This course will begin with a discussion of the electrophysiology of ventricular arrhythmias, including premature ventricular contractions, ventricular tachycardia, and ventricular fibrillation. Related pathophysiology, including the impact of ventricular arrhythmias on cardiac output, signs and symptoms, and underlying diseases will be discussed. Guidelines for the immediate management of life-threatening ventricular arrhythmias in the hospital and community setting will be explained with emphasis on the use of the automated external defibrillator (AED) and current advanced cardiac life support (ACLS) guidelines. Current therapies for the long-term management of ventricular arrhythmias, including ICD therapy, the use of antiarrhythmic medications, the role of electrophysiology, the use of radiofrequency ablation, and surgical procedures designed to eliminate ventricular arrhythmias will be presented. The module will conclude with simulated case studies.
- INTRODUCTION
- BASIC CONCEPTS: A REVIEW
- PREMATURE VENTRICULAR CONTRACTIONS
- VENTRICULAR TACHYCARDIAS
- VENTRICULAR FIBRILLATION
- OVERVIEW: AMERICAN HEART ASSOCIATION "CHAIN OF SURVIVAL"
- EMERGENCY MANAGEMENT OF VENTRICULAR ARRHYTHMIAS
- LONG-TERM MANAGEMENT OF VENTRICULAR ARRHYTHMIAS
- MANAGEMENT OF PVCs AND NONSUSTAINED VT
- IMPLANTABLE CARDIOVERTER-DEFIBRILLATOR THERAPY
- MANAGING WITH AN ICD AT HOME
- LONG-TERM MANAGEMENT OF ICD FUNCTION
- ANTIARRHYTHMIC DRUG THERAPY
- ABLATION OF VENTRICULAR TACHYCARDIA
- OPTIMIZATION OF UNDERLYING DISEASE MANAGEMENT
- SURGICAL RECONSTRUCTION
- TRENDS AND ISSUES IN THE MANAGEMENT OF VENTRICULAR ARRHYTHMIAS
- SIMULATED CASE STUDIES
- CONCLUSION
- GLOSSARY OF TERMS
- RESOURCES
- Works Cited
- Evidence-Based Practice Recommendations Citations
This course is designed for physicians, physician assistants, nurse practitioners, and nurses seeking to enhance their knowledge of ventricular arrhythmias. The course is of particular importance for clinicians in the primary care and emergency settings.
The purpose of this course is to provide physicians, nurses, and other healthcare professionals with up-to-date knowledge of risk factors for development of ventricular arrhythmias, recommended therapies for the immediate and long-term management of arrhythmias, and indications of complications or side effects of therapy necessary to facilitate effective patient management, early identification of problems, and appropriate patient and family education.
Upon completion of this course, you should be able to:
- Define basic concepts of normal cardiac conduction, including cellular events, and normal electrocardiogram (ECG) waveform.
- Identify premature ventricular complexes (PVCs), including electrophysiologic characteristics, patterns of occurrence, related symptoms and common precipitating causes.
- Describe monomorphic ventricular tachycardia (VT), including electrophysiologic characteristics, impact on physiologic functioning, and underlying pathophysiology.
- Identify polymorphic VT, including electrophysiologic characteristics, impact on physiologic functioning, and underlying pathophysiology.
- Describe ventricular fibrillation, including electrophysio-logic characteristics, impact on physiologic functioning, and underlying pathophysiology.
- List key points in the American Heart Association's Chain of Survival, including the use of automated external defibrillators (AEDs).
- Summarize key points in the emergency management of life-threatening ventricular arrhythmias for patients, as recommended by Advanced Cardiovascular Life Support (ACLS) guidelines.
- Outline the assessment, diagnosis, and risk stratification process for patients who have, or are deemed to be at risk for, ventricular arrhythmias.
- Describe the management of PVCs and nonsustained VT in persons with and without heart disease.
- State major indications for implantable cardioverter-defibrillators (ICDs) and outline the basic components, functions, therapy options, and implantation procedure for current generation ICDs.
- Discuss basic discharge education, home safety measures, psychosocial considerations, and medical follow-up for a person post-ICD implantation.
- Evaluate problems that may occur with ICD functioning, including ICD storm, delivery of inappropriate shocks, and failure to deliver therapy.
- Describe the use of antiarrhythmic medications in the long-term management of ventricular arrhythmias.
- Describe additional therapies that may be used in the management of ventricular arrhythmias, including the optimization of medical therapy for underlying heart disease and the use of ventricular reconstruction surgery.
- Analyze current trends and issues in the management of persons who have, or are at risk for, ventricular arrhythmias.
- Using simulated clinical data, discuss the clinical management of a person with ischemic cardiomyopathy and nonsustained VT, and the clinical management of a person who experiences multiple ICD shocks.
Karen Majorowicz, RN, is currently employed in the Cardiac Intermediate Care Unit at Shands Healthcare at the University of Florida, Gainesville. She received her Master's in Medical-Surgical Nursing in 1978 from the University of Maryland. Karen has created numerous instructional manuals on Medicare and has conducted educational programs on cardiovascular assessment.
Contributing faculty, Karen Majorowicz, RN, has disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
John M. Leonard, MD
Mary Franks, MSN, APRN, FNP-C
The division planners have disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
Sarah Campbell
The Director of Development and Academic Affairs has disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
The purpose of NetCE is to provide challenging curricula to assist healthcare professionals to raise their levels of expertise while fulfilling their continuing education requirements, thereby improving the quality of healthcare.
Our contributing faculty members have taken care to ensure that the information and recommendations are accurate and compatible with the standards generally accepted at the time of publication. The publisher disclaims any liability, loss or damage incurred as a consequence, directly or indirectly, of the use and application of any of the contents. Participants are cautioned about the potential risk of using limited knowledge when integrating new techniques into practice.
It is the policy of NetCE not to accept commercial support. Furthermore, commercial interests are prohibited from distributing or providing access to this activity to learners.
Supported browsers for Windows include Microsoft Internet Explorer 9.0 and up, Mozilla Firefox 3.0 and up, Opera 9.0 and up, and Google Chrome. Supported browsers for Macintosh include Safari, Mozilla Firefox 3.0 and up, Opera 9.0 and up, and Google Chrome. Other operating systems and browsers that include complete implementations of ECMAScript edition 3 and CSS 2.0 may work, but are not supported. Supported browsers must utilize the TLS encryption protocol v1.1 or v1.2 in order to connect to pages that require a secured HTTPS connection. TLS v1.0 is not supported.
The role of implicit biases on healthcare outcomes has become a concern, as there is some evidence that implicit biases contribute to health disparities, professionals' attitudes toward and interactions with patients, quality of care, diagnoses, and treatment decisions. This may produce differences in help-seeking, diagnoses, and ultimately treatments and interventions. Implicit biases may also unwittingly produce professional behaviors, attitudes, and interactions that reduce patients' trust and comfort with their provider, leading to earlier termination of visits and/or reduced adherence and follow-up. Disadvantaged groups are marginalized in the healthcare system and vulnerable on multiple levels; health professionals' implicit biases can further exacerbate these existing disadvantages.
Interventions or strategies designed to reduce implicit bias may be categorized as change-based or control-based. Change-based interventions focus on reducing or changing cognitive associations underlying implicit biases. These interventions might include challenging stereotypes. Conversely, control-based interventions involve reducing the effects of the implicit bias on the individual's behaviors. These strategies include increasing awareness of biased thoughts and responses. The two types of interventions are not mutually exclusive and may be used synergistically.
#90374: Clinical Management of Ventricular Arrhythmias
A large number of adults require medical treatment for the management of ventricular arrhythmias. Many of these adults have ischemic heart disease and have survived at least one myocardial infarction (MI). A connection between angina and sudden cardiac death was first recognized in the 18th century; by the mid-19th century, researchers had identified a link between ventricular arrhythmias and acute coronary blockage. Clinical experience has shown that ventricular arrhythmias may range from asymptomatic premature ventricular complexes (PVCs) to life-threatening ventricular tachycardia (VT) and ventricular fibrillation. Although ventricular arrhythmias can occur in persons with "normal" hearts, the risk of life-threatening ventricular arrhythmias and sudden cardiac death increases in persons with ischemic heart disease and various forms of cardiomyopathy. Researchers have focused on identification of the most effective therapies for the prevention of sudden cardiac death from life-threatening ventricular arrhythmias. The data from multiple, large international clinical trials have resulted in significant changes in recommended medical management [1,2,3,4,5,6]. Consider these simulated patient scenarios based on historical data:
In the 1970s, Patient A is admitted to the hospital and undergoes triple vessel coronary artery bypass grafting (CABG). Postoperatively, his course is complicated by frequent episodes of ventricular arrhythmias. To control the arrhythmias, Patient A is placed on oral procainamide and quinidine. Six weeks after surgery, Patient A is discharged home on significant doses of both oral procainamide and oral quinidine. One week after his return home, his wife finds him collapsed on the bedroom floor. On arrival to the home, emergency medical services (EMS) find him in ventricular fibrillation but is unable to resuscitate him. Patient A is pronounced dead on arrival at a local hospital.
In the late 1980s, Patient B is admitted to the hospital for management of recurrent nonsustained VT. He has a history of coronary artery disease and MI. An electrophysiology test is performed to guide the selection and dosage of antiarrhythmic drug therapy. Following initiation of drug therapy, Patient B undergoes serial electrophysiology testing to evaluate the efficacy of the drug therapy in reducing his risk of sudden cardiac death. Despite optimal drug therapy, electrophysiology testing shows that Patient B remains at high risk for sudden cardiac death. The decision is made to insert an implantable cardioverter defibrillator (ICD). Patient B is taken to the operating room, where a thoracotomy is performed to allow placement of the ICD's leads. The ICD generator is implanted in a "pocket" in Patient B's abdomen. Following surgery, the patient develops some atelectasis that requires aggressive pulmonary treatment to resolve.
In 2002, Patient C is exercising in a local health club when he suddenly collapses. One health club employee, following the club's policy and procedure, establishes unresponsiveness while a second employee notifies EMS and obtains the club's automated external defibrillator (AED) located in the exercise room. The employees initiate a defined protocol established by the medical authority for the health club, and attach the portable defibrillator to Patient C. The AED advises that the patient should receive a defibrillatory shock, and the shock is administered. EMS arrives, and Patient C is successfully resuscitated. He is admitted to a local hospital for diagnosis and evaluation of his sudden cardiac arrest episode.
In 2008, Patient D experiences nagging chest pain at home that is not relieved by her sublingual nitroglycerin. Family members summon EMS, and Patient D is rushed to a local emergency room. Her cardiac enzymes and electrocardiogram (ECG) are both positive for a severe ST-segment elevation myocardial infarction (STEMI). Patient D is taken emergently to the cardiac catheterization lab, where she is placed on an intra-aortic balloon pump and undergoes percutaneous coronary interventions including balloon angioplasty and placement of two stents in her left anterior descending coronary artery. Her recovery is uneventful, and she is discharged home. Subsequent evaluation shows that the MI damaged her left ventricle and has significantly impaired the left ventricle's pumping ability. Her ejection fraction (EF) is approximately 25%. During an admission for congestive heart failure, Patient D spontaneously develops sustained VT. The arrhythmia is terminated with IV amiodarone. Because of Patient D's cardiac history and low EF, the decision is made to implant a biventricular pacer-cardioverter defibrillator.
As illustrated by these simulated case studies, antiarrhythmic medications were initially the mainstay of management for ventricular arrhythmias. Serial electrophysiology testing was performed to guide the selection and to evaluate the effectiveness of the medications prescribed. Implantable defibrillators, first developed under the trade name of automatic internal cardio-defibrillators, were first approved for use in the 1980s. Early ICDs required general anesthesia and a surgical procedure for implantation; associated morbidity and mortality were significant, and the type of therapy that the device could provide was limited to the delivery of a high-energy shock. Since the 1980s, a number of changes have occurred. To begin with, there has been a significant increase in the number of patients requiring treatment for the chronic management of ventricular arrhythmias. It is thought that this increase is related to the improved survival rate for persons suffering from coronary artery disease and acute MI. Advances in the management of coronary artery disease, including the availability of procedures for revascularization and restoration of blood flow to the myocardium, have reduced mortality but increased the number of people who experience complications from myocardial damage. Common complications post-MI include congestive heart failure, ischemic cardiomyopathy, and ventricular arrhythmias. In addition, rapid advances in technology have resulted in major changes in implantable defibrillators. Current generation ICDs are smaller, easier to implant, and generally do not require a thoracotomy procedure or general anesthesia for implantation. ICDs have evolved from a device capable only of delivering an electrical shock to the heart in response to identified ventricular fibrillation/VT to extremely sophisticated dual-chamber devices capable of providing a range of therapy for the management of both ventricular and atrial arrhythmias. They also have been combined with biventricular pacemakers to provide not only management of ventricular arrhythmias and antibradycardia pacing but synchronized pacing of the ventricles as well. Also, as the need for clinical data to direct the selection of therapy for persons with ventricular arrhythmias has increased, many large clinical trials have been initiated. Data from some of those trials has shown that Class IA antiarrhythmic drugs, once commonly used in the management of arrhythmias, are associated with an increased risk of death and should not be routinely used. ICDs, once reserved for persons who had survived one or two sudden cardiac arrest episodes, have been identified as the treatment of choice for secondary prevention in certain groups of high-risk patients [1,7,8,9].
Finally, new and/or revised guidelines have been developed to provide evidence-based recommendations that impact the management of persons with ventricular arrhythmias. These guidelines include [4,5,6,10]:
The American Heart Association (AHA) Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care
The ACC/AHA/Heart Rhythm Society (HRS) 2012 Guidelines for Device-Based Therapy of Cardiac Rhythm Abnormalities
The European Society of Cardiology (ESC) 2022 Guidelines for the Management of Patients With Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death
The American College of Cardiology (ACC)/AHA/HRS 2017 Guidelines for Management of Patients With Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death
Each set of guidelines organizes the recommendations into classifications based on the strength of the supporting evidence available. In brief, Class I procedure/treatment recommendations are supported by evidence and/or general agreement that the recommendation is beneficial, useful, and effective. Class II recommendations are those that have conflicting evidence and/or a difference in expert opinion about the usefulness and efficacy. Class III recommendations include treatments/procedures that are not recommended or that may be harmful [5,6,10].
Before looking specifically at the major types of ventricular arrhythmias, their underlying pathophysiology and their impact on cardiac output, a brief review of the basic concepts of normal electrical conduction and cardiac output is necessary.
Some readers may consider this section an extended review, but it provides an introduction to the management of patients with ventricular arrhythmias.
In the normal heart, the heart beat is initiated by the sinoatrial (SA) node. From the SA node, the electrical impulse travels through both right and left atria, causing depolarization of the atria. Atrial depolarization is followed by atrial contraction and atrial repolarization. The electrical impulse travels from the atria to the atrioventricular (AV) node located in the inferior wall of the right atrium. The speed of conduction slows in the AV node to allow time for the atria to depolarize, contract, and complete ventricular filling. From the AV node, the electrical impulse travels through the bundle of His located in the septum of the heart between the right and left ventricles. The bundle of His divides into the right and left bundle branches. These branches divide further into the smaller fibers of the Purkinje system. Electrical conduction through the His-Purkinje system is rapid, causing rapid depolarization of both right and left ventricles. Depolarization of the ventricular cells spreads from the apex of each ventricle to the base and moves from the inside layer of the heart (i.e., endocardium) to the outer (i.e., epicardium). Ventricular depolarization is followed by ventricular contraction and ventricular repolarization [11].
When an electrical impulse stimulates a cardiac cell, a series of events is initiated that cause the cell to depolarize and repolarize. This generates an action potential that allows the electrical impulse to propagate, ultimately resulting in the contraction of the cells of the myocardium. The basic events that occur during the formation of the action potential are as follows [12]:
Phase 0: When an electrical impulse stimulates a cardiac cell, the cell depolarizes. Positively charged sodium ions from the extracellular space flood rapidly into the intracellular space. This increases the total number of positively charged ions in the intracellular space, and the charge in the intracellular space becomes less negative. The potential or voltage in the cell increases.
Phase 1: The flood of sodium ions into the intracellular space stops very quickly; it is followed by a brief and incomplete period of repolarization. This period is mediated by a temporary movement of potassium ions from the intracellular to the extracellular space.
Phase 2: This phase is characterized by a balance of inward and outward movement of ions. Calcium ions move slowly through select channels into the intracellular space while potassium ions move out through multiple channels into the extracellular space. This initiates a slow repolarization, and creates a plateau in the action potential. Cardiac contraction is mediated by phase 2.
Phase 3: The calcium channels close. The process of repolarization is accelerated.
Phase 4: Electrical diastole occurs. Except for the SA node, the heart rests. The SA node begins the process of initiating the next electrical impulse.
After the myocardial cell has depolarized, there is a period of time that the cell cannot generate an action potential in response to another electrical impulse; this is referred to as the "absolute refractory period." As the cell continues to repolarize, an "effective refractory period" occurs in which the cell can transiently depolarize in response to an electrical impulse but generally will not develop enough of an action potential to propagate the impulse to surrounding cells. As repolarization nears completion, the cell is said to be in a "relative refractory period;" in this period, a strong electrical stimulus can trigger the cell to depolarize and create another action potential [13,14].
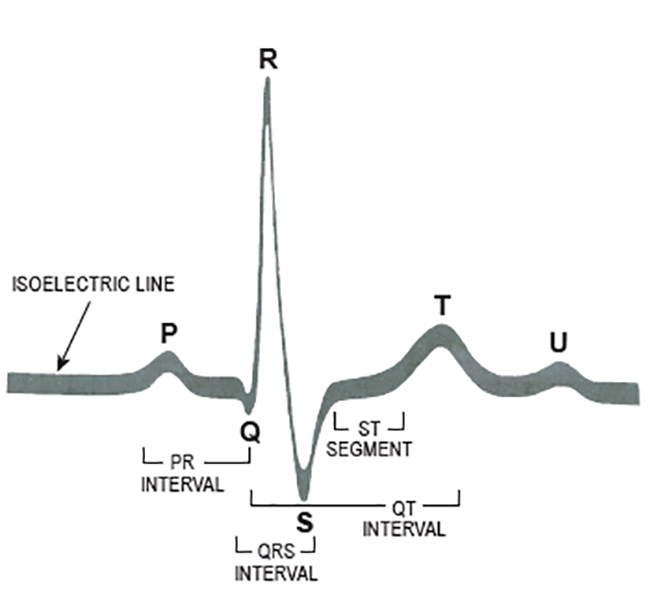
The electrical events that occur in the heart are reflected in the ECG waveform. Components of a normal beat are illustrated in Figure 1[15].
The P wave represents atrial depolarization.
The PR interval represents the amount of time the electrical impulse takes to travel from the SA node through the AV node. The normal PR interval is 0.12 to 0.20 seconds.
The QRS represents the amount of time it takes the ventricles to depolarize. In normal conduction, ventricular depolarization occurs rapidly; this rapid conduction is reflected in a narrow QRS interval. The normal duration of a QRS interval is <0.10 seconds.
The T wave represents ventricular repolarization.
The QT interval represents the amount of time that it takes the ventricles to depolarize and repolarize; it is measured from the beginning of ventricular depolar-ization (i.e., the start of the QRS complex) to the end of repolarization (i.e., the end of the T wave). During the early part of the QT interval, the ventricles are completely refractory and unable to respond to another electrical impulse. During the latter part of the interval, the ventricles are only partially refractory and may respond to some impulses but not to others. The normal QT interval is <0.44 seconds.
When changes occur in the normal cardiac cycle, the normal ECG waveform is altered to reflect them. For example, prolonged ventricular repolarization is reflected in a prolonged QT interval. A slowing of conduction from the SA node through the AV node may be reflected in a prolonged PR interval. Abnormal conduction of the electrical impulse through the ventricles results in a QRS that is wider than usual or bizarre in shape. Careful analysis of the changes in a patient's ECG can provide valuable information in the diagnosis and treatment of an arrhythmia [11,16].
For the heart to contract effectively and maintain adequate cardiac output, electrical conduction through the heart must occur normally. Normal conduction at a normal rate:
Results in the coordinated contraction of the atria
Allows the atria time to contract and complete ventricle filling before the ventricles depolarize
Allows adequate time for the ventricles to fill before contracting
Results in coordinated contraction of the right and left ventricles
When an arrhythmia occurs, normal conduction of the electrical impulse is altered and the pattern of depolarization is changed. As a result, the chambers of the heart may not contract normally and cardiac output may drop. When cardiac output is reduced, predictable signs and symptoms occur. These may include [17]:
Hypotension or a drop in blood pressure from the patient's norm
Complaints of dizziness or light-headedness
Syncopal or near syncopal episodes
Drop in oxygen saturation
Decrease in exercise tolerance
Dyspnea
Exacerbation of signs, such as rales and rhonchi, in persons with congestive heart failure
Acute angina attacks in persons with underlying coronary artery disease
How much the cardiac output drops is dependent on the severity of the change in conduction.
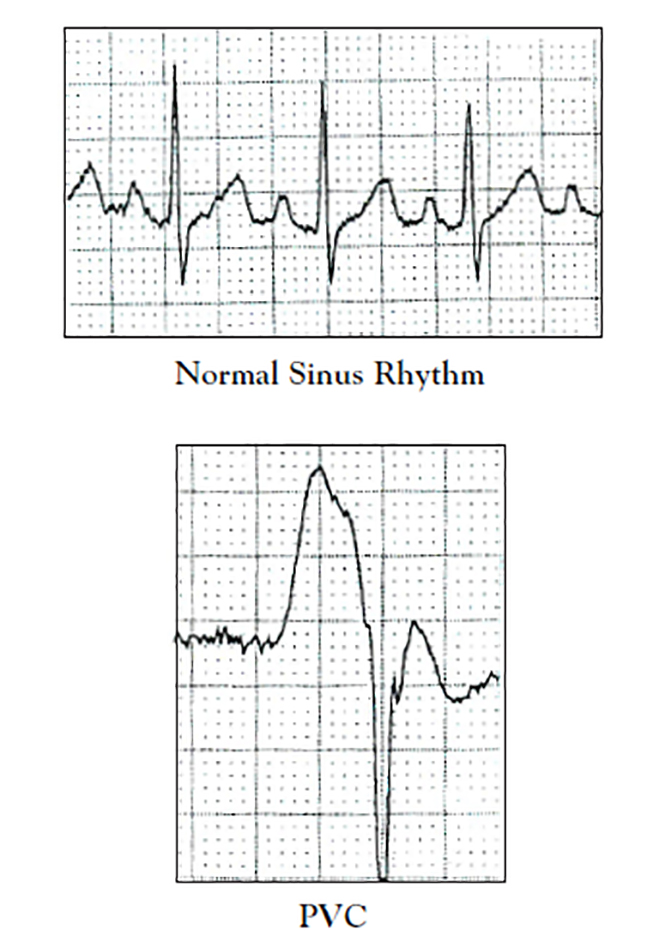
A PVC is an abnormal beat that is initiated at some point in the ventricles of the heart. With PVC, the electrical impulse is not conducted through the ventricles normally. As a result, ventricular depolarization is abnormal and ventricular contraction is impaired. On a surface ECG, the abnormal ventricular depolarization is represented by a QRS complex that is wider and more bizarre in shape than the normal QRS complex. Frequently, PVC occurs earlier in the cardiac cycle than the next normal beat would occur and interrupts the person's underlying heart rhythm. Other names for PVCs include ventricular ectopic beats, ventricular premature depolarizations (VPDs), or ventricular extra systoles. ECG characteristics of PVC include [1,11,12,18]:
Absence of a normal P wave or normal PR interval. As PVC starts in the ventricle and is not initiated by the sinus node, coordinated depolarization and contraction of the atria do not occur to complete ventricular filling. On surface ECG, this is reflected by the absence of a normal P wave and the absence of a normal PR interval.
A widened, bizarre-appearing QRS complex. Normal conduction through the ventricles takes no more than 0.10 seconds; with PVC, conduction takes longer than 0.10 seconds and often lasts 0.14 sec.
In Figure 2, note the absence of a normal sinus P wave and normal PR interval in the PVC; also note the change in appearance and width of the QRS complex from the normal QRS.
PVCs may occur infrequently and in isolation, or they may occur in an identifiable pattern. Terms commonly used to describe PVC patterns of occurrence include [19]:
Isolated: PVCs occur very infrequently with no repeating or identifiable pattern
Bigeminy: A repeating pattern of a normal beat followed by a PVC
Trigeminy: Repeating pattern of two normal beats followed by a PVC
Couplet: Two PVCs in a row
R-on-T: PVC that occurs so early in the cardiac cycle that it falls on the T wave of the preceding beat. Especially in persons experiencing an acute MI or hypokalemia, an R-on-T PVC carries the risk of causing the heart to go into ventricular fibrillation.
PVCs may also be described by their appearance. PVCs that have a similar shape or configuration in the same ECG lead are thought to arise from the same place or "focus" in the ventricles and are referred to as "uniform" or "unifocal" PVCs. PVCs that have different shapes or configurations in the same ECG lead are referred to as "multifocal" or "multiform;" they are thought to arise from different places or foci in the ventricles [11,18,19].
Because PVC alters the normal pattern of conduction and contraction, it may result in a drop in cardiac output for that beat. Isolated PVCs rarely cause symptoms; they occur too infrequently to create a discernible impact on the patient's cardiac output. PVCs that occur more frequently may cause troublesome symptoms related to the decreased cardiac output [1,5,18,20].
In healthy individuals, isolated PVCs may occur at rest or during exertion; these PVCs are usually clinically insignificant and do not require follow-up or treatment. Transient PVCs may occur as a result of excessive caffeine intake, smoking, drinking alcohol, the use of illicit drugs (e.g., cocaine, amphetamines), or the use of over-the-counter medications (e.g., diet pills, antihistamines) that contain ingredients that mimic the effects of sympathetic nervous system stimulation. In individuals with and without heart disease, hypokalemia and hypomagnesemia often contribute to the development of PVCs. On occasion, the abrupt development of PVCs or a marked increase in PVCs may be an indicator of myocardial ischemia [20].
Ventricular tachycardia is a rapid, abnormal rhythm that originates in the ventricles and takes over or usurps the heart's normal rhythm. Ventricular tachycardia can occur in persons with normal hearts but is more likely to occur in the presence of ischemic or nonischemic heart disease. It may also develop as a side effect of certain medications [3,5,11,19,21].
By definition, VT consists of three or more PVCs occurring at a rapid rate of more than 100 beats per minute (bpm) [6,21]. In the past, VT was thought to be a life-threatening arrhythmia with relatively uniform characteristics. There are different types of VT that may vary greatly in the following characteristics:
Duration
Appearance of the QRS complexes
Underlying mechanism
Site or origin of the VT
Impact on cardiac output
VT may be classified by clinical presentation, duration, or shape and configuration of the QRS complex[5]. In terms of clinical presentation, VT may be described as stable or unstable. In hemodynamically stable VT, the patient is asymptomatic or experiences only minimal mild symptoms, such as racing heartbeat, palpitations, feeling skipped beats, or awareness of heart beats. In hemodynamically unstable VT, the patient may report symptoms ranging from dizziness, lightheadedness, or feeling faint to more severe symptoms of syncope, sudden loss of consciousness, and sudden cardiac death.
When classified according to duration, VT may be described as sustained or nonsustained. Nonsustained VT is defined as VT that lasts less than 30 seconds and spontaneously terminates. Sustained VT lasts longer than 30 seconds and requires intervention to terminate or is so unstable that immediate intervention should be employed[5,22].
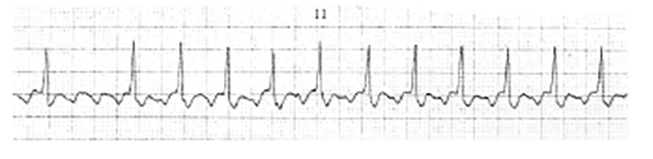
Shape and configuration of the QRS complex may also be used to classify VT. Using these criteria, VT may be described as monomorphic or polymorphic [22]. Bidirectional VT (Figure 3) is a rare form of ventricular arrhythmia, characterized by a beat-to-beat alternation in the QRS frontal plane axis and a heart rate of 140–180 bpm. Bidirectional VT is most commonly associated with severe digoxin toxicity [5,23].
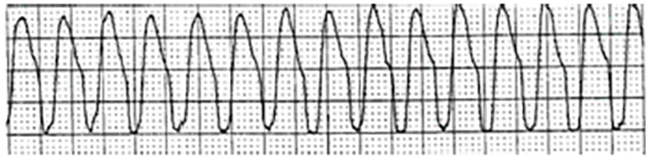
Monomorphic VT is commonly seen in persons with ischemic heart disease. It gets its name from the uniform appearance of the QRS complexes in a single ECG lead. Monomorphic VT may be sustained (duration >30 seconds) or nonsustained (duration <30 seconds) [24].
The defining ECG characteristics of monomorphic VT include:
Absence of normal P waves
Absence of a normal PR interval
A ventricular rate that ranges from 120 bpm to 250 bpm
Regular ventricular rhythm
QRS complexes that are wider than normal and bizarre in appearance
QRS complexes that remain constant in shape and configuration (in the same ECG lead)
Figure 4 is a sample strip of monomorphic VT. Note the absence of P waves, the bizarre QRS complexes, and the rapid ventricular rate [1,3,25].
Ventricular tachycardia is one type of sustained ventricular arrhythmia. Two other sustained ventricular arrhythmias that may develop in the clinical setting are slow idioventricular escape rhythm and accelerated idioventricular escape rhythm. Both are characterized by the absence of P waves and by widened, bizarre QRS complexes. They differ from VT in terms of rate.
Slow idioventricular escape rhythm is a protective rhythm generated by the ventricles in an attempt to produce adequate cardiac output when the normal sinus and AV nodal (junctional) pacemakers have failed. It occurs at a very slow rate of 30 to 40 bpm and is characterized by wide QRS complexes. It may result in a significant drop in cardiac output and generally requires immediate treatment. Unlike the treatment for VT, the treatment for a ventricular escape rhythm does not focus on suppressing the arrhythmia but on increasing the heart rate through pharmacologic means or temporary external pacing.
Accelerated idioventricular rhythm occurs when cells in the bundle branches or the Purkinje fibers in the ventricles act as a pacemaker for the heart. It is characterized by widened QRS complexes that occur at a rate of 40 to 120 bpm; normal sinus P waves are absent. Referred to as slow VT or accelerated ventricular rhythm (AVR), accelerated idioventricular rhythm may be an escape rhythm that occurs when the sinus node fails to generate an effective heart rhythm. As with rapid VT, it may be a usurping rhythm that takes over the pacemaker function from the sinus node [11,19,26].
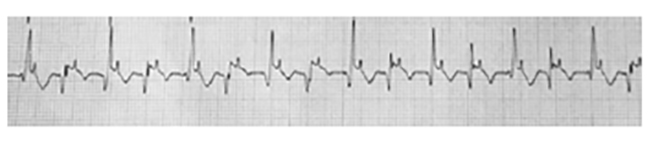
On occasion, differentiating a VT from a supraventricular atrial tachycardia can be extremely difficult. By definition, a supraventricular tachycardia is an abnormal rapid rhythm that is initiated at or above the level of the AV node (but outside the SA node). Conduction through the ventricles is typically normal. This differs from a sinus tachycardia with rapid firing of the SA node. The defining characteristics of a supraventricular tachycardia include a heart rate in excess of 120 bpm, an absence of normal sinus P waves, and narrow QRS complexes. Note the absence of the P waves and the normal configuration of the QRS complexes in Figure 5.
In theory, one of the major differences between the ECG pattern of supraventricular tachycardia and that of VT is the width and appearance of the QRS complexes. However, due to conduction abnormalities present in the bundle branch system, in some cases the QRS complexes in supraventricular tachycardia may be wider than normal and bizarre in appearance. Differentiating a VT from a supraventricular tachycardia may be impossible from a surface 12-lead ECG. Treatment of a wide complex tachycardia that has not been clearly identified as either VT or supraventricular tachycardia is discussed in a later section on emergency management of ventricular arrhythmias [11].
A re-entry mechanism is responsible for most clinical monomorphic VTs. In re-entry, the normal transmission of the electrical impulse is altered in part of the heart. Instead of the wave of depolarization spreading evenly through the heart, it is slowed or blocked in one area while proceeding normally in an adjoining area. The mix of abnormal and normal conduction in adjoining areas of the heart causes electrophysiologic conditions that create an abnormal conduction loop that allows the depolarization to repeatedly and continuously re-excite the heart at rapid rates. The risk that a re-entry tachycardia will develop increases with factors that increase the risk of a PVC, such as electrolyte imbalances, increased sympathetic nervous system activity, recurrent myocardial ischemia, or uncontrolled heart failure [27,28].
Some persons with monomorphic VT will be completely asymptomatic. Others will experience mild-to-moderate symptoms of decreased cardiac output including a drop in blood pressure, a reduction in activity tolerance, dyspnea, and dizziness or light-headedness. Still others will experience severe symptoms of syncope or near syncope, and some will develop cardiac and respiratory arrest. VT may degenerate into ventricular fibrillation. In persons with already compromised left ventricular function, persistent VT may result in signs of increased heart failure, angina, and acute MI. The impact of the VT on the patient is mediated by a number of factors, including [1]:
The rate of the VT
How long the VT lasts
How frequently the VT recurs
The presence and extent of any heart disease present
Ischemic Heart Disease
Sustained monomorphic VT commonly occurs in individuals with ischemic heart disease. According to data from clinical trials, the combination of a healed MI, nonsustained VT, and a reduced left ventricular ejection fraction (LVEF) greatly increases a person's risk for developing prolonged runs of VT [1]. In an MI, the damage to the heart muscle creates the necessary conditions for a re-entry VT to develop. Areas of the myocardium die and are replaced with scar tissue. The resulting mix of normal tissue and abnormal scar tissue create adjoining areas of normal and abnormal conduction that allow a re-entry rhythm to occur [29]. Damage from an MI can also impair the ability of one or both ventricles to pump effectively. When the left ventricle is damaged, its ability to pump blood into the systemic circulation is reduced. LVEF describes how well the left ventricle is functioning. LVEF is defined as the percentage of the total volume of blood contained in the left ventricle that is pumped from the ventricle with each heartbeat. A normal LVEF at rest falls between 50% and 75%. EF may be measured noninvasively through the use of an echocardiogram or nuclear medicine study or invasively during a cardiac catheterization procedure [30]. Although polymorphic VT is more common during the acute stage of an MI, sustained monomorphic VT may develop. Aside from coronary artery disease, causes of myocardial ischemia that have been found to trigger monomorphic VT include coronary vasospasm, cocaine-induced vasospasm, severe hypertension, and aortic stenosis [1,3,18].
Nonischemic Heart Disease
The occurrence of monomorphic VT also occurs in nonischemic heart disease. It is estimated that dilated cardiomyopathy is the second most frequent type of cardiac disease linked to monomorphic VT, the third most common cause of heart failure, and the most frequent reason for heart transplantation [1,3,31]. Dilated cardiomyopathy is a syndrome in which one or both ventricles dilate abnormally, resulting in enlarged, sluggish chamber(s) that contract poorly [32,33]. Cardiac output is compromised. Dilated cardiomyopathy is a progressive disease characterized by a fairly rapid deterioration in ventricular function. As left ventricular function decreases, the risk of serious ventricular arrhythmias increases. Multifocal PVCs, couplets, and runs of nonsustained VT commonly occur. Sustained monomorphic VT may develop from a re-entry circuit involving the bundle of His and the bundle branches. Causes of dilated cardiomyopathy include underlying genetic disorders and abnormalities, viral infection, excessive alcohol consumption over time, pregnancy, and environmental factors [31,32,33]. Another form of nonischemic heart disease, hypertrophic cardiomyopathy, has also been linked with sudden cardiac death episodes and the development of sustained VT. A disorder of the myocardium, it is characterized by hypertrophy or enlargement of the left ventricular muscle [34]. The enlargement occurs without dilatation of the left ventricular chamber and is not associated with any cardiac or systemic cause. The hypertrophy of the left ventricle impairs normal functioning of the heart during diastole, and the development of arrhythmias and ischemia is common. In some persons with hypertrophic cardiomyopathy, the septum between the right and left ventricle also enlarges, causing significant obstruction of blood flow from the left ventricle during systole [34]. Runs of nonsustained VT occur commonly and are thought to be a marker of poor prognosis and increased risk of sudden death in people older than 25 years of age. Another form of cardiomyopathy, arrhythmogenic right ventricular dysplasia (ARVD), has also been linked to ventricular arrhythmias including PVCs, sustained monomorphic VT, and in some cases, ventricular fibrillation. ARVD is a progressive form of cardiomyopathy in which fatty infiltrates invade and damage the right ventricle. Scarring occurs, and the combination of scarred and normal areas creates the conditions for a re-entry circuit and monomorphic VT to develop. ARVD can also damage the left ventricle [1,3,33].
Idiopathic VT
Although monomorphic VT occurs more often in people with heart disease, it may also occur in the absence of heart disease. Two types of idiopathic VT that may be seen in persons with normal hearts are right ventricular outflow tract VT and idiopathic left VT. In right ventricular outflow tract VT, exercise or some other hyperadrenergic state may induce repetitive bursts of nonsustained VT or episodes of sustained monomorphic VT. Unlike the VT seen commonly in ischemic heart disease, the VT in right ventricular outflow tract is thought to be caused by abnormal impulse formation in the ventricle. The ventricular arrhythmias seen in right ventricular outflow tract have not been strongly linked to sudden cardiac death episodes but may create debilitating symptoms of palpitations, dizziness, or syncope [35]. Right ventricular outflow tract in the absence of a family history for cardiomyopathies and without evidence for structural heart disease are suggestive for idiopathic VT [6].
Idiopathic left VT also develops in the absence of obvious heart disease. Its underlying mechanism is thought to be a re-entry circuit involving the left side of the His bundle and bundle branch system. Unlike right ventricular outflow tract VT, idiopathic left VT is not precipitated by exercise. It occurs more commonly in younger men and is associated with symptoms of palpitations and (less commonly) syncope. This form of VT rarely leads to sudden cardiac death. However, if the VT occurs incessantly, it may eventually cause a tachycardia-induced cardiomyopathy [3,35].
The second category of VT is the polymorphic type. Characterized by a normal QRS during sinus rhythm, polymorphic VT most commonly occurs during acute myocardial ischemia or infarct. However, it may also occur in patients with cardiomyopathy and heart failure. It is characterized by QRS complexes that continuously change in configuration from one beat to the next (in the same ECG lead). A particularly severe form of polymorphic VT is torsades de pointes [1,9,36].
Torsades de pointes gets its name from the characteristic pattern of its QRS complexes. As the complexes change from upward to a downward deflection, they give the appearance of "twisting" around the baseline [36]. The amplitude of each successive complex gradually increases then decreases, creating an identifiable spindle-shaped pattern. Other ECG characteristics associated with torsades de pointes include [33,36]:
A rate of 200 to 250 bpm
No P wave or PR interval present
An irregular ventricular rhythm
Torsades de pointes is an unstable form of VT. Episodes are often nonsustained; however, torsades de pointes may quickly degenerate into ventricular fibrillation [36]. Most persons who develop torsades de pointes experience noticeable symptoms of decreased cardiac output, including hypotension or a decrease in blood pressure, decreased activity tolerance, syncope or near-syncope, and dyspnea [1,9,11,18,19,33,36].
Pathophysiology of Torsades de Pointes
The occurrence of torsades de pointes is linked to a prolonged period of repolarization. On surface ECG, prolonged repolarization is reflected by a prolonged QT interval. With a prolonged period of repolarization, the effective refractory period is also prolonged and there is an increased risk that an abnormal electrical impulse arising during this period will trigger a rapid ventricular arrhythmia. Factors that increase the risk that torsades de pointes will develop include hypomagnesemia, hypokalemia, or a pre-existing bradycardia. Prolonged QT intervals may be congenital or acquired [36].
Acquired prolonged QT syndrome is usually caused by administration of one or more medications that prolong action potential duration and refractory period of ventricles. Certain antiarrhythmic medications are well known to prolong the QT interval. Less well known is the fact that a number of noncardiac medications have also been found to prolong QT interval and increase the risk of developing torsades de pointes. Table 1 lists examples of medications known to prolong the QT interval. Administration of multiple medications that prolong the action potential duration and refractory period can produce a cumulative effect on the QT interval and can increase a person's risk for developing torsades de pointes [1,18,36].
DRUGS LINKED TO PROLONGED QT INTERVAL AND/OR TORSADES DE POINTES
| Category | Agents | |||||||
|---|---|---|---|---|---|---|---|---|
| Class I antiarrhythmic medications |
| |||||||
| Class III antiarrhythmic medications |
| |||||||
| Antibiotics |
| |||||||
| Antipsychotic |
| |||||||
| Other |
|
Less common than acquired long QT syndrome, congenital long QT syndrome is linked to several genetic abnormalities [6]. The initial clinical presentation may be syncope or cardiac arrest that is precipitated by exertion, such as vigorous activity or intense emotions [36]. Symptoms may begin as early as preteen or teenage years (average age is 14 years), or they may not develop until the person is 40 or 50 years of age. Persons with congenital long QT syndrome usually have a first-degree family history of syncopal episodes, sudden cardiac arrest, or unexpected death at a young age. Genetic-specific triggers include emotional stress, exercise, and sleep [6]. In younger persons, syncopal symptoms may initially be misdiagnosed as a seizure disorder. Diagnostic evaluation of prolonged QT syndrome may include screening for prolonged QT intervals in other family members and assessment for any episodes of sudden cardiac death or syncopal spells among other family members [36,38].
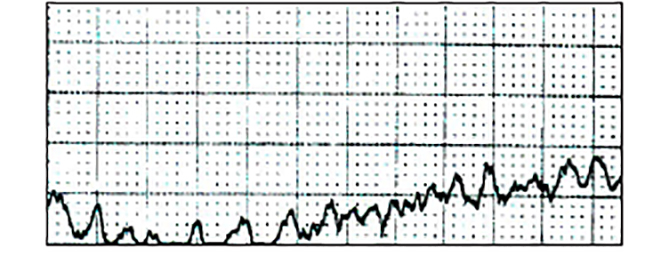
The most lethal and disorganized ventricular arrhythmia is ventricular fibrillation. In ventricular fibrillation, electrical activity is completely chaotic. No effective depolarization occurs, and there is absolutely no effective contraction of the heart. Complete circulatory collapse occurs, and emergency intervention is required [39]. Defining ECG characteristics include:
An absence of any identifiable P, QRS, or T waves
The total absence of any organized rhythm
An indiscernible rate
Figure 6 is an example of ventricular fibrillation. Note the absence of organized, identifiable QRS complexes [1,10,18].
Within 10 to 20 seconds of the onset of ventricular fibrillation, the patient experiences a total loss of blood pressure and pulse and loses consciousness. Respiratory arrest quickly follows. Unless the arrhythmia is quickly terminated and circulation restored, damage to the brain and heart will occur. Ventricular fibrillation that is untreated or that does not respond to treatment results in death [18,39,40].
Coronary artery disease (CAD) is the single most common etiologic factor predisposing patients to ventricular fibrillation [40]. Other common causes include the deterioration of VT and severe bradycardia caused by MI [40]. Ventricular fibrillation may also develop in persons with severe heart failure. Other conditions that are linked to ventricular fibrillation include [1,18,36]:
Multiple electrolyte imbalances that prolong the relative refractory period
Congenital or acquired long QT syndrome
Proarrhythmic effect of medications, especially antiarrhythmic medications
The ventricles are not stimulated to contract due to very rapid, minute impulses from multiple foci.
Lethal arrhythmias can occur in any setting. A person's chance of surviving a sudden death episode with neurologic and cardiac function intact increases when emergency measures are employed rapidly and in an organized fashion. The AHA, based on years of research into the causes and management of sudden cardiac death episodes, has identified a specific sequence of events that has been found to improve survival and outcomes following cardiac arrest. These events have been grouped together in a conceptual model referred to as the Chain of Survival. Although the general principles are applicable to hospital and out-of-hospital settings, a major focus of this chain of survival has been on improving emergency care provided in the community setting and postcardiac arrest care [10,41,42,43].
The first link in the Chain of Survival concept is recognition and activation. In simple terms, this means that a cardiac arrest victim receives help as soon as possible. Two actions are critical: recognition of the emergency and activation of the emergency medical system. Efforts have been made to strengthen the link of early access through increasing public education that targets individuals who are most likely to witness a cardiac arrest in a community or home setting. Research has shown that a person who witnesses a cardiac arrest is more likely to call other people (e.g., friends, neighbors, physicians) before activating the emergency communication system. Consequently, additional public education has been directed toward a "phone first" campaign that instructs people to notify EMS first before making other calls or initiating cardiopulmonary resuscitation (CPR). Other efforts have been directed toward the establishment of an easily dialed emergency dispatch system and toward public education to ensure that people living in any given community are familiar with the local emergency number to call. Healthcare professionals can help to strengthen the link of early access for patients through patient and family education. Pertinent patient/family education may include helping high-risk patients and family members to make plans for emergencies. When to call for emergency help, how to access the emergency medical system in their local area, and identifying an appropriate, easily visible place to post emergency numbers are key points to include. When appropriate, health professionals can encourage family members to enroll in local CPR courses [41,42,43].
The prompt initiation of CPR as soon as a cardiac arrest is recognized is critical [6]. The structured pattern of chest compressions and ventilations can produce adequate blood flow to the brain and the heart until EMS can arrive and initiate more advanced measures. To strengthen this link, the American Heart Association and American Red Cross have implemented training programs for citizens in an attempt to ensure that bystanders who witness a cardiac arrest will know how to perform CPR. Some data collected suggest that mass training of the civilian population to perform CPR may not be the most effective, because many people who choose to attend training will never see a cardiac arrest. Alternative suggestions for increasing the availability of trained lay rescuers include [6,41,42,43]:
Targeting persons or groups of persons who are most likely to need to perform CPR. Because many victims of sudden cardiac death are middle-aged adults and older, targeted groups would include persons who, as part of their routine family surroundings, work, or social life, are frequently in contact with middle to older age adults. Examples of targeted groups include staff in senior centers and family members and friends of persons who are at risk for cardiac arrest. Training of personnel in gyms or "workout" facilities would be another example of targeted group education.
Training EMS dispatchers to give instructions for hands-only CPR (i.e., compression only) over the telephone. Hands-only CPR is substantially easier for EMS dispatchers to instruct and shows similar survival rates among victims when compared with traditional CPR.
It has been shown that early initiation of CPR is most effective when it has been followed rapidly by defibrillation [43]. The purpose of defibrillation is to restore the heart to a normal, effective rhythm. When the heart is defibrillated, the electrical shock causes the heart cells to completely depolarize. This interrupts the pattern of chaotic electrical activity, represented on the monitor as a brief period of asystole. If defibrillation is effective, normal electrical activity will then resume. Effective defibrillation is partially dependent on the presence of adequate high-energy phosphate stores in the myocardium. If phosphate stores are adequate, the heart will have the necessary energy resources to restart in an effective rhythm. Rapid arrhythmias, such as ventricular fibrillation, consume high-energy stores very quickly; for that reason, it is critical to shock the heart out of ventricular fibrillation before the heart's energy sources are depleted [10,41,42,43].
To provide more rapid defibrillation in an out-of-hospital setting, public access defibrillation programs have been established. In a public access defibrillation program, a trained lay rescuer uses an AED to analyze the patient's rhythm and to deliver a defibrillatory shock if indicated. An AED is reasonably simple to operate and does not require the operator to master complex skills of rhythm interpretation. Initially, there was some opposition to the use of AEDs by trained lay rescuers; some believed that defibrillation was an intervention that should only be performed by physicians or physician extenders. However, as the need for early defibrillation became obvious and the efficacy of the AED by trained lay rescuers was documented, opposition to the use of AEDs in the United States decreased [41,44,45]. Despite these gains, fewer than 12% of adults have an AED applied before EMS arrival [43]. Because an AED is considered a "restricted medical device" and its use falls under state regulations governing health practice and use of medical devices, public access defibrillation programs must meet certain regulatory requirements. Although details may vary from state to state, in general, these requirements specify that the public access defibrillation program should be overseen by a medical authority that is responsible for supervising the purchase of the AEDs, establishing the treatment protocols, and the training of individuals who will use the equipment. Another problem in the establishment of a public access defibrillation program was the potential liability to the lay rescuer, the physician overseeing the program, and the business or organization providing the program. To reduce liability and promote use of public access defibrillation programs, all 50 states have passed or amended legislation to address the issue of the use of AEDs [46]. In general under the amended Good Samaritan Acts, lay rescuers may be immune from lawsuits resulting from the use of AEDs if [41,44]:
The lay rescuer has completed an appropriate training program, such as the Heartsaver AED course from the AHA, or a comparable course that has identical knowledge, objectives, and performance requirements.
Treatment protocols are used that are approved by an appropriate, recognized medical authority.
The AED equipment is checked and maintained according to the manufacturer's guidelines.
Local EMS authorities are notified of the placement of the AED.
Obviously, emergency medical technicians and paramedics responding as part of an EMS system should be trained to use an AED in the community setting. In addition, groups identified or labeled as public access defibrillation responders should be trained. These include [44]:
Nontraditional responders. These are people who are not healthcare personnel but who are employed in a setting or an occupation that requires them to respond to emergency situations. Included in this group are police, firefighters, security staff, and airline flight attendants.
Citizen or worksite responders. These may be employees of a company, business, or public facility whose leadership has implemented public access defibrillation programs.
Responders to high-risk individuals. This usually includes friends or family members who live with or visit a person who is deemed to be at high risk for sudden cardiac death.
Data support the need for increased availability of public access defibrillators and community training in basic life support that is initiated in childhood and repetitive [6].
Before moving on, a more in-depth discussion of AEDs and how they function may be helpful.
AEDs come in various shapes and sizes. Those designed for use in public places by trained lay rescuers are relatively small portable units, approximately the size of a laptop computer. The AED consists of several major parts: a main unit, defibrillator cables, and detachable electrode pads. On the outside of the main unit are the power switch, controls for operating the AED, and a screen for visual display. The microcircuitry that analyzes the victim's heart rhythm and initiates a shock for defibrillation, if it is indicated according to the AEDs programmed algorithms, is housed inside the main unit along with the batteries that provide the necessary power. The electrode pads attach to the defibrillation cables and are placed on the victim's body. The pads detect and transmit information about the victim's heart rhythm back to the circuitry in the main unit. They are subsequently available to deliver a shock for defibrillation if indicated. Specific characteristics of individual AEDs will vary by manufacturer, and individuals using AEDs must be familiar with the specific device that they are likely to use. AEDs may also be combined with conventional defibrillators for use by medical personnel in the hospital setting. These defibrillators generally provide a variety of available therapies including automated external defibrillation, standard defibrillation, cardioversion, and in some devices, the availability of external transcutaneous pacing [47,48].
How an AED Works
As noted, an AED is designed to analyze a victim's heart rhythm to determine if a defibrillatory shock is indicated and then to deliver that shock. The electrode pads placed on the patient's chest pick up information about the patient's heart rhythm and transmit it back to the circuitry in the main unit. The AED analyzes the transmitted information, matching the characteristics of the patient's current heart rhythm to its programmed parameters and determines if a defibrillation shock is indicated. After it completes its analysis, the AED then prompts the rescuer to the appropriate action using audible or visual cues. If the AED determines that a shock is indicated, it initiates a "shock advised" message. If the AED determines that a shock is not indicated, it initiates a "no shock, check pulse" message instead[48]. The use of an AED involves a few basic steps. These steps are discussed here to provide a review or an overview for the reader. However, for rescuers to learn how to actually use an AED requires a structured training program that includes hands-on practice as well as classroom lecture and discussion material. Appropriate courses are offered through AHA locations or local chapters of the American Red Cross. For specific information about available training programs, consult the local chapter or access information available on the websites of the AHA or the American Red Cross (Resources).
Basic Steps in AED Operation
If an AED is available, it should be obtained immediately after the rescuer has determined that the victim is unresponsive and the emergency medical system has been activated. Some state legislation governing the use of AEDs requires that the rescuer notify EMS that an AED is available at the site. To use the AED, the rescuer first turns the device on, then attaches the electrodes to the victim in the designated locations. Diagrams showing proper electrode placement are printed on many of the electrode packages to guide the rescuer in determining proper placement. Electrode pads should not be placed directly over implanted pacemaker or ICD sites or over transdermal medication patches. If needed, transdermal patches may be removed and the skin wiped clean before the electrode pads are applied. After the electrodes are attached, the rescuer must make sure that no one is touching the patient. Once the rescuer is sure that the patient is "cleared," the rescuer depresses the "analyze" button on the AED. If prompted that a shock is indicated, the rescuer again verifies that no one is touching the patient and depresses the "shock" button [41,44]. According to CPR guidelines, delivery of the shock should be followed immediately by chest compressions [10,43].
Safety Measures
Despite its simplicity, an AED is a powerful tool. Used correctly, it can promote survival and facilitate resuscitation efforts. Used incorrectly, it can result in harm to the victim or to the rescuers attempting use of the equipment. Specific safety measures should be employed, including [10,41,43,44]:
Adequate training. Most state regulations limit use of an AED to persons who have completed a prescribed training program that meets specified content and performance criteria. The "criterion standard" is the AHA course. Other programs are permissible but must meet the same training and performance objectives as the AHA course.
Routine equipment checks. As specified by the manufacturer, routine maintenance checks of the AED should be performed and documented. Some recommended equipment checks are designed to be performed by the individuals who will be using the equipment. Lay rescuers should become familiar with the specific AED provided at their location and review and practice troubleshooting to improve performance during an actual emergency situation.
Use of age-appropriate equipment. Although not common, ventricular fibrillation can occur in children. Many AEDs are able to distinguish "shockable" from "nonshockable" rhythms and can identify ventricular fibrillation in children. Use of pediatric-specific equipment (i.e., pediatric attenuator system) to modify the delivered dose is recommended for children between 1 and 8 years of age. However, if the rescuer does not have an AED with a pediatric attenuator system, a standard AED may be used. According to CPR guidelines, a manual defibrillator may be used on and is preferred for infants younger than 1 year of age.
Additional safety measures. Unresponsive victims who are lying in water should be moved to a dry area, and their chests briskly wiped before electrodes are applied. For a patient whose chest area is drenched in water or who is extremely diaphoretic, the chest should also be briskly wiped before patches are applied.
The advanced care life support link addresses advanced airway skills, including different forms of intubation, and the use of intravenous medications. To assist medical personnel in implementing this link in an organized, orderly, effective manner, advanced cardiovascular life support (ACLS) guidelines have been developed for different emergency situations. For detailed information about ACLS guidelines, algorithms, and protocols, please consult your local AHA chapter or the AHA website [10,41,43].
The final link in the Chain of Survival concept is integrated post-cardiac arrest care. Post-cardiac arrest care can significantly reduce morbidity and mortality due to hemodynamic instability, brain injury, and/or multi-organ failure. The key objectives of integrated post-cardiac arrest care include [10,43]:
Ensure that cardiopulmonary function and vital organ perfusion have been optimized.
Transport the victim to a hospital or critical care unit with a post-cardiac arrest treatment system.
Prevent a recurrent arrest by identifying and treating the cause of the victim's current arrest.
Control the victim's body temperature for optimal survival and neurologic recovery.
Anticipate, treat, and prevent multi-organ dysfunction.
Patients experiencing ventricular arrhythmias may present in a multitude of ways. Consider these clinical scenarios:
Patient E collapses unexpectedly in the bathroom of her hospital room. A quick assessment by the nursing staff shows that she has no palpable pulse or spontaneous respiration. When she is attached to a cardiac monitor, the monitor shows that she is in ventricular fibrillation.
Patient F is ambulating in the halls when the central telemetry monitoring station notes that he has gone into monomorphic VT. Patient F remains alert and oriented and denies symptoms. Despite sustained monomorphic VT, his vital signs remain essentially unchanged from his baseline and he continues to deny any symptoms of dizziness, dyspnea, or light-headedness.
Patient G comes to the clinic for a scheduled appointment. She tells the staff that she made the appointment because she noticed that her heartbeat sometimes becomes very irregular and seems to skip beats. A 12-lead ECG shows that she is in sustained monomorphic VT.
The nurse notifies the physician that Patient H's vital signs have changed. The patient's blood pressure has dropped from 140/70 mm Hg to 90/40 mm Hg, and his pulse is difficult to palpate. He complains of feeling dizzy and light-headed. When he is attached to a cardiac monitor, the nurse sees that he is having frequent, long runs of nonsustained VT.
The telemetry station notifies the nurse that Patient I has gone into VT. Just as staff members reach her room to assess her status, they see her eyes roll back in her head and she loses consciousness.
Patient J complains of feeling like her heart is "acting funny." When her physician palpates her pulse, he finds that it skips beats and is irregular. A 12-lead ECG shows short bursts of nonsustained VT.
As illustrated in these scenarios, patients with serious ventricular arrhythmias may display a wide range of signs and symptoms. Immediate and careful assessment is required to determine the patient's status, identify the arrhythmia present, and implement the most appropriate interventions. Key points to assess include:
The patient's level of consciousness. Is the patient responsive or unresponsive? If the patient is responsive, does the patient show any subtle or mild signs of decreased cerebral perfusion, such as loss of orientation and confusion? Do family members report any change in the patient's behavior?
The patient's respiratory status. Does the patient have an open airway? Is the patient breathing spontaneously? If so, what is the patient's respiratory rate? Are the respirations labored, shallow, or slow? What is the patient's oxygen saturation? Is it normal for the patient? Has it dropped significantly?
The patient's blood pressure and pulse. Does the patient have a blood pressure? If so, has it remained at a normal or usual level for that patient? Is hypotension present? Has the blood pressure dropped significantly from the patient's norm? What is the patient's pulse rate and rhythm? What is the quality of the patient's pulse?
The patient's cardiac rhythm. What is the patient's ECG rhythm?
The patient's status. Are there any other changes in the patient's status from the patient's norm?
Consider the following patient situations:
The central telemetry station notifies the staff that Patient K appears to have gone into ventricular fibrillation. When personnel reach the room, they find Patient K sitting up in bed, discussing the results of the last high school football game with his family. His color is good, he is alert and oriented, and the only "distress" he voices is his disappointment in the team's performance. The telemetry desk informs personnel that the monitor shows he is still in ventricular fibrillation. What should they do?
Because the patient is awake, alert, and talking, he is obviously not in ventricular fibrillation. Ventricular fibrillation is never a perfusing rhythm; patients in ventricular fibrillation become unresponsive within seconds of developing the rhythm. The most likely cause of the apparent ventricular fibrillation on the central monitor screen is a partial disconnection of the patient's monitor leads. Many monitors are capable of detecting a disconnect from the monitor (e.g., when the monitoring leads become completely detached). However, if part of a lead remains on the patient and one or more leads are dislodged and become stuck to something else (e.g., the sheets), it can produce a pattern that looks like ventricular fibrillation on the monitor. The appropriate action is to check the patient's monitor leads and re-attach any disconnected ones.
The telemetry station notifies the staff that Patient L has gone into a "funny-looking" VT at a rate of 200 or more bpm. When personnel reach the room, they find the respiratory therapist in the room performing chest physical therapy; the patient acknowledges the staff members with a wave and a grimace. As personnel enter the room, the therapist stops the chest physical therapy to see what they wanted. The telemetry desk notifies the staff that the patient has resumed a normal rhythm.
From the information presented, chances are that Patient L has gone into what might be described as "chest physical therapy VT." When a patient is on ECG monitoring, any regular rhythmic activity that involves the chest may create interference or artifact on the monitor screen that appears to be VT but is not.
After the patient's status has been assessed, appropriate intervention may be identified and implemented. There are several recommended protocols for different situations.
In 2010, the AHA published updated guidelines for cardiopulmonary resuscitation and emergency cardiovascular care. These guidelines were updated in 2015 and again in 2020 [10,43]. In 2022, the ESC published updated guidelines for the management of patients with ventricular arrhythmias and prevention of sudden cardiac death [6]. Major points are summarized below; for a comprehensive discussion of the recommendations, changes, and accompanying rationale, the published guidelines are available through the AHA and ESC websites (Resources).
The definitive treatment of choice to suppress ventricular fibrillation or pulseless VT is direct current defibrillation [6,10,43]. Chest compressions should be initiated while waiting for the defibrillator to be set up and charged. Then, administer a single defibrillation shock. Most defibrillators currently in use deliver a biphasic waveform. The recommended energy dose for biphasic waveform is 120–200 joules (i.e., the specific dose within the range that has been found effective in terminating ventricular fibrillation/pulseless VT with the specific device in use). If the effective dose for a specific biphasic device is unknown, 200 joules should be used. Alternatively, if the defibrillator is monophasic, use 360 joules. Compressions should not be delayed to check the pulse or rhythm immediately after a shock is delivered.
Immediately resume chest compressions at a rate of 100 to 120 compressions per minute, and continue CPR for approximately two minutes, or five cycles. Resuscitation efforts should be organized to minimize interruptions to chest compressions [43].
If ventricular fibrillation/pulseless VT persists after delivery of CPR and one shock, the use of epinephrine every three to five minutes should be considered. General guidelines for administering medications during CPR include [10,43]:
Do not stop chest compressions to give medications.
Medications should be administered as soon as possible (ideally within five minutes) after the victim's heart rhythm is checked. The person responsible for preparing drugs should prepare medication before rhythm check to ensure that the dose is available to give as soon as rhythm check is complete.
The recommended sequence of emergency interventions for persistent ventricular fibrillation/pulseless VT is [43]:
Resume compressions and ventilate.
Check rhythm.
If still in ventricular fibrillation/pulseless VT, resume CPR while administering medication and charging defibrillator.
Administer another defibrillation shock.
Resume CPR and administer epinephrine.
If still in ventricular fibrillation/pulseless VT, resume CPR while administering medication and charging defibrillator.
Administer another defibrillation shock.
Resume CPR and consider administering amiodarone or lidocaine. Treat reversible causes.
Epinephrine has been a treatment of choice in ventricular fibrillation for many years. Epinephrine acts by stimulating adrenergic receptors. This results in an increase in blood pressure and heart rate, which re-routes blood flow to the heart and brain and produces peripheral vasoconstriction. Ironically, its negative effects include an increase in myocardial ischemia, increased myocardial irritability, and increased risk for development of new ventricular fibrillation. The usual initial dose of epinephrine is 1 mg given as a rapid IV push. Administration of epinephrine should be followed by another defibrillator shock. One mg doses of epinephrine may be repeated every three to five minutes during the cardiac arrest. Although higher doses may be considered if 1 mg doses are unsuccessful, higher cumulative doses have been associated with a worse neurologic outcome for the patient [10,37].
If ventricular fibrillation/pulseless VT persists after several shocks, CPR, and administration of epinephrine, the administration of an antiarrhythmic should be considered. The initial drug of choice is amiodarone; the second choice is lidocaine [10,43].
Amiodarone is a Class III antiarrhythmic that exerts multiple effects on the circulatory system. For pulseless VT or ventricular fibrillation, the recommended dose is 300 mg given as a rapid IV bolus [10,37]. If needed, a supplemental dose of 150 mg can be given, followed by 1 mg/min for 6 hours and then 0.5 mg/min for 18 hours [10]. If defibrillation shocks temporarily restore a perfusing rhythm that degenerates again into ventricular fibrillation or pulseless VT, the administration of an additional amiodarone bolus may be considered [10].
Lidocaine may be initiated by a loading dose of 1–1.5 mg/kg IV push, followed by a continuous infusion [10]. Additional doses of 0.5–0.75 mg/kg bolus may be administered every 5 to 10 minutes, with a maximum cumulative dose of 3 mg/kg [10,37].
If a patient has gone into VT but maintains a palpable pulse and blood pressure, the selection of interventions depends on an assessment of the patient's status [6]. As illustrated by the clinical scenarios in the beginning of this section, persons with VT may display symptoms that range from asymptomatic to severe. These symptoms result from a drop in cardiac output. Remember that in VT the normal diastolic filling time is reduced, and ventricular contraction is impaired by the rapid rate coupled with the altered conduction. The severity with which VT impacts an individual person's physiologic functioning may be influenced by many factors, such as the patient's age, presence of co-existing cardiac and noncardiac diseases, the rate of the tachycardia, and the length of time that the VT has persisted. VT may be considered unstable if the patient is experiencing significant symptoms of decreased cardiac output and/or there is a high risk that the rhythm will deteriorate into a pulseless or nonperfusing rhythm. VT may be considered stable if the patient experiences only mild symptoms of decreased cardiac output. (It is important to note that stable VT that persists over time can fatigue the heart and produce symptoms.) Determining whether VT is stable or unstable in an individual patient is not always an easy task. The patient's symptoms may fall somewhere between mild and severe, and the clinician will have to make a decision about whether the patient's status leans more toward unstable or stable. When assessing the impact of the arrhythmia on the patient, look for the following signs and symptoms of a decreased cardiac output:
Hypotension or a drop in patient's blood pressure from patient's norm
Dyspnea, shortness of breath, or change in oxygen saturation
Chest pain (especially in persons with underlying coronary artery disease)
Confusion or altered mental status
Rales in lung fields or other signs of congestive heart failure
Complaints of weakness or light-headedness
Syncope or near syncope
Diaphoresis
Patient complains of feeling apprehensive or anxious
Also, it is important to consider the patient's overall status and presence of cardiovascular disease to evaluate the risk that the symptoms will exacerbate other underlying problems [6]. For example, does the patient have congestive heart failure? If so, the combination of rapid rhythm seen in VT coupled with a pre-existing ventricular dysfunction can rapidly lead to severe symptoms [1,6,10].
For stable sustained monomorphic VT, recommended interventions include [5,6,10,37]:
Class I Recommendation
If patient's status changes from stable to unstable at any point, patient should be immediately cardioverted. If unstable status persists or recurs, IV amiodarone should be administered to attempt to achieve a stable rhythm.
Class IIa Recommendations
Administration of IV procainamide is the initial treatment of choice. For suppression of sustained monomorphic VT, procainamide may be initiated with a loading dose of 20–50 mg/min (or 100 mg every five minutes) until the VT is suppressed, or until a maximum loading dose of 17 mg/kg has been reached or until the patient develops the side effects of hypotension or a widened QRS complex (more than 50% over patient's baseline QRS complex). It may be followed by a maintenance infusion of 1–4 mg/min.
Administration of IV lidocaine may be considered for VT specifically associated with acute MI. Lidocaine for suppression of sustained monomorphic VT may be initiated with a 1–1.5 mg/kg bolus IV push. If necessary, a lidocaine infusion may be repeated at a dose of 0.5–0.75 mg/kg every 5 to 10 minutes up to a maximum total dose of 3 mg/kg. A bolus may be followed by a maintenance infusion of 1–4 mg/min.
Class IIb Recommendation
Administration of IV amiodarone or IV sotalol is indicated for VT that recurs despite procainamide. Amiodarone may be initiated with a rapid loading dose of 150 mg over 10 minutes. The loading dose may be repeated every 10 minutes, as needed. Typically, a rapid loading dose is followed with an infusion of 360 mg over six hours. This can then be followed by a maintenance infusion of 0.5 mg/min over 18 hours. Sotalol may be initiated with a dose of 75 mg over five hours twice daily. Dose may be increased after three days to 112.5 mg twice daily and again after another three days to a usual maximum dose of 150 mg twice daily.
If assessment shows that the patient's status is unstable (or more unstable than stable), synchronized cardioversion or defibrillation is the treatment of choice [5,6]. Defibrillators in use today in the acute care setting are cardioverter defibrillators, with the capability of delivering either a synchronized or an unsynchronized shock.
In synchronized cardioversion, the machine delivers the shock in synchrony with the patient's own rhythm; this avoids the risk that the shock will fall during the vulnerable period of repolarization (represented by the T wave on the ECG) and send the heart into ventricular fibrillation. Cardioversion has been found to be effective for arrhythmias caused by re-entry mechanisms (e.g., those underlying most episodes of monomorphic VT). Setting up for cardioversion usually takes a few minutes longer than preparing for defibrillation. The machine uses a sophisticated sensor to identify the highest point of the R wave in the patient's cardiac cycle to time the delivery of the synchronized shock. For the machine to properly identify the R wave, the electrodes (not the "quick-look" paddles) must be used. Additionally, delivery of a DC shock through the chest wall is extremely painful; if possible, the patient should be sedated before the shock is administered. If the patient's status is rapidly deteriorating or is so unstable that the delay of a few minutes to set up and prepare the patient for synchronized cardioversion jeopardizes his/her status, defibrillation should be used instead [49].
In defibrillation, an electrical shock is delivered at a random point in the cardiac cycle; if the patient has no organized, underlying rhythm (e.g., in ventricular fibrillation), delivery of a random electrical shock poses no additional risk to the patient. The electrical shock depolarizes the entire defibrillating myocardium. For persons with a perfusing rhythm, such as VT with a pulse, the heart has some degree of organized electrical activity. Theoretically, the random delivery of an electric shock may allow the shock to fall on the T wave (i.e., during the vulnerable period of repolarization) and cause the heart to go into ventricular fibrillation. If that occurs, the patient should be immediately shocked again to abolish the ventricular fibrillation and restore a normal rhythm [10]. If the VT does not respond to direct current shock or if it recurs, administration of IV amiodarone may be used [5].
Consider this clinical situation:
Patient M is admitted to the emergency room complaining of palpitations, shortness of breath, and light-headedness. She is 23 years of age and reports that she has been in good health. She has no cardiac history or history of arrhythmias; she denies any cocaine use. The monitor shows that she is in a wide complex tachycardia at a rate of 160–170 bpm. The rhythm is slightly irregular, and the width of the QRS complexes is slightly greater than 0.14 seconds (normal is less than 0.10 seconds). Her blood pressure is 85/50 mm Hg, and her oxygen saturation on room air is 91%.
From the information presented above, it is difficult to determine if Patient M is in a sustained monomorphic VT or a supraventricular tachycardia with aberrant (abnormal) conduction through the ventricles. In some cases, precise identification of a wide QRS complex tachycardia cannot be readily determined from a 12-lead ECG. Expert consultation and/or electrophysiologic testing may be required to diagnose the exact mechanism [6,10].
Recommended interventions for wide complex tachycardia of unknown etiology are based on assessment of the patient's status as stable or unstable [5,6,10]. If the patient's status is unstable, the treatment of choice is immediate cardioversion or defibrillation (as described for unstable monomorphic VT). Adenosine should not be given if the patient's status is unstable as it can lead to ventricular fibrillation. If the patient is stable, assess the rhythm to determine if it is regular or irregular. Regular wide complex tachycardias are most commonly caused by VT, supraventricular tachycardia, and pre-excitation tachycardias that involve an extra conducting pathway. Appropriate interventions include adenosine IV, amiodarone IV, and synchronized cardioversion. Amiodarone can be effective in terminating either VT or supraventricular tachycardia. Adenosine can be effective in terminating a form of re-entry tachycardia or may be used to slow the rhythm down sufficiently so that a clear diagnosis of the underlying arrhythmia may be made. Use of beta blockers and calcium channel blockers should be avoided until the rhythm is definitively identified as supraventricular. Use of these drugs in VT can result in cardiovascular collapse [5,6,10].
Irregular wide complex tachycardias are frequently caused by atrial fibrillation with an uncontrolled ventricular response or multifocal atrial tachycardia [50].
Consider this clinical situation:
Patient N is admitted to the emergency room complaining of severe substernal chest pain. He is diaphoretic, and complains of nausea. His blood pressure is 100/50 mm Hg. The ECG monitor shows that he is having frequent episodes of nonsustained VT. On analysis, the QRS complexes are not uniform in configuration; however, they do not appear to be "twisting" around the baseline in the characteristic pattern of torsades de pointes. His ECG also shows signs consistent with myocardial injury and possible infarct. He is diagnosed with polymorphic VT.
Class I recommendations for the management of polymorphic VT include [5]:
For hemodynamically unstable patients, immediate direct current cardioversion or defibrillation is the treatment of choice to suppress the arrhythmia.
If myocardial ischemia is present or suspected, the use of IV beta blockers is indicated. Urgent angiography leading to revascularization is recommended.
Other points to consider in the management of polymorphic VT include [1,3,5,10]:
Identify and correct any electrolyte imbalances, especially potassium and magnesium.
Determine if signs of myocardial ischemia are present; if so, measures to relieve ischemia should be employed.
Consider use of lidocaine, amiodarone, or procainamide to suppress the ventricular arrhythmias if ventricular function is normal.
If ventricular function is abnormal, amiodarone or lidocaine should be used.
If torsades de pointes is caused by a prolonged QT interval caused by a single medication or the interaction of several QT prolonging medications, the offending medication(s) should be immediately stopped [5,10]. Recommended emergent therapies include direct current cardioversion or defibrillation. This may be achieved by [1,5,10,37]:
Overdrive pacing. Overdrive pacing attempts to interrupt the arrhythmia and restore a normal pattern of depolarization and repolarization.
Isoproterenol. Isoproterenol is used as a pharmacologic form of overdrive pacing. When administered by IV infusion, isoproterenol increases the patient's heart rate. Hopefully, the increase in the patient's heart rate will eliminate the torsades de pointes and restore a normal rhythm. To administer isoproterenol, mix 1 mg of isoproterenol in 250 mL of normal saline, D5W, or lactated Ringer's, and infuse at a rate of 2–10 micrograms per minute. Titrate the dose up (to the maximum of 10 micrograms per minute) until the torsades de pointes is abolished.
The AHA/ACC/HRS 2017 guideline recommends IV magnesium as a first-line therapy in patients with torsades de pointes [5].
Consider this clinical situation:
Patient O presents to the emergency room with symptoms of exacerbation of her congestive heart failure. She has a history of end-stage renal disease, peritoneal dialysis, insulin-dependent diabetes, coronary artery disease, and past coronary artery bypass graft surgery. As Patient O is walking into the emergency room, she suddenly collapses. ECG monitoring shows that she is in what appears to be a slow ventricular rhythm. Her QRS complexes are wide with abnormally tall and peaked T waves. Her rhythm rapidly deteriorates, and a sine wave pattern appears on the monitor. Lab analysis shows that her serum potassium level is over 10 mEq/liter (normal is 3.5–5.0 mEq/liter).
Unlike the management of some other ventricular arrhythmias, the management of rhythm changes caused by hyperkalemia must focus on the immediate correction of the potassium level. If the potassium level is not corrected, interventions such as defibrillation and the use of IV medications to restore blood pressure and heart rhythm will be ineffective. Possible urgent/emergency interventions for severe hyperkalemia include the administration of calcium chloride 500–1,000 mg IV or the administration of one ampule of D50W (50% dextrose in water) plus 5–10 units of regular insulin IV to restore a normal serum potassium level [10,19].
Note: Table 2 summarizes drugs used in the emergency management of life-threatening ventricular arrhythmias.
ANTIARRHYTHMIC MEDICATIONS USED IN EMERGENCY MANAGEMENT OF LETHAL VENTRICULAR ARRHYTHMIAS
| Medication | VF Pulseless VT | Stable Monomorphic VT | Stable Wide Complex Tachycardia Etiology Uncertain | Unstable Monomorphic VT | Torsades de Pointes | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Amiodarone |
| 150 mg diluted in 100 mL D5W over 10 min. Follow with continuous infusion 1 mg/min for 6 hr, then 0.5 mg/min for 18 hr | 150 mg diluted in 100 mL D5W over 10 min. Follow with continuous infusion 1 mg/min for 6 hr, then 0.5 mg/min for 18 hr | Not first treatment of choice | Not treatment of choice | |||||||||||
| Lidocaine |
| 1.5 mg/kg IV push. May follow with repeat bolus of 0.5–0.75 mg/kg every 5 to 10 mins to max total dose of 3 mg/kg or maintenance infusion of 2–4 mg/min (30–50 mcg/kg/min) | Not treatment of choice | Not first treatment of choice | Not treatment of choice | |||||||||||
| Magnesium sulfate | Only indicated if hypomagnesemia is suspected or known | Only indicated if hypomagnesemia is suspected or known | Only indicated if hypomagnesemia is suspected or known | Only indicated if hypomagnesemia is suspected or known | For cardiac arrest: 1–2 grams over a maximum of 15 mins given IV push. | |||||||||||
| Procainamide |
|
|
| Not first treatment of choice | Not treatment of choice |
The primary goal in the long-term management of ventricular arrhythmias is to reduce the patient's risk of sudden cardiac death and to control or eliminate symptoms. A detailed patient history and a variety of diagnostic tests may be used in the assessment and evaluation of individual patients.
Key points to cover in a patient history include:
Has the patient experienced an arrhythmia episode before? If so, when? What happened? What symptoms did the patient have? How was it treated?
Is the patient taking any antiarrhythmic medications at this time? If so, which ones? At what dose? Has the patient missed doses? Is there any indication that the patient might have been taking extra doses? In some cases, evaluation of serum drug levels may be indicated to check for the presence of therapeutic or toxic levels.
What other medications is the patient taking? Have there been any recent changes in the medications that the patient takes? Do any of the patient's noncardiac medications have cardiac side effects? Inquire about prescribed, over-the-counter, and natural or herbal medications that the patient may be taking.
Does the patient have any history of heart disease? Coronary artery disease? Stents? Percutaneous transluminal coronary angioplasty (PTCA)? CABG? Heart valve disease, especially mitral valve or aortic valve disease? Has the patient had an MI? Does the patient have congestive heart failure? Cardiomyopathy? If heart disease is present, how is it being treated? How well are the patient's symptoms controlled?
What symptoms is the patient experiencing? Does the patient have any symptoms of reduced cardiac output, such as weakness, light-headedness, unexplained falls or near falls, syncopal episodes, dyspnea, or reduced activity tolerance? Do family members report any changes in the patient's usual functioning?
Does the patient have a history of a close family member (e.g., parent, sibling) who died unexpectedly at a young age or who has/had a history of unexplained syncopal episodes or a known history of sudden cardiac death episodes?
When a patient does not speak the same language as the clinician, a professional interpreter should be consulted to ensure accurate communication. A systematic review of the literature has shown that the use of professional interpreters provides better clinical care than the use of "ad hoc" interpreters, with the former improving the quality of care for patients with limited English language skills to a level equal to that for patients with no language barriers [51]. Use of professional interpreters has been associated with improvements in communication, utilization, clinical outcomes, and satisfaction with care [51]. Individuals with limited English language skills have indicated a preference for professional interpreters rather than family members [52].
Diagnostic tests may be used to confirm the presence of an arrhythmia, link the arrhythmia to symptoms that the patient is experiencing, and identify causes underlying the occurrence of the arrhythmia. Following diagnosis and implementation of treatment, diagnostic tests may also be used to monitor the efficacy of the prescribed therapy.
A resting 12-lead ECG is strongly recommended for all patients with ventricular arrhythmias [5,6,40]. In addition, ambulatory ECG monitoring, either by a Holter or event monitor, should be used for 1) clarification of diagnosis through detection of arrhythmias, QT interval changes, and ST changes, 2) evaluation of effectiveness of therapy, and 3) determination of a diagnosis when symptoms are intermittent or suspected to be caused by transient arrhythmias [5,6].
A 24-to 48-hour Holter monitor may be used for evaluation of arrhythmias thought to occur at least once a day [6]. This monitor is a small portable battery-powered device that is worn by the patient for an extended period. Electrodes connected to the monitor are attached and taped to the patient's chest. A belt or shoulder strap holds the monitor in position. The patient is not allowed to bathe or shower during the period of the test, but otherwise the patient is encouraged to perform his/her usual daily activities. The patient is instructed to keep a diary to document the occurrence, type, and time of any symptoms that he or she experiences. In some cases, if the patient is elderly or might otherwise have problems completing a written log, a family member is asked to assist the patient to document the occurrence of any symptoms. Wireless Holter monitors have a longer recording time than standard Holter monitors and they work for days or even weeks. The down side to wireless monitors is that the patient must remember to write down the time symptoms occurred so that his or her physician can match it to the recording. These monitors also have a shorter battery life [53].
For suspected arrhythmias that occur at less frequent intervals, an event monitor is indicated. An event recorder may be used for monitoring the patient's heart rhythm over a one-to two-month period. Several types are available. Post-event recorders are among the smallest event monitors—about the size of a thick credit card. They can be worn by the patient like a wristwatch or carried in a pocket. There are no wires connecting them to chest sensors, and the patient must hold the device to the chest when a symptom is felt. Post-event recorders only record what happens after started by the patient, so they may miss a heart rhythm problem that occurs before and during the onset of symptoms. Additionally, it may be difficult for the patient to start the device while experiencing symptoms [53]. Autodetect recorders are also small—about the size of the palm of the hand. They do have wires connected to chest sensors and need not be started by the patient. They detect abnormal heart rhythms and automatically record and send the data to the patient's physician [53]. Pre-symptom memory loop recorders (also called continuous loop event recorders) are the size of a small cell phone. They are worn constantly, either clipped to a belt or carried in a pocket. A loop recorder records the patient's heart rhythm continuously, but it only stores and saves the data when "instructed" to do so by the patient [53]. When the event monitor is applied, the patient is told to depress a switch on the recorder any time he or she feels symptoms. When the switch is activated, the recorder automatically stores a few minutes of data from before, during, and after the onset of symptoms, making it possible for the patient's physician to see even very brief changes in heart rhythm [53]. This type of event recorder is indicated for persons whose symptoms are so brief and fleeting that they would disappear before any type of recording device could be attached. The loop recorder may also be used for persons whose symptoms cause loss of consciousness (or near loss of consciousness) as long as the person is not severely disoriented upon returning to consciousness and can still depress the event switch on the outside of the recorder. The data obtained by the event recorder can be readily transferred to a physician via telephone for analysis.
Another type of external event recorder is only attached when the patient experiences symptoms. During a clinic visit, physician appointment, or prior to discharge from an inpatient hospital stay, the patient is given the recorder and instructed on how to apply the electrodes. Commonly, the electrodes attach as bracelets, finger clips, or patches applied under the arms. When the device is attached to the patient, it records and stores ECG data. This type of event recorder is indicated for persons whose symptoms last long enough to permit attachment of the device; it is not indicated for persons who experience loss of consciousness or near loss of consciousness during an episode. To capture the data, the patient should be capable of finding and applying the device during symptomatic episodes. As with the loop recorder, data recorded and stored by this type of event monitor may be readily transmitted to a physician via telephone for analysis [54,55]. An event monitor that may be implanted under the skin has also been developed.
Mobile cardiac telemetry (MCT) has real-time capability to detect any ECG abnormalities and automatically transmit the information via cell phone to an accredited diagnostic laboratory for review [46,56]. The system includes an external, wire-free, adhesive heart monitor that can be worn for up to 30 days. The MCT monitor can transmit both symptoms felt by the patient and abnormalities not felt by the patient [46]. Studies have shown satisfactory patient compliance and arrhythmia detection; however, with some monitors, detected arrhythmias are not discovered until the system is returned for analysis [5].
Wearable patches involve electrodes embedded in adhesive patches that can be worn up to 14 days. These water-resistant patches need not be removed during the monitoring period, which leads to greater wear time, more analyzable data, and no lead reversal errors. Most wearable patch systems have an integrated button to mark the timing of symptoms on the recorded rhythm trace. After the monitoring period is complete, the patch device is returned to the manufacturer, who extracts the data and then sends a diagnostic report to the patient's treating physician. Newer patch-based systems transmit data to the cloud, which facilitates more rapid data collection and analysis [57].
Textile-based systems (e.g., wearable vests and elastic bands) adapt easily to patients' movements. These devices capture electrocardiographic signals via electrodes that are integrated into the garment. This allows for noninvasive ECG monitoring for up to 30 days. ECG signals can either be stored in memory cards for later analysis or transmitted real-time via Bluetooth to a smartphone (and subsequently to a cloud-based platform) [57].
Left ventricular imaging may also be required. According to the ACC/AHA/HRS and ESC guidelines, class 1 recommendations include transthoracic echocardiogram ultrasound exam, exercise testing, cardiac catheterization, and electrophysiology [5,6].
A transthoracic echocardiogram diagnostic ultrasound exam is recommended for patients with ventricular arrhythmias who are also suspected of having structural heart disease and those whose underlying disease processes make them at high risk to develop serious ventricular arrhythmias or sudden cardiac death. This exam may be used to evaluate the valves and structures of the heart as well as the heart's ability to contract. It is a noninvasive test that may be performed on an inpatient or outpatient basis. During the test, electrodes are placed on the patient's chest. Conducting gel is applied to the chest area, and an ultrasound transducer is used to scan the chest. The patient may be studied in the supine or decubitus positions as part of the procedure.
Exercise testing should be considered for adults who have an intermediate or higher probability of coronary heart disease and for persons who are known or suspected to have exercise induced ventricular arrhythmias [5]. Exercise testing with imaging is also recommended to detect silent ischemia in patients who meet specified criteria, including presence of ventricular arrhythmias, intermediate probability of coronary heart disease, and factors that make ECG assessment of ischemia less reliable [5,6]. Types of imaging may include echocardiography or nuclear perfusion (i.e., single-photon emission computed tomography). Pharmacologic stress testing is recommended for patients who have ventricular arrhythmias and an intermediate probability of coronary heart disease but who are physically unable to perform a symptom limited exercise test. The use of MRI and CT may be useful in patients with ventricular arrhythmias when an echocardiogram does not provide accurate assessment of left and right ventricular function or structural changes. Coronary angiography is a possible adjunct to confirm or exclude the presence of significant coronary heart disease in persons with life-threatening ventricular arrhythmias (including survivors of sudden cardiac death) who have at least an intermediate probability of having coronary heart disease [5,6].
Cardiac catheterization may be performed to evaluate coronary blood flow and identify areas of blockage that are accessible to percutaneous interventions such as PTCA, placement of intracoronary stents, rotablation, and atherectomy. Cardiac catheterization may also provide information about left ventricular function and intracardiac pressures.
The role of electrophysiology in the management of persons with ventricular arrhythmias continues to evolve based on data from clinical trials. Electrophysiologic studies can be used both for diagnostic purposes and to guide therapy. The guidelines include electrophysiology testing for evaluation of patients with [6]:
History of an MI and symptoms such as palpitations, presyncope, and syncope
Coronary heart disease who have wide QRS-complex tachycardias of unclear etiology
Impaired LV function or structural heart disease and syncope
Electrophysiologic studies are controversial and not without risk. A multicenter pooled analysis showed that induction of a sustained ventricular arrhythmia during an electrophysiologic study is associated with a higher future risk of ventricular arrhythmia [58].
Data obtained from the patient's history and diagnostic tests are used to evaluate the patient's risk of sudden cardiac death from VT or ventricular fibrillation and to identify the presence of debilitating or troublesome symptoms. Medical intervention is then directed toward the control of any debilitating or troublesome symptoms and the reduction of sudden cardiac death risk in persons evaluated to be at high risk for such events.
Analysis of data from multiple large clinical trials has led to the development of recommended therapies for both primary and secondary prevention of sudden cardiac death in persons at risk. Table 3summarizes findings of major clinical trials investigating the efficacy of various therapies; Table 4 andTable 5summarize current recommendations grouped by ECG presentation and pathology.
OVERVIEW OF CLINICAL TRIALS RELATED TO MANAGEMENT OF VENTRICULAR ARRHYTHMIAS
| Study | Study Population | Findings and Clinical Significance |
|---|---|---|
| Research Focus: Efficacy of ICDs versus Conventional Drug Therapy | ||
| Multicenter Automatic Defibrillator Implantation Trial (MADIT) | Persons with CAD, an EF <35%, documented nonsustained VT, arrhythmia inducible during EPS | ICD is more effective in reducing mortality |
| Multicenter Unsustained Tachycardia Trial (MUSTT) | Persons with CAD, EF of 40% or less, and episodes of nonsustained VT, and inducible sustained VT | ICD is more effective in reducing mortality |
| Multicenter Automatic Defibrillator Implantation Trial (MADIT II) | Persons with previous MI, and an EF of <30% | ICD is more effective in reducing mortality |
| Antiarrhythmic Versus Implantable Defibrillators (AVID) Trial | Persons who had sustained VT, VF or survived a sudden cardiac arrest episode | ICD is more effective in reducing mortality |
| Canadian Implantable Defibrillator Study (CIDS) | Similar to above | ICD is more effective in reducing mortality |
| Cardiac Arrest Study Hamburg (CASH) | Similar to above | ICD is more effective in reducing mortality |
| COMPANION trial | Patients with Class III–IV heart failure, ischemic or nonischemic dilated cardiomyopathy, and QRS duration >120 ms | Cardiac resynchronization therapy with ICD reduced all-cause mortality compared with pharmacologic therapy alone or in patients with dilated cardiomyopathy |
| Sudden Cardiac Death in Heart Failure Trial (SCD-HeFT) | Patients with coronary artery disease or nonischemic cardiomyopathy with NYHA functional class II–III, and LVEF <35% | ICD group showed reduced risk of death (absolute mortality lower in nonischemic group than those with prior MI) |
| Defibrillator in Acute Myocardial Infarction Trial (DINAMIT) | Patients 6 to 40 days post MI with LVEF <35% and impaired cardiac autonomic function | Reduced death from arrhythmias in ICD group No difference in total mortality |
| Defibrillators in Nonischemic Cardiomyopathy Treatment Evaluation (DEFINITE) | Patients with nonischemic cardiomyopathy, NYHA Class I–III heart failure, LVEF <35%, and more than 10 PVCs per hour or nonsustained VT | Data showed trend toward reduced mortality in group with optimized medical therapy plus ICD (versus group with only optimized medical therapy and no ICD) |
| Research Focus: Ambulatory Monitoring versus Electrophysiology Study for Guiding Antiarrhythmic Drug Therapy | ||
| Electrophysiologic Study Versus Electrocardiographic Monitoring (ESVEM) | Patients with ventricular arrhythmias, syncope or cardiac arrest | No difference between group managed with EPS and group managed with Holter monitoring at end point. Sotalol found to be more effective than Class I drugs in preventing death and recurrent arrhythmias. |
| Research Focus: Efficacy of Antiarrhythmias | ||
| Cardiac Arrhythmia Suppression Trial (CAST) | Patients post MI | Higher rate of cardiac and arrhythmic mortality in patients treated with encainide, flecainide, and moricizine than patients treated with placebo |
| Sotalol with Oral D-Sotalol (SWORD) | Patients post MI | Higher rate of cardiac and arrhythmic mortality with patients taking d-isomer sotalol than patients treated with placebo |
| Danish Investigations of Arrhythmia and Mortality on Dofetilide-Congestive Heart Failure (DIAMOND-CHF) Study | Persons with history of heart failure and an EF of less than 35% | Dofetilide did not increase mortality in study population. |
| Congestive Heart Failure-Survival Trial of Antiarrhythmic Therapy (CHF-STAT) | Persons with symptomatic heart failure, fewer than 9 PVCs per hour, and an EF less than 40% | No differences between group treated with amiodarone and group treated with placebo in regard to total mortality or sudden death. Subgroup of persons with nonischemic cardiomyopathy, there was a trend toward reduced mortality in group treated with amiodarone. |
| Grupo de Estudio de la Sobrevida en la Insuficencia Cardiaca en Argentina (GESICA) | Persons with symptomatic CHF, many with nonischemic CH and an EF of 35% | Found a reduction in overall mortality and reduction in SCD in group treated with amiodarone over group treated with triple drug therapy. |
| European Myocardial Infarction Amiodarone Trial (EMIAT) | Persons post MI with asymptomatic ventricular arrhythmia | Amiodarone did not decrease overall mortality. Suggestion that amiodarone did reduce death from arrhythmia. |
| Canadian Amiodarone Myocardial Infarction Trial (CAMIAT) | Persons post MI with asymptomatic ventricular arrhythmia | Amiodarone did not improve overall mortality but reduced combined endpoint of SCD and VF. |
| Ventricular Tachycardia Ablation versus Escalated Antiarrhythmic Drug Therapy in Ischemic Heart Disease (VANISH) | Persons with prior MI and recurrent sustained monomorphic VT despite antiarrhythmic medications | The composite of death, VT storm, or ICD shocks occurred in 59.1% of the ablation group and 68.5% of the escalated-therapy group, with no difference in mortality between the two groups. |
| CAD=coronary artery disease; EF=ejection fraction; EPS=electrophysiology study; ICD=implantable cardioverter defibrillator; MI=myocardial infarction; NYHA=New York Heart Association; PVC=premature ventricular contraction; SCD=sudden cardiac death; VF=ventricular fibrillation; VT=ventricular tachycardia. | ||
RECOMMENDED THERAPIES FOR MANAGEMENT OF VT
| ECG Presentation | Recommendations | Comments | ||
|---|---|---|---|---|
| Sustained monomorphic VT | Class I | — | ||
| If tachycardia is wide complex, presume VT if diagnosis uncertain. Direct current cardioversion if patient is unstable | ||||
| Class IIa | ||||
| ||||
| Class IIb | ||||
| Consider IV amiodarone for VT that is unstable, refractory to counter-shock, or recurrent despite procainamide. For patients in cardiac arrest, epinephrine 1 mg every 3 to 5 mins during CPR may be reasonable. | ||||
| Polymorphic VT (QT interval normal during intervening SR) | Class I | Polymorphic VT with a normal QT interval is most commonly seen during acute ischemia or myocardial infarct, but it may also occur in cardiomyopathy and heart failure. | ||
| ||||
| Class IIb | ||||
| IV lidocaine may be reasonable when acute MI cannot be ruled out. | ||||
| Torsades de pointes | Emergent treatment | When replenishing potassium, a target level of 4.5–5.0 mmol/L may be considered. | ||
| ||||
| Class I | ||||
| ||||
| Class IIa | ||||
| ||||
| Incessant VT | Class I | — | ||
| For patients with incessant or recurrent polymorphic VT due to myocardial ischemia, use IV amiodarone followed by VT ablation. | ||||
| Class IIa | ||||
| IV amiodarone followed by VT ablation may be used in the management of recurring or incessant monomorphic VT storm. | ||||
| ACLS=advanced cardiovascular life support; MI=myocardial infarction; VT=ventricular tachycardia. | ||||
RECOMMENDED PRIMARY AND SECONDARY PREVENTIONa OF SUDDEN CARDIAC DEATH BY EXISTING PATHOLOGY
| Pathology | Primary Prevention Recommendations | Secondary Prevention Recommendations | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Prior MI with left ventricular dysfunction | Class I | ||||||||
|
| ||||||||
| Class IIa | |||||||||
|
| ||||||||
| Cardiac sarcoidosis | Class I | ||||||||
| ICD for patients who have sustained VT, or have survived sudden cardiac arrest, or who have an LVEF ≤35%, and have a reasonable expectation of survival with good functional status for more than one year. | — | ||||||||
| Class IIa | |||||||||
| AAD treatment should be considered in patients with recurrent symptomatic VA. | ||||||||
| Dilated cardiomyopathy | Class I | ||||||||
| ICD for patients with an LVEF 30% to 35% and NYHA Class II or III who are receiving optimal medical therapy and have a reasonable expectation of survival with good functional status for more than one year. (This is a class IIa primary prevention recommendation in the ESC guidelines.) | ICD for patients with significant left ventricular dysfunction and sustained VT or ventricular fibrillation who are receiving optimal medical therapy and have a reasonable expectation of survival with a good functional status for more than one year. | ||||||||
| Class IIa | |||||||||
|
| ||||||||
| Hypertrophic cardiomyopathy | ICD for patients with an LVEF 30% to 35% and NYHA Class II or III who are receiving optimal medical therapy and have a reasonable expectation of survival with good functional status for more than one year. (This is a class IIa primary prevention recommendation in the ESC guidelines.) | ICD for patients with significant left ventricular dysfunction and sustained VT or ventricular fibrillation who are receiving optimal medical therapy and have a reasonable expectation of survival with a good functional status for more than one year. | |||||||
| ICD for patients with hypertrophic cardiomyopathy and sustained VT or ventricular fibrillation who are receiving optimal medical therapy and have a reasonable expectation of survival with good functional status for more than one year. | ||||||||
| Class IIa | |||||||||
|
| ||||||||
| Heart failure | Class I | ||||||||
|
| ||||||||
| Class IIa | |||||||||
| ICD combined with resynchronization therapy (biventricular pacing) for patients with NYHA Class III or IV who are receiving optimal medical therapy, are in sinus rhythm with a QRS of at least 120 ms, and have a reasonable expectation of survival with good functional status for more than one year. | — | ||||||||
| Class IIb | |||||||||
| — | Amiodarone, sotalol, other beta blockers may be used to suppress symptomatic VT in patients with heart failure with are being optimally treated but for whom an ICD is not an option. | ||||||||
| Nonischemic cardiomyopathy | Class I | ||||||||
|
| ||||||||
| Class IIa | |||||||||
| An ICD can be beneficial for patients with Lamin A/C mutation, who have 2 or more risk factors (NSVT, LVEF <45%, nonmissense mutation, male sex), and who have a reasonable expectation of survival for more than one year. | ICD or electrophysiologic study for risk stratification for sudden cardiac death in patients who experience syncope, presumed due to VA, who do not meet indications for a primary prevention ICD, and who have a reasonable expectation of survival for more than one year. | ||||||||
| Long QT syndrome | Class I | ||||||||
|
| ||||||||
| Class IIa | |||||||||
| — | ||||||||
| Drug-induced long QT syndrome | Class I | ||||||||
| — | ||||||||
| Class IIa | |||||||||
| Discontinue use of offending medication(s). Reprogram pacing device. | — | ||||||||
| Adult congenital heart disease | Class I | ||||||||
| — | ||||||||
| Class IIa | |||||||||
| — | ||||||||
| |||||||||
Therapies recommended for the management of ventricular arrhythmias include implantable cardioverter-defibrillators, antiarrhythmic drug therapy, VT ablation, and surgical reconstruction [5,6]. Selection of appropriate therapy can be complex and involves an understanding of the etiology and mechanism of the patient's arrhythmia, the underlying disease processes that may exacerbate the arrhythmia, the risk that the arrhythmia poses to the person, and the risk to benefit ratio of the proposed therapy [5,6].
The presence of PVCs and episodes of nonsustained VT is an indicator of high risk of sudden cardiac death in some patient populations. Its significance depends on whether or not the patient has underlying heart disease, the type of heart disease, and the severity of symptoms [25]. Medical evaluation and treatment are geared toward determining the significance of PVCs/nonsustained VT in the individual patient and controlling troublesome symptoms. Consider these examples of persons diagnosed with PVCs or nonsustained VT:
Patient P is a woman 24 years of age who is admitted to the emergency room complaining that she feels "ready to pass out." Her pulse is irregular; 12-lead ECG shows frequent PVCs with bursts of nonsustained VT. Patient P has no history of cardiac disease and no family history of sudden cardiac death. Subsequent evaluation rules out myocardial ischemia, but a urine toxicology screen positive for cocaine links her arrhythmias to cocaine use.
Patient Q is a man 46 years of age who makes an appointment with his primary care physician because he has been experiencing palpitations. A 12-lead ECG shows no arrhythmias. When Patient Q's symptoms persist, ambulatory ECG (Holter) monitoring is done; it shows frequent PVCs. Further medical evaluation rules out myocardial ischemia and shows that Patient Q has a normal EF. Skillful questioning during the medical evaluation elicits the information that Patient Q has been working two jobs, sleeps less than four hours per night, and drinks a substantial amount of coffee (along with stimulant tablets) to stay awake. Patient Q's arrhythmias are linked to the combination of his stress level, lack of sleep, and excessive caffeine intake.
Patient R is a man 52 years of age who is seen in the preoperative clinic prior to undergoing surgery for a hernia repair. A routine 12-lead ECG shows a short run of nonsustained VT. Patient R has a history of a MI; an echocardiogram a few months earlier showed that he has a near normal EF. Patient R denies any symptoms; the 12-lead ECG and laboratory tests are negative for recent MI. Patient R's arrhythmias are linked to past damage from an MI.
Patient S is a woman 65 years of age who has had a total hip replacement. Her preoperative ECG was normal. She has a history of hyperlipidemia and mild coronary artery disease. Twenty-four hours postoperatively, she develops frequent PVCs and runs of nonsustained VT. She denies symptoms of chest pain but complains of shortness of breath. A 12-lead ECG shows changes characteristic of acute MI, and cardiac enzymes are positive for infarction. Patient S's arrhythmias are linked to STEMI.
Patient T is a man 57 years of age who is admitted to the hospital for dehydration secondary to prolonged nausea, vomiting, and diarrhea. In the emergency room, his continuous ECG monitoring shows frequent PVCs and runs of nonsustained VT. A 12-lead ECG shows no indications of myocardial ischemia, and an initial set of cardiac enzymes is negative for MI. However, his initial electrolyte panel shows a serum potassium level of 2.8 and a serum magnesium level of 1.3. Patient's symptoms are linked to his hypokalemia and hypomagnesemia.
For people with PVCs and nonsustained VT, medical care includes identification and correction of the underlying cause, assessment of risk for sudden cardiac death, and management of troublesome symptoms.
In persons with structurally normal hearts, isolated PVCs can occur normally and without symptoms. These PVCs carry minimal risk of producing sudden cardiac death and do not require treatment. If the patient experiences symptoms such as palpitations, light-headedness, near syncope, or reduced activity tolerance, treatment to control the PVCs to decrease symptoms may be implemented. Initial treatment may focus on explanation of what the arrhythmia is and why it is causing symptoms coupled with reassurance that the arrhythmia is not dangerous and will not lead to serious problems. Given the serious side effects associated with antiarrhythmic drug therapy, antiarrhythmics are often not the first treatment of choice for these patients. The patient may be assessed for the use of substances such as caffeine, nicotine, cocaine, and some over-the-counter medications that have been known to trigger PVCs. Often, reduction or elimination of the offending substance(s) is sufficient to eliminate the PVCs and related symptoms. For persons whose symptoms persist, the use of antiarrhythmic medications or radiofrequency ablation may be considered. Particularly in hospitalized patients (or persons who are ill), the abrupt occurrence of PVCs or nonsustained VT in persons without heart disease may be triggered by electrolyte imbalances such as hypokalemia or hypomagnesemia. Often, correction of the electrolyte imbalance eliminates the arrhythmias [1,3,25].
In persons with known risk factors for heart disease, the abrupt occurrence of PVCs or nonsustained VT may be an indicator of myocardial ischemia or infarction. In these cases, further assessment is necessary. If ischemia is present, measures to reduce the ischemia are indicated. If the arrhythmias are severe, medications such as IV lidocaine may be used to suppress them. In persons with known heart disease who are not experiencing acute ischemia, the occurrence of PVCs and nonsustained VT does not usually lead directly to the development of a sudden cardiac arrest; however, their occurrence may be an indication that the patient's underlying disease has progressed to the point that the patient's overall risk of developing sudden cardiac death has increased. In these patients, the long-term goal of therapy is not the suppression of the PVCs or the nonsustained VT but rather the prevention of sudden cardiac death [1,3,25].
Based on data from several major clinical trials, an ICD is the treatment of choice for persons at high risk for developing sudden cardiac death. Since the advent of the first ICDs in the 1980s, the technology has evolved rapidly. The third- and fourth-generation ICDs are compact, implantable devices capable of delivering a range of therapies based on sophisticated programmed algorithms. Advances in ICD therapy have resulted in devices that include multiple therapy options for the management of ventricular arrhythmias as well as dual-chamber, rate-responsive antibradycardia pacing and cardiac resynchronization therapy. All modern ICDs also function as pacemakers [8,59,60].
Guidelines from the AHA, ACCF, and HRS regarding device-based therapy for cardiac rhythm abnormalities make extensive recommendations for the appropriate use of ICD therapy. In brief, the strongest (i.e., category I) indications for ICD implantation includes [61]:
Persons who have LVEF ≤35%, sinus rhythm, left bundle branch block with a QRS duration ≥150 ms, and NYHA class II, III, or ambulatory IV symptoms on guideline-directed medical therapy
ICD implantation can be useful (i.e., class IIa recommendation) for persons who have [61]:
LVEF ≤35%, sinus rhythm, left bundle branch block with a QRS duration of 120–149 ms, and NYHA class II, III, or ambulatory IV symptoms on guideline-directed medical therapy
LVEF ≤35%, a non-left bundle branch block pattern with a QRS duration ≥150 ms, and NYHA class III/ambulatory class IV symptoms on guideline-directed medical therapy
Atrial fibrillation and LVEF ≤35% on guideline-directed medical therapy if the patient requires ventricular pacing or otherwise meets cardiac resynchronization therapy criteria and AV nodal ablation or pharmacologic rate control will allow near 100% ventricular pacing with CRT
LVEF ≤35% on guideline-directed medical therapy and are undergoing new or replacement device placement with anticipated requirement for significant (>40%) ventricular pacing
ICD implantation may be "considered" (i.e., class IIb recommendation) for persons who have [61]:
LVEF ≤30%, ischemic etiology of heart failure, sinus rhythm, left bundle branch block with a QRS duration of ≥150 ms, and NYHA class I symptoms on guideline-directed medical therapy
LVEF ≤35%, sinus rhythm, a non-left bundle branch block pattern with QRS duration 120–149 ms, and NYHA class III/ambulatory class IV on guideline-directed medical therapy
LVEF ≤35%, sinus rhythm, a non-left bundle branch block pattern with a QRS duration ≥150 ms, and NYHA class II symptoms on guideline-directed medical therapy
ICD implantation is not indicated (i.e., "no benefit," a category III recommendation) for persons with [61]:
NYHA class I or II symptoms and non-left bundle branch block pattern with QRS duration <150 ms.
Comorbidities and/or frailty that limits survival with good functional capacity to less than one year
The basic components of an ICD are one or more leads, a generator, a power source, and a programmer.
The ICD lead is a small, flexible insulated wire that connects the ICD generator to the patient's myocardium. An ICD lead is used to relay information about electrical activity in the heart and to deliver electrical therapy when indicated. Most commonly, ICD leads are inserted transvenously and positioned into the endocardium. Leads may have a small amount of a steroid in a reservoir at the tip of the lead. When the lead is positioned and attached, the steroid is released. The steroid, through its anti-inflammatory action, reduces the amount of inflammation and scar tissue that could develop around the tip of the lead. Minimizing scar tissue allows the electrode at the end of the lead to deliver electrical pacing impulses more effectively. Lead systems in an ICD can be very complex. For basic ICD function, a single lead, consisting of sense/pace electrodes and defibrillation electrode(s), is implanted into the right ventricle. For dual-chamber systems, both atrial and ventricular leads are implanted. ICD systems that provide dual-chamber biventricular pacing may use three leads, one positioned in each ventricle and one in the right atrium. The specific configuration of ICD leads may vary from manufacturer to manufacturer.
The generator houses the power source, microprocessor, and circuitry for the ICD. The microprocessor interprets signals transmitted from the patient's heart through a lead and determines what, if any, electrical therapy is indicated. Arrhythmia detection and selection of therapy are based on sophisticated programming that makes use of logic circuitry and complex algorithms. The generator is also responsible for generating and delivering electrical therapy through the lead system. Both a cardioverting shock and a defibrillating shock require more energy than the energy required by a pacemaker to send a pacing stimulus to the heart. To permit the higher energy shocks, an ICD generator contains capacitors that accumulate and store the necessary energy. The generator also houses a component that stores data about arrhythmias detected and electrical therapy delivered. This stored data may be retrieved noninvasively for analysis through the use of telemetered communication. Generators are small enough to be implanted in the pectoral muscle.
To function as a defibrillator, an ICD must have the capability of building up a high-energy charge rapidly. ICDs use lithium vanadium pentoxide or lithium silver vanadium pentoxide batteries. The batteries are sealed in titanium to prevent leakage. The average battery life is 8 years or longer and is influenced by the type and frequency of electrical therapy delivered [60]. As with a permanent pacemaker, when the battery is depleted the entire ICD generator must be replaced.
As with a permanent pacemaker, an external programmer may be used to communicate with the ICD to retrieve stored data or to program changes in the ICD's functioning. The programmer consists of a relatively portable specialized computer that is capable of communicating with a transceiver housed in the ICD generator through the use of remote telemetric communication technology. This is sometimes referred to as interrogation of the ICD. To interrogate the implanted ICD, the attending physician or other person trained in the use of programmers holds a wand directly over the generator site. When directed to do so, the wand permits data to be exchanged between the ICD and the programmer. Information that may be retrieved includes the current settings and information about arrhythmias detected and electrical therapy delivered. Analysis of stored data allows the physician to evaluate ICD programming to determine if the ICD is delivering electrical therapy appropriately. Data about the status of the battery and remaining battery life may also be obtained. The retrieved data may be printed for later analysis and/or documentation in the patient's medical record. Changes in ICD settings may also be transmitted from the programmer through the wand to the generator; this permits the physician to noninvasively adjust settings based on analysis of the ICD's past functioning and on assessment of the patient's current status, clinical signs, and symptoms [1,8,59].
In simple terms, an ICD has the ability to continuously monitor the heart's rate and rhythm through data obtained from the sensing electrode that is transmitted to the microcircuitry housed in the generator. Using complex algorithms and logic circuitry, the ICD processes the transmitted data to identify episodes of life-threatening arrhythmias. When a life-threatening arrhythmia is detected, the ICD selects the appropriate therapy (based on its programming). The generator rapidly builds up the necessary electrical charge and delivers the programmed amount of electrical energy through the ICD leads directly to the myocardium. Available electrical therapy options that may be programmed include antitachycardia pacing (ATP), lower energy shock for cardioversion, and high-energy shock for defibrillation.
Antitachycardia pacing involves the rapid delivery of multiple pacing stimuli to the myocardium designed to disrupt the VT re-entry circuit by depolarizing the site of the VT during its excitable period. This makes the myocardial tissue refractory to the repeating electrical impulse that is sustaining the arrhythmia. ATP has been found to be effective in terminating sustained monomorphic VT. Depending on the characteristics of the individual patient's arrhythmia, various pacing algorithms may be programmed. Because the amount of electrical energy required for ATP pacing shocks is similar to the amount of energy used for normal pacing, the patient generally experiences no sensation from ATP. This makes ATP a desirable option as an initial response for sustained monomorphic VT. In some patients, the use of ATP can reduce the frequency of the more painful high-energy shocks. Unlike defibrillation, ATP does not disrupt the patient's normal function and its delivery does not produce symptoms discernible to the patient or to other people in the room. ATP should not be a programmed response for detected ventricular fibrillation. Electrophysiology studies may provide data about the specific characteristics of the patient's VT that can facilitate effective programming of ATP.
Low-energy cardioversion involves the delivery of an electrical shock to the myocardium at a precise point in the cardiac cycle. It may be effective in the termination of VT. Low-energy cardioversion was originally developed because it was noted that VT might respond to a lower energy shock than the high-energy shock used to terminate ventricular fibrillation. Unlike ATP, cardioversion shocks are painful to the patient. Low-energy cardioversion can be programmed to act as a back-up when ATP pacing is not effective. Like ATP, low-energy cardioversion should not be programmed for detected ventricular fibrillation.
In the past, high-energy defibrillation was the only type of therapy delivered by ICDs. It remains a standard feature on ICDs. With defibrillation, the ICD delivers a high-energy shock to the myocardium that is sufficient to depolarize the fibrillating myocardium. This interrupts the arrhythmia and gives the heart an opportunity to return to a normal rhythm. Defibrillation shocks are very painful to the patient; some people describe it as a kick in the chest. ICDs are now capable of monitoring the heart's rhythm after a defibrillation shock is delivered and of delivering one or more subsequent shocks if indicated by the patient's heart rhythm. Because some patients experience bradycardia after defibrillation, back-up bradycardia pacing at a low rate is a standard feature on current ICDs. High-energy defibrillation is the only appropriate ICD therapy for ventricular fibrillation. The amount of energy programmed for high-energy defibrillation shocks is based, in part, on a determination of the patient's defibrillation threshold. The defibrillation threshold is defined as the minimum shock strength that terminates ventricular fibrillation that is determined either during implantation of the ICD or during follow-up testing. A patient's defibrillation threshold may change during the life of the ICD; factors that can alter the defibrillation threshold include initiation of or changes in antiarrhythmic drug therapy and poorly controlled ischemia.
Programming therapy options in an ICD is a complex process. Specific therapy should be individualized to the patient and to the actual device implanted. In the past, a major problem in ICD therapy was the inappropriate delivery of electrical therapy for a sinus tachycardia or a supraventricular tachycardia that the device incorrectly detected as VT. ICDs now have features designed to enhance the device's ability to discriminate between VT and supraventricular tachycardia. Dual-chamber ICDs permit sensing in both the atria and the ventricles, thus increasing the device's ability to differentiate a supraventricular arrhythmia from a ventricular arrhythmia [1,8,59].
The magnet feature is a programmable means to temporarily disable the device and may be programmed on or off. When programmed "on," the ICD will respond to the application of the magnet. In general, when the magnet mode is programmed "on," the direct application of a magnet over an ICD generator can inhibit the generator's ability to detect arrhythmias and to deliver electrical therapy until the magnet is removed. Depending on the manufacturer, programming of other magnet functions may be possible [8,60,62].
A routine feature in any modern ICD is back-up ventricular demand pacing. Sometimes referred to as VVI pacing, back-up ventricular demand pacing was primarily designed to pace the heart if a slow heart rate occurred following the heart's recovery from a defibrillation shock. In persons whose ventricular arrhythmias were thought to result when the patient's heart rate dropped, the VVI function in the ICD could also provide intermittent pacing. However, back-up bradycardic pacing was never designed to function as a full-time pacemaker for the heart. One of the primary limiting factors was battery life; continuous pacing would significantly decrease the battery life of the ICD. For a period of time, if a patient required ICD therapy and pacemaker therapy, two separate devices were implanted. This worked adequately, but device interactions created some programming problems. Now ICDs are designed to provide both full pacemaker and ICD therapies. Options include [8,60]:
Dual-chamber sensing that monitors electrical activity in both atria and ventricles.
Dual-chamber pacing that provides a pacing impulse to trigger depolarization in the atrium, the ventricle, or both in proper sequence.
Rate-responsive feature that triggers the pacemaker to pace the heart at a faster or slower rate (within programmed parameters) in response to the body's metabolic demands.
Cardiac resynchronization therapy that uses pacing stimuli to recoordinate the depolarization and contraction of the right and left ventricles (referred to as biventricular pacing). Cardiac resynchronization therapy is a rapidly expanding treatment option for some patients with severe congestive heart failure.
A dual-chamber ICD that is capable of treating atrial and ventricular arrhythmias.
Once the decision to implant an ICD is made, a specific device is selected. As outlined, available devices include single- and dual-chamber ICDs, with options of single- and dual-chamber pacing and biventricular pacing. In selecting a device, the patient's overall status and possible needs for additional pacing support or cardiac resynchronization therapy should be considered. Points to consider in the selection of a device include [8]:
Does the patient have conduction problems such as sinus node dysfunction or symptomatic bradycardia that would benefit from dual-chamber pacing?
Does the patient have congestive heart failure or a form of cardiomyopathy that would benefit from resynchronization of ventricular contraction through biventricular pacing?
Is the patient showing signs of early sinus node dysfunction that may be aggravated if antiarrhythmic therapy is later required to reduce the frequency of arrhythmic attacks?
Would programming of the ICD be more effective for this patient if the device sensed electrical activity in both the atrium and the ventricle?
Does the patient currently have a separately implanted permanent pacemaker?
When the patient is scheduled for implantation of an ICD, any underlying medical conditions, such as congestive heart failure or recurrent myocardial ischemia, should be treated and under control. Electrolyte imbalances, particularly potassium and magnesium, should be corrected. The length of stay for ICD implantation is usually very short and often involves only an overnight stay in an acute care facility. In most cases, ICD implantation may be safely performed in the cardiac catheterization/electrophysiology laboratory. Occasionally, an ICD will be implanted in the operating room during surgery for coronary artery bypass grafting. The patient should be NPO for six to eight hours prior to the procedure. Basic preimplantation lab work is obtained, including a complete blood count (CBC), serum electrolytes, blood urea nitrogen, creatinine, prothrombin time, partial thromboplastin time, and a urinalysis. A baseline 12-lead ECG, arterial pressure, and lateral chest x-ray are also obtained. Baseline vital signs and physical assessment are performed and documented. The planned site for the ICD generator (usually the right or left subclavicular area) is scrubbed and shaved according to institutional protocol. The signed informed consent form is reviewed. The patient may receive a mild sedative prior to leaving the room, although additional sedation and/or analgesics will be administered during the procedure. Some institutions administer prophylactic antibiotics just prior to the procedure [8,62].
Education for the patient and the patient's family begins in the preoperative period and continues throughout the patient's life. Patient education in the preoperative period focuses on addressing the patient's immediate questions and concerns and on providing the patient and family with information about what to expect during implantation and in the period immediately following implantation. Many patients and their families are very anxious about ICD implantation. They may express fears of the ICD firing or the ICD failing to fire when needed. When interacting with them, there are some general points to keep in mind. Many patients who receive ICDs have had a sudden cardiac arrest; this usually represents a major crisis to the patient and to the patient's family. Similarly, because implantation of an ICD is often a graphic reminder that the patient has a life-threatening condition, ICD implantation is also often a time of crisis for the patient and family. During crisis, people do not learn new information or new skills well. They require time to adapt to the changes in the patient's health status and in his or her lifestyle. The short length of stay permitted for ICD implantation limits the amount of information that can be presented or learned. Written instructions should be provided as well as information on outside resources and community support groups (Resources). Patients and their families often have specific concerns about how an ICD may interfere with their normal activities or with their ability to use certain types of equipment. Eliciting any concerns or questions the patient has and responding specifically to those during the preoperative period is very important. Providing specific information about what the patient can expect before, during, and immediately after ICD implantation may also help to reduce the patient's anxiety. The following topics are often helpful to cover in the preoperative period [62,63,64]:
A brief explanation of how an ICD works
A brief discussion about why the device is necessary for the patient. The explanation should not be overly technical and should be directly linked to the patient's specific situation. It is important to stress that the ICD does not prevent arrhythmias from occurring but it monitors the heart continuously and will provide immediate treatment should a life-threatening arrhythmia occur.
A brief description of what sensations the patient may experience when the ICD discharges. Patients have described the high-energy shock as "very painful" or like a "kick in the chest."
A brief description of what the patient's partner or family may experience when the patient's ICD fires. With a high-energy shock, the patient may appear startled, go limp and fall, lose consciousness briefly, or have some body movements that look similar to seizure activity. Following discharge of the ICD, some patients may be disoriented for a short period of time. If someone is touching the patient when the ICD delivers a high-energy shock, he or she may feel a tingling sensation.
A brief description of what an ICD generator and lead system look like. ICD manufacturers are a good source of pictures and diagrams of their products. Some also provide informational videos directed at patients and their families.
A brief discussion about where the ICD will be implanted and how the leads will be attached
A brief discussion about the use of moderate sedation during the procedure to minimize discomfort
A brief description of the usual preoperative preparation procedures
As always, it is important to allow time for the patient and family to ask questions and clarify concerns.
After informed consent is obtained and the preoperative preparation is completed, the patient is taken to the cardiac catheterization/electrophysiology lab. Intravenous access is obtained, and continuous monitoring of ECG rhythm, oxygen saturation, and blood pressure is initiated. The patient receives moderate sedation; commonly used agents for sedation include midazolam and fentanyl. Once the patient is sedated, the physician begins the procedure by creating a small surgical incision that is approximately 2–3 inches in length; this incision is placed under the patient's clavicle, on the left side if possible. The incision site is positioned to provide the physician with ready access to the subclavian vein for transvenous insertion of the lead(s). Although the right ventricular/defibrillation lead is often larger than a typical pacemaker lead, the ICD lead(s) are inserted in similar fashion to pacemaker leads. As the leads are inserted transvenously and passed through the vessels into the chambers of the heart, the physician monitors their passage using fluoroscopy. Once leads are properly positioned, the physician creates a small pocket under the skin in the area of the original incision. Sometimes referred to as the "generator pocket," this area is designed to hold the pulse generator. In most people, the pocket is created in the subcutaneous tissue superior to the pectoralis muscle. For extremely thin individuals, it may be created under pectoral muscle to help provide adequate support and stability for the generator. The lead ends are firmly attached to the pulse generator to ensure good contact and a stable position. The ICD generator is then inserted into the pocket, and the skin is sutured shut. A dressing is applied. (In some institutions, a pressure dressing is preferred.) Antibiotics are frequently administered to reduce the risk of infection. The use of antibiotics varies from institution to institution; some administer a single dose before or during the procedure while others administer IV antibiotics for a 24-hour period following implantation. While the patient is still sedated, the function of the ICD is tested to be certain that the ICD monitors the heart correctly and will deliver a shock if a life-threatening ventricular arrhythmia is detected [8].
Once the procedure is completed, the patient is generally moved to a recovery area until he/she has recovered from the moderate sedation. Once the patient is awake and stable, he/she may be transferred back to the general unit. The goals of care in the immediate 24 to 48 hours postoperative period are to:
Monitor the patient for signs of postoperative complications
Manage the patient's postprocedure pain
Monitor ECG rhythm for indications of normal ICD functioning
Complications that may develop following permanent ICD implantation include bleeding into the generator pocket, hemothorax, pneumothorax, and dislodgment of the leads. Upon return to his or her room, the patient's vital signs are monitored periodically. Continuous ECG monitoring is initiated. Spot checks of the patient's oxygen saturation level may be performed as needed. The generator site is assessed frequently for signs of excessive bleeding or swelling; if these are present, the attending physician should be notified immediately. Posterior-anterior and lateral chest x-rays are taken to evaluate the position of the leads as well as to rule out the presence of hemothorax or pneumothorax [8,59,60,62,63].
Many patients will not experience an arrhythmic episode during their short stay following ICD implantation. However, in the event that the patient does have an arrhythmia, it is important to know how the ICD has been programmed to respond. That information can be obtained by the staff from the patient's chart or by reviewing the summary sheet from the interrogation of the ICD that occurred at the end of the implantation procedure. The physician may choose to include information about the basic parameters as part of the patient's post-procedure orders. Information important to note includes:
Is the ICD programmed to respond initially with antitachycardia pacing?
Is the ICD programmed to respond with lower energy cardioversion?
Does the device provide pacing functions? If so, what kind? What is/are the rate parameter(s)?
Although complete assessment of ICD function requires specialized training in ICD therapy and arrhythmia management, some indications of abnormal (or inadequate) function may be noted by telemetry monitoring and observation of the patient. The most obvious indications are failure to deliver a shock when the patient goes into VT or ventricular fibrillation and a shock delivered inappropriately when the patient is not having a life-threatening arrhythmia.
Failure to deliver a shock may occur because the device failed to recognize the arrhythmia when it occurred. This may reflect a problem with the lead, which may have become dislodged or loose. It may also reflect a problem with the programmed parameters. Please review the following clinical example:
Patient U has returned to his bed following the implantation of an ICD with back-up bradycardia pacing capability. According to the progress notes, Patient U has ATP programmed for VT followed by a high-energy shock if the ATP is unsuccessful or if the patient goes into ventricular fibrillation. Patient U goes into VT at a rate of 180–190 bpm.
If the ICD is functioning properly, the telemetry monitor would show a group of pacing stimuli delivered rapidly; the spikes would be visible and would be intermixed with the VT beats. If the ATP is effective, the monitor would show the resumption of the patient's normal rhythm. If ATP is not effective and VT continues, the telemetry monitor should show a sharp spike on the screen, indicating the delivery of a high-energy shock. If the ICD failed to deliver therapy, the arrhythmia would continue until spontaneously terminated or terminated by external intervention; no evidence of pacing spikes of or defibrillation shock would be present on telemetry monitoring. Delivery of inappropriate therapy may occur as a result of sensing problems or problems in the ICD's programming.
Patient U's ICD is programmed to deliver ATP first, followed by a defibrillation shock if needed. Telemetry monitoring notes that Patient U's rhythm is his "usual" atrial fibrillation at a rate of 80–90 bpm. Patient U calls the desk abruptly to report that his ICD "just fired." A check of the telemetry record shows that the ICD has fired. The physician is notified; interrogation of the ICD and analysis of the data show that a high-energy shock was delivered inappropriately for atrial fibrillation.
Problems with programming are corrected through noninvasive interrogation and reprogramming of the generator. If a lead has become dislodged, an additional surgical procedure may be required to replace the lead in the correct position.
Assessment of function of an ICD with dual-chamber pacing capabilities can be complex. Consider these situations:
Patient V returns to the telemetry unit following implantation of a dual-chamber ICD. His upper rate limit for pacing is 110 bpm; his low rate limit is 60 bpm. The device is capable of pacing in the atrium, the ventricles, or both. His ICD is programmed for both ATP and a high-energy defibrillation shock. Patient V's rhythm on telemetry when he first returns to the unit is sinus rhythm at a rate of 60 bpm. A few hours later, personnel note intermittent spikes appearing in Patient V's ECG tracing. In some cases, the spikes are present just before a widened QRS, and other times they appear just before a slightly abnormal P wave; sometimes, the spikes appear at both times. Patient V's heart rate on ECG is 75 bpm; the spikes are not appearing at a rapid rate. Does this represent normal functioning for this device?
Yes. Patient V's device is also a dual-chamber pacemaker. A spike before the P wave indicates that the pacemaker has fired to depolarize the atria; often, P waves following a pacer spike look slightly different in shape from a normal P wave. A spike before the QRS complex indicates that the pacemaker has fired to depolarize the ventricles; paced QRS complexes are normally wider and more bizarre in shape than normal QRS complexes. Because a dual-chamber pacer can pace both atria and ventricles, it is normal to see pacer spikes in front of P waves, QRS complexes, or both on the same patient over a short space of time. Patient V's rate is within the programmed limits for his pacemaker.
Patient W is admitted to the unit for management of an exacerbation of congestive heart failure. He has an ICD with rate-responsive ventricular demand pacing capability; his ICD is programmed to deliver ATP as well as a high-energy defibrillation shock. On his admission, it is noted that his ECG rhythm on telemetry monitoring shows wide and bizarre-appearing QRS complexes at a rate of 66 bpm. Careful examination in other leads shows evidence of a small ventricular pacing spike present before each QRS. Patient records show that Patient W's pacemaker has a low rate setting of 60 bpm and a high rate limit for rate responsiveness of 100 bpm. Later in the evening, on telemetry monitoring, personnel note that Patient W's QRS complexes have changed deflection (now pointing down instead of up). They remain wide and bizarre but occur at a rate of 110–120 bpm. No pacemaker spikes are visible in any lead. Patient W reports "feeling bad" and a little "light-headed." What is occurring?
The initial ECG strip obtained on admission shows normal ventricular demand pacing function. The rate is within the rate parameters programmed for the pacing function, and there is a pacing spike in front of each QRS complex. Paced QRS complexes are frequently wider and more bizarre than normal complexes. The second strip, obtained later, does not show normal characteristics for Patient W's device. The rate exceeds the programmed limit, the QRS complexes are wide and bizarre, and there are no pacemaker spikes in front of each QRS. The abnormal shape of the QRS complexes coupled with the rate and change in deflection suggest that this rhythm is probably ventricular. The absence of any response from the ICD may indicate abnormal function; however, more probably, the rate of this rhythm is too slow for the ICD to recognize it as VT.
Patients often experience postoperative pain in the generator site. Depending on multiple factors, patients may describe pain as mild to very severe. The patient's pain level should be routinely assessed using a 10-point pain scale (1 for least, 10 for most). The physician's orders should include analgesic medications; prescribed medications may range from acetaminophen to stronger narcotic analgesics. The effectiveness of pain medication should be assessed and documented. Any complaint of increasing pain despite adequate analgesic medication should be carefully investigated. Increased pain in the generator site may be an indication that bleeding is occurring into the pocket.
Infection related to cardiac device implantation has become a serious and costly issue. The incidence of pocket infection and/or bloodstream infection or device-related endocarditis has risen markedly. Hospitalizations for device-related infections increased out of proportion to the number of new devices implanted. Additionally, the presence of infection increases the risk of in-hospital death by more than twofold [65].
In 2010, the AHA published updated guidelines for the management of cardiovascular implantable electronic device (CIED) infections [65]. In this scientific statement, the writing group presented evidence-based recommendations for prevention and management of non-valvular device infections. Key elements of these guidelines include [65]:
Prophylactic use of an antibiotic that has in vitro activity against staphylococci at the time of device implantation is recommended.
Antimicrobial prophylaxis to prevent CIED infection is not recommended for dental or other invasive procedures not directly related to device manipulation.
Diagnosis of suspected CIED infection should include at least two sets of blood cultures.
Complete removal of the device and leads is recommended for patients who meet specific clinical criteria. These criteria include but are not limited to:
Definite CIED infection evidenced by valvular and/or lead endocarditis or sepsis
Evidence of pocket infection
Presence of occult staphylococcal bacteremia
Valvular endocarditis
Duration of antimicrobial therapy after device removal depends on the location and complexity of the infection. Duration for uncomplicated pocket-site infections should be 10 to 14 days, and at least 14 days for bloodstream infections. Complicated infections may require four to six weeks of therapy.
The need for reimplantation should be evaluated carefully on a patient-by-patient basis.
If blood cultures were positive before the device is removed, blood cultures should be drawn after device removal and should be negative for at least 72 hours before a new device is placed.
Although infection with Staphylococcus spp. accounts for the majority of cases of CIED infections, other opportunistic pathogens (e.g., Burkholderia cepacia, S. aureus, S. marcescens, Pseudomonas aeruginosa) have also been implicated in CIED infection [66,67].
Early complications associated with ICD implantation include infection of the incision site or the pocket and accidental dislodgment of the leads. To lessen the risk of infection, instruct the patient to avoid getting the incision site wet. Also, the site should be inspected routinely for signs of redness, warmth, or drainage. During the patient's hospital stay, a dressing should be maintained over the incision site. If the dressing becomes dislodged or saturated with blood during this initial period, it may be changed using sterile technique. To lessen the risk of lead dislodgment, instruct the patient to limit the use and movement of the affected arm. Caution the patient to avoid extending the arm above the level of the shoulder. Use of a sling on the affected arm can serve to promote comfort and to remind the patient to limit use of that arm [8].
Basic patient education prior to the patient's discharge from the hospital should focus on incision care, arm precautions, resumption of activity (including sexual activity), and basic safety measures. Written instructions should be provided. During every subsequent follow-up visit, the patient's understanding of previous instructions should be assessed, and additional information provided as needed. Key points to consider when implementing patient education for a person following ICD implantation are outlined in this section.
To decrease the patient's risk of developing a wound infection, include these instructions [8,63]:
Keep the incision clean and dry for the first five to seven days.
You may shower as long as you do not let the water hit the incision line directly. Do not immerse the incision in water.
After bathing, carefully pat your incision line dry. Do not rub. Do not apply lotions or powders to the incision line.
Look at your incision line every day. Use a mirror if necessary. Look for any signs of redness, drainage, or swelling around the incision line. Notify your physician immediately if any of these signs are present or if you develop a fever (>101°F or 38°C).
To decrease the risk that an ICD lead will accidentally become dislodged during the initial four to six weeks after implantation, instruct the patient to [8,63,68]:
Avoid excessive arm movement on the side that an ICD was implanted for at least four weeks.
Avoid vigorous activities (including golf or tennis) for four to six weeks.
Avoid lifting, pushing, or pulling more than 5 pounds for the first four to six weeks. Activities that should be restricted include carrying groceries in from the car, carrying laundry baskets, and pushing vacuum cleaners.
Patients frequently have questions about resumption of activities. Following ICD implantation, a patient may gradually resume his or her usual level of activity. Moderate physical activity is permitted as long as it does not cause dyspnea, chest pain, or other medical problems. To reduce the risk that the ICD may be triggered to fire by an increase in pulse rate caused by exertion, some patients undergo a treadmill stress test to assess their heart rate response to activity. If the patient's maximum heart rate is too close to the rate used in programming the ICD to deliver electrical therapy, the ICD may be reprogrammed or beta blockers may be prescribed for rate control [8,62,63,64].
Sexual activity may also be resumed with no restrictions as long as strain on the generator site is avoided during the immediate post-op period. As with any activity, the patient should discuss with his/her physician any dyspnea, chest discomfort, or dizziness that occurs with sexual activity. Many patients and their partners have concerns about the resumption of sexual activity. These include the concern that sexual activity will trigger the ICD to fire, concern that sexual activity will trigger the arrhythmia and the ICD will fail to fire, and concern about what the partner might experience if he/she is in contact with the patient when the ICD fires. Patients and their partners should be advised that someone touching them is not harmed if the ICD discharges [60]. Reassurance and a common sense approach can be very helpful [68,69].
Basic safety measures to cover with the patient and family include the ICD identification card, notification of other physicians and healthcare providers, restricted activities, what to do in the event that the patient receives a shock from the ICD, and when and how emergency help should be summoned [62,63,70].
At the time that an ICD is implanted, a temporary identification card is issued. This card lists specific information about the patient's device, including the type, manufacturer, date of insertion, and healthcare provider information. Make sure that the patient receives the temporary card before discharge. Explain that a permanent card will be mailed by the device manufacturer to the patient within a few weeks following discharge. Instruct the patient to always carry the identification card, and encourage a family member to keep and carry an additional copy. Explain that the card contains vital information that a physician would need to know to assess both the patient and the ICD function accurately in the event of a problem.
Instruct the patient and family to let all other healthcare providers, including dentists, know that the patient has an ICD. Some medical and dental procedures may temporarily affect the functioning of the device and will need to be modified. Other procedures, such as magnetic resonance imaging (MRI), are contraindicated for a person with an implanted device. Also instruct the patient to obtain a medic-alert bracelet or other appropriate ID jewelry. If possible and available, assist the patient to complete the form for the identification bracelet prior to the patient's discharge.
Unlike a permanent pacemaker, implantation of an ICD is usually accompanied by restrictions on patient activities. Because the patient may lose consciousness during an episode of VT or ventricular fibrillation, he or she should refrain from activities in which loss of consciousness could result in injury to themselves or to someone else. Driving is a major issue for persons with an ICD. At this time, state laws regarding driving for ICD recipients are not consistent. Commercial driving is generally prohibited. Some authorities recommend that people should not drive for a six-month period following the implantation of an ICD or for six months following the discharge of the ICD [60]. Loss of driving privileges can impose a significant hardship on the patient, and it is thought that many do not adhere to the six-month driving restriction. Some research has been done to attempt to quantify the risks of allowing persons with known life-threatening ventricular arrhythmias to drive. In one study, data showed that patients often had symptoms of arrhythmias while driving but that the occurrence of accidents was lower than the annual national accident rate [71]. In addition to driving restrictions, persons with ICDs are encouraged to avoid performing any activities in isolation (e.g., swimming, boating) where a loss of consciousness could result in injury. Generally, the use of heavy equipment or piloting an aircraft is also prohibited [68,72].
Many patients with an ICD will experience a discharge or shock from the ICD at some time. The experience can be very frightening. It is important to discuss in advance what the patient might expect and what the patient should do in the event of a shock. Make sure to provide written instructions and to review the information verbally. When discussing this with the patient and family, keep these points in mind:
It is likely that the patient's ICD will fire and the patient will receive a shock.
When the arrhythmia occurs, the patient may develop symptoms of light-headedness, dizziness, or loss of consciousness. These symptoms will disappear or reverse once the ICD has fired to terminate the rhythm.
Although it is possible for the patient to be completely asymptomatic and unaware of the arrhythmia until the ICD unexpectedly delivers a shock, most people have at least some subtle signs that they can learn to recognize as warnings that their arrhythmia is occurring and that a shock may be imminent.
When the patient receives the first shock, the physician should be notified within 24 hours. The physician will schedule an immediate appointment to assess the patient and the functioning of the device.
Following a shock, the patient should note the details in a journal, including a brief description of the circumstances. What was the patient doing? What symptoms occurred before the device fired? What happened after it fired?
Following a shock, emergency help should be summoned if the patient:
Receives numerous shocks
Remains unresponsive or symptomatic following the shock
Develops worsening symptoms following delivery of the shock
Plans should be made in advance for accessing emergency help. Emergency numbers for EMS or local ambulance companies should be visibly posted near a telephone along with contact numbers for the local hospital emergency room, the patient's physician, and a designated friend or family member.
If the patient's heart rate is frequently paced by the implanted device, the patient or a family member should be taught how to check the pulse. When teaching a patient to check his/her pulse, consider covering these points [73]:
Check your pulse regularly at least once a week (or more often as directed).
Count your pulse for a full minute.
Each time that you count your heart rate, record the rate and the date in a notebook. Bring this notebook to your follow-up appointments.
Find out from your physician what your lowest acceptable heart rate is. Notify the physician if your heart rate drops abruptly or significantly below that level.
If your pacemaker has an upper rate limit, determine what your highest acceptable heart rate is. If your heart rate dramatically increases over that limit, contact your physician.
Your pulse might feel a little irregular. As long as your pulse rate falls within the acceptable range established by your physician, do not worry about an occasional irregularity.
Patients with ICDs should exercise caution to avoid exposing the device to strong electromagnetic interference (EMI) [63]. By definition, EMI occurs when the functioning of one electronic device disrupts or interferes with the normal functioning of another device. With an implanted ICD, if the EMI is strong enough, the patient may receive an inappropriate shock or the device may fail to deliver a shock when needed. In general, EMI does not permanently damage the device; usually, moving away from the source of the EMI is sufficient to restore normal ICD functioning. Many tools and electronic devices use both electricity and magnets; these can create an electromagnetic field around the device. The size and strength of the field vary with the device. A large piece of equipment, such as a magnetic resonance imaging machine or an arc welding device, generates a very strong, powerful electromagnetic field. Household items, such as microwave ovens, mixers, and electric blankets, generate a very weak electromagnetic field [63,74].
Portable cordless telephones and many cell phones do not interfere with ICDs. However, as cellular phone technology is constantly changing, it is prudent to instruct patients to use some precautions when using or carrying a cell phone. Consider covering the following points [74]:
Maintain at least a 6-inch distance between the cell phone and the device. Use the ear on the opposite side from the device when talking on a cell phone.
Carry the cell phone in a pocket away from the generator; avoid carrying the phone in the pocket that is right over the generator site when the phone is turned on.
Cordless household telephones are not the same as cellular phones and should not interfere with the device's function.
Generally, a person with an ICD can safely use normal household electrical devices as long as those devices are in good repair and properly grounded. Devices that may be safely operated include [74]:
Microwave ovens
Gas or electric ovens
Televisions, DVD players, CD players, AM/FM radios, video games, and stereos (except for those with large speakers)
Washing machines, dryers, and refrigerators
Electric blankets, heating pads, and portable space heaters
Toasters and electric can openers
Vacuum cleaners
Treadmills
Handheld remote controls for televisions, radios, DVD, garage doors, or cars
Computers and other small office equipment, such as printers, fax machines, copiers, and scanners
The following cautions and precautions should be noted:
A remote control that has an antenna should not be used.
Small household appliances that contain an AC motor should be kept 6 inches from the ICD generator. These include electric toothbrushes, handheld dryers or shavers with an electric cord, and pagers.
When using a smooth-top range (i.e., induction range), the patient should keep the defibrillator generator two feet away.
For more specific information about the use of a particular device, the patient may review the material provided about his/her defibrillator by the manufacturer or, in some cases, obtain additional information from the manufacturer's website.
With the heightened emphasis on security checks at airports, patients and families may have concerns about how to get through airport security safely and with as little hassle as possible. Studies have shown that while airport security devices do not damage an ICD, the security devices will detect the implanted metal and trigger the alarm. To facilitate getting through airport security, instruct the patient to [68,74]:
Always carry your ICD identification card.
Present the identification card to the security agent before walking through a security screening device.
Request a hand search. Avoid the use of a handheld screening wand. Screening wands contain large magnets; movement of the wand back and forth over the area of the defibrillator can cause the device to discharge inappropriately.
Commercial antitheft devices are commonly used in many department stores in the United States. These devices have the potential to disrupt the functioning of the pacemaker temporarily. Instruct the patient to walk through the security screening device at a reasonable pace; also instruct the patient to avoid lingering in the area around the antitheft devices.
Most medical and dental diagnostic procedures will not interfere with the proper functioning of a person's ICD. For example, dental x-rays, dental drills, dental ultrasonic probes used in dental cleaning, chest x-rays, CT scans, nuclear medicine studies, mammograms, and fluoroscopy do not cause interference and are considered "safe." Some procedures, however, will disrupt normal ICD functioning. For example, MRI (which uses a very large, extremely powerful magnet) is contraindicated for anyone with an ICD. Other procedures have the potential to disrupt ICD functioning and may cause extra shocks. Instruct the patient to let all healthcare providers know that he/she has an ICD before a procedure is performed. If the procedure has a risk of disrupting the ICD's functioning, the patient should discuss the risks and benefits of having that procedure done with the physicians. In some instances, specific precautions may be taken to reduce the risk that a given procedure will disrupt the ICD's function. ICD manufacturers also recommend that the pacemaker be interrogated to verify proper programming following performance of a procedure that could potentially generate enough EMI to disrupt programming or functioning.
Research has shown that patients with ICDs report strong feelings of fear, terror, and anxiety [60]. If the patient experiences a shock from the ICD, his or her anxiety and fear may increase. If the ICD fires without warning, patients may become additionally anxious and fearful. Common fears include fear of a shock, fear that the device will fail, and fear of death. Persons with an ICD also have been found to experience a strong sense of loss of control. They struggle with the realization that the device may fire without warning and that they have no control over when the device fires. Some people who experience a shock may respond by restricting their activities in an attempt to avoid "triggering" the device another time. They may repeatedly analyze the circumstances of the initial shock, attempting to establish a "cause and effect" that will help them avoid a future shock. Occasionally, people with an ICD develop the phenomenon of a "phantom shock." Briefly defined, a phantom shock occurs when the patient feels that the ICD has fired although the device actually has not discharged an electrical shock. People may experience anger and depression over their loss of independence and their decline in health status [60]. Other issues include employability, maintenance of their current job, obtaining a different job, eligibility and maintenance of insurance coverage, and the costs involved in ICD follow-up and medical care. Patients also report that they experience a variety of physical complaints during the months following ICD implantation. These include insomnia and sleep disturbances, lack of energy, dizziness, weakness, decreased exercise tolerance, and weight loss or gain. Short-term memory loss, difficulties in coping, and problems with family functioning may be experienced. The patient's partner and close family and friends also report anxiety and fear related to the implantation of the ICD. Some express the fear that they "will not know what to do if the ICD fires." Some partners report changes in sexual activity brought about by the fear that such activity will trigger the ICD. Some people are concerned that they might experience a shock if they are touching the patient when the patient's ICD fires. Persons with ICDs report that their partners and other family members often become overprotective [60,62,64,69,70]. Interventions that are helpful to the patient and family include:
Referral to a local support group for ICD patients and their families
Assistance in helping the patient and family develop and rehearse a plan for what to do when the patient receives a shock and what to do in the event of an emergency
Recommendation that partners or family members take a community-based CPR course
Referral for professional counseling for management of anxiety and depression and development of increased skills in coping with a life-threatening illness
People often experience major lifestyle alterations following ICD implantation. Depending on the type of job and the patient's overall status, the patient may or may not return to work. Some studies have shown that many people who return to work choose to retire within the following 6 to 12 months. Reasons individuals give for not continuing to work include a decrease in exercise tolerance, impaired concentration and short-term memory, and poor overall status. The patient's normal patterns of interaction with family and close friends may change; many patients report that they are forced to become dependent on other people for assistance if they cannot drive themselves to the store, medical appointments, and other routine activities. Especially for younger people, participation in previous hobbies and sports may be limited or no longer possible [70].
Patients with ICDs should be observed in a dedicated ICD clinic. These patients generally are seen one week after implant for a wound check, one month after implant for device interrogation, and three months post-implant for repeat device interrogation. The follow-up interval then can generally be increased to once every six months if the patient is clinically stable [60]. The generator site should be carefully assessed for indications of impaired healing or signs of complications. Complications that may occur include infection in the pocket, development of a hematoma in the pocket site, and erosion of the generator or the lead through the tissue and skin. Persons with diabetes are more at risk to develop an infection. Persons taking anticoagulants are at risk to form a hematoma in the generator pocket [75]. Signs of impaired healing may include:
Subjective complaint of increased pain at the generator site
Increased swelling present in the pocket area
Elevated temperature or other signs of systemic infection
Purulent drainage from the incision site
Failure of the incision line to heal
Warmth or redness around the incision line or pocket
Infection, hematoma, and erosion are serious complications. Infection in the ICD site or along the leads necessitates the removal of the device. Because of the risk of sudden cardiac death, the patient may have to remain in the hospital, where emergency equipment is readily available, until the infection resolves and the device can be re-implanted. In addition to wound assessment, the functioning and programming of the ICD should be assessed. Noninvasive interrogation of the generator is done to retrieve stored information and information about any arrhythmia events detected or any delivered therapy. The follow-up visit also provides an opportunity to obtain and document patient information as required by law. U.S. regulations require that medical device manufacturers maintain a correct and current database of all persons in the United States who have an implantable medical device [8,64]. Recorded information includes the patient's address, telephone number, and physician. Patients should be instructed to notify their specific device manufacturer when they change addresses, when they get a new telephone number (or when their area is code is changed), or if they change their physician [8,64].
Long-term follow-up of patients should be done by a physician who is experienced in ICD and arrhythmia management. It should occur in a setting that also provides the services of an experienced cardiovascular nurse and experienced technical support staff. Available equipment should include a programmer, ECG recorder, and an emergency cart with a defibrillator capable of external defibrillation. Facilities for ambulatory monitoring, exercise stress testing, and echocardiography should be readily available if needed for extended evaluation of the ICD function or management of the patient's arrhythmias. There is no universal agreement about a set pattern or frequency of follow-up visits. In general, follow-up should occur at one-to four-month intervals or when indicated by suspected problems in device functioning or changes in the patient's status [4,8,59,63]. Elements of long-term follow-up include:
Questioning the patient about his/her perception of his/her general status and any change in overall symptoms or condition since the last appointment.
Obtaining a patient history related to any arrhythmia episodes and ICD discharges. Data to obtain include a description of symptoms, any precipitating factors, and a description of what occurred after the shock. The device should be interrogated to obtain data about detected events and device function; the data obtained should be correlated with the patient's verbal report of the event(s).
Performing noninvasive interrogation of the device to check battery status and the status of the leads.
Checking the pacing and sensing thresholds.
Performing a chest x-ray to verify that the leads are still properly positioned.
Taking the patient's history and performing a physical assessment to evaluate the status of any co-existing cardiovascular disease. If changes are detected, the patient may be referred back to his/her primary physician for evaluation and management.
Changing the programmed parameters, if indicated from patient assessment and/or interrogation of the device, to improve the device's functioning.
Because assessment of the patient's defibrillation threshold involves programmed stimulation to induce ventricular fibrillation, the defibrillation threshold is not routinely checked at follow-up visits. It may be checked at the time of implantation or approximately one month following implantation. Changes in the patient's antiarrhythmic therapy or significant medication changes may require a recheck of the defibrillation threshold at some point.
Over the last decade, an increased number of patients have received ICDs for primary or secondary prevention of sudden cardiac death. Although ICD implantation is effective in reducing a person's risk for sudden cardiac death, long-term device therapy poses additional patient management issues. Common issues may include device malfunction, delivery of inappropriate shocks, frequent appropriate firing of the device for VT or ventricular fibrillation, "electrical storm," and decisions related to end-of-life care.
Failure to deliver therapy results in an emergency situation for the patient. Clinical indications of a failure to deliver therapy include cases in which the patient:
Becomes unresponsive
Goes into cardiac arrest
Complains of continuing or worsening symptoms similar to those experienced before the device was implanted
When failure to deliver therapy occurs, immediate interventions should focus on the termination of the arrhythmia. Emergency assistance should be summoned. If necessary, the patient may be externally defibrillated or cardioverted. Defibrillation pads or paddles should not be placed directly over the ICD generator. Following termination of the acute event and stabilization of the patient, evaluation of device function and possible contributing factors to the therapy failure should be analyzed. Therapy failure may occur because of device problems, such as fractured or dislodged leads, breaks in lead insulation, or battery depletion, or, in rare cases, it may be caused by an inadvertent deactivation of the ICD. Inappropriately programmed settings, such as the sensitivity level for sensing electrical activity, and, less commonly, intradevice interactions, may lead to failure to deliver therapy [8,60,76].
In the past, a large number of people with ICDs received inappropriate electrical shocks for rhythms other than VT or ventricular fibrillation [77,78]. Non-life-threatening rhythms that the ICD commonly mistook for VT included sinus tachycardia and supraventricular tachycardia [59,79]. Given the enhancements incorporated into current generation ICDs, the risk of shocks for sinus tachycardia or supraventricular tachycardias is much less. In addition, the use of dual-chamber ICDs that can sense electrical activity in the atria has improved the device's ability to discriminate between VT and rapid atrial fibrillation or atrial flutter. When the patient suspects an inappropriate shock in the absence of persisting severe symptoms (e.g., chest pain, shortness of breath, rapid palpitations), he/she should contact the physician and schedule an appointment for interrogation of the device for stored data and testing of lead function [79]. Device problems that may result in delivery of inappropriate shocks include EMI, T-wave oversensing, a dislodged lead, a fractured lead, or a break in the insulation covering the lead [59]. Replacement of the lead may be required.
If interrogation shows that the device fired in error, programming adjustments may be made noninvasively to improve detection and discrimination. If interrogation shows that the device did not fire in error, the patient should be questioned carefully about the circumstances of the shock and why the patient thought the shock was inappropriate. A patient may suspect an inappropriate shock when the ICD fires without warning or when it fires in the absence of symptoms that the patient commonly associates with an arrhythmia occurrence. The patient may also suspect an inappropriate shock when the device fires more than once in close succession. While these circumstances do not indicate device malfunction, they may indicate a change in the patient's status. There may be a new pattern of arrhythmic episodes that should be evaluated to ensure that device programming and other medical therapy are optimally maintained [8,79].
As a person's cardiac function declines, the likelihood that he/she will experience a life-threatening arrhythmia and receive an appropriate shock from the ICD increases. ICD shocks can be very painful, and patients who are shocked may become very anxious about the possibility of another shock. To reduce the need for high-energy shocks, ICDs may be programmed to deliver ATP as an initial therapy when the ICD recognizes VT. (Note: Electrical external defibrillation remains the most successful treatment for ventricular fibrillation [80].) As discussed, ATP attempts to disrupt the VT and return the patient to a normal rhythm by firing a rapid burst of pacing spikes. Antiarrhythmic medications may also be used to reduce the need for high-energy defibrillator shocks [5,10].
In an "electrical storm," a patient receives multiple appropriate shocks from his or her ICD for VT or ventricular fibrillation within a relatively short period of time. Most sources define an ICD storm more precisely as at least three or more appropriate shocks within 24 hours [81,82,83,84]. Factors that may trigger electrical storm include modification of or noncompliance to drug therapy, worsening of heart failure, early postoperative period, emotional stress and anger, alcohol excess, electrolyte abnormalities, and myocardial ischemia. Most cases, however, occur without any apparent cause [79]. More than 80% of cases of electrical storm are caused by episodes of monomorphic VT; however, it can also occur due to polymorphic VT/ventricular fibrillation. Knowledge of the type of underlying arrhythmia is important when selecting a management strategy. Electrical storm has been reported in 10% to 40% of patients in secondary prevention; the incidence is lower in primary prevention [79].
ICD storm can be extremely frightening and painful for the patient. Patients may be concerned that the device is malfunctioning while at the same time they may be realistically frightened that the increase in ICD shocks indicates deterioration in their cardiac status [79]. Patients who experience ICD storm should be instructed to contact their physician immediately and should expect to come to the clinic or the hospital for evaluation [63,64,79].
During medical evaluation for ICD storm, the patient's ICD is interrogated and stored data about any events detected and therapy or therapies delivered is retrieved and analyzed. From the data and the analysis, one can determine if the ICD accurately detected the arrhythmia and provided the appropriate electrical therapy as programmed. Sharing the information that the ICD functioned as programmed with the patient may reduce some of the patient's anxiety about the reliability of the device's functioning, but it may also increase concerns about changes in his or her health status that may be causing the increase in episodes of life-threatening arrhythmias [63,64]. It is important, therefore, to assess the patient's psychologic reaction by asking about his/her perception of the shocks and addressing any misconceptions; determining whether the patient is feeling anxious or helpless or seems panicked; and ensuring that the patient has a source of support (e.g., family, friends, clinical). If the patient is extremely distressed, stabilizing interventions such as anxiety-reducing medications may be offered, particularly if distress is considered to be a contributing cause of repeated arrhythmias [79].
After ICD interrogation confirms that the shocks were appropriate and that no device malfunction occurred, clinical management focuses on strategies to prevent high-energy shocks through other means of arrhythmia control. The treatment option of choice is the addition of antiarrhythmic medications. Although some antiarrhythmic medications have been linked to increased mortality in patients with heart disease who do not have an ICD, many antiarrhythmic medications may be safely used in persons with an ICD to prevent repeated defibrillation shocks [5]. Antiarrhythmic drugs may be used in persons with an ICD to suppress episodes of nonsustained VT, suppress episodes of sustained VT, slow the rate of VT and increase the efficacy of ATP in terminating the arrhythmia, and prevent the syncope that may be associated with faster rates of VT.
When antiarrhythmic drugs are used, the potential interaction between the actions of the antiarrhythmic drugs and the defibrillator's ability to depolarize the heart effectively should also be considered. Proarrhythmic effects of antiarrhythmic medications can result in the increased frequency of VT, and they can also convert nonsustained VT to sustained VT. Due to changes in conduction caused by antiarrhythmic medications, use of these drugs may make it more difficult for the ICD to sense the heart's rhythm accurately and may result in an increase in the defibrillation threshold (i.e., the energy required to depolarize the fibrillating heart effectively) [79]. Defibrillation threshold testing may be indicated to identify the appropriate setting after the addition of antiarrhythmic drugs. Because of negative inotropic effects on myocardial contraction, the use of some antiarrhythmic drugs can also exacerbate congestive heart failure, creating more arrhythmias or making it more difficult for the ICD to terminate the arrhythmias. To facilitate the efficacy of antiarrhythmic drugs and limit negative side effects, only a single antiarrhythmic drug should be used, if possible. Research has found that the use of multiple antiarrhythmic medications is not helpful, as it can increase the risk of new ventricular arrhythmias and may exacerbate underlying congestive heart failure [1,8,59,63,64].
Factors that increase the likelihood that ventricular arrhythmias will reoccur include the presence of decompensated heart failure, poorly controlled coronary artery disease or recurrent ischemia, and electrolyte imbalances [22]. "Fine-tuning" the patient's medical regimen for optimal management of underlying cardiovascular disease and correction of electrolyte imbalances can help to limit recurrent episodes of VT and ventricular fibrillation. In addition, for a select group of patients, the use of radiofrequency ablation may be used to eliminate re-entry circuits and reduce the frequency of ventricular arrhythmias [79].
Patient Z, a man 75 years of age with ischemic cardiomyopathy, presents to the emergency room with the complaint that his ICD had fired three times over the last 24 hours. When asked, he reports one shock in the early morning and two shocks about three hours apart in the late afternoon. He is placed on continuous telemetry monitoring; the monitor shows frequent runs of nonsustained VT. When his ICD is interrogated, the data confirm that his ICD discharged at the approximate times Patient Z reported and that the discharges were appropriate. On physical exam, Patient Z appears ill and slightly dehydrated. He has a low-grade fever, but his other vital signs are relatively unchanged from his norm. When questioned, Patient Z reports that he has had "the flu" for a couple of days; he reports severe episodes of vomiting and diarrhea. He admits to continuing to take his furosemide but says he has been unable to "keep down" food or his potassium supplements. Lab tests show that his potassium level is 2.8 mEq/L and his magnesium level is 1.2 mg/dl. Immediate IV supplements of potassium and magnesium are administered. The patient is admitted to an inpatient unit for monitoring and stabilization. Patient Z's electrolyte levels return to normal, and he recovers from his illness. No further ectopy is noted. A review of his record finds that Patient Z tends to go into VT when his potassium levels drop. Due to the patient's sensitivity to hypokalemia, the treating physician decides to add an oral antiarrhythmic to Patient Z's regimen. Patient Z is subsequently discharged home.
As the prevalence of device implantation has risen, healthcare providers in community and hospital settings have begun to see an increase in terminal patients with ICDs. Questions arise about the impact of the device during the dying process. Will it prolong the patient's pain and suffering? Will it prolong the dying process? Will the device fire as the patient is dying? Is it legal or ethical to deactivate an ICD? Under what circumstances should the device be deactivated? How does a do not resuscitate (DNR) order and deactivation of an ICD relate? Who should deactivate the device? What documentation should be placed in the patient's medical record? Who can make the decision to deactivate the device?
As with many decisions about end-of-life care, the decision to deactivate an ICD can be a complex and difficult one for patients and families. Consider these clinical scenarios:
Patient X is a man 86 years of age with a long history of ischemic heart disease and severe congestive heart failure. He had a biventricular pacemaker/ICD implanted three years ago. Until recently, he has remained active. Now he is admitted to the hospital with increasing dementia and severe pneumonia. Over time, he fails to respond to medical therapy; his pneumonia worsens, and his kidneys begin to fail. After discussion with the patient and family, the attending physician writes a DNR order; however, the patient and family state that they are not ready to turn off the ICD at this point. Aggressive therapy is stopped, and only comfort measures are continued. The patient becomes unresponsive; the physician tells the family that the patient's status has deteriorated and that the patient is likely to go into VT or ventricular fibrillation. The physician then asks the family if they would like to turn the patient's ICD off at this point, gently pointing out that allowing the ICD to shock the patient at this point resuscitates him, but the patient is likely to die from respiratory or other complications.
Patient AA is admitted from the emergency department with a diagnosis of ICD storm. He is 50 years of age and has NYHA Class IV ischemic cardiomyopathy. Patient AA has a combination biventricular pacemaker/ICD. The patient has reported 10 shocks over a 12-hour period. The cardiologist interrogates the device and determines that all shocks were appropriate (8 for VT, 2 for ventricular fibrillation). The device history also shows that the patient received ATP therapy 10 times over a two-week period for VT. ATP was successful in terminating the arrhythmia. Patient AA has a history of multiple MIs, previous CABG surgery and multiple stent placements, and ischemic cardiomyopathy. An echocardiogram shows that the patient's EF has dropped from 25% to approximately 10%. Patient AA is not a candidate for heart transplant. Over a period of few days, the physicians assess Patient AA's status and adjust medications in an attempt to optimize heart function. Despite optimization of his medical therapy, the patient continues to show signs of worsening heart failure and has persistent runs of nonsustained VT and VT terminated by antitachycardia pacing. The medical team approaches Patient AA about a DNR order, and he consents. But, because Patient AA understands that turning off the ICD also turns off the antitachycardia pacing, he refuses to have his ICD deactivated at this time, stating that he is not ready to die. After further discussion with the medical team and his family, the patient specifies a DNR order that states his ICD should be deactivated if it fires three high-energy shocks in succession.
Guidelines published by the Device-Based Therapy Writing Group acknowledge issues related to end-of-life care for persons with ICDs and note the following points [4]:
Some research has shown that dying patients can experience painful defibrillations throughout the process of dying, including the final moments before death.
Deactivation of an ICD at the request of a dying patient or the patient's surrogate decision maker is legal and ethical in the United States.
The decision to deactivate an ICD should be preceded by a discussion with the patient and/or healthcare surrogate about the consequences and alternatives to device deactivation. A summary of the discussion and its outcome should be recorded in the medical record.
The patient's medical record should include both a DNR order and an order for deactivation of the ICD.
At the time of ICD implantation, device education should include information about end-of-life issues.
Persons with ICDs should be encouraged to express their wishes related to device management and deactivation as part of an advance directive.
Antiarrhythmic drug therapy for the management of ventricular arrhythmias has moved from a primary role in prevention of sudden cardiac death and reduction of arrhythmias to, in many cases, an adjunct therapy prescribed to facilitate the functioning of an ICD. In the past, PVCs were thought to be a reliable indicator of the patient's risk for lethal arrhythmias. Because Class I antiarrhythmic medications were known to suppress PVCs, the assumption was made that their routine use would prevent episodes of ventricular arrhythmias and sudden cardiac death. The Cardiac Arrhythmia Suppression Trial (CAST) looked at the efficacy of several Class I antiarrhythmic medications in patients following MI [85]. The data indicated that Class I antiarrhythmics were harmful in the study population and that their use increased morbidity and mortality. Other clinical trials looked at the efficacy of implantable defibrillators versus antiarrhythmic medications. Data from those trials indicated that antiarrhythmic therapy, whether guided by electrophysiology testing, ambulatory ECG (Holter) monitoring, or empiric choice, was not as effective as an ICD in the patient populations involved in the studies [85]. As a result of the data from these trials, the recommendations for the use of antiarrhythmic drugs and the specific agents of choice changed. The recommendations included the following points [85]:
Use of Class I antiarrhythmic drugs should be avoided in persons with episodes of nonsustained VT, ischemic heart disease, and left ventricular dysfunction.
The routine use of Class I antiarrhythmic drugs is not recommended for use in persons who do not have an ICD.
Consideration may be given to the use of Class I antiarrhythmic drugs for suppression of troublesome ventricular arrhythmias in persons who have an ICD.
Asymptomatic PVCs that occur in persons following recovery from MI should not be routinely suppressed with antiarrhythmic drugs; the risks of serious side effects and proarrhythmic effects outweigh the potential benefits.
Antiarrhythmic drugs may be prescribed for persons who are at high risk for sudden cardiac death and who are not candidates for ICD therapy or other intervention to terminate the arrhythmias.
However, later studies questioned some of these results [86].
As negative data regarding the use of Class I antiarrhythmics mounted, clinical and research interest turned to the use of amiodarone, a Class III antiarrhythmic. Several studies reported favorable data with the use of amiodarone in patients following acute MI; these included the Canadian Amiodarone Myocardial Infarction Arrhythmia Trial (CAMIAT) and the European Myocardial Infarction Amiodarone Trial (EMIAT) [87]. Favorable results with amiodarone sparked interest in some of the Class III antiarrhythmic drugs, including sotalol and dofetilide. Results with sotalol were not encouraging. One study, the Survival with Oral D-Sotalol (SWORD) trial involving persons with a history of a previous MI, reduced left ventricular function, and overt heart failure, was terminated early because of an excessive mortality rate [87,88]. Dofetilide, another Class III antiarrhythmic drug approved for use in the management of atrial and supraventricular arrhythmias, was studied in two clinical trials to evaluate its use in reducing the risk of sudden cardiac death in high-risk patients [89]. The data obtained did not find evidence that dofetilide reduced risk, but it did show that the use of dofetilide was not associated with increased mortality in persons with structural heart disease. Dofetilide increases the action potential and refractory period; it has been found to terminate re-entry tachycardias. It has also been found to reduce the defibrillation threshold for VT and ventricular fibrillation in persons undergoing ICD implantation [1,3,9,25,90].
The use of antiarrhythmic drugs in the management of ventricular arrhythmias should be influenced by five major factors: the patient's overall status, the presence or absence of co-existing cardiovascular and other medical conditions, the presence or absence of an ICD, the goals of medical therapy, and an analysis of the efficacy of the medication versus its potential risks. Different classes of antiarrhythmic medications may be used in the management of different forms of ventricular arrhythmias. Before looking at specific drugs, it may be helpful to quickly review the classification system of antiarrhythmic drugs and the general characteristics of each class.
In the early 1970s, a classification system for antiarrhythmic drugs was developed by Williams and later modified by Harrison [91]. An antiarrhythmic medication was classified according to its specific effect on the normal cardiac cycle. When the classification system was developed, it was believed that each antiarrhythmic medication had only one action. Modern electrophysiologic research has shown, however, that the mechanisms involved in the generation and spread of an electrical impulse throughout the heart are complex and that antiarrhythmic medications may impact more than one of these mechanisms. Although the classification system does not reflect the advances in the understanding of electrophysiology, it is still in use today. Table 6 summarizes the classification system [5,92].
CLASSIFICATION OF ANTIARRHYTHMIC MEDICATIONS
| Class | Action/Properties | Class Proarrhythmic Effects | Specific Agents | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ia |
|
|
| ||||||||||
| Ib |
|
|
| ||||||||||
| Ic |
| Re-entry arrhythmias |
| ||||||||||
| II |
|
|
| ||||||||||
| III |
|
|
| ||||||||||
| IV |
|
|
| ||||||||||
| |||||||||||||
When administering antiarrhythmic drugs and monitoring the patient's response, keep these points in mind [1,13,93]:
Because many antiarrhythmic drugs have a very narrow therapeutic range, small changes in dose or absorption may increase the serum level from therapeutic to toxic, resulting in dangerous side effects and the development of other arrhythmias. Likewise, small changes in dose or absorption may reduce the serum level from therapeutic to ineffective, resulting in a loss of control of the arrhythmia.
Multiple factors influence the absorption, metabolism, and excretion of antiarrhythmic medications. These may include the patient's renal function, liver function, and age-related physiologic changes. Elderly patients are more susceptible to the effects and side effects of antiarrhythmic medications than younger individuals.
Drug interactions can have a significant impact on the patient. Serious interactions may occur between two antiarrhythmic medications, between an antiarrhythmic and other cardiovascular medications, and between antiarrhythmics and noncardiac medications. Interactions that may occur include: A drug potentiates or inhibits the effectiveness of the antiarrhythmic drugs
A drug interferes with the normal absorption of an antiarrhythmic drug, thus reducing the serum level and the drug's effectiveness in controlling an arrhythmia
A drug interferes with the normal metabolism or excretion of the medication, thus increasing the serum levels and increasing the risk of toxicity
An antiarrhythmic drug potentiates or inhibits the therapeutic effects of other medications
An antiarrhythmic drug interferes with the normal absorption, metabolism, or excretion of another medication, thus reducing the serum level and the drug's effectiveness
When given concurrently with other antiarrhythmic drugs, an antiarrhythmic drug can have cumulative effects on heart rate and blood pressure.Table 7 lists some common drug-to-drug interactions.Table 8 provides an overview of available antiarrhythmic drugs.
DRUG-TO-DRUG INTERACTIONS OF ANTIARRYTHMIC DRUGS (AADs)
| AAD | AAD Increases the Level of Action | AAD Inhibits the Action | AAD Serum Levels Increased | AAD Serum Levels Decreased | Other Considerations and Comments | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Amiodarone |
| — | — | — |
| |||||||||||||
| Digoxin | — | — |
|
| Concurrent administration of medications such as thiazide diuretics, ticarcillin, or amphotericin B that cause hypokalemia can increase risk of digitalis toxicity. | |||||||||||||
| Disopyramide | Warfarin | — | Quinidine |
| Additive effects on heart rate, blood pressure, force of cardiac contraction can occur when administered concurrently with calcium channel blockers, beta blockers, or flecainide. | |||||||||||||
| Flecainide | Digoxin | — |
| — | Additive effects on heart rate, blood pressure, force of cardiac contraction when given concurrently with beta blockers, calcium channel blockers, or disopyramid. | |||||||||||||
| Procainamide | — | — |
| Alcohol | — | |||||||||||||
| Propafenone |
| — |
| — | — | |||||||||||||
| Propranolol | May alter effectiveness of insulin and oral hypoglycemic agents |
| — | Thyroid preparations | Additive myocardial depression possible when administered concurrently with general anesthesia, IV phenytoin, or verapamil. | |||||||||||||
| Quinidine |
| — |
|
| — |
ORAL ANTIARRYTHMIC THERAPY USED IN THE LONG-TERM MANAGEMENT OF VENTRICULAR ARRHYTHMIAS
| Drug (Class) | Action | Dosage | Side Effects | Comments | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Amiodarone (III) |
|
|
|
| ||||||||||||||||||||||
| Sotalol (II and III) |
|
|
|
| ||||||||||||||||||||||
| Procainamide (Ia)a |
| Initial IV loading dose: up to 17 mg/kg administered at 20–50 mg/minute or 100 mg at a rate of 20–50 mg/minute every 5 minutes. Administer until arrhythmia controlled, hypotension occurs, QRS complex widens by 50% of original width, or until maximum dose of 17 mg/kg is given. After arrhythmia is controlled, may continue 1–4 mg/minute for ongoing control. |
|
| ||||||||||||||||||||||
| Mexiletine (Ib) |
|
|
| |||||||||||||||||||||||
| Quinidine (Ia) |
|
|
|
| ||||||||||||||||||||||
| Propafenone (Ic) |
|
|
|
| ||||||||||||||||||||||
| Disopyramide (Ia) |
|
|
|
| ||||||||||||||||||||||
| Flecainide (Ic) |
|
|
|
| ||||||||||||||||||||||
| ||||||||||||||||||||||||||
Amiodarone has become one of the drugs of choice in the management of ventricular arrhythmias. Categorized as a Class III antiarrhythmic, amiodarone has been found to reduce sudden cardiac death episodes and improve mortality rates in some clinical trials [1,93,94]. It is approved for the treatment of documented, life-threatening recurrent ventricular fibrillation or hemodynamically unstable VT that has not responded to other available agents or when other agents are not well tolerated [37]. Amiodarone is an oral antiarrhythmic drug of choice for use in persons with compromised ventricular functioning.
Amiodarone prolongs action potential duration and the refractory period and slows sinus rate. On ECG, amiodarone can increase PR and QT intervals. Amiodarone has a slow and variable onset of action. A therapeutic effect may be achieved in several days, but it usually takes one to three weeks, even with the use of loading doses. It is eliminated slowly and remains for days following the discontinuation of oral therapy [37].
Chronic amiodarone therapy is associated with severe side effects, including pulmonary fibrosis, liver function abnormalities, photosensitivity, hyper- or hypothyroidism, acute respiratory distress syndrome, corneal microdeposits, and photophobia [1,93,94]. Other side effects include dizziness, fatigue, malaise, anorexia, and constipation [37].
It is recommended that amiodarone therapy be initiated in the hospital setting with continuous ECG monitoring. A loading dose may be administered initially by IV or by mouth, followed by decreasing oral doses at a set interval. Usual dosing includes [1,93,94]:
Rapid IV loading dose: 150 mg IV in 100 mL of D5W administered over 10 minutes
Oral loading dose: 400 mg every 8 to 12 hours for one to two weeks
Followed by: 300–400 mg per day in single or divided doses
Reduce to: 200–400 mg/day
Followed by: 200 mg per day as a maintenance dose
The lower loading and maintenance dosage should be considered for women, elderly patients, and all patients with low body weight [37]. Electrolyte imbalances, especially hypokalemia and hypomagnesemia, should be corrected before amiodarone therapy is initiated. Electrolyte levels should be periodically monitored in patients who are at risk to develop recurrent electrolyte imbalances, such as those with renal failure or those on chronic diuretic therapy [37].
Sotalol is categorized as both a Class II and a Class III antiarrhythmic with beta-adrenergic effects. It is a combination of two isomers: l-sotalol and d-sotalol. It is approved for use in the management of documented ventricular arrhythmias that are considered life-threatening [37]. It should not be used for the treatment of asymptomatic ventricular arrhythmias.
Class II effects slow the heart rate and the rate of conduction through the AV node. Class III effects increase the refractory period of the AV node, prolong ventricular action potential, and increase the effective refractory period in both atrial and ventricular muscle. On surface ECG, it can prolong the QT interval. At low doses, only beta blocking effects are seen. Doses of 160 mg per day or more are required before Class III actions are seen [37].
Proarrhythmic effects associated with sotalol include torsades de pointes, the development of new ventricular arrhythmias, and bradycardia. Sotalol may exacerbate congestive heart failure. The patient should be monitored for an excessively prolonged QT interval or signs of increasing renal compromise [37,93,95].
Only the oral form of sotalol is approved for use in the United States. The initial oral dose is 80–120 mg twice a day. The dose may be gradually increased to a maintenance dose of 160–320 mg in divided doses. The maximum recommended oral dose is 320 mg per day [37,93,95].
Sotalol should not be used in the presence of uncompensated congestive heart failure or bradycardia. It should be used cautiously in persons with renal impairment, pulmonary disease, or diabetes. Lower dosage should be considered with the elderly [37]. If the benefit outweighs the risk, the patient should be admitted to an inpatient setting with telemetry monitoring for the initiation of therapy.
Procainamide is a Class Ia antiarrhythmic medication approved for use in the treatment of documented ventricular arrhythmias, including PVCs and VT [37]. Treatment with procainamide has not been found to improve survival [1,93,94].
Procainamide increases the refractory period of the atria, the bundle of His-Purkinje system, and the ventricles. It slows conduction through most myocardial tissue, with variable effects in the AV node. Procainamide also decreases myocardial excitability. When metabolized, it converts to N-acetylprocainamide (NAPA), a metabolite with active antiarrhythmic properties [37].
Some persons who take procainamide can develop a positive antinuclear antibody titer with or without the symptoms of a lupus-like syndrome. Significant proarrhythmic effects may be seen with procainamide, including heart block and new or worsening ventricular arrhythmias [1,93,94]. Other side effects include diarrhea, anorexia, agranulocytosis, and seizures [37].
As stated, the oral forms of procainamide are no longer available in the United States. The initial IV loading dose is up to 17 mg/kg administered at 20–50 mg/minute or 100 mg at a rate of 20–50 mg/minute every 5 minutes [1,37,93,94].
The serum level of procainamide should be monitored; the therapeutic level is 4–10 mcg/mL. Toxicity develops with levels over 10–12 mcg/mL. NAPA levels should be monitored as well [37]. Clinical signs of toxicity can include confusion, dizziness, drowsiness, nausea, vomiting, and arrhythmias [93,94].
Mexiletine is a Class Ib antiarrhythmic drug. It is approved for the treatment of life-threatening ventricular arrhythmias. Due to proarrhythmic effects, mexiletine is not recommended for treatment of lesser ventricular arrhythmias or for asymptomatic PVCs [37].
Mexiletine alters sodium ion transport across the myocardial cell membrane; it decreases the rate of rise of the action potential and the effective refractory period in Purkinje fibers [37]. In patients with normal conducting systems, it does not prolong the QRS or QT.
Mexiletine has significant proarrhythmic effects. Other side effects include dizziness, dyspnea, heartburn, nausea, and vomiting [37,93,94].
An initial loading dose of 150–200 mg is given orally every 8 or 12 hours (may load with 400 mg if necessary). The dose may be increased by 50–100 mg every two to three days to a maximum of 1,200 mg per day. The usual maintenance dose is 200–300 mg every eight hours [37,93,94].
Use of mexiletine is contraindicated in second-or third-degree heart block unless the patient has a pacemaker [37].
Quinidine is a Class Ia antiarrhythmic medication with a long history of use in the management of ventricular arrhythmias. Due to its proarrhythmic effects, quinidine should be reserved for life-threatening arrhythmias; it has largely been replaced by safer and more effective agents and/or nonpharmacologic therapies (e.g., radiofrequency ablation) [37].
Quinidine decreases myocardial excitability and slows conduction velocity [37].
As mentioned, quinidine can have significant proarrhythmic effects, including the development of torsades de pointes. Side effects may be extremely troublesome and cause the patient to discontinue the medication independently. Side effects include nausea, anorexia, abdominal cramping, and diarrhea [37].
Quinidine sulfate in a dose of 200 mg is equal to 267 mg of quinidine gluconate [37]. A test dose of 200 mg taken orally may be administered to adults several hours prior to the full dose to test for adverse effects. Quinidine gluconate is rarely used parenterally. Common doses of quinidine sulfate are 200 mg every six hours or every eight hours for long-acting forms. The dose may be calculated as 15–60 mg/kg/day in four to five divided doses or 6 mg/kg every four to six hours [37,93,94].
Propafenone is a Class Ic antiarrhythmic medication. It is approved for the treatment of life-threatening ventricular arrhythmias. Due to the high risk of proarrhythmias, it should not be used in the treatment of lesser arrhythmias [37].
In addition to proarrhythmias, propafenone may cause dizziness, constipation, nausea, and vomiting [37].
Propafenone may be initiated at a dose of 150–300 mg every eight hours; the dose may be increased at three-to four-day intervals as needed to up to maximum dose of 300 mg every eight hours. Propafenone should be initiated in an inpatient setting with telemetry monitoring [37,93,94].
Use of propafenone is contraindicated in conduction disorder (unless the patient has a pacemaker), electrolyte imbalances, and uncontrolled congestive heart failure. Lower doses may be used in elderly patients [37].
Disopyramide is categorized as a Class Ia antiarrhythmic. It is approved for the suppression of unifocal and multifocal PVCs, couplets, and VT [37].
Disopyramide suppresses ventricular arrhythmias by reducing myocardial excitability and decreasing the speed of conduction. It has negative inotropic effects and therefore can reduce the force of contraction of the heart [37].
Common side effects include constipation, dry mouth, urinary hesitancy, and urinary retention [37].
The initial dose is 300 mg (immediate-release form) followed by 150–300 mg every 6 hours (immediate-release) or 300 mg every 12 hours (controlled-release) [37]. The total dose per day should not exceed 600 mg. For persons with compromised left ventricular function, the dose should be reduced to 200 mg (immediate-release) as an initial dose followed by 100 mg every 6 to 8 hours (immediate-release) or 200 mg every 12 hours (controlled-release) [37,93,94].
Use of disopyramide is contraindicated in the presence of severe conduction abnormalities, such as sick sinus syndrome and second-or third-degree heart block, unless the patient has a permanent pacemaker [37].
Flecainide is categorized as a Class Ic antiarrhythmic. It is approved for the suppression of documented life-threatening ventricular arrhythmias [37].
Flecainide suppresses ventricular arrhythmias by prolonging the refractory period, and slowing conduction in the atria, AV node, His-Purkinje system, and ventricles [37].
Common side effects include the increased risk of proarrhythmias, headache, and dizziness [37].
The initial oral dose is 50–100 mg twice a day. If indicated, the dose may be increased to 150 mg twice a day. The maximum dose is 400 mg/day [37,93,94].
Flecainide should be used very cautiously in persons with congestive heart failure. It may reduce left ventricular function and exacerbate episodes of congestive heart failure [37].
Radiofrequency ablation for VT is performed in the electrophysiology lab and may be used to reduce or suppress ventricular arrhythmias in the presence of cardiomyopathy. It is used to treat symptomatic VT rather than to reduce the risk of sudden cardiac death [22]. The use of radiofrequency ablation in the management of VT is increasing. It has been found to be successful for persons with structurally normal hearts and idiopathic VT, especially right ventricular outflow tract VT and left ventricular idiopathic VT caused by a re-entrant tachycardia involving the Purkinje network. Less frequently, radiofrequency ablation has been performed in persons with structural heart disease who may have multiple types of VT including a VT caused by a re-entrant circuit that is amenable to ablation. Candidates for possible ablation should be able hemodynamically to tolerate their VT for long periods of time. Mapping and ablation is usually most feasible in the presence of slower, monomorphic, hemodynamically stable VT. In radiofrequency ablation, either the origin or a critical point of conduction is identified. Once a location has been identified, radiofrequency energy is delivered for 30 to 60 seconds. The energy creates heat, and the heat damages the target tissue, thus breaking or disrupting the arrhythmia's conduction pathway [7,9,27,97].
Ablation of VT is recommended for patients who [5,97]:
Have sustained monomorphic VT that is drug resistant
Are intolerant of antiarrhythmic medications
Do not wish to use long-term drug therapy and do not have additional risk factors for sudden cardiac death
Are receiving multiple appropriate shocks for sustained VT that does not respond to programming changes or optimal drug therapy
The preparation for radiofrequency ablation is similar to that for cardiac catheterization. The physician should explain the procedure and obtain informed consent. Risks associated with radiofrequency ablation include bleeding at the insertion site(s), hematoma formation, pneumothorax, and risk of death from arrhythmias that are invoked during the test and not terminated. Prior to the procedure, the electrophysiologist will review any antiarrhythmic medications that the patient is taking and make a recommendation about tapering or stopping those medications before the study. The patient should be NPO for six to eight hours prior to the procedure. Routine lab work, including a CBC, electrolyte panel, prothrombin time/international normalized ratio, and urinalysis should be done. A chest x-ray and ECG are also performed [7,9,27,97]. The ECG is usually performed to assess the location of scarring, if any, and to assess the overall pumping function of the heart [97,98].
A VT ablation is carried out in a specially equipped cardiac catheterization lab. As with cardiac catheterization, the patient is monitored continuously for blood pressure, ECG, and oxygen saturation. Because electrophysiology procedures are frequently very long, a Foley catheter is inserted for comfort and to prevent complications from urinary retention. Intravenous heparin is usually administered to help prevent the formation of blood clots before, during, and after ablation [98]. The patient undergoes moderate sedation, usually with agents such as midazolam and fentanyl [97]. After the patient is sedated, vascular access, most commonly using the femoral vein, is obtained and catheters containing multiple electrodes are passed transvenously to desired positions inside the heart [97].
VT is induced and then a complex process of mapping the arrhythmia occurs. Mapping may include the identification of the arrhythmia's site of origin, spread of activation, and critical site(s) [97]. Only patients who can hemodynamically tolerate sustained VT for a prolonged period can undergo electrophysiology for mapping and possible ablation [7,9,27]. The procedure generally lasts from three to six hours [98].
Patient BB, a man 71 years of age, has been admitted to the hospital for management of ICD storm. He reports a several-month history of increased ICD firings that have persisted despite the addition of antiarrhythmic medications and changes in his ICD programming. Patient BB is very anxious and reports a significant decrease in activity tolerance. He has underlying congestive heart failure and coronary artery disease. Based on evaluation and assessment, the electrophysiologist decides to schedule Patient BB for electrophysiology for possible mapping and ablation of VT. His antiarrhythmic medications are discontinued for a 24-hour period prior to the scheduled test. He is anxious that the withdrawal of his medications will provoke episodes of his arrhythmia and frequently calls the telemetry desk to verify that his heart rhythm is normal. Patient BB's cardiac status is optimized through use of additional diuretic therapy and some alterations in medications for his congestive heart failure. He is taken to the electrophysiology lab. One form of his VT is successfully induced and mapped; mapping shows a re-entrant mechanism that is amenable to ablation. The pathway is ablated, and Patient BB is returned to his room following recovery from moderate sedation. Antiarrhythmic therapy is re-initiated. During the remainder of his hospital stay, the patient experiences no further episodes of VT and is discharged home.
Poorly controlled coronary artery disease and/or poorly controlled congestive heart failure greatly increase the risk that a patient will develop recurrent ventricular arrhythmias that are difficult to control. Overall management of a patient with ventricular arrhythmias includes optimal management of any underlying disease process.
In general, for persons with reduced left ventricular function, recommended medical management includes the use of a combination of angiotensin-converting enzyme (ACE) inhibitors, beta blockers, diuretic therapy, and often digitalis [99,100]. In addition to these medications, for persons with symptomatic heart failure, consideration may be given to the use of spironolactone (with normal renal function), angiotensin receptor blockade (especially when an ACE inhibitor is not tolerated because of side effects), or the use of a nitrate with hydralazine in persons who cannot take an ACE inhibitor because of renal insufficiency and/or hypotension. The AHA/ACC have developed updated guidelines for the evaluation and management of adults with chronic heart failure [101,102,103].
For persons with coronary artery disease, recommended medical management includes the use of beta blockers, aspirin, cholesterol-lowering agents, and (depending on the patient's status) long-acting nitrates. The AHA/ACC updated guideline for the management of patients with chronic stable angina includes the recommendation that ACE inhibitors be given to persons with a previous MI or who have significant coronary artery disease identified by cardiac catheterization [99,100]. In addition, the AHA/ACC assert that ACE inhibitors are a reasonable option for use in lower-risk patients (e.g., mildly reduced or normal LVF, well-controlled cardiovascular risk factors) in whom revascularization has been performed [100]. As always, the optimal management of this group also includes measures to reduce risk, including pharmacotherapy and lifestyle changes to reduce low-density lipoprotein cholesterol levels, cessation of smoking, increased physical activity, etc. [100,101].
Surgery to resect or reconstruct a left ventricle that has been severely damaged from a MI has been used to improve the heart's pumping ability and to reduce ventricular arrhythmias. When the left ventricle is damaged by an MI, an area of scar tissue forms; the size of the scar depends on the extent or severity of the MI. The presence of scar tissue in the heart interferes with the heart's ability to contract normally, producing signs of reduced cardiac output. The scar tissue also interferes with normal conduction of electrical impulses and creates conditions for ventricular arrhythmias to develop. Theoretically, reconstruction of the ventricle to remove the scar tissue permanently from the areas of functioning myocardium should improve the heart's pumping ability and reduce the incidence of arrhythmias. Various techniques have been developed to identify and separate the area of scar. If the area of scar is large, the dead tissue may be surgically removed and a patch applied. In some cases, at least two rows of circulation stitches may be placed around the border of the scar tissue; the stitches are pulled together so the scarred area is permanently separated from the functioning tissue. Reconstructive surgery is a high-risk procedure involving heart-lung bypass and is not indicated for all patients who have reduced left ventricular function following MI. Other names for reconstructive surgery (or similar procedures) include endoventricular circular patch plasty repair, Dor procedure, surgical ventricular restoration, and left ventricular aneurysmectomy reconstruction [104,105].
Long-term follow-up studies have shown that medical care costs are higher in patients with ICDs than in those without [4]. Some clinical trials have included measurements of both cost and clinical effectiveness. Based on available data, the Writing Group for Device-Based Therapy notes that [4]:
Proper patient selection is crucial to cost-effective device therapy. Improper patient selection increases medical care costs.
Theoretically, ICD therapy will be more cost effective when used for patients who have an increased risk for sudden cardiac death but a low risk for death from other causes.
For patients with reduced left ventricular function, additional risk stratification to better identify specific groups of patients who would benefit from ICD therapy might improve cost effectiveness.
Further improvement of the reliability and length of life of the device itself coupled with a reduction in cost of the device would also improve cost effectiveness.
Data from the COMPANION trial indicates that combination cardiac resynchronization therapy biventricular pacemaker and ICD is cost effective with regards to accepted benchmarks for therapeutic interventions [4,106]. Risk-benefit ratio becomes an issue when considering low-risk patients (i.e., asymptomatic or mildly symptomatic heart failure patients) and data is lacking on the risk-benefit ratio of combination therapy in NYHA class I and class II patients [4,106].
AEDs have been found to be effective in reducing sudden cardiac death when external defibrillation occurs within minutes of the onset of ventricular fibrillation [5,10]. Because a large percentage of cardiac arrests occur in the home setting, the placement of AEDs in the home setting is receiving increased attention. The U.S. Food and Drug Administration (FDA) has approved AEDs for home use in families with inherited arrhythmias, such as long QT syndrome and hypertrophic cardiomyopathy. The FDA has also approved over-the-counter sales of AEDs [5]. Insurance coverage and cost of devices may be a barrier to obtaining a home AED for some individuals and families.
Wearable external defibrillators have been approved by the FDA for prevention of sudden cardiac death in persons with a transient high risk for ventricular fibrillation. Examples include patients at high risk for ventricular fibrillation following MI or invasive cardiac procedure and those who require temporary removal of an infected ICD and antibiotic therapy before re-implantation of a permanent device. A wearable defibrillator is a vest-like device that is worn under an individual's clothing. Through multiple external electrodes, the device continuously monitors heart rhythm and automatically delivers a shock when ventricular fibrillation is detected. Proper function requires that the device electrodes be properly placed per the manufacturer's guidelines. The device should be worn continuously except while bathing or showering [5]. Use of wearable defibrillators can provide a cost-effective alternative to a prolonged hospital stay for patients who require a lengthy course of antibiotics before undergoing re-implantation of an ICD [5].
In November 2017, the FDA announced that it is requiring manufacturers to obtain premarket approval for all future and currently marketed AEDs and necessary AED accessories (e.g., pad electrodes, batteries). The FDA action is the result of manufacturing problems and the recall of several AED devices. AEDs currently on the market will remain available while manufacturers work to meet the new premarket approval process. The FDA has published a list of all AEDs that have received premarket approval [107,108].
Patient CC schedules an appointment with his primary care physician because he has noticed that his heartbeat is irregular at times. Patient CC is 74 years of age and retired with a history of coronary artery disease, an MI (12 years prior), CABG surgery (11 years prior), and stents placed in his coronary arteries (two years prior). During the appointment, Patient CC explains that he exercises regularly and always monitors his pulse rate during and after exercising. Recently, he says, his pulse has started "skipping beats," sometimes a few and sometimes "a lot." Since he noticed the missed beats, Patient CC has been checking his pulse off and on when he gets up and when he is resting and has noted that it has been increasingly irregular. Physical examination is negative for any changes or new abnormal findings. A routine 12-lead ECG is performed and shows no changes from previous ECGs. Routine lab work is done, and all values returned within normal limits. The physician prescribes a 48-hour Holter monitoring test.
Rationale and comments: Patient CC appears to have a type of arrhythmia that occurs relatively frequently. Ambulatory ECG monitoring such as a Holter monitor can provide helpful information about the type of arrhythmia and its link to any symptoms that Patient CC is having.
Patient CC completes the 48-hour monitoring test. As requested, he maintained a diary of any symptoms and his activities. Analysis of the Holter monitoring data showed that Patient CC was experiencing frequent runs of nonsustained monomorphic VT. Patient CC is notified of the results and is admitted to a local hospital for further testing and evaluation.
Rationale and comments: Asymptomatic (or relatively asymptomatic) runs of nonsustained VT may develop in persons with ischemic heart disease. Patient CC has had a MI; the damage from the infarction creates conditions that predispose him to develop a re-entry VT, such as monomorphic VT. The onset nonsustained VT may be an indicator that Patient CC is experiencing more ischemia. Diagnostic tests to assess for the presence of reversible ischemia are often indicated.
Patient CC is admitted to a telemetry unit for continuous telemetry monitoring. Serial cardiac enzymes obtained at eight-hour intervals are negative. Serum electrolytes are obtained; Patient CC's potassium and magnesium levels are within normal limits. Given Patient CC's past history of coronary artery disease, bypass surgery, and stent placement for re-occlusion of one of his grafts, the decision is made to perform a left heart catheterization. His catheterization shows a reduced EF of 30%; the stent and previous bypass grafts were found to be open with no significant blockages.
Rationale and comments: Myocardial ischemia from inadequate blood flow to the myocardium may cause an increase in ventricular arrhythmias. If ischemia is present, the treatment of choice is restoration of blood flow through percutaneous coronary procedures or coronary artery bypass graft surgery. However, Patient CC is not experiencing myocardial ischemia. His increasing ectopy is probably related to the damage from his previous MI and his increasing heart failure.
Over a period of days, Patient CC's medications are adjusted to optimize his heart function and halt further ventricular remodeling and progression of his heart failure. Following accepted standards for heart failure management, Patient CC is placed on a beta blocker and an ACE inhibitor along with furosemide for diuresis and potassium for replenishment as needed[109,110]. Despite optimization of his medications, Patient CC continues to experience frequent runs of nonsustained VT.
Rationale and comments: For patients with heart failure, primary prevention of sudden cardiac death involves aggressive management of heart failure symptoms and disease progression.
The medical team decides to evaluate Patient CC for device therapy. Because he has significant heart failure, the team chooses to evaluate him for combination cardiac resynchronization therapy and ICD. A 12-lead ECG and echocardiogram show that Patient CC would benefit from resynchronization therapy in addition to the ICD.
Rationale and comments: Patient CC has risk factors for sudden cardiac death, including reduced left ventricular function, ischemic heart disease, the continued presence of PVCs, and episodes of nonsustained VT after CABG surgery. Electrophysiology is indicated to determine if he is at high risk. If he is, implantation of an ICD is indicated. Some patients with heart failure who require an ICD may benefit from cardiac resynchronization therapy as well. In cardiac resynchronization therapy, a biventricular pacemaker is implanted, leads are placed in the right and left ventricles, and the device is programmed to coordinate the contraction of the right and left ventricles to restore a more normal pattern of ventricular contraction. Re-synchronizing ventricular contraction has been shown to improve functional status.
The physician explains the proposed procedure to Patient CC and his wife, including the purpose of the test, its possible risks, and the different results that might be obtained. The physician also explains the purpose of a biventricular pacemaker/ICD, how it works, why it might be needed in Patient CC's case, and how it would be implanted. In addition, he provides Patient CC and his wife with written material as well as a video on device therapy provided by one of the ICD manufacturers for use in patient education. Patient CC is very anxious, and his wife questions the physician privately about whether the defibrillator "will really work." The physician spends time with Patient CC and his wife, answering their questions and listening to their concerns.
Rationale and comments: People who learn that they may have a life-threatening arrhythmia requiring the placement of an ICD are frequently very anxious and frightened and may express anger, frustration, denial, or other emotions. The patient and his family may have a lot of questions about whether the device is really necessary and whether it will actually work. This is a crisis time for Patient CC and his family. Taking time to listen to the patient's and family's concerns and providing basic information about an ICD and its function are appropriate in the preprocedure period.
Patient CC is taken to the electrophysiology lab. Monitoring catheters containing multiple electrodes are inserted into the venous system and advanced to the appropriate sites. Using an established protocol, programmed electrical stimulation is performed. Sustained monomorphic VT is induced. Based on the electrophysiologist's assessment of Patient CC's current and potential needs for pacing and defibrillation, a dual-chamber biventricular pacemaker/ICD is implanted. Following implantation, before Patient CC is returned to his room, the ICD is tested and found to respond appropriately and effectively to a life-threatening arrhythmia.
Rationale and follow-up: An approved indication for implantation of an ICD is the presence of inducible sustained VT in a person with coronary artery disease and a previous MI. The selection of a specific device is the responsibility of the individual implanting the device and is based on an assessment of the patient's current and potential needs for defibrillation therapy as well as for permanent pacing. A dual-chamber ICD provides sensing in both the atrium and the ventricle; the atrial-sensing capability facilitates its ability to recognize and differentiate between atrial and ventricular arrhythmias. ICD function is tested before the procedure is completed, while the patient is still sedated.
Patient CC is returned to the telemetry unit. His vital signs are monitored frequently, and the generator site under his left clavicle is checked for signs of swelling or bleeding. The bilateral femoral sites used for insertion of venous catheters for the electrophysiology test are monitored for signs of bleeding or hematoma formation. Patient CC is placed on continuous telemetry monitoring and remains in the hospital for an additional 48 hours. During this time, his vital signs are stable and he develops no complications from the procedures. Telemetry monitoring shows that he has occasional PVCs and some runs of monomorphic VT that are terminated by the programmed antitachycardia pacing.
Rationale and comments: ATP is used to terminate VT. A group of pacing stimuli is rapidly delivered to the heart through the pacing electrode at a rate faster than the rate of the VT to depolarize the site of the arrhythmia, thus terminating the re-entry cycle. Because ATP uses about the same amount of electrical energy as a permanent pacemaker, patients usually experience minimal sensation. Patients may notice a period of light-headedness or "fluttering" in the chest from the rapid rate of the VT.
Discharge education for Patient CC includes incision care, arm restrictions, safety measures, driving restrictions, and instructions regarding appropriate actions if the device fires. The importance of Patient CC keeping all scheduled follow-up appointments and adhering to prescribed therapy for the ongoing management of his ischemic heart disease is stressed. Patient CC and his wife are provided with information about a local support group for ICD recipients and their families and are given written information in addition to verbal instructions.
Rationale and comments: Keeping follow-up appointments is critical for persons with ICDs to permit assessment of device function, identification of any programming or device problems, and assessment of the status of the battery to permit timely replacement. The amount of information that people need following ICD implantation is considerable, and the short hospital stays associated with ICD implantation do not permit time to review all the necessary material. In addition, immediately following ICD implantation, patients and their families may still seem to be in a crisis situation; their ability to hear and remember detailed information may be lessened. Providing outside resources and written resources is very important. Education should be continued in outpatient settings when the patient returns for follow-up visits.
Patient DD is admitted via EMS to the emergency room of the local hospital. He reports that he has an ICD and that it has fired six to eight times over the last three to four days. A cardiologist experienced with electrophysiology interrogates the ICD and retrieves stored data on all detected arrhythmias and device therapy delivered. The interrogation confirms that the patient has received a total of six shocks over a period of 72 hours for polymorphic VT and ventricular fibrillation. In two instances, multiple shocks were required to terminate the arrhythmia. Patient DD is diagnosed with "ICD storm."
Rationale and comments: When a patient reports multiple discharges from an ICD, the ICD should be interrogated to determine if the shocks actually occurred as the patient reported and if the shocks were delivered correctly (i.e., did the device correctly detect the arrhythmia and deliver therapy appropriately). Shocks that the patient reports feeling but that do not appear in stored data when the device is interrogated are sometimes referred to as "phantom shocks." Both counseling and measures to reduce anxiety may be effective in reducing the patient's sensation of phantom shocks. Shocks that are delivered inappropriately may reflect programming issues or equipment problems. Multiple shocks that are delivered over a short period for detected VT/ventricular fibrillation reflect appropriate ICD function; the condition is referred to as ICD or electrical storm.
A history and physical are performed. Patient DD is assessed for symptoms of recurrent ischemia or exacerbation of congestive heart failure. A 12-lead ECG is ordered along with an electrolyte panel and cardiac enzymes. A list of Patient DD's current medications is obtained, and Patient DD's adherence to his prescribed medications is evaluated.
Rationale and comments: In a person with an ICD and known ischemic heart disease, recurrent arrhythmias requiring ICD therapy may be triggered by changes in the patient's status, including recurrent or worsening ischemia and decompensated congestive heart failure. Indications of ischemia may include elevated cardiac enzymes and identifiable changes on an ECG from the patient's previous ECG. Indications of decompensated failure may include rales in the lung fields, edema in dependent sites, pulmonary edema present on chest x-ray, obvious weight gain, hypotension, report of diminished activity tolerance, dyspnea, and reduced oxygen saturation. Lack of adherence to prescribed antiarrhythmic therapy may cause an increase in arrhythmic episodes. In addition, failure to follow prescribed therapy for congestive heart failure or ischemic heart disease may result in recurrent ischemia or a congestive heart failure exacerbation.
Patient DD is admitted to an inpatient telemetry monitoring unit for further assessment, evaluation, and management. The 12-lead ECG shows no new changes, and his cardiac enzymes are negative. His serum electrolytes show slightly decreased serum potassium and serum magnesium levels. A chest x-ray shows mild signs of pulmonary edema. Patient DD denies any decrease in activity tolerance or other subjective symptoms of decompensated heart failure but does admit that the swelling in his ankles has gotten "a little worse." Patient DD's pre-admission medications are continued; these include digoxin, an ACE inhibitor, and a beta blocker for management of his heart failure and ischemic heart disease along with low-dose amiodarone for suppression of episodes of VT/ventricular fibrillation. The physician places Patient DD on IV diuretic therapy (instead of the patient's usual oral dose) for a brief period and restricts his fluid limit to 1,500 mL/day. Doses of his medications are adjusted, and consideration is given to adding an angiotensin receptor blocker in addition to Patient DD's other medications. Supplemental potassium and magnesium are ordered to restore Patient DD's serum levels to normal.
Rationale and comments: Appropriate initial intervention for ICD storm includes evaluating the efficacy of measures to control heart failure and ischemic heart disease. Correction of underlying electrolyte abnormalities, especially potassium and magnesium imbalances, is also important in decreasing the risk that the patient will develop recurrent arrhythmias.
With IV diuretic therapy and adjustments to his medications, Patient DD's signs of heart failure resolve. His activity tolerance improves, his chest x-ray is clear, and the swelling in his feet and ankles diminishes. As Patient DD and his family talk about his probable discharge one afternoon, Patient DD suddenly loses consciousness and slumps forward in his chair. Simultaneously, the ventricular fibrillation lethal arrhythmia alarm sounds at the central monitor station. Responding to the emergency, the staff note that Patient DD is unresponsive and without palpable pulse. Patient DD's ICD delivers a high-energy shock; the energy spike is noted on telemetry. The staff in Patient DD's room note seizure-like movement of his body. Patient DD does not wake up, and the monitor shows that he is remaining in ventricular fibrillation. As the staff begins to prepare to return Patient DD to bed in anticipation for the need for external defibrillation, the ICD fires a second time, terminating the arrhythmia and restoring a normal, perfusing rhythm. Patient DD is placed on a lidocaine drip and transferred to an intermediate coronary care unit.
Rationale and comments:People with ICDs who go into ventricular fibrillation may lose consciousness from the absence of a perfusing rhythm before the ICD has time to fire. Multiple ICD discharges for a single event of arrhythmia do not indicate a problem with ICD function. ICD programming includes a monitoring function. Once the ICD has fired, it monitors the patient's response. If the patient remains in ventricular fibrillation, the ICD is programmed to deliver another shock rapidly to terminate the rhythm. Depending on the programming, an ICD can deliver at least three shocks to terminate ventricular fibrillation. In the event that an ICD does not terminate a ventricular arrhythmia, external defibrillation may be performed. The defibrillator pads/paddles should not be applied directly over the ICD generator site. Lidocaine is an appropriate medication to use for continued suppression of ventricular arrhythmias after ventricular fibrillation has been terminated. The usual rate of infusion is 2–4 mg/min.
Patient DD remains on the lidocaine infusion for several days. His oral amiodarone dose is increased. He is evaluated for the presence of active myocardial ischemia, but cardiac catheterization shows no areas of reversible ischemia. Following discontinuation of his lidocaine drip, Patient DD is transferred back to a general telemetry unit for monitoring. Telemetry monitoring shows no additional episodes of ventricular fibrillation, and Patient DD's overall status remains stable. However, because his amiodarone dose has been significantly increased, the physicians decide to test his ICD to make sure that the defibrillation threshold has not been altered by the increased medication. Patient DD is taken to the electrophysiology lab, where he is sedated. Ventricular fibrillation is induced, the arrhythmia is detected, and his ICD fires, terminating the arrhythmia. Patient DD is returned to the telemetry unit and is discharged on the following day.
Rationale and comments: Increasing the dose of an antiarrhythmic medication or adding additional antiarrhythmic medications can change the amount of energy the defibrillator must deliver to the myocardium in order to defibrillate the heart. ICD testing is a useful procedure that can be performed in the electrophysiology lab. Because high-energy shocks are painful, patients are sedated for this procedure. Adjustments in programming can be made noninvasively if indicated by the results of the testing procedure.
Clinical management of a person with ventricular arrhythmias focuses on evaluation of the individual's risk for sudden cardiac death and on control of troublesome symptoms. The recommended intervention for people at high risk for sudden cardiac death is the implantation of an ICD. ICD therapy has evolved to include not only delivery of a high-energy shock for defibrillation, but also ATP and cardioversion for the termination of VT. ICDs combine sophisticated pacing features, such as dual-chamber pacing, rate-responsive pacing, and pacing for cardiac resynchronization. Other therapies available for management of ventricular arrhythmias include the careful use of antiarrhythmic drug therapy, radiofrequency ablation, left ventricular reconstruction surgery, and optimization of medical management for any underlying cardiovascular disease. Clinicians should have a working knowledge of the management of ventricular arrhythmias as this patient population grows.
Action potential: term used to describe the electrical activity that occurs in individual myocardial cells.
Accelerated idiopathic ventricular rhythm: abnormal ventricular rhythm characterized by an absence of P waves, widened QRS complexes, and a rate of 50–100 bpm.
Cardioversion, synchronized: method of applying a direct current shock through the chest to the myocardium; in synchronized cardioversion, the shock is timed to fall at the peak of the R wave.
Electromagnetic interference: occurs when one electronic device disrupts the functioning of another electronic device.
ICD storm: occurrence of multiple appropriate shocks delivered by an ICD for VT or ventricular fibrillation within a 24-hour period (also called "electrical storm").
Inducible: term used to describe the outcome of an electrophysiology test when programmed electrical stimulation is successful in causing sustained VT or ventricular fibrillation.
Monomorphic: type of VT characterized by QRS complexes that are uniform in configuration in a single ECG lead.
Non-inducible: term used to describe the outcome of an electrophysiology test when programmed electrical stimulation is unsuccessful in producing sustained VT or ventricular fibrillation.
Polymorphic: type of VT characterized by continuously changing QRS complexes in a single ECG lead.
Proarrhythmia: the ability of an antiarrhythmic drug to produce new or worsening arrhythmias as a side effect.
Programmed electrical stimulation: specific protocols used during electrophysiology testing to elicit specific arrhythmias.
Radiofrequency ablation: invasive interventional procedure performed in the electrophysiology lab using radiofrequency waves to destroy a specific group of myocardial cells; the purpose of radiofrequency ablation is to interrupt an abnormal conduction pathway to eliminate an arrhythmia.
Rate-responsive pacing: pacemaker feature that allows the pacemaker to pace the heart at a more rapid rate when indicated by the body's metabolic demands.
Re-entry mechanism: abnormal conduction pattern in which an electrical impulse is able to repeatedly stimulate the heart to depolarize, thus creating an arrhythmia.
Refractory period: period of time in the cardiac cycle when the myocardial cell is unable to respond to an electrical stimulus.
Supraventricular tachycardia: type of arrhythmia that is initiated at or above the level of the AV junction in the heart.
| American Heart Association |
| 7272 Greenville Avenue |
| Dallas, TX 75231 |
| https://www.heart.org |
| (800) 242-8721 |
| European Society of Cardiology (ESC) |
| https://www.escardio.org/Guidelines/Clinical-Practice-Guidelines |
| Heart Rhythm Society (HRS) |
| 1325 G Street NW, Suite 400 |
| Washington, DC 20005 |
| https://www.hrsonline.org |
| (202) 464-3400 |
| Medtronic |
| 710 Medtronic Parkway |
| Minneapolis, MN 55432-5604 |
| https://www.medtronic.com |
| (763) 514-4000 |
1. Podrid PJ, Kowey PR. Cardiac Arrhythmia: Mechanisms, Diagnosis, and Management. 2nd ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2001.
2. Eckardt L, Haverkamp W, Breithardt G. Antiarrhythmic therapy in heart failure. Heart Fail Monit. 2002;2(4):110-119.
4. Tracy CM, Epstein AE, DiMarco JP, et al. 2012 ACCF/AHA/HRS focused update of the 2008 guidelines for device-based therapy of cardiac rhythm abnormalities: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2013;61(3):e6-e75.
5. Al-Khatib SM, Stevenson WG, Ackerman MJ, et al. 2017 AHA/ACC/HRS guideline for management of patients with ventricular arrhythmias and the prevention of sudden cardiac death: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2018;72(4):e91-e220.
6. Zeppenfeld K, Tfelt-Hansen J, de Riva M, et al. 2022 ESC guidelines for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death: developed by the Task Force for the Management of Patients with Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death of the European Society of Cardiology (ESC). Endorsed by the Association for European Paediatric and Congenital Cardiology (AePC). Eur Heart J. 2022;43(40):3997-4126.
7. Curtis AB. Demise of the routine electrophysiologic study for survivors of life-threatening ventricular arrhythmias. Am Heart J. 2002;144(3):380-382.
8. Bing Liem L. Implantable Cardioverter-Defibrillator: A Practical Manual. Boston, MA: Kluwer Academic Publishers; 2001.
9. Brodsky MA, Mitchell LB, Halperin BD, Raitt MH, Hallstrom AP. Prognostic value of baseline electrophysiology studies in patients with sustained ventricular tachyarrhythmia: the Antiarrhythmics Versus Implantable Defibrillators (AVID) trial. Am Heart J. 2002;144(3):478-484.
10. American Heart Association. 2015 American Heart Association guidelines update for cardiopulmonary resuscitation and emergency cardiovascular care. Circulation. 2015;132(18 Suppl 2):S315-S573.
12. Toth P, Cannon CP. Comprehensive Cardiovascular Medicine in the Primary Care Setting. New York, NY: Springer Science Business Media; 2010.
13. Woods SL, Froelicher ES, Motzer SA, Bridges EJ. Cardiac Nursing. 6th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2010.
17. Mayo Clinic Staff. Heart Arrhythmia: Symptoms and Causes: Overview. Available at https://www.mayoclinic.org/diseases-conditions/heart-arrhythmia/symptoms-causes/syc-20350668. Last accessed October 31, 2023.
18. Bennet DH. Cardiac Arrhythmias: Practical Notes in Interpretation and Treatment. 7th ed. London: Hodder Arnold; 2006.
19. Wagner GS. Marriott's Practical Electrocardiography. 12th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2013.
20. Keany JE. Premature Ventricular Contraction. Available at https://emedicine.medscape.com/article/761148-overview. Last accessed October 31, 2023.
21. MedlinePlus. Ventricular Tachycardia. Available at https://medlineplus.gov/ency/article/000187.htm. Last accessed October 31, 2023.
22. Compton SJ. Ventricular Tachycardia. Available at https://emedicine.medscape.com/article/159075-overview. Last accessed October 31, 2023.
23. Chapman M, Hargreaves M, Schneider H, Royle M. Bidirectional ventricular tachycardia associated with digoxin toxicity and with normal digoxin levels. Short Comm. 2014;11(7):1222-1225.
24. Merck Manuals. Ventricular Tachycardia (VT). Available at https://www.merckmanuals.com/home/heart-and-blood-vessel-disorders/abnormal-heart-rhythms/ventricular-tachycardia. Last accessed October 31, 2023.
25. Weigner MJ, Buxton AE. Nonsustained ventricular tachycardia: a guide to the clinical significance and management. Med Clin North Am. 2001;85(2):305-320.
26. Pezeshkian NG. Accelerated Idioventricular Rhythm. Available at https://emedicine.medscape.com/article/150074-overview. Last accessed October 31, 2023.
28. Thaler MS. The Only EKG Book You'll Ever Need. 8th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2015.
29. Koplan BA, Stevenson WG. Ventricular tachycardia and sudden cardiac death. Mayo Clin Proc. 2009;84(3):289-297.
30. Habash-Bseiso DE, Rokey R, Berger CJ, Weier AW, Chyou PH. Accuracy of noninvasive ejection fraction measurement in a large community-based clinic. Clin Med Res. 2005;3(2):75-82.
31. Nguyen VQ, Celebi MM, Suleman A, Sander GE. Dilated Cardiomyopathy. Available at https://emedicine.medscape.com/article/152696-overview. Last accessed October 31, 2023.
32. Brieler J, Breeden MA, Tucker J. Cardiomyopathy: an overview. Am Fam Physician. 2017;96(10):640-646.
33. Braunwald E, Zipes DP, Libby P, Bonow R. Braunwald's Heart Disease: A Textbook of Cardiovascular Medicine. 9th ed. Philadelphia, PA: Elsevier; 2012.
34. American Heart Association. Hypertrophic Cardiomyopathy (HCM). Available at https://www.heart.org/en/health-topics/cardiomyopathy/what-is-cardiomyopathy-in-adults/hypertrophic-cardiomyopathy. Last accessed October 31, 2023.
35. Diez P, Brugada J. How to recognize and manage idiopathic ventricular tachycardia. Eur Soc Cardiol. 2010;8(25).
36. Dave J. Torsade de Pointes. Available at https://emedicine.medscape.com/article/1950863-overview#aw2aab6b5. Last accessed October 31, 2023.
37. Lexicomp Online. Available at https://online.lexi.com. Last accessed October 31, 2023.
38. Sovari AA, Kocheril AG, Assadi R, Baas AS. Long QT Syndrome. Available at https://emedicine.medscape.com/article/157826-overview. Last accessed October 31, 2023.
39. MedlinePlus. Ventricular Fibrillation. Available at https://medlineplus.gov/ency/article/007200.htm. Last accessed October 31, 2023.
40. Goyal SK, Rottman JN. Ventricular Fibrillation. Available at https://emedicine.medscape.com/article/158712-overview. Last accessed October 31, 2023.
41. Cummins RO, Ornato JP, Thies WH, Pepe PE. Improving survival from sudden cardiac arrest: the "chain of survival" concept: a statement for health professionals from the Advanced Cardiac Life Support Subcommittee and the Emergency Cardiac Care Committee, American Heart Association. Circulation. 1991;83(5):1832-1847.
42. American Heart Association. Out-of-Hospital Chain of Survival. Available at https://cpr.heart.org/en/resources/cpr-facts-and-stats/out-of-hospital-chain-of-survival. Last accessed October 31, 2023.
43. American Heart Association. 2020 American Heart Association Guidelines for CPR and ECC. Available at https://cpr.heart.org/en/resuscitation-science/cpr-and-ecc-guidelines. Last accessed October 31, 2023.
44. Aufderheide TP, Stapleton E, Hazinski MF, Cummins R. Instructor's Manual: Heartsaver AED. Dallas, TX: American Heart Association; 1998.
45. Koster RW. Automatic external defibrillator: key link in the chain of survival. J Cardiovasc Electrophysiol. 2002;13(1 Suppl): S92-S95.
46. Cardiac Monitoring.com. What is Mobile Cardiac Telemetry? Available at http://cardiacmonitoring.com/patient-information/what-is-mobile-cardiac-telemetry. Last accessed October 31, 2023.
47. Martinez-Rubio A, Kanaan N, Borggrefe M, et al. Advances for treating in-hospital cardiac arrest: safety and effectiveness of a new automatic external cardioverter-defibrillator. J Am Coll Cardiol. 2003;41(4):627-632.
48. American Heart Association. What Is an Automated External Defibrillator? Available at https://www.heart.org/-/media/files/health-topics/answers-by-heart/what-is-an-aed.pdf. Last accessed October 31, 2023.
49. Beinart SC. Synchronized Electrical Cardioversion. Available at https://emedicine.medscape.com/article/1834044-overview. Last accessed October 31, 2023.
50. January CT, Wann LS, Calkins H, et al. 2019 AHA/ACC/HRS focused update of the 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation. J Am Coll Cardiol. 2019;74:104-132.
51. Karliner LS, Jacobs EA, Chen AH, Mutha S. Do professional interpreters improve clinical care for patients with limited English proficiency? A systematic review of the literature. Health Serv Res. 2007;42(2):727-754.
52. Ngo-Metzger Q, Massagli MP, Clarridge BR, et al. Linguistic and cultural barriers to care: perspectives of Chinese and Vietnamese immigrants. J Gen Intern Med. 2003;18(1):44-52.
53. National Heart, Lung and Blood Institute. Holter and Event Monitors. Available at https://www.nhlbi.nih.gov/health/heart-tests. Last accessed October 31, 2023.
54. Medtronic. Daily Living: Pacemakers. Available at https://www.medtronic.com/uk-en/patients/treatments-therapies/pacemaker/living-with/daily-living.html. Last accessed October 31, 2023.
55. Heart Rhythm Society. Atrial Fibrillation. Available at https://upbeat.org/heart-rhythm-disorders/atrial-fibrillation#axzz3Iz01qUAn. Last accessed October 31, 2023.
56. Mayo Clinic. Mobile Cardiac Telemetry. Available at https://cardiovascularservices.mayoclinic.com/mobile-cardiac-telemetry-tech/. last accessed October 31, 2023.
57. Varma N, Cygankiewicz I, Turakhia MP, et al. 2021 ISHNE/HRS/EHRA/APHRS expert collaborative statement on mHealth in arrhythmia management: digital medical tools for heart rhythm professionals: from the International Society for Holter and Noninvasive Electrocardiology/Heart Rhythm Society/European Heart Rhythm Association/Asia-Pacific Heart Rhythm Society. Circulation. 2021;14(2):e009204.
58. Sroubek J, Probst V, Mazzanti A, et al. Programmed ventricular stimulation for risk stratification in the Brugada syndrome: a pooled analysis. Circulation. 2016;133:622-630.
60. Beyerbach DM. Pacemakers and Implantable Cardioverter-Defibrillators. Available at https://emedicine.medscape.com/article/162245-overview#a1. Last accessed October 31, 2023.
61. Epstein AE, DiMarco JP, Ellenbogen KA, et al. 2012 ACCF/AHA/HRS focused update incorporated into the ACCF/AHA/HRS 2008 guidelines for device-based therapy of cardiac rhythm abnormalities. A report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2013;61(3):e6-e75.
62. Knight L, Livingston NA, Gawlinski A, DeLurgio DB. Caring for patients with third-generation implantable cardioverter defibrillators: from decision to implant to patient's return home. Crit Care Nurse. 1997;17(5):46-61.
63. White E. Patients with implantable cardioverter defibrillators: transition to home. J Cardiovasc Nurs. 2000;14(3):42-52.
64. Doughtery CM. The natural history of recovery following sudden cardiac arrest and internal cardioverter-defibrillator implantation. Prog Cardiovasc Nurs. 2001;16(14):163-168.
65. Baddour LM, Epstein AE, Erickson CC, et al. Update on cardiovascular implantable electronic device infections and their management: a scientific statement from the American Heart Association. Circulation. 2010;121(3):458-477.
66. Maskarinec SA, Thaden JT, Cyr DD, Ruffin F, Souli M, Fowler VG. The risk of cardiac device-related infection in bacteremic patients is species specific: results of a 12-year prospective cohort. Open Forum Infect Dis. 2017;4(3):ofx132.
67. Ben Abid F, Al-Saoub H, Howadi F, Aibishawi A, Thapur M. Delayed pacemaker generator pocket and lead primary infection due to Burkholderia Cepacia. Am J Case Rep. 2017;18:855-858.
68. Mayo Clinic. Implantable Cardioverter-Defibrillators (ICDs). Available at https://www.mayoclinic.org/tests-procedures/implantable-cardioverter-defibrillators/about/pac-20384692. Last accessed October 31, 2023.
69. Steinke EE. Sexual concerns of patients and partners after an implantable cardioverter defibrillator. Dimens Crit Care Nurs. 2003;22(2):89-96.
70. Ocampo CM. Living with an implantable cardioverter defibrillator: impact on the patient, family, and society. Nurs Clin North Am. 2000;35(4):1019-1030.
71. Bleakley JF, Akiyama T. Driving and arrhythmias: implications of new data. Card Electrophysiol Rev. 2003;7(1):77-79.
72. Akiyama T, Powell JL, Mitchell LB, Ehlert FA, Baessler C. Resumption of driving after life-threatening ventricular tachyarrhythmia. N Engl J Med. 2001;345(6):391-397.
73. Schulmeister L. Pacemakers and environmental safety: what your patient needs to know. Nursing. 1998;28(7):58-60.
74. American Heart Association. Living with Your Implantable Cardioverter Defibrillator. Available at https://www.heart.org/en/health-topics/arrhythmia/prevention--treatment-of-arrhythmia/living-with-your-implantable-cardioverter-defibrillator-icd. Last accessed October 31, 2023.
75. Al-Khadra AS. Implantation of pacemakers and implantable cardioverter defibrillators in orally anticoagulated patients. Pacing Clin Electrophysiol. 2003;26(1 Pt 2):511-514.
76. Swerdlow CD, Friedman PA. Advanced ICD troubleshooting: part II. Pacing Clin Electrophysiol. 2006;29(1):70-96.
77. Rosenqvist M, Beyer T, Block M, den Dulk K, Minten J, Lindemans F. Adverse events with transvenous implantable cardioverter-defibrillators: a prospective multicenter study. Circulation. 1998;98(7):663-670.
78. Korte T, Jung W, Spehl S, et al. Incidence of ICD lead system related complications during long-term follow-up: comparison of epicardial and endocardial electrode system. Pacing Clin Electrophysiol. 1995;18(11):2053-2061.
79. Braunschweig F, Boriani G, Bauer A, et al. Management of patients receiving ICD shocks: electrical storm. Europace. 2010;12(12):1673-1690.
80. Marill KA. Ventricular Fibrillation in Emergency Medicine. Available at https://emedicine.medscape.com/article/760832-overview. Last accessed October 31, 2023.
81. Reddy VY, Reynolds MR, Neuzil P, et al. Prophylactic catheter ablation for the prevention of defibrillator therapy. N Engl J Med. 2007;357(26):2657-2665.
82. Israel CW, Barold SS. Electrical storm in patients with an implanted defibrillator: a matter of definition. Ann Noninvasive Electrocardiol. 2007;12(4):375-382.
83. Credner SC, Klingenheben T, Mauss O, Sticherling C, Hohnloser SH. Electrical storm in patients with transvenous implantable cardioverter-defibrillators: incidence, management and prognostic implications. J Am Coll Cardiol. 1998;32(7):1909-1915.
84. Exner DV, Pinski SL, Wyse DG, et al. Electrical storm presages nonsudden death: the antiarrhythmics versus implantable defibrillators (AVID) trial. Circulation. 2001;103(16):2066-2071.
85. The Cardiac Arrhythmia Suppression Trial (CAST) Investigators. Preliminary report: effect of encainide and flecainide on mortality in a randomized trial of arrhythmia suppression after myocardial infarction. N Engl J Med. 1989;321(6):406-412.
86. Domanski MJ, Zipes DP, Schron E. Treatment of sudden cardiac death: current understandings from randomized trials and future research directions. Circulation. 1997;95(12):2694-2699.
87. Borggrefe M, Haverkamp W, Breithardt G. Clinical implications of the CAMIAT, EMIAT and SWORD trials: a European perspective. Card Electrophysiol Rev. 1998;2(1):34-35.
88. Pratt CM. Clinical implications of the Survival with Oral D-Sotalol (SWORD) Trial: an investigation of patients with left ventricular dysfunction after myocardial infarction. Card Electrophysiol Rev. 1998;2(1):28-29.
89. Torp-Pedersen C, Møller M, Bloch-Thomsen PE, et al. Dofetilide in patients with congestive heart failure and left ventricular dysfunction. N Engl J Med. 1999;341(12):857-865.
90. Steinberg JS, Martins J, Sadanandan S, et al. Antiarrhythmic drug use in the implantable defibrillator arm of the Antiarrhythmics Versus Implantable Defibrillators (AVID) study. Am Heart J. 2001;142(3):520-529.
91. Vaughan Williams EM. A classification of antiarrhythmic actions reassessed after a decade of new drugs. J Clin Pharmacol. 1984;24(4):129-147.
92. Williams ES, Viswanathan MN. Current and emerging antiarrhythmic drug therapy for ventricular tachycardia. Cardiol Ther. 2013;2(1):27-46.
93. Deglin JH, Vallerand AH, Sanoski CA. Davis's Drug Guide for Nurses. 17th ed. Philadelphia, PA: FA Davis Company; 2020.
94. RxList. Available at https://www.rxlist.com/script/main/hp.asp. Last accessed October 31, 2023.
95. Heidenreich PA, Keeffe B, McDonald KM, Hlatky MA. Overview of randomized trials of antiarrhythmic drugs and devices for prevention of sudden cardiac death. Am Heart J. 2002;144(3):422-430.
96. Daily Med. Available at https://dailymed.nlm.nih.gov/dailymed/index.cfm. Last accessed October 31, 2023.
97. Greenberg ML. Catheter Ablation. Available at https://emedicine.medscape.com/article/1828541-overview. Last accessed October 31, 2023.
98. Tung R, Boyle NG, Shivkumar K. Catheter ablation of ventricular tachycardia. Circulation. 2010;122:e389-e391.
99. Gibbons RJ, Abrams J, Chatterjee K, et al. ACC/AHA 2002 guideline update for the management of patients with chronic stable angina—summary article: a report of the American College of Cardiology/American Heart Association Task Force on practice guidelines (Committee on the Management of Patients With Chronic Stable Angina). J Am Coll Cardiol. 2003;41(1):159-168.
100. Fraker TD Jr, Fihn SD, Gibbons RJ, et al. 2007 chronic angina focused update of the ACC/AHA 2002 guidelines for the management of patients with chronic stable angina: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines Writing Group to Develop the Focused Update of the 2002 Guidelines for the Management of Patients with Chronic Stable Angina. Circulation. 2007;116(23):2762-2772.
101. Hunt SA, Abraham WT, Chin MH, et al. ACC/AHA 2005 guideline update for the diagnosis and management of chronic heart failure in the adult: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Update the 2001 Guidelines for the Evaluation and Management of Heart Failure): developed in collaboration with the American College of Chest Physicians and the International Society for Heart and Lung Transplantation: Endorsed by the Heart Rhythm Society. Circulation. 2005;112(12):e154-e235.
102. Jessup M, Abraham WT, Casey DE, et al. 2009 focused update: ACCF/AHA guidelines for the diagnosis and management of heart failure in adults. J Am Coll Cardiol. 2009;53(15):1343-1382.
103. Hunt SA, Abraham WT, Chin MH, et al. 2009 focused update incorporated into the ACC/AHA 2005 guidelines for the diagnosis and management of heart failure in adults: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines: developed in collaboration with the International Society for Heart and Lung Transplantation. Circulation. 2009;119(14):e391-e479.
104. Wellens F, Geelen P, Demirsoy E, et al. Surgical treatment of tachyarrhythmias due to postinfarction left ventricular aneurysm with endoaneurysmorrhaphy and cryoablation. Eur J Cardiothorac Surg. 2002;22(5):771-776.
105. Di Donato M, Sabatier M, Dor V, RESTORE Group. Surgical ventricular restoration in patients with postinfarction coronary artery disease: effectiveness on spontaneous and inducible ventricular tachycardia. Semin Thorac Cardiovasc Surg. 2001;13(4):480-485.
106. Daubert JC, Leclercq C, Mabo P. Cardiac resynchronization therapy in combination with implantable cardioverter-defibrillator. Europace. 2009;11(Suppl 5):v87-v92.
107. U.S. Food and Drug Administration. Automated External Defibrillators (AEDs). Available at https://www.fda.gov/medical-devices/cardiovascular-devices/automated-external-defibrillators-aeds. Last accessed October 31, 2023.
108. U.S. Food and Drug Administration. Press Release. FDA Reaches Agreement with Automatic External Defibrillator Manufacturer Over Quality Control Issues. Available at https://www.fda.gov/news-events/press-announcements/fda-reaches-agreement-automatic-external-defibrillator-manufacturer-over-quality-control-issues. Last accessed October 31, 2023.
109. Heart Failure Society of America. Executive summary: HFSA 2010 comprehensive heart failure practice guideline. J Cardiac Failure. 2010;16(6):475-539.
110. Yancy CW, Jessup M, Bozkurt B, et al. 2017 ACC/AHA/HFSA focused update of the 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Failure Society of America. Circulation. 2017;1-75.
1. National Institute for Health and Care Excellence. Implantable Cardioverter Defibrillators and Cardiac Resynchronization Therapy for Arrhythmias and Heart Failure (Review of TA95 and TA120). London: National Institute for Health and Care Excellence; 2014. Available at https://www.nice.org.uk/guidance/ta314. Last accessed December 15, 2023.
2. Epstein AE, DiMarco JP, Ellenbogen KA, et al. 2012 ACCF/AHA/HRS focused update incorporated into the ACCF/AHA/HRS 2008 guidelines for device-based therapy of cardiac rhythm abnormalities: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2013;61(3):e6-e75. Available at https://www.ahajournals.org/lookup/doi/10.1161/CIR.0b013e318276ce9b. Last accessed December 15, 2023.
Mention of commercial products does not indicate endorsement.