Visual impairment and injuries to the eye are major health concerns, affecting more than 5 million individuals collectively each year. The severity of eye disorders range from conditions that cause irritating symptoms to those that are vision-threatening; the most severe conditions (ocular tumors) can be life-threatening. This course addresses all of these conditions, providing details on the pathophysiology of disease, etiology, diagnosis, treatment, and prevention (when feasible). To provide a better context for the discussion of eye disorders and injuries, the course begins with an overview of the anatomy and physiology of the normal eye and eyelid and provides a comprehensive discussion of the clinical examination of the eye, including recommended guidelines for eye examinations for a variety of populations and preparing the clinical setting for the routine and emergency examination and treatment of the eye.
- INTRODUCTION
- OVERVIEW OF THE ANATOMY AND PHYSIOLOGY OF THE EYE
- CLINICAL EXAMINATION OF THE EYE
- OFFICE PREPAREDNESS FOR EYE EXAMINATION AND TREATMENT IN THE PRIMARY CARE SETTING
- SYMPTOMATIC CONDITIONS OF THE EYE AND EYELID
- EYE INJURIES AND EMERGENCIES
- COMMON EYE CONDITIONS IN CHILDREN
- VISION IMPAIRMENT IN ADULTS
- OCULAR MANIFESTATIONS OF DISEASES AND MEDICATIONS
- MALIGNANT CONDITIONS OF THE EYE AND EYELID
- CASE STUDIES
- CONCLUSION
- Works Cited
- Evidence-Based Practice Recommendations Citations
This course is designed for physicians, physician assistants, nurse practitioners, nurses, and surgical technologists and assistants seeking to enhance their knowledge of the pathophysiology of eye diseases and more effectively diagnose and treat eye disorders and injuries.
Although ophthalmologists are responsible for the diagnosis and treatment of eye disease and vision loss, interprofessional teams in the emergency room and primary care settings are often involved in initial evaluation and long-term management. The purpose of this course is address knowledge gaps, enhance nurse/physician clinical skills, and improve the care of patients with disorders of the eye that threaten vision. The goal is to achieve better clinical outcomes and a reduction in the prevalence of vision impairment.
Upon completion of this course, you should be able to:
- Describe the normal anatomy and physiology of the eye and eyelid.
- Outline the physiology of vision.
- Describe vision screening recommendations and barriers to their implementation.
- Identify appropriate skills for the clinical examination of the adult eye.
- Describe the components of pediatric eye examinations.
- Discuss the assessment of refraction and preparedness for eye examinations in the primary care setting.
- Differentially diagnose inflammatory conditions of the eye and eyelid, and describe the pharmacologic and nonpharmacologic treatment options.
- Define the causes, treatment options, and preventive strategies for noninflammatory conditions of the eye and eyelid.
- Identify the causes and treatment of mechanical disorders of the eyelid.
- Identify the most common causes of eye injuries and sudden loss of vision.
- Explain the appropriate diagnosis and treatment for eye emergencies, including interventions for non-English-proficient patients.
- Describe eye conditions that commonly occur in children.
- Outline the impact of vision impairment in adults and interventions to improve quality of life for vision-impaired individuals.
- Describe the ocular manifestations of several chronic diseases and the recommendations for eye follow-up.
- Identify ocular side effects that may result with the administration of some common medications and herbal supplements.
- Discuss the diagnosis and treatment of malignant conditions of the eye and eyelid.
Lori L. Alexander, MTPW, ELS, MWC, is President of Editorial Rx, Inc., which provides medical writing and editing services on a wide variety of clinical topics and in a range of media. A medical writer and editor for more than 30 years, Ms. Alexander has written for both professional and lay audiences, with a focus on continuing education materials, medical meeting coverage, and educational resources for patients. She is the Editor Emeritus of the American Medical Writers Association (AMWA) Journal, the peer-review journal representing the largest association of medical communicators in the United States. Ms. Alexander earned a Master’s degree in technical and professional writing, with a concentration in medical writing, at Northeastern University, Boston. She has also earned certification as a life sciences editor and as a medical writer.
John M. Leonard, MD, Professor of Medicine Emeritus, Vanderbilt University School of Medicine, completed his post-graduate clinical training at the Yale and Vanderbilt University Medical Centers before joining the Vanderbilt faculty in 1974. He is a clinician-educator and for many years served as director of residency training and student educational programs for the Vanderbilt University Department of Medicine. Over a career span of 40 years, Dr. Leonard conducted an active practice of general internal medicine and an inpatient consulting practice of infectious diseases.
Contributing faculty, Lori L. Alexander, MTPW, ELS, MWC, has disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
Contributing faculty, John M. Leonard, MD, has disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
John V. Jurica, MD, MPH
Mary Franks, MSN, APRN, FNP-C
Shannon E. Smith, MHSC, CST, CSFA
The division planners have disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
Sarah Campbell
The Director of Development and Academic Affairs has disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
The purpose of NetCE is to provide challenging curricula to assist healthcare professionals to raise their levels of expertise while fulfilling their continuing education requirements, thereby improving the quality of healthcare.
Our contributing faculty members have taken care to ensure that the information and recommendations are accurate and compatible with the standards generally accepted at the time of publication. The publisher disclaims any liability, loss or damage incurred as a consequence, directly or indirectly, of the use and application of any of the contents. Participants are cautioned about the potential risk of using limited knowledge when integrating new techniques into practice.
It is the policy of NetCE not to accept commercial support. Furthermore, commercial interests are prohibited from distributing or providing access to this activity to learners.
Supported browsers for Windows include Microsoft Internet Explorer 9.0 and up, Mozilla Firefox 3.0 and up, Opera 9.0 and up, and Google Chrome. Supported browsers for Macintosh include Safari, Mozilla Firefox 3.0 and up, Opera 9.0 and up, and Google Chrome. Other operating systems and browsers that include complete implementations of ECMAScript edition 3 and CSS 2.0 may work, but are not supported. Supported browsers must utilize the TLS encryption protocol v1.1 or v1.2 in order to connect to pages that require a secured HTTPS connection. TLS v1.0 is not supported.
The role of implicit biases on healthcare outcomes has become a concern, as there is some evidence that implicit biases contribute to health disparities, professionals' attitudes toward and interactions with patients, quality of care, diagnoses, and treatment decisions. This may produce differences in help-seeking, diagnoses, and ultimately treatments and interventions. Implicit biases may also unwittingly produce professional behaviors, attitudes, and interactions that reduce patients' trust and comfort with their provider, leading to earlier termination of visits and/or reduced adherence and follow-up. Disadvantaged groups are marginalized in the healthcare system and vulnerable on multiple levels; health professionals' implicit biases can further exacerbate these existing disadvantages.
Interventions or strategies designed to reduce implicit bias may be categorized as change-based or control-based. Change-based interventions focus on reducing or changing cognitive associations underlying implicit biases. These interventions might include challenging stereotypes. Conversely, control-based interventions involve reducing the effects of the implicit bias on the individual's behaviors. These strategies include increasing awareness of biased thoughts and responses. The two types of interventions are not mutually exclusive and may be used synergistically.
#90564: Disorders and Injuries of the Eye and Eyelid
Optimal eye health and vision are essential to overall functioning, quality of life, well-being, and productivity in children and adults. In 2015, a total of 1.02 million people in the United States were blind, and approximately 11.4 million people in the United States had vision impairment[1]. Of those with vision impairment, more than 70% were due to uncorrected refractive error. In addition, at least one-half of all conditions that cause vision impairment are preventable or treatable[1]. In the United States, the leading causes of blindness and low vision are age-related macular degeneration, cataract, diabetic retinopathy, glaucoma, amblyopia, and strabismus[1]. Eye injuries also have a substantial effect on vision, with more than 2 million eye-related emergency department visits occurring each year, with approximately 50,000 of these injuries resulting in permanent vision impairment[2,4]. Studies have consistently demonstrated that approximately 90% of all eye injuries could have been prevented[5]. The economic burden of adult vision loss and eye disorders is high: the estimated cost in 2017 was estimated at $134.2 billion; this includes $98.7 billion in direct medical costs and $35.5 billion in indirect costs (e.g., lost wages, lost household production, informal care)[3]. The prevalence of vision impairment in the United States is expected to increase in future years because of an aging population. Thus, eye health and safety are major public health concerns, and primary care clinicians have an integral role in preserving the vision of their patients.
Based on data from the 2017 National Health Interview Survey, approximately 93 million adults in the United States (about 40%) are at high risk for vision loss, compared with 65 million high-risk individuals in 2002[6]. High-risk groups include people with diabetes, adults older than 65 years of age, and those with pre-existing vision or eye problems. The analysis involved 32,886 survey respondents and excluded adults who were blind or otherwise unable to see. Among respondents at risk of vision loss, 40% had not visited an eye doctor or received an eye examination in the previous year. Among adults who reported needing eyeglasses, nearly 1 in 10 said they could not afford them. The authors concluded that improved access, awareness, and affordability of eye care services could reduce unnecessary vision loss in the United States[6].
The American Academy of Ophthalmology defines visual impairment as 20/40 vision, or worse, in the better eye even with eyeglasses[7]. A person is considered legally blind if visual acuity is 20/200, or worse, with best correction in the better eye. Nearly 1 million people older than 40 years of age in the United States are legally blind[5]. The potential burden of visual impairment varies in relation to demographic differences (e.g., age, biologic sex, race/ethnicity) in disorders of vision that may lead to blindness. Among non-Hispanic White individuals, common causes of visual impairment are cataract (42.2%), age-related macular degeneration (28.1%), others (22.7%), diabetic retinopathy (4.7%), and glaucoma (2.3%)[7]. Among African Americans, the common causes of vision impairment are cataract (41.7%), others (27%), diabetic retinopathy (12.2%), glaucoma (11.3%) and age-related macular degeneration (7.8%). Among Hispanics, the common causes are cataract (48%), others (16.2%), diabetic retinopathy (15%), age-related macular degeneration (14.5%), and glaucoma (6.4%)[7].
Amblyopia and strabismus are the most common causes of vision impairment in children. Recommended screening guidelines have been found to reduce the prevalence of these two conditions, yet a high percentage of children do not have appropriate screening, and some who do have screening do not receive appropriate follow-up by an eye specialist[9,11,37]. In addition, screening is not universal and compliance with screening guidelines for children is low[37]. There is also lack of adherence to guidelines for eye examinations among adults, which contributes substantially to the high rate of vision impairment[6]. Screening and comprehensive eye examinations are needed to identify adults at risk for age-related conditions such as cataracts, glaucoma, and macular degeneration, which together account for most cases of vision impairment[14]. Symptomatic eye conditions, such as conjunctivitis and dry-eye syndrome, are common reasons individuals seek eye care, often at their primary care physician's office. A variety of other inflammatory, infectious, and noninflammatory conditions of the eye and eyelids, as well as mechanical disorders of the eyelids are also frequently seen in the primary care setting. In addition, research has shown that approximately 39% of individuals with an eye injury or emergency seek medical attention in the primary care setting[6]. Thus, primary care professionals should be skilled in the initial management of a wide variety of eye disorders and emergencies, including providing timely referral.
Clinicians should also be knowledgeable about symptomatic eye conditions associated with diseases such as diabetes mellitus, systemic hypertension, autoimmune disorders, and human immunodeficiency virus (HIV) infection. For example, diabetic retinopathy is a primary cause of vision impairment, and optimal management of the diabetes and close follow-up of the eyes are essential for preserving vision in individuals with this disease. In addition, many medications, especially several used to manage chronic diseases, have been associated with adverse ocular side effects[16,17,18,20]. Although most of these effects are minor, some are vision-threatening and require close follow-up with frequent eye examinations. Knowledge of these side effects can help healthcare professionals better recognize and treat symptomatic conditions and recommend appropriate follow-up. Malignant conditions of the eyelid and ocular tumors are rare and are best treated by specialists, but recognizing signs and symptoms and making appropriate referrals are essential.
This course addresses a range of eye disorders and their treatment, drawing primarily from evidence-based guidelines developed by the American Academy of Ophthalmology (AAO) (Table 1) [11]. To provide a better context for the discussion of eye disorders and injuries, the course begins with an overview of the anatomy and physiology of the normal eye and eyelid and provides a comprehensive discussion of the clinical examination of the eye, including recommended guidelines for eye examinations and screening for a variety of populations and preparing the clinical setting for the routine and emergency examination and treatment of the eye.
TOPICS OF SELECT EVIDENCE-BASED PREFERRED PRACTICE PATTERNS (PPPs) OF THE AMERICAN ACADEMY OF OPHTHALMOLOGY (AAO)
| |||||||||||||||||
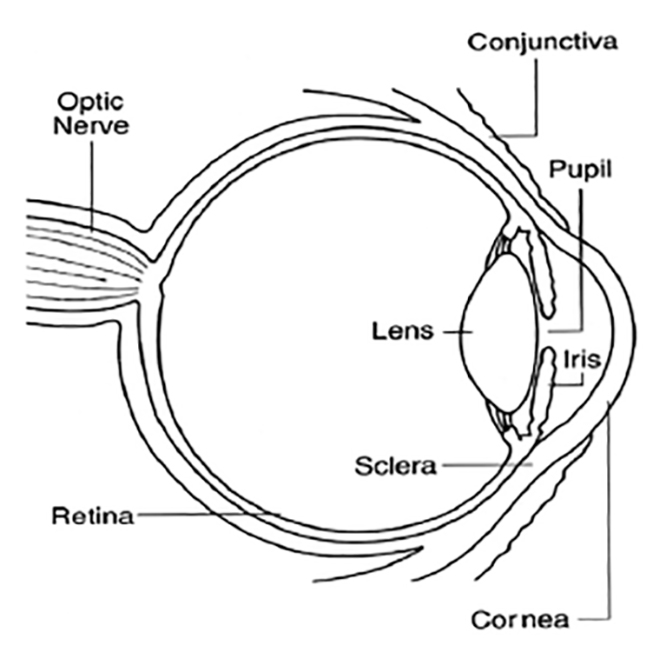
The human eye is complex, both anatomically and physiologically, with an intricate system of specialized structures, nerves, and muscles that work together as the image-forming organ (Figure 1). The primary components of the eye include the cornea, iris, pupil, lens, choroid, fovea, immaculate, retina, and optic nerve. Two unequal spheres make up the eyeball; the anterior segment is a small sphere that forms about one-sixth of the eyeball, and the posterior segment forms the remaining five-sixths[21]. The anterior pole refers to the central point of the anterior curvature of the eyeball, and the posterior pole is the central point of the posterior curvature. The posterior pole corresponds to the area of the retina between the optic disc and the immaculate[21].
Each eye has three primary membranes: the cornea and sclera (external membrane), the uvea (middle membrane), and the retina (innermost membrane).
The primary function of the external membrane is to protect the eye; other functions are filtration of ultraviolet light and refraction. The cornea comprises the anterior portion of the external membrane, and the sclera makes up the posterior portion [21,22]. The cornea has no lymphatic or blood vessels, is approximately 0.55 mm thick, and is made up of five layers. Anterior to posterior, these layers are the epithelium, Bowman's layer, corneal stoma, Descemet's membrane, and endothelium [21]. The epithelium, which is about five to six cells thick, covers the surface of the cornea. Bowman's layer is a strong, thick membrane made up of collagen fibrils arranged in a parallel pattern. This layer protects the cornea from injury. The corneal stroma is the thickest part of the cornea, comprising 90% of the total thickness. It consists primarily of lamellated connective tissue. Descemet's membrane is a layer of specialized basement membrane of endothelial cells that lies between the corneal stroma and the endothelium. A one-cell thick layer, the endothelium keeps the cornea clear by removing water from it.
The epithelium (i.e., the outer surface of the cornea) is coated with a multilayer tear film. The innermost layer of the tear film is the mucoid layer, which provides a hydrophilic layer of the cornea, allowing the tear film to remain on the corneal surface, which is naturally hydrophobic [23]. The mucoid layer is produced by goblet cells, which are found in the bulbar conjunctiva. The middle layer—the thickest layer—is produced by the lacrimal gland and sebaceous glands, primarily the meibomian glands (located in the tarsal plates along the margins of the eyelid) and the glands of Zeis (which open into the hair follicles of the eyelashes). The most external layer of the cornea, which is only a few molecules thick, is the lipid layer. This layer protects the lacrimal layer from evaporation and provides a smooth surface for the cornea.
The sclera, or "white" of the eye, is a dense membrane of collagen-elastic tissue. In the adult eye, the posterior portion of the sclera is approximately 1 mm thick, and it becomes thinner at the equator (a line that divides the eye into anterior and posterior halves). The sclera extends from the limbus at the margin of the cornea to the optic nerve, where it becomes fused with the dural and arachnoid sheaths of the optic nerve [21]. The most anterior portion of the sclera is coated with the bulbar conjunctiva, a thin transparent membrane.
The middle membrane of the eye is the uvea (i.e., uveal tract), a richly vascularized layer with pigmented tissue. The most anterior of the three components of the uvea is the iris, which surrounds the pupil, the black-looking aperture that determines the appropriate amount of light to enter the eye. Pigment in the iris gives the eye color and prevents light from passing through, thus protecting the retina from too much light. Intrinsic muscles on the pupil—the sphincter pupillae and the dilator pupillae—constrict and dilate the pupil, respectively, to allow the appropriate amount of light to enter the eye [24].
Behind the iris is the lens (i.e., the crystalline), a transparent body that is held in place by the suspensory ligaments attached to the anterior part of the ciliary body, a ring made up of epithelial, stromal, and ciliary muscle tissue. Intrinsic muscles on the ciliary body contract and relax to change the shape of the lens, a process referred to as accommodation. This process helps the eye to focus, allowing for a sharp image to form on the surface of the retina [24].
The posterior aspect of the ciliary body is attached to the choroid, a highly vascular layer that extends to the optic nerve. The choroid is thicker at the posterior pole (the area of the retina between the optic disc and the macula) and thinnest at the optic nerve head (optic disc) [22]. In the area of the optic nerve, the outer surface of the choroid is firmly attached to the sclera. The choroid consists of four layers, which are (from internal to external): Bruch's membrane, the choriocapillaris, the stroma, and the suprachoroid [21].
The innermost membrane of the eye is the retina, a thin (0.5 mm), transparent lining made up of several layers of nerve cells and photoreceptors (rods and cones) as well as a single layer of retinal pigment epithelium. The external surface of the retina is in contact with the choroid, and the internal surface is in contact with the vitreous. The center of the retina is the fovea, and the macula is a small area (representing approximately 5% of the retina) around the fovea. Approximately 3 mm to the nasal side of the fovea is the optic disc, where ganglion cell axons exit the eye to form the optic nerve [21]. A central depression in the optic disc is the optic cup. The area of the eye that includes the retina, optic disc, macula, and posterior pole is referred to as the fundus.
There are three fluid-filled structures of the eye: the anterior and posterior chambers and the vitreous body. The anterior chamber occupies the space between the cornea and the iris, and the posterior chamber lies between the iris, suspensory ligaments, and lens. Both of these chambers are filled with aqueous fluid (humor), which is produced in the posterior chamber by the ciliary body and circulates through the pupil into the anterior chamber, supplying oxygen and nutrients to the cornea and iris. Aqueous fluid also helps to maintain the pressure of the eye.
The vitreous is attached to the lens and occupies the space between the lens and the retina; the attachment is firm in young individuals and becomes weaker with age. This structure represents two-thirds of the volume of the eye and is filled with vitreous fluid, a clear, jelly-like substance made up primarily of water, with approximately 1% to 2% of the components being soluble proteins, salts, and hyaluronic acid [22]. Vitreous fluid has many functions: it transmits light, supports the posterior surface of the lens, helps to hold the retina against the retinal pigment epithelium, and transports nutrients to the cornea. During natural aging, vitreous fluid may shrink or thicken, causing tiny strands or clumps to form, the cause of muscae volitantes or so-called floaters (small spots or threads that appear in the field of vision).
The first branch of the internal carotid artery is the ophthalmic artery, and branches of this artery bring blood to each eyeball and the extraocular muscles. The two greatest sources of blood to the retina are the choroidal blood vessels and the central retinal artery [24]. Blood flows through the choroidal vessels to the innermost layer of the retina, the retinal pigment epithelium, to nourish the photoreceptors. Blood is also supplied to the retina through the central retinal artery, which has four main branches in the retina. There is a ring of blood vessels in the macular area, and the fovea contains no blood vessels or capillaries. Blood is drained from the eyeball through the superior and inferior ophthalmic veins and the infraorbital vein, which drain to vorticose veins, and the central retinal vein, which drains to the ophthalmic vein [24].
It is important to note that the retinal arteries are end arteries, meaning they have no means for collateral circulation. Therefore, in the case of central retinal artery occlusion, the retina necroses and blindness results.
The eyeball sits in an osseous socket, shaped like a pyramid, and is formed by seven articulating bones: the frontal, zygomatic, maxillary, sphenoid, ethmoid, palatine, and lacrimal bones. The orbit is also formed by a fascial sheath, referred to as the capsule of Tenon (fascia bulbi)[21]. This capsule is separated from the sclera by the episcleral space, blends with the sclera behind the junction of the sclera and cornea, and is fused to the sclera and the sheath of the optic nerve around the entrance of the optic nerve. The capsule of Tenon has two perforations: one near the equator for the veins that drain the choroid and sclera, and one at the optic nerve.
As noted, intrinsic muscles in the eye act to control the size of the pupil and to change the shape of the lens. In addition, external (extraocular) muscles enable the movement of each eye in the direction of a point of interest in any direction (side to side and up and down). The lateral and medial rectus muscles move the eye in the horizontal plane, with the lateral rectus muscle responsible for abduction of the eye (away from the nose) and the medial rectus muscle responsible for adduction (toward the nose)[24]. Movement in the vertical plane is controlled by the superior and inferior rectus and superior and inferior oblique muscles. All four of these muscles contribute to vertical movement when the eye is looking straight ahead. However, when the eye is in abduction, vertical motion is controlled by the superior and inferior rectus muscles. When the eye is in adduction, vertical motion is controlled by the superior and inferior oblique muscles. Normally, both eyes move together in the same direction.
The left and right third, fourth, and sixth cranial nerves are involved in innervating the muscles of the left and right eye, respectively (Table 2). In addition, the second cranial nerve, the optic nerve, carries afferent fibers from the retina to the primary visual cortex in the occipital lobe. The two optic nerves (one from each eye) unite in the optic chiasm and combine into the optic tract. At the optic chiasm, the optic nerves cross over to project images from each eye to the opposite side of the brain[24].
CRANIAL NERVES INNERVATING THE EXTRAOCULAR MUSCLES OF THE EYE AND EYELID
| Cranial Nerves | Muscles Innervated | Action | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Third (oculomotor nerve) |
|
| ||||||||||
| Fourth (trochlear nerve) | Superior oblique | Rotates eyeball downward and outward | ||||||||||
| Sixth (abducent nerve) | Lateral rectus | Moves eyeball laterally (abduction) | ||||||||||
| Seventh (facial nerve) | Orbicularis oculi | Closes eyelids |
The diameter of the optic nerve becomes larger as the distance from the eye increases. The fibers of the optic nerve are protected by the three meningeal layers of the central nervous system (dura, arachnoid, and pia mater). Because central nervous system fibers are incapable of regeneration, damage to the optic nerve results in irreversible blindness.
A high concentration of nerve fibers is found in the cornea. These fibers enter the cornea at its margins and radiate toward the center. They are associated with many pain receptors and have a low threshold.
The eye is primarily protected by the upper and lower eyelids, or palpebrae. The eyelids can be closed to protect the eye from too much light or from foreign substances and to aid in sleeping. In addition, the act of blinking allows for lubrication of the cornea by helping to distribute and eliminate tears. Form and support of the eyelids are provided by the tarsal plate, a thick fold of connective tissue. This plate is one of several layers; overlying the tarsal plate are (from anterior to posterior) the epidermis, dermis, subcutaneous tissue, and fibers of the orbicularis oculi muscle. The palpebral conjunctiva lines the innermost part of the eyelid.
Each tarsal plate includes a row of sebaceous glands known as the meibomian glands; these glands, together with the pilosebaceous glands of Zeis and the apocrine glands of Moll (also located in the tarsal plate), produce the lipid layer of the corneal tear film. The eyelid is closed through action of the orbicularis oculi muscle, which is innervated by the seventh cranial nerve, and is opened as a result of action of the levator palpebrae muscle, which is innervated by the third cranial nerve.
Lacrimal fluid, or tears, is produced by the lacrimal gland, a bi-lobed secretory gland located behind the upper eyelid in the superolateral angle of the orbit. The gland is about the size of an almond; the larger of the two lobes is the orbital lobe, and the smaller lobe is the palpebral lobe. The inferior surface of the latter lobe lies close to the eye and can be seen when the eyelid is everted. Excretory ducts in the orbital lobe merge with ducts in the palpebral lobe and carry lacrimal fluid onto the conjunctiva of the upper eyelid. The act of blinking spreads the lacrimal fluid medially over the surface of the eyeball. The fluid then flows into the lacrimal punctum, an opening on the medial, superior, or inferior eyelid margins, and into one of two lacrimal canals (one above the lacrimal punctum and one below). From there, the fluid flows into the nasolacrimal duct and, lastly, into the nasal cavity.
As noted, the lacrimal gland is a secondary supplier to the lacrimal layer of the corneal tear film. Lacrimal fluid contains salts, proteins, and lysozyme (a bacterial enzyme) and serves to supply nutrients (oxygen) to the cornea, carry waste products away from the cornea, and moisten and lubricate the eyeball [24]. Each lacrimal gland secretes approximately 1 mL of lacrimal fluid each day. If the conjunctiva is irritated, such as by a foreign substance or body, the lacrimal glands are stimulated to produce more lacrimal fluid, which causes tears to accumulate on the surface of the eyeball (watery eyes) and wash away the substance causing the irritation. Tears can also accumulate if the nasolacrimal ducts become blocked or if an emotional response triggers parasympathetic stimulation of the lacrimal gland to overproduce lacrimal fluid (the process of crying). The parasympathetic nerve supply originates from the lacrimal nucleus of the facial nerve in the pons.
The blood supply to the lacrimal gland is transported through the lacrimal artery, a branch of the ophthalmic artery. The superior ophthalmic vein drains blood from the gland to the cavernous sinus.
The act of vision is made possible by a process that is similar to the function of a camera. The cornea transmits and focuses light into the eye, and the intrinsic muscles of the iris constrict and expand to control the size of the pupil, allowing the appropriate amount of light to enter the eye. The contraction and relaxation of the muscles of the ciliary body change the shape of the lens, helping the eye to focus and allowing for a sharp image to form on the surface of the retina. The central point for focusing of the image (the visual axis) is the fovea.
Light enters the retina and travels through its two outermost layers, the nerve fiber layer and the ganglion cell layer, to the layer of rods and cones. Light is absorbed by photopigment in these photoreceptors and converted into electrical impulses. These impulses are sent through nerve fibers that exit the eyeball at the optic disk and are transmitted via the optic nerve to the visual cortex, where they are converted to images.
The human retina contains approximately 100 million rods and 5 million cones. Rods allow vision in dim light, and cones facilitate vision in bright light (daylight). Cones also provide color perception and high spatial resolution. Rods are responsible for peripheral vision, and the macula provides the central 10 degrees of vision. Most cones are located within the macula. The fovea is made up exclusively of cones and provides for the best visual acuity. In contrast, there are no rods or cones in the area of the retina where the optic nerve leaves the eye; as such, this area is known as the eye's blind spot.
All of the essential structures of the eye are evaluated in a comprehensive eye examination performed by an eye specialist (optometrist or ophthalmologist). Screening eye examinations, which do not include as many detailed tests, are also necessary for eye health and can be carried out by primary care clinicians. Screening eye examinations in the primary care setting can help detect visual impairment and identify individuals at high risk for occult eye disease, enabling clinicians to help prevent vision loss in their patients. Eye examinations are also important as part of overall health assessment, as the eye and its associated structures are affected by systemic diseases and medications. Indications of systemic hypertension, diabetes mellitus, collagen vascular diseases, central nervous system abnormalities, and other conditions can be detected on funduscopy, making eye examination a fundamental diagnostic component[8,25]. The AAO has established recommendations for eye examinations in children and adults, according to age and risk factors for ocular disease[8,37]. According to these recommendations, a baseline comprehensive eye examination by an ophthalmologist should be done before 40 years of age for individuals who have no signs or risk factors for ocular disease and at certain intervals depending on the age of the adult. The results of a baseline comprehensive eye examination enable the eye specialist to categorize individuals into one of three groups according to their risk for ocular disease: patients with no risk factors, patients with risk factors, or patients with established conditions that require intervention[8]. Recommendations for further screening are made according to the patient's risk category (Table 3)[8,25,26,27,28,37]. Regardless of risk, a comprehensive eye exam should be done every one to two years beginning at 65 years of age, with a focus on the identification of such age-related diseases as cataracts, glaucoma, and macular degeneration. The American Optometric Association (AOA) has also established guidelines for eye and vision examinations in adults and children[25,27].
RECOMMENDATIONS FOR EYE EXAMINATIONS AND SCREENING
| Age Groups | Interval for Examination | Comments | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Asymptomatic, No Risk | |||||||||||
| Newborn to 6 months |
|
| |||||||||
| 6 months to 3 years |
|
| |||||||||
| 3 to 5 years |
|
| |||||||||
| 6 to 18 years |
|
| |||||||||
| 19 to 39 years |
| — | |||||||||
| 40 to 54 years |
| Baseline comprehensive exam at 40 years of age | |||||||||
| 55 to 64 years |
| — | |||||||||
| ≥65 years |
| Comprehensive eye examination | |||||||||
| At Risk | |||||||||||
| Adults with diabetes | Every year (AAO) | First examination 1 to 5 years after onset of type 1 diabetes or at time of diagnosis of type 2 diabetes | |||||||||
| Risk Factors for Glaucomaa | |||||||||||
| <40 years | Every 1 to 2 years (AAO) | — | |||||||||
| 40 to 54 years | Every 1 to 3 years (AAO) | — | |||||||||
| 55 to 64 years | Every 1 to 2 years (AAO) | — | |||||||||
| >65 years | Every 1 to 2 years (AAO) | — | |||||||||
| |||||||||||
Despite the importance of eye examinations, the percentage of adults who have appropriate eye examinations is low. According to data from the National Health Interview Survey (2017), 40% of individuals at high risk for vision loss did not receive an eye exam in the previous year[6]. Rates of routine eye care vary according to race/ethnicity, socioeconomic status, and availability of vision insurance[6].
Among children, the rate of appropriate eye examinations is also low, as is the rate of follow-up examinations for children who failed visual acuity screening[33]. For example, a survey of pediatricians showed that preschool vision screening was done on 36% of children 3 years of age, 58% of children 4 years of age, and 73% of children 5 years of age[33]. Respondents gave many reasons for the lack of screening, with the most common reasons being that children were not cooperative (39%) and that testing is time-consuming (21%)[33].
Surveys have shown that primary care providers lack confidence in performing eye examinations on adults as well, perhaps because of the low number of such examinations they actually perform. In one survey, 10% of family physicians and 45% of general internists had performed a fluorescein eye examination, and 8% and 15%, respectively, had performed 10 or more[35]. This lack of experience has led to even fewer procedures precepted by family physicians and general internists, which may provide an explanation for why most residents have also reported difficulty in performing eye examinations[36].
As integral as they are to eye health, screening eye examinations in the primary care setting are not enough, especially for children and adults at high risk for ocular disease. The AAO and the American Association for Pediatric Ophthalmology and Strabismus (AAPOS) recommend that an ophthalmologist examine high-risk infants and children who have abnormal results of screening examinations[37].
Primary care clinicians should take several steps toward better serving the eye care needs of their patients, especially those of racial/ethnic minority and low socioeconomic status (Table 4). First, clinicians should enhance their knowledge and skills related to eye examinations in the wide range of populations within their practice and should carry out examinations as recommended by evidence-based guidelines. Second, each primary practice should have all the equipment, ophthalmic medications, and eye charts necessary to perform basic eye examinations, vision screening, and to treat minor eye conditions and provide initial management of eye injuries. Third, educational materials should be made available for patients to help them gain a better understanding of the importance of appropriate eye care as well as the risk and symptoms associated with eye diseases and conditions. Patient education is imperative to help improve screening rates and appropriate care, and clinicians should take advantage of each visit to encourage their patients to have their eyes and their children's eyes examined by an eye care specialist, as appropriate[38]. Lastly, establishing professional relationships with local eye specialists can provide an important resource to primary care clinicians and help facilitate timely referrals.
WAYS PRIMARY CARE CLINICIANS CAN BETTER SERVE THE EYE CARE NEEDS OF THEIR PATIENTS
|
The purpose of screening eye examinations is to detect occult disease and to identify individuals who are at high risk for such disease. Routine screening eye examinations include external assessment (orbit, lids, conjunctiva, cornea, iris, and pupil), evaluation of corneal reflexes, and assessment of red reflexes on funduscopy (Table 5)[8,25]. Visualization of the fundus with the use of a direct ophthalmoscope is essential for detecting occult eye disease. However, examination of other aspects of the eye can provide important information about the presence or risk of ocular disease. In addition, testing of vision is important, especially for patients with vision-related symptoms or diabetes. In general, a screening eye examination in the primary care setting includes the following components[8,25]:
History
External examination
Evaluation of extraocular muscle motility
Assessment of pupil function
Testing for visual acuity
Testing of visual field
Direct ophthalmoscopy (funduscopy)
COMPONENTS OF EYE EXAMINATIONS FOR ADULTS
| Component of Examination | Description of Testing | Indicators | ||
|---|---|---|---|---|
| Screening Examination | ||||
| History | Elicit details regarding eye health, overall health, and use of medications. | Age, race/ethnicity, family history, eye history, lifestyle habits, and use of medications all may contribute to the risk of ocular disease. | ||
| Examination of external structures | Evaluate eyebrows, eyelashes, eyelids, orbital area, lacrimal apparatus, cornea, and sclera. | Abnormalities may suggest ocular disease. | ||
| Assessment of pupils | Assess size, symmetry, and reaction to light. | Abnormal findings may suggest trauma, neurologic disorder, or intraocular problem. | ||
| Eye muscle test | Evaluate eye movements in all directions. | Abnormal movement may indicate systemic disease or neurologic lesion. | ||
| Visual acuity test | Examine vision with Snellen chart (at 20 feet) or Rosenbaum card (at 14 inches). | Lack of "normal vision" (20/20) denotes refractive error and need for corrective lenses. | ||
| Confrontational exam (perimetry) | Examiner moves hand in and out of quadrants of visual field while individual covers one eye and looks straight ahead. | An abnormal finding indicates loss of peripheral vision due to lesion of the retina, optic nerve, or optic chiasm. | ||
| Tangent screen exam (perimetry) | Screen with target for individual to focus on; another object on the screen moves in and out of the periphery. | |||
| Automated perimetry | Individual looks into an instrument while a computer program flashes small lights in the periphery. | |||
| Ophthalmoscopy (funduscopy) | Evaluate fundus (i.e., retina, optic disk, and choroid); usually done after dilating the pupils with eye drops. | Abnormalities in fundus may indicate a wide variety of ocular disorders or signs of systemic disease. | ||
| Direct ophthalmoscopy | Examine fundus with ophthalmoscope. | |||
| Indirect ophthalmoscopy | Examine fundus with use of forehead apparatus, which provides brighter light than ophthalmoscope (for visualization in greater detail and in three dimensions). | |||
| Comprehensive Examination | ||||
| Slit-lamp examination | Evaluate with slit-lamp microscope, which enlarges and illuminates the front of the eye. | Staining with fluorescein eye drops can aid in the detection of lacerations, abrasions, tears, foreign objects, or infections on the cornea. | ||
| Applanation tonometry | Tonometer used in slit-lamp microscope measures the amount of force needed to temporarily flatten a part of the cornea; fluorescein eye drops and eye drops with an anesthetic are used. | High intraocular pressure is a sign of glaucoma. | ||
| Noncontact tonometry | A puff of air is used to measure intraocular pressure. | |||
| Refraction assessment |
| — | ||
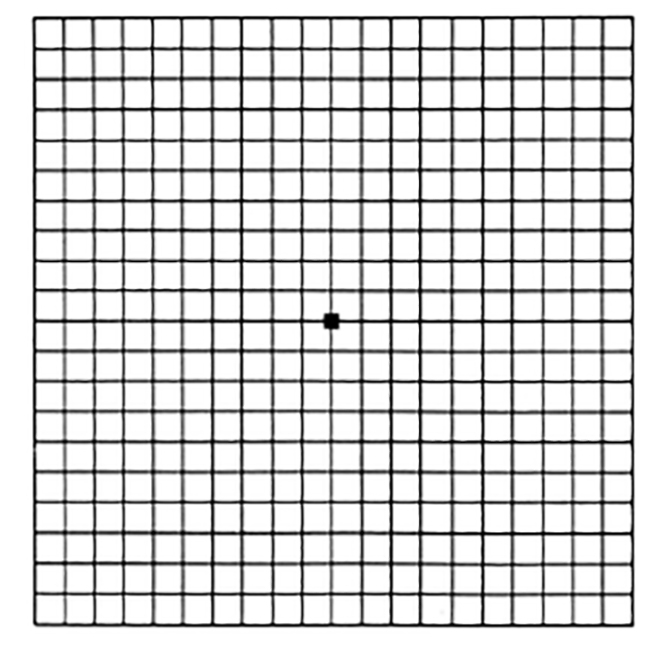
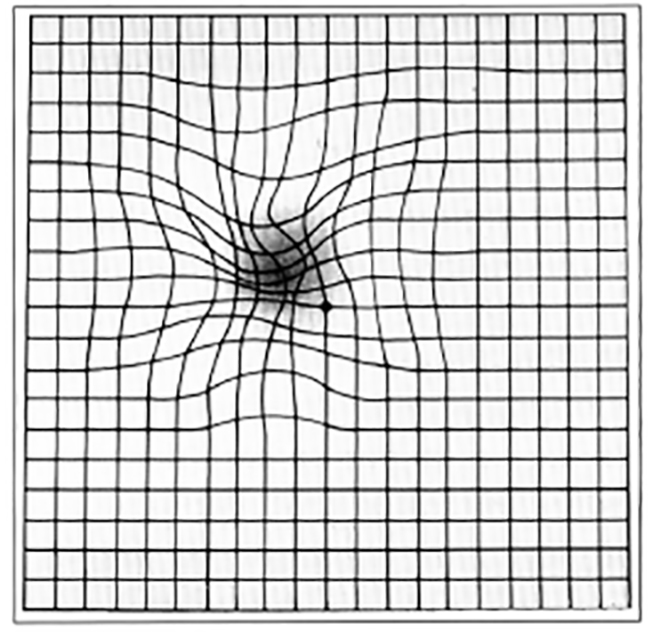
| Amsler grid (test of central visual field) | Patient covers one eye and is asked to fixate on point at center of grid, which is held 14 inches away. | Central areas of distortion suggest macular (rather than optic nerve) disorder. | ||
| Gonioscopy | Visualize structures of anterior chamber angle. | Visualization of angle is essential for evaluation of patients with glaucoma. | ||
| Fundus photography | Camera used to take color photographs of fundus | Photos provide clear views to assess signs of retinopathy or macular degeneration. | ||
As with the clinical evaluation for most disease settings, the patient's history provides details that are essential for making an accurate diagnosis. The history should include not only information specific to the eye but also details related to overall health, family eye health, systemic diseases, use of medications (including prescription and nonprescription drugs as well as herbal or vitamin supplements), and vocational and avocational vision requirements [8]. Eye health history should include the patient's self-assessment of his or her current visual function, including the use of corrective lenses; eye-related symptoms; and previous eye disease, injury, surgery, or medications. The family history as it relates to both ocular and systemic disease should be elicited. Because of the influence of lifestyle and environmental factors on the risk of many ocular diseases, the patient should also be asked about his or her occupation, smoking history, alcohol use, and living situation [8,25].
The external examination is directed at evaluating the eyebrows, eyelashes, orbital area, eyelids, lacrimal apparatus, and cornea and sclera [8]. The eyelids are evaluated to ensure that the upper eyelids overlap the iris but not the pupil, that they open and close completely, and that they move symmetrically. Signs of edema, sagging, or lesions in the orbital area should be noted. Retraction of the upper or lower eyelid and use of a penlight allows for examination of the conjunctiva and sclera.
To evaluate eye movement, the clinician should ask the patient to follow a target (light from a penlight) with both eyes into the cardinal fields of gaze. Normal movement is smooth, symmetric, and full [8]. Problems with eye movement may be a sign of systemic disease or a neurologic lesion. For example, poor abduction may be an indicator of Graves eye disease, myasthenia gravis, or palsy of the sixth cranial nerve.
Pupils should be equal, round, and reactive to light (which is documented as the acronym PERRL). It is fairly common for there to be subtle inequalities (up to 0.4 mm) in the size of the two pupils, but this inequality is normal only if the asymmetry remains constant during changes in ambient light [39]. In assessing anisocoria (unequal pupils), the first two questions should be: Is there a normal light reaction? Is anisocoria worse in darkness or light? If the light reaction is poor in the eye with a larger pupil, the pupillary constrictor of that eye is abnormal; if the light reaction is good in both eyes, the pupillary dilator of the eye with smaller pupil is abnormal [39]. Anisocoria that is worse in light and accompanied by ptosis and paresis of extraocular muscles may be indicative of intracranial aneurysm. Anisocoria may indicate Horner syndrome if ipsilateral ptosis and anhidrosis are also present. Other possible causes of unequal pupils include trauma-related damage to the iris and pupil and palsy of the third cranial nerve. Irregularly shaped pupils are most commonly related to an intraocular problem, such as congenital anomaly, inflammation, or trauma.
To assess pupillary reaction to light, a penlight is used while the patient fixates on a distant target; dim lighting in the room is best for the assessment. In individuals with normal pupils, there is consensual response to light; that is, both pupils will have equal constriction in response to light shined in one eye. Poor reaction of pupils is suggestive of many conditions, including diabetes, neurologic disease, optic nerve disease, or damage from trauma or surgery. In addition, abnormal reaction may be a sign of a serious ocular problem, such as acute angle-closure glaucoma or uveitis [39].
The size of the pupils can be affected by the use of systemic medications or drugs. Morphine, heroin, and parasympathetic agents (e.g., pilocarpine, demecarium bromide) cause constriction (miosis) of the pupils, and anticholinergic agents cause the pupils to dilate (mydriasis). When evaluation of the pupils demonstrates abnormality, examination with slit-lamp microscopy should be done to rule out causative factors, which may include an occult foreign body, intraocular inflammation or adhesions, a perforating injury, surgical trauma to the iris, and angle-closure glaucoma.
An integral component of all eye examinations is testing of visual acuity, or the quantitative measure of the ability of the eye to see an image in clear focus at a certain distance. Beyond assessing visual acuity in order to determine the need for correction of refractive errors, such testing is important because a loss of visual acuity is a sensitive indicator of occult eye disease [8,25].
Visual acuity is usually measured with a wall-mounted Snellen chart. Patients are asked to cover one eye and read the line of the smallest letters they can at a distance of 20 feet; the test is then repeated with the other eye covered. The lines of letters on the chart correspond to specified visual acuity based on what a person with "normal vision" can see at various distances. A smaller version (to scale) of the Snellen chart is the Rosenbaum card, which can be held 14 inches away from the patient.
The term 20/20 vision applies to the ability to see an image at 20 feet that a person with "normal vision" can see at 20 feet. The ability to see an image at 20 feet that a person with normal vision can see at 40 feet is defined as 20/40 vision. When a patient has visual acuity that is worse than 20/20, the primary care clinician should provide a referral to an eye specialist for assessment of the refractive error and prescription of corrective lenses 8; 25].
In the primary care setting, the patient's visual field is usually assessed with the confrontation visual field exam. With this test, the patient is asked to cover one eye and fixate the contralateral eye on the clinician. The clinician holds up fingers in various quadrants of the field of vision to determine whether there is a deficiency in peripheral vision. Technology available in the specialty setting enables assessment of the visual field with use of the tangent screen exam or automated periphery (a computer program).
A deficiency in the visual field may indicate a lesion of the retina (macular degeneration), optic nerve (optic neuritis, glaucoma), or optic chiasm (tumors, stroke, trauma). The site of the lesion can be identified by mapping the visual field deficit.
The use of a direct ophthalmoscope enables visualization of the retina and the blood vessels of the eye. The fundus is best visualized after dilation of the pupil with eye drops, but a limited view can be achieved through an undilated pupil. In the latter situation, the room should be darkened and the patient should be asked to focus on a far corner of the room.
The red reflex is the term used to describe the reddish-orange reflection of the retina. This reflex can be found by looking through both pupils with the ophthalmoscope at a distance of about 2 feet and then moving to within a few inches of the patient. Evidence of decreased brightness, asymmetry, or the existence of a focal defect should prompt referral to an eye specialist for a comprehensive eye examination [40].
The primary objective of direct ophthalmoscopy is to evaluate the optic disc and the retinal vasculature. The outline of the disc should be clear, and the central area of the physiologic cup should be pale. The retina appears as reddish-orange, and the macula is dark. The retinal arteries should appear bright red, and veins should be slightly purple. It is vital to recognize swelling of the optic disc, as this is usually an indication of a serious condition.
A comprehensive eye examination for adults is done to detect ocular disease, visual dysfunction, or ocular signs of systemic disease [8]. This examination includes all of the components of a screening examination, as well as examination with slit-lamp microscopy and tonometry; other specialized tests may be warranted on the basis of symptoms or findings. A slit-lamp microscope allows for magnified viewing of the eye structures from the cornea to the vitreous. Fluorescein eye drops can be used to aid in the detection of tiny corneal tears or scrapes, foreign objects, or signs of infection.
Tonometry provides a measurement of intraocular pressure, a sign of glaucoma. Two methods are available to measure pressure. With contact applanation tonometry, fluorescein drops and anesthetic eye drops are used and the tonometer is slowly moved toward the eye and makes contact with the cornea. The amount of force needed to temporarily flatten a part of the cornea is measured. This method is preferred to noncontact tonometry (emitting a puff of air to test intraocular pressure) [8,25].
Other tests that may be incorporated into an eye examination include optical coherence tomography, central visual field testing (Amsler grid), gonioscopy, fundus photography, keratometry, pachymetry, glare testing, contrast sensitivity testing, and dry eye assessment [8,25].
According to guidelines established jointly by the American Academy of Pediatrics (AAP), the AAO, the AAPOS, and other eye-related professional organizations, an examination for eye problems should be done in the newborn period and at all of the child's subsequent routine health visits [27,37]. The eye examination for children consists of essentially the same components as for adults, with age-appropriate modifications [27,37]. Several approaches can help to maintain the child's cooperation and provide optimum results [27]:
Perform the examination when the child is alert (e.g., early morning or after a nap)
Arrange the office visit so that the eye examination is done first or near the beginning (e.g., before the patient has endured other aspects of the physical examination or has received vaccinations)
Have young children sit on the parent's or caregiver's lap
Demonstrate, with the assistance of the parent or caregiver, the testing procedure before carrying it out
Carry out each component of the evaluation rapidly
Perform the least-threatening examinations first (e.g., external examination, red reflex test, assessment of binocular motility and vision, then assessment of monocular motility and vision)
Provide a variety of interesting targets for the child
Examine the eye in normal light if darkening the room frightens the child
A screening eye examination that includes the red reflex test should be done by 6 to 12 months of age [37]. When examining an infant for the first time, the clinician should ask the parent or caregiver about the prenatal, perinatal, and postnatal history; on subsequent examinations, the developmental history of the child should be elicited [27]. The clinician should also ask about the family eye history and should encourage parents or caregivers to describe their observations about their children's eyes and vision. For example, does the child hold objects close to the face when trying to focus? Do the child's eyes seem to cross or drift? Does one eyelid (or both) droop or tend to close?
The clinician should examine the eyes and external structures thoroughly for signs of inflammatory conditions and infection. Evaluation of ocular motility is of particular importance when examining children of preschool or early school age in order to detect strabismus[37]. Ophthalmoscopy and age-appropriate measurement of visual acuity should be attempted for children who are 3 years of age or older. The cross cover test should be used to evaluate binocular vision and motility in children of all ages[27].
Testing of visual acuity in children who cannot communicate verbally is done by evaluating the ability to fix and follow an object (cross-cover test), and this may be carried out binocularly and monocularly[27,37]. For older children, visual acuity can be assessed with vision testing machines or a variety of picture cards and wall charts (e.g., LEA symbols, Snellen letters or numbers, tumbling E test, or HOTV test)[27,37]. The guideline developed by the AAP in conjunction with other organizations includes detailed information on these tests and how to perform and interpret them[41].
When visual acuity cannot be measured in children 3 to 4 years of age, a second attempt should be made after four to six months. For children 4 years of age and older, a second attempt should be made after one month[37]. If visual acuity cannot be measured after two attempts in the primary care setting, the child should be referred to an ophthalmologist.
The results of the eye examination may warrant further evaluation by a pediatric ophthalmologist or an eye specialist trained to treat children, and for some signs or suspected conditions, urgent referral is needed to preserve vision (Table 6)[27,37,41,42]. The findings of the examination should be clearly communicated to the parent or caregiver, with special emphasis on the need for follow-up care or for evaluation by an ophthalmologist. A professional interpreter should be used if patients and/or their caregivers do not speak the same language as the clinician.
FINDINGS IN THE PEDIATRIC PATIENT THAT WARRANT REFERRAL TO AN OPHTHALMOLOGIST
| Sign, Symptom, or Condition | Indication for Referral |
|---|---|
| Signs | |
| Anisocoria | Expedited |
| Asymmetry in terms of color, brightness, or size on the red reflex test | Expedited |
| Poor binocular fixation after 3 months of age | Routine |
| Cataract | Expedited |
| Cloudy or asymmetrically enlarged corneas | Expedited |
| Corneal opacities (congenital) | Expedited |
| Corneal ulcer | Urgent |
| Detection of an eye muscle imbalance on assessment of ocular motility | Routine |
| Movement in or out when shifting the cover on the cross cover test | Routine |
| Nystagmus | Within few weeks of onset |
| Optic disk abnormalities | Urgent if papilledema is suspected; expedited if associated with decreased vision; routine otherwise |
| Proptosis | Urgent if orbital cellulitis is suspected; expedited otherwise |
| Ptosis | Expedited if Horner syndrome or palsy of the third cranial nerve is suspected |
| Retinal detachment (suspected) | Urgent |
| Conditions | |
| Blepharitis | If persistent or resistant to local treatment with lid hygiene and topical antibiotics |
| Chalazion | If persistent or resistant to local treatment with lid hygiene and topical antibiotics |
| Conjunctivitis (allergic) | If severe or persistent after treatment with topical antihistamines or mast-cell stabilizers |
| Conjunctivitis (infectious) | If severe or persistent after treatment with topical antibiotics |
| Dacryocele | Expedited |
| Herpes simplex virus keratitis | Urgent |
| Iritis | Expedited |
| Other | |
| Inability to read letters or lack of verbal skills as appropriate for age (when eye disease is suspected) | Expedited |
| Presence of congenital or genetic ocular anomalies or infections (e.g., aniridia, toxoplasmosis) | Routine |
| Presence of systemic syndromes, metabolic disorders, or chromosomal abnormalities with possible ocular involvement (e.g., juvenile rheumatoid arthritis, galactosemia, diabetes mellitus, Marfan syndrome, Down syndrome) | Routine |
| Suspected abuse | Urgent |
For adults and children who have visual acuity that is worse than 20/20, a comprehensive eye examination also includes assessment of the refraction. A computerized refractor may be used to measure the eyes and estimate the prescription needed to correct the refractive error. An apparatus containing wheels of different lenses is then used to determine the combination of lenses that provides the sharpest image. If testing with these lenses cannot attain 20/20 vision, evaluation should be carried out to determine if an underlying pathologic condition is the cause of the abnormal vision.
With normal refraction (emmetropia), light focuses directly on the retina. With a refractive error (ametropia), an irregular shape of the cornea causes light rays to focus either in front of or behind the retina. In myopia, close images are seen clearly but images farther away are blurry because light rays focus in front of the retina. In contrast, with hyperopia, close images are blurry and images at a distance are clear, as light rays focus behind the retina. Often developing in conjunction with either myopia or hyperopia is an astigmatism, in which the cornea has an ellipsoidal shape, causing light to focus on two points on the retina rather than one. As a result, the eye has different focal points in different planes, and it is difficult for the individual to see fine detail. Presbyopia is a condition that develops as part of the normal aging process, usually affecting individuals 40 to 50 years of age. With this condition, the lens loses elasticity and thus its ability to accommodate on close objects. While these changes are typically treated by corrective lenses or surgery, in 2021 the FDA approved pilocarpine (1.25% solution) eye drops to improve age-related deficits in near vision [215].
Approximately 75% of adults older than 40 years of age have ametropia requiring refraction correction [8,43]. However, many refractive errors are not sufficiently corrected and are, as such, the leading cause of mild visual impairment [43]. Among all adults, myopia occurs more frequently than hyperopia (25% vs. 10%), and hyperopia is more common in women than men; both myopia and hyperopia have been found to be more common in White adults than in Hispanic or Black adults [43]. A high degree of myopia has been associated with a higher risk of ocular diseases that may lead to vision loss (e.g., glaucoma, cataract, retinal detachment).
The Multi-Ethnic Pediatric Eye Disease Study (MEPEDS) group and the Baltimore Pediatric Eye Disease Study group have carried out several studies to determine the prevalence of vision disorders in children. In contrast to the findings in adults, hyperopia is the more common refractive error among children [44]. Significant racial/ethnic differences have been found in refractive errors, with the prevalence of hyperopia highest among non-Hispanic White and Hispanic children and lowest among Black and Asian children [44,45,46]. Hyperopia occurs most often in infants and older children and more often in girls than boys [44]. In contrast, the prevalence of myopia is highest among Asian, Black, and Hispanic children and lowest among non-Hispanic White and American Indian children [44,45,46]. The prevalence of astigmatism is highest in Hispanic and Asian children and lower in Black, non-Hispanic White, and American Indian children [44,46,47,48]. Astigmatism occurs most often in infants, with one study showing that it is three times as likely in children 6 to 12 months of age compared with children 5 to 6 years of age [44,47,48]. Astigmatism and a high degree of hyperopia are risk factors for bilateral decreased visual acuity in children [49]. Maternal smoking during pregnancy has been found to be associated with both astigmatism and hyperopia [45,48]. The findings of these studies can help clinicians identify children at highest risk for vision disorders.
Equipping the primary care office setting with visual acuity testing materials, a well-stocked eye tray, and basic ophthalmic instruments, medications, and supplies, helps to prepare the clinician for routine and emergency examinations and interventions. In addition to the basic visual acuity charts, such as the wall-mounted Snellen chart and the handheld Rosenbaum card, a variety of charts and cards for children of different ages should be available. Among the basic instruments needed are a visual acuity chart, penlight, direct ophthalmoscope, cobalt light source, and a tonometer[12]. A direct ophthalmoscope is adequate for performing a thorough examination in most cases, but a slit-lamp microscope is useful if the clinician wishes to examine the cornea and anterior chamber, remove foreign bodies from the cornea, or measure intraocular pressure[12].
A small supply of topical medications to rapidly decrease intraocular pressure (e.g., beta-blockers, carbonic anhydrase inhibitors, prostaglandin F2αanalogs) should be kept on hand. Other medications that should be kept in stock are short-acting mydriatic agents (e.g., tropicamide, cyclopentolate), miotic agents (e.g., pilocarpine), anesthetics (e.g., tetracaine or proparacaine), topical nonsteroidal anti-inflammatory drugs (e.g., ketorolac or diclofenac), topical antibiotics (e.g., bacitracin, erythromycin, gentamicin), and lubricants/irrigants (e.g., Lacri-Lube or BBS)[12]. An eye tray should also be readily available (Table 7), and it should be stocked independently to avoid missing items in an emergency[12]. Because of the limited use of most ophthalmic medications, the drug labels should be routinely checked and medications should be replaced according to the expiration date[12].
CONTENTS OF EYE TRAY FOR EMERGENCY TREATMENT
|
Primary care clinicians should provide their patients with education about the need for screening and comprehensive eye examinations, especially with regard to patient risk factors and health status, and encourage patients to have an ophthalmologist as a primary eye care physician [40]. Clinicians should also discuss the importance of fulfilling prescriptions of corrective lenses. Educational materials in English and other languages appropriate for the clinician's specific patient population should be readily available. These materials can enhance the clinician's discussion of specific eye-related disorders as well as emphasize the need to adhere to recommended eye examinations and treatment for diseases or conditions with ocular manifestations.
Establishing professional relationships with local eye specialists is beneficial for both primary care clinicians and their patients. Through such relationships, clinicians can establish the timing of appropriate referrals (i.e., when referrals should be same-day, urgent [within two or three days], or routine [within seven days]). Eye specialists can also be a valuable resource in determining the indications for diagnostic testing and implementing specific interventions, such as patching and topical steroid therapy. Eye specialists can include general and subspecialty ophthalmologists, pediatric ophthalmologists, and optometrists. The AAO has established guidelines for signs, symptoms, and diseases that warrant referral to an ophthalmologist for definitive diagnosis and treatment (Table 8)[40].
AAO GUIDELINES FOR REFERRAL TO AN OPHTHALMOLOGIST
|
Most patients first seek medical attention in the primary care setting for symptomatic conditions of the eye and eyelid. In general, these conditions can be managed effectively by the primary care clinician, although referral may be necessary when there is no or limited response to treatment. Symptomatic conditions include a wide range of inflammatory and noninflammatory conditions of the eye and eyelid and mechanical disorders of the eyelid. The conditions described here are those occurring most commonly in adults. Further information on the condition in children, when different, is provided.
Conjunctivitis is inflammation of the membrane that lines the underneath surface of the eyelids and the exposed surface of the sclera. Of the so-called red-eye-associated conditions, conjunctivitis is encountered most often by primary care clinicians and is one of the most common diagnoses in family practice [52]. One study found that more than 80% of all cases of acute conjunctivitis are diagnosed by non-ophthalmologists [52]. The condition is most often caused by the highly contagious adenoviruses; other causes include infectious micro-organisms, chemicals, mechanical eye irritation, allergies, immune-mediated factors, and neoplastic diseases [10]. In general, bacterial conjunctivitis is uncommon, and fungal or parasitic cases are rare. Any ocular chemical irritants, foreign bodies, or trauma can introduce conjunctivitis. These noninfectious agents cause inflammation in only the affected eye. The features and treatment differ according to the type of conjunctivitis. Conjunctivitis rarely causes permanent vision loss or structural damage but is a socioeconomic burden because of lost work or school time and the cost of medical visits and treatment [10].
The typical characteristics of conjunctivitis are a red eye, blurred vision that can be cleared with a blink, purulent or watery discharge, and itching [10]. Primary care clinicians should promptly refer patients with conjunctivitis to an ophthalmologist if any of the following is present [10]:
Loss in vision
Moderate or severe pain
Severe, purulent discharge
Corneal involvement
Conjunctival scarring
Lack of response to treatment
Recurrent episodes
History of herpes simplex virus eye disease
Immunocompromised status
Adenoviral Conjunctivitis
Adenoviral conjunctivitis can be difficult to distinguish from bacterial conjunctivitis. Adenoviral conjunctivitis is more likely to affect both eyes (often sequentially), and the eye discharge is either serous or clear [10]. Preauricular lymphadenopathy and petechial and subconjunctival hemorrhage are also distinctive signs. A rapid, in-office immunodiagnostic test is available to detect adenoviral antigen in specimens of conjunctival fluid [55].
Antimicrobial agents are not effective against adenoviruses, and corticosteroids should also be avoided, as they may actually prolong adenoviral infection [10]. The management of adenoviral conjunctivitis is directed toward symptom relief combined with scrupulous hygiene and other measures to prevent secondary spread of adenovirus conjunctivitis [10]. Artificial tears, cold compresses, and topical vasoconstrictors relieve symptoms. The AAO has noted that there is insufficient evidence to support the use of antiviral agents to treat adenoviral conjunctivitis [10]. Patients and caregivers should be told to wash their hands often, with soap and water rather than sanitizer only; to avoid touching their eyes; and to avoid close contact with others for at least seven days after the onset of symptoms [10].
Conjunctivitis related to herpes simplex or herpes zoster virus should be treated with topical and/or antiviral agents to prevent corneal infection [10]. Options include topical ganciclovir (0.15% gel) or trifluridine (1% solution), or oral acyclovir; oral valacyclovir and famciclovir may also be used [10]. Topical corticosteroids should be avoided. Herpes zoster vaccination should be strongly recommended in patients older than 50 years of age [10].
Bacterial Conjunctivitis
Bacterial conjunctivitis affects either one or both eyes, and eye drainage is either purulent or mucopurulent. Bacterial cultures of eye drainage may be helpful for recurrent, severe, or chronic purulent conjunctivitis [10]. Although bacterial conjunctivitis usually resolves spontaneously and does not cause any serious harm, a systematic review demonstrated that the use of antibiotic eye drops was associated with modest improvements in the rates of clinical and microbiologic remission [55]. No one broad-spectrum antibiotic covers all potential conjunctival bacterial pathogens, and the choice the antibiotic is usually empiric [10]. No specific antibiotic has been found to be superior to another, and factors for choosing may thus focus on convenience and cost [10].
Allergic Conjunctivitis
Primary itchiness is the hallmark of allergic conjunctivitis; drainage from the eye is usually serous or mucoid, and the condition is typically bilateral [10,56]. Allergic conjunctivitis may be treated with over-the-counter antihistamine/vasoconstrictor agent, second-generation topical histamine H1-receptor antagonists, mast-cell stabilizers, or medications that combine antihistamine activity with mast-cell inhibitors [10]. Second-generation topical histamine H1-receptor antagonists are more effective than vasoconstrictor agents [10]. Mast-cell inhibitors include cromolyn, lodoxamide, nedocromil, and pemirolast. Among the combination medications are azelastine, bepotastine, epinastine, ketotifen, and olopatadine. Ketorolac ophthalmic solution, a nonsteroidal anti-inflammatory drug, is also U.S. Food and Drug Administration (FDA) approved for the treatment of allergic conjunctivitis [10]. If such treatment does not adequately control symptoms, a short (one- to two-week) course of low-potency topical corticosteroids may be added to treatment [10]. Other measures to alleviate symptoms include cold compresses, artificial tears, and oral antihistamines, as well as allergen avoidance. If allergic conjunctivitis cannot be controlled with topical medications and oral antihistamines, consultation with an allergist or dermatologist may be helpful [10].
Keratitis is an inflammatory condition of the cornea caused by an adenovirus, herpes simplex virus, bacteria, or fungi[56]. Moderate-to-severe adenoviral keratitis presents with edema of the eyelid, preauricular lymphadenopathy, and chemosis; a punctate pattern is seen on fluorescein staining[56]. Keratitis caused by herpes simplex virus frequently occurs after conjunctivitis, usually involves only one eye, and is often associated with uveitis. Blisters may be present on the eyelid, and fluorescein staining shows classic dendritic corneal lesion[56].
The primary risk factor for bacterial and fungal keratitis is contact lens use; other common risk factors are pre-existing ocular disease and ocular trauma[55,58].Pseudomonas aeruginosa,Staphylococcus aureus, and other coagulase-negative staphylococci are the most common causative micro-organisms[58]. Treatment with topical antibiotic should be empiric, and the eye should not be patched[10,59]. Bacterial keratitis carries a heavy burden: in 2010, 930,000 physician office and outpatient clinic visits and 58,000 emergency department visits were for bacterial keratitis or contact lens disorders[60]. The estimated cost was $175 million in direct healthcare expenditures annually[60]. This burden calls for clinicians to heighten awareness of proper hygiene among their patients who wear contact lenses, emphasizing several points (Table 9) [61].
PATIENT EDUCATION TO HELP PREVENT EYE INFECTION IN CONTACT LENS WEARERS
|
Ultraviolet radiation, as from tanning beds, welding, and photographic flood lamps, can burn the cornea, causing keratitis or keratoconjunctivitis [59]. Symptoms usually occur 8 to 12 hours after exposure [56]. The most common symptoms are pain, redness, swollen eyelids, photophobia, and headache. Treatment includes short-acting cycloplegic drugs, antibiotic solution or ointment, and a patch (over the more severely affected eye) [59]. Ultraviolet keratitis typically resolves within 24 to 48 hours without vision loss; and the eye should be rechecked in 24 hours [59].
Dacryocystitis is the acute or chronic inflammation/infection of the lacrimal sac and is usually accompanied by a blocked nasolacrimal passage [57]. Among adults, dacryocystitis is most often caused by chronic sinusitis or facial trauma; in infants, the most common cause is a nasolacrimal passage that has not formed or opened normally [57]. The medial lower lid location differentiates the swelling from styes and internal hordeolum. Typically, there is redness and swelling over the lacrimal sac and the surrounding lid tissue. An ophthalmologist should be consulted promptly [57].
In North America, the most common causative pathogen is S. aureus. Methicillin-resistant S. aureus (MRSA) is four times more likely to be associated with acute versus chronic dacryocystitis [63].
The most common therapies are oral cephalosporins and amoxicillin/clavulanic acid [63]. These antibiotics are useful against a spectrum of Gram-positive bacteria and some Gram-negative bacteria. Amoxicillin/clavulanic acid is also effective against anaerobes. As almost 30% of bacteria cultured from patients with dacryocystitis are resistant to these empiric antibiotics, further antibiotic therapy should be tailored to lacrimal sac culture results and sensitivity testing [63]. Lacrimal drainage surgery should be considered if nasolacrimal duct obstruction contributes to the patient's dacryocystitis, especially if MRSA is identified [63,64,65].
Uveitis is inflammation of the uvea, or the iris, ciliary body, and choroid; the condition is categorized by site: anterior, intermediate, and posterior uveitis and panuveitis [59]. The cause of uveitis is usually unclear. It may follow certain acute infections, toxin exposures, or bruising of the eye. Cytomegalovirus (CMV), herpes viruses, Pneumocystis jiroveci, and toxoplasmosis are the most common causes of infectious uveitis [59]. On occasion, uveitis is a "herald sign" of an unrecognized autoimmune disorder or granulomatous disease (e.g., sarcoidosis, tuberculosis, histoplasmosis).
Symptoms usually develop rapidly, in one or both eyes, and vary according to the location. Anterior uveitis is usually the most symptomatic, causing pain, redness, photophobia, and decreased vision. Intermediate and posterior uveitis are not usually associated with pain, but floaters may develop and vision may decrease. Panuveitis may be associated with all symptoms and signs [59]. The clinical evaluation to diagnose uveitis includes visual acuity testing, funduscopic examination, measurement of ocular pressure, and slit-lamp examination [59]. With anterior uveitis, cells and flare are found in the aqueous humor and keratic precipitates may be present. Features of intermediate uveitis include cells in the vitreous humor and aggregates and condensations of inflammatory cells over the pars plana. Posterior uveitis is also associated with cells in the vitreous humor, as well as with white or yellow-white lesions in the retina and/or choroid, retinal vasculitis, and optic disk edema [59]. Intraocular pressure may be substantially elevated or decreased [62].
Because uveitis is a leading cause of blindness in the United States, urgent referral to an ophthalmologist is recommended [62]. A cycloplegic-mydriatic drug and corticosteroids (given topically or by periocular injection) is the usual treatment, and an agent may be needed to lower intraocular pressure; immunosuppressants may also be part of therapy. Systemic corticosteroids are used for refractory uveitis [59,62]. Systemic treatment with corticosteroids and immunosuppressants was compared with local therapy with a fluocinolone acetonide implant for noninfectious intermediate and posterior uveitis and panuveitis. Compared with systemic therapy, implant therapy was associated with some benefits, including a lower rate of residual active uveitis and improved vision-related quality of life, but improvement in visual acuity did not significantly differ between the two groups, and higher rates of risk for cataract surgery, treatment for high intraocular pressure, and risk for glaucoma were associated with implant therapy [66]. The authors suggested that treatment be individualized according to a patient's specific circumstances. In a seven-year extended follow-up to the study, the authors found that participants randomized to receive systemic therapy had better visual acuity than those who received implant therapy [67].
Infection causes endophthalmitis, an acute, diffuse uveitis [59]. Localized endophthalmitis most commonly occurs as a complication several days after cataract surgery but may also be induced by ocular trauma or foreign bodies [50,62]. Additionally, bacterial endophthalmitis may be endogenous, with the liver, lung, endocardium, urinary tract, and meninges being the most common sources of infection [68]. Symptoms include periocular pain, vision loss, diffuse redness of the conjunctiva, and swollen eyelids [62]. The cornea is often hazy, and a layer of pus may occur on the meniscus (hypopyon).
Most cases are caused by Gram-positive bacteria (most frequently, Gram-positive coagulase-negative micrococci) as well as staphylococcal and streptococcal species [59,69]. Traumatic endophthalmitis occurs more frequently in rural areas than urban settings, as individuals are more likely to be exposed to contaminated soil and animal matter [70]. Staphylococcus and Bacillus cereus infection are usually associated with endophthalmitis following outdoor penetrating trauma to the eye [71].
Patients with endophthalmitis should be referred to an ophthalmologist immediately, as prompt treatment is crucial to preserving vision [62]. Cultures of aspirate from the aqueous and vitreous should be done to determine the causative organism [62]. Treatment of endophthalmitis after cataract surgery usually consists of intravitreal injection of antibiotics, and primary vitrectomy may be done, although its benefit is unclear [69]. Treatment outcomes depend on the causative organism, with outcomes best for cases of Gram-positive coagulase-negative infections, followed by Gram-negative and S. aureus infections [69]. Endogenous endophthalmitis is most often treated with both systemic and intravitreal antibiotics, and vitrectomy is sometimes done [68]. The most commonly used intravitreal antibiotics are vancomycin (for Gram-positive infections) and ceftazidime (for Gram-negative infections) [59,68]. Other potential treatments being investigated for the management of endophthalmitis include perioperative prophylaxis with antibiotics and adjunctive steroid therapy [72,73].
Scleritis defines focal or diffuse scleral inflammation. It is often linked to systemic autoimmune diseases such as rheumatoid arthritis, HLA-B27 spondyloarthropathy, relapsing polychondritis, systemic lupus erythematosus, rheumatoid arthritis, and necrotizing systemic vasculitis [59,62]. However, at least one-half of the cases are idiopathic. Scleritis usually involves the anterior segment and is defined as diffuse, nodular, or necrotizing [59].
Scleritis causes severe pain, which is usually bilateral, and eye movement or palpation will exacerbate the pain [50]. The eye will appear red as a result of inflamed vessels, and symptoms may include tearing, photophobia, tenderness, and decreased visual acuity [59]. Mucopurulent discharge from the eye occurs in approximately 25% of cases [50].
Urgent referral to an ophthalmologist is recommended, as the diagnosis and treatment are challenging [62]. Initial treatment is a systemic corticosteroid. Nonsteroidal anti-inflammatory drugs may be given to manage the overall inflammatory response, and analgesic agents may be needed for pain [59]. Systemic immunosuppressant drugs may be used if there is no response from initial treatment; a rheumatologist should be consulted in such cases [59].
Inflammation of the optic nerve is most often associated with a demyelinating disease, especially multiple sclerosis. In fact, optic neuritis is considered to be an early indicator of multiple sclerosis, with the disease subsequently developing in more than 50% of individuals [76]. Other possible causes include infection (e.g., meningitis, Lyme disease, syphilis, viral infection), tumor metastasis, and chemicals or medications; the cause is often unknown [59]. Optic neuritis occurs most often in individuals 20 to 40 years of age [59].
The clinical features of optic neuritis have been thought to differ between adults and children. Among adults, optic neuritis usually involves only one eye, whereas children often have involvement of both eyes [59,77]. In addition, most pediatric cases follow a viral illness. Signs and symptoms among both children and adults include eye pain and vision loss (partial or complete) [59,76,78].
On funduscopy, the optic disk usually appears pale in late stages of optic neuritis; the disk appears normal in early stages of the condition. Magnetic resonance imaging (MRI) is a highly sensitive and specific tool for assessing changes in the optic nerves due to inflammation. Additionally, MRI may help predict the future development of multiple sclerosis in patients presenting for the first time with acute optic neuritis [79].
Treatment has typically involved oral or intravenous corticosteroids, but a systematic review demonstrated no conclusive evidence of benefit of corticosteroids with respect to recovery of normal visual acuity, visual field, or contrast sensitivity [80]. The AAO acknowledges this lack of benefit but notes that high-dose intravenous corticosteroids followed by oral corticosteroids may be appropriate in helping to achieve a shorter time to recovery of vision [13]. In addition, high-dose corticosteroids may also delay the onset of subsequent demyelination among patients with multiple sclerosis [13].
Prognosis regarding vision outcomes differs between adults and children, with the prognosis poorer for children [77]. Visual acuity among adults improves to at least 20/40 within a year in most cases (90% to 95%); 15-year follow-up of adults with optic neuritis demonstrated visual acuity of at least 20/20 in 72% of patients [76,81]. Among children, sustained vision impairment has been reported in 22% [82,83].
Blepharitis, or inflammation of the sebaceous glands or lash follicles of eyelids, affects both adults and children. The condition is categorized anatomically by anterior and posterior blepharitis and may be either acute or chronic. Typical diseases such as atopic allergies, seborrheic dermatitis, rosacea, HIV, and some inflammatory ocular conditions predispose individuals to blepharitis [15,84]. Bacterial infection (usually with staphylococci) is the most common cause of acute blepharitis, and infection may be caused by a virus, most notably herpes simplex or varicella zoster) [15,59]. Chronic blepharitis is usually noninfectious.
Signs and symptoms of blepharitis include redness, a gritty feeling, burning sensation, itchiness, loss of eyelashes, and crusting together of the eyelids during sleep [15]. Photophobia and blurred vision may also develop. Slit-lamp examination is used to diagnose the condition, but there are no specific clinical diagnostic tests to confirm blepharitis [15]. Cultures of the eyelid margin may be useful for patients who have recurrent anterior blepharitis with severe inflammation or for patients who do not have a response to therapy.
The treatment plan usually consists of eyelid margin hygiene (application of warm compresses to the eyes to drain the secretory glands and gentle cleaning of the eyelid margin with a wet then dry cotton-tipped applicator) [15]. If the condition does not resolve, nightly application of a topical antibiotic (e.g., bacitracin or erythromycin) to the eyelid margin may be prescribed [15]. Oral tetracycline or doxycycline is indicated for blepharitis that does not respond to topical antibiotics. Chronic blepharitis cannot be permanently cured; successful management is dependent on patient education and compliance [15]. A systematic review of treatment for chronic blepharitis demonstrated that eyelid margin hygiene often provided symptomatic relief in anterior and posterior blepharitis and that topical antibiotics provided some relief of symptoms and eradicated bacteria in cases of anterior blepharitis [84]. Data on the effectiveness of other treatments, such as topical steroids and oral antibiotics, were inconclusive [84].
A hordeolum, commonly known as a stye, is a focal inflammation of the eyelid, usually caused by bacterial infection [62,85]. An external hordeolum involves the eyelash follicles or accessory glands. A chalazion results from noninfectious occlusion of a meibomian gland [59]. It is difficult to distinguish the two conditions, as both cause swelling of the eyelid and pain. An internal hordeolum is rare and results from infection of a meibomian gland [59].
Treatment with warm compresses will help alleviate the symptoms of an external hordeolum, and the condition usually resolves spontaneously within a few days [62]. No treatment has been found to shorten the time to resolution. An external hordeolum does not grow; if the mass becomes enlarged, the patient should be evaluated for a neoplasm [62]. A systematic review of treatments for acute internal hordeolum showed no evidence for or against a variety of treatments that included warm (or hot) compresses, lid scrubs, antibiotics, or glucocorticoids [85].
Dry-eye syndrome—also known as keratoconjunctivitis sicca or tear film dysfunction—is a complex, multifactorial condition defined by clinical evidence of corneal and conjunctival dryness and reduced tear secretion [59]. Risk factors for chronic dry-eye syndrome include aging, female gender, cataract surgery, connective tissue disease, vitamin A deficiency, and androgen deficiency [19]. Prolonged use of antihistamines or estrogen replacement therapy is a reversible risk factor. In the United States, an estimated 3.2 million women and 1.7 million men older than 50 years of age are affected by dry-eye syndrome [5]. Approximately 10% of people with significant dry eye have primary Sjögren syndrome [19].
Most patients report feeling a gritty irritation or the sensation of a foreign body in the eye [19,59]. Other symptoms include itching, burning, and photophobia; blurred vision and sharp, stabbing pain may also occur [59]. Symptoms tend to worsen late in the day and after exposure to adverse environmental conditions [19]. No single test is adequate for diagnosing dry-eye syndrome; diagnosis is based on the characteristic symptoms and the clinical appearance [19]. The Schirmer test may also be useful; with this test, a strip of filter paper is placed at the junction of the middle and lateral third of the lower eyelid, and the wetness on the filter paper is measured [59].
Dry eye is classified as mild, moderate, severe, and severe and/or disabling, according to several factors, including severity and frequency of discomfort, visual symptoms, conjunctival injection and staining, corneal staining, corneal/tear signs, and lid/meibomian gland abnormalities, as well as the fluorescein tear break-up time and the Schirmer score [19]. The AAO recommends treatment according to the severity of the condition. For mild dry eye, recommendations include environmental modifications (avoidance of dry, drafty environments; use of a humidifier; and avoidance of secondary smoke), discontinuation of offending topical or systemic medications, artificial tears, warm compresses and eyelid scrubs, treatment of contributing ocular conditions (such as blepharitis), and correction of eyelid abnormalities [19]. In addition to these treatments, anti-inflammatory agents, such as topical cyclosporine, lifitegrast, and corticosteroids; systemic omega-3 fatty acids supplements and punctal plugs are options for moderate dry eye [86]. Although omega-3 fatty acid products have been used for treatment of dry eye, a prospective randomized 12-month trial of 3,000 mg omega-3 fatty acids failed to show any clinical benefit over placebo [19]. Punctal occlusion, with either silicone or collagen plugs, may provide relief for severe dry eye [87]. However, in its Choosing Wisely campaign, the AAO notes that punctal plugs should not be used until other options (artificial tears, lubricants, and compresses) have been used [88] Additional options for severe dry eye include systemic cholinergic agonists, systemic anti-inflammatory agents, mucolytic agents, autologous serum tears, contact lenses, and tarsorrhaphy [19].
Entropion is a condition in which the eyelid margin (usually the lower lid) turns inward so that the eyelashes rub against the cornea and conjunctiva. The primary symptom is irritation of the ocular surface; other signs and symptoms may include redness, excessive tearing, eye discomfort or pain, photophobia, and stringy white mucoid discharge [59].
Although entropion is usually a result of advanced age, people of all ages can be affected [89]. Other risk factors are anatomical anomalies of the eye, chemical burn, trauma, scarring, and ocular surgeries. In determining the etiology, the clinician must distinguish entropion from other conditions that simulate it; appropriately classify it as congenital or acquired; and then subclassify it as involutional, cicatricial, or acute spastic; involutional entropion is the most common form [89]. Antibiotics, lubricating ointments, chemodenervation of the orbicularis muscle, or everting of the eyelid with adhesive tape may help to alleviate symptoms of involutional entropion, but only temporarily. The only effective treatment is surgery to repair or advance the lower eyelid retractors [89].
Ectropion is the abnormal outward turning of the eyelid margin (usually the lower lid) so that the inner surface is exposed. The primary morbidity is associated with corneal/conjunctival exposure. Other symptoms include excess tearing, redness of the lid and white part of the eye, and ocular irritation [59].
Although ectropion can occur at any age, it is most commonly seen in older adults. Other risk factors include a history of skin cancer removal, increased skin sun sensitivity, lighter iris color and presence of pinguecula, current smoking, hypertension, diabetes, and stroke [89]. Like entropion, treatment depends on the ectropion being properly classified as congenital/acquired and then as involutional, cicatricial, paralytic, or mechanical [89]. Involutional ectropion is the most common form of the condition.
Treatment consists of tear drops and lubricating ointment to provide symptomatic relief and prevent corneal complications; definitive treatment is surgery [89].
Dermatochalasis is a common, normal, physiologic condition clinically seen as the sagging of the upper and (to some degree) lower eyelids. Although it is most often seen in patients older than 65 years of age, the most frequently noted age of onset is between 40 and 50 years of age, and it may occur in younger adults as well [92]. The sagging is due to lax, redundant skin and fat with poor adhesion to the underlying connective tissue and muscle [92]. The major causes of dermatochalasis are, unsurprisingly, gravity and the loss of elastic tissue over many years. Other predisposing factors include heredity, some systemic diseases, trauma, and renal failure [92]. Dermatochalasis often results in ptosis.
In addition to being a cosmetic problem, dermatochalasis can result in a functional loss if the tissue obstructs the superior visual field by hanging over the eyelid margin into the visual axis [92]. Surgery is definitive treatment.
Ptosis (blepharoptosis) is drooping of the upper eyelid and is one of the most common involutional eyelid changes. The drooping is a result of underaction of the eyelid protractors relative to the eyelid retractors, which cause the eyelid to be lower than its normal position. Lowering of the upper eyelid obstructs the superior visual field, which may cause functional vision loss depending on the severity of the condition.
Etiology includes third cranial nerve palsy, age-related dehiscence of the levator muscle, Horner syndrome, or myasthenia gravis [93]. Injection of botulinum toxin (Botox) into the upper third of the face may also cause ptosis [94].
Patients with chronic ptosis can be referred to an ophthalmologist nonemergently, but patients with acute ptosis should be referred immediately, to rule out third cranial nerve palsy [93]. Although treatment is usually surgical, the many types of ptosis and the degree of levator function determine which of a variety of surgical interventions is most appropriate for a given patient.
Eyelid retraction is the shrinking or drawing back of the eyelid from its normal resting position. In the case of the upper lid, the normal resting position is 2 mm below the junction of the sclera and the superior cornea. Positioning is abnormal if a white band of sclera between the lid margin and the upper corneal limbus is exposed. Whereas the normal resting position of the lower lid is at the junction of the sclera and the inferior cornea, the retracted lower lid lies below the inferior corneal margin and is tethered to the orbital margin [95].
Eyelid retraction is symptomatic of many congenital and acquired diseases, and its multifactorial etiology includes neurogenic, myogenic, mechanical, and miscellaneous causes [95]. As the most common cause is thyroid ophthalmopathy, the patient's thyroid status should be evaluated.
Due to its multifactorial etiology, treatment is dependent on a differential diagnosis. Several surgical techniques and materials are available for the repair of eyelid retraction, and the patient should be referred to an ophthalmology specialist [96].
Blepharospasm is a focal dystonia characterized by involuntary chronic intermittent or persistent involuntary eyelid closure. Closure is due to spasmodic contractions of the orbicularis oculi muscles, and the spasms may be unilateral or bilateral [97]. Severity ranges from frequent blinking to forceful eyelid closure. Most cases are idiopathic [59]. Other facial and neck muscles are often involved. Nearly three-quarters of patients report that a stressful event occurred immediately prior to symptoms; photophobia may also precede the development of spasms [98]. The condition occurs more frequently in women than men, and approximately one-third of patients have a family history of blepharospasm [98]. Type A botulinum toxin injections is the treatment of choice, and surgery may be done if the injections fail [97].
The human eye has several inherent protectors, including the bony orbit that covers most of the eye; anatomical structures on the anterior portion of the eye; and accessory structures, such as the eyebrows, eyelids, and eyelashes. According to data from the U.S. Nationwide Emergency Department Sample (NEDS), an estimated 128.9 million emergency department visits occurred in 2010, including more than 1.9 million eye-related visits, indicating that 646.7 emergency department visits per 100,000 population are related to eye injuries [99]. According to the AAO, about 2.4 million eye injuries occur each year, of which 35% are in persons 18 to 45 years of age [5]. The National Institute for Occupational Safety and Health reports that about 2,000 workers sustain job-related eye injuries every day that require medical treatment and estimates that nearly 90% of these injuries can be prevented or severity lessened by using proper eye protection [1]. The prompt and accurate diagnosis and treatment of eye injuries is essential for preserving vision, especially among children, for whom eye injuries are the leading cause of monocular blindness [100]. Minor eye injuries are also a healthcare burden, leading to loss of productivity, decreased quality of life, and increased health costs [6]. The NEDS data also show that the average charge per eye-related emergency department visit was more than $900; the total charge for all eye-related visits was nearly $2 billion [99].
Nearly 50% of individuals with an eye injury are first seen at an emergency department, and a private physician's office is the second most frequent setting for the initial treatment of eye injuries [101]. Thus, healthcare professionals in an office setting should be knowledgeable about the types of eye injuries that can occur, about which injuries can be treated in the primary care setting and which need immediate referral, and about how to effectively treat eye injuries. Primary care clinicians also have an important role in educating patients about eye safety.
Eye injuries cover a spectrum of severity, from corneal abrasions caused by minor trauma (e.g., vigorous rubbing or fingernails) to high-velocity penetrating injuries and injuries that cause sudden loss of vision. Corneal abrasions are the most common eye injury treated in the emergency department, followed by foreign body in the external eye. Other injuries include laceration of skin of the eyelid and around the eye, unspecified bruise of the eye, bruise of orbital tissues, and other specified open wound of the ocular adnexa) [101]. The rates of eye injury, as well as the causes of injury, vary according to patient demographics. Understanding the populations at greatest risk of injury and the types of injuries incurred in distinct populations can help clinicians and other healthcare professionals to better target education and prevention strategies.
According to NEDS data, men account for 54.2% of eye-related emergency department visits, compared with women [101]. The mean age of eye injury is 32 years of age. An estimated 67% of all reported eye injuries among those 19 to 64 years of age, constituting more than the proportion of the U.S. population represented by this group [101]. Eye injury also appears to be seasonal, with most injuries occurring in spring and summer months when construction, gardening, landscaping, and sports and recreation activities are more prevalent [101].
Several causes of eye injury have been identified as occurring most frequently, and the causes vary according to age and the site of injury. Many consumer products cause eye injuries in the home, sports and recreation also cause a large proportion of eye injuries, and occupational hazards cause eye injuries in the workplace. The home is the most common setting for eye injuries among adults and children, accounting for approximately 69% to 78% of all eye injuries [100,103,105,106]. Among children and adolescents, close to 50% of eye injuries occur in sports and recreational activities [107].
Eye injuries in the home are caused by a wide variety of items, including chemicals, tools, outdoor equipment, paint, debris generated by power tools and lawn equipment, automobile mechanic materials, and toys. The most common cause overall is chemicals, with cleaning agents, personal care products, and automotive chemicals being the most common agents[101]. Across all ages, eye injuries related to consumer products are more common among male individuals than female individuals[100,101,103,105,108,109]. Among men, injuries attributable to hardware and lawn equipment have been found to be more common, whereas among women, more injuries are attributable to chemicals, housewares, storage and organization, and bed and bath items[110]. The cause of injury varies among age groups (Table 10)[100,103,105,108,109,112].
CONSUMER-PRODUCT-RELATED CAUSES OF EYE INJURIES ACCORDING TO AGE
| Age Group | Most Common Causes |
|---|---|
| 0–4 years | Chemical product contact, non-chemical product contact, toys |
| 5–9 years | Non-chemical product contact, non-powder firearmsa, chemical contact, desk/school supplies |
| 10–24 years | Non-chemical product contact, non-powder firearmsa, chemical contact |
| 25–64 years | Cutting and construction tools |
| 65 years and older | Chemicals, cutting and construction tools, furniture, gardening |
| aNon-powder firearms include air, airsoft, paintball, pellet, and BB guns. | |
The most common consumer products causing eye injury among children 0 to 12 years of age are toys, pens and pencils, household cleaners and chemicals, adhesives, golf equipment, baseballs and basketballs, cigarettes/cigars/pipes/tobacco, furniture, and fireworks [105,109]. The rate of injury related to toys is proportionately higher among children 2 to 4 years of age and the rate of injury related to desk and school supplies is proportionately higher among children 5 to 8 years of age [105,109].
Chemicals are one of the most common causes of injury among individuals 10 to 24 years of age [103,108]. Aerosol cans have emerged as another cause of eye injury occurring at a high rate among children and adolescents. In one study, individuals younger than 18 years of age accounted for more than half of patients seen in the emergency department for eye injuries related to aerosol cans (5,927 of 10,765 patients) [111]. Spray paint was the most common product involved, and self-inflicted spray to the eye was the most common mechanism of injury [111]. Non-powder firearms (e.g., air, airsoft, paintball, pellet, BB guns) are an increasingly common cause of eye injury among children, adolescents, and young adults, having risen more than 50% since the 1990s, and are associated with a high risk of hospital admission, severe damage, and permanent vision impairment [105,112]. Nearly every eye injury with non-powder firearms (98%) occurs in individuals who are not wearing protective eyewear, highlighting the need for clinicians to emphasize the importance of preventing this type of injury [112].
Among individuals 25 to 64 years of age, cutting and construction tools are the most common causes of eye injury, and chemicals are the leading cause of injury among individuals 65 years of age and older; cutting and construction tools, furniture, and gardening are other common causes in this latter age group [106].An additional hazard to eyes for both adults and children is fireworks. Injuries related to fireworks have increased over the years, from 8,500 in 1998 to 11,500 in 2022; and the eye is the third leading affected part of the body (15%) [115]. Approximately 73% of these injuries occur during a one-month period from mid-June to mid-July [115]. Nearly one-half of the eye injuries were contusions or lacerations, with burns accounting for one-fifth of the injuries [115]. As with other causes of eye injuries, fireworks-related injuries occur much more frequently among male individuals (65%) [115]. Individuals 25 to 44 years of age account for 36% of the estimated injuries; 28% occur among children younger than 15 years of age [115].
Healthcare professionals should heighten awareness of the need for eye safety in the home. The AAO and the American Society of Ocular Trauma recommend that each home should have at least one pair of American National Standards Institute (ANSI)-approved protective eyewear, to be worn during activities related to a risk of eye injury [118]. Clinicians should also emphasize eye safety for children, specifically noting the following:
Teach children not to run while holding items that can cause injury (e.g., utensils, pencils, scissors).
Keep detergents, cleaning supplies, bath and hygiene items, and tools in locked cabinets or out of reach.
Keep clothes hangers in the closet.
Keep younger children away from areas where power tools, lawn equipment, or automobile mechanical items are being used.
Discourage the use of fireworks.
Be a role model for children by wearing appropriate eye protection when performing tasks with tools or equipment.
As stated, among children and adolescents, eye injuries are most often related to sports activities[100,107,108]. In a joint policy statement, the AAP and the AAO classified sports as eye safe, low-risk, moderate-risk, and high-risk in terms of the potential for eye injury (Table 11)[119]. Most sports are considered to be moderate risk when eye protection is not worn. Among athletes 5 to 24 years of age, baseball, basketball, and golf are the most common causes of eye injuries[105,119].
CLASSIFICATION OF SPORTS ACCORDING TO RISK OF EYE INJURY
| Level of Risk | Sport | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Eye safe |
| ||||||||||||
| Low risk |
| ||||||||||||
| Moderate risk |
| ||||||||||||
| High risk |
|
The most common mechanism of eye injury in sports is blunt trauma, which is defined as an injury that occurs when a blunt object that is larger than the orbital opening exerts force on the floor of the orbit or the medial wall, with resultant fractures of the thin bones [120]. Blunt trauma can cause several different types of injury, such as:
Orbital blowout fracture
Contusion of the orbit and lid
Rupture of the globe
Injury to the iris
Traumatic iritis
Subconjunctival hemorrhage
Hyphema (blood in the anterior chamber)
Retinal or vitreous hemorrhage
Choroidal rupture
Retinal tear or detachment
Other types of eye injuries occur less frequently in sports. Penetrating injuries are less common than blunt trauma and range from mild (abrasions) to severe (serious lacerations). Such injuries can be caused by broken glass (including eyeglasses), a finger, or a fishing hook. Participants in snow skiing and water sports may be at some risk for radiation injuries because of exposure to ultraviolet light [50,120].
It has been shown that protective eyewear for athletes substantially reduces the number of eye injuries. While some sports mandate use of protective eyewear, rules and laws vary among sports and regions, so it is important for healthcare professionals (as well as coaches and athletic trainers) to know local mandates and ensure that athletes, especially those who are young, wear eye protection appropriate for the sport[122,123]. The proper eye protection for sports is made of polycarbonate, which is resistant to high impacts. According to the AAO/AAP statement, the types of eyewear that are satisfactory for eye-injury risk sports include [119]:
Safety sports eyewear that conforms to the requirements of the American Society for Testing and Materials (ASTM) standard F803 for selected sports (i.e., racket sports, baseball fielders, basketball, women's lacrosse, and field hockey)
Sports eyewear that is attached to a helmet or for sports in which ASTM standard F803 eyewear is inadequate
There are standard specifications for youth baseball batters and base runners, paintball, skiing, ice hockey, football, and men's lacrosse.
Eye injuries in the workplace range from minor, such as computer-related eyestrain and dry-eye syndrome, to severe injuries caused by flying objects. The overall lifetime prevalence of occupational eye injuries has been reported to be 4.4% [124]. In 2020, eye injuries accounted for 1.6% of the nearly 1.18 million occupational injuries involving days off from work [51,126]. The rate of eye injury varies according to many factors, including gender, age, socioeconomic status, and work setting. The rate of injury is more than three times higher among men than women and is highest (112.8 cases per 10,000 full-time workers) among individuals 45 to 54 years of age [51,125]. Individuals with less than a high school education and men in lower income brackets are more likely to have an eye injury [101]. These injuries are a substantial health and economic burden, with an estimated total cost of approximately $300 million annually [118].
Eye injuries occur in virtually every work setting. In 2020, the rates were highest among heavy and tractor-trailer truck drivers; laborers and freight, stock, and material movers; and nursing assistants [51]. Most workplace-related eye injuries are caused by flying objects (e.g., bits of metal or glass), tools, particles, and chemicals, resulting in corneal abrasion or burn. However, the type of injury ranges widely across work settings [118]. Approximately 64% to 90% of individuals who use a computer or digital screen devices have eye-related symptoms, particularly eyestrain, tired eyes, dry eye, blurred vision, and headache; this complex of symptoms has been termed computer vision syndrome or digital eye strain [127]. In addition, symptoms of dry eye have been associated with indoor office environments, and it has been postulated that the cause is dehydration of the corneal tear film as a result of low relative humidity and high room temperature, as well as decreased blinking during near-task activities [127]. Ergonomic adjustments, such as modification of lighting and glare, frequent breaks, eyeglasses specifically made for computer screen use, and lubricating eye drops can be helpful in reducing symptoms [127].
Allergic conjunctivitis can occur in food handlers and agriculture workers who are exposed to spices, fruits, and vegetables and in outdoor workers exposed to grass, ragweed, and rhus [51]. The absence of symptoms when the individual is not at work confirms the diagnosis. Outdoor workers are at increased risk for radiation injury and age-related macular degeneration, and sunglasses that provide protection from ultraviolet rays should be worn [51].
The Occupational Safety and Health Administration (OSHA) standards mandate that employers must provide suitable eye (and face) protection to all employees to protect against chemical, environmental, radiologic hazards, or mechanical irritants [131]. In addition, all employees must be trained to work safely. Safety spectacles (which may include side shields) or safety goggles should be worn when there is potential for impact injuries, as from flying/falling objects or heat hazards (e.g., high temperatures, splashes of molten metal, hot sparks). Safety goggles should fit tightly around the eyes, forming a protective seal to prevent objects from entering under or around the goggles [131]. Face shields should be used in conjunction with safety goggles when there is a high potential for exposure to impact injuries, heat hazards, or chemicals or when working with lasers and welding equipment [131]. Lenses for goggles and face shields may be specially filtered. Eyewashes should be readily available in occupational environments with risk for injuries from chemical exposure [131].
Approximately 90% of occupational eye injuries are preventable with protective eyewear [118]. As such, clinicians should help to reinforce the importance of eye safety in the workplace.
One of the most important aspects of evaluating eye injuries is a knowledge of which injuries should be referred and how urgent referral should be. In some cases, primary care clinicians will be able to treat the injury, as in corneal abrasions, removal of foreign bodies, and minor lacerations of the conjunctiva or eyelid; in other cases, immediate referral to an ophthalmologist is essential. All patients presenting with an eye injury in the primary care setting, however, should be evaluated to determine the nature of the trauma and the potential injury.
In many cases, the exact injury is not known, and the clinician should seek specific details regarding the injury from the patient or parent. If the patient (or parent) does not speak the same language as the clinician, a professional interpreter should be consulted to ensure accurate communication. A systematic review of the literature has shown that the use of professional interpreters provides better clinical care than the use of so-called ad hoc interpreters, with professional interpreters improving the quality of care for patients with limited English language skills to a level equal to that for patients with no language barriers [132]. Use of professional interpreters has been associated with improvements in communication (errors and comprehension), utilization, clinical outcomes, and satisfaction with care [132]. Despite the clear benefit of professional interpreters, a survey of more than 900 pediatricians showed that professional interpreters were the third most commonly used resource (40% of respondents); bilingual family members were used most often (70%) [133]. However, individuals with limited English language skills have indicated a preference for professional interpreters rather than family members [134].
The patient with an eye injury may be anxious or hysterical about the injury, especially if loss of vision is involved. It is important for clinicians to be calm and compassionate and to provide information to help the patient and family understand the eye injury. Clinicians should be realistic in estimating the severity of the injury and describing the treatment options and prognosis. Again, a professional interpreter should be used if the clinician does not speak the same language as the patient and/or family.
In addition to obtaining information about the cause of the injury, the primary care clinician should carry out an eye examination consisting of several components. First, visual acuity should be evaluated, both to determine loss of vision related to the injury and to document a baseline vision for comparison over time [8]. Evaluation of confrontation visual fields should also be done, as a defect suggests injury to the retina, optic nerve, or central nervous system. The anterior chamber should be examined with a penlight to check for foreign bodies, hyphema, abrasions, or lacerations. The pupils should be assessed, and anisocoria may indicate damage to the retina or optic nerve (if the affected pupil constricts consensually but not to direct light).
It is essential to administer analgesic agents both to provide adequate pain relief for the patient and to facilitate examination of the eye. Antiemetics may also be needed for injuries that are associated with nausea and vomiting [135]. The clinician should determine whether the patient was wearing contact lenses or corrective glasses at the time of the injury. If still present, contact lenses should be removed before swelling makes removal difficult [51].
Corneal Abrasions and Removal of Foreign Bodies
A corneal abrasion is a defect in the corneal epithelial surface that is usually caused by trauma. A foreign body may or may not be in the eye at the time of examination. In diagnosing a corneal abrasion, the history is important in determining whether there has been recent trauma to the eye, bearing in mind that the cause can be as minor as aggressive eye rubbing or accidental scratching with a fingernail, or can be related to poor contact lens technique or overuse. If no clear history of trauma is present, infectious keratitis should be suspected [136]. Symptoms of corneal abrasion include sensation of a foreign body in the eye, photophobia, excessive tearing, and pain [137].
A topical anesthetic is helpful for pain-free examination of the eye and/or removal of a foreign body [136]. Visual acuity should be measured, and the cornea should be examined with use of a penlight and loupe or biomicroscope magnification [136]. The patient should be referred immediately to an ophthalmologist if any of the following is present [137]:
Vision loss of more than 20/40
Corneal infiltrate or ulcer
Hypopyon or hyphema
Evidence of penetrating eye injury (irregular, dilated, or fixed pupil or extension of ocular contents)
The diagnosis of abrasion is confirmed by applying fluorescein to the eye and examining the eye with use of cobalt-blue filtered light; the abrasion will appear green [136,137].
Examination of the eye should also be directed toward determining if there is a foreign body, even if the patient does not recall a foreign body entering the eye. The lower lid should be pulled down gently to allow for more complete examination of the eye, and the upper eyelid can be inverted to search for a subtarsal foreign body, but only if a penetrating injury is not suspected [136,137]. Vertical linear corneal abrasion on fluorescein staining may indicate a hidden foreign body, and the upper eyelid and lower fornix should be evaluated carefully [137].
Irrigation with saline can remove minor foreign bodies (e.g., soot particles). If the foreign body is larger, a moistened cotton-tipped applicator may be used to remove the foreign body [137]. A more deeply embedded (but nonperforating) foreign body can be removed with use of an eye spud or a 25-gauge needle, but primary care clinicians should attempt removal of such foreign bodies only if they are comfortable performing the procedure and if these instruments are available; otherwise, referral to an eye specialist should be made [137].
A metallic foreign body may be more difficult to remove because it can become "rusted" on the cornea. Rust residue may be present on the eye after removal of a metallic foreign body. This so-called rust ring can increase inflammation, prevent healing, and cause scarring. Patients should be referred to an ophthalmologist for removal of a rust ring within the next few days [137].
The treatment of corneal abrasions (with or without a foreign body) has simplified over the past several years, with adequate healing achieved with minimal treatment. Most corneal abrasions heal without complications, and relieving pain is the primary concern. Oral analgesics and topical nonsteroidal anti-inflammatory drugs (NSAIDs) may be used, but oral analgesics are more cost-effective [137]. One 2017 systematic review of five topical NSAIDs (i.e., 0.1% indomethacin, 0.03% flurbiprofen, 0.5% ketorolac, 1% indomethacin, 0.1% diclofenac) found no strong evidence to support their use [138]. However, other studies indicate that diclofenac 0.1% is effective [137]. Topical NSAIDs should be used for no more than one or two days [137]. Topical mydriatics and cycloplegics were once used to relieve pain, but they are no longer recommended, as evidence has shown no benefit in cases of uncomplicated corneal abrasion [139,140]. Evidence for the use of topical antibiotics is lacking, but they are often prescribed to prevent infection [137,141,142]. If a topical antibiotic is used, an ointment is preferred to drops because it is more lubricating and soothing; erythromycin 0.5% ophthalmic ointment and sulfacetamide 10% ophthalmic ointment are two recommended options [137]. A systematic review found that patching does not reduce pain; in addition, the use of a patch results in a loss of binocular vision [143]. A tetanus booster is not needed for patients with a superficial corneal abrasion when there is no evidence of perforation, infection, or devitalized tissue [144].
For patients who wear contact lenses, a topical fluoroquinolone or aminoglycoside should be prescribed because of their antipseudomonal activity [137]. In addition, the contact lenses should be discarded, and new ones should not be worn until the abrasion has healed. All patients should be followed up in 24 hours; if symptoms have improved, no further follow-up is necessary. Patients should be referred to an ophthalmologist if symptoms do not improve, if the abrasion does not heal within three to four days, if the size of the abrasion increases, if a corneal infiltrate or ulcer develops, or if vision worsens [137].
Other Minor Injuries
Other minor injuries that are encountered in the primary care setting are subconjunctival hemorrhage, small conjunctival lacerations, and minor eyelid lacerations. Subconjunctival hemorrhage is typically a focal, bright red lesion in the underlying white of the sclera. It is usually related to minor trauma or to a contact lens-induced injury [145]. In evaluating the eye, a globe rupture should be ruled out; subconjunctival hemorrhage in the area surrounding the cornea is a sign of globe rupture [135]. In older patients, spontaneous subconjunctival hemorrhage is often associated with hypertension [145]. The condition usually resolves on its own in a few days to a few weeks, and patients should be reassured that the condition is not serious. In infants and children with subconjunctival hemorrhage, nonaccidental trauma should be considered [146].
Primary care clinicians can treat small conjunctival lacerations (less than 1 cm) that do not involve the underlying sclera with a topical antibiotic and close follow-up [147]. Patients with larger lacerations should be referred to an eye specialist, as sutures may be necessary. Examination with a slit-lamp microscope can help to distinguish a conjunctival laceration from a globe rupture [135].
Eyelid lacerations may be sutured using 6-0 interrupted sutures, with nonabsorbable sutures used for adults and absorbable sutures used for children; if the laceration is small and the tissue edges are opposed, suturing is not necessary [148]. Patients with complicated eyelid lacerations should be referred to an ophthalmologist for repair [148].
Five eye injuries require urgent referral and immediate treatment to preserve vision [50]:
Chemical injuries
Mechanical globe injury
Retinal detachment
Central retinal artery occlusion
Angle-closure glaucoma
Chemical Injuries
Exposure of an eye to chemicals (liquid, powder, or gas) represents a true ocular emergency, and it is the only injury for which treatment should not be delayed until visual acuity is tested. The eye should be treated immediately on the basis of the history alone, if necessary[50,135]. The severity of an injury caused by exposure to a chemical can range from reversible edema to stromal scarring, calcific band keratopathy, and fibrosis of the iris or ciliary body. Severity will depend on the pH concentration of the chemical, the nature of the chemical, the volume of direct exposure, and the duration of exposure[50,135,149]. An alkali (pH of more than 10) can penetrate the cornea more readily than an acid (pH of less than 4) and thus presents the greater danger; alkali burns are also more common[147,149]. Determining the type of chemical is important, as the mechanism of injury differs between acidic and alkali agents. Examples of alkalis are household cleaning fluids, fertilizer, pesticides, and sparklers and firecrackers[135]. Hydrofluoric acid is the most common cause of acid burns[50,135]. When possible, the patient should be asked to bring in the container or a sample of the chemical. Identification of the chemical can also be obtained from Material Safety Data Sheets, which are required for some occupational sites[51].
Signs and symptoms related to a chemical injury include pain or burning sensation, red eye, photophobia, blurred vision, and sensation of a foreign body in the eye[135,147,149]. Treatment begins with topical anesthetics to the eye and immediate copious irrigation of the eye with normal saline or lactated Ringer's solution (tap water may be used if necessary) for at least 15 minutes (at least 2 liters of fluid)[51,147,149]. The pH of the ocular surface should be checked periodically during irrigation. Once the physiologic pH has been restored (pH of 7.0–7.5), the clinician should refer the patient immediately to an ophthalmologist or an emergency department[147]. The next steps are a thorough medical history and a complete eye exam. The exam includes assessment of visual acuity, pupil symmetry, and pupillary light response; evaluation of extraocular muscles; slit lamp examination with fluorescein staining and cobalt blue light; measurement of intraocular pressure; and dilated funduscopic examination[149]. Some findings on examination include swelling and burns of the eyelid, a cloudy cornea, a corneal epithelial defect, conjunctival injection, or a white eye (indicating conjunctival ischemia[147]. The extent of injury can be defined with the Roper-Hall classification system (Table 12)[150].
Roper-Hall Classification of Ocular Burns
| Grade | Clinical Findings | Prognosis | |
|---|---|---|---|
| Cornea | Limbal Ischemia | ||
| I | No opacity or mild epithelial damage | None | Good |
| II | Haziness, but iris details are visible | Less than one-third of limbus | Good |
| III | Haziness that obscures iris details | One-third to one-half of limbus | Guarded |
| IV | Opaque; no view of iris or pupil | More than one-half of limbus | Poor |
The severity of a chemical injury dictates treatment. Topical antibiotics and artificial tears are usually sufficient for grade I and II burns; a topical steroid is added for grade II and IV burns [147]. There is no evidence to support the routine use of ascorbate in the treatment of alkali burns [151]. Patients should be referred to an ophthalmologist if visual acuity is abnormal, if intraocular pressure remains high, or if epithelial defects are present [149].
Mechanical Globe Injury
Blunt trauma or a high-velocity injury can cause a full-thickness rupture or laceration through the cornea and/or sclera. Because of the high risk of infection and vision loss associated with a mechanical globe injury, it should be considered in every case of eye trauma [51,135]. Blunt trauma to the eye, as from a thrown ball, motor vehicle accident, or assault, can cause a globe rupture, whereas penetrating injury, as from a knife or a small high-velocity projectile, can cause a globe laceration [147]. The most common signs/symptoms are eye pain, redness, tearing, and decreased vision, but a globe injury should be suspected after blunt trauma or a penetrating injury even if these symptoms are absent [147].
Examination of the anterior segment with a slit lamp is preferred for diagnosing a globe injury; penlight examination may also be done. Indicators of a globe injury are subconjunctival hemorrhage, irregular pupil, and iris prolapse through a corneal or scleral wound [147,152]. If a diagnosis of globe injury is not clear, the Seidel test should be done. This test is performed by applying a moistened fluorescein dye strip to the area of possible injury and using a slit lamp and cobalt blue light to examine the site. The test is positive (presence of a globe injury) if the fluorescein is diluted by aqueous flow from the eye; if darker, diluted fluorescein dye streams from the injury site, or if bright-green concentrated dye surrounds the leak site [147,153]. Dilated pupil exam and tonometry are not recommended. Computed tomography (CT) (axial and coronal views) of the head and orbits is the preferred imaging modality and can be used to detect an intraocular foreign object or a fracture of the orbital wall [135,147,152]. If there is a protruding foreign body, the clinician should not attempt to remove it if a globe rupture is suspected [135,152].
If a globe injury is confirmed (or suspected), a metal shield should be placed over the eye and the patient should be referred immediately to an ophthalmologist [147,152]. The patient should be told to limit coughing or straining, and an antiemetic may be given to prevent the Valsalva maneuver. A tetanus booster should be given if tetanus immunization is not up to date [147,152].
The treatment of a globe injury consists of continued analgesic agents to maintain adequate pain relief and antibiotics to prevent endophthalmitis [135,147,152]. The preferred antibiotics are the fluoroquinolones levofloxacin and moxifloxacin, as they target the most common causes of post-traumatic endophthalmitis and have excellent vitreous penetration [147]. Another option is intravenous vancomycin or ceftazidime. If organic material (such as hay, grain, or leaves) was involved, an antibiotic to treat infection with Gram-negative bacteria or fungi should be chosen [135]. The risk of endophthalmitis is substantially reduced if surgical repair and/or removal of a foreign body is performed with 24 hours of the injury, further emphasizing the need for immediate referral to an ophthalmologist [147].
Retinal Detachment
Retinal detachment occurs when the neurosensory layer of the retina separates from the underlying choroid and retinal pigment epithelium. Retinal detachment can occur with a retinal tear (rhegmatogenous detachment) or without a retinal tear (nonrhegmatogenous). Rhegmatogenous detachment is most often associated with myopia, cataract surgery, and ocular trauma [59]. Nonrhegmatogenous detachment is caused by vitreoretinal traction or transudation of fluid into the subretinal space; detachment caused by traction is often associated with diabetic retinopathy, sickle cell disease, and advanced retinopathy of prematurity, whereas detachment caused by transudation of fluid is most often the result of inflammation, malignant effusions, or severe acute hypertension [59].
Retinal detachment is uncommon but can cause rapid degeneration of photoreceptors, and loss of vision can be permanent if diagnosis and treatment are not prompt. Signs and symptoms of retinal detachment include unilateral flashing lights, an increasing number of floaters, and blurred vision [59,147]. The patient may also note that it appears as if a dark curtain covers part of the visual field [154]. There is usually no pain. Involvement of the macula is associated with a substantial loss of central visual acuity [59,147,154]. Dilated funduscopic examination is used to diagnose retinal detachment; if the retina cannot be visualized because of vitreous hemorrhage, cataract, or corneal scar, B-scan ultrasonography should be done to identify a tear [59,147,154].
Patients who are suspected of having a retinal detachment should be referred urgently to an ophthalmologist [59,147]. If left untreated, a retinal detachment can result in permanent and severe vision loss; treatment with surgery is successful in approximately 90% to 95% of cases [59,147]. Patients should be followed up closely, as retinal detachment in the contralateral eye develops in about 25% of patients; in addition, monitoring for recurrent retinal detachment is necessary [147,154].
Central Retinal Artery Occlusion
Occlusion of the central retinal artery is usually caused by an embolism from atherosclerotic plaque, endocarditis, fat emboli, atrial myxoma, or thrombosis of a retinal artery [59]. Occlusion can also be caused by temporal arteritis. Immediate treatment of acute central retinal artery occlusion is needed, as complete loss of vision may develop as soon as 90 minutes after injury [50,135]. Older age, diabetes, atherosclerosis, hypercholesterolemia, hypertension, and hypercoagulable states are risk factors for central retinal artery occlusion [135,155]. Approximately 5% to 10% of cases are associated with giant cell arteritis [50,155]. Other diseases or disorders associated with central retinal artery occlusion include systemic lupus erythematosus, acute lymphoblastic leukemia, primary central nervous system vasculitis, iron deficiency anemia, and Wegener granulomatosis [50]. It may also result from blunt trauma to the eye or as a complication after coronary angiography, laryngectomy, or spinal surgery.
Central retinal artery occlusion is usually painless, with sudden blindness or visual field defect. In some cases, the patient may note a history of transient, painless loss of vision in one eye (amaurosis fugax) [135,155]. Transient, bilateral vision loss is usually caused by conditions other than central retinal artery occlusion, such as migraine auras, heart failure, and hypertensive emergencies [135].
During pupillary examination, the pupil may react slowly or poorly to light, but the pupil will constrict quickly when the contralateral eye is illuminated [59]. Findings on funduscopic examination that indicate central artery occlusion include a pale, opaque fundus and a cherry-red spot at the fovea; however, this spot may take hours to develop [59,155].
Immediate treatment should be given if the occlusion occurred within the past 24 hours. Because of the need for immediate restoration of blood flow to the retina in order to preserve vision, interventions should be implemented before the patient can be seen by an eye specialist. Digital massage of the closed eyelid should be done immediately (for 15 minutes), as it may dislodge an embolus, allowing it to move to a smaller branch of the artery [59]. In addition, the intraocular pressure should be lowered with either a topical hypotensive drug, such as timolol 0.5%, or acetazolamide (oral or intravenous) [59,155]. However, treatment rarely improves visual acuity, and treatment given 72 hours or more after occlusion is unlikely to improve vision [59]. Systemic corticosteroids should be given to patients who have central retinal artery occlusion caused by temporal arteritis.
Acute Angle-Closure Glaucoma
Acute angle-closure glaucoma is associated with a closed anterior chamber angle and may be caused by many mechanisms. With this type of glaucoma, the iris is pushed (or pulled) up into the angle, blocking the drainage of aqueous humor, which in turn increases the intraocular pressure [59]. Angle-closure glaucoma accounts for only 10% of glaucoma cases, but without treatment, angle-closure glaucoma can cause vision loss within several hours to days [34,59]. Thus, a suspected attack of acute angle-closure glaucoma requires emergent referral to an ophthalmologist.
A shallow anterior chamber and hyperopia are the ocular factors that most predispose individuals to primary angle-closure glaucoma; other factors include steep corneal curvature, thick crystalline lens, and a short axial length [34]. Demographic risk factors include advancing age, strong family history/heredity, and female gender [34]. The highest rates of primary angle-closure glaucoma have been found in Asian populations, especially Taiwanese; Inuit populations; and African or African-derived populations [34,59].
The signs and symptoms of acute angle-closure glaucoma are classic. The most common symptoms include [34,59]:
Ocular and facial pain (mild to severe)
Eye redness
Blurred vision
Colored halos around lights
Photophobia
Nausea and vomiting
Headache
Patients usually have a mid-dilated pupil, which is fixed and reacts sluggishly to light. The conjunctiva may have signs of hyperemia, and corneal swelling may be present [34,59].
Treatment consists of decreasing intraocular pressure, reversing the angle closure, and preventing damage to the optic nerve [34]. Initial treatment usually involves medication to reduce intraocular pressure and reduce pain. The AAO recommended options include topical beta-adrenergic antagonists, topical alpha2-adrenergic agonists, topical or systemic carbonic anhydrase inhibitors, topical mitotics, and systemic hyperosmotic agents [34]. The preferred surgical treatment is laser peripheral iridotomy (LDI) because of its favorable risk-benefit ratio [34,59]. Because the chance of having an acute attack in the contralateral eye is 80%, LDI should be done on both eyes [59].
Many eye conditions can develop in children, and a discussion of all of them is beyond the scope of this course. Provided here is an overview of the two most common eye conditions in children: strabismus and amblyopia. Although strabismus can occur in adults, it is either a recurrence of childhood strabismus or a complication of a neurologic event or in conjunction with a systemic disease (e.g., diabetes). Early detection and treatment of these conditions is important, as the rate of successful outcomes decreases with increasing age [157,163].
In strabismus, the eyes are not properly aligned with each other, with one eye in either constant or intermittent horizontal deviation (esotropia or exotropia) or vertical deviation (hypertropia or hypotropia). Because both eyes are not directed to the same point, no image of a fixated object is formed on the fovea of the strabismic eye. The exact cause of strabismus is often unknown, and it is thought to be the result of several factors. Strabismus is the most common cause of amblyopia[26].
Horizontal deviations are much more common than vertical deviations[157]. Horizontal strabismus causes the eyes either to turn out (exotropia) or cross in (esotropia). Exotropia usually begins and remains intermittent, whereas esotropia usually begins intermittently but becomes constant over a few weeks[156].
Strabismus usually occurs before 6 years of age, and the peak age at the time of onset differs slightly for esotropia and exotropia. Most cases of esotropia occur in children 2 to 3 years of age, while exotropia has a usual onset in children 1 to 4 years of age[156]. Infantile esotropia occurs in a neurologically normal child between the ages of 3 and 6 months[157].
In MEPEDS, which included children 6 to 72 months of age in Los Angeles County, the prevalence of strabismus was highest for Asian children (3.6%) and lowest for Hispanic/Latino children (2.4%) (Table 13)[48,49]. Different rates were found in the Vision in Preschoolers (VIP) Study, which included children 3 to 5 years of age in five geographically diverse locations. In this older population, the prevalence of strabismus was highest among non-Hispanic White (4.6%) and lowest among Asian children (1.0%)[46].
PREVALENCE OF STRABISMUS AND AMBLYOPIA IN THE VISION IN PRESCHOOLERS (VIP) STUDY AND MULTI-ETHNIC PEDIATRIC EYE DISEASE STUDY (MEPEDS)a
| Race/Ethnicity | Strabismus | Amblyopia | ||
|---|---|---|---|---|
| VIP | MEPEDS | VIP | MEPEDS | |
| Non-Hispanic White | 4.6% | 3.2% | 5.4% | 1.8% |
| Black | 2.5% | 2.5% | 3.3% | 1.5% |
| Hispanic/Latino | 2.5% | 2.4% | 5.0% | 2.6% |
| Asian | 1.0% | 3.6% | 3.0% | 1.8% |
| American Indian | 2.9% | — | 3.5% | — |
| aThe VIP Study included children who were 3 to 5 years of age in Head Start in five different geographic locations. The MEPEDS included children who were 6 to 72 months of age in Los Angeles County. | ||||
There are few risk factors for strabismus. Children are at risk for the condition if there is a family history of strabismus, if there is neurodevelopment impairment, if there was maternal exposure to smoking or alcohol during pregnancy, if the child was born prematurely, and if the child's birth weight was low [157]. A more recent study has indicated that very low birth weight may be more of a risk factor than gestational age [158].
Often, a parent's observation of deviation of one eye will prompt him or her to bring the child to the clinician for an eye examination. Children in whom strabismus is noted on screening eye examination in the primary care setting should be referred to an ophthalmologist for further evaluation and treatment [37].
The goals of treatment in young children with strabismus are good vision, correct alignment of the eyes, and the development of stereopsis (i.e., binocular depth perception) [156]. The AAO notes that the initial treatment for children with esotropia or exotropia is eyeglasses to correct any significant refractive error but emphasizes that restoration of good alignment does not ensure elimination of amblyopia; surgery may be indicated for some children [157]. The results of a Cochrane review (three studies) indicated that the true effect of correction of hyperopia on strabismus is uncertain [159]. Improved alignment may take several weeks. For children with intermittent exotropia, part-time patching therapy (two to six hours daily) may provide benefit [157]. If eyeglasses and treatment of amblyopia fail to align the eyes in children with esotropia, surgical realignment should be done. The AAO states that surgery is not needed for young children with intermittent exotropia and good fusional control [157].
Follow-up is essential after treatment for either esotropia or exotropia. If the eyes are well aligned and amblyopia is not present, children treated for esotropia should have follow-up visits every four to six months to monitor for amblyopia, loss of binocular vision, and recurrence of strabismus. Follow-up visits are recommended every 6 to 12 months for children with good fusional control of intermittent exotropia and no amblyopia [157].
When developing a treatment plan, the eye specialist will consider the goals and expectations of the parents and the child, when possible, and will emphasize the importance of compliance to treatment in order to achieve goals [157]. The primary care clinician should take advantage of the child's well-care visits to assess whether the child and parents are complying with the recommended treatment course as well as with follow-up examinations.
Prompt diagnosis and treatment of strabismus in children are crucial, as the condition can delay developmental milestones, adversely affect academic achievement, and have a lower psychosocial quality of life [157]. The AAPOS and AAO recommend that primary care clinicians screen for strabismus in children 3 to 5 years of age at all well-child visits; the USPSTF recommends screening for strabismus just once during these ages [26,157]. The USPSTF notes that the cover-uncover test and the Hirschberg light reflex text are feasible in primary care for assessing ocular alignment/strabismus [26].
Amblyopia (or "lazy eye") is a condition in which the vision in one eye is reduced because the eye and the brain are not working together properly [1]. Although there is no apparent structural abnormality, best-corrected visual acuity is less than 20/20 because the brain is favoring the other eye. Amblyopia is classified as strabismic (usually related to esotropia), refractive (a result of untreated unilateral or bilateral refractive errors), or visual deprivation (caused by a congenital or early-onset cataract, corneal opacities, intraocular inflammation, or vitreous hemorrhage) [163].
In MEPEDS, the prevalence of amblyopia was highest for Hispanic/Latino children (2.6%) and lowest for Black children (1.5%) [48,49]. Among the older population in the VIP Study, the prevalence of amblyopia was highest among non-Hispanic White (5.4%) and lowest among Asian children (3.0%) [46].
The presence of strabismus increases the risk of amblyopia, with amblyopia occurring in approximately 50% of children with strabismus [160,163]. Significant refractive errors are risk factors for unilateral amblyopia, and bilateral astigmatism and bilateral hyperopia are risk factors for bilateral amblyopia [160,163]. Other identified risk factors include prematurity, low birth weight, developmental delay, and a first-degree relative with amblyopia [163]. As with strabismus, maternal smoking and use of drugs or alcohol have also been associated with an increased risk for amblyopia [163].
Amblyopia is suggested if a child has difficulty performing tasks that require binocular depth perception. Due to the high rate of amblyopia in association with strabismus, all children who have esotropia or exotropia should be evaluated closely for amblyopia. A cooperative child is necessary for reliable visual acuity testing, and the optimal tests vary according to age. The AAO notes that the optotypes on an eye chart can significantly affect the visual acuity score and recommends that optotypes be clear, standardized, have similar characteristics, and be free of cultural bias [163]. LEA Symbols (Good-Lite; Elgin, IL) are useful for young children, as is the HOTV chart [163]. Children who are too young to name the symbols on these charts may be able to match them to the symbols on a hand-held card. LEA numbers and Sloan letters are preferred for older children [163]. Children in whom amblyopia is noted on screening eye examination in the primary care setting should be referred to an ophthalmologist for further evaluation and treatment [163].
The goal of treatment is equal visual acuity in the two eyes [163]. Treatment of amblyopia is highly successful in approximately 75% of children who are younger than 7 years of age when treatment begins [161]. Although a younger age at the start of treatment is associated with better final visual acuity, treatment should be attempted in older children who have not been previously treated, as nearly half of such children have sustained improvement in visual acuity [161,162].
The treatment plan varies according to the type of amblyopia. Treatment of refractive error alone can improve visual acuity in children who have untreated strabismic or anisometropic amblyopia or bilateral refractive amblyopia [157,163]. Occlusion of the better eye to stimulate the amblyopic eye has been the cornerstone of treatment for amblyopia, and a systematic review demonstrated that occlusion in combination with refractive correction was more effective than refractive correction alone [164]. Occlusion is achieved with either patching or administration of 1% atropine drops (one drop daily); both have been found to be effective for children younger than 7 years of age, with one study showing that both treatment approaches led to good visual acuity up to at least 15 years of age in children with moderate amblyopia who were treated before the age of 7 [165,166]. Six hours of patching daily has been shown to be as effective as all-day patching (all but one waking hour), and initial treatment with two hours of daily patching has been as effective in improving visual acuity as six hours for children with moderate amblyopia [163]. A drawback of patching is compliance, with one study demonstrating a mean compliance of 44% [167]. The selection of a treatment approach should be discussed with the parents and the child, if feasible, to determine the best option.
Visual acuity (in both eyes) and treatment compliance should be assessed during follow-up, with the first follow-up visit within two to three months [163]. If visual acuity is unchanged, treatment intensity or modality should be considered.
Vision impairment (i.e., uncorrectable low vision and blindness) affects more than 4.2 million individuals 40 years of age or older in the United States [1]. Vision impairment carries a high cost, both in terms of quality of life and economic burden; the financial cost of low vision in the United States is approximately $139 billion and is expected to increase to $373 billion by 2050 [6,14].
The primary causes of vision impairment are age-related. Because of this, the rate of vision impairment is expected to increase by 50% or more by 2050 as the U.S. population continues to age[155]. Overall, the primary causes of low vision and blindness are cataract, diabetic retinopathy, glaucoma, and age-related macular degeneration[14].
There are substantial differences in the prevalence rates of these conditions among ethnic/racial populations. According to data from the 2005–2008 NHANES, the prevalence of cataract surgery ranged from 13.5% for the non-Hispanic Black population to 16.4% for the Mexican American population and 18.4% for the non-Hispanic White population[168]. The prevalence of age-related macular degeneration is also lowest in the non-Hispanic Black population (3.1%) than in the Mexican American and non-Hispanic White populations (9.8% and 8.3%, respectively)[168]. In contrast, glaucoma is more prevalent among non-Hispanic Black individuals (11.5%) than among non-Hispanic White and Mexican American individuals (6.9 and 6.5%, respectively)[168].
Any level of impaired vision has a major adverse impact on quality of life, affecting safety, independence, and well-being. Vision impairment increases the risk of falling and has been associated with higher rates of difficulty with mobility, social isolation, depression, admission to nursing homes, and mortality[169,172]. In addition, vision impairment is associated with difficulty reading, which has far-reaching effects, including the potential for medication errors because of difficulty reading prescription labels[170].
The AAO recommends that people with impaired vision be referred for multidisciplinary vision rehabilitation[172]. This vision rehabilitation should consider the following factors: reading, activities of daily living, safety, increasing participation in the community, and psychosocial well-being[172]. Addressing these factors has been shown to be effective, although there is insufficient evidence on the effect of different types of low-vision aids on reading performance[171].
Occupational therapists certified in low-vision rehabilitation emerged in the 1990s as an important component in the management of individuals with vision impairment, and their services are reimbursable by Medicare[172]. These therapists can provide focused, individualized strategies and training with devices to help improve daily living activities, such as [172]:
Optical devices
Technology assistance
Computer adaptations (e.g., magnification, audio-screen readers, text-to-speech)
Cell phone accessibility options (e.g., magnification, audio readers, using virtual assistants on smartphones, identification of useful apps)
Other non-optical devices
Audio devices (for watches, liquid level indicators)
Large-format or large-print items (e.g., bank checks, telephones)
Alternative strategies (e.g., signature templates, needle threaders)
Tactile, audio, or braille labeling
Alternative strategies and training
Modification of lighting and contrast to increase visibility
Training with new strategies for completing desired daily activities, including personal care, home management, financial management, meal preparation, and shopping
Cataracts—opacifications of the eye's crystalline lens—are the leading cause of blindness among adults worldwide and are the second leading cause in North America; they are also the overall leading cause of moderate and severe vision impairment[14]. Cataracts affect more than 24 million people older than 40 years of age in the United States, and by 75 years of age, half of all people have cataracts[5,14]. Slow, painless vision loss occurs over years in the eye with a cataract. Due to the cataract's opacity, the patient's pupil may appear gray. The different types of adult-onset cataracts are nuclear, cortical, and subcapsular (posterior and anterior), and mixed. Of the three types, nuclear cataracts develop most frequently in older adults[29]. With nuclear cataracts, central opacification and coloration interfere with visual function. Nuclear cataracts affect distance vision more often than near vision[29]. Cortical cataracts are characterized by light-blocking spokes extending from the outer rim of the lens to its center (Image 1), and patients most often report glare. Posterior subcapsular cataracts, granular opacities in the posterior pole of the cortex that develop under the capsule, cause visual impairment in bright light and glare. With posterior subcapsular cataracts, near vision is affected more often than distance vision[29].
Advanced age is the most common cause of cataracts. As a person ages, the thickness and weight of the lens increase and nuclear sclerosis (hardening and compression of the nucleus) develops. Several risk factors have been evaluated, and overall, the most common factors are diabetes, smoking, use of inhaled or oral corticosteroids, and exposure to ultraviolet-B light [29]. Other potential risk factors vary according to the type of cataract. For example, a family history of cataracts and hypertension have been associated with an increased risk for nuclear and cortical cataracts; obesity has been associated with an increased risk for cortical and posterior subcapsular cataracts, and ocular trauma has been associated with posterior subcapsular cataracts [29].
The most common early symptoms associated with cataracts include loss of contrast, glare, the need for more light to see well, and decreased color perception. Blurring often occurs as the cataract progresses [59]. Primary care clinicians should refer patients suspected of having a cataract to an ophthalmologist for evaluation. According to the AAO, the ophthalmic evaluation for cataracts should include a complete history; measurement of visual acuity with current correction; examination of external aspects of the eye; examination of ocular alignment and motility; assessment of pupillary function; measurement of intraocular pressure; slit-lamp biomicroscopy of the anterior segment; and dilated examination of the lens, macular, peripheral retina, optic nerve, and vitreous [29].
The only effective treatment for cataracts is surgery; clinicians should emphasize to patients that no nutritional supplements (such as antioxidants), herbal medications, or pharmacologic agents have shown benefit in the treatment of cataracts [20,29]. Surgery involves replacement of the affected lens with an intraocular lens (artificial lens). In most cataract surgeries in the United States, the lens is removed by phacoemulsification, which has resulted in significantly better visual acuity and fewer postoperative complications [29]. A Cochrane review indicated that, compared with manual small incision cataract surgery, phacoemulsification was associated with better uncorrected visual acuity up to three months after surgery, but similar best-corrected visual acuity [174]. No data were available on long-term outcomes. The cost of manual small incision cataract surgery is lower than the cost of phacoemulsification, leading the authors to conclude that manual small incision cataract surgery may be a reasonable option for some patients. The AAO recommends joint decision-making regarding cataract surgery. Criteria typically considered during decision-making include the patient's visual acuity, the likelihood that surgery will improve vision, the effect of vision loss on the patient's lifestyle, and the patient's amenability to surgery [29].
When surgery is planned, the ophthalmologist is responsible for confirming the diagnosis and designing the treatment plan. The primary care clinician can play an important role in alleviating patient concerns preoperatively and monitoring improvement postoperatively. Cataract surgery leads to improved visual acuity in approximately 85% to 90% of patients [29]. Overall, cataract surgery is very cost-effective, with benefits in quality-adjusted life years [29].
Smoking cessation and protection from ultraviolet-B radiation have been shown to reduce the risk of cataracts, and the AAO notes that these are reasonable precautions to recommend to patients [29]. However, several randomized, long-term, clinical studies have demonstrated that using supplements such as multivitamins, vitamin C and E, beta carotene, or herbal medications does not prevent the incidence or progression of cataracts [20,29]. Prevention and appropriate treatment of type 2 diabetes can help to reduce the risk of cataract formation, as can avoidance of long-term use of oral or inhaled corticosteroids [29].
Primary open-angle glaucoma is characterized by progressive optic neuropathy, usually caused by increased intraocular pressure [173]. In some cases, however, intraocular pressure is normal. The disease slowly reduces peripheral vision and excavates the optic disc. If not properly treated, vision in the glaucomatous eye can be lost over time. The condition is associated with aging.
Glaucoma is the fourth leading cause of blindness and moderate and severe vision impairment in the United States and worldwide [14]. An estimated 2.7 million people older than 40 years of age in the United States have glaucoma, and the number is expected to reach 7.3 million by the year 2050. The prevalence of glaucoma is highest among people 70 to 79 years of age (32%), women (50%), and Hispanics (50%) [7,14]. It has been estimated that an additional 2.4 million people in the United States have undetected (and untreated) glaucoma [14]. The prevalence of glaucoma increases with age and occurs twice as frequently in the non-Hispanic Black population (the highest prevalence) than in the non-Hispanic White population [176]. The rate of undiagnosed glaucoma is higher among minority populations compared with the non-Hispanic White population [14].
Elevated intraocular pressure is the most important—and only modifiable—risk factor for primary open-angle glaucoma [173]. Other strong risk factors include older age, family history of glaucoma, Black race, Latino/Hispanic ethnicity, thinner central cornea, myopia, and type 2 diabetes (and fasting glucose levels) [173,179]. Some studies have suggested that there may be gender differences in the prevalence and pathophysiology of glaucoma, with hormone therapy, oral contraceptive use, and menopausal status being associated with glaucoma [180]. A meta-analysis published in 2014 demonstrated that hypertension was associated with increased intraocular pressure, with a strong association between hypertension and primary open-angle glaucoma [181].
More than half of people with glaucoma have no symptoms; about 24% note ocular surface disease symptoms and 25% report visual problems [182]. Some people with glaucoma note vision difficulty while reading or driving [59]. The presence of symptoms has been associated with increased severity of glaucoma [182].
The AAO recommends prompt referral to an ophthalmologist for an abnormal intraocular pressure or a family history of glaucoma, especially in patients of Black or Hispanic race/ethnicity [40]. A diagnosis of glaucoma requires a complete history and an ophthalmic evaluation that includes measuring visual acuity and intraocular pressure and performing gonioscopy to rule out other causes of elevated intraocular pressure [173].
There are three types of computer-based optic nerve imaging devices that have been shown to be highly effective diagnostic tools, especially when coupled with functional tests: confocal scanning laser ophthalmoscopy, optical coherence tomography, and scanning laser polarimetry [173]. These devices have been found to be similar in their ability to distinguish between glaucoma and controls [173].
The goals of treating glaucoma are to control intraocular pressure, stabilize the status of the optic nerve/retinal nerve fiber layer, and stabilize visual fields [173]. Treatment options include medication, laser therapy, or incisional glaucoma surgery, all of which lower intraocular pressure. The choice of treatment depends on many factors, such as the effects of treatment, quality of life, and life expectancy, and the patient should be involved in the decision-making process [173].
Medical treatment is typically used initially, and prostaglandin analogs and beta-blockers are the eye drops most often prescribed; prostaglandin analogs have been the most effective at lowering intraocular pressure [173]. A comparative effectiveness study indicated that medical and surgical treatments for primary open-angle glaucoma reduce intraocular pressure and reduce the risk of damage to the optic nerve in the short- to medium-term setting [184]. However, the comparative effectiveness of different treatments is not clear. In addition, it is not clear which treatments are best in preventing visual disability and improving patient-reported outcomes [184]. Although marijuana has been shown to lower intraocular pressure, the American Glaucoma Society does not recommend the use of marijuana in any form to treat glaucoma because of the drug's side effects, short duration of action, and lack of evidence that it alters the course of glaucoma [185]. No data support the use of antioxidants or herbal supplements as treatment [20].
Primary care clinicians should ask their patients with glaucoma about adherence to medical treatment, as a high level of adherence is necessary for adequate treatment.
There is no agreed-on standard of diagnosis for glaucoma, and no single screening method can identify a significant number of patients with glaucoma [186]. The AAO notes that population-based screening is not practical or cost-effective and therefore does not recommend it [173]. Similarly, the USPSTF concluded that the evidence is insufficient to assess the balance of benefit and harms of screening for glaucoma [175]. Additionally, there is limited evidence that screening can reduce either intermediate or final health outcomes [186]. The AAO does note that screening may be useful for populations at high risk for glaucoma, including older adults, people with a family history of glaucoma, and people of Black or Latino/Hispanic race/ethnicity [173].
Several lifestyle risk factors have been explored as potential contributors to glaucoma, including consumption of dietary fat, cholesterol, and caffeine, but none of these has been found to be associated with the risk of glaucoma [187]. As a preventive measure, antioxidants or herbal supplements have shown no effect [20]. Early detection, through regular and complete eye exams, is the key to protecting vision from damage caused by glaucoma.
As noted, an eye examination should be completed every two to four years for patients younger than 40 years of age who have risk factors for glaucoma, with more frequent examinations as the patient ages [173].
Age-related macular degeneration, the leading cause of blindness in North America, is a degenerative disease that causes loss of central visual acuity by its effects on the Bruch's membrane, choroid, outer neural retina, and retinal pigment epithelium [188]. Age-related macular degeneration is classified into two forms: neovascular (otherwise known as wet or exudative) and non-neovascular (otherwise known as dry or nonexudative). The slowly progressing, atrophic non-neovascular form is more common than the wet form, affecting approximately 80% of people with age-related macular degeneration; however, the neovascular form is responsible for almost 90% of the severe vision loss due to age-related macular degeneration [188]. Age-related macular degeneration is classified as early, intermediate, and advanced, based on the size and extent of drusen (small yellow deposits that form under the macula) [188].
The prevalence of age-related macular degeneration varies according to race/ethnicity. Studies have shown that age-related macular degeneration is most prevalent in White individuals (5.3%), intermediate in Hispanic and Asian individuals (4.5% and 3.3%, respectively), and lowest in Black individuals (1.6%) [190]. Nearly 2.1 million persons in the United States 50 years of age and older have advanced age-related macular degeneration, the stage that can lead to severe vision impairment [5].
As with cataracts and glaucoma, increasing age is the primary risk factor for macular degeneration. A variety of other risk factors have been evaluated, but smoking and ethnicity are the only two that have been shown consistently to be risk factors for age-related macular degeneration[188]. The risk of age-related macular degeneration doubles with smoking, and the risk increases as the number of pack-years smoked increases[188]. Hypertension, other cardiovascular disease, low levels of antioxidants, and dietary fat have also been associated with advanced age-related macular degeneration, but the findings have been conflicting[188]. A meta-analysis identified several risk factors with varying levels of association with age-related macular degeneration (Table 14)[193].
RISK FACTORS ASSOCIATED WITH AGE-RELATED MACULAR DEGENERATION
| Level of Association | Risk Factor | ||||||
|---|---|---|---|---|---|---|---|
| Strong and consistent |
| ||||||
| Moderate and consistent |
| ||||||
| Weaker and inconsistent |
|
The non-neovascular form of age-related macular degeneration is associated with a slow and painless loss of central visual acuity and central blind spots. With the neovascular form, the first sign is usually visual distortion in one eye, and vision loss is rapid[59]. Any signs of age-related macular degeneration should result in prompt referral to a specialist[188]. Although therapies can slow the visual deterioration associated with age-related macular degeneration, available treatments cannot restore vision lost to the condition. Drusen and/or pigmentary changes in the macula associated with early non-neovascular age-related macular degeneration can be identified using funduscopy (Image 2) [194]. In the neovascular form of macular degeneration, the earliest sign of choroidal neovascularization is the appearance of semi-translucent punctate dots in retroillumination (Image 3)[194]. Fluorescein angiography and optical coherence tomography are imaging modalities used to detect new or recurrent neovascular disease activity and guide therapy[188].
A combination of antioxidant vitamins (vitamin C, beta carotene, vitamin E), zinc oxide, and cupric oxide can slow the progression of intermediate age-related macular degeneration to the advanced form, but there is no evidence to support use of this combination for early age-related macular degeneration or prophylaxis in family members [188]. Evidence suggests that lutein supplementation is associated with a mild improvement in visual acuity and a significant improvement in macular pigment optical density [195].
The first-line treatment of neovascular macular degeneration is an antivascular endothelial growth factor (VEGF) agent, and three have been approved for use by the FDA: pegaptanib (Macugen), ranibizumab (Lucentis), and aflibercept (Eylea) [188]. All three drugs are administered by intravitreal injection. A biosimilar to ranibizumab (ranibizumab-nuna, Byooviz) was approved for the treatment of neovascular macular degeneration in 2021 [54]. Another anti-VEGF agent, bevacizumab (Avastin), has also been shown to be effective for neovascular age-related macular degeneration, but its use is off-label in this setting [188]. A Cochrane review and meta-analysis demonstrated that pegaptanib, ranibizumab, and bevacizumab are effective in maintaining visual acuity, with ranibizumab and bevacizumab also effective in improving visual acuity [196]. Aflibercept has been shown to be as effective as ranibizumab, with the need for fewer injections [197]. Thermal laser photocoagulation surgery and photodynamic therapy with verteporfin are approved treatments for subfoveal lesions [188]. Conbercept is a novel anti-VEGF approved for the treatment of neovascular macular degeneration in China. Its chemical structure differs slightly from the agents available in the United States, but the drug appears to have the same safety and efficacy profile, with reduced dosing frequency. The drug's developer is in discussions with the FDA about the possibility of introducing the drug into phase 3 testing in the United States [198,199].
Routine, comprehensive eye exams are critical for the early detection of age-related macular degeneration [188]. Smoking cessation reduces the risk of age-related macular degeneration, and clinicians should encourage their patients who are smokers to stop smoking and should provide resources on cessation options [188]. Early cessation is optimal; the risk of age-related macular degeneration in former smokers who have not smoked for more than 20 years is comparable to the risk in nonsmokers [188].
Several systemic diseases have ocular manifestations, and tight management of the disease can help to minimize the adverse effect on the eye. The ocular effects of some systemic diseases also warrant more frequent eye examinations to help prevent damage through early detection of eye disease. In addition, many medications and herbal supplements have been shown to cause eye-related symptoms ranging from minor to severe. Understanding the complete health and medication history of a patient is necessary when evaluating the eye either routinely or in response to symptoms. Knowledge of systemic diseases and medications that affect eye health is important for planning appropriate screening and follow-up to minimize the potential for serious eye disorders or loss of vision.
Diabetic retinopathy is a progressive vascular complication of diabetes mellitus and the leading cause of moderate-to-severe vision loss in working adults 20 to 65 years of age[212]. Globally, the prevalence of diabetes has been increasing, and with it the burden of diabetic retinopathy. In 2021, approximately 540 million people were living with diabetes; by 2045, this will rise to 783 million[211]. A meta-analysis of published population studies found that from 1990 to 2010, visual impairment caused by diabetic retinopathy increased by 64% and blindness by 27%[209]. By 2010, approximately 3.7 million cases of vision impairment and more than 800,000 cases of blindness worldwide were caused by diabetic retinopathy. In the United States and other countries with nationwide screening programs, advances in retinal imaging technologies and new therapies have improved the evaluation, treatment, and visual outcomes of patients with diabetic retinopathy[212].
Some degree of diabetic retinopathy develops in nearly all people with long-standing type 1 or type 2 diabetes. In general, the threat to vision arises from increased retinal vascular permeability, complications of retinal neovascularization, or vascular insufficiency in the central retina[212]. The underlying mechanisms for this retinal vasculopathy are unclear. Intravitreal glucocorticoids have been shown to reduce diabetic macular edema, suggesting that inflammation may be an important mechanism. Thinning of the inner retinal layers and abnormal neural function are known to precede clinical evidence of diabetic vascular lesions, indicating that diabetic neuropathy may be involved in pathogenesis of retinopathy[212]. Once retinopathy is established, the driver of disease progression is retinal ischemia and tissue hypoxia, potent inducers of vascular endothelial growth factor (VGEF). Elevated retinal concentrations of VEGF stimulate angiogenesis and promote vascular permeability, precursors to retinal neovascularization and macular edema, respectively[212].
Diabetic retinopathy is classified according to clinical stage of disease. The earliest stage, nonproliferative diabetic retinopathy (also known as background retinopathy), is characterized by retinal vascular abnormalities such as microaneurysms, intraretinal hemorrhages, and cotton-wool spots (white spots on the retinal surface, often having an irregular edge)[213]. Nonproliferative retinopathy is categorized as mild (microaneurysms only), moderate (more than microaneurysms but less than signs of severe retinopathy), and severe (severe intraretinal hemorrhages and microaneurysms in each of the four quadrants, definite venous beading in two or more quadrants, or moderate intraretinal microvascular abnormalities in one or more quadrants)[213]. An increase in vascular permeability can result in macular edema, which is characterized by retinal thickening and hard exudates (lipid deposits). Macular edema is the most common cause of vision loss associated with nonproliferative diabetic retinopathy[213]. As retinopathy progresses, retinal vessels gradually close, leading to impaired perfusion and retinal ischemia. Proliferative retinopathy, which poses the most threat to vision, is characterized by neovascularization on the retina and posterior surface of the vitreous[213]. The new blood vessels have weak walls, do not form in orderly branching, and bleed easily into the retina or vitreous cavity[201]. With advanced proliferative retinopathy, excess blood in the vitreous causes the retina to detach.
The prevalence of diabetic retinopathy in the United States has decreased since the early 2000s, from 40.3% reported in 2004 to 28.5% in the 2005–2008 NHANES; only a slight decline (26.4% in 2021) has been recorded in the decade between NHANES studies[43,176,177]. The prevalence is higher among non-Hispanic Black individuals (approximately 9%) compared with Mexican American and non-Hispanic White individuals (approximately 7% and 3%, respectively)[168,176]. Visual symptoms are rare with nonproliferative diabetic retinopathy; blurred vision and black spots or flashing lights in the field of vision are associated with proliferative retinopathy[59].
The primary risk factor for developing diabetic retinopathy is chronicity of disease coupled with sustained unsatisfactory control of metabolic derangements associated with diabetes. In short, the risk of retinopathy increases with duration of diabetes and poor glycemic control. In most patients, retinopathy develops 10 to 15 years after diabetes has been diagnosed[212,213].
Studies of individuals with type 1 diabetes have shown that the prevalence of diabetic retinopathy increases from 25% at 5 years to 60% at 10 years to 80% at 25 years[202,213]. The prevalence of retinopathy also increases with duration of type 2 diabetes, with rates that increase to 84% in patients who take insulin and 53% in patients who do not take insulin at 19 years[213]. These findings demonstrate a need to follow up patients more closely as the number of years with diabetes increases.
Studies have provided strong evidence that decreasing hemoglobin A1clevels in patients who have either type 1 or type 2 diabetes substantially reduces the development of new diabetic retinopathy or the progression of existing retinopathy[203,204]. Among people with type 2 diabetes, intensive glycemia treatment (target hemoglobin A1c level of less than 6.0%) was associated with a lower rate of progression of retinopathy than standard treatment (target hemoglobin A1c level of 7.0–7.9%)[203].
Hyperlipidemia has been associated with decreased visual acuity among patients with diabetic retinopathy. However, the benefit of treatment to decrease lipid levels has not been demonstrated[205].
The AAO recommends that eye examinations should begin five years after the diagnosis of type 1 diabetes and at the time of diagnosis of type 2 diabetes; examinations should be yearly thereafter[213]. Pregnant women with pre-existing type 1 or type 2 diabetes should have examinations prior to conception, if possible, or early in the first trimester of pregnancy, as pregnancy can potentiate rapid progression of retinopathy[213]. If retinopathy is severe, follow-up examination should be done every 1 to 3 months; otherwise, follow-up examination should be done every 3 to 12 months[213]. The AOA also provides a guideline for the frequency of eye examinations, recommending yearly examinations for people with mild nonproliferative diabetic neuropathy (every four to six months if macular edema is also present); examinations every six to eight months for moderate nonproliferative retinopathy (every four to six months if macular edema is also present); examinations every three to four months for severe retinopathy (every two to three months if macular edema is also present); and examinations every two to three months for proliferative retinopathy[206]. All eye examinations in people with diabetic retinopathy should be carried out by an eye specialist.
Focal or panretinal photocoagulation is the recommended treatment for proliferative diabetic retinopathy[213,206,212]. While panretinal (scatter) laser photocoagulation is the preferred modality for high-risk proliferative retinopathy, it is sometimes used in patients with non-high risk disease and for cases of severe nonproliferative retinopathy[206]. With this technique, neovascularization is targeted, with delivery of laser in a scatter pattern, throughout the peripheral fundus, sparing the macula (Image 4).
Although laser photocoagulation is effective for managing non-center involved macular edema, intravitreal anti-VEGF agents are standard of care for treatment of center-involved diabetic macular edema and accompanying vision loss [213,212]. Before the use of anti-VEGF agents, intravitreal sustained-release glucocorticoid therapy was shown to reduce retinal thickening and improve vision; however, treatment with glucocorticoids increases the risk of cataracts and other complications such as intraocular pressure elevation and glaucoma [212]. A systematic review demonstrated high-quality evidence of benefit for the use of intravitreal anti-VEGF agents for diabetic macular edema compared with grid laser photocoagulation, with improvement in visual acuity and patient-reported outcomes [214]. Most experts now recommend intravitreal anti-VEGF agents as standard of care for diabetic macular edema, and two such drugs—ranibizumab and aflibercept—are approved by the U.S. Food and Drug Administration [210,212]. Small studies have documented some efficacy of bevacizumab for treatment of diabetic macular edema; however, this is an off-label use [215,216]. There is no evidence to support the use of antioxidants or herbal medications for the prevention or treatment of diabetic retinopathy [20].
When baseline visual acuity in an eye with diabetic macular edema is 20/25 or better, initial management with observation is a reasonable strategy rather than immediate anti-VEGF therapy or laser treatment [212]. This strategy requires that patients are followed closely and anti-VEGF therapy is initiated promptly if vision worsens. In a randomized, controlled trial of initial management approaches, similar rates of vision loss at two years were observed with aflibercept (16%), laser treatment (17%), and clinical observation (19%) [208].
Despite the availability of treatment to prevent the progression of diabetic retinopathy, a large percentage of patients with diabetes have vision loss. A primary reason for this is poor patient compliance with eye examinations, with studies showing that 38% to 50% of people with diabetes do not adhere to the guidelines for eye examinations [213]. Lack of primary care physicians' referral for eye examinations is a contributing factor to low patient compliance, and a Canadian study demonstrated that printed educational messages for primary care physicians did not increase examination uptake [217]. Other strategies to enhance awareness of the need for close follow-up eye examinations for patients with diabetes are needed.
Over time, sustained hypertension is a risk factor for complications that threaten vision, including retinal vascular occlusion, retinal microaneurysm, and ischemic optic neuropathy [53,207]. In addition, hypertension exacerbates progression of diabetic retinopathy and the risk of developing glaucoma and age-related macular degeneration. The most common ocular presentation of hypertension is hypertensive retinopathy.
The eye is the one organ in the body where vascular changes of systemic hypertension can be observed and monitored. On visualization of the retina, an early sign of hypertension is generalized narrowing of arteriolar vessels due to vasospasm and increased vascular tone [207]. Intimal thickening of the vessel wall and hyaline degeneration are late changes of chronic hypertension. The combination of cotton-wool spots and optic disc edema is indicative of severe systemic hypertension [201]. Cotton-wool spots represent microinfarcts of the retinal nerve fiber layer, and optic disc edema is caused by leakage of optic disc arterioles [201]. Vision is usually not affected. When moderate hypertension (a systolic pressure higher than 160 mm Hg and a diastolic pressure higher than 100 mm Hg) is chronic (five years or more), the walls of retinal arterioles become stiff and the lumen narrows [201]. Arteriovenous nicking and an increase in light reflection from the vessel wall seen on ophthalmoscopic examination are signs of chronic hypertension in individuals younger than 70 years of age; these signs are not reliable indicators of chronic hypertension in older individuals, as these abnormalities may be caused by advanced age alone [59].
According to earlier population-based epidemiologic studies representing more than 26,000 participants, various signs of retinopathy occur in 2% to 15% of individuals older than 40 years of age, regardless of blood pressure status [218,219]. The prevalence of retinopathy has also been higher in the Black population than in the White population, which may be a reflection of higher blood pressure levels in the Black population [218]. No differences in the prevalence of hypertensive retinopathy have been found between men and women [218].
There are no specific treatments to reverse the retinal changes associated with systemic hypertension, but studies show that timely intervention and control of blood pressure can halt progression and reduce risk of ocular complications [53,218]. The evidence is insufficient to recommend routine ophthalmoscopic consultation for all patients with hypertension [218]. Referral to an eye specialist may be useful for patients who have borderline or inconsistent hypertension. Due to the association between retinopathy and stroke, it may be beneficial to closely monitor individuals with these signs for cerebrovascular risk and implement interventions to reduce the risk [218]. In addition, routine examination of the retina may be of particular value for risk stratification of Black individuals [218].
Several autoimmune disorders have ocular manifestations, primarily involving the cornea (Table 15) [201]. The conjunctiva, uvea, sclera, retina, and surrounding structures may also be affected. Individuals with such disorders as rheumatoid arthritis, systemic lupus erythematosus, seronegative spondyloarthropathy, multiple sclerosis, giant cell arteritis, or Graves disease should have routine ophthalmologic examination to prevent ocular impairment. The use of NSAIDs, corticosteroids, and immunosuppressive agents to treat the systemic effects of these diseases is usually sufficient in managing the ocular symptoms; but additional specific treatment may sometimes be necessary[201]. To ensure appropriate management of ocular symptoms, primary care physicians should make timely referrals to eye specialists.
MOST COMMON OCULAR DISORDERS
| Autoimmune Disorder | Most Common Ocular Manifestation | Subjective Symptoms | |||
|---|---|---|---|---|---|
| Rheumatoid arthritis |
| Burning, pain, blurred vision, sensation of foreign body or grittiness, redness, photophobia, ocular tenderness | |||
| Systemic lupus erythematosus (SLE) |
| Burning; pain; blurred vision; sensation of foreign body or grittiness; redness; sudden painless loss of vision | |||
| Multiple sclerosis |
| Pain with eye movement, photophobia, vision loss | |||
| Giant cell arteritis | Ischemic optic neuropathy | Sudden, painless loss of vision; constitutional symptoms | |||
| Graves disease | Proptosis, lid retraction, optic nerve compression | Gritty feeling, blurred vision, diplopia, photophobia, dry eye |
Several ocular conditions have been associated with HIV infection. Retinopathy is the most common ocular condition in individuals with HIV or acquired immune deficiency syndrome (AIDS), and cytomegalovirus (CMV) retinitis is the most common HIV-associated retinopathy [222]. Since the advent of highly active antiretroviral therapy (HAART), the prevalence of CMV retinitis has decreased substantially, but the infection remains the leading cause of poor vision and a predictor of mortality [222,226]. CMV retinitis may be asymptomatic or be associated with floaters, scotomata, or peripheral visual field defects [228].
Non-CMV opportunistic infections of the eye are uncommon but may be associated with high rates of vision loss or mortality [74]. HIV retinal microangiopathy is another common HIV-related ocular disease and was significantly associated with mortality in one study [226]. Visual dysfunction (abnormal visual field, reduced contrast sensitivity, and altered color vision) is common among individuals with AIDS who do not have retinitis, with nearly 40% of such individuals having an abnormal visual field [75]. Retinal vasculopathy may contribute to vision dysfunction [90].
HIV-related CMV retinitis is treated with anti-CMV agents, such as valganciclovir (oral), ganciclovir (intravenous), ganciclovir followed by valganciclovir, foscarnet (intravenous), or cidofovir (intravenous) [228]. In a guideline on the prevention and treatment of HIV-related opportunistic infections, the Centers for Disease Control and Prevention, the National Institutes of Health, and the HIV Medicine Association of the Infectious Diseases Society of America recommend that the choice of treatment should be individualized, based on the location and severity of the lesions and the level of immunosuppression [228]. Ophthalmologic examination is recommended every three months for individuals with CMV retinitis in whom maintenance therapy has been discontinued; examinations may be done annually after immune reconstitution [228].
The eye is particularly susceptible to medication-related adverse events because of its relatively small mass and rich blood supply[17]. As such, ocular toxicity is an adverse event associated with a wide variety of systemic medications as well as herbal supplements[16,17,18,90]. The National Registry of Drug-Induced Ocular Side Effects was established in 1976 as a database for the collection of reports specifically documenting ocular side effects of systemic and ocular medications. (The registry can be accessed by members of the AAO.)
The medications with ocular side effects are typically those that are taken over a long period of time, as for a chronic condition such as arthritis or cardiovascular disease. Ocular side effects span a range of severity from sensitivity to light and color changes to retinopathy and optic neuritis (Table 16). The effects are usually time- and dose-dependent, and they often resolve or stabilize when use of the drug is discontinued[17]. However, side effects may be irreversible, as with the visual deficits associated with chloroquine and hydroxychloroquine, antimalarial drugs that are often used to treat rheumatoid arthritis and lupus erythematosus[17,102]. In 2016, the AAO published recommendations for screening and management of ocular side effects of chloroquine and hydroxychloroquine. The recommendations include using a lower than routine dose for individuals of short stature, a baseline eye examination, and annual screening beginning no more than five years after beginning use[104]. In 2020, as a result of differences between the AAO and American College of Rheumatology recommendations, a joint guideline was released to address the importance of an interdisciplinary approach to determining the appropriate drug regimen based on risk factors and screening for drug-induced retinopathy. The ACR and AAO are joined in this statement by the American Academy of Dermatology (AAD) and Rheumatologic Dermatology Society (RDS)[113]. Close follow-up with periodic eye examinations may be warranted for other drugs used to manage chronic conditions.
MEDICATIONS WITH OCULAR SIDE EFFECTS
| Drug or Supplement | Side Effects | Treatment and Indication for Referral | Recommended Follow-Up |
|---|---|---|---|
| Amiodarone | Blurred vision, halos, corneal changes, optic neuropathy, loss of eyelashes or eyebrows, photosensitivity | Refer promptly to ophthalmologist for vision changes and discontinue drug if optic neuropathy is suspected | Baseline eye examination and every 6 to 12 months (or more frequently depending on findings) |
| Anticholinergics | Blurry near vision | Tell patients about potential difficulty focusing and possible need for glasses | — |
| Angle-closure glaucoma (rare) | Refer emergently to ophthalmologist for acute vision loss, eye redness or pain, or cloudy cornea | — | |
| Antihistamines (e.g., loratadine, cetirizine) | Dry-eye syndrome, keratoconjunctivitis, photosensitivity | Consider discontinuation of medication or use of non-drying antihistamine | — |
| Antimalarial drugs (e.g., hydroxychloroquine, chloroquine) | Loss of color vision, visual field, visual acuity, corneal deposits, bull's eye appearance of macula | — | Baseline comprehensive eye examination and annual eye examination beginning 5 years after the start of treatment |
| Corticosteroids (oral and inhaled) | Cataracts, glare, decreased visual acuity, halos, glaucoma, loss of peripheral vision | Maintain close follow-up | Measurement of intraocular pressure every 6 months (by eye specialist) |
| COX-2 inhibitors | Conjunctivitis, blurred vision | Educate patients regarding possible side effects | — |
| Digoxin | Yellowish-orange or snowy, flickering vision | Maintain dose in therapeutic range | — |
| Erectile dysfunction drugs | Changes in color perception, blurred vision, sensitivity to light | Consider decreasing dose or discontinuing medication | — |
| Ethambutol | Color vision changes, visual field defects, optic neuritis (usually bilateral) | Discontinue drug for vision changes and refer to ophthalmologist | Baseline eye examination and monthly examinations (for patients taking a dose >15 mg/kg/day) |
| Nicotinic acid (niacin) | Decreased visual acuity, cystoid macular edema, dry eyes, discoloration of eyelids, loss of eyelashes and eyebrows, proptosis | Discontinue medication and refer to ophthalmologist for vision changes | — |
| Phenothiazines (e.g., thioridazine) | Blurred vision, changes in color vision, difficulty seeing at night | Reduce dose or discontinue medication | — |
| Retinoids (e.g., isotretinoin) | Intracranial hypertension, meibomian gland dysfunction, dry-eye syndrome, blepharoconjunctivitis | Prompt referral to ophthalmologist for unexplained headache or blurred vision | Ophthalmic examinations should be performed at least every 6 months. |
| Tamoxifen | Retinopathy, cystoid macular edema, loss of visual acuity, retinal hemorrhage, optic disc swelling | — | Baseline comprehensive eye examination (including testing of color vision) within first year of initiation of therapy and at least every 2 years. |
| Topiramate | Acute angle-closure glaucoma, blurred vision, conjunctival hyperemia, corneal edema, shallow anterior chamber, cataracts, pupil changes, elevated intraocular pressure, visual field defects, blindness | Maximal medical therapy (oral and topical aqueous suppressants) is needed | — |
| Bisphosphonates | |||
| Pamidronate | Uveitis, episcleritis, and scleritis (rare); conjunctivitis | Refer to ophthalmologist for persistent decrease in vision or if patient has ocular pain; topical ocular or systemic medication may be needed to reduce inflammation | — |
| Alendronate | Blurred vision, ocular pain, conjunctivitis, uveitis, scleritis | ||
| Etidronate | Conjunctivitis, blurred vision | ||
Several herbal supplements have also been associated with ocular side effects [17,20,90]. Canthaxanthin can leave deposits in the retina. Severe conjunctivitis has been associated with the application of chamomile around the eye, and allergic conjunctivitis has been found with use of Echinacea purpurea. Datura can cause dilation of the pupils, and Ginkgo biloba has led to spontaneous hyphema and retinal hemorrhage. Licorice root has caused ocular migraine-like visual symptoms without a headache, although large amounts of licorice root are needed to produce the effect [90].
Patients who routinely take medications with documented ocular side effects should be monitored closely to detect early signs of ocular toxicity and avoid damage to the eye or vision impairment. Patients should be reminded to tell their primary care clinician and their eye specialist about all their prescribed medications, as well as any over-the-counter drugs and herbal supplements they take [90]. Patients should be especially encouraged to disclose whether they take herbal supplements, as approximately 40% of patients who do so do not tell their clinician [17].
Malignant conditions of the eye and eyelid are rare in both children and adults. The most common ocular cancer among children is retinoblastoma [114]. Among adults, an estimated 3,490 new cases of primary ocular cancers will be diagnosed in 2020, with an estimated 430 deaths [116]. The two most common primary cancers affecting the adult eye are melanoma and lymphoma, and they are most often the result of metastasis. The most common ocular malignancy is metastasis from either breast cancer or primary melanoma. Metastasis to intraocular structures is more common than metastasis to extraocular structures [117].
Although retinoblastoma is rare, it is the 10th most common childhood cancer overall, representing approximately 2% of all childhood cancers [114]. Retinoblastoma occurs primarily in young children; approximately 66% of cases are diagnosed in children younger than 2 years of age, and 95% of all cases are diagnosed before the age of 5 years [27,121]. The age-adjusted annual incidence among children 0 to 4 years of age is 10 to 14 cases per one million [121]. Surveillance, Epidemiology, and End Results (SEER) data demonstrate that retinoblastoma occurs significantly more often in boys than girls, and the tumor is bilateral in significantly more White Hispanic boys than in White non-Hispanic boys and White Hispanic girls [128]. Approximately 25% to 30% of retinoblastoma cases are heritable, with a germline mutation of the RB1 gene [121]. A positive family history of bilateral or multifocal disease is suggestive of heritable disease.
Leukocoria, or an abnormal white reflection from the retina, is the most common sign of retinoblastoma, occurring in 50% to 60% of children [129]. The presence of strabismus is the second most common sign (20% to 25%), and often correlates with macular involvement; inflammatory signs occur in 6% to 10% [121,129].
Evidence of retinoblastoma can be detected with the red reflex examination. The AAO recommends that this examination be done on infants before they are discharged from the nursery and again at all well-child visits [157]. Despite these recommendations, most children with retinoblastoma had initial detection of leukocoria by a parent [129]. The examination consists of direct ophthalmoscopy done in a darkened room, with the eyes viewed separately at 18 to 30 inches. A white reflex indicates possible retinoblastoma, and the patient should be referred to a pediatric ophthalmologist [157]. Additional procedures and imaging studies are necessary to confirm the diagnosis. Genetic counseling is recommended for all patients with retinoblastoma [121].
Advances in treatment have led to higher rates of ocular survival. Current treatment includes the use of chemotherapy to reduce the size of the tumor (chemoreduction) in combination with focal therapies [121]. Enucleation remains an option and is typically used for advanced tumors. The risk of subsequent neoplasms is higher for children with retinoblastoma, and so they should be followed up closely. Follow-up is also essential for detecting the potential late effects of treatment, which include diminished orbital growth, visual-field defects, and hearing loss [121].
Uveal melanoma affects approximately 4.3 per 1 million individuals each year; although rare, the cancer is the most common primary intraocular malignancy in adults. Individuals are usually older at the time of diagnosis, with a peak incidence at 70 years of age. Men/boys have a higher incidence than women/girls (4.9 vs. 3.7 per million). Among the patient-related risk factors for uveal melanoma are White race, light eye color, and fair skin [130]. Although these risk factors are similar to those for malignant melanoma of the skin, there are no consistent data to suggest that exposure to ultraviolet rays increases the risk for uveal melanoma [130]. One epidemiologic study demonstrated higher rates of uveal melanomas in states with lower prevalences of water fluoridation; the theory is that fluoridation protects against choroidal melanoma by inhibiting microbial agents that cause choroiditis and/or choroidal lesions in humans [183].
Among the structures in the uveal tract, the choroid is the most often affected by melanoma (90%) and the iris is the least often affected (2%) [130]. Small tumors may not produce symptoms, depending on their location with respect to the macula, and are often undetected or an incidental finding on routine eye examination. Metastatic melanoma can be distinguished from primary uveal melanoma by the presence of symptoms, vitreous seeding, and rapidly growing multifocal tumors [130].
Uveal melanoma frequently metastasizes, with metastasis developing in approximately 50% of patients [189]. The liver is the most common site of metastasis [191]. No treatments have been effective for metastatic uveal melanoma, making the prognosis particularly poor for tumors that have already metastasized by the time of diagnosis [189,191,192].
The diagnosis and treatment of uveal melanoma requires the expertise of an ophthalmologist, especially one who has experience with intraocular tumors. Diagnosis is typically determined through examination with such tools as ophthalmoscopy, slit-lamp microscopy, gonioscopy, and transillumination of the globe, depending on the site of the lesion [130].
Ancillary studies such as fluorescein, indocyanine green angiography, and ultrasonography are useful for detecting the characteristics of melanoma [130]. Recent research has enhanced knowledge of the biology of uveal melanoma, leading to a better understanding of the molecular biology, and molecular testing and genetic profiling is helping to allow for better prognostic and predictive testing and to expand therapeutic options [189,192,200].
Intraocular lymphoma is rare. Unlike the situation with uveal melanoma, intraocular lymphoma rarely results from metastasis of a primary lymphoma elsewhere in the body [220]. Primary vitreoretinal lymphoma is the most common tumor, with involvement of the retinal pigment epithelium and vitreous. With this type of tumor, 50% to 90% of patients develop central nervous system lymphoma and/or spinal cord lymphoma within one year [220,221]. Primary choroidal lymphoma is another distinct form of intraocular lymphoma, which is also rare.
The diagnosis of primary vitreoretinal lymphoma includes examination with direct ophthalmoscopy and slit-lamp microscopy. Ancillary studies such as fluorescein angiography and ultrasonography are often useful [221]. Cytologic analysis is the criterion standard for diagnosis, and other tests such as immunocytochemistry, flow cytometry, and polymerase chain reaction testing are also useful [221]. A complete medical and neurologic examination should be carried out to determine the presence of central nervous system lymphoma and to rule out other causes of uveitis, such as sarcoidosis, toxoplasmosis, and tuberculosis. Diagnostic testing should include a chest x-ray, laboratory studies (complete blood count, erythrocyte sedimentation, chemistry levels), neuroimaging of the brain and orbits, vitreous testing, and a lumbar puncture [221].
The most common treatments are chemotherapy (systemic and intravitreal) and external-beam radiation therapy [221]. The prognosis is poor because of the malignant potential of the tumor [220,221].
The two most common malignant lesions of the eyelid are basal cell carcinoma and squamous cell carcinoma, with the former accounting for 80% to 90% of such lesions [223]. There are several risk factors for the development of both types of skin cancer, including fair complexion, light-colored eyes (blue, green, or gray), blond or red hair, tendency to burn or freckle with exposure to the sun, history of exposure to the sun, use of tanning booths, family history of skin cancer, excessive alcohol consumption, and weakened immune system [223,224]. The incidence appears to be higher in men (33% to 39%) than in women (23% to 28%), and the likelihood of developing basal cell carcinoma increases with age [225]. Due to the link to sun exposure, primary care clinicians should emphasize the importance of protecting the eyes from the sun and other sources of ultraviolet rays.
Among periocular basal cell carcinomas, most lesions initially occur on the lower eyelid (>50%) and the medial canthus (30%); 15% of lesions develop on the upper eyelid, and 5% are found on the lateral canthus [223]. Periocular squamous cell carcinoma also develops most often on the lower eyelid (49% to 61%), with 18% to 36% on the medial canthus, 12% on the lateral canthus, and 10% to 23% on the upper eyelid [30,223].
The primary challenges in diagnosing skin cancers are to distinguish between benign and malignant lesions and to identify lesions with malignant potential. The first steps in diagnosing basal cell or squamous cell carcinoma involve obtaining a history and physical examination and evaluating the clinical features of the lesion [223]. Symptomatology does not play a large role in the detection and diagnosis, as early-stage lesions are usually asymptomatic. Advanced lesions may be associated with nonhealing ulceration, bleeding, or pain [31]. A sore that does not heal may be a sign of either basal cell or squamous cell carcinoma [31].
The classic appearance of basal cell carcinoma is a pearly, waxy, or translucent papule or nodule with small telangiectatic vessels on its surface (nodular type). This type occurs frequently in the periocular area [31,227]. Superficial basal cell carcinomas are thinner than nodular lesions and often appear as scaly erythematous patches or plaques that may slowly enlarge. Care should be taken to distinguish this type of lesion from a benign inflammatory skin disorder, such as psoriasis or nummular eczema [31]. Both the superficial and the nodular types may look brown, blue, or black as a result of the presence of melanin. The morpheaform type of basal cell carcinoma usually presents as a whitish or yellowish, indurated, scar-like plaque, usually with indistinct borders [31]. The clinical course of this type is aggressive, and left untreated, it can lead to extensive destruction of soft tissue [227].
The various clinical presentations of basal cell carcinoma make diagnosis a challenge, with the lesion being undetected or misidentified in 20% to 40% of cases [227]. The lesions that are most often confused with basal cell carcinoma include papilloma, nevus, hidrocystoma, epidermal inclusion cyst, and squamous cell carcinoma [31]. The clinical features of squamous cell carcinoma also vary widely [223]. Squamous cell carcinoma usually presents as an ulcerated erythematous nodule or superficial erosion on the skin but can also appear as a verrucous papule or plaque. Color also varies, and the lesions may be reddish-brown, pink, or flesh-colored. They typically present as exophytic tumors, ranging in size from a few millimeters to centimeters. Larger lesions may appear crusted, erythematous, or eroded. In contrast to basal cell carcinoma, overlying telangiectasias are uncommon. The margins may be ill defined or well circumscribed [223].
According to the National Comprehensive Cancer Network (NCCN), the goal of treatment is elimination of the tumor with maximal preservation of function and physical appearance. Treatment decisions should be individualized in accordance with the patient's particular risk factors and preferences [225]. The treatment of choice for periocular basal cell and squamous cell carcinomas is surgical excision, and biopsy should be done preoperatively to confirm the diagnosis and identify the histologic subtype [31]. Mohs microsurgery is preferred for high-risk lesions, such as recurrent tumors, larger lesions, lesions near the medial canthus, and morpheaform basal cell carcinomas, as these lesions are associated with incomplete resection, the primary risk factor for recurrence [227]. The American Academy of Dermatology, in collaboration with the American College of Mohs Surgery, the American Society for Dermatologic Surgery Association, and the American Society for Mohs Surgery, has developed appropriate use criteria for Mohs micrographic surgery [32]. Mohs surgery is associated with the lowest five-year recurrence rate of any of the treatment modalities (1% to 2% for primary tumors) [223].
Radiotherapy may be useful for patients who are not candidates for surgery or who have an unresectable lesion [31,227]. Cryotherapy for periocular basal cell or squamous cell carcinoma is not usually recommended because removal of the lesion cannot be confirmed. However, the findings of several studies have indicated that this approach can be effective, and it is best used to treat small lesions in patients who are not good candidates for surgery. Recurrence rates are high for curettage and laser removal and photodynamic therapy [227].
Patient A, a woman 20 years of age, arrives at her primary care clinician's office with red eyes, profuse epiphora, and edematous eyelids. She noticed redness and tearing in her left eye three days ago. These symptoms develop in her right eye the next day, so she calls in sick to her gymnastics teaching job at a local youth center. She notes no blurred vision, photophobia, halos, or pain/burning sensations, but she feels like "sand is in my left eye." She also has a slight headache, sneezing, and a runny nose. Patient A initially assumed her condition was caused by allergies and self-medicated (unsuccessfully) with an over-the-counter antihistamine and ibuprofen. She is unsure if any of the children at the youth center have similar symptoms. Based on the symptoms and history, the clinician suspects adenoviral conjunctivitis and prepares to examine Patient A.
Rationale and comments: When diagnosing a red eye, the primary care clinician should first determine if a referral to an ophthalmologist is necessary to protect the patient's vision. Serious conditions associated with a red eye (e.g., scleritis, uveitis, angle-closure glaucoma, keratitis) typically affect vision and/or cause severe pain, which Patient A does not have. Although conjunctivitis is primarily diagnosed on the basis of the patient's history and symptoms, further evaluation is necessary for confirmation and to ensure no other eye conditions are occurring simultaneously.
Assessment and evaluation: Physical examination shows that Patient A's pupils are normal, and no ciliary flush is detected when evaluated in natural light. The perilimbal region is clear. The only additional feature observed during her ocular evaluation is preauricular adenopathy. With use of a rapid immunochromatography test, adenoviral antigen is found in tear fluid obtained from Patient A's left eye.
Rationale and comments: The rapid immunochromatography test detects adenoviral antigen in human eye fluid. This point-of-care test, which shows results within 10 minutes, has been found to be as effective as polymerase chain reaction or cell culture. Ciliary flush and pupillary abnormalities are not characteristic of conjunctivitis but are indicative of red-eye conditions such as iritis, angle-closure glaucoma, or keratitis. These features should be assessed during the examination of a red eye. Little or no injection around the iris is indicative of conjunctivitis. Preauricular adenopathy is characteristic of viral conjunctivitis.
Ongoing management: Patient A is informed of the diagnosis of adenoviral conjunctivitis and is told that the condition is self-limiting and will likely resolve itself in 10 to 14 days. Antibiotics are not offered to her, and she is told that applying cool compresses for 10 to 15 minutes may provide some relief. The clinician also tells Patient A that she likely contracted this highly contagious condition from one of the children at the youth center and that she should not return to work until she is symptom free (likely 5 to 10 more days). To promote resolution as well as to prevent reinfection and spread of adenoviral conjunctivitis, Patient A should practice very strict hygiene—especially frequent hand washing; avoidance of sharing towels/soap, pillowcases, and keyboards; and washing of sheets, towels, and other items that may spread the infection. She is advised to avoid touching her eyes, and is given an appointment for a check-up in seven days.
If possible, the youth center should be contacted to inform individuals working at or attending the center that they should refrain from coming to the center if they have conjunctival symptoms. Additionally, the use of good hygiene should be stressed to the children and staff to avoid acquiring conjunctivitis.
Rationale and comments: Patient A is not offered antibiotics because there is no evidence of bacterial involvement and antibiotics will not be effective against the viral infection.
The incidence of adenoviral conjunctivitis peaks during the summer months. Individuals exposed to child-oriented facilities are at the highest risk, perhaps because the spread of the condition is fostered by the concentration of children in combination with their lack of appropriate hygiene.
Follow-up management: At the seven-day visit, Patient A's eyes appear free of redness, tearing, discharge, and are no longer itchy. She is not referred to an ophthalmologist.
Rationale and comments: Primary care clinicians can treat most individuals with conjunctivitis without referral. Patient A would require referral to an ophthalmologist if her condition was unchanged or worsened or if regions other than the conjunctiva were affected.
Patient C, an active White man 83 years of age, was diagnosed with non-neovascular (dry) age-related macular degeneration at 77 years of age. Patient C attempted to quit smoking cigarettes nine years ago, and although he smokes much less, he still smokes periodically. The ophthalmologist gave Patient C an Amsler grid to self-monitor his vision. Last year, some centralized gridlines appeared distorted to the patient, and the ophthalmologist determined that age-related macular degeneration had progressed significantly (Figure 2 and Figure 3). (Patient C had drusen of about 100 micrometers bilaterally.) While at his primary care clinician's office for a check-up, Patient C tells the clinician that some lines on the grid appear to be missing and he can no longer read his watch. He states that his next age-related macular degeneration-related visit with the ophthalmologist is five months away.
Rationale and comments: Patient C's dry age-related macular degeneration was diagnosed in an early stage. The Amsler grid was given to help determine when the disease progressed to the intermediate stage, when a combination of antioxidant vitamins might be effective in slowing progression.
Assessment and evaluation: After completing Patient C's check-up, the clinician refers him to his ophthalmologist for an appointment earlier than the one scheduled. The physician reminds Patient C that age-related macular degeneration will not take his sight away completely; it will affect only his central vision, and his peripheral vision will remain intact. A smoking cessation program is recommended. Patient C is also asked about his ability to perform everyday functions and what activities he enjoys. He notes that his children take care of the utility and other bills with an electronic payment online. A few months ago, his wife forbade him from driving anymore. But he enjoys cooking meals for his wife a few times a week, solving crossword puzzles, reading the newspaper daily, looking at pictures of his great grandchildren, and watching sports on television. To prime him for a discussion with his ophthalmologist, the clinician briefly tells him about low-vision devices that may be of benefit to him, such as magnifiers, closed-caption television, high-contrast watch faces, and a computer that reads printed material. The physician also tells Patient C that the ophthalmologist may prescribe treatment with a combination of antioxidants (vitamin C vitamin E, beta carotene, zinc oxide, and cupric oxide). The physician explains, however, that Patient C should not take beta carotene supplementation because he continues to smoke.
Rationale and comments: A large trial has shown that the combination of vitamins C and E, zinc, copper, and beta carotene slows central vision loss in individuals with intermediate dry age-related macular degeneration. However, beta carotene should be removed from this supplement regimen for patients who smoke. Additionally, other large studies have shown that beta carotene may accelerate progression of age-related macular degeneration. Patient C should be encouraged to quit smoking, as smoking accelerates the progression of age-related macular degeneration.
Ongoing management: Patient C's primary care clinician continues to monitor quality of life associated with his vision loss for two years. The age-related macular degeneration evolved into the neovascular (wet) form, and he is being treated by his ophthalmologist accordingly. Patient C, now 86 years of age, is no longer able to cook and has difficulties with other activities. His wife, who is 76 years of age, tells the clinician that she fears he may fall at home because his favorite area in the house has stairs. The clinician has also noticed that Patient C exhibits signs of depression. At each appointment, the clinician has discussed information about government and private agencies that can help with redesigning the home for Patient C's needs, low-vision rehabilitation programs, support and activity groups, and psychotherapy. The clinician has also asked questions to determine the patient's psychosocial status. The physician prescribes an antidepressant for Patient C.
Rationale and comments: A devastating consequence of age-related macular degeneration-induced central vision loss is the accompanying loss of independence. As vision declines, an individual with age-related macular degeneration and his or her family find that tasks such as cooking or navigating the home are dangerous. Studies suggest that loss of visual acuity correlate to increased injuries and falls. Individuals' dissatisfaction with the inability to perform valued activities increases the risk of depression.
Follow-up management: Patient C's primary care clinician continues to monitor his functional and quality-of-life needs. Although his central vision has declined, Patient C is satisfied with the support he has from his family and others and his functioning with low-vision devices. An occupational therapist certified in low-vision rehabilitation has helped the patient and his wife make small modifications to their home to increase his independence. Patient C is happy that he can enjoy cooking occasionally again.
Rationale and comments: At each visit, Patient C's primary care clinician provided education to the patient and his family about services to help him adapt to his vision impairment. This information played a major role in protecting the patient's quality of life and independence. The low-vision rehabilitation program and services helped the patient's family become involved in preparing the couple for age-related macular degeneration-associated life changes. For instance, because the couple's health was generally good and they wished to stay in their home as long as possible, their children completed a series of practical home modifications to help with Patient C's low-vision problem through a state-funded program. To reduce the caregiving burden, a caretaker was hired to perform weekly light cleaning and to precook nutritional meals. Low-vision devices were purchased as needed. Also, Patient C and his wife continue to attend weekly support group sessions hosted by their local hospital.
In their position at the frontline of health care, primary care providers have an important role in ensuring proper eye health and preserving vision in their patients. Healthcare professionals in the primary care setting should be skilled in performing screening eye examinations in children and adults to rule out common disorders and to detect signs of occult eye disease as well as early signs of some systemic diseases. They should also be knowledgeable about the most recent recommended guidelines for comprehensive eye examinations (performed by an eye specialist) and encourage their patients to adhere to these guidelines. In addition, clinicians should be competent in diagnosing and treating minor symptomatic conditions of the eyes and eyelids, educating patients about eye safety, providing immediate treatment for eye injuries and emergencies, managing chronic diseases that have consequences in the eye, and identifying early signs of ocular tumors. Perhaps most important, clinicians should recognize the signs, symptoms, and diagnoses that require referral to an ophthalmologist.
1. Centers for Disease Control and Prevention. Vision Health Initiative (VHI). Available at https://www.cdc.gov/visionhealth. Last accessed October 1, 2023.
2. Channa R, Zafar SN, Canner JK, et al. Epidemiology of eye-related emergency department visits. JAMA Ophthalmol. 2016;134(3):312-319.
3. ٣. Rein DB, Wittenborn JS, Zhang P, et al. The Economic Burden of Vision Loss and Blindness in the United States. Ophthal. 2022;129(4):369-378.
4. Owens PL, Mutter R. Emergency department visits related to eye injuries. Statistical Brief #112. Rockville, MD: Agency for Healthcare Research and Quality; 2011.
5. American Academy of Ophthalmology. Eye Health Statistics. Available at https://www.aao.org/newsroom/eye-health-statistics. Last accessed October 1, 2023.
6. Saydah SH, Gerzoff RB, Saadine JB, et al. Eye care among U.S. adults at high risk for vision loss in the United States in 2002 and 2017. JAMA Ophthalmol. 2020;138:479-489.
7. Flaxman AD, Wittenborn JS, Robalik T, et al. Prevalence of visual acuity loss or blindness in the U.S. JAMA Ophthalmology.2021;139(7):171-123.
8. American Academy of Ophthalmology. Comprehensive Adult Medical Eye Evaluation PPP 2020. Available at https://www.aao.org/education/preferred-practice-pattern/comprehensive-adult-medical-eye-evaluation-ppp. Last accessed October 1, 2023.
9. Anzeljc S, Ziemnik L, Koscher S, et al. Preschool vision screening collaborative: successful uptake of guidelines in primary care. Ped Qual Saf. 2019;4(6):e241.
10. American Academy of Ophthalmology. Conjunctivitis PPP 2018. Available at https://www.aao.org/education/preferred-practice-pattern/conjunctivitis-ppp-2018. Last accessed October 1, 2023.
11. American Academy of Ophthalmology. Preferred Practice Pattern Guidelines. Available at https://www.aaojournal.org/content/preferred-practice-pattern. Last accessed October 1, 2023.
13. 13.Newman NJ. The Optic Neuritis Treatment Trial. Available at https://www.aaojournal.org/article/S0161-6420(19)32364-4/pdf. Last accessed October 1, 2023.
14. Wittenborn JS, Rein DB. The Future of Vision: Forecasting the Prevalence and Cost of Vision Problems. Available at https://preventblindness.org/wp-content/uploads/2020/04/Future_of_Vision_final_0.pdf. Last accessed October 1, 2023.
15. American Optometric Association. Blepharitis. Available at https://www.aoa.org/healthy-eyes/eye-and-vision-conditions/blepharitis. Last accessed October 1, 2023.
16. Fraunfelder FW, Fraunfelder FT. Adverse ocular drug reactions recently identified by the National Registry of Drug-Induced Ocular Side Effects. Ophthalmology. 2004;111(7):1275-1279.
17. Santaella RM, Fraunfelder FW. Ocular adverse effects associated with systemic medications: recognition and management. Drugs. 2007;67(1):75-93.
19. 19. American Academy of Ophthalmology. Dry Eye Syndrome PPP 2018. Available at https://www.aao.org/education/preferred-practice-pattern/dry-eye-syndrome-ppp-2018. Last accessed October 1, 2023.
20. Wilkinson JT, Fraunfelder FW. Use of herbal medicines and nutritional supplements in ocular disorders: an evidence-based review. Drugs. 2011;71(18):2421-2434.
22. Mafee MF, Karimi A, Shah JD, Rapoport M, Ansari SA. Anatomy and pathology of the eye: role of MR imaging and CT. Magn Reson Imaging Clin North Am. 2006;14(2):249-270.
23. McIlwain JT. An Introduction to the Biology of Vision. Cambridge: Cambridge University Press; 1996.
24. Moses K, Banks JC, Nava PB, Petersen D. Atlas of Clinical Gross Anatomy. 2nd ed. Philadelphia, PA: Saunders; 2012.
25. American Optometric Association. Comprehensive Adult Eye and Vision Examination. Available at https://aoa.uberflip.com/i/578152-aoa-clinical-practice-guidelines-adult-eye-exam. Last accessed October 1, 2023.
26. U.S. Preventive Services Task Force. Vision in Children Ages 6 Months to 5 Years: Screening. Available at https://www.uspreventiveservicestaskforce.org/uspstf/recommendation/vision-in-children-ages-6-months-to-5-years-screening. Last accessed October 1, 2023.
27. American Optometric Association. Pediatric Eye and Vision Examination. Available at https://aoa.uberflip.com/i/807465-cpg-pediatric-eye-and-vision-examination. Last accessed October 1, 2023.
28. U.S. Preventive Services Task Force. Impaired Visual Acuity in Older Adults: Screening. Available at https://www.uspreventiveservicestaskforce.org/uspstf/recommendation/impaired-visual-acuity-screening-older-adults. Last accessed October 1, 2023.
29. American Academy of Ophthalmology. Cataract in the Adult Eye PPP 2021. Available at https://www.aao.org/education/preferred-practice-pattern/cataract-in-adult-eye-ppp-2021-in-press. Last accessed October 1, 2023.
30. Faustina M, Diba R, Ahmadi MA, Esmaeli B. Patterns of regional and distant metastasis in patients with eyelid and periocular squamous cell carcinoma. Ophthalmology. 2004;111(10):1930-1932.
31. Bader RS. Basal Cell Carcinoma. Available at https://emedicine.medscape.com/article/276624-overview. Last accessed October 1, 2023.
32. Connolly SM, Baker DR, Coldiron BM, et al. AAD/ACMS/ASDSA/ASMS 2012 appropriate use criteria for Mohs micrographic surgery: a report of the American Academy of Dermatology, American College of Mohs Surgery, American Society for Dermatologic Surgery Association, and the American Society for Mohs Surgery. J Am Acad Dermatol. 2012;67(4):531-550.
33. Kemper AR, Clark SJ. Preschool vision screening by family physicians. J Pediatr Ophthalmol Strabismus. 2007;44(1):24-27.
34. American Academy of Ophthalmology. Primary Angle-Closure Disease PPP 2020. Available at https://www.aao.org/education/preferred-practice-pattern/primary-angle-closure-disease-ppp. Last accessed October 1, 2023.
35. Wickstrom GC, Kelley DK, Keyserling TC, et al. Confidence of academic general internists and family physicians to teach ambulatory procedures. J Gen Intern Med. 2000;15(6):353-360.
36. Bonds DE, Mychaleckyj JC, Watkins R, Palla S, Extrom P. Ambulatory care skills: do residents feel prepared? Med Educ Online. 2002;7.
37. American Academy of Ophthalmology. Joint Policy Statement: Vision Screening for Infants and Children—2022. Available at https://www.aao.org/education/clinical-statement/vision-screening-infants-children-2022. Last accessed October 1, 2023.
38. Goldzweig CL, Rowe S, Wenger NS, MacLean CH, Shekelle PG. Preventing and managing visual disability in primary care: clinical applications. JAMA. 2004;291(12):1497-1502.
40. American Academy of Ophthalmology. Referral of Persons with Possible Eye Diseases or Injury - 2014. Available at https://www.aao.org/education/clinical-statement/guidelines-appropriate-referral-of-persons-with-po. Last accessed October 1, 2023.
41. American Academy of Pediatrics, the American Association for Pediatric Ophthalmology and Strabismus, and the American Association of Certified Orthoptists. Visual system assessment in infants, children, and young adults by pediatricians: policy statement, 2016. Pediatrics. 2016;137(1):e20153596.
42. Friedman LS, Kaufman LM. Guidelines for pediatrician referrals to the ophthalmologist. Pediatr Clin North Am. 2003;50(1):41-53.
43. Kempen JH, Mitchell P, Lee KE, et al. The prevalence of refractive errors among adults in the United States, Western Europe, and Australia. Arch Ophthalmol. 2004;122(4):495-505.
44. Wen G, Tarczy-Hornoch K, McKean-Cowdin R, et al. Prevalence of myopia, hyperopia, and astigmatism in non-Hispanic white and Asian children: Multi-ethnic Pediatric Eye Disease Study. Ophthalmology. 2013;120(10):2109-2116.
45. Borchert MS, Varma R, Cotter SA, et al. Risk factors for hyperopia and myopia in preschool children the Multi-ethnic Pediatric Eye Disease and Baltimore Pediatric Eye Disease studies. Ophthalmology. 2011;118(10):1966-1973.
46. Ying GS, Maguire MG, Cyert LA, et al. Prevalence of vision disorders by racial and ethnic group among children participating in Head Start. Ophthalmology. 2014;121(3):630-636.
47. Fozailoff A, Traczy-Hornoch K, Cotter S, et al. Prevalence of astigmatism in 6- to 72-month-old African American and Hispanic children: the Multi-ethnic Pediatric Eye Disease Study. Ophthalmology. 2011;118(2):284-293.
48. McKean-Cowdin R, Cotter SA, Tarczy-Hornoch K, et al. Prevalence of amblyopia or strabismus in Asian and non-Hispanic white preschool children: Multi-ethnic Pediatric Eye Disease Study. Ophthalmology. 2013;120(10):2117-2124.
49. Tarczy-Hornoch K, Varma R, Cotter SA, et al. Risk factors for decreased visual acuity in preschool children: the Multi-ethnic Pediatric Eye Disease and Baltimore Pediatric Eye Disease studies. Ophthalmology. 2011;118(11):2262-2273.
50. Shah SM, Khanna CL. Ophthalmic emergencies for the clinician. Mayo Clin Proc. 2020;95(5):1050-1058.
51. Bureau of Labor Statistics. Workers suffered 18,510 eye-related injuries and illnesses in 2020. Available at https://www.bls.gov/opub/ted/2023/workers-suffered-18510-eye-related-injuries-and-illnesses-in-2020.htm. Last accessed October 1, 2023.
52. Azari AA, Arabi A. Conjunctivitis: a systematic review. J Ophthalmic Vis Res. 2020;15(3):372-395.
53. Taukikewa M, Stacey AW. A review of hypertensive retinopathy and chorioretinopathy. Clin Optom (Auckl). 2020;12:67-73.
54. U.S. Food and Drug Administration. FDA Approves First Biosimilar to Treat Macular Degeneration Disease and Other Eye Conditions. Available at https://www.fda.gov/news-events/press-announcements/fda-approves-first-biosimilar-treat-macular-degeneration-disease-and-other-eye-conditions. Last accessed October 1, 2023.
55. Centers for Disease Control and Prevention. Conjunctivitis (Pink Eye). Available at https://www.cdc.gov/conjunctivitis/clinical.html. Last accessed October 1, 2023.
56. Porter RS, Kaplan JL, Homeier BP (eds). The Merck Manual of Patient Symptoms. Whitehouse Station, NJ: Merck Research Laboratories; 2008.
57. Gilliland GD. Dacryocystitis. Available at https://emedicine.medscape.com/article/1210688-overview?form=fpf. Last accessed October 1, 2023.
58. Green M, Apel A, Stapleton F. Risk factors and causative organisms in microbial keratitis. Cornea. 2008;27(1):22-27.
59. Beers MH, Porter RS, Jones TV, Kaplan JL, Berkwits M (eds). The Merck Manual. 20th ed. Whitehouse Station, NJ: Merck Research Laboratories, 2018.
60. Collier SA, Gronostaj MP, MacGurn AK, et al. Estimated burden of keratitis—United States, 2010. MMWR. 2014;63(45):1027-1030.
61. Centers for Disease Control and Prevention. Healthy Contact Lens Wear and Care. Available at https://www.cdc.gov/contactlenses/fast-facts.html. Last accessed October 1, 2023.
62. American Academy of Ophthalmology. Red Eye. Available at https://www.aao.org/eye-health/symptoms/red-eye-3. Last accessed October 1, 2023.
63. Mills DM, Bodman MG, Meyer DR, Morton AD 3rd. The microbiologic spectrum of dacryocystitis: a national study of acute versus chronic infection. Ophthal Plast Reconstr Surg. 2007;23(4):302-306.
64. Kubo M, Sakuraba T, Arai Y, Nakazawa M. Dacryocystorhinostomy for dacryocystitis caused by methicillin-resistant Staphylococcus aureus: report of four cases. Jpn J Ophthalmol. 2002;46(2):177-182.
65. Kotlus BS, Rodgers IR, Udell IJ. Dacryocystitis caused by community-onset methicillin-resistant Staphylococcus aureus. Ophthal Plast Reconstr Surg. 2005;21(5):371-375.
66. Multicenter Uveitis Steroid Treatment (MUST) Trial Research Group. Randomized comparison of systemic anti-inflammatory therapy versus fluocinolone acetonide implant for intermediate, posterior, and panuveitis: the Multicenter Uveitis Steroid Treatment Trial. Ophthalmology. 2011;118(10):1916-1926.
67. Writing Committee for the Multicenter Uveitis Steroid Treatment (MUST) Trial and Follow-Up Study Research Group. Association between long-lasting intravitreous flucinolone acetonide implant vs systemic anti-inflammatory therapy and visual acuity at 7 years among patients with intermediate, posterior, or panuveitis. JAMA. 2017;317(19):1993-2005.
68. Jackson TL, Paraskevopoulos T, Georgalas I. Systematic review of 342 cases of endogenous bacterial endophthalmitis. Surv Ophthalmol. 2014;59(6):627-635.
69. Pijl BJ, Theelen T, Tilanus MA, Rentenaar R, Crama N. Acute endophthalmitis after cataract surgery: 250 consecutive cases treated at a tertiary referral center in the Netherlands. Am J Ophthalmol. 2010;149(3):482-487.
70. Essex RW, Yi Q, Charles PG, Allen PJ. Post-traumatic endophthalmitis. Ophthalmology. 2004;111(11):2015-2022.
71. Callegan MC, Engelbert M, Parke DW 2nd, Jett BD, Gilmore MS. Bacterial endophthalmitis: epidemiology, therapeutics, and bacterium-host interactions. Clin Microbiol Rev. 2002;15(1):111-124.
72. Gower EW, Lindsley K, Tulenko SE, Nanji AA, Leyngold I, McDonnell PJ. Perioperative antibiotics for prevention of acute endophthalmitis after cataract surgery. Cochrane Database Syst Rev. 2017;2:CD006364.
73. Kim CH, Chen ME, Coleman AL. Adjunctive steroid therapy versus antibiotics alone for acute endophthalmitis after intraocular procedure. Cochrane Database Syst Rev. 2017;2:CD012131.
74. Gangaputra S, Drye L, Vaidya V, et al. Non-cytomegalovirus ocular opportunistic infections in patients with acquired immunodeficiency syndrome. Am J Ophthalmol. 2013;155(2):206-212.
75. Freeman WR, Van Natta ML, Jabs D, et al. Vision function in HIV-infected individuals without retinitis: report of the Studies of Ocular Complications of AIDS Research Group. Am J Ophthalmol. 2008;145(3):453-462.
76. Atkins EJ, Biousse V, Newman NJ. The natural history of optic neuritis. Rev Neurol Dis. 2006;3(2):45-56.
77. Yeh EA, Graves JS, Benson LA, et al. Pediatric Optic Neuritis. Neurology. 2016;87(9 Suppl 2):S53-S58.
78. Herce H. Childhood Optic Neuritis. Available at https://emedicine.medscape.com/article/1217290-overview. Last accessed October 1, 2023.
79. Bonhomme GR, Waldman AT, Balcer LJ, et al. Pediatric optic neuritis: brain MRI abnormalities and risk of multiple sclerosis. Neurology. 2009;72(10):881-885.
80. Gal RL, Vedula SS, Beck R. Corticosteroids for treating optic neuritis. Cochrane Database Syst Rev. 2015;8:CD001430.
81. Optic Neuritis Study Group. Visual function 15 years after optic neuritis: a final follow-up report from the Optic Neuritis Treatment Trial. Ophthalmology. 2008;115(6):1079-1082.
82. Wilejto M, Shroff M, Buncic JR, Kennedy J, Goia C, Banwell B. The clinical features, MRI findings, and outcome of optic neuritis in children. Neurology. 2006;67(2):258-262.
83. Brady KM, Brar AS, Lee AG, Coats DK, Paysse EA, Steinkuller PG. Optic neuritis in children: clinical features and visual outcome.J AAPOS. 1999;3(2):98-103.
84. Lindsley K, Matsumura S, Hatef E, Akpek EK. Interventions for chronic blepharitis. Cochrane Database Syst Rev. 2012;5:CD005556.
85. Lindsley K, Nichols JJ, Dickersin K. Interventions for acute internal hordeolum. Cochrane Database Syst Rev. 2017;1:CD007742.
86. U.S. Food and Drug Administration. FDA Approves New Medication for Dry Eye Disease. Available at https://www.fda.gov/news-events/press-announcements/fda-approves-new-medication-dry-eye-disease. Last accessed October 1, 2023.
87. Ervin A-M, Law A, Pucker AD. Punctal occlusion for dry eye syndrome. Cochrane Database Syst Rev. 2017;6:CD006775.
88. American Academy of Ophthalmology. Choosing Wisely: Five Things Ophthalmologists and Patients Should Question. Available at https://www.aao.org/education/choosing-wisely. Last accessed October 1, 2023.
89. DeBacker C. Entropion or Ectropion Repair. Available at https://emedicine.medscape.com/article/1844045-overview. Last accessed October 1, 2023.
90. Kalyani PS, Fawzi AA, Gangaputra S, et al. Retinal vessel caliber among people with acquired immunodeficiency syndrome: relationships with visual function. Am J Ophthalmol. 2012;153(3):428-433.
91. Fraunfelder FW. Ocular side effects from herbal medicines and nutritional supplements. Am J Ophthalmol. 2004;138(4):639-647.
92. Gilliland GD. Dermatochalasis. Available at https://emedicine.medscape.com/article/1212294-overview. Last accessed October 1, 2023.
93. Morris CL, Chesnutt DA. Acquired Ptosis: Evaluation and Management. Available at https://www.aao.org/eyenet/article/acquired-ptosis-evaluation-management. Last accessed October 1, 2023.
94. Scheinfeld N. The use of apraclonidine eyedrops to treat ptosis after the administration of botulinum toxin to the upper face. Dermatol Online J. 2005;11(1):9.
95. Popovic Suic S, Cerovski B, Tojagic M. Lid retraction etiology. Acta Med Croatica. 2004;58(5):407-409.
96. Aldave AJ, Maus M, Rubin PA. Advances in the management of lower eyelid retraction. Facial Plast Surg. 1999;15(3):213-224.
97. Hallett M, Evinger C, Jankovic J, Stacy M. Update on blepharospasm: report from the BEBRF International Workshop. Neurology. 2008;71:1275-1282.
98. Peckham EL, Lopez G, Shamim EA, et al. Clinical features of patients with blepharospasm: a report of 240 patients. Eur J Neurol. 2011;18(3):382-386.
99. Vaziri K, Schwartz SG, Flynn HW Jr, Kishor KS, Moshfeghi AA. Eye-related emergency department visits in the United States, 2010. Ophthalmology. 2016;123(4):917-919.
100. Matsa E, Shi J, Wheeler KK, et al. Trends in U.S. emergency department visits for pediatric acute ocular injury. JAMA Ophthalmol. 2018;136(8):895–903.
101. Channa R, Zafar SN, Canner JK. Epidemiology of eye-related emergency department visits. JAMA Ophthalmol. 2016;134(3):312-319.
102. Michaelides M, Stover NB, Francis PJ, Weleber RG. Retinal toxicity associated with hydroxychloroquine and chloroquine: risk factors, screening, and progression despite cessation of therapy. Arch Ophthalmol. 2011;129(1):30-39.
103. Chen AJ, Linakis JG, Mello MJ, Greenberg PB. Epidemiology of infant ocular and periocular injuries from consumer products in the United States, 2001–2008. J AAPOS. 2013;17(3):239-242.
104. American Association of Ophthalmologists. Recommendations on Screening for Chloroquine and Hydroxychloroquine Retinopathy, 2016. Available at https://www.aao.org/education/clinical-statement/revised-recommendations-on-screening-chloroquine-h. Last accessed October 1, 2023.
105. Chen T, Kistamgari S, Smith GA. Consumer product-related pediatric eye injuries treated in United States emergency departments. Ophthalmic Epidemiol. 2023 Aug;30(4):424-433.
106. Chen AJ, Kim JG, Linakis JG, Mello MJ, Greenberg PB. Eye injuries in the elderly from consumer products in the United States: 2001–2007. Graefes Arch Clin Exp Ophthalmol. 2013;251(3):645-651.
107. American Association for Pediatric Ophthalmology and Strabismus. Eye Safety. Available at https://aapos.org/viewdocument/eye-safety. Last accessed October 1, 2023.
108. Chen AJ, Chan JJ, Linakis JG, Mello MJ, Greenberg PB. Age and consumer product-related eye injuries in the United States. R I Med J. 2014;97(1):44-48.
109. Moren Cross J, Griffin R, Owsley C, McGwin G. Pediatric eye injuries related to consumer products in the United States, 1997–2006. J AAPOS. 2008;12(6):626-628.
110. McGwin G Jr, Hall TA, Seale J, Xie A, Owsley C. Consumer product-related eye injury in the United States, 1998–2002. J Safety Res. 2006;37(5):501-506.
111. Seidman CJ, Linakis JG, Mello MJ, Greenberg PB. Aerosol container-related eye injuries in the United States: 1997–2009.Am J Ophthalmol. 2011;151(6):1041-1046.
112. American Academy of Ophthalmology. Eye Injuries from Paintball Guns, Air Guns and Other Projectile Toys. Available at https://www.aao.org/eye-health/news/combating-eye-injuries-from-air-guns. Last accessed October 1, 2023.
113. American Academy of Ophthalmology. ACR, AAD, RDS, and AAO 2020 Joint Statement on Hydroxychloroquine Use with Respect to Retinal Toxicity. Available at https://www.aao.org/education/clinical-statement/acr-aad-rds-aao-2020-joint-statement-on-hydroxychl-2. Last accessed October 1, 2023.
114. National Cancer Institute. Cancer in Children and Adolescents. Available at https://www.cancer.gov/types/childhood-cancers/child-adolescent-cancers-fact-sheet. Last accessed October 1, 2023.
115. Smith B, Pledger D. 2022 Fireworks Annual Report. Bethesda, MD: U.S. Consumer Product Safety Commission; 2022.
116. Siegel RL, Miller KD, Wagle NS, Jemal A. Cancer statistics, 2023. CA Cancer J Clin. 2023;73(1):17-48.
117. Ahmad SM, Esmaeli B. Metastatic tumors of the orbit and ocular adnexa. Curr Opin Ophthalmol. 2007;18(5):405-413.
118. American Academy of Ophthalmology. Recognizing and Treating Eye Injuries. Available at https://www.aao.org/eye-health/tips-prevention/injuries. Last accessed October 1, 2023.
119. American Academy of Ophthalmology, American Academy of Pediatricians. Joint Policy Statement: Protective Eyewear for Young Athletes. San Francisco, CA: American Academy of Ophthalmology; 2013.
120. Rodriguez JO, Lavina AM, Agarwal A. Prevention and treatment of common eye injuries in sports. Am Fam Physician. 2003;67(7): 1481-1488.
121. National Cancer Institute. Retinoblastoma Treatment (PDQ). Available at https://www.cancer.gov/types/retinoblastoma/hp/retinoblastoma-treatment-pdq. Last accessed October 1, 2023.
122. Kriz PK, Comstock RD, Zurakowski D, et al. Effectiveness of protective eyewear in reducing eye injuries among high school field hockey players. Pediatrics. 2012;130(6):1069-1075.
123. Lincoln AE, Caswell AV, Almquist JL, et al. Effectiveness of the women's lacrosse protective eyewear mandate in the reduction of eye injuries. Am J Sports Med. 2012;40(3):611-614.
124. Forrest KY, Cali JM. Epidemiology of lifetime work-related eye injuries in the U.S. population associated with one or more lost days of work. Ophthalmic Epidemiol. 2009;16(3):156-162.
125. Bureau of Labor Statistics. Nonfatal Occupational Injuries and Illnesses Requiring Days Away from Work, 2015. Available at https://www.bls.gov/news.release/pdf/osh2.pdf. Last accessed October 1, 2023.
126. Centers for Disease Control and Prevention. The National Institute for Occupational Safety and Health (NIOSH): Traumatic Occupational Injuries. Available at https://www.cdc.gov/niosh/injury/fastfacts.html. Last accessed October 1, 2023.
127. American Optometric Association. Computer Vision Syndrome. Available at https://www.aoa.org/healthy-eyes/eye-and-vision-conditions/computer-vision-syndrome?sso=yhttps://www.aoa.org/healthy-eyes/eye-and-vision-conditions/computer-vision-syndrome. Last accessed October 1, 2023.
128. Wong JR, Tucker MA, Kleinerman RA, Devesa SS. Retinoblastoma incidence patterns in the U.S. Surveillance, Epidemiology, and End Results program. JAMA Ophthalmol. 2014;132(4):478-483.
129. Abramson DH, Beaverson K, Sangani P, et al. Screening for retinoblastoma: presenting signs as prognosticators of patient and ocular survival. Pediatrics. 2003;112(6):1248-1255.
130. National Cancer Institute. Intraocular (Uveal) Melanoma Treatment (PDQ). Available at https://www.cancer.gov/types/eye/hp/intraocular-melanoma-treatment-pdq. Last accessed October 1, 2023.
131. Occupational Safety and Health Administration. Eye and Face Protection eTool. Available at https://www.osha.gov/SLTC/etools/eyeandface. Last accessed October 1, 2023.
132. Karliner LS, Jacobs EA, Chen AH, Mutha S. Do professional interpreters improve clinical care for patients with limited English proficiency? A systematic review of the literature. Health Serv Res. 2007;42(2):727-754.
133. Kuo DZ, O'Connor KG, Flores G, Minkovitz CS. Pediatricians' use of language services for families with limited English proficiency. Pediatrics. 2007;119(4):920-927.
134. Ngo-Metzger Q, Massagli MP, Clarridge BR, et al. Linguistic and cultural barriers to care. J Gen Intern Med. 2003;18(1):44-52.
136. MedlinePlus. Corneal Ulcers and Infections. Available at https://medlineplus.gov/ency/article/001032.htm. Last accessed October 1, 2023.
137. Wipperman JL, Dorsch JN. Evaluation and management of corneal abrasions. Am Fam Physician. 2013;87(2):114-120.
138. Wakai A, Lawrenson JG, Lawrenson AL, et al. Topical non-steroidal anti-inflammatory drugs for analgesia in traumatic corneal abrasions. Cochrane Database Syst Rev. 2017;5:CD009781.
139. Carley F, Carley S. Towards evidence-based emergency medicine: best BETs from the Manchester Royal Infirmary: mydriatics in corneal abrasion. Emerg Med J. 2001;18(4):273.
140. Meek R, Sullivan A, Favilla M, Lamour I, Guastalegname S. Is homatropine 5% effective in reducing pain associated with corneal abrasion when compared with placebo? A randomized controlled trial. Emerg Med Australas. 2010;22(6):507-513.
142. Thiel B, Sarau A, Ng D. Efficacy of topical analgesics in pain control for corneal abrasions: a systematic review. Cureus. 2017;9(3):e1121.
143. Lim CH, Turner A, Lim BX. Patching for corneal abrasion. Cochrane Database Syst Rev. 2016;7:CD004764.
144. Mukherjee P, Sivakumar A, Mackway-Jones K. Tetanus prophylaxis in superficial corneal abrasions. Emerg Med J. 2003;20(1):62-64.
145. Mimura T, Usui T, Yamagami S, et al. Recent causes of subconjunctival hemorrhage. Ophthalmologica. 2010;224(3):133-137.
146. DeRidder CA, Berkowitz CD, Hicks RA, Laskey AL. Subconjunctival hemorrhages in infants and children: a sign of nonaccidental trauma. Pediatr Emerg Care. 2013;29(2):222-226.
148. Ing EB. Eyelid Laceration Treatment and Management. Available at https://emedicine.medscape.com/article/1212531-treatment. Last accessed October 1, 2023.
149. Kosoko A, Vu Q, Kosoko-Lasaki O. Chemical ocular burns: a case review. Am J Clin Med. 2009;6(3):41-49.
151. Mackway-Jones K, Marsden J. Ascorbate for alkali burns to the eye. Emerg Med J. 2003;20(5):465-466.
152. Acerra JR. Globe Rupture. Available at https://emedicine.medscape.com/article/798223-overview. Last accessed October 1, 2023.
153. Cain W Jr, Sinskey RM. Detection of anterior chamber leakage with Seidel's test. Arch Ophthalmol. 1981;99(11):2013.
154. Pandya HK. Retinal Detachment. Available at https://emedicine.medscape.com/article/798501-overview. Last accessed October 1, 2023.
155. National Institutes of Health. Visual Impairment, Blindness Cases in U.S. Expected to Double By 2050. Available at https://www.nih.gov/news-events/news-releases/visual-impairment-blindness-cases-us-expected-double-2050. Last accessed October 1, 2023.
156. Boyd K. Strabismus in Children. Available at https://www.aao.org/eye-health/diseases/strabismus-in-children. Last accessed October 1, 2023.
157. American Academy of Ophthalmologists. Pediatric Eye Evaluations PPP 2022. Available at https://www.aao.org/education/preferred-practice-pattern/pediatric-eye-evaluations-ppp-2022. Last accessed October 1, 2023.
158. Gulati S, Andrews CA, Apkarian AO, et al. Effect of gestational age and birth weight on the risk of strabismus among premature infants. JAMA Pediatr. 2014;168(9):850-856.
159. Jones-Jordan L, Wang X, Scherer RW, Mutti DO. Spectacle correction versus no spectacles for prevention of strabismus in hyperopic children. Cochrane Database Syst Rev. 2014;8:CD007738.
160. Boyd K. Amblyopia: What is Lazy Eye? Available at https://www.aao.org/eye-health/diseases/amblyopia-lazy-eye. Last accessed October 1, 2023.
161. Gunton KB. Advances in amblyopia: what have we learned from PEDIG trials? Pediatrics. 2013;131(3):540-547.
162. Pediatric Eye Disease Investigator Group. Randomized trial of treatment of amblyopia in children aged 7 to 17 years. Arch Ophthalmol. 2005;123(4):437-447.
163. American Academy of Ophthalmologists. Amblyopia PPP 2022. Available at https://www.aao.org/education/preferred-practice-pattern/amblyopia-ppp-2022. Last accessed October 1, 2023.
164. Taylor K, Elliott S. Interventions for strabismic amblyopia. Cochrane Database Syst Rev. 2014;7:CD006461.
165. Li T, Shotton K. Conventional occlusion versus pharmacologic penalization for amblyopia. Cochrane Database Syst Rev. 2009;4:CD006460.
166. Repka MX, Kraker RT, Holmes JM, et al. Atropine vs patching for treatment of moderate amblyopia: follow-up at 15 years of age of a randomized clinical trial. JAMA Ophthalmol. 2014;132(7):799-805.
167. Wallace MP, Stewart CE, Moseley MJ, et al. Compliance with occlusion therapy for childhood amblyopia. Invest Ophthalmol Vis Sci. 2013;54(9):6158-6166.
168. Zhang X, Cotch MF, Ryskulova A, et al. Vision health disparities in the United States by race/ethnicity, education, and economic status: findings from two nationally representative surveys. Am J Ophthalmol. 2012;154(6 Suppl):S53-S62.
169. Hong T, Mitchell P, Burlutsky G, Samarawickrama C, Wang JJ. Visual impairment and the incidence of falls and fractures among older people: longitudinal findings from the Blue Mountains Eye Study. Invest Ophthalmol Vis Sci. 2014;55(11):7589-7593.
170. Feinberg JL, Rogers PA, Sokol-McKay D. Age-related eye disease and medication safety. Ann Longterm Care. 2009;17:17-22.
171. Virgili G, Acosta R, Grover LL, Bentley SA, Giacomelli G. Reading aids for adults with low vision. Cochrane Database Syst Rev. 2013;10:CD003303.
172. American Academy of Ophthalmologists. Vision Rehabilitation PPP 2022. Available at https://www.aao.org/education/preferred-practice-pattern/vision-rehabilitation-ppp-2022. Last accessed October 1, 2023.
173. American Academy of Ophthalmologists. Primary Open-Angle Glaucoma PPP 2020. Available at https://www.aao.org/education/preferred-practice-pattern/vision-rehabilitation-ppp-2022. Last accessed October 1, 2023.
174. Riaz Y, de Silva SR, Evans JR. Manual small incision cataract surgery (MSICS) with posterior chamber intraocular lens versus phacoemulsification with posterior chamber intraocular lens for age-related cataract. Cochrane Database Syst Rev. 2013;10:CD008813.
175. Mangione CM, Barry MJ, Nicholson WK. Screening for primary open-angle glaucoma: U.S. Preventive Services Task Force recommendation statement. JAMA. 2022;327(20):1992-1997.
176. Klein R, Klein BE. The prevalence of age-related eye diseases and visual impairment in aging: current estimates. Invest Ophthalmol Vis Sci. 2013;54(14):ORSF5-ORSF13.
177. Lundeen EA, Burke-Conte Z, Rein DB, et al. Prevalence of diabetic retinopathy in the U.S. in 2021. JAMA Ophthalmol. 2023;141(8):747-754.
178. Shaikh Y, Yu F, Coleman AL. Burden of undetected and untreated glaucoma in the United States. Am J Ophthalmol. 2014;158(6):1121-1129.
179. Zhao D, Cho J, Kim MH, Friedman DS, Guallar E. Diabetes, fasting glucose, and the risk of glaucoma: a meta-analysis. Ophthalmology. 2014;158(6):1121-1129.
180. Tehrani S. Gender difference in the pathophysiology and treatment of glaucoma. Curr Eye Res. 2014;6:1-10.
181. Zhao D, Cho J, Kim MH, Guallar E. The association of blood pressure and primary open-angle glaucoma: a meta-analysis. Am J Ophthalmol. 2014;158(3):615-627.
182. Fernandez Lopez E, Karaca EE, Ekici F, Waisbourd M, Spaeth GL. Symptoms reported by patients with varying stages of glaucoma: review of 401 cases. Can J Ophthalmol. 2014;49(5):420-425.
183. Schwartz GG. Eye cancer incidence in U.S. States and access to fluoridated water. Cancer Epidemiol Biomarkers Prev. 2014;23(9):1707-1711.
184. Boland MV, Ervin AM, Friedman DS, et al. Comparative effectiveness of treatments for open-angle glaucoma: a systematic review for the U.S. Preventive Services Task Force. Ann Intern Med. 2013;158(4):271-279.
185. American Glaucoma Society. Position Statement on Marijuana and the Treatment of Glaucoma. Available at https://www.americanglaucomasociety.net/about/statements. Last accessed October 1, 2023.
186. Chou R, Selph S, Blazina I, et al. Screening for Glaucoma in Adults: Updated Evidence Report and Systematic Review for the US Preventive Services Task Force. JAMA. 2022;327(20):1998–2012.
187. National Glaucoma Research. Is There a Connection Between Diet, Eye Pressure and Glaucoma? Available at https://www.brightfocus.org/glaucoma/article/there-connection-between-diet-and-glaucoma. Last accessed October 1, 2023.
188. American Academy of Ophthalmologists. Age-Related Macular Degeneration PPP 2019. Available at https://www.aao.org/education/preferred-practice-pattern/age-related-macular-degeneration-ppp. Last accessed October 1, 2023.
189. Patel M, Smyth E, Chapman PB, et al. Therapeutic implications of the emerging molecular biology of uveal melanoma. Clin Cancer Res. 2011;17(8):2087-2100.
190. Fisher DE, Klein BE, Wong TY, et al. Incidence of age-related macular degeneration in a multi-ethnic United States population: the multi-ethnic study of atherosclerosis. Ophthalmology. 2016;123(6):1297-1308.
191. Luke JJ, Triozzi PL, McKenna KC, et al. Biology of advanced uveal melanoma and next steps for clinical therapeutics. Pigment Cell Melanoma Res. 2015;28(2):135-147.
192. Harbour JW, Chao DL. A molecular revolution in uveal melanoma: implications for patient care and targeted therapy. Ophthalmology. 2014;121(6):1281-1288.
193. Chakravarthy U, Wong TY, Fletcher A, et al. Clinical risk factors for age-related macular degeneration: a systematic review and meta-analysis. BMC Ophthalmol. 2010;10:31.
194. Gehrs KM, Anderson DH, Johnson LV, Hageman GS. Age-related macular degeneration—emerging pathogenetic and therapeutic concepts. Ann Med. 2006;38(7):450-471.
195. Wang X, Jiang C, Zhang Y, et al. Role of lutein supplementation in the management of age-related macular degeneration: meta-analysis of randomized controlled trials. Ophthalmic Res. 2014;52(4):198-205.
196. Solomon SD, Lindsley K, Vedula SS, Krzystolik MG, Hawkins BS. Anti-vascular endothelial growth factor for neovascular age-related macular degeneration. Cochrane Database Syst Rev. 2014;8:CD005139.
197. Browning DJ, Kaiser PK, Rosenfeld PJ, Stewart MW. Aflibercept for age-related macular degeneration: a game-changer or quiet addition? Am J Ophthalmol. 2012;154(2):222-226.
198. Retina Today. Another Anti-VEGF Agent for Treatment of Neovascular AMD. Available at https://retinatoday.com/articles/2015-july-aug/another-anti-vegf-agent-for-treatment-of-neovascular-amd. Last accessed October 1, 2023.
199. Ophthalmology Times. Novel Anti-VEGF Agent Approved in China May Reduce Injection Frequency. Available at https://www.ophthalmologytimes.com/view/novel-anti-vegf-agent-approved-china-may-reduce-injection-frequency. Last accessed October 1, 20230.
200. Field MG, Harbour JW. Recent developments in prognostic and predictive testing in uveal melanoma.Curr Opin Ophthalmol. 2014;25(3):234-239.
201. Glover K, Mishra D, Singh TRR. Epidemiology of ocular manifestations in autoimmune disease.Front Immunol. 2021;12:744396.
202. Fong DS, Aiello LP, Gardner TW, et al. Diabetic retinopathy.Diabetes Care. 2003;26(suppl 1):S99-S102.
203. ACCORD Eye Study Group. Effects of medical therapies on retinopathy progression in type 2 diabetes.N Engl J Med. 2010;363(3):233-244.
204. 204. Chew EY, Davis MD, Danis RP, et al. The effects of medical management on the progression of diabetic retinopathy in persons with type 2 diabetes: The Action to Control Cardiovascular Risk in Diabetes Eye Study.Ophthalmology. 2014;121(12):2443-2451.
205. Mohamed Q, Gillies MC, Wong TY. Management of diabetic retinopathy: a systematic review.JAMA. 2007;298(8):902-916.
206. American Optometric Association. Clinical Practice Guidelines: Evidence-Based Clinical Practice Guideline Eye Care of the Patient with Diabetes Mellitus. Available at https://aoa.uberflip.com/i/374890-evidence-based-clinical-practice-guideline-diabetes-mellitus. Last accessed October 1, 2023.
207. Fraser-Bell S, Symes R, Vaze A. Hypertensive eye disease: a review.Clin Exp Ophthalmol. 2017;45:45-53.
208. Baker CW, Glassman AR, Beaulieu WT, et al. Effect of initial management with aflibercept vs laser photocoagulation vs observation on vision loss among patients with diabetic macular edema involving the center of the macula and good visual acuity: a randomized clinical trial. JAMA. 2019;321:1880-1894.
209. Leasher JL, Bourne RRA, Flaxman SR, et al. Global estimates on the number of people blind or visually impaired by diabetic retinopathy: a meta-analysis from 1990 to 2010.Diabetes Care. 2016;39:1643-1649.
210. Stewart MW. Critical appraisal of ranibizumab in the treatment of diabetic macular edema.Clin Ophthalmol. 2013;7:1257-1267.
211. International Diabetes Federation. Diabetes Facts and Figures. Available at https://www.idf.org/aboutdiabetes/what-is-diabetes/facts-figures.html. Last accessed October 1, 2023.
212. Jampol LM, Glassman AR, Sun J. Evaluation and care of patients with diabetic retinopathy.N Engl J Med. 2020;382:1629-1637.
213. American Academy of Ophthalmologists. Diabetic Retinopathy PPP 2019. Available at https://www.aao.org/education/preferred-practice-pattern/diabetic-retinopathy-ppp. Last accessed October 1, 2023.
214. Virgili G, Parravano M, Evans JR, Gordon I, Lucenteforte E. Anti-vascular endothelial growth factor for diabetic macular oedema: a network meta-analysis.Cochrane Database Syst Rev. 2017;6:CD007419.
215. Lexicomp Online. Available at https://online.lexi.com. Last accessed October 1, 2023.
216. 216. Mavrikakis E. Macular Edema in Diabetes. Available at https://emedicine.medscape.com/article/1224138-overview. Last accessed October 1, 2023.
217. Zwarenstein M, Shiller SK, Croxford R. Printed educational messages aimed at family practitioners fail to increase retinal screening among their patients with diabetes: a pragmatic cluster randomized controlled trial [ISRCTN72772651].Implement Sci. 2014;9:87.
219. Wong TY, Klein R, Sharrett AR, et al. The prevalence and risk factors of retinal microvascular abnormalities in older people: the Cardiovascular Health Study.Ophthalmology. 2003;110(4):658-666.
221. Sobolewska B, Chee SP, Zaguia F, et al. Vitreoretinal lymphoma.Cancers (Basel). 2021;13(16):3921.
222. Goldberg DE, Smithen LM, Angelilli A, Freeman WR. HIV-associated retinopathy in the HAART era.Retina. 2005;25(5):633-649.
223. Shi Y, Jia R, Fan X. Ocular basal cell carcinoma: a brief literature review of clinical diagnosis and treatment.Onco Targets Ther. 2017;10:2483-2489.
224. BaşZ, Sharpe J, Yaghy A, et al. Prevalence of and associated factors for eyelid cancer in the American Academy of Ophthalmology Intelligent Research in Sight Registry.Ophthalmol Sci. 2022;3(1):100227.
225. National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology: Basal Cell Skin Cancer Version 2.2024. Available at https://www.nccn.org/professionals/physician_gls/pdf/nmsc.pdf. Last accessed October 1, 2023.
226. Lai TY, Wong RL, Luk FO, et al. Ophthalmic manifestations and risk factors for mortality of HIV patients in the post-highly active anti-retroviral therapy era.Clin Experiment Ophthalmol. 2011;39(2):99-104.
227. Prabhakaran VC, Gupta A, Huilgol SC, Selva D. Basal cell carcinoma of the eyelids.Compr Ophthalmol Update. 2007;8(1):1-14.
228. Panel on Opportunistic Infections in HIV-Infected Adults and Adolescents. Guidelines for the Prevention and Treatment of Opportunistic Infections in Adults and Adolescents with HIV. Available at https://clinicalinfo.hiv.gov/en/guidelines/hiv-clinical-guidelines-adult-and-adolescent-opportunistic-infections/introduction. Last accessed October 1, 2023.
1. American Optometric Association.Comprehensive Adult Eye and Vision Examination. St. Louis, MO: American Optometric Association; 2015. Available at https://www.aoa.org/AOA/Documents/Practice%20Management/Clinical%20Guidelines/EBO%20Guidelines/Comprehensive%20Adult%20Eye%20and%20Vision%20Exam.pdf. Last accessed November 29, 2023.
2. Wippold FJ II, Cornelius RS, Berger KL, et al.ACR Appropriateness Criteria: Orbits, Vision, and Visual Loss. Reston, VA: American College of Radiology; 2017. Available at https://acsearch.acr.org/docs/69486/Narrative. Last accessed November 29, 2023.
3. Cruz OA, Repka MX, Hercinovic A, et al. Amblyo6420pia preferred practice pattern.Ophthalmology. 2023;130:136-178. Available at https://www.aaojournal.org/article/S0161-6420(22)00865-X/fulltext. Last accessed November 29, 2023.
4. American Optometric Association.Evidence-Based Clinical Practice Guideline: Eye Care of the Patient with Diabetes Mellitus. 2nd ed. St. Louis, MO: American Optometric Association; 2019. Available at https://aoa.uberflip.com/i/1183026-evidence-based-clinical-practice-guideline-eye-care-of-the-patient-with-diabetes-mellitus-second-edition/1?m4=. Last accessed November 29, 2023.
Mention of commercial products does not indicate endorsement.