes for upper limb functional recovery following stroke. Cochrane Database Syst Rev. 2012;5:CD006755.
368. Schwartz I, Meiner Z. The influence of locomotor treatment using robotic body-weight-supported treadmill training on rehabilitation outcome of patients suffering neurological disorders. Harefuah. 2013;152(3):166-171.
369. Macko RF, Smith GV, Dobrovolny CL, Sorkin JD, Goldberg AP, Silver KH. Treadmill training improves fitness reserve in chronic stroke patients. Arch Phys Med Rehabil. 2001;82(7):879-884.
370. Yang YR, Wang RY, Lin KH, Chu MY, Chan RC. Task-oriented progressive resistance strength training improves muscle strength and functional performance in individuals with stroke. Clin Rehabil. 2006;20(10):860-870.
371. Stewart JC, Yeh SC, Jung Y, et al. Intervention to enhance skilled arm and hand movements after stroke: a feasibility study using a new virtual reality system. J Neuroeng Rehabil. 2007;4:21.
372. Brunner I, Skouen JS, Hofstad H, et al. Virtual reality training for upper extremity in subacute stroke (VIRTUES): a multicenter RCT. Neurology. 2017;89(24):2413-2421.
373. Schuster-Amft C, Eng K, Suica Z, et al. Effect of a four-week virtual reality-based training versus conventional therapy on upper limb motor function after stroke: a multicenter parallel group randomized trial. PLoS One. 2018;13(10):e00204455.
374. Jamieson DG. Secondary prevention of ischemic stroke: evolution from a stepwise to a risk stratification approach to care. Dis Manag. 2007;10(5):273-284.
375. Hankey GJ. Long-term outcome after ischaemic stroke/transient ischaemic attack. Cerebrovasc Dis. 2003;16(Suppl 1):14-19.
376. Yusuf S, Sleight P, Pogue J, Bosch J, Davies R, Dagenais G. Effects of an angiotensin-converting-enzyme inhibitor, ramipril, on cardiovascular events in high-risk patients. The Heart Outcomes Prevention Evaluation Study Investigators. N Engl J Med. 2000;342(3):145-153.
377. Lawes CM, Bennett DA, Feigin VL, Rodgers A. Blood pressure and stroke: an overview of published reviews. Stroke. 2004;35(4):1024-1033.
378. Rashid P, Leonardi-Bee J, Bath P. Blood pressure reduction and secondary prevention of stroke and other vascular events: a systematic review. Stroke. 2003;34(11):2741-2748.
379. Petty GW, Brown RD Jr, Whisnant JP, Sicks JD, O'Fallon WM, Wiebers DO. Survival and recurrence after first cerebral infarction: a population-based study in Rochester, Minnesota, 1975 through 1989. Neurology. 1998;50(1):208-216.
380. Hillen T, Coshall C, Tilling K, Rudd AG, McGovern R, Wolfe CD. Cause of stroke recurrence is multifactorial: patterns, risk factors, and outcomes of stroke recurrence in the South London Stroke Register. Stroke. 2003;34(6):1457-1463.
381. Heart Outcomes Prevention Evaluation Study Investigators. Effects of ramipril on cardiovascular and microvascular outcomes in people with diabetes mellitus: results of the HOPE study and MICRO-HOPE substudy. Lancet. 2000;355(9200):253-259.
382. Burns DM. Epidemiology of smoking-induced cardiovascular disease. Prog Cardiovasc Dis. 2003;46:11-29.
383. Fagerstrom K. The epidemiology of smoking: health consequences and benefits of cessation. Drugs. 2002;62(Suppl 2):1-9.
384. You RX, Thrift AG, McNeil JJ, Davis SM, Donnan GA. Ischemic stroke risk and passive exposure to spouses' cigarette smoking: Melbourne Stroke Risk Factor Study (MERFS) Group. Am J Public Health. 1999;89(4):572-575.
385. Tobacco Use and Dependence Guideline Panel. Treating Tobacco Use and Dependence: 2008 Update. Rockville, MD: U.S. Department of Health and Human Services, Public Health Service; 2008.
386. Reynolds K, Lewis B, Nolen JD, Kinney GL, Sathya B, He J. Alcohol consumption and risk of stroke: a meta-analysis. JAMA. 2003;289(5):579-588.
387. Thijs VN, Albers GW. Symptomatic intracranial atherosclerosis: outcome of patients who fail antithrombotic therapy. Neurology. 2000;55(4):490-497.
388. Lopes RD, Crowley MJ, Shah BR, et al. Stroke Prevention in Atrial Fibrillation: Comparative Effectiveness Review No. 123. Rockville, MD: Agency for Healthcare Research and Quality; 2013.
389. You JJ, Singer DE, Howard PA, et al. Antithrombotic therapy for atrial fibrillation. Antithrombotic therapy and prevention of thrombosis, 9th ed. American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 2012;141 (2 Suppl):e531S-e575S.
390. Lexicomp Online. Available at http://online.lexi.com. Last accessed March 9, 2023.
391. Connolly SJ, Ezekowitz MD, Yusuf S, et al. Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med. 2009;361(12):1139-51.
392. Drugs@FDA. Available at https://www.accessdata.fda.gov/scripts/cder/daf. Last accessed March 9, 2023.
393. Uchino K, Hernandez AV. Dabigatran association with higher risk of acute coronary events: meta-analysis of noninferiority randomized controlled trials. Arch Intern Med. 2012;172(5):397-402.
394. Howard PA, New oral anticoagulants for stroke prevention in atrial fibrillation: more choices bring more challenges. Hosp Pharm. 2013;48(5):366-371.
395. Wann LS, Curtis AB, Ellenbogen KA, et al. 2011 ACCF/AHA/HRS focused update on the management of patients with atrial fibrillation (update on dabigatran): a report of the American College of Cardiology Foundation/American Heart Association Task Force on practice guidelines. Circulation. 2011;123(10):1144-1150.
396. LaMori JC, Mody SH, Gross HJ, et al. Burden of comorbidities among patients with atrial fibrillation. Ther Adv Cardiovasc Dis. 2013;7(2):53-62.
397. U.S. Food and Drug Administration. FDA Approves First-of-Its-Kind Stroke Rehabilitation System. Available at https://www.fda.gov/news-events/press-announcements/fda-approves-first-its-kind-stroke-rehabilitation-system. Last accessed March 9, 2023.
398. Amarenco P, Lavallée PC, Monteiro Tavares L, et al. Five-year risk of stroke after TIA or minor ischemic stroke. N Engl J Med. 2018;378:2182-2190.
1. U.S. Preventive Services Task Force. Asymptomatic Carotid Artery Stenosis: Screening. Available at https://www.uspreventiveservicestaskforce.org/uspstf/recommendation/carotid-artery-stenosis-screening. Last accessed March 22, 2023.
2. Brown MD, Burton JH, Nazarian DJ, Promes SB, American College of Emergency Physicians. Clinical policy: use of intravenous tissue plasminogen activator for the management of acute ischemic stroke in the emergency department. Ann Emerg Med. 2015;66(3):322-333. Available at https://www.acep.org/globalassets/new-pdfs/clinical-policies/tpa-for-stroke_new.pdf. Last accessed March 22, 2023.
3. Lansberg MG, O’Donnell MJ, Khatri P, et al. Antithrombotic and thrombolytic therapy for ischemic stroke: antithrombotic therapy and prevention of thrombosis, 9th ed.: American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 2012;141(2 Suppl):e601S-e636S. Available at https://www.ahajournals.org/doi/10.1161/STR.0000000000000211. Last accessed March 22, 2023.
4. Price CIM, Pandyan AD. Electrical stimulation for preventing and treating post-stroke shoulder pain. Cochrane Database Syst Rev. 2000;(4):CD001698. Available at https://www.cochranelibrary.com/cdsr/doi/10.1002/14651858.CD001698/full. Last accessed March 22, 2023.
5. National Clinical Guideline Centre. Long-Term Rehabilitation After Stroke. London: National Institute for Health and Care Excellence; 2013. Available at https://www.nice.org.uk/guidance/CG162. Last accessed March 22, 2023.
Mention of commercial products does not indicate endorsement.