Pharmacogenetics is the study of the impact that inheritance and genetic variability can have on drug response and ultimately the selection of a specific drug for a particular individual. The use of pharmacogenetic testing is becoming an increasing well recognized process in the medical and scientific communities. The ability to determine the types of responses may be elicited by a particular drug serves as a monumental breakthrough in terms of optimizing drug therapy and improving healthcare outcomes. Because it has become possible to identify specific genes and their connection to drug response in certain individuals, this has both garnered both supporters and opponents who view significant ethical impactions with the growing adoption of this new healthcare advancement.
This course is designed for physicians, nurses, physician assistants, pharmacists, and pharmacy technicians who assess and make decisions regarding the appropriate pharmacotherapy for their patients.
The purpose of this course is to educate healthcare professionals about pharmacogenetics and its application in drug selection and therapeutic interventions.
Upon completion of this course, you should be able to:
- Describe the background of pharmacogenetic testing.
- Discuss the types of pharmacogenetic testing and their applications.
- Examine the ethical implications of pharmacogenetic testing.
Abimbola Farinde, PharmD, PhD, is a clinical pharmacy specialist who has gained experience in the field and practice of psychopharmacology/mental health and geriatric pharmacy. She has worked with active-duty soldiers with dual diagnoses of a traumatic brain injury and a psychiatric disorder, providing medication therapy management and disease state management. Dr. Farinde has also worked with mentally impaired and developmentally disabled individuals at a state-supported living center. Her different practice experiences have allowed her to develop and enhance her clinical and medical writing skills over the years. Dr. Farinde always strives to maintain a commitment toward achieving professional growth as she transitions from one phase of her career to the next.
Contributing faculty, Abimbola Farinde, PharmD, PhD, has disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
John M. Leonard, MD
Mary Franks, MSN, APRN, FNP-C
Randall L. Allen, PharmD
The division planners have disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
Sarah Campbell
The Director of Development and Academic Affairs has disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
The purpose of NetCE is to provide challenging curricula to assist healthcare professionals to raise their levels of expertise while fulfilling their continuing education requirements, thereby improving the quality of healthcare.
Our contributing faculty members have taken care to ensure that the information and recommendations are accurate and compatible with the standards generally accepted at the time of publication. The publisher disclaims any liability, loss or damage incurred as a consequence, directly or indirectly, of the use and application of any of the contents. Participants are cautioned about the potential risk of using limited knowledge when integrating new techniques into practice.
It is the policy of NetCE not to accept commercial support. Furthermore, commercial interests are prohibited from distributing or providing access to this activity to learners.
Supported browsers for Windows include Microsoft Internet Explorer 9.0 and up, Mozilla Firefox 3.0 and up, Opera 9.0 and up, and Google Chrome. Supported browsers for Macintosh include Safari, Mozilla Firefox 3.0 and up, Opera 9.0 and up, and Google Chrome. Other operating systems and browsers that include complete implementations of ECMAScript edition 3 and CSS 2.0 may work, but are not supported. Supported browsers must utilize the TLS encryption protocol v1.1 or v1.2 in order to connect to pages that require a secured HTTPS connection. TLS v1.0 is not supported.
The role of implicit biases on healthcare outcomes has become a concern, as there is some evidence that implicit biases contribute to health disparities, professionals' attitudes toward and interactions with patients, quality of care, diagnoses, and treatment decisions. This may produce differences in help-seeking, diagnoses, and ultimately treatments and interventions. Implicit biases may also unwittingly produce professional behaviors, attitudes, and interactions that reduce patients' trust and comfort with their provider, leading to earlier termination of visits and/or reduced adherence and follow-up. Disadvantaged groups are marginalized in the healthcare system and vulnerable on multiple levels; health professionals' implicit biases can further exacerbate these existing disadvantages.
Interventions or strategies designed to reduce implicit bias may be categorized as change-based or control-based. Change-based interventions focus on reducing or changing cognitive associations underlying implicit biases. These interventions might include challenging stereotypes. Conversely, control-based interventions involve reducing the effects of the implicit bias on the individual's behaviors. These strategies include increasing awareness of biased thoughts and responses. The two types of interventions are not mutually exclusive and may be used synergistically.
#95103: An Introduction to Pharmacogenetic Testing
The role of genetic factors in an individual's response to drug therapy has become an area of increasing interest in medicine and pharmacy[1]. Over the years, significant progress has been made toward the application of pharmacogenetic testing when it comes to the selection of drug therapies for a variety of patient populations [2]. The U.S. Food and Drug Administration's (FDA's) Center for Drug Evaluation and Research (CDER) has begun to require some manufacturers to provide pharmacogenetic-specific information on product labels to alert prescribers that pharmacogenetic testing may be required prior to the first dose of a drug or drug class [2,3]. A proficiency testing program for pharmacogenetics was introduced by the College of American Pathologists (CAP), in part with the goal of identifying drugs that should be relabeled by the FDA CDER. Since the inception of the program, five genes with significant impact on the response to drug testing have been recognized [2]. These initial steps have been instrumental in fostering a growing awareness of the application of pharmacogenetic testing, which, while limited, has the opportunity to grow with ongoing and future research [4].
Pharmacogenetics is the study of how an individual's genetic makeup affects his or her response to drugs. It is a well-defined discipline of pharmacology, with principles that can be applied to the practice of medicine and nursing [5]. All genes in the body can be analyzed, and each drug interacts with numerous proteins in each gene. The presence of genetic variations in a large number of genes can affect the response to the drug. Eventually, the use of pharmacogenetics may lead to genotype-based individualized therapy instead of the traditional approach of one-size-fits-all pharmacotherapy [5].
The basis of pharmacogenetics is the belief that drug response can be fueled by pharmacokinetic and pharmacodynamic changes spurred by an individual's genotype. The discovery of a new disease susceptibility gene can lead to more individualized prescriptions and an increased likelihood that an individual is able to receive the most effective drug and dose during initial therapy [5]. It is estimated that genetic factors can be attributed to 20% to 95% of the variations that occur in individual responses to medications, and the identification of these gene variants has the potential to impact an individual's ultimate outcome [6]. This serves as the basis for pharmacogenetic testing as a method of determining which drugs can provide optimal treatment for any given patient [6].
During the years since the development of the proficiency testing program for pharmacogenetics, several gene variants with significant impact on response to drug therapy have been identified, including [7,8,9]:
Variants in the gene for uridine diphosphate (UDP) glucuronosyltransferase 1A1 (UGT1A1)
Patients with variant alleles for cytochrome P450 or CYP2C9 (*2, *3)
Vitamin K epoxide reductase complex subunit
There are specific instances in which the identification and testing of these genes can prove to be important to clinical decision making. For example, testing for variations in UGT1A1 can be vital in the identification of individuals who are at increased risk for serious adverse effects with the administration of the chemotherapy drug irinotecan, including diarrhea and hematopoietic toxicity [2,4,10].
The clinical implementation of pharmacokinetic testing will be highly dependent on the applicability, usability, and accuracy of genotyping. There were initially few clinical guidelines that linked the result of pharmacogenetic tests to specific therapeutic recommendations, but systematic literature reviews have led to recommendations for 53 different drugs that are associated with the gene coding for CYP2D6, CYP2C19, CYP2C9, thiopurine-S-methyltransferase (TPMT), dihydropyrimidine dehydrogenase (DPD), vitamin K epoxide reductase (VKORC1), UGT1A1, HLA-B44, HLA-B*5701, CYP3A5, and factor V Leiden (FVL) [11,12]. Variability in the coding by these genes can help guide the selection and development of more effective therapeutic interventions with less potential for serious adverse effects.
With the increasing number of pharmacogenetic tests becoming available, the requirement by the FDA for drug labels to possess pharmacogenetic information may become a standard of practice over time [12,13]. The FDA maintains a table of data on nearly 300 drugs and 100 molecular biomarkers. The labeling for some, but not all, of the products listed in the table includes specific actions to be taken based on the biomarker information [3].
There is a divergence in the stance that regulatory authorities and clinicians take when it comes to pharmacogenetic testing. On one hand, there is an increased expectation for more personalized medicine development [14,15]. On the other hand, some experts recommend exercising caution, with a focus more on pharmacogenetic testing's ability to improve the likelihood of achieving benefit effects and lowering specific toxic effects rather than its potential to direct drug development [16].
Some of the more commonly used medications that have proven benefits from pharmacogenetic testing include tricyclic antidepressants, tamoxifen, atomoxetine, and other oncologic agents [2,4,9].
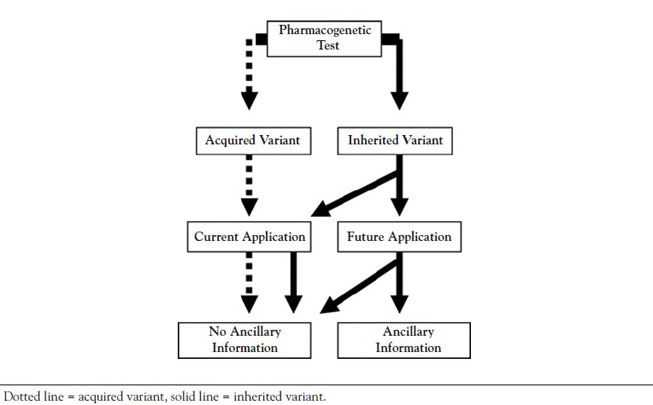
The ability to apply the concepts of pharmacogenetic testing to clinical practice is one of the most promising aspects of genomic research. The two key components in clinical settings are the type of genetic variation identified by the test (i.e., inherited or acquired) and the goal of the testing process (i.e., to address a specific issue or to gather information for future care) [17]. A classification framework has been developed to guide testing and treatment decisions related to pharmacogenetic testing (Figure 1) [17].
This classification consists primarily of tests for either acquired or inherited variants. Acquired genetic variants are involved in testing for possible tumor and/or other disease tissue growth prior to the selection of the most beneficial drug therapy [17]. For instance, tests for mutation in the epidermal growth factor gene may be done for patients with non-small cell lung cancer to determine which patients are more likely to respond to various tyrosine kinase inhibitors (e.g., afatinib, erlotinib, gefitinib, lapatinib, osimertinib, rociletinib, sunitinib, dasatinib) [18,19]. Testing for inherited genetic variations is intended to improve the future level of care. Patients will ultimately require drug treatment, which may be guided by information gleaned from the test results. As the use of prescription drugs continues to increase in the United States, the requirement for pharmacogenetic testing may as well. For example, there has been an increase in the use of warfarin algorithms to estimate the therapeutic dose for those with either existing genetic or nongenetic factors [19]. The algorithm uses linear regression models (y=ax+b) in order to arrive at the therapeutic warfarin dose, with factors identified as independent variables (i.e., the x variables) included in the model [20]. Take for example a white woman who is 70 years of age, is 5 feet 5 inches tall, weighs 70 kilograms, and carries VKORC1-1639AA and CYP2C9*1/*1 genotypes. She is a nonsmoker and is being treated for atrial fibrillation with a target INR of 2.5. Based on the model estimates, the warfarin dose is set at 3.74 mg/day. The algorithm provides an estimated warfarin dose, while a pharmacogenetic dosing table can assist with the selection of the first dose of warfarin if a patient has a CYP2CP and VKORC1 genotype [21,22]. This practice of pharmacogenetic testing is not required for the initiation of warfarin dosing; it is at the discretion of the clinician to order the test. Further research is necessary to determine the effectiveness of genotype-guided care over adherence to established clinical algorithms and guidelines.
Besides testing for acquired or inherited genetic variation to assist with the improvement of current care, there is also combined testing for inherited/acquired variants. In special circumstances, an individual may exhibit with both an acquired and an inherited variant affecting drug selection. For example, a drug belonging to the class of a tyrosine kinase inhibitor may be metabolized by a highly variable CYP3A4 enzyme. Therefore, in addition to testing for particular genetic predisposition, testing should also be performed to assess the level of CYP3A4 activity [21,22]. Whereas CYP3A4 activity may be affected by an inherited variant, an acquired variant may also impact how the patient reacts to the selected drug.
It is important that testing for acquired variants, inherited variants, or a combination of acquired/inherited variants is performed in a timely manner to help to guide and make treatment decisions. For example, if an elderly patient has been diagnosed with rectal cancer, the rapid testing process to produce a pharmacogenetic profile may lead to the immediate initiation of effective cancer treatment. The pharmacogenetic profile may guide the selection of a chemotherapeutic treatment plan that is more appropriate for this patient given that host genetic variability has been shown to be a vital component in the individualization of cancer treatment [23,24].
Various clinical practice guidelines recommend that pharmacogenetic tests should be performed for certain diseases or conditions in order to achieve optimal dosing or to guide drug/agent selection (Table 1)[4].
SELECTED CLINICAL PHARMACOGENETIC TESTING AND APPLICATION
| Disease/Condition | Drug/Agent | Genotype Test | Therapeutic Consideration |
|---|---|---|---|
| Anticoagulation therapy | Warfarin | CYP2C9 and VKORC1 | Optimal dose |
| Anti-platelet therapy for acute coronary syndrome | Clopidogrel | CYP2C19 | Switch to prasugrel or other alternative therapy |
| Tuberculosis | Isoniazid | NAT2 | Establish minimum effective dose |
| Metastatic colon cancer and other solid tumor cancers | High-dose irinotecan | UGT1A1 | Optimal initial dose |
| Immune diseases | Thiopurine family drugs (azathioprine, mercaptopurine, and thioguanine) | TPMT | Optimal initial dose |
| Non-small-cell lung cancer | Tyrosine kinase inhibitors | EGFR | Treatment selection |
| Breast cancer | Anti-HER2 therapies (e.g., lapatinib, pertuzumab, trastuzumab, trastuzumab emtansine) | HER2 | Treatment selection (i.e., decide use of HER2-targeted therapies) |
| Hormone-dependent breast cancer | Tamoxifen | CYP2D6 | Optimal dose |
| Metastatic colorectal cancer | Anti-EGFR-targeted drugs (e.g., cetuximab, panitumumab) | KRAS | Predict therapeutic effect |
| Attention deficit hyperactivity disorder | Atomoxetine | CYP2D6 | Optimal dose |
| Analgesia | Codeine | CYP2D6 | Optimal dose |
| Depression (and other uses) | Tricyclic antidepressants | CYP2D6 | Optimal dose |
Although pharmacogenetic testing can be a valuable tool to predict an individual's response to the administration of a drug, there are ethical issues regarding the growing use of this testing process [25]. Genome sequencing has the potential to remove the need for individual genetic formation tests, but there are concerns that sequencing may reveal information that some would prefer to not know (e.g., misattributed paternity, findings that cannot be addressed medically) [26]. It is important that all patients provide informed consent prior to any genetic testing. In addition to the right to know, patients also have the right not to know. There are risks associated with knowing (e.g., discovering disturbing information) and with not knowing (e.g., missing an opportunity to address an actionable issue), and these points should be explained and understood by each patient [25,27]. For patients for whom English is not their first language, forms should be presented in the language they feel most comfortable with. The use of a trained interpreter is also recommended in order to minimize any potential misunderstandings and liabilities.
Another ethical issue is whether the use of pharmacogenetic testing constitutes a reasonable allocation of scarce resources and whether it will widen the disparities in access to health care. In order to support the clinical utility of widespread pharmacogenetic testing, cost savings, such as the avoidance of adverse reactions and hospitalizations through the identification of genetic variants, should be demonstrated [28]. Due to the additional cost and potential reimbursement issues, pharmacogenetic testing may end up being unequally available to those with the means to pay for the services. In addition, drug companies may cease developing drugs to treat conditions that affect a small number of patients (as identified through genetic markers) [28].
There has also been debate as to whether pharmacogenetic testing should be performed on newborns, infants, and children. While newborn screening has become commonplace, genetic tests in children have not. The rationale for genetic testing should be clearly defined so patients and guardians are able to make informed consent [29]. If there is clinical relevance and a therapeutic intent behind the use of pharmacogenetic testing in infants and children, the test may be used, but a discussion should occur about the broader implications (e.g., how to proceed with secondary or ancillary findings) [25,29].
The confidentiality of stored genetic information is also a concern. There is a risk that insurance providers and/or employers may gain access to test results, violating patient confidentiality. Researchers reviewed 111 laboratories that offer clinical pharmacogenetic testing in the United States. Of these, 76 offered pharmacogenetic testing services, including 31 that offered tests only for specific genes, 30 that offered tests for multiple genes, and 15 that offered both types of tests. A total of 45 laboratories offered 114 multigene panel tests that cover 295 genes; however, no clinical guidelines were available for most of the tests [30]. Despite the lack of consensus on preventive pharmacogenetic testing, many healthcare organizations have implemented testing programs to obtain information regarding clinical validity and usefulness [31,32,33,34].
In 2008, U.S. Congress passed the Genetic Information Nondiscrimination Act (GINA). GINA makes it illegal for employers to require the purchase of genetic tests, prohibits insurance companies from using genetic information to adjust premiums, deny coverage, or impose restrictions related to pre-existing conditions, and bars companies with 15 or more employees from requiring or using genetic information for the purpose of employment [35]. The Patient Protection and Affordable Care Act of 2010 contains provisions that complement GINA and does little to alter GINA's approach to preventing genetic discrimination [35].
Because guidelines for testing and interpreting results are constantly changing, several organizations provide current data and integration with pharmacogenetic testing programs [36,37].
Although ethical and scientific concerns remain regarding this relatively new genetic technology, pharmacogenetic testing is undergoing substantial growth and may allow researchers and clinicians to engage in gene profiling in order to better predict patients' response to pharmacotherapy. This could ultimately result in the development of safer and more effective drug therapies and/or treatment protocols. The application of pharmacogenetic testing is becoming a reality for a variety of clinical conditions, and the subsequent drug developments may foster the widespread adoption of pharmacogenetic testing as a valid diagnostic and prescriptive tool in health care [2]. Some areas of pharmacogenetic testing have yet to be fully examined. However, to date it has helped to shape clinical guidelines and has the potential to provide greater insight for healthcare providers when it comes to effectively treating patients.
1. Gervasini G, Benítez J, Carrillo J. Pharmacogenetic testing and therapeutic drug monitoring are complementary tools for optimal individualization of drug therapy. Eur J Clin Pharmacol. 2010;66(8):755-774.
2. Wu AH. Genotype and phenotype concordance for pharmacogenetic tests through proficiency survey testing. Arch Pathol Lab Med. 2013;137(9):1232-1236.
3. U.S. Food and Drug Administration. Table of Pharmacogenomic Biomarkers in Drug Labeling. Available at https://www.fda.gov/drugs/science-and-research-drugs/table-pharmacogenomic-biomarkers-drug-labeling. Last accessed March 21, 2024.
4. Kim S, Yun YM, Chae HJ, et al. Clinical pharmacogenetic testing and application: laboratory medicine clinical practice guidelines.Ann Lab Med. 2017;37(2):180-193.
5. Shah RR, Shah DR. Personalized medicine: is it a pharmacogenetic mirage? Br J Clin Pharmacol. 2012;74(4):698-721.
6. Haga SB, Burke W, Ginsburg GS, Mills R, Agans R. Primary care physicians' knowledge of and experience with pharmacogenetic testing. Clin Genet. 2012;82(4):388-394.
7. Gage BF, Eby C, Johnson JA, et al. Use of pharmacogenetic and clinical factors to predict therapeutic dose of warfarin. Clin Pharmacol Ther. 2008;84(3):326-331.
8. Mega JL, Close SL, Wiviott SD, et al. Cytochrome P450 polymorphism and response to clopidogrel. N Engl J Med. 2009;360(4):354-362.
9. Goetz MP, Knox SK, Suman VJ, et al. The impact of cytochrome P450 2D6 metabolism in women receiving adjuvant tamoxifen.Breast Cancer Res Treat. 2007;101(1):113-121.
10. Innocenti F, Ratain MJ. Pharmacogenetics of irinotecan: clinical perspectives on the utility of genotyping. Pharmacogenomics. 2006;7(8):1211-1221.
11. Swen JJ, Nijenhuis M, de Boer A, et al. Pharmacogenetics: from bench to byte: an update of guidelines. Clin Pharmacol Ther. 2011;89(5):662-673.
12. Pop TR, Chirită DN, Buzoianu AD. Pharmacogenetics of oral anticoagulant therapy. Hum Vet Med. 2013;5(1):9-13.
13. Flockhart DA, Skaar T, Berlin DS, Klein TE, Nguyen AT. Clinically available pharmacogenomics tests. Clin Pharmacol Ther. 2009;86(1):109-113.
14. Alonso SG, de la Torre Díez I, Zapiraín BG. Predictive, personalized, preventive and participatory (4P) medicine applied to telemedicine and eHealth in the literature. J Med Syst. 2019;43(5):140.
15. Cardon LR, Harris T. Precision medicine, genomics and drug discovery. Hum Mol Genet. 2016;25(R2):R166-R172.
16. Hood E. Pharmacogenetics: the promise of personalized medicine. Environ Health Perspect. 2003;111(11):A581-A589.
17. Haga SP, Burke W. Pharmacogenetic testing: not as simple as it seems. Genet Med. 2008;10(6):391-395.
18. Tsao MS, Sakurada A, Cutz JC, et al. Erlotinib in lung cancer—molecular and clinical predictors of outcome. N Engl J Med. 2005;353(2):133-144.
19. Jackman DM, Yeap BY, Sequist LV, et al. Exon 19 deletion mutations of epidermal growth factor receptor are associated with prolonged survival in non-small cell lung cancer patients treated with gefitinib or erlotinib. Clin Cancer Res. 2006;12(13):3908-3914.
20. Cavallari L, Shin J, Perera, MA. Role of pharmacogenetics in the management of traditional and novel oral anticoagulants. Pharmacotherapy. 2011;31(12):1192-1207.
21. Limdi NA, Wadelius M, Cavallari L, et al. Warfarin pharmacogenetics: a single VKORC1 polymorphism is predictive of dose across three racial groups. Blood. 2010;115(18):3827-3834.
22. Li J, Zhao M, He P, Hidalgo M, Baker SD. Differential metabolism of gefitinib and erlotinib by human cytochrome P450 enzymes.Clin Cancer Res. 2007;13(12):3731-3737.
23. Cecchin E, Agostini M, Pucciarelli S, et al. Tumor response is predicted by patient genetic profile in rectal cancer patients treated with neo-adjuvant chemo-radiotherapy. Pharmacogenomics J. 2011;11(3):214-226.
24. Dreussi E, Cecchin E, Polesel J, et al. Pharmacogenetics biomarkers and their specific role in neoadjuvant chemoradiotherapy treatments: an exploratory study on rectal cancer patients. Int J Mol Sci. 2016;17(9):1482.
25. Carrieri D, Bewshea C, Walker G, et al. Ethical issues and best practice in clinically based genomic research: Exeter Stakeholders Meeting report. J Med Ethics. 2016;42(11):695-697.
26. Vogenberg FR, Barash CI, Pursel M. Personalized medicine, part 2: ethical, legal and regulatory issues. P T. 2010;35(11):624-626, 628-631, 642.
27. Hoop JG, Lapid MI, Paulson RM, Roberts LW. Clinical and ethical considerations in pharmacogenetic testing: views of physicians in three "early adopting" departments of psychiatry. J Clin Psychiatry. 2010;71(6):745-753.
28. Barash CI. Ethical Issues in Pharmacogenetics. Available at https://theatreofdebate.co.uk/wp-content/uploads/2020/12/Ethical-Issues-in-Pharmacogenetics-ActionBioscience.pdf. Last accessed March 21, 2024.
29. American Academy of Pediatrics. Ethical and policy in genetic testing and screening of children. Pediatrics. 2013;131(3):620-622.
30. Haga SB, Kantor A. Horizon scan of clinical laboratories offering pharmacogenetic testing. Health Aff (Millwood). 2018;37(5):717-723.
31. Janssens AC, Deverka PA. Useless until proven effective: the clinical utility of preemptive pharmacogenetic testing. Clin Pharmacol Ther. 2014;96(6):652-654.
32. Lazaridis KN. Improving therapeutic odyssey: preemptive pharmacogenomics utility in patient care. Clin Pharmacol Ther. 2017;101(1):39-41.
33. Bielinski SJ, Olson JE, Pathak J, et al. Preemptive genotyping for personalized medicine: design of the right drug, right dose, right time-using genomic data to individualize treatment protocol. Mayo Clin Proc. 2014;89(1):25-33.
34. Dunnenberger HM, Crews KR, Hoffman JM, et al. Preemptive clinical pharmacogenetics implementation: current programs in five U.S. medical centers. Annu Rev Pharmacol Toxicol. 2015;55:89-106.
35. Feldman EA. The Genetic Information Nondiscrimination Act (GINA): public policy and medical practice in the age of personalized medicine. J Gen Intern Med. 2012;27(6):743-746.
1. Schaefer GB, Mendelsohn NJ, Professional Practice and Guidelines Committee. Clinical genetics evaluation in identifying the etiology of autism spectrum disorders: 2013 guideline revisions. Genet Med. 2013;15(5):399-407. Available at https://www.acmg.net/PDFLibrary/Austism-Spectrum-Genetics-Evaluation.pdf. Last accessed April 19, 2024.
Mention of commercial products does not indicate endorsement.