No substance, legal or illegal, has a more paradoxical mythology than alcohol. It is undeniably one of the most widely and safely used intoxicants in the world; however, it is also potent and dangerous, both from a psychologic and physiologic viewpoint. Alcohol is currently responsible for more deaths and personal destruction than any other known substance of abuse, with the exception of tobacco. All of this is known with scientific certainty. Alcohol is legal, easily obtained, and supported by a multi-billion-dollar worldwide industry. Alcohol consumption reduces social inhibitions and produces pleasure and a sense of well-being. It also can have some rather impressive positive medical effects, such as a reduced risk of cardiovascular disease. This course will include core competencies related to alcohol use and abuse as well as knowledge, assessment, and treatment-based competencies.
- INTRODUCTION
- HISTORY
- CURRENT ESTIMATES OF ALCOHOL USE
- BENEFITS
- RISK AND PROTECTIVE FACTORS
- ALCOHOL USE DISORDER
- COMPLICATIONS
- OTHER PSYCHIATRIC DISORDERS ASSOCIATED WITH ALCOHOL USE DISORDERS
- DETECTING ALCOHOL USE DISORDERS
- BRIEF INTERVENTION
- TREATMENT
- CONCLUSION
- RESOURCES
- Works Cited
- Evidence-Based Practice Recommendations Citations
This course is designed for physicians, nurses, and allied health professionals involved in the treatment or care of patients who consume alcohol.
The purpose of this course is to address the ongoing alcohol competency educational needs of practicing physicians, nurses, and other healthcare providers. The material will include core competencies as well as knowledge, assessment, and treatment-based competencies.
Upon completion of this course, you should be able to:
- Review facts about the history, costs, and prevalence of alcohol use and abuse.
- Define moderate drinking and take a history of alcohol use as defined by the standard drink equivalency.
- Identify benefits reported in the literature for moderate alcohol consumption.
- Distinguish between genetic and environmental risk and protective factors for developing alcohol problems.
- Describe clinical characteristics of alcohol use disorder, intoxication, and withdrawal.
- List complications associated with alcohol use disorders.
- Recognize mental health diagnoses associated with alcohol use disorders.
- Discuss screening instruments for detecting alcohol use disorders, including considerations for non-English-proficient patients.
- Explain brief intervention efficacy and techniques.
- Describe and evaluate treatment modalities.
Mark S. Gold, MD, DFASAM, DLFAPA, is a teacher of the year, translational researcher, author, mentor, and inventor best known for his work on the brain systems underlying the effects of opiate drugs, cocaine, and food. Dr. Gold was a Professor, Eminent Scholar, Distinguished Professor, Distinguished Alumni Professor, Chairman, and Emeritus Eminent Scholar during his 25 years at the University of Florida. He was a Founding Director of the McKnight Brain Institute and a pioneering neuroscience-addiction researcher funded by the NIH-NIDA-Pharma, whose work helped to de-stigmatize addictions and mainstream addiction education and treatment. He also developed and taught courses and training programs at the University of Florida for undergraduates and medical students.
He is an author and inventor who has published more than 1,000 peer-reviewed scientific articles, 20 text books, popular-general audience books, and physician practice guidelines. Dr. Gold was co-inventor of the use of clonidine in opioid withdrawal and the dopamine hypothesis for cocaine addiction and anhedonia. Both revolutionized how neuroscientists and physicians thought about drugs of abuse, addiction, and the brain. He pioneered the use of clonidine and lofexidine, which became the first non-opioid medication-assisted therapies. His first academic appointment was at Yale University School of Medicine in 1978. Working with Dr. Herb Kleber, he advanced his noradrenergic hyperactivity theory of opioid withdrawal and the use of clonidine and lofexidine to ameliorate these signs and symptoms. During this time, Dr. Gold and Dr. Kleber also worked on rapid detoxification with naloxone and induction on to naltrexone.
Dr. Gold has been awarded many state and national awards for research and service over his long career. He has been awarded major national awards for his neuroscience research including the annual Foundations Fund Prize for the most important research in Psychiatry, the DEA 30 Years of Service Pin (2014), the American Foundation for Addiction Research’s Lifetime Achievement Award (2014), the McGovern Award for Lifetime Achievement (2015) for the most important contributions to the understanding and treatment of addiction, the National Leadership Award (NAATP) from addiction treatment providers for helping understand that addiction is a disease of the brain, the DARE Lifetime Achievement Award for volunteer and prevention efforts, the Silver Anvil from the PR Society of America for anti-drug prevention ads, the PRIDE and DARE awards for his career in research and prevention (2015), and the PATH Foundation’s Lifetime Achievement Award (2016) as one of the “fathers” of addiction medicine and MAT presented to him by President Obama’s White House Drug Czar Michael Botticelli. He was awarded Distinguished Alumni Awards at Yale University, the University of Florida, and Washington University and the Wall of Fame at the University of Florida College of Medicine. Gold was appointed by the University President to two terms as the University’s overall Distinguished Professor, allowing him to mentor students and faculty from every college and institute. The University of Florida College of Medicine’s White Coat Ceremony for new medical students is named in his honor.
Since his retirement as a full-time academic in 2014, Dr. Gold has continued his teaching, mentoring, research, and writing as an Adjunct Professor in the Department of Psychiatry at Washington University and an active member of the Clinical Council at the Washington University School of Medicine’s Public Health Institute. He regularly lectures at medical schools and grand rounds around the country and at international and national scientific meetings on his career and on bench-to-bedside science in eating disorders, psychiatry, obesity, and addictions. He continues on the Faculty at the University of Florida College of Medicine, Department of Psychiatry as an Emeritus Distinguished Professor. He has traveled extensively to help many states develop prevention, education, and treatment approaches to the opioid crisis.
Contributing faculty, Mark S. Gold, MD, DFASAM, DLFAPA, has disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
John M. Leonard, MD
Mary Franks, MSN, APRN, FNP-C
The division planners have disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
Sarah Campbell
The Director of Development and Academic Affairs has disclosed no relevant financial relationship with any product manufacturer or service provider mentioned.
The purpose of NetCE is to provide challenging curricula to assist healthcare professionals to raise their levels of expertise while fulfilling their continuing education requirements, thereby improving the quality of healthcare.
Our contributing faculty members have taken care to ensure that the information and recommendations are accurate and compatible with the standards generally accepted at the time of publication. The publisher disclaims any liability, loss or damage incurred as a consequence, directly or indirectly, of the use and application of any of the contents. Participants are cautioned about the potential risk of using limited knowledge when integrating new techniques into practice.
It is the policy of NetCE not to accept commercial support. Furthermore, commercial interests are prohibited from distributing or providing access to this activity to learners.
Supported browsers for Windows include Microsoft Internet Explorer 9.0 and up, Mozilla Firefox 3.0 and up, Opera 9.0 and up, and Google Chrome. Supported browsers for Macintosh include Safari, Mozilla Firefox 3.0 and up, Opera 9.0 and up, and Google Chrome. Other operating systems and browsers that include complete implementations of ECMAScript edition 3 and CSS 2.0 may work, but are not supported. Supported browsers must utilize the TLS encryption protocol v1.1 or v1.2 in order to connect to pages that require a secured HTTPS connection. TLS v1.0 is not supported.
The role of implicit biases on healthcare outcomes has become a concern, as there is some evidence that implicit biases contribute to health disparities, professionals' attitudes toward and interactions with patients, quality of care, diagnoses, and treatment decisions. This may produce differences in help-seeking, diagnoses, and ultimately treatments and interventions. Implicit biases may also unwittingly produce professional behaviors, attitudes, and interactions that reduce patients' trust and comfort with their provider, leading to earlier termination of visits and/or reduced adherence and follow-up. Disadvantaged groups are marginalized in the healthcare system and vulnerable on multiple levels; health professionals' implicit biases can further exacerbate these existing disadvantages.
Interventions or strategies designed to reduce implicit bias may be categorized as change-based or control-based. Change-based interventions focus on reducing or changing cognitive associations underlying implicit biases. These interventions might include challenging stereotypes. Conversely, control-based interventions involve reducing the effects of the implicit bias on the individual's behaviors. These strategies include increasing awareness of biased thoughts and responses. The two types of interventions are not mutually exclusive and may be used synergistically.
#96564: Alcohol and Alcohol Use Disorder
No substance, legal or illegal, has a more paradoxical mythology than alcohol. It is undeniably one of the most widely and safely used intoxicants in the world; however, it is also potent and dangerous, both from a psychological and a physiologic viewpoint. Alcohol is currently responsible for more deaths and personal destruction than any other known substance of abuse, with the exception of tobacco. All of this is known with scientific certainty. Alcohol is legal, easily obtained, and supported by a multi-billion-dollar worldwide industry. Alcohol consumption at reasonable doses reduces social inhibitions and produces pleasure and a sense of well-being. It also can have some rather impressive positive medical effects, such as a reduced risk of cardiovascular disease[1,2].
Alcohol is defined as a substance of abuse by self-administration in lab animals and man. All drugs of abuse affect the brain's reward pathways. The effects of alcohol appear to be related to complex multiple interactions with the dopamine, gamma-aminobutyric acid (GABA), serotonin, opioid, and N-methyl-D aspartate (NMDA) neurotransmitter systems [3,4]. Studies suggest that the reinforcing effect of alcohol is partially mediated through nicotinic receptors in the ventral tegmental area, which when combined with nicotine may be a factor in the high incidence of smoking among those with alcohol use disorder [5,6,7]. Alcohol, food, and other drugs of abuse have similar effects on dopamine receptors. The development of addiction, including to alcohol, is affected by genetic predisposition and influenced by alterations in the rewarding chemicals released per dose.
Substances of abuse are often put into categories based on their effects. Alcohol has effects similar to other depressants. Characteristics include:
Decreased cognitive function while intoxicated
Decreased inhibition and increased impulsivity
Risk of overdose
Development of depressive symptoms in heavy users
Withdrawal symptoms similar to other depressants
Symptoms of anxiety during withdrawal
Substance-induced psychoses in some heavy users
The established criteria for the diagnosis of alcohol abuse and dependence will be discussed in detail later in this course.
From the earliest days of colonial settlement to the present, Americans have been drinking alcohol. The early American experience with alcohol provides a glimpse of patterns of use, as well as controversies involving alcohol. The argument could be made that alcohol, in one form or another, was used more in early revolutionary America than it is today. Drinking had almost religious support, with alcohol portrayed as a gift from God; a gift that could be abused by excessive drinking or drunkenness.
The story of alcohol in America begins with the Mayflower. The Mayflower dropped anchor in Plymouth, Massachusetts, in February 1621. The passengers were out of beer, and the crew was in no mood to share. Running out of beer or spirits was no laughing matter. So seriously did the crew take this lack of spirits that they quickly dropped the passengers off the Mayflower into very harsh conditions at Plymouth. This preference for beer was at least partly because it was a reliable source of nonpathogenic hydration, as bacteria and parasites are killed during the boiling stage of brewing.
In 1741, Benjamin Franklin, when listing the thirteen cardinal virtues, started with "Temperance: Drink not to elevation." Still, he noted that beer, applejack, and other alcohol-containing beverages of the day were safer to drink than the water in Boston or Philadelphia. Like Londoners, the colonists drank beer with breakfast after a sherry eye-opener, and drank beer at lunch and brandy if it was cold. They would have wine with dinner and punch or other liqueurs thereafter. Dr. Benjamin Rush, a signer of the Declaration of Independence who is also known as the father of American psychiatry, became alarmed by what he viewed as rampant health problems caused by alcohol and called for temperance. He described addiction and identified alcohol as an addictive substance. He argued that addiction was like a disease, and that the alcoholic victim was completely unable to control his consumption. Dr. Rush, a citizen of the Enlightenment Age, accurately described and anticipated the fifth revised edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-5-TR) description of alcohol use disorder. However, he also suggested that alcoholism could be treated by whippings, bleeding, shame, emetics, oaths, and plunging the patient in cold water.
American attitudes about alcohol have flip-flopped from a free marketplace in the 18th and 19th centuries, to Prohibition in the 1920s, to the repeal of Prohibition in the 1930s, to lowering of the legal drinking age in most states during the late 1960s and early 1970s, and a return to the 21-years-of-age limit with the National Minimum Drinking Age Act of 1984. Alcohol consumption tends to be high during war years and was lowest during the Great Depression [8]. It was very high in the early 1980s, perhaps due to the lowered drinking age and poor economy. Like other consumer goods, alcohol consumption is generally inversely affected by changes in taxes and prices [9]. But interesting trends have been noted in the United States. While inflation-adjusted prices of alcohol declined between the late 1970s through the late 1990s, per capita alcohol consumption has also been declining since the mid-1980s [8,10]. One study revealed that changing demographics, such as a shift to an older population that consumes less alcohol, could have more of an impact on consumption levels than falling prices. Other sources cite increased health awareness, national drunk-driving campaigns, and a less tolerant public attitude toward heavy drinking and youth intoxication [8].
This roller coaster of historical attitudes toward alcohol use results from conflicting sociologic and psychological factors. For centuries, alcohol has been part of our social fabric and part of holidays and traditions. Simultaneously, our society has either shunned or punished those who succumbed to alcohol abuse, treating dependence as a legal issue or a moral failing rather than as a mental health problem. On one hand, alcohol is readily and cheaply accessible, safe for most people, moderately beneficial to health, and an important sector of our economy. However, we also understand that some individuals are at a high risk of losing control over alcohol.
As many as 90% of adults in the United States have had some experience with alcohol [11]. People drink alcohol for a variety of reasons:
The pleasurable feeling that often accompanies drinking, including reduced tension and/or anxiety
Enjoyment of the taste
Social inclusion
Self-medication
Peer pressure
Behavioral and physical addiction
Nearly one-half (47.4%) of all Americans older than 12 years of age reported being current consumers of alcohol in the 2022 National Survey on Drug Use and Health [12]. This translates to an estimated 137.5 million people, slightly less than the 2019 estimate of 139.7 million people [12]. An estimated 21.7% of Americans participated in binge drinking at least once in the 30 days prior to the survey. This represents approximately 61.1 million people. Heavy drinking was reported by 5.7% of the population 18 years of age and older (16.0 million people) [12]. The 2022 estimates for binge and heavy drinking are lower than the 2019 estimates [12].
There is some evidence that drinking and alcohol-related injuries increased during the COVID-19 pandemic [13]. National mortality data revealed a 25% increase in alcohol-related deaths from 2019 to 2020, outpacing the all-cause increase in mortality over the same period (16.6%).
Binge drinking among various races is 10.9% for Asian Americans, 21.3% for Black Americans, 26.5% for persons reporting two or more races, 22.2% for White Americans, and 23.1% for Hispanic Americans [12].
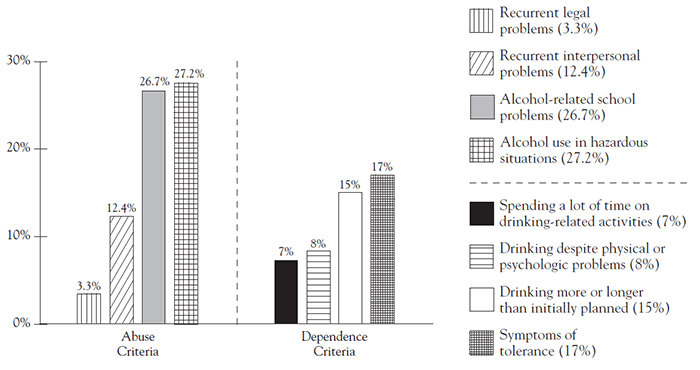
Use of alcohol is higher for college graduates compared to those with only a high school diploma (66.2% and 33.8%, respectively) [12]. However, binge and heavy use is slightly higher for young adults 18 years of age and older who have not completed college [12]. The pattern of higher rates of current alcohol use, binge alcohol use, and heavy alcohol use among full-time college students, compared with rates for others 18 to 22 years of age, has remained consistent since 2002 [14]. In a 2002 study of alcohol use on college campuses, researchers at Harvard University reported that of the more than 14,000 students surveyed, 31% met the criteria for alcohol abuse and an additional 6% met the criteria for diagnosis of alcohol dependence [15]. In the study, alcohol abuse was defined as a positive response to any one of the four abuse criteria and the absence of dependence. Alcohol dependence was defined as a positive response to any three or more of seven dependence criteria. Percentages of students meeting specific alcohol abuse and dependence criteria are presented in Figure 1[15]. Male students are at greater risk than female students. Almost 10% of male students and 5% of female college students younger than 24 years of age met the criteria for a 12-month diagnosis of alcohol dependence [15].
About 40% of people who drink have experienced an alcohol-related problem [11]. Between 3% and 8% of women and 10% to 15% of men will develop alcohol use disorder at some point in their lives. While alcohol use disorders can develop at any age, repeated intoxication at an early age increases the risk of developing an alcohol use disorder [11]. Usually, dependence develops in the mid-twenties through age forty.
The National Institute on Drug Abuse (NIDA) and the National Institute on Alcohol Abuse and Alcoholism (NIAAA) estimated that the annual economic cost of alcohol and drug abuse was $365.4 billion in 1998 [9]. This estimate represents roughly $1,350 each year for every man, woman, and child living in the United States. Alcohol use disorders generated about half of the estimated costs ($184.6 billion). This figure rose to $249 billion in 2010, representing approximately $807 for every man, woman, and child living in the United States [16].
Nearly three-fourths (72%) of the costs of alcohol abuse are related to lost workplace productivity ($179 billion); 11% are related to healthcare expenses for treating problems caused by excessive drinking ($28 billion); 10% are law enforcement and other criminal justice expenses ($25 billion); and 5% are losses from motor vehicle crashes related to excessive alcohol use ($13 billion) [16]. Binge drinking is responsible for the majority of the cost at $191 billion [16]. Alcohol use disorder generally reduces the lifespan by 15 years [11]. Approximately $99.6 billion of the total costs of alcohol abuse is paid by federal, state, and local governments [16]. When both direct and indirect costs are included, the estimated annual cost of alcohol-related problems alone may be much greater [16].
Tolerance: Either (1) a need for markedly increased amounts of the substance to achieve intoxication or desired effect; or (2) a markedly diminished effect with continued use of the same amount of the substance [17].
A Standard Drink: 1.5 ounces of 80-proof distilled spirits, 5 ounces of table wine, or 12 ounces of standard beer [18,19].
Alcohol Intoxication: Clinically significant problematic behavioral or psychological changes (e.g., inappropriate sexual or aggressive behavior, mood lability, impaired judgment) that developed during, or shortly after, alcohol ingestion [17]. Changes include slurred speech, loss of coordination, unsteady walking or running, impairment of attention or memory, nystagmus, stupor, or coma.
Alcohol Withdrawal: The presence of certain symptoms after stopping or reducing heavy and prolonged alcohol use [17]. The symptoms of alcohol withdrawal may develop within a few hours to a few days after stopping or reducing use and symptoms cause significant physical and emotional distress in social, occupational, or other important areas of functioning. Symptoms include increased hand tremor, sweating, increased pulse rate, nausea, vomiting, insomnia, temporary hallucinations or illusions, anxiety, psychomotor agitation, and generalized tonic-clonic seizures. Fewer than 5% of persons who develop alcohol withdrawal experience severe symptoms such as seizures and death [20].
Blood Alcohol Concentration (BAC): The percentage of alcohol present in the bloodstream. The BAC is usually what is measured by police officers to determine legal intoxication. It can be measured directly from a blood sample or a breath sample collected by a "Breathalyzer." The national legal limit for intoxication is a BAC of 0.08.
Moderate Drinking: No more than one drink per day for women and no more than two drinks per day for men [19].
Current Use: At least one drink in the past 30 days [16].
Binge Drinking: Consuming five or more drinks on the same occasion in the past 30 days [16,19].
Heavy Drinking: Five or more drinks on the same occasion on each of 5 or more days in the past 30 days [16].
Fetal Alcohol Syndrome (FAS): A severe fetal alcohol spectrum disorder (FASD), FAS is a lifelong syndrome in children with confirmed prenatal exposure to alcohol. Signs include growth deficiencies, facial abnormalities, and neurocognitive deficits that may lead to problems with vision, hearing, attention, learning, memory, or any combination thereof [21]. There is no safe recommended level of alcohol use in pregnancy.
Alcohol is consumed sensibly by the vast majority, but it can also be a cause of considerable damage and death when used excessively. Alcohol is part of many cultures, and most individuals learn from their bad experiences to moderate their drinking. Consequently, the majority of people do not have accidents or develop alcohol use disorder. Additionally, data suggests that moderate consumption of alcohol does have some health benefits.
The French consume large amounts of wine and high-cholesterol foods, yet they have a low incidence of heart disease. The Japanese drink large amounts of sake, but eat basically low-cholesterol foods and have a low incidence of heart disease. Other cultures traditionally drink whiskey and beer. Should we be drinking more, more regularly, or less on both counts?
Data for health benefits associated with low-to-moderate drinking appear to be common in many medical journals [22]. Light-to-moderate alcohol intake from beer, wine, or spirits is associated with a reduction in all-cause mortality, possibly due to its ability to decrease cardiovascular diseases, especially coronary heart disease (CHD). The relationship between alcohol intake and reduced risk of coronary disease is generally accepted as a U-shaped curve of low-dose protective effect and higher doses producing a loss of protective effects and increased all-cause deaths [23,24,25,26,27,28,29,30]. The World Health Organization (WHO) reported that there is convincing evidence that low-to-moderate alcohol intake decreases risk for heart disease [31].
Many researchers have replicated the finding that moderate alcohol consumption is associated with a reduced risk of coronary artery disease, peripheral artery disease, sudden death, and stroke and suggest that this effect is to a large extent mediated by increases in high-density lipoproteins (HDLs) [1]. A 2011 meta-analysis inclusive of 84 out of 4,235 studies on the benefits of alcohol concluded that the lowest risk of CHD mortality was conferred by one to two drinks per day and that the lowest stroke mortality risk was conferred by consuming one or fewer drinks per day [2]. Research suggests that the protective effect may be a result of an interaction between diet and genetics, specifically related to a genetic variation in alcohol dehydrogenase (ADH) [32]. Moderate drinkers who are homozygous for the slow-oxidizing ADH3 allele have higher HDL levels and a substantially decreased risk of myocardial infarction [32]. An acute protective effect of alcohol consumption was also found for regular drinkers who consumed one or two drinks in the 24 hours preceding the onset of cardiac symptoms. Risk of a major coronary event is lowest among men who report daily drinking and among women who report one or two drinks daily. Alcohol does have effects on several markers for coronary risk factors, such as blood pressure, HDL cholesterol, low-density-lipoprotein (LDL) cholesterol, fibrinogen, clotting factors, and insulin sensitivity.
Prescribing alcohol to patients is not recommended, but research should continue in an attempt to identify the beneficial effects of alcohol alone. The psychiatric and other medical costs associated with drinking should be considered. Epidemiologists and other researchers are weighing the benefits of moderate alcohol consumption against the risks of addiction and accidents.
Alcohol clearly causes detrimental effects on a number of critical organs and systems in the human body when taken in large doses over time. Excessive alcohol consumption increases cardiovascular risk factors and mortality. Alcohol abuse is often considered the second most common cause of preventable death in the United States [33]. However, light-to-moderate drinking may protect against ischemic stroke and abstaining from alcohol may increase the risk of stroke [2]. A prospective study of moderate alcohol consumption and risk of peripheral arterial disease in U.S. male physicians found that any alcohol consumption decreases the risk of peripheral artery disease [1]. No evidence exists for a reduction in cardiovascular mortality in anyone younger than 40 years of age. Because almost no one dies of coronary artery disease before age 40, the studies to see if drinking in individuals younger than 40 years of age is particularly protective in later life have yet to be done.
Cardiovascular protection occurs primarily through blood lipids such as HDL, especially HDL subfraction 2 [1]. Moderate alcohol consumption inhibits platelets, especially after a fatty meal, suggesting an aspirin-like effect for moderate alcohol consumption [34]. Alcohol's effects on clotting appear to be related to the findings that drinking reduces acute heart attack risk. Certain alcoholic beverages, namely red wine, may also have an additional positive antioxidant effect as it contains flavonoids, which possibly slow oxidation of unsaturated fatty acids [35]. Additionally, low amounts of drinking can also enhance insulin sensitivity, reduce fasting insulin, and may also reduce stress.
Risk-to-benefit analysis should take into account a person's age, sex, family history, likelihood of an adverse effect on blood pressure, cancer risk, medication interaction, accidents, and dependency. Light-to-moderate alcohol consumption reduces overall risk of ischemic stroke; however, greater alcohol consumption has no additional benefit and can be harmful [2].
It has been questioned whether the cardiac protective effects can be easily generalized to women, in whom the risk of breast cancer complicates alcohol risks. For example, the consumption of seven or more drinks per week is associated with a twofold increase in postmenopausal hormone-sensitive breast cancers; however, several studies have shown that moderate alcohol consumption reduces the mortality of breast cancer [36,37,38]. It should also not be forgotten that alcohol increases the risk of certain other cancers (e.g., liver, mouth, esophageal, laryngeal, pharyngeal) that affect both men and women. Multiple case-control studies and meta-analyses have reported on the relationship between alcohol and cardiovascular disease and mortality. Many of these studies reported that low-to-moderate alcohol consumption decreases the number of adverse cardiovascular events and deaths when compared with abstinence. Beginning in the early 2000s, the cardioprotective effects of low doses of alcohol were refuted by the results of large epidemiological studies. Since that time, alcohol use is consistently associated with cardiac arrhythmias, dilated cardiomyopathy, arterial hypertension, atherosclerotic vascular disease, and type 2 diabetes [39,40].
Moderate drinking is heart-healthy for diabetics in the same way it is for other people, easing concerns that alcohol may disrupt diabetics' blood-sugar balance. In a 12-year study, diabetics who had one or two drinks daily were up to 80% less likely to die of heart disease than diabetics who did not drink [41]. However, alcohol consumption is a marker for poorer adherence to diabetes self-care behaviors [42,43].
Although alcohol appears to have some moderate health benefits, physicians need not alter the drinking habits of those who consume low-to-moderate amounts of alcohol. It is problematic to advise a patient who is abstinent or who drinks infrequently to begin or increase alcohol consumption. In addition, social and religious factors may already dictate the patient's drinking habits.
Vulnerability to alcohol use disorders, depression, and alcohol-related pathologies varies greatly among individuals and cannot always be predicted before a patient begins or escalates drinking. Some individuals may be genetically predisposed to acquiring problems with alcohol use disorder. Similarly, excessive consumption often escapes detection before the onset of related health consequences. The balance of risk to benefit appears to favor encouraging some patients in midlife who are very infrequent drinkers to increase slightly the frequency of drinking. Again, this is debatable and will vary with the individual patient. Consuming alcohol is not the only means to reduce the risk of cardiovascular disease. Exercising, not smoking, lowering fat intake and lipids, and other health-related lifestyle issues should also be addressed.
For those who already have heart disease, it is clear that heavy drinkers should reduce their consumption or abstain and that everyone should avoid heavy and binge drinking. Data does not support advising abstainers with a history of myocardial infarction or decreased left ventricular function to start drinking for their health [44]. In general, moderate drinkers with these conditions should be able to continue to drink alcohol in moderation [44].
Alcohol is not without risks. Alcohol abuse worsens the course of psychiatric disorders. In countries with high alcohol consumption, the suicide rate is also high. One should ask whether the promotion of moderate alcohol consumption, justified on the basis of a biomedical effect (e.g., a reduction in all-cause mortality), might change a patient's quality of life or cause them to take offense. However, existing public educational efforts that target reductions in hazardous and harmful drinking and at the same time encourage drinkers to consume alcohol at responsible levels are appropriate and ethical.
Research has shown that genetic factors play a strong role in whether a person develops alcohol use disorder, accounting for 40% to 60% of the risk [45,46]. In fact, family transmission of alcohol use disorder has been well established. Individuals who have relatives with alcohol use disorder are at three- to five-times greater risk of developing alcohol use disorder than the general population. The presence of alcohol use disorder in one or both biologic parents is more important than the presence of alcohol use disorder in one or both adoptive parents. The genetic risk of alcohol use disorder increases with the number of relatives with alcohol use disorder and the closeness of the genetic relationship [46]. However, most children of parents with alcohol use disorder do not become alcoholics themselves, and some children from families where alcohol is not a problem develop alcohol use disorders when they get older. Alcohol use disorder is seen in twins from alcoholic parents, even when they are raised in environments where there is little or no drinking. Identical twins adopted into households with an alcoholic stepfather do not show more alcohol use disorders than the general population. Children with close biologic relatives with alcohol use disorder, who are adopted into a never drinking, even religiously opposed family, can readily develop alcohol problems [47].
As mentioned previously, genetic factors are thought to account for 40% to 60% of the risk of developing alcohol use disorder [25,46]. Animal studies have shown that genetic factors may be responsible for enhanced brain reward produced by alcohol, decreased initial impairment, or even altered metabolism of alcohol [48,49,50,51,52,53,54].
Genetic factors appear to influence the level of response (LR) to alcohol, as measured by the intensity with which one reacts to a given quantity [55]. The level of response to alcohol varies from individual to individual depending on the tolerance. Low LR at an early age contributes to the risk of alcohol use disorder later in life [55,56].
Genetic differences in metabolic or other biologic processes may play a role in the development of alcohol use disorder in specific individuals. Studies using a self-rated scale have shown consistent results in sons of alcoholic fathers scoring themselves lower than sons of nonalcoholic fathers on feelings of drunkenness, dizziness, drug effect, and sleepiness following alcohol consumption [57]. This suggests that sons of alcoholic fathers have a less intense reaction to alcohol than sons of nonalcoholic fathers. Low reaction to alcohol suggests tolerance and impaired ability to recognize even modest levels of alcohol intoxication, indicators of tendency towards dependence [58,59]. High alcohol sensitivity in men is associated with substantially decreased risk of alcohol use disorder. Understanding reactions to alcohol could establish a better understanding of future risk of developing alcohol use disorder in these men.
Studies have found similar results of higher tolerance for alcohol among daughters of parents with alcohol use disorder. One study examined the drinking patterns of 38 daughters of alcoholics compared with 75 family-history-positive men from the same families and 68 men with no family history of alcohol use disorder [60]. Family-history-positive men and women both displayed low reaction to alcohol. This indicates that the degree of genetic influence on alcohol-related behavior is similar for both men and women with family history of alcohol use disorder. In a study of adolescent and young adult offspring from families where alcohol use disorders are prevalent, researchers found both neurophysiologic and neuroanatomical differences, such as reduced right amygdala volume, when comparing these offspring to controls [61]. Another study assessed the relationship between amygdala and orbitofrontal cortex volumes obtained in adolescence and substance use disorder outcomes in young adulthood among high-risk offspring and low-risk controls [62]. A total of 78 participants 8 to 19 years of age (40 high-risk, 38 low-risk) from a longitudinal family study underwent magnetic resonance imaging. Volumes were obtained with manual tracing. Outcomes were assessed at approximately one-year intervals. The ratio of orbitofrontal cortex volume to amygdala volume significantly predicted substance use disorder survival time across the sample. A reduction in survival time was seen in participants with smaller ratios; this was true for both high-risk and low-risk participants [62].
Native Americans and Alaskan Natives have a lower level of response and an increased risk of alcohol use disorder [46]. The alcohol metabolizing enzymes are another important genetic influence, especially for persons of Asian descent. About 50% of Japanese, Chinese, and Korean persons flush and have a more intense response to alcohol because they have a form of alcohol dehydrogenase (ADH) that causes high levels of acetaldehyde. Forms of ADH and aldehyde dehydrogenase (ALDH) (e.g., homozygous or heterozygous) contribute to a higher rate of alcohol metabolism, intensify the response to alcohol, and lower the risk of alcohol use disorder. High levels of impulsivity/sensations seeking/disinhibition are also genetically influenced and may impact alcohol use disorder risk [46].
An estimated 178,000 people (approximately 120,000 men and 59,000 women) die from alcohol-related causes annually. According to NIAAA, alcohol is a significant cause of death, disease, and disability, currently ranked as one of the leading preventable causes of death in the United States [63]. According to a 2022 SAMSHA survey reported by NSDUH, 29.5 million Americans 12 years of age and older (10.5% of this age group) have alcohol use disorder [63]. Almost 1 in 4 adults have had a heavy drinking event in the past year (defined as five or more drinks for men and four or more drinks for women). The NIH and the CDC report increasing alcohol problems, deaths, and alcohol use disorders. An analysis of death certificates from 2019 and 2020 showed that deaths involving alcohol rose from approximately 79,000 to more than 99,000, a 25.5% increase [64]. Increases in alcohol-related deaths are consistent with reports of increased alcohol sales, consumption, alcohol-involved emergency department visits, and hospitalizations. The most recent alcohol data provide more evidence of increasing heavy alcohol use and associated consequences during the COVID-19 pandemic [65]. A 2023 study demonstrated that only individuals with prepandemic history of alcohol use disorder reported greater increases in drunkenness frequency during the COVID-19 pandemic [66]. Increased alcohol use may also worsen medical and mental health problems. Among people who die by suicide, alcohol use disorder is the second most common mental disorder and involved in an estimated one in four deaths by suicide [64].
Researchers who study risk factors have developed models of how known risk factors may interact to create pathways in children that lead to alcohol use disorders.
One model focuses on children who have temperaments that make it difficult for them to regulate their emotions and control their impulses. Clearly, these children are difficult to parent, and if one or both of their parents have alcohol use disorder, it is likely that they will be poorly socialized and have trouble getting along in school [67,68]. Poor academic performance and rejection by more mainstream peers at school may make it more likely for these children to join peer groups where drinking and other risky behaviors are encouraged. Parents with alcohol use disorders will likely not monitor their children closely and will lose control over them at an early age. These children will begin drinking early, often before 15 years of age [69]. If such a child is genetically predisposed to alcohol use disorders, these environmental factors may further increase the tendency [70].
Another model of risk factors leading to alcohol use disorder focuses on drinking to regulate inner distress [71]. Some children have temperaments that make them highly reactive to stress and disruption. This type of child may be born into a family with history of alcohol use disorder, where the stressors may be intense, or a nonalcoholic family, with everyday types of low-level stressors. Regardless of the child's family environment, he or she maintains higher levels of inner distress (anxious and depressed feelings) than other children. When they take their first drink, the inner distress dissipates for a while. This leads to more drinking and may lead to alcohol use disorder. However, for some individuals, at certain doses, alcohol may induce rather than reduce the stress response. Research demonstrates that alcohol actually induces the stress response by stimulating hormone release by the hypothalamus, pituitary, and adrenal glands [72]. Research also demonstrates a bidirectional relationship between alcohol and stress [73]. More research is required before the role of stress as a risk factor in alcohol use disorders is understood.
A third risk factor model focuses on sensitivity to the effects of alcohol, both to its sedative properties and its stimulating qualities [74]. The stimulant-like (increased heart rate and blood pressure) and sedative properties (impaired vigilance and psychomotor performance) depend on the quantity of alcohol consumed, the time elapsed since consumption, and individual differences in response [75,76]. Researchers believe that this subjective response to alcohol may be an important endophenotype in understanding genetic influences on drinking behavior and alcohol use disorders. While subjective response predicts alcohol use and problems, the exact pattern of association remains unclear [77,78,79]. Two prominent models of subjective response have been discussed in the literature. The low level of response model suggests that high-risk individuals experience decreased sensitivity to the full range of the effects of alcohol. The differentiator model suggests that high risk for alcohol problems is associated with increased sensitivity to alcohol's positive effects but decreased sensitivity to its negative effects [77,78]. A literature review of studies that employed challenge paradigms to assess a range of the effects of alcohol (i.e., impairment, stimulation, sedation) found some support for both models [77]. Results of a quantitative review and meta-analysis suggest that the two models may describe two distinct sets of phenotypic risk with different etiologies and predictions for development of alcohol use disorder [78]. A total of 32 independent samples were combined to produce estimates of the effects of risk-group status (i.e., positive family history of alcohol use disorder or heavy alcohol consumption). Groups with positive family history for alcohol experienced reduced overall subjective response relative to groups with negative family history, as predicted by the low level of response model. In contrast, consistent with the differentiator model, heavy drinkers of both genders responded less on measures of sedation than did lighter drinkers, but more on measures of stimulation [78].
The effects of alcohol on the electroencephalogram (EEG) of subjects at risk for developing alcoholism are well known [80,81,82]. Researchers found that low EEG response to small amounts of alcohol may be associated with future development of alcohol use disorder. Additionally, differences in EEG response to alcohol may have ethnic variations [82]. Other studies have shown that heavy drinkers had less sedation and cortisol response after alcohol consumption than light drinkers. In addition, heavy drinkers were more sensitive to the positive stimulant-like properties as blood alcohol levels increased [74,83].
With these three models in mind, a review of some of the research findings on genetic and psychosocial risk factors may provide a better understanding of the factors leading to alcohol use disorders [11,84]:
Temperament: Moodiness, negativity, and provocative behavior may lead to a child being criticized by teachers and parents. These strained adult-child interactions may increase the chances that a child will drink.
Hyperactivity: Hyperactivity in childhood is a risk factor for the development of adult alcohol use disorders. Children with attention deficit hyperactivity disorder (ADHD) and conduct disorders have increased risk of developing an alcohol use disorder. Childhood aggression also may predict adult alcohol abuse.
Parents: The most compelling and largest body of research shows parents' use and attitudes toward use to be the most important factor in an adolescent's decision to drink.
Gender: Among adults, heavy alcohol use is almost three times more common among men than women and also more common among boys in middle or high school than among girls. Men with ADHD and/or conduct disorders are more likely to use alcohol than men without these disorders, while women who experience more depression, anxiety, and social avoidance as children are more likely to begin using alcohol as teens than women who do not experience these negative states.
Psychology: Bipolar disorder, schizophrenia, antisocial personality disorder, and panic disorder all also increase the risk of a future alcohol use disorder.
Childhood abuse is a significant risk factor for later alcohol and substance abuse [85]. Women who were physically abused are 1.5 to 2 times more likely to abuse alcohol than non-abused adults. Children from crowded, noisy, and disorderly homes without rules or religion are more likely to abuse alcohol as teens. Children who are quick to anger, who perceive themselves to be highly stressed, who are resentful of parents' absences, or who have repeated conflicts at home are more likely to abuse alcohol as teens.
An exciting area of research is focused on protective factors and poses the question, "What protects children from taking one of the risk pathways to alcohol use disorder?"
In 1997, some good news came from the National Longitudinal Study on Adolescent Health, a survey in which nearly 12,000 students in grades 7 through 12 were given lengthy interviews timed one year apart. The researchers were trying to determine what kept children, over the course of that year, from taking health risks in four areas: substance abuse (cigarettes, alcohol, and marijuana), sexuality, violence, and emotional health [86]. The researchers found two factors that protected these children in all four areas. They named the factors: parent-family connectedness and school connectedness.
Children identified as having parent-family connectedness said they felt close to their mother or father, felt that their mother or father cared about them, felt satisfied with their relationship with their mother or father, and felt loved by family members [86]. School connectedness was experienced as a feeling of being part of one's school and a belief that students were treated fairly by the teachers.
There is broad evidence of the protective role of parenting on adolescent health risks. Another well-established protective factor is adolescents' perceived disapproval of alcohol use by their parents [87,88,89,90,91]. In 2022, the National Survey on Drug Use and Health asked children 12 to 17 years of age about their perceptions of the level of parental disapproval of substance use initiation, including alcohol [12]. Most adolescents (88.9%) reported that their parents would strongly disapprove of them having one or two alcoholic drinks nearly every day. This percentage was similar to percentages in most years since 2002, with rates ranging from 88.5% to 91.2% [12]. The number of past-year initiates 12 years of age or older for alcohol also remained stable between 2002 and 2022 [12].
Alcohol use disorder, also referred to as alcohol abuse and/or alcohol dependence, is defined in the DSM-5-TR as a problematic pattern of use with two or more of the following criteria over a one-year period [17]:
Alcohol often taken in larger amounts or over a longer period than was intended
A persistent desire or unsuccessful efforts to cut down or control alcohol use
A great deal of time spent in activities necessary to obtain alcohol, use alcohol, or recover from its effects
Craving, or a strong desire or urge to use alcohol
Recurrent alcohol use resulting in a failure to fulfill major role obligations at work, school, or home
Continued alcohol use despite having persistent or recurrent social or inter-personal problems caused or exacerbated by the effects of alcohol
Important social, occupational, or recreational activities given up or reduced because of alcohol use
Recurrent alcohol use in situations in which it is physically hazardous
Alcohol use is continued despite knowledge of having a persistent or recurrent physical or psychological problem that is likely to have been caused or exacerbated by alcohol
Tolerance
Withdrawal
Alcohol use disorder is extremely amenable to brief intervention. Brief intervention usually includes giving patients information about problems associated with excessive drinking and advising them to cut down on their drinking or abstain. Without intervention, 10% will likely progress to dependence and 50% to 60% will continue to experience problems over the next five years [92,93].
Alcohol use disorder is a primary and chronic disease that is progressive and often fatal; it is not a symptom of another physical or mental condition. It is a disease in itself, like cancer or heart disease, with a very recognizable set of symptoms that are shared by others with the same disorder. About 29.5 million people in the United States met DSM-5-TR criteria for alcohol use disorder in 2022, with an additional 5.3 million abusing or dependent on both alcohol and illicit drugs [12,94].
Like cancer and many other chronic diseases, alcohol use disorder progresses over time. People with alcohol use disorder experience physical, emotional, and other changes in their lives and relationships. These changes may worsen if drinking continues and if treatment specifically targeted to alcohol use disorder is not initiated. Left untreated, alcohol use disorders may lead to premature death through overdose or through damage to the brain, liver, heart, and many other organs. Excessive alcohol consumption is highly associated with suicide, motor vehicle accidents, violence, and other traumatic events [94]. People with untreated alcohol use disorders often lose their jobs, their families, their relationships, and other freedoms that were once important to them.
As noted, alcohol problems can often be prevented by early identification and brief intervention. A weak link in the early identification of problems is the lack of skill and competencies necessary to perform such an assessment and the experience to confidently move to more specific questions and suggestions for change.
Alcohol dependence is included in the DSM-5-TR umbrella definition of alcohol use disorder [17]. The symptoms of withdrawal and tolerance have been the hallmarks of more severe disease, though alone they are neither necessary for nor sufficient to make the diagnosis.
Healthcare professionals should understand the criteria and warning signs of alcohol use disorder. This enables confrontation and intervention earlier in the course of the illness rather than relying on toxic liver markers. Verifying the facts that show a person is at risk for alcohol use disorder and confronting the impaired individual with those facts is the definition of an office or brief intervention. Brief intervention is most effective before dependence is reached. Once diagnosable, the patient needs more comprehensive intervention.
Individuals with alcohol use disorder often experience a severe, potentially fatal withdrawal syndrome when they either abruptly discontinue or sharply reduce their alcohol consumption. The symptoms may include sweating, rapid heartbeat, hypertension, tremors, anorexia, insomnia, agitation, anxiety, nausea, and vomiting. Tremors of the hands are usually the earliest symptom of alcohol withdrawal. Hallucinosis, seizures, and delirium tremens (DTs) are the most severe form of alcohol withdrawal. Hallucinosis, when it occurs, occurs one to two days after decreasing or abstaining from alcohol. While the effects of DTs can be life threatening, all other symptoms, with or without treatment, usually resolve several hours or days after appearance. Alcohol withdrawal in tolerant individuals can occur before the BAC has dropped below the established legal limit for intoxication. Some persons with alcohol use disorder have symptoms of irritability, emotional lability, insomnia, and anxiety that persist for weeks to months after alcohol withdrawal. The symptoms may be due to the residual effects of alcohol toxicity on the central nervous system and can be post-acute withdrawal symptoms; members of Alcoholics Anonymous (AA) refer to this as being a "dry drunk." AA considers alcoholics who are only abstaining from alcohol but who are not working a recovery program and remaining in essentially the same emotional state as they were when they were drinking to be "dry drunks."
Long-term heavy drinking and genetic predisposition can result in the development of tolerance, which is the body's adaptation to the presence of alcohol. As tolerance develops, the drinker requires increasing amounts of alcohol to feel the same effect. For this reason, the usual reported effects for various BAC levels do not apply to individuals with tolerance. In our society, people are often admired for their ability to "hold their drinks." But the fact is, tolerance may be an early warning sign that a physical dependence on alcohol is developing. During the late stages of alcohol use disorder, reverse-tolerance occurs, meaning the individual becomes intoxicated more quickly and with less alcohol.
Impaired control over drinking means that a person is consistently unable to limit the number of occasions when alcohol is used or the amount of alcohol ingested on those occasions. Often, because of the damage alcohol causes in their lives, people with alcohol use disorder will express a strong and persistent desire to cut down or stop drinking. Often they may be able to do so, sometimes for a matter of weeks, a month, or even longer. One does not need to be a daily drinker to meet criteria for alcohol use disorder, as even those who go weeks or months without a drink may binge and meet diagnostic criteria. However, because alcohol use disorder is a chronic progressive disease, once patients with alcohol use disorder resume drinking, even after years of sobriety, they typically return to the previous quantities of consumption, with worsening adverse consequences.
Individuals with alcohol use disorder may have a preoccupation with alcohol, defined as a noticeable shift in priorities, with a focus on obtaining and consuming an adequate supply of alcohol. Drinking alcohol becomes a central focus in their lives. Over time, the energies of individuals with alcohol use disorder are diverted from people, places, and things that were once important to them.
Another highly noticeable feature of the preoccupation with alcohol may be the large amount of time that drinking consumes. Thinking about alcohol, obtaining alcohol, drinking alcohol, and recovering from the effects of alcohol take more and more of the individual's time. Hobbies and other activities once enjoyed are abandoned one by one, and the only pleasure seems to come from drinking.
Continued drinking despite adverse consequences is characterized by the inability of individuals with alcohol use disorder to stop drinking even when they recognize that their family, interpersonal, spiritual, occupational, legal, and financial problems are the result of their drinking. Furthermore, alcohol may be causing serious health and psychological problems (e.g., anemia, gastritis, liver disease, neurologic disorders, depression) and still the dependent individual cannot stop.
Denial is a common characteristic distortion in thinking that becomes profound in people with alcohol use disorder. For decades, those who have treated individuals with alcohol use disorder, and recovering alcoholics themselves, have puzzled over why these persons continue to drink when the link between alcohol and the losses they suffer is so clear. Denial is an integral part of the disease of alcohol use disorder and a major obstacle to recovery. Although the term denial is not specifically used in the wording of the diagnostic criteria, it underlies the primary criteria described as, "drinking despite adverse consequences."
Because alcohol use disorder is a chronic disease, another symptom that is increasingly being recognized and treated is relapse. Although alcohol use disorder is a treatable, chronic disease, as yet, no cure has been found. This means that even if individuals with alcohol use disorder have been sober for a long time and have regained their health and reclaimed other important aspects of their lives, they may experience a relapse that will require further treatment in order to return to remission.
While a strong attachment to alcohol is the hallmark of early dependency, if the patient refuses to acknowledge a problem and no one from home or work helps to confirm the diagnosis, healthcare professionals are often left with nothing more than clinical intuition, resulting in a missed diagnosis. However, late in the course of alcohol use disorder, physical clues typically become increasingly apparent and suggestive of alcohol abuse and/or dependence. Alcohol abuse and dependence are often referred to as the "Great Masquerader" because many of the signs and symptoms are also commonly found in other conditions [95].
Serum glutamic oxaloacetic transaminase (SGOT)
Lactic acid dehydrogenase (LDH)
Cholesterol
Gamma-glutamyltransferase (GGT)
Mean corpuscular volume (MCV)
Alkaline phosphatase
Triglycerides
Blood alcohol concentration (BAC)
Urinary ethyl glucuronide (EtG) and ethyl sulfate (EtS)
Whole blood phosphatidylethanol (PEth)
Serum transferrin
Uric acid
Anxiety
Insomnia
Memory impairment
Depression
Irritability
Panic
Suicide attempt(s)
Suicidal thinking
Loss of interest in previously favorite activities and people
Marital and financial problems
Positive family history
Cigarette smoking
Problems at home and work
Anger when someone asks about drinking
Legal difficulties
Higher than normal scores on screening questionnaires, such as the Michigan Alcohol Screening Test (MAST) and CAGE
Alcohol use disorders are often associated with physical disorders and related problems.
The liver is a particularly vulnerable organ to alcohol consumption, in large part because it is where alcohol is metabolized prior to elimination from the body. As few as six drinks a day for men have been found to be associated with liver damage. The most common manifestation among persons with alcohol use disorder is called "fatty liver." Among heavy drinkers, the incidence of fatty liver is almost universal. For some, a fatty liver may precede the onset of alcoholic cirrhosis. Fatty deposits have been associated with men who have six or more drinks a day and women who have only one or two drinks daily.
Alcoholic hepatitis is a condition that, when severe, is characterized by jaundice, fever, anorexia, and right upper-quadrant pain. Between 10% and 35% of heavy drinkers (those drinking five or six standard drinks a day or more) develop alcoholic hepatitis and 10% to 20% develop cirrhosis [96,97]. More than 60% of persons who develop both alcoholic hepatitis and cirrhosis will die within four years. Drinking 12 beers a day for 20 years has been associated with a 50% incidence of cirrhosis. It is not known which individuals will develop cirrhosis. Studies have shown that women develop liver disease faster and at lower levels of alcohol consumption than men [97,98]. Women also have a higher incidence of alcoholic hepatitis and higher mortality rate from cirrhosis [99].
Alcohol use disorder is also a strong predictive factor for the development of hepatocellular cancer [100]. The presence of other hepatic risk factors, including hepatitis C, fatty liver disease, smoking, and obesity, further increases this risk.
The leading indication for liver transplantation in the United States is chronic hepatitis C [101]. Cirrhosis due to alcoholic liver disease is the second most common cause for a person to require a liver transplantation [101]. Candidates for liver transplantation should be adequately screened for alcohol use disorders and receive appropriate treatment both perioperatively and as part of long-term follow-up. Patient survival after transplantation for both of these conditions is surprisingly good, with 72% of patients surviving after five years [101]. Short-term survival is similar; however, long-term survival for patients with hepatitis C now appears to be compromised by universal recurrence. When patients have both alcohol use disorder and chronic hepatitis C, they do worse than when both diseases occur independently. One study demonstrated that patients' short-term survival is the same for those who have alcohol use disorder, hepatitis C, or both diseases [102].
Chronic heavy drinking appears to activate the enzyme CYP2E1, which may be responsible for transforming the over-the-counter pain reliever acetaminophen into toxic metabolites that can cause liver damage [103]. Even when acetaminophen is taken in standard therapeutic doses, liver damage has been reported in this population [104,105]. A review of studies of liver damage resulting from acetaminophen-alcohol interaction reported that, in individuals with alcohol use disorder, these effects may occur with as little as 2.6 grams of acetaminophen (four to five "extra-strength" pills) taken over the course of the day by persons consuming varying amounts of alcohol [106]. The damage caused by alcohol-acetaminophen interaction is more likely to occur when acetaminophen is taken after, rather than before, the alcohol has been metabolized [107]. Moderate drinkers should also be made aware of this potential for interaction. There is now a warning label on the bottle that states, "If you consume three or more alcoholic drinks every day, ask your doctor whether you should take acetaminophen or other pain relievers/fever reducers." Further, in 2014 the U.S. Food and Drug Administration (FDA) issued a statement that combination prescription pain relievers containing more than 325 mg acetaminophen per dosage unit should no longer be prescribed due to reported severe liver injury with acetaminophen in patients who took more than the prescribed dose in a 24-hour period; took more than one acetaminophen-containing product at the same time; or drank alcohol while taking acetaminophen products [108].
Alcohol can have a detrimental effect on the heart, including a decrease in myocardial contractility, hypertension, atrial and ventricular arrhythmias, and secondary nonischemic dilated cardiomyopathy [109]. A common complication in alcohol use disorder is elevated pulse and blood pressure, often in the hypertension range. Younger people with alcohol use disorder and those without existing hypertension are less likely to have an elevation than those who are older and predisposed to some hypertension. When drinking stops, the blood pressure often returns to normal over a period of a few days. One study found that people who had six or more drinks a day were twice as likely to suffer from hypertension than moderate drinkers (two or fewer drinks per day) or nondrinkers. Increased serum GGT levels may be an indicator of an individual's susceptibility to the hypertensive effect of alcohol [110].
Aside from hypertension, chronic heavy drinking can adversely affect the heart primarily through direct toxicity to striated muscle, leading to a form of cardiomyopathy [109,111]. Alcoholic cardiomyopathy is probably more common than is currently thought because of underdiagnosis of alcohol use disorder in general. The reported prevalence of alcoholic cardiomyopathy has varied widely from 4% to 40% or more, depending on the characteristics of the study population and the threshold of alcohol consumption used to identify the disorder [112].
The association between heavy alcohol consumption and rhythm disturbances, particularly supraventricular tachyarrhythmias in apparently healthy people, is called "holiday heart syndrome" [111,113]. The syndrome was first described in persons with heavy alcohol consumption, who typically presented on weekends or after holidays, but it may also occur in patients who usually drink little or no alcohol [111,114]. The most common rhythm disorder is atrial fibrillation, which usually converts to normal sinus rhythm within 24 hours. The incidence of holiday heart syndrome depends on the drinking habits of the studied population, but it continues to be a prevalent occurrence in emergency department settings, with alcohol serving as a precipitating factor for atrial fibrillation in 35% to 62% of cases, particularly 12 to 36 hours post-binge drinking [115]. Additionally, the observed trend for increased binge alcohol use in younger adults may be associated with an increased incidence of atrial fibrillation in this population [116]. Holiday heart syndrome should be considered as a diagnosis particularly in patients without overt heart disease presenting with new onset atrial fibrillation. Though recurrences occur, the clinical course is benign and specific antiarrhythmic therapy is usually not warranted [111,113,114].
Abnormally high plasma levels of the amino acid homocysteine have been shown in studies to increase the risk for cardiac and other vascular diseases [117]. Even small increases in homocysteine appear to increase the risk of heart disease. Vitamins like folate, B12, and B6 are required for homocysteine disposal within cells. The lower the concentration of these and other vitamins, the greater the concentration of homocysteine. A number of nutritional problems have been reported in people with alcohol use disorder. Malnourished persons with alcohol use disorder and liver diseases have been found to have B6 and folate deficiencies. In addition, average homocysteine levels are twice as high in patients with chronic alcohol use disorder when compared to nondrinking controls. Thus, homocysteine may contribute to the cardiovascular complications experienced by many with chronic alcohol use disorder. Lowering homocysteine with B vitamin supplementation may reduce cardiovascular risk [118,119]. Further research is necessary to determine whether abstinence and recovery reverses the risk of cardiovascular disease, and whether folate and vitamins B12 and B6 should be considered as appropriate nutritional supplements for patients with alcohol use disorder [120].
Heavy drinking increases the risk of cancer of the upper gastrointestinal and respiratory tracts [121]. Almost 50% of cancers of the mouth, pharynx, and larynx and approximately 75% of esophageal cancers in the United States are associated with chronic, excessive alcohol consumption [122,123,124]. When alcohol consumption is combined with tobacco use, the risk of esophageal cancer increases markedly, as much as 130-fold in one study [125,126]. Alcohol increases production of estradiol, and increased levels of estradiol have been linked to an increased risk of breast cancer in women who drink [127].
Alcohol produces irritation and inflammation of the mucosal lining of the gastrointestinal tract and influences the motility in the esophagus, stomach, and small bowel [128]. Frank ulceration may occur with chronic excessive alcohol use. This well-known alcohol related "heartburn" is due to esophageal reflux with esophagitis that commonly occurs with irritation and inflammation of the gastroesophageal junction. Severe vomiting from alcohol gastritis may result in mucosal tears at the gastroesophageal junction, resulting in frank, usually transient pain in the upper gastrointestinal tract.
Short-term and long-term alcohol ingestion are associated with gastritis, erosive gastritis, gastric ulceration, atrophic gastritis, and gastric hemorrhage. Furthermore, duodenitis and duodenal ulcerations are a direct result of chronic excessive alcohol irritation and inflammation.
Patients who have undergone gastric bypass surgery for obesity have higher breath-alcohol levels after drinking the same amount as other people. Many bypass surgeries attach the jejunum directly to the stomach, allowing delivery of alcohol more rapidly to the jejunal site of primary absorption as well minimizing the effect of the stomach's alcohol dehydrogenase. Findings from a small study suggest that it takes much longer for their levels to return to zero [129].
Alcohol consumption is the leading cause of chronic pancreatitis, accounting for approximately 70% of cases in the United States; however, fewer than 10% of heavy alcohol drinkers develop the disease [130,131,132,133,134]. While there are many theories regarding the pathophysiology of chronic pancreatitis, the most prevalent for alcohol-induced chronic pancreatitis involves the effect of toxic metabolites on the pancreas. This theory suggests that inflammation and fibrotic changes in the pancreas are the direct result of premature activation of enzymes due to ethanol's effect on the Golgi complex [132,134,135]. Another theory suggests that pancreatic hypoxia results from decreased blood flow to the pancreas. Alcohol-induced acinar injury may reduce capillary flow and result in edema and capillary compression [132]. Individuals with alcohol use disorder may develop diabetes mellitus or hyperglycemia as a result of chronic pancreatitis, when the islet cells in the pancreas are eventually destroyed. Once alcohol-induced chronic pancreatitis has developed, ingestion of even small amounts can result in severe flare-up requiring hospitalization.
Although alcohol has a relatively high caloric value, 7.1 calories per gram (1 gram of fat contains 9 calories), alcohol consumption does not necessarily result in increased body weight. Moderate, regular doses of alcohol added to the diets of lean men and women do not seem to lead to weight gain. However, in some studies obese patients have gained weight when alcohol is added to their diets.
An analysis of data collected from the first National Health and Nutrition Examination Survey (NHANES I) found that although drinkers had significantly higher intakes of total calories than nondrinkers, drinkers were not more obese than nondrinkers. In fact, women drinkers had significantly lower body weight than nondrinkers. As alcohol intake among men increased, their body weight decreased. An analysis of data from the second National Health and Nutrition Examination Survey (NHANES II) and other large U.S. studies found similar results for women [136]. When chronic heavy drinkers substitute alcohol for food in their diets, they typically lose weight and weigh less than their nondrinking counterparts [137].
Many older studies, such as those discussed, have focused on total volume of alcohol based on intake over time (e.g., number of drinks per week), an average that reveals little about the actual drinking habits of individuals. This has led to a very inconsistent array of data on the relationship of drinking and body mass index (BMI). One study sought a better understanding of the relationship between BMI and regular/moderate versus infrequent binge drinking [138]. Researchers found that although individuals of similar height might consume the same weekly average of alcohol (e.g., 14 drinks per week), individuals who consume two drinks each day of the week typically have low BMIs and individuals who consume seven drinks on each of two days of the week typically have high BMIs. A 2018 study examined the associations of alcoholic beverage consumption with dietary intake, waist circumference, and BMI [139]. A total of 7,436 men and 6,939 women 20 to 79 years of age were included in the study. By average daily drinking volume, the differences in waist circumference and BMI between former and moderate drinkers were +1.78 cm and +0.65, respectively, in men and +4.67 cm and +2.49, respectively, in women. Compared with moderate drinking, heavier drinking volume (three drinks/day or more in men, two drinks/day or more in women) was not associated with higher waist circumference or BMI, whereas drinking five or more drinks/day was associated with higher waist circumference and BMI in men. There were no significant differences in women who consumed four or more drinks/day compared with women who consumed one drink/day [139].
It is also important to note those individuals who have undergone bariatric surgery. According to a research study conducted at a substance abuse treatment facility, bariatric surgery patients were more likely to be diagnosed with alcohol withdrawal than those who had not had the surgery [140]. In another study of patients in active weight management being considered for bariatric surgery, an inverse relationship was found between BMI and alcohol consumption—the more overweight the patient, the less alcohol was consumed [141]. Past-year alcohol consumption actually decreased as BMI increased. Surgeons felt it rare to have a patient excluded for bariatric surgery due to excessive alcohol consumption. The authors concluded that it is likely that food and alcohol compete at brain reward sites.
Excessive drinking may interfere with the absorption, digestion, metabolism, and utilization of nutrients, particularly vitamins. Individuals with alcohol use disorder often use alcohol as a source of calories to the exclusion of other food sources, which may also lead to a nutrient deficiency and malnutrition. In the late stage of the disease, patients may develop anorexia or severe loss of appetite, and refuse to eat. Persons with alcohol use disorder account for a significant proportion of patients hospitalized for malnutrition [137].
Direct toxic effects of alcohol on the small bowel causes a decrease in the absorption of water-soluble vitamins (e.g., thiamine, folate, B6). Studies have suggested that alcoholism is the most common cause of vitamin and trace-element deficiency in adults in the United States. Alcohol's effects are dose dependent and the result of malnutrition, malabsorption, and ethanol toxicity [142]. Vitamins A, C, D, E, K, and the B vitamins are deficient in some individuals with alcohol use disorder. All of these vitamins are involved in wound healing and cell maintenance. Because vitamin K is necessary for blood clotting, deficiencies can cause delayed clotting and result in excess bleeding. Vitamin A deficiency can be associated with night blindness, and vitamin D deficiency is associated with softening of the bones. Deficiencies of other vitamins involved in brain function can cause severe neurologic damage (e.g., deficiencies of folic acid, pyridoxine, thiamine, iron, zinc).
Thiamine deficiency from chronic heavy alcohol consumption can lead to devastating neurologic complications, including Wernicke-Korsakoff syndrome, cerebellar degeneration, dementia, and peripheral neuropathy [143]. Thiamine deficiency in patients with alcohol use disorder who are suffering from Wernicke-Korsakoff syndrome leads to lesions and increased microhemorrhages in the mammillary bodies, thalamus, and brainstem. This syndrome can also be associated with diseases of the gastrointestinal tract when there is inadequate thiamine absorption. All patients with alcohol use disorders should receive supplemental thiamine whenever entered into hospitalization or treatment to reduce this possibility.
Alcohol abuse is a major risk factor for many infectious diseases, especially pulmonary infections [144]. Studies have shown that alcohol abuse increases the risk for acute respiratory distress syndrome and chronic obstructive pulmonary disease [145,146,147,148]. Pneumonia, tuberculosis, and other pulmonary infections are frequent causes of illness and death among patients with alcohol use disorder [149]. Other infectious diseases that are over-represented among individuals with alcohol use disorder are bacterial meningitis, peritonitis, and ascending cholangitis. Less serious infections are chronic sinusitis, pharyngitis, and other minor infections.
Acute and chronic alcohol abuse also increase the risk for aspiration pneumonia. Alcohol use disorders are associated with increased risk of aspiration of gastric acid and/or oropharyngeal flora, decreased mucus-facilitated clearance of bacterial pathogens from the upper airway, and impaired pulmonary host defenses [150]. In addition, pathogenic colonization of the oropharynx is more common in patients with alcohol use disorder.
The consumption of alcohol alters T-lymphocyte functions, immunoglobulin production by B cells, NK cell function, and neutrophil and macrophage activities making patients with alcohol use disorder more susceptible to septic infection [151,152,153]. Studies have shown that animals given ethanol are unable to suppress infections that can ultimately result in progressive organ damage and death [154,155,156].
Although some people believe that alcohol helps them sleep, chronic excessive drinking can induce sleep disorders by disrupting the sequence and duration of sleep states and by altering total sleep time, as well as the time required to fall asleep [157,158]. Specifically, drinking within an hour of bedtime appears to disrupt the second half of the sleep period [159]. The person may sleep poorly during the second half of sleep, awakening from dreams and returning to sleep with difficulty, resulting in daytime fatigue and sleepiness [157,160].
Individuals with alcohol use disorder may be at increased risk for sleep apnea, a disorder in which the upper air passage narrows or closes during sleep [161,162,163,164]. The combination of alcohol, obstructive sleep apnea, and snoring increases a person's risk for heart attack, arrhythmia, stroke, and sudden death [165]. Obstructive sleep apnea significantly increases the risk of stroke or death from any cause, independent of other risk factors, including hypertension [166,167].
The most common neurologic abnormality among patients with alcohol use disorder is dementia syndrome, which manifests primarily as impairment in recent memory, and more subtle fluctuations in abstractions, calculations, and other aspects of cognitive functions. As previously stated, one specific neurologic complication resulting from thiamine deficiency is Wernicke-Korsakoff syndrome, which involves delirium, clouded sensorium, confusion, ophthalmoplegia, nystagmus, and ataxia [168]. Immediate administration of thiamine is usually successful in treating the symptoms, but in some cases permanent memory loss occurs [168]. Once delirium and confusion resolve, there is sometimes a profound loss in recent memory (out of proportion to the other cognitive deficits) and alcoholic peripheral neuropathy, which results in diminished sensitivity to touch, pinprick, and vibration (objectively, and paraesthesias subjectively).
The acute effects of alcohol on the nervous system are signs people commonly think of when they envision an intoxicated person, such as slurred speech, loss of coordination, unsteady gait, impairment of attention or memory, nystagmus, stupor, or coma. The degree to which the central nervous system is impaired is directly proportional to the BAC and degree of tolerance.
Alcohol affects most neurochemical systems including NMDA, GABA, serotonin, dopamine (DA), and opioid systems.
Alcohol inhibits NMDA systems, which may contribute to feeling intoxicated. NMDA receptors change as tolerance develops. These receptor systems are overactive during withdrawal. Alcohol also enhances the action of the GABA system, producing some of the symptoms of acute intoxication. GABA receptors are especially sensitive to alcohol. The GABA system is underactive during withdrawal, and the genes that control these receptors may have an impact on the risk of alcohol use disorder [169,170].
Alcohol causes the release of 5-HT, or serotonin. Lower 5-HT levels in the brain are associated with increased alcohol intake in animals and humans, while higher 5-HT levels are associated with slightly reduced alcohol intake. Several 5-HT genes may be related to the genetic risk of alcohol use disorder [11,46].
Alcohol activates DA in the reward system in the ventral tegmental area of the brain. Alcohol also causes the release of DA. Several DA receptors may be related to the genetic risk of alcohol use disorder [11,46].
Finally, alcohol causes the release of endogenous opioids. Opioid receptors change with tolerance and withdrawal. Some receptors may affect genetic predisposition for alcohol use disorder, and opioid antagonists can decrease voluntary alcohol consumption. Alcohol may also affect acetylcholine, norepinephrine, and steroids.
Most people who drink do not develop brain damage. However, studies do indicate that impaired cognition and motor abilities occur in some individuals who are heavy drinkers. Older persons with alcohol use disorder exhibit more brain tissue loss than both older and younger persons without alcohol use disorder. These results suggest that aging may render a person more susceptible to the effects of chronic excessive alcohol. Most studies suggest that, following long-term abstinence, most brain changes resolve.
Magnetic resonance imaging has been used to measure changes in the brain structure and volume in persons with alcohol use disorder at three weeks after abstinence from alcohol [171]. The results indicated that the brain volume in men and women with alcohol use disorder was significantly reduced as compared with healthy men and women. The differences, however, were much more significant in women than in men [172]. These results indicate that alcohol inflicts greater neurotoxic effects in women with alcohol use disorder than men, but again, these brain changes may resolve with long-term abstinence. Results from a 2021 study found significant impacts of age, but not sex, on white matter microstructure in individuals with alcohol use disorder [173].
Although the literature on gender differences in addiction can appear at times to be inconsistent, as a whole men are more substance dependent than women for all substances except benzodiazepines and analgesics, on which women are equally or more frequently dependent [174]. However, on average, women show the effects of alcohol more immediately, more intensely, and for longer periods of time than men. They achieve higher concentrations of alcohol in the blood after drinking the same amounts of alcohol [175]. Women also produce a lower level of the enzymes required to break down alcohol. In addition, female hormones make women's bodies more susceptible to alcohol at certain times of the menstrual cycle. Women also tend to be shorter and weigh less than men. Because women generally have a higher percentage of body fat, they reserve alcohol in the body for longer periods of time. This is important because when a person drinks a large amount of alcohol, it is deposited in fatty tissues. Neurophysiology is more compromised in women with alcohol use disorder than men [176].
It may be because of these factors that women develop alcohol problems more quickly than men, and their progression to severe complications, such as liver disease, is more rapid. The death rate among women with alcohol use disorder is 50% to 100% greater than that of men because of their increased risk for suicide, alcohol-related accidents, cirrhosis, and hepatitis [177]. It is important to note, however, that women are more likely than men to obtain help, participate in treatment, and have long-term involvement in AA, and therefore are more likely to have better life outcomes [178].
International studies of gender differences indicate that the greater the societal gender equality in a country, the smaller the gender differences in drinking behavior. The gender gap in alcohol drinking is one of the few universal gender differences in human social behavior [179].
The dangers of drinking while pregnant are well-documented. Pregnant women who drink risk the chance of their child developing FASD. Prenatal alcohol exposure is known to be toxic to the developing fetus and is one of the leading known preventable causes of intellectual disability. Excess fetal mortality secondary to drinking is most prevalent during the first trimester of pregnancy. Even drinking as little as one beer a day has been associated with decreased birth weights and spontaneous abortions. Although FASD has received a great deal of publicity, the majority of people may not understand it correctly. For example, one large study of adults 18 to 44 years of age found that the majority of respondents incorrectly assumed that FAS referred to infants born with an addiction to alcohol.
FASDs refer to the whole range of conditions that can affect the offspring of mothers who drank alcohol during pregnancy. These conditions can affect each person in different ways and can range from mild to severe. A person with an FASD might have [180]:
Abnormal facial features, such as a smooth ridge between the nose and upper lip (the philtrum)
Small head size
Shorter-than-average height
Low body weight
Poor coordination
Hyperactive behavior
Difficulty with attention
Poor memory
Difficulty in school (especially with math)
Learning disabilities
Speech and language delays
Intellectual disability or low IQ
Poor reasoning and judgment skills
Sleep and sucking problems as an infant
Vision or hearing problems
Problems with the heart, kidneys, or bones
There are a variety of conditions that are considered FASDs. Alcohol-related neurodevelopmental disorder (ARND) is associated with intellectual difficulties and problems with behavior and learning. Patients with ARND may do poorly in school, with particular issues with math, memory, attention, judgment, and impulse control [180]. Offspring of mothers who consumed alcohol, during pregnancy may also develop alcohol-related birth defects, including congenital malformations of the heart, kidneys, and/or bones or hearing problems.
The most commonly studied FASD is FAS. FAS is defined by the existence of certain physical characteristics of children whose mothers drank during pregnancy. These characteristics include [180]:
Intellectual disability
Growth deficiencies
Central nervous system dysfunction
Decreased brain size
Low birth weight
Distorted facial features
Behavioral maladjustments
Abnormal joints and limbs
Other less visible symptoms of FAS include [181,182,183,184,185,186,187,188,189,190]:
Verbal learning and memory problems
Visual-spatial learning problems
Attention deficits and hyperactivity
Increased reaction time/slow information processing
Executive function problems
Structural and functional changes in the brain
Alcohol apoptotic neurodegeneration has been shown to appear in the forebrain when rats are injected with alcohol. Seven-day-old rats were divided into a group receiving saline solution and another group receiving alcohol solution. The brains were examined after 24 hours of ingesting alcohol or saline. The alcohol group showed a very dense, widely distributed area of deterioration (cell death). When alcohol is administered, various neurons in the forebrain show sensitivity. Also, the brain weight of the alcohol-treated rats was much lower than the saline group. Exposure of the developing rat brain to alcohol for a certain period of time during a specific developmental stage induces destruction of brain cells that deletes large numbers of neurons from several areas of the brain. This period of time in humans is the last three months of gestation [191].
Alcohol is especially neurotoxic to the developing fetus. Vulnerability is highest at six months' gestation to several years after birth. During this period, alcohol exposure can kill millions of neurons in the developing brain. This helps to explain reduced brain size and behavior disturbances associated with FAS. The most disabling effects are hyperactivity and learning disabilities, depression, and psychosis. Depending on the time of exposure to alcohol, different neurons are depleted, which shows evidence of alcohol being an agent that can contribute to many mental disabilities.
The Centers for Disease Control and Prevention (CDC) reported in 2022 that nearly 14% (or 1 in 7) pregnant women 18 to 44 years of age used alcohol and 5.0% (1 in 20) were binge drinkers [192]. According to the CDC, the cost to the United States for FAS alone is more than $4 billion annually [192]. Additionally, an estimated 6 to 9 out of 1,000 U.S. school children may have FASDs [180]. Binge drinking among pregnant women increased from 4.7% in 2021 to 5.3% in 2022[12]. FASD is 100% preventable when pregnant women abstain from drinking alcohol [180,193].
Living with a non-recovering family member with alcohol use disorder can contribute to stress for all members of the family. Children raised in these families have different life experiences than children raised in nonalcoholic families. For example, children living with a non-recovering alcoholic score lower on measures of family cohesion, intellectual cultural orientation, active recreational orientation, and independence. They also experience higher levels of conflict within the family. Many children of alcoholics experience other family members as distant and noncommunicative and may be hampered by their inability to grow in developmentally healthy ways. The level of dysfunction or resiliency of the nonalcoholic spouse is a key factor in the effects of problems impacting the children. Support groups, such as Children of Alcoholics, are available to help people deal with these issues.
Alcohol use disorder usually has strong negative effects on marital relationships. Separated and divorced men and women were three times as likely as married men and women to say they had been married to a person with alcohol use disorder or problem drinker. Almost two-thirds of separated and divorced women and almost one-half of separated or divorced men younger than 45 years of age have been exposed to alcohol use disorder in the family at some time. As of 2022, approximately 29.7 million Americans met the diagnostic criteria for alcohol use disorder; this number represents an increase from 18.1 million Americans in 2002 [12].
The majority of studies suggest an increased prevalence of alcohol use disorder among parents who abuse children. Existing research suggests that alcoholism is more strongly related to child abuse than are other disorders, such as parental depression, but the most important factor is whether the abusive parent was abused themselves or witnessed a parent or sibling being abused. Although several studies report very high rates of alcoholism among the parents of incest victims, much additional research in this area is needed [194,195].
Among some individuals and subgroups, excess alcohol consumption is associated with the risk of violent behavior. Alcohol may encourage aggression or violence by disrupting normal brain function, especially in levels of serotonin [196]. There is considerable overlap among nerve cell pathways in the brain that regulate aspects of aggression, sexual behavior, and alcohol consumption. Alcohol may weaken brain mechanisms that normally restrain impulsive behaviors, including inappropriate aggression.
Drinking and violence may occur together by chance. Also, violent criminals who drink heavily are more likely to be caught and consequently are over-represented in samples of people arrested for violent behavior. Antisocial personality disorder (ASPD) and early-onset alcoholism are common traits in many criminals. A person who intends to engage in a violent act may drink to bolster his or her courage or in hopes of evading punishment or censure. The motive of drinking to avoid censure is encouraged by the popular view of intoxication as a "time-out," during which a person is not subject to the same rules of conduct as when sober. Such alcohol-violence interactions are not readily treated. However, ongoing research has identified medications that have the potential to reduce violent behavior in both alcoholic and nonalcoholic subjects.
Young men who exhibit violent and antisocial behaviors often "burn out" with age [197]. By the time they reach 40 years of age, serotonin concentrations are increasing and testosterone concentrations are decreasing, both of which help to restrain violent behavior [198].
Research suggests that increasing the unit price of alcohol by raising alcohol taxes is an effective strategy for reducing excessive alcohol consumption and related harms, including violent behavior [199]. An examination of the impact of the price of alcoholic beverages on violence and other delinquent behavior among college students found that an increase in the price of beer could reduce the overall number of students involved in some sort of violent behavior by 4% [200]. In a study that used data from the National Household Survey on Drug Abuse, higher taxes on beer led to significant reductions in crime (e.g., property damage, use of force), with the largest impact among individuals younger than 21 years of age [201]. Another study that examined the impact of tax increases and advertising bans on reducing the prevalence of underage drinking and subsequent alcohol-related harms found both interventions to be effective [202]. A literature review of studies of underage populations found that increased taxes were significantly associated with reduced consumption and alcohol-related harms [203]. Public policies that affect the price of alcohol appear to have significant effects on alcohol-related disease and injury rates. The results of one systematic review suggest that doubling the tax on alcoholic beverages could reduce alcohol-related violence by 2% and crime by 1.4% [204].
According to the National Council on Alcoholism and Drug Dependence, on college campuses each year an estimated 696,000 students 18 to 24 years of age are assaulted by another student who has been drinking, and 97,000 students report experiencing alcohol-related sexual assault or date rape [205]. Four out of every five juvenile and teen arrestees are under the influence of alcohol or drugs while committing their crimes, test positive for drugs, are arrested for committing an alcohol- or drug-related offense, admit having substance abuse problems, or share some combination of these characteristics [206].
In 2022, 10,317 people died in alcohol-related traffic fatalities, accounting for 32% of all traffic-related deaths in the United States [207]. Of the 1,090 traffic-related fatalities in 2020 among children 0 to 14 years of age, 229 (21%) involved an alcohol-impaired driver [208]. Of the estimated 127 million episodes of alcohol-impaired driving among U.S. adults, 1 million drivers were arrested for driving under the influence [208]. The CDC estimates that 32 people in the United States die in alcohol-related crashes every day [208]. In a study of persons who have been convicted of driving while impaired, 85% of women and 91% of men reported a lifetime alcohol use disorder [209]. Psychiatric comorbidity may be a key element distinguishing driving under the influence (DUI) offenders from others and in distinguishing repeat offenders from first-time offenders [210].
A study found that although marijuana's effects on driving performance were small or moderate when taken alone, the effects were severe when combined with even a low dose of alcohol. These findings are very serious considering the frequency with which these two substances are combined, especially in young inexperienced drivers [211,212,213,214,215]. One study conducted at seven trauma centers involving 4,243 drivers who were seriously injured in crashes found that 54% of these drivers tested positive for alcohol and/or drugs from September 2019 to July 2021. Of these, 22% of the drivers tested positive for alcohol, 25% tested positive for cannabis, 9% tested positive for opioids, 10% tested positive for stimulants, and 8% tested positive for sedatives [216].
Persons with alcohol use disorder, like other addicts, generally have comorbid disorders, meaning they have alcohol problems as well as other illnesses or conditions [217]. These problems may include personality disorders (formerly Axis II disorders), other drug use (especially tobacco use disorders), or a number of psychiatric disorders, from major depression and bipolar illness to eating disorders and anxiety disorders. One study reported that 50% of women and 33% of males with a history of alcohol use disorders have at least one other psychiatric disorder [209]. Treatment of the comorbid disorder is absolutely essential in preventing relapses to drinking and in preventing other adverse consequences, such as suicide among patients with depression and alcohol use disorder.
Alcohol is both a stimulant and a depressant, depending on the levels and time after drinking. Patients with alcohol use disorder are often misdiagnosed with depression because of the many symptoms that mimic depression. Insomnia, reduced appetite, and decreased energy are just a few of the symptoms that can occur in both diseases. Alcohol can cause temporary depressive symptoms, even in persons who have no history of depression. In fact, as many as 80% of men and women with alcohol use disorder complain of depressive symptoms, and at least one-third meet the criteria for a major depressive disorder (excluding, of course, criterion D) [218]. Depression is often a comorbid disorder but can also be solely or partially due to alcohol. This carries important implications in the way depressive symptoms are evaluated and treated in patients with alcohol use disorders. Alcohol intoxication, especially binge drinking, can also cause mood swings that mimic the "highs" of people with manic depression/bipolar disorder. Thirty to fifty percent of persons with alcohol use disorder suffer from major depression at the same time [218,219]. Studies have found that many cases initially diagnosed as substance-induced depression were later reclassified as independent depression (i.e., not substance-induced) because the condition persisted after a period of abstinence [220].
How alcohol use disorder is related to depression is not clear. Some studies have suggested that both conditions may share common risk factors. For example, both problems may run in families. Co-occurrence is very common, but likely has independent though inter-related etiology.
Treatment professionals have found that after two to three weeks of abstinence from alcohol and with good nutrition, the temporary depressive effects of alcohol dissipate. However, there are subgroups of individuals with alcohol use disorder who have a co-occurring depression or manic depression, and it is critically important to diagnose and treat these illnesses during alcohol treatment. If true co-occurring depression is left untreated, many patients will drop out of treatment and relapse to drinking. Alcohol use disorders and depression are important risk factors for suicidal thinking or actions. Because alcohol can increase impulsivity and make depression worse, even intolerable, alcohol is often a factor in suicides.
Suicide is the 2nd leading cause of death among persons 25 to 34 years of age and the 3rd leading cause of death among persons 15 to 24 years of age [221]. Most people who attempt suicide and 90% of suicide victims have a diagnosable psychiatric disorder [222]. Alcohol is the number one drug of abuse associated with suicide. In 2021, 48,183 people in the United States died by suicide and an estimated 1.7 million attempted suicide [221]. Among people who attempt suicide, alcohol use disorder is a common diagnosis. Major depression and alcohol use disorder, respectively, are the most commonly diagnosed psychiatric disorders in patients who die by suicide. They also are more common among planned suicide attempts than among impulsive attempts [223]. Next to age, alcohol and drug addictions are the second most important risk factors in suicide. As many as 85% of individuals who die by suicide suffer from depression or alcohol use disorder, and 70% of patients with comorbid alcohol use disorder and depression report that they have made a suicide attempt at some point in their lives [224]. The reported likelihood of suicide in diagnosed alcoholism is between 60 and 120 times that of persons without mental illness [225]. Suicide attempts among women with alcohol use disorder are more mental health-related, whereas suicide attempts among men are more related to the severity of alcohol use disorder [226].
Alcohol intoxication can exaggerate depression and increase the likelihood of an impulsive act like suicide or other forms of violence. Alcohol use is frequently detected in suicide methods involving firearms, driving a vehicle, or overdosing. Alcohol impairs judgment and lowers the threshold to attempt suicide, explaining its association with suicide methods that involve a high level of pain [227]. In a case-control study, researchers examined the relationship between near fatal suicide attempts and aspects of alcohol consumption, such as amount and frequency of drinking, alcoholism, binge drinking, and drinking within three hours of a suicide attempt, and found a J-shaped relationship between alcohol exposure and near lethal attempts for all measures [228].
In a comprehensive review of the subject, it is estimated that the lifetime suicide risk among individuals with alcohol use disorder is 10% to 15%, a figure 5 to 10 times greater than seen in the general population [227,229]. Between 15% and 20% of persons with alcohol use disorder will attempt suicide, and of those who have attempted in the past, 15% to 20% will attempt suicide again in the next five years [229]. Approximately 40% of all patients seeking treatment for alcohol use disorder report at least one suicide attempt at some point in their lives [227].
One study conducted in Japan showed that, among drinkers, the risk of suicide increased with the amount of alcohol consumed. An unusual finding of this study was a U-shaped relationship between alcohol and suicide. Abstainers also have a significantly increased risk, similar to heavy drinkers. Among middle-aged males, moderate drinkers had the lowest risk for suicide [230].
In order to be most effective at the prevention of suicide, healthcare providers should be adept at eliciting both a substance use history and a psychiatric history. Risk factors associated with completed suicide with alcohol use disorder include comorbid major depression, active drinking, serious medical illness, living alone, and interpersonal loss and conflict.
Male, alcoholic, and depressed are the most common descriptors for suicide attempters. Always evaluate persons with alcohol use disorder for depression, suicide, and appropriate referral to a psychiatrist or psychologist. Depression and alcohol use disorder are common problems in the United States. Both are at the top of the list of problems that commonly require psychiatric treatment. Unfortunately, both problems are difficult to diagnose by physicians due to patient fears and stigma and the realities of a busy medical office. Treating one problem but not the other is also very common. In order to successfully treat alcohol use disorder and depression it is important that healthcare providers diagnose and treat both problems.
Treatment of alcohol use disorder begins with evaluation, stabilization, and detoxification and the appropriate level of treatment, which may include a 12-step program. Adding an antidepressant and treating the depression requires a number of subtle changes in thinking. First, the physician must be convinced that the depression is not transient and related to alcohol or detoxification or so severe that the patient is unable to do treatment work. Next, the patient must be willing to accept and adhere to simultaneous, coordinated treatment.
The next issue is determining which antidepressant to use. Lithium and tricyclics used to treat depression alone may not be effective or could have serious adverse effects when used in patients with comorbid depression and alcohol use disorder. Another class of antidepressants, selective serotonin reuptake inhibitors (SSRIs), has been studied to treat depression after failing to treat alcohol use disorder. SSRIs generally cause less serious adverse effects than tricyclics, but some, like fluoxetine, work slowly and cause sexual performance side effects. SSRIs, such as fluoxetine, sertraline, and paroxetine, and herbal remedies such as St. John's wort have been tried in a variety of studies and are generally able to help alleviate depression, but do not appear to help with drinking outcomes. Results of a systematic review found only low-quality evidence to support the use of antidepressants for the treatment of co-occurring depression and alcohol use disorder [231]. A Japanese study observed lower response to antidepressant treatment in patients with comorbid depression and alcohol use disorder [232]. Venlafaxine and bupropion appear to be especially effective in treating patients with depression and alcohol use disorder. Venlafaxine is well suited to treat alcohol use disorder with depression and even depression with anxiety [233]. Venlafaxine is effective in mild and severe depression with anhedonia. Bupropion is effective as well, but it has seizure risks in this population [234]. One study that evaluated treatment outcomes in patients with comorbid alcohol use disorder and depression found venlafaxine and bupropion to be less effective than antidepressants [235]. Men with depression who are using alcohol appear very sensitive to the sexual side effects of the SSRIs and may discontinue their use and drop out of treatment. Both pharmacologic and behavioral treatments have demonstrated efficacy for patients with comorbid depression and alcohol use disorder; however, treatment response is modest, particularly for drinking outcomes [236]. Transcranial magnetic stimulation is now available for refractory depression, and studies are in progress for its use in treating substance use disorder [237].
A 2000 study analyzed the substance/alcohol abuse patterns of 89 patients with a confirmed diagnosis of bipolar disorder (71 with bipolar I and 18 with bipolar II) [238]. The diagnosis was confirmed by a structured clinical interview for DSM-IV Axis I, an attending psychiatrist, a medical records review, and family members. The age of the patients ranged from 18 to 65 years. Among those with bipolar disorder I, 41 patients (57.8%) abused or were dependent on one or more substances (including alcohol), 28.2% abused or were dependent on two substances, and 11.3% abused or were dependent on three or more substances. Among those with bipolar disorder II, 39% of patients abused or were dependent on one or more substances, 17% were dependent on two or more substances, and 11% were dependent on three or more substances. The risk for substance or alcohol abuse was higher among patients with bipolar I disorder than with bipolar disorder II. Patients with both bipolar disorders I and II abused alcohol more often than any other substances [238]. One study sought to identify the demographic and clinical differences between patients with bipolar disorder both with and without alcohol use disorder [239]. Data from 238 patients with bipolar disorder included alcohol use, social demographics, longitudinal course of bipolar disorder, clinical features of depressive episodes, comorbid physical diseases, anxiety disorder, and other substance use disorder. Of the 238 patients with bipolar disorder, 74 had alcohol use disorder, with the best predictors of alcohol use disorder being male sex, younger age, and comorbidity with other unclassified substance dependence [239].
Alcohol withdrawal causes many of the signs and symptoms of anxiety and can even mimic panic attacks. Alcohol works much like a benzodiazepine; many people who abuse and are dependent on alcohol have learned to drink to temporarily relieve anxious feelings.
Special problems exist for people who drink to self-medicate the symptoms of a true generalized anxiety disorder, social phobia, or panic disorder. Alcohol may provide temporary relief, but it is not a good treatment for shyness or an anxiety disorder. The price a person may pay for self-medication are two diseases: anxiety and alcohol use disorder. Social anxiety can be a major impediment to active participation and even attendance to group therapy and 12-step meetings.
Pain is a subjective experience, and the perception of being in pain is an important factor of the alcohol use disorder. It is hypothesized, as well as established in some research, that individuals in pain will drink as a means to decrease their perception of pain or as a reaction to painful stimuli [240]. According to the National Institute on Alcohol Abuse and Alcoholism, an estimated one in four adults in chronic pain reports self-medicating with alcohol and 43% to 73% of people with alcohol use disorder report experiencing chronic pain [241].
All drugs of abuse, including alcohol, cause dopamine release in the mesolimbic system in the brain. This dopamine system, sometimes referred to as the neuroanatomy of pleasure or reinforcement, starts in the ventral tegmental area and projects to the nucleus accumbens. Alcohol- or drug-taking results in a dopamine reward that stimulates its taking. Pavlovian conditioning to environmental cues (e.g., sights, smells, and sounds of a bar) that precede use become associated with use of the drug. Notably, this sense of "reward," which confers evolutionary fitness, is more likely to be perceived as crucial than even that produced by natural, survival-oriented stimuli (e.g., food, sex). This conditioning is reflective of synaptic strengthening mediated by the glutamatergic system, with neuroplasticity changes in brain areas thought to mediate drug-taking behavior, including the amygdala (stress and anxiety), hippocampus (memory), and dorsal striatum (routine motor movements). Natural stimuli (e.g., food, sex, other previously pleasurable activities) become less enjoyable, resulting in a profound state of anhedonia. With time, alcohol use disorders become ingrained. Ultimately, this preference for alcohol compared to natural rewards is mediated through a process of "bad learning," or neuroplasticity changes in the extended amygdala, also referred to as the antireward system. The anti-reward system involves stress-response hormones, including corticotrophin-releasing hormone and dynorphin. Long- or short-term abstinence activates the antireward system, and with more abstinence, it becomes even more difficult to ignore with the attendant anxiety, dysphoria, craving, and anhedonia. Over time, with repeated administration, nucleus accumbens dopamine receptors desensitize, leading to a functional decrease in available dopamine, anhedonia, and decreased sense of pleasure. Real-world examples include an individual with alcohol dependence developing a sudden craving for a drink when watching a beer commercial, walking by a bar, or seeing a place where s/he drinks. This stage reveals one of the remarkable properties of addiction; the act of drug-taking transitions from being impulsive (i.e., pleasure-seeking without afterthought) to compulsive (i.e., undertaken to relieve stress, tension, or physical signs such as pain).
Alcohol use disorders are often associated with dependence on or abuse of other substances, such as marijuana, cocaine, opioids, amphetamines, anxiolytics, designer or "club drugs," and tobacco. Alcohol may be used to alleviate the unwanted effects of these other substances or to augment their effects or substitute for them when they are not available.
According to the most recent National Survey on Drug Use and Health, about 5.2 million Americans 12 years of age and older were past year cocaine users in 2019 [12]. Many cocaine addicts also use alcohol to enhance euphoria, to reduce the mania associated with intoxication, or to calm or reduce the impact of dysphoria caused by cocaine withdrawal. Use of cocaine impairs both mental and physical functions, including learning and memory, hearing and seeing, motor coordination, speed of information processing, and problem-solving ability. Alcohol use has its own set of impairments, but many overlap with cocaine use. The negative impact exerted by alcohol and cocaine on either mental or physical activities has been found to be greater than when either is used alone. This is due to the production of a compound called cocaethylene. Cocaethylene is a novel compound that is produced in the bodies of individuals using cocaine and alcohol. Cocaethylene has been linked to cardiotoxicity, neurotoxicity, overdose deaths, and acute functional impairment [242,243]. The combination of cocaine and alcohol may be associated with other neurologic changes, including poor memory and poorer judgment. Alcohol use can also be a trigger for cocaine relapse.
As many as 50% of persons with alcohol use disorder smoke, compared with about 18% of the general population [12,244]. In a cohort study of 845 persons who had been treated for alcohol use disorder, more than 25% of the sample had died within 12 years [245]. Approximately one-half of the deaths were related to tobacco use and one-third were related to alcohol. Smoking and excessive alcohol use are risk factors for cardiovascular and lung diseases and some forms of cancer. Compared to nonsmoking nondrinkers, the risk for developing mouth and throat cancer is seven times greater for those who use tobacco, six times greater for those who use alcohol, and 300 times greater for those who use both tobacco and alcohol [246].
Both nicotine and alcohol consumption cause the release of dopamine in the nucleus accumbens. Neurobiology may make the combination of the two substances more rewarding than if either substance was taken alone. Certain enzymes in the liver (i.e., microsomal enzymes) convert some of the ingredients found in tar from cigarette smoke into chemicals that can cause cancer [247]. Long-term excessive alcohol consumption may activate these enzymes as well as decrease the body's ability to respond to infections or abnormal states. Smoking and excessive alcohol use are significant risk factors for cancer of the mouth, throat, and esophagus [246].
A 2000 study revealed that people who smoke, drink alcohol (one or more drinks per day) and develop non-small cell lung cancer had more mutations in the p53 gene when compared to those who smoked only or did not smoke or drink [248]. Mutations in the p53 gene have been seen in smoking-associated tumors and were present more often in alcohol drinkers who smoked cigarettes, than in nondrinkers who smoked cigarettes or in nondrinkers who did not smoke. Seventy-six percent of patients who consumed one or more alcoholic drinks per day and smoked were found to have mutations in the p53 gene. In contrast, 42% of smokers who did not drink (consuming less than one drink per day) had gene mutations [248].
A 2006 study sought to determine how nicotine delivered by tobacco smoke influences alcohol intake. Findings suggest that smoking increases alcohol consumption in at least a subset of smokers [249]. Animal studies have found that chronic nicotine use leads to escalation of alcohol self-administration through a dysregulation in opioid signaling [250,251].
One of the major barriers to treating tobacco dependence in patients with a co-occurring alcohol use disorder is the notion that it is too difficult to quit both alcohol and tobacco and that attempts to quit tobacco might adversely affect the patient's recovery from alcoholism [252,253]. Treatment facilities often concentrate on the "primary" addiction to alcohol and treat tobacco use as a more benign addiction. Fewer than 1 in 10 treatment facilities ban tobacco use on their grounds and many treatment facilities do not screen for or treat tobacco dependence [254]. Moreover, many treatment facilities enable patient smoking by adjourning meetings for "smoke breaks" and allowing staff to smoke openly with patients [255]. In fact, studies show that quitting smoking does not cause abstinent alcoholics to relapse and may actually decrease the likelihood of relapse [256]. Further, quitting smoking has been found to facilitate drinking cessation among tobacco and alcohol co-users [257].
Alcohol use disorder and eating disorders are commonly comorbid conditions, with patterns of comorbidity differing by eating disorder subtype [258]. A community-based sample of women found that those with lifetime alcohol use disorder or nicotine dependence were at higher risk for eating disorders [259]. The process of alcohol detoxification and treatment is often accompanied by overeating with weight gain, and in some cases food becomes a replacement for alcohol [141,260].
As lotteries proliferate and states legalize casino gambling, pathologic or compulsive gambling is being recognized as a major public health problem. Alcohol use disorder is often a comorbid condition among compulsive gamblers. As with depression, each disorder can make the other more serious. Individuals with alcohol use disorder may bet more money and may be reluctant to quit chasing their losses. In one study, subjects received either three alcoholic drinks or an equal volume of a nonalcoholic beverage (placebo) [261]. The alcohol group persisted for twice as many gaming trials as the placebo group. One-half of the alcohol group lost their entire cash stake, compared with 15% of the placebo group [261].
Another study examined how alcohol affects judgment and decision-making during gambling, with a focus on sequential decision-making, including the gambler's fallacy (i.e., thinking that a certain event is more or less likely, given a previous series of events) [262]. Thirty-eight male participants completed a roulette-based gambling task 20 minutes after receiving either an alcoholic or placebo beverage. The task measured color choice decisions (red/black) and bet size, in response to varying lengths of color runs and winning/losing streaks. Color choice affected run length in line with the gambler's fallacy, which further varied by previous wins or losses. Bet size increased particularly for losing streaks. The alcohol group placed higher bets following losses than did the placebo group [262].
Problem drinking described as severe is given the medical diagnosis of alcohol use disorder. An estimated 28.8 million adults 18 years of age and older in the United States have an AUD, including 17.1 million men and 11.7 million women. In addition, an estimated 753,000 adolescents 12 to 17 years of age had an alcohol use disorder [63].
AUD is a chronic relapsing addiction previously called alcoholism and characterized by an impaired ability to stop or control alcohol use despite adverse social, occupational, or health consequences. To be diagnosed with alcohol use disorder, individuals must meet the specific DSM criteria. Using the DSM-5-TR, anyone meeting any 2 of the 11 criteria during the same 12-month period receives a diagnosis of alcohol use disorder. The severity of the disorder—mild, moderate, or severe—is assigned based on the number of criteria met.
A variety of screening instruments are available to detect unhealthy alcohol use in adults. After conducting a systematic evidence review of trials published between 1985 and 2011 on screening and behavioral counseling interventions for unhealthy alcohol use in adults, the U.S. Preventive Services Task Force (USPSTF) recommends that clinicians screen all patients 18 years of age or older for alcohol abuse using one of the following tools [263,264]:
The abbreviated three-question AUDIT-Consumption (AUDIT-C)
The NIAAA-recommended Single Alcohol Screening Question (SASQ)
The USPSTF concludes that there is insufficient evidence to determine the benefits and harms of screening for unhealthy alcohol use in adolescents 12 to 17 years of age [263].
The three questions on the Alcohol Use Disorders Identification Test-Concise (AUDIT-C) inquire about frequency of alcohol use, typical amount of alcohol use, and occasions of heavy use. The test takes one to two minutes to administer. Preliminary evidence suggests that the USAUDIT-C (based on U.S. standards) may be more valuable in identifying at-risk college drinkers [265]. In contrast, the SASQ inquires about past-year alcohol use and takes less than one minute to administer [263].
The CAGE questionnaire is the best known and most often studied screening tool used to detect alcohol problems. In an office setting, the four CAGE questions are often used to detect alcohol problems [266]. The first question, "Have you ever felt the need to cut down on your drinking?" is an easy question to ask. It is not threatening and at the same time suggests to the patient that you understand their pathologic attachment to alcohol [266]. A positive answer to the first and second questions strongly suggests further evaluation and brief intervention [263]. However, by itself, the CAGE questionnaire is not an adequate screening for alcohol use problems; it should trigger more intensive screening if positive [266,267].
Ask current drinkers the CAGE questions:
Have you ever felt that you should cut down on your drinking?
Have people annoyed you by criticizing your drinking?
Have you ever felt bad or guilty about your drinking?
Have you ever had a drink first thing in the morning to steady your nerves or get rid of a hangover (eye opener)?
If there is a positive response to any of these questions:
Ask if this occurred during the past year.
A patient may be at risk for alcohol-related problems if:
Alcohol consumption is:
Men:
14 drinks per week or 4 drinks per occasion Women:
7 drinks per week or 3 drinks per occasion
Or
One or more positive responses to the CAGE that have occurred in the past year
When is screening for alcohol problems appropriate?
As part of a routine health examination
Before prescribing a medication that interacts with alcohol
In response to presenting problems that may be alcohol related
One "yes" response to the CAGE questionnaire suggests an alcohol use problem. More than one "yes" is a strong indication that a problem exists [267,268].
If a patient is CAGE positive, or if clinical suspicion remains high, the AUDIT questionnaire may be administered and can be extremely useful in detecting alcohol problems [252]. The AUDIT was developed by the WHO to identify persons whose alcohol consumption has become problematic to their health [253]. Research has shown that the AUDIT may be especially useful when screening women and minorities and has shown promise when tested in adolescents and young adults [254]. The AUDIT consists of 10 screening questions with three questions about the frequency and amount of drinking, three about dependence, and four questions about problems caused by alcohol [246,249,255].
How often do you have a drink containing alcohol?
0 Never
1 Monthly or less
2 2 to 4 times a month
3 2 to 3 times a week
4 4 or more times a week
How many drinks containing alcohol do you have on a typical day when you are drinking?
0 1 or 2
1 3 or 4
2 5 or 6
3 7 or 8
4 10 or more
How often do you have 6 or more drinks on one occasion?
0 Never
1 Less than monthly
2 Monthly
3 Weekly
4 Daily or almost daily
How often during the past year have you found that you were not able to stop drinking once you had started?
0 Never
1 Less than monthly
2 Monthly
3 Weekly
4 Daily or almost daily
How often during the past year have you failed to do what was normally expected from you because of drinking?
0 Never
1 Less than monthly
2 Monthly
3 Weekly
4 Daily or almost daily
How often during the past year have you needed a first drink in the morning to get yourself going after a heavy drinking session?
0 Never
1 Less than monthly
2 Monthly
3 Weekly
4 Daily or almost daily
How often during the past year have you had a feeling of guilt or remorse after drinking?
0 Never
1 Less than monthly
2 Monthly
3 Weekly
4 Daily or almost daily
How often during the past year have you been unable to remember what happened the night before because you had been drinking?
0 Never
1 Less than monthly
2 Monthly
3 Weekly
4 Daily or almost daily
Have you or someone else been injured as a result of your drinking?
0 No
2 Yes, but not in the past year
4 Yes, during the past year
Has a relative or friend or a doctor or other health worker been concerned about your drinking or suggested you cut down?
0 No
2 Yes, but not in the past year
4 Yes, during the past year
The minimum score is 0 and the maximum possible score is 40. A score of 8 or more indicates a strong likelihood of hazardous or harmful alcohol consumption [203].
The AUDIT-C is a 3-question screening tool that can help identify persons who are at-risk drinkers (who may not be alcohol dependent) or who have active alcohol use disorders, including alcohol abuse or dependence [256].
How often do you have a drink containing alcohol?
0 Never
1 Monthly or less
2 2 to 4 times a month
3 2 to 3 times a week
4 4 or more times a week
How many standard drinks containing alcohol do you have on a typical day?
0 1 or 2
1 3 or 4
2 5 or 6
3 7 or 8
4 10 or more
How often do you have six or more drinks on one occasion?
0 Never
1 Less than monthly
2 Monthly
3 Weekly
A score of 4 or more in men and 3 or more in women (when not all points are from question 1) is considered positive for hazardous drinking or alcohol use disorder [273].
The SASQ consists of one question: "How many times in the past year have you had X or more drinks in a day?" [263]. The question is individualized based on sex, with X being five for men and four for women. A response of more than one is considered positive and requires additional assessment.
The Michigan Alcohol Screening Test (MAST) continues to be a good screening test for alcohol abuse and dependence, but for optimal results it should be used with a questionnaire that asks about the amount and frequency of alcohol consumption. The following questions are from the 13-item Short MAST (SMAST) regarding the respondent's involvement with alcohol during the past 12 months [249]:
Do you think you are a normal drinker? (By normal we mean you drink less than or as much as most other people.)
No = 1 Yes = 0
Does your wife, husband, a parent, or other near relative ever worry or complain about your drinking?
No = 0 Yes = 1
Do you feel guilty about your drinking?
No = 0 Yes = 1
Do friends or relatives think you are a normal drinker?
No = 1 Yes = 0
Are you able to stop drinking when you want to?
No = 1 Yes = 0
Have you ever attended a meeting of Alcoholics Anonymous?
No = 0 Yes = 1
Has drinking ever created problems between you and your wife, husband, a parent, or other near relative?
No = 0 Yes = 1
Have you ever gotten into trouble at work because of your drinking?
No = 0 Yes = 1
Have you ever neglected your obligations, your family, or your work for two or more days in a row because you were drinking?
No = 0 Yes = 1
Have you ever gone to anyone for help about your drinking?
No = 0 Yes = 1
Have you ever been in a hospital because of drinking?
No = 0 Yes = 1
Have you ever been arrested for drunken driving, driving while intoxicated, or driving under the influence of alcoholic beverages?
No = 0 Yes = 1
Have you ever been arrested, even for a few hours, because of drunken behavior?
No = 0 Yes = 1
Key: There are two definitions for this test.
| Seltzer Definition: |
| 0-1 points = Nonalcoholic |
| 2 points = Possibly alcoholic |
| 3 or "yes" to 6, 10, or 11 = Alcoholic |
| Ross Definition: |
| 5 points = Alcohol abuse |
There are certain risks and comorbidities (e.g., psychiatric and medical conditions requiring pharmacologic treatment) that may modify the criteria of at-risk drinking, especially within the geriatric population [275]. It is important for healthcare providers to assess each patient's threshold for alcohol use, taking into account their level of risk and comorbidities. The Comorbidity-Alcohol Risk Evaluation Tool (CARET) may be helpful in this task, with comorbidity-specific measures to place patients in "at-risk" or "not-at-risk" groups [276].
Communication with patients regarding history and current alcohol use patterns is a necessary step in determining if alcohol use has become a problem. When there is an obvious disconnect in the communication process between the practitioner and patient due to the patient's lack of proficiency in the English language, an interpreter is required. Frequently, this may be easier said than done, as there may be institutional and/or patient barriers.
If an interpreter is required, the practitioner should acknowledge that an interpreter is more than a body serving as a vehicle to transmit information verbatim from one party to another. Instead, the interpreter should be regarded as part of a collaborative team, bringing to the table a specific set of skills and expertise [277]. Several important guidelines should be adhered to in order to foster a beneficial working relationship and a positive atmosphere.
When interpreters are enlisted and treated as part of the interdisciplinary clinical team, they serve as cultural brokers, who ultimately enhance the clinical encounter. When providing care for patients for whom English is a second language, the consideration of the use of an interpreter and/or patient education materials in their native language may improve patient understanding and outcomes.
In addition, several organizations provide information and toolkits in languages other than English. The National Hispanic Medical Association offers an alcohol screening kit in Spanish, including patient education sheets [278]. The National Institute on Alcohol Abuse and Alcoholism also provides patient education brochures and pamphlets in English and Spanish [279].
The FDA has approved a test to detect alcohol use disorder and alcohol-related diseases. The test detects the level of carbohydrate-deficient transferrin (CDT) in the body, which is elevated in persons with alcohol use disorder and remains elevated even several weeks after drinking is stopped [280]. The advantages of the CDT test are reliability and the availability of automated test results within four hours [281,282]. The CDT is often used in combination with other screening tests, such as the gamma-glutamyl transferase (GGT) test. While both CDT and GGT are independently associated with alcohol abuse, combining tests may dramatically increase sensitivity [267,283]. CDT is less sensitive/specific in women than in men [267].
The relationship between alcohol and the liver serves as the basis for many of the tests that identify possible alcohol abusers. Alcohol markers for recent alcohol ingestion include urine/breath/blood, AlcoPatch, methanol, urinary ethyl glucuronide (EtG) and ethyl sulfate(ES), whole blood phosphatidylethanol, and the ratio of 5-hydroxytryptophol (5-HTOL) to 5-hydroxyindole-3-acetic acid (5-HIAA) [267,284].
The CDT test is often used to assess prolonged ingestion of high amounts of alcohol (more than 50–80 g/day for two to three weeks) [267]. Another test examines hemoglobin or whole blood acetaldehyde adducts. In a study of almost 3,000 women and 4,000 men, the combination of CDT and GGT compared with either alone shows a higher diagnostic sensitivity and specificity and is correlated more strongly with alcohol consumption than either test alone (Table 1) [285,286,287].
Tests in this category look at the classic toxic markers that use of alcohol leaves on the body. They include [267]:
Liver function tests
GGT
Aspartate aminotransferase (AST)
Alanine aminotransferase (ALT)
Red blood cell index
Mean corpuscular volume (MCV)
Despite the fact that alcohol abuse complications have caused grave illness and many deaths, physicians are not always good at detecting alcohol and other drug abuse in their patients. Even when physicians and other health professionals identify an individual with alcohol use disorder, they are sometimes unsure of how to proceed. At times, the physician will offer help but the patient refuses. Nevertheless, the addiction specialist or the primary care physician with a continuous, comprehensive, patient-centered approach to the medical, psychosocial, and family issues is the ideal person to offer intervention, treatment, and recovery support.
Almost 20% of patients treated in a primary care setting drink at levels that may place them at risk for developing alcohol-related problems [267,288]. Brief intervention, as part of primary healthcare, can help reduce this risk. Brief intervention is generally conducted over one to a few visits with each session lasting from just a few minutes up to one hour. The type of brief intervention varies depending on how severe the problem. Brief intervention is often used with patients who have not yet developed alcohol use disorder and the goal may be to reduce drinking rather than abstinence. For persons with alcohol use disorder, the goal of brief intervention is abstinence, and for these individuals, referral to a more comprehensive treatment may be necessary. The USPSTF recommends that clinicians provide patients who are engaged in risky or hazardous drinking with brief behavioral counseling interventions to reduce alcohol misuse [263].
Miller and Sanchez proposed six elements, summarized by the acronym FRAMES, to describe the key elements of brief intervention: feedback, responsibility, advice, menu of strategies, empathy, and self-efficacy [289]. How these elements enhance effectiveness has been supported in other reviews [290,291]. Goal setting, follow-up, and timing are also important in brief intervention [292,293].
Feedback of Personal Risk: Health professionals use current drinking behaviors, lab test results, and actual or potential consequences of drinking to provide patients with feedback on the risk of developing a problem.
Responsibility of the Patient: Brief intervention often includes encouraging the patient to recognize that it is his or her responsibility and choice to change the behavior. This gives patients a sense of personal control in the process of change.
Advice to Change: Brief intervention may also include recommendations about moderate- or low-risk drinking and advice on cutting down or eliminating alcohol consumption.
Menu of Ways to Reduce Drinking: Patients are advised about how to cut back or avoid alcohol consumption. Health professionals can help patients set limits, recognize reasons for drinking, and acquire skills to avoid high risk drinking. Often, self-help materials such as drinking diaries are given to patients to help monitor their progress.
Empathetic Counseling Style: Confrontational methods of brief intervention are not as effective as when health professionals use a more empathetic counseling approach.
Self-Efficacy or Optimism of the Patient: Patients should be encouraged during brief intervention to help themselves by creating a plan to change their behavior and to think positively about their ability to reduce or stop drinking. Health professionals often use motivation-enhancing techniques.
In addition to FRAMES, the following items are important:
Establishing a drinking goal: Patients should be encouraged to set a drinking goal with help from their physician. In some cases it is helpful to put the goal in writing. The drinking goal may be abstinence.
Follow-up: It is important that the healthcare provider follows the patient's progress by telephone calls, or repeat tests or visits.
Patients are more likely to change their behavior when they recognize they have a problem and when they are optimistic about prospects for change; therefore, evaluating readiness to change is an important part of brief intervention. Some patients are not ready to change at the start of brief intervention, but may be ready when they experience adverse consequences of alcohol use. One study found that 77% of patients who were very confident and motivated were able to reduce their alcohol consumption by using self-help instructions and drinking diaries [291]. Motivation techniques are more useful to the resistant patient than self-help instructions.
Motivational interviewing is a method of brief intervention that is used to help move individuals from the precontemplation, contemplation, or determination/preparation stage into the action stage of change related to their drinking. In addition to focusing on the patient's view of the problem and consequences of the behavior, the interview often includes a comprehensive assessment of drinking behaviors with personalized feedback. Motivational interviewing therapists emphasize personal responsibility and support their patients' feelings of self-efficacy for making a change in their drinking. This method has demonstrated empirical efficacy with problem drinkers [294].
Many studies have documented that brief intervention can assist patients without clinical alcohol use disorder to reduce alcohol consumption [295,296,297,298,299]. Brief intervention can also help motivate the patient with alcohol use disorder to enter treatment. One study in an emergency care setting found that 65% of individuals with alcohol use disorder who received brief counseling kept a follow-up appointment for treatment, compared with only 5% percent of those who did not have counseling [295]. Some studies have found that brief intervention may be as effective as more specialized treatment for some patients with alcohol use disorder [296,300].
Researchers conducted a comprehensive meta-analysis of controlled studies of brief intervention in treatment and nontreatment settings [301]. In this meta-analysis, effect sizes were calculated for several outcome variables, including amount and timing of alcohol consumed, abstinence rates and duration, proportion and duration of non-problem drinking, frequency of intoxication, laboratory markers, ratings of drinking severity, ratings of improvements, dependence symptoms, and problems resulting from drinking [301]. Two types of investigations were examined. The first group of studies (N=34) compared brief intervention to control conditions in non-treatment-seeking patients and generally did not include persons with severe alcohol problems. The second group of investigations (N=20) included persons who had more severe alcohol problems in a treatment setting. The effect size for brief intervention on alcohol consumption was noted at between three to six months in studies that excluded more severe alcohol problems [301]. This meta-analysis provides further documentation of the effectiveness of brief intervention provided by healthcare professionals to patients with alcohol problems, especially during the first three to six months after intervention. Effectiveness may decrease over time, so progress should be monitored and referrals made if necessary. For those with more severe alcohol problems in a treatment setting, brief intervention may be appropriate as an initial treatment with nonresponders receiving more extensive/intensive treatment [301].
In summary, brief intervention can help patients without disordered alcohol consumption reduce or stop drinking, can help motivate patients with alcohol use disorder to enter treatment, and can be used to treat some patients with alcohol use disorder. One study reported that brief intervention is associated with decreased alcohol consumption and decreased healthcare utilization, motor vehicle events, and other related costs [302]. The study also reported that the cost-benefit analysis suggests that for every $10,000 invested in early intervention, there will be a $43,000 decrease in future healthcare costs [302]. A meta-analysis of brief alcohol interventions for adolescents and young adults found that the interventions yielded modest, but clinically significant positive effects on problematic alcohol use trajectories among youth [303].
Readiness to Change was Dimension 4 of the third edition of the American Society of Addiction Medicine's (ASAM's) Six Dimensions of Multidimensional Assessment (also known as the ASAM Criteria) that is the standard for placement, continued stay, transfer, or discharge of patients with substance use disorder and co-occurring conditions [304]. Readiness to Change is now considered within each of the six dimensions in the fourth edition of the ASAM Criteria. The six dimensions of the fourth edition include: intoxication, withdrawal, and addiction medications; biomedical conditions; psychiatric and cognitive conditions; substance use-related risks; recovery environment interactions; and person-centered considerations (new). This new sixth dimension considers barriers to care, patient preferences, and need for motivational enhancement [304]. Several factors influence a person's readiness and ability to change behaviors. It is useful to help patients to weigh the risks of continued alcohol consumption and benefits of decreasing or eliminating alcohol consumption. Physicians can help motivate the patient to become ready for treatment if the patient appears ready to change.
Is the patient ready to change? The role of motivation is an important part of changing behavior. Table 2 summarizes the "pros" and "cons" of changing [305].
MOTIVATIONS FOR CHANGE
|
Patients are more likely to seek treatment if:
Patients are less likely to seek treatment if:
|
The Stages of Change Model is also useful in determining where a patient is in the process of change [306]. The stages of change include:
Precontemplation
Contemplation
Preparation
Action
Maintenance
Relapse
Treatment works. People who make the decision to stop drinking will be able to find the treatment and support they need to quit, remain sober, and regain their lives. However, as with treatment for any other disease, it is important to have a good idea of the options available in order to make informed choices.
To understand treatment and make the right treatment choices, it helps to have an overview. Treatment should be seen as having three phases.
Phase 1: Assessment and evaluation of disease symptoms and accompanying life problems including co-occurring medical and psychiatric conditions utilizing ASAM Criteria, detoxification (withdrawal management), acute stabilization of comorbid conditions, making treatment choices, and developing a plan
Phase 2: Residential treatment or therapeutic communities, intensive and regular outpatient treatment, medications to help with alcohol craving and to discourage alcohol use, medications to treat concurrent psychiatric illnesses, treatment of concurrent medical conditions, trauma and family therapy, 12-step programs, other self-help and mutual-help groups
Phase 3: Maintaining sobriety and relapse prevention with ongoing outpatient treatment as needed, facilitated group meetings, contingency management, 12-step programs, other self-help and mutual-help groups
Drug testing frequently, randomly, and for-cause should be a mandatory component of all phases. Transition from one phase to the next should not be based on time but on individual symptoms and progress.
First, the individual with alcohol use disorder must overcome denial and distorted thinking and develop the willingness to begin treatment—what AA calls the desire to stop drinking. At this stage, it is important to obtain the help of someone knowledgeable about treatment and the options available.
When getting started, some people have lost control over alcohol to such an extent that they will only be able to make immediate decisions and set the most basic goal of quitting drinking. Development of a detailed treatment plan with goals and choices may have to wait until after detoxification. On the other hand, getting started is exactly the place where some people with alcohol problems get stuck. In being stuck, denial is always a problem, but complete denial is not universal: people have various levels of awareness of their alcohol use problems, which means they are in different stages of readiness to change their drinking behavior. Professionals have taken advantage of this insight about alcohol use disorder to develop treatment approaches that are matched to a person's readiness to change. Addiction specialists can best decide which treatment is best and which is the less restrictive at specific times during recovery.
Individuals with alcohol use disorder must stop using in order to be able to progress in treatment, which can be done on either an inpatient or outpatient basis. Medical evaluation and treatment are particularly important at this stage. A large proportion of persons with alcohol use disorder develop dangerous withdrawal symptoms that must be medically managed either in a hospital or on an outpatient basis.
Although detoxification is a critical step for many with alcohol use disorder, most treatment professionals are reluctant to call it treatment, and for good reason. Treatment is what helps a person develop a commitment to change, keep the motivation to change, create a realistic plan to change, and put the plan in action. Successful treatment means a person begins to experience the rewards of seeing the plan work. Just taking away the alcohol does not automatically produce any of these outcomes.
Withdrawal Symptoms and Medical Management
Abrupt discontinuation or even cutting down on the amount of drinking by persons who are physiologically dependent on alcohol produces a characteristic withdrawal syndrome with sweating, rapid heartbeat, hypertension, tremors, anorexia, insomnia, agitation, anxiety, nausea, and vomiting [307]. In some ways, alcohol withdrawal resembles withdrawal from opioids, but unlike opioid withdrawal, which is rarely life-threatening in and of itself, alcohol withdrawal can be fatal. As many as 15% of persons with alcoholism progress from the autonomic hyperactivity and agitation common to withdrawal from other drugs to seizures and, for some, even death. In some cases, DT may occur within the first 48 to 72 hours and can include disorientation, confusion, auditory or visual hallucinations, and psychomotor hyperactivity [307].
The Revised Clinical Institute Withdrawal Assessment for Alcohol Scale (CIWA-Ar) is a symptom-triggered, 10-item scale that quantifies the risk and severity of alcohol withdrawal [307]. However, in order to be most useful, it requires patient input, which may not be feasible in patients undergoing severe DTs. If the patient is able, the assessment takes only minutes and aids in identification of patients who may need immediate pharmacologic treatment to prevent further complications. Very mild withdrawal usually corresponds with a score of 9 or less, mild withdrawal with a score between 10 and 15, modest withdrawal with a score between 16 and 20, and scores greater than 20 indicate severe withdrawal [308]. Patients scoring less than 9 may not require pharmacologic intervention. However, reassessment of symptoms should be performed every one to two hours until withdrawal is resolved.
Pharmacologic management of acute alcohol withdrawal generally involves the use of benzodiazepines, which reduce related anxiety, restlessness, insomnia, tremors, DT, and withdrawal seizures [307]. While benzodiazepines may have abuse liability in some patients, they have been safely used for years [309,310,311]. These medications may be administered either on a fixed interval or symptom-triggered schedule. However, both short-acting and long-acting benzodiazepines have their problems. The long-acting benzodiazepines can decrease rebound symptoms and work for long periods of time, but intramuscular absorption can be very erratic. Short-acting benzodiazepines have less risk of oversedation, no active metabolites, and considerable utility in patients with liver problems or disease. Yet, breakthrough symptoms can and do occur, and risk of seizure is imminent.
Patients with withdrawal symptoms are generally treated with diazepam or chlordiazepoxide until withdrawal subsides [307,309,310]. These medications are preferred due to their long action, which decreases the risk of rebound symptoms. If intramuscular administration is necessary, lorazepam is the drug of choice. More severe withdrawal is generally treated in a hospital setting. In patients with severe hepatic dysfunction, benzodiazepines that are metabolized outside the liver (lorazepam and oxazepam) are preferred. Treatment-resistant withdrawal warrants the use of phenobarbital or propofol, both with demonstrated efficacy in management [311].
Other medications may be used in conjunction with benzodiazepines for the treatment of withdrawal. Anticonvulsants, especially carbamazepine, are used safely to treat withdrawal [309]. They do not have abuse liability and have anticonvulsant and antikindling effects. Nevertheless, they also have problems. They do not reduce delirium and can have liver toxicity. The anticonvulsant gabapentin has demonstrated efficacy for mild alcohol withdrawal and early abstinence, but there is concern about its potential for abuse [312]. Alpha-adrenergic agonists like clonidine can reverse many of the behavioral symptoms of withdrawal but do not prevent seizure and can cause hypotension. However, for those patients with coronary artery disease, use of an alpha-adrenergic agonist or beta blocker may be indicated. It is important to note that these agents do not prevent seizures and can mask some signs of worsening withdrawal. They should be used only in conjunction with benzodiazepines [313]. More research is necessary regarding the efficacy of calcium channel antagonists in the treatment of alcohol withdrawal [314]. Studies have shown that those who have withdrawal seizures may have a worse prognosis than those who do not [92,93].
In the earliest views of alcohol use disorder, relapse to alcohol use was primarily seen as the patient's failure to respond to withdrawal treatment. After all, if the addicted person's primary problem was the trap of withdrawal, it would be reasonable to expect that the newly freed prisoner would gratefully and persistently grasp onto alcohol-free status, never to return voluntarily to the prison of addiction. But many people returned repeatedly for detoxification.
The medical profession was remarkably slow to recognize the ineffectiveness of repeated detoxification. Rather than question the underlying assumption that medical diagnosis and treatment of withdrawal was the solution to the problem of dependence, physicians seemed content to recycle people through one emergency room or detoxification experience after another for what often proved to be an addiction-shortened lifetime. Detoxification is only the first step in the treatment process, and the beginning of a lifelong process.
As the detoxification process occurs, careful evaluation should be done to identify co-occurring medical and psychiatric conditions that require acute stabilization. This should be done before facilitating a smooth transition to phase 2.
It is crucial to decide if the patient requires acute hospitalization or inpatient detoxification. It has been established that hospitalization can be cost-effective, but this is not always a possibility. However, if a patient appears to have acute intoxication, exhibits or will exhibit withdrawal symptoms that will require medical management, has failed outpatient detoxification, appears to be depressed or suicidal, relapses shortly after previous detoxification, has an extremely unstable home situation, or has the possibility of family disruption or job loss, then inpatient hospitalization is likely indicated. If you are in doubt, call a physician who is a member of the ASAM or the American Academy of Addiction Psychiatry who specializes in these problems.
The next step is what has been commonly known as "active treatment." Relapse to alcohol use disorder is most likely to occur in the first three to six months after a person stops drinking; a period characterized by physiologic abnormalities, mood changes, and complaints of anxiety, depression, insomnia, and hormone and sleep problems. Getting active help and support during the early months of sobriety is critical for treatment to succeed.
This is the stage in which a person gains the motivation necessary to maintain a commitment to sobriety, the knowledge and skills necessary to stay sober, and the support systems necessary to cope with all the problems of daily life (the problems that everyone has to face) without resorting to the old "solution" of drinking. This is when the assistance of a treatment professional is important. A professional can help patients better understand how alcohol has affected their lives, so they can set goals and develop a plan to stay sober. In addition, the treatment professional can assist the patient in choosing the treatment options that are right for them.
Some proven medications are available to help with alcohol craving and to discourage alcohol use and will be discussed in detail later in this course. The treatment professional will also need to choose medications and treatments for concurrent psychiatric illnesses, like depression or anxiety, if appropriate, or for a variety of health problems that often accompany alcohol use disorder.
Research has shown that the longer people stay in treatment, remain sober, and are actively committed to sobriety, the more likely it is that they will maintain sobriety. Some treatment professionals think of the phase of active treatment as lasting from 6 to 12 months. During the first critical months of treatment, people often need a variety of supports, especially drug testing and AA or other self-help groups, to achieve and maintain lasting sobriety.
It is often difficult to pinpoint when the active treatment phase ends and a person enters the maintenance phase of recovery. In phase 2, people learn what they need to do to stay sober and they develop the many skills they will use to avoid relapse. A person could be said to enter this maintenance and growth stage when he or she is comfortable with these skills and has had a chance to rely on them to stay sober when life throws them the inevitable curveballs, either as a crisis or an everyday problem. Many people in recovery attribute their ongoing sobriety to participation in a support group such as AA or Women for Sobriety.
A promising approach to maintain gains made in active treatment is a low-intensity, telephone-based approach. In a 2005 study, this program of follow-up care was compared with two more intensive face-to-face continuing care interventions. Patients with alcohol use disorder who had completed 4-week intensive outpatient programs were provided three 12-week continuing care treatments. Telephone-based continuing care was found to be an effective form of step-down treatment for most patients with alcohol use disorder who complete an initial stabilization treatment, compared with more intensive face-to-face interventions [315].
The grandfather of successful alcohol treatment is Alcoholics Anonymous, a self-help organization founded in 1935 that changed the way professionals thought about alcohol use disorder and treatment. AA developed a very successful 12-step program that combines self-help with a spiritual foundation and is based on the fellowship of recovering alcoholics. Although there is a spiritual foundation in AA, one is not required to be religious. The organization is run entirely by recovering alcoholics and reaches into virtually every community with a specific program as well as around-the-clock assistance. Membership is available to anyone wishing to join, and there are no financial dues. AA has probably done more to promote the self-help concept than any other organization.
For many people with alcohol use disorder, attending an AA meeting is like brushing their teeth. Prevention of relapse is an active daily process. AA provides fellowship that can be exceptionally positive and counterbalance the feelings of loss, grief, and shame often associated with alcohol use disorder.
AA and other 12-step programs are effective treatment programs that facilitate long-term abstinence after treatment, especially for patients with low psychiatric severity [316]. AA provides important peer-led support for individuals with alcohol use disorder. AA also helps individuals with relapse and relapse prevention by prescribing that people keep it simple, take it one day at a time, and avoid the people, places, and things associated with their use. They also help recovering alcoholics to develop positive lifestyles and find new ways to solve old problems. The feeling of fellowship, the support, and guidance to sobriety makes recovery more likely. Reduction of shame and guilt and acceptance of powerlessness over drinking may be reported by individuals with alcohol use disorder after attending meetings every day. An AA meeting may take one of several forms, but at any meeting you will find alcoholics talking about what drinking did to their lives and personalities, what actions they took to help themselves, and how they are living their lives today.
Patients can find the listing for a nearby AA group in the telephone book or online. Typically, a person in recovery will answer the telephone. Websites provide printable lists of all local meetings with time, location, types of meeting, and often directions. One of AA's principles is the value of performing services that will help other alcoholics. Answering the telephone at the local AA office is one of these services, reserved for those who have been in recovery long enough to answer questions in a knowledgeable manner and provide a nonjudgmental ear.
A Cochrane review found that AA, the premier mutual aid peer-recovery program definitely helps people get sober [317]. In addition, AA has significantly higher rates of continuous sobriety compared with evidence-based professional mental health therapy, such as cognitive-behavioral therapy, alone. AA was often was found to be markedly better than other interventions or quitting cold turkey. One study found the program 60% more effective than alternatives [317].
The study by Harvard and Stanford addiction researchers of 10,565 subjects determined that AA was nearly always found to be more effective than psychotherapy in achieving abstinence [317].This review concluded that AA participation improved the duration of abstinence and the amount they reduced their drinking (if they continued drinking). AA had harm reduction features as well, reducing the medical consequences of drinking and related healthcare costs. While not a random assignment treatment comparison study, in this analysis, AA was never found less effective than other treatments. As such, AA could be a helpful addition to any treatment for alcohol use disorder. For example, adding AA to naltrexone would be expected to be better than pharmacotherapy alone. Recent studies and the preponderance of evidence supports the effectiveness of 12-step program involvement in sustaining abstinence [10]. Stable and long-term abstinence was associated with living longer, better mental health, better marriages, being more responsible parents, and being successful employees.
A brain imaging study by Yale researchers showed that those diagnosed with alcohol use disorder showed disruptions of activity between the ventromedial prefrontal cortex and striatum, a brain network linked to decision making [318]. Time is necessary for re-learning how to be sober but also for brain recovery. The more recent the last drink, the more severe the disruption, and the more likely the individual will relapse to drinking. The Yale researchers also found that the severity of disruption between these brain regions recovers very slowly, day after day, gradually over time. They conclude the longer subjects with alcohol use disorder abstain from alcohol, the better. The number of days of alcohol abstinence at treatment initiation significantly affected functional disruption of the prefrontal-striatal responses to alcohol cues in patients with alcohol use disorder and brain imaging abnormalities [318].
Cognitive-behavioral therapies (CBTs) are among the most frequently evaluated approaches used to treat substance use disorders [319,320]. CBTs have been shown to be effective in several clinical trials of substance users [321]. Characteristics of CBTs include:
Social learning and behavioral theories of drug abuse
An approach summarized as "recognize, avoid, and cope"
Organization built around a functional analysis of substance use (i.e., understanding substance use with respect to its antecedents and consequences)
Skill training focused on strategies for coping with craving, fostering motivation to change, managing thoughts about drugs, developing problem-solving skills, planning for and managing high-risk situations, and cultivating drug refusal skills
Basic principles of CBTs are that [322,323]:
Basic skills should be mastered before more complex ones are given.
Material presented by the therapist should be matched to patient needs.
Repetition fosters the development of skills.
Practice is needed for mastery of skills.
The patient is an active participant in treatment.
Skills taught are general enough to be applied to a variety of problem areas.
Structured behavior therapy techniques can be effective components of alcohol use disorder treatment. Contingent incentive procedures are designed to enhance a patient's motivation to meet treatment goals by offering concrete rewards for specific performance outcomes.
Behavioral therapy techniques are often part of CBT. In this approach, substance use is believed to develop from changes in behavior and a reduction in opportunities for reinforcement of positive experience. The goal is to increase the person's engagement in positive or socially reinforcing activities. Techniques such as having patients complete a schedule of weekly activities, engaging in homework to learn new skills, role-playing, and behavior modification are used. Activity, exercise, and scheduling are major components of this approach based on the following:
Drug abuse patients need motivation and skills to succeed in stopping drug use.
Research has shown that drug abuse behavior can be reduced by offering contingent incentives for abstinence.
The most striking successes have come from positive reinforcement programs that provide contingent incentives for abstinence using money-based vouchers as rewards.
Research provides examples, but treatment providers may need to be creative in discovering reinforcers that can be used for contingency management in their own clinical settings.
Family therapy is a highly effective treatment for alcohol use disorder, especially in adolescents. While most treatments emphasize the individual as the target of intervention, the defining characteristic of family therapy is the transformation of family interactions. Repetitive patterns of family interactions are the focus of treatment. Changing these patterns results in diminished antisocial behavior including alcohol abuse. Family therapy can work with a broad range of family and social network populations. Family therapy approaches have developed specific interventions for engaging and keeping reluctant, unmotivated adolescents and family members in treatment.
Alcohol drinking is an immensely complex human behavior, but it has been modeled in laboratory animals. Two similar strains of alcoholic rats, the alcohol-preferring (P) rats and the high-alcohol-drinking (HAD) rats, have been successfully used to study alcohol use disorders. Like patients with DSM-5-TR qualifying alcohol use disorders, these rats self-administer alcohol, show tolerance, lose control over alcohol, and spend a lot of time. They also have cravings and physical stigmata of withdrawal, providing psychopharmacologic researchers with excellent face validity with animal models. Models have helped us develop anti-withdrawal, anti-craving, and harm-reducing treatments.
Several medications are available to help treat alcohol use disorder [324,325]. Some are used for detoxification and others are used to prevent relapse. Research has shown that medications are most effective when used in conjunction with other therapies.
Disulfiram, commonly known as Antabuse, was the first drug to be made available for the treatment of alcohol use disorder. It was approved for treatment of alcohol use disorder by the FDA in 1951 and has been used safely and effectively for more than half a century. It works by blocking an enzyme, aldehyde dehydrogenase, that helps metabolize alcohol. Taking even one drink while on disulfiram causes the alcohol at the acetaldehyde stage to accumulate in the blood. This produces nausea, vomiting, sweating, and even difficulty breathing. More alcohol in the patient's system produces more severe reactions (e.g., respiratory depression, cardiovascular collapse, unconsciousness, convulsions, death) [325,326]. Patients must also be mindful of consuming even minute amounts of alcohol in foods, over-the-counter medications, mouthwash, and even topical lotions. Disulfiram can be effective for people who have completed alcohol withdrawal, are committed to staying sober, and are willing to take the medication under the supervision of a family member or treatment program [325]. Due to more modern and improved medication modalities, many clinicians prescribe disulfiram as a last resort intervention. Although widely used, it is less clearly supported by clinical trial evidence [327,328,329].
The recommended dose for disulfiram is 250 mg/day, which can be increased to 500 mg based upon whether a patient experiences the disulfiram-ethanol reaction [330]. Doses may need to be reduced in patients older than 60 years of age [325]. Labeling for disulfiram includes several precautions regarding drug-drug interactions; therefore, caution should be used when prescribing it to older adults at risk for polypharmacy [325]. Due to the physiologic changes that occur with use, use of disulfiram is not recommended in patients with diabetes, cardiovascular or cerebrovascular disease, or kidney or liver failure. It also is contraindicated in the presence of psychoses and pregnancy and in those with high levels of impulsivity and suicidality [325].
Naltrexone (ReVia) is an opioid antagonist that interferes with the rewarding or pleasurable effects of alcohol and reduces alcohol craving [331,332,333]. The exact mechanisms by which naltrexone induces the reduction in alcohol consumption observed in patients with alcohol use disorder is not entirely understood, but preclinical data suggest involvement of the endogenous opioid system [325]. Naltrexone has been shown to reduce alcohol relapses, decrease the likelihood that a slip becomes a relapse, and decrease the total amount of drinking [325]. The FDA approved the use of oral naltrexone in alcohol use disorder in December 1994 [325,333]. In 2006, the FDA approved an extended-release injectable formulation, which is indicated for use only in patients who can refrain from drinking for several days prior to beginning treatment [325]. In 2010, the FDA approved the injectable naltrexone for the prevention of relapse to opioid dependence following opioid detoxification [325]. Naltrexone, which has long been used to treat heroin addicts, was not known as a treatment that could reduce alcohol relapse until the 1980s. In 1980, researchers reported reductions in monkey ethanol self-administration when they were pretreated with naltrexone [334].
By 1992, researchers reported a six-week, double-blind placebo-controlled outpatient naltrexone trial with 70 individuals with alcohol use disorder. They found that the naltrexone-treated patients had a lower relapse rate, fewer drinking episodes, longer time to relapse, and reduced tendency for a slip to become a relapse [335]. These and other data suggested that endogenous opioids were important in alcohol reinforcement.
Also in 1992, researchers compared naltrexone with placebo and found that naltrexone-treated patients had lower rates of relapse to heavy drinking, consumed fewer drinks per drinking-day, and had lower dropout rates than placebo-treated patients with alcohol use disorder [336]. These results have since been supported by other studies [337]. Research suggests that naltrexone may be most effective for individuals with alcohol use disorder and a family history of alcohol use disorder [338]. However, one study found no significant effects for naltrexone in individuals with a family history of alcoholism on percentage of days abstinent, drinks per drinking day, and percentage of heavy drinking days [339].
Another study investigated pretreatment social network variables as potential moderators of naltrexone's treatment effects [340]. The study sample included 1,197 participants from the COMBINE study, the largest pharmacotherapy trial conducted for alcoholism in the United States. In treatment conditions involving combined CBT and medical management, the effects of naltrexone on heavy drinking were significantly greater for individuals with frequent drinkers in their social network and greater frequency of contact with those drinkers, indicating patterns of environmental exposure to alcohol [40,341]. In an attempt to replicate and extend the findings of the COMBINE study, researchers conducted a secondary analysis of a 12-week randomized clinical trial of daily or targeted naltrexone among problem drinkers, with a focus on those who received a daily dose of either naltrexone or placebo [342]. Four reward/relief phenotypes were identified: low reward/low relief; low reward/high relief; high reward/low relief; and high reward/high relief. When compared with the placebo group, individuals in the high reward/low relief phenotype who received daily naltrexone had significantly fewer drinks per drinking day and a lower proportion of heavy drinking days [342].
After a complete history, physical exam, and laboratory testing, most patients are started on 50 mg orally per day [234]. For most patients, this is the safe and effective dose of naltrexone. However, in a four-month study period, the COMBINE study demonstrated efficacy of naltrexone at a dose of 100 mg daily [343]. Some treatment providers give patients a naltrexone identification card or ask them to order a MedicAlert bracelet that clearly indicates that they are maintained on an opioid antagonist, so if they need an opiate drug or medication for pain relief, the dose of the pain medication can be adjusted higher. Meta-analyses have revealed that approximately 70% of previous clinical trials that measured reductions in "heavy or excessive drinking" demonstrated an advantage for prescribing naltrexone over placebo [344]. In another trial, naltrexone was determined to have the greatest impact on reducing daily drinking when craving for alcohol was highest [345]. The approved dose of the extended-release formulation is 380 mg IM once per month. Pretreatment with oral naltrexone is not required before induction onto extended-release injectable naltrexone [325].
The most common side effects of naltrexone are light-headedness, diarrhea, dizziness, and nausea. Pain or tenderness at the injection site is a side effect unique to the extended-release injectable formulation [325]. Most side effects tend to disappear quickly in most patients. Naltrexone is not recommended for patients with acute hepatitis or liver failure, for adolescents, or for pregnant or breastfeeding women [325,343]. Weight loss and increased interest in sex have been reported by some patients. In general, patients maintained on opioid antagonists should be treated with nonopioid cough, antidiarrheal, headache, and pain medications. The patient's family or physician should call the treating physician if questions arise about opioid blockade or analgesia. It is important to realize that naltrexone is not disulfiram; drinking while maintained on naltrexone does not produce side effects or symptoms.
Naltrexone works best when it is used in the context of a full spectrum of treatment services, possibly including traditional 12-step fellowship-based treatments. Studies show also that naltrexone is effective when coupled with CBT. Patients receiving medical management with naltrexone, CBT, or both fared better on drinking outcomes [343].
Acamprosate (Campral) is a synthetic compound that has a chemical structure similar to that of the naturally occurring amino acid neurotransmitters taurine and GABA [234]. Because chronic alcohol use is associated with decreased GABA and glutamate activity, a hyperexcitable glutamate system is one possible alcohol withdrawal mechanism. Glutamate systems may become unstable for 12 months after a person stops drinking. In a review of published, double-blind, placebo-controlled clinical trials evaluating the safety and efficacy of acamprosate in the treatment of alcohol use disorder, Mason reported that acamprosate appeared to improve treatment completion rate, abstinence rate and/or cumulative abstinence during treatment, and time to first drink, than placebo [346]. The effect on abstinence, combined with an excellent safety profile, lend support to the use of acamprosate across a broad range of patients with alcohol use disorder. A dose of 2,000 mg/day is associated with the greatest efficacy regardless of body weight [347]. It is important to note that medication in combination with therapies can improve outcomes.
In July 2004, after many years of safe use in Europe and around the world, the FDA approved the use of acamprosate for the maintenance of alcohol abstinence [333]. As in the case of naltrexone, acamprosate reduces the reinforcing (pleasurable) effects of alcohol to reduce craving. Oral dosing is two 333-mg delayed-release tablets three times daily [234,325]. Common side effects include diarrhea, anxiety, insomnia, nausea, dizziness, and weakness. Some research indicates that acamprosate may worsen depression and/or suicidal ideation; so, patients with a history of major depression should be monitored closely or prescribed a different medication [234]. Acamprosate is contraindicated in patients with severe renal impairment [234,325]. Due to risk of diminished renal function in patients 65 years of age and older, baseline and frequent renal function tests should be performed in this population. Dose reductions also may be necessary [325].
The effectiveness of acamprosate in promoting abstinence has not been demonstrated in individuals who have not completed detoxification or who have not achieved alcohol abstinence before beginning treatment [325]. An analysis of many studies of acamprosate showed a benefit in maintaining abstinence when coupled with CBT [343]. A systematic review found similar benefit [348]. Results of other research into the effectiveness of acamprosate have been mixed. One study showed no improvement in measures of psychological well-being or health status when compared to treatment with placebo. Another study demonstrated both safety and effectiveness of acamprosate for treating alcohol use disorder [349].
Baclofen is a GABA agonist that may prove to be a unique therapeutic alternative to reduce alcohol craving and consumption. In a small, 12-week trial, patients with alcohol use disorder were given 10 mg of baclofen three times daily paired with motivational enhancement therapy. Patients experienced a reduction in number of drinks, drinking days, anxiety, and craving [350]. In a study of patients with alcohol use disorder and liver cirrhosis, baclofen was also found to work favorably in maintenance of alcohol abstinence. Seventy-one percent of baclofen-treated patients maintained abstinence as compared with 29% of the placebo group [351]. A 2018 meta-analysis of 12 randomized controlled trials that compared the efficacy of baclofen to placebo found that baclofen was associated with higher rates of abstinence than placebo but that its effects were not superior to placebo in increasing the number of abstinent days or in decreasing heavy drinking, craving, depression, or anxiety [352]. A 2023 systematic review of 17 randomized controlled trials involving 1,818 participants found that baclofen reduces the risk of relapse to any drinking and increases the percentage of abstinent days, mainly among detoxified participants. The agent was not found to reduce the number of heavy drinking days or the number of drinks per drinking days [353].
Research has demonstrated that topiramate is efficacious in decreasing heavy drinking among individuals with alcohol use disorder [354]. In a controlled study, topiramate produced significant and meaningful improvement in a wide variety of drinking outcomes [355]. Topiramate may suppress the craving and rewarding effects of alcohol [356]. In a double-blind, controlled trial, 150 patients with alcohol use disorder were randomized to escalating doses of topiramate (25–300 mg/day) or placebo. Those on topiramate had a reduction in self-reported drinking (number of drinks and drinking days), alcohol craving, and plasma y-glutamyl transferase (an indicator of alcohol consumption) [357]. Side effects of topiramate include numbness in the extremities, fatigue, confusion, paresthesia, depression, change in taste, and weight loss. Use of topiramate for alcohol use disorder is off-label [234].
Carbamazepine has proven effective for treating acute alcohol withdrawal [358]. Its side effects include nausea, vomiting, drowsiness, dizziness, chest pain, headache, trouble urinating, numbness in extremities, liver damage, and allergic reaction [234]. In a 12-month, double-blind, placebo-controlled trial, 29 patients were assigned to carbamazepine three times daily (to reach an average blood level of 6 mg/liter) or placebo. Those treated with carbamazepine showed a delay in time to first drink and a decrease in number of drinks and drinking days [359].
Oxcarbazepine is a carbamazepine derivative, with fewer side effects and contraindications, used to prevent relapse in patients with alcohol use disorder by blocking alcohol withdrawal [358]. A group of 84 patients with alcohol use disorder following detoxification were randomized to 50 mg naltrexone, 1,500–1,800 mg oxcarbazepine, or 600–900 mg oxcarbazepine for 90 days. Approximately 58.6% of the high-dose oxcarbazepine patients remained alcohol-free, a significantly larger number as compared to the low-dose (42.8%) and naltrexone groups (40.7%) [360].
Ondansetron is a serotonin antagonist and antiemetic that may block the rewarding effects of alcohol, specifically in the early-onset alcoholic subgroup. Early-onset alcoholism differs from late-onset in its association with abnormal serotonin and antisocial behavior. In a double-blind, controlled trial of ondansetron as an adjunct to cognitive-behavioral therapy, ondansetron was shown to reduce self-reported drinking and increase abstinence as compared to placebo. These results were confirmed by measure of plasma carbohydrate deficient transferring, a biomarker of alcohol consumption [361]. One hypothesis suggests that ondansetron may reduce drinking in individuals with alcohol use disorder with the LL genotype [362].
Buspirone hydrochloride is a dopamine antagonist and partial agonist for serotonin, exhibiting anxiolytic properties. In a 12-week randomized, placebo-controlled trial among 61 patients with alcohol use disorder and anxiety, buspirone was associated with slower return to heavy alcohol consumption and fewer drinking days [363]. One study found buspirone to be effective in treatment of comorbid anxiety disorder and alcohol use disorder [364].
Clozapine is an atypical antipsychotic approved to treat schizophrenia and its resultant symptoms (e.g., hallucinations, suicidal behavior). In case studies, it has shown promise in the treatment of comorbid substance use. In a study of 151 individuals with schizophrenia with comorbid substance use, 36 were given clozapine [365]. Those who abused alcohol experienced a reduction in drinks and drinking days.
Other drugs under trial for use in the treatment of alcohol use disorder include varenicline and lithium. Varenicline does appear to help reduce drinking in some individuals with alcohol use disorder; however, concerns exist regarding reports of an association between the drug and an increased risk for suicidal thoughts and cardiovascular events [366]. Studies have demonstrated that varenicline helps reduce alcohol craving and consumption in patients with alcohol use disorder and in individuals with alcohol use disorder who also smoke [367,368,369]. None of the medications mentioned for alcohol use disorder are recommended for women who are pregnant or breastfeeding.
Benzodiazepines have been used for 30 years in the United States as the primary medical treatment for alcohol withdrawal syndrome. All benzodiazepines appear similarly effective in the treatment of alcohol withdrawal syndrome [370]. Although benzodiazepines are the drugs of choice, there are concerns about the side effects and, as stated, problems of abuse, especially for outpatient detoxification. Benzodiazepines are sedatives and cause deficiencies in psychomotor abilities that, when combined with alcohol, can cause accidents and affect the ability to think clearly. However, benzodiazepines are, and have been, effective in treating alcohol withdrawal symptoms and preventing most seizures. Other regimens for alcohol withdrawal syndrome include barbiturates, propofol, and ethanol [371,372,373].
A desirable alternative to benzodiazepines would be a nonsedative anticonvulsant that has less potential for abuse and dependence. Valproic acid has been used in Europe safely and successfully for many years for alcohol withdrawal syndrome, but is only approved by the FDA for the treatment of mania, seizures, and migraines. Valproic acid should be used as an adjunctive therapy, not as monotherapy [370]. According to clinical reports, valproic acid is an anticonvulsant with no potential for abuse and is better tolerated by patients. Valproic acid also has less cognitive impairment and causes fewer deficiencies of psychomotor abilities than benzodiazepines; however, benzodiazepines have allowed for safe detoxification for patients with alcohol use disorder since they were approved. While detoxification is not treatment, and detoxification problems have not been the most important problem area in successful treatment of the patient with alcohol use disorder, these are important findings.
Recognizing that relapse prevention and harm-reducing medications are safe and effective in alcohol use disorders, fewer than 10% of these patients are given medication-assisted treatment. In a 2018 meeting of the American Psychiatric Association, experts suggested [374]:
Naltrexone or acamprosate should be offered to those patients with moderate- to-severe alcohol use disorder that have a goal of reducing consumption or achieving abstinence, prefer pharmacotherapy, or have not responded to nonpharmacologic therapies, and have no contraindications.
Disulfiram should be offered to patients with severe alcohol use disorder that seek to achieve abstinence, prefer the therapy, or have not responded (or are intolerant) to naltrexone or acamprosate, and have no contraindications. Additionally, patients must understand the risks associated with consuming alcohol while on disulfiram.
Topiramate or gabapentin should be offered to patients with moderate-to-severe alcohol use disorder when they aim to reduce or achieve abstinence, prefer them to other medications, or have not responded to naltrexone or acamprosate and have no contraindications.
Benzodiazepine use is discouraged except in patients with alcohol use disorder who require treatment for acute alcohol withdrawal.
Even coerced or court-mandated treatment for alcohol use disorder can work. In a follow-up study (six months to one year) of Florida physicians with alcohol use disorder, 84% had positive outcomes, defined as positive counselor and physician assessment, negative alcohol testing, group attendance, and full return to work [375].
In a society where alcohol use is ubiquitous, it is important for healthcare professionals to recognize the signs and symptoms of alcohol abuse and intervene before a state of dependence is reached. It is critical to stress upon patients the negative health effects of excessive alcohol consumption, especially the synergistic effects of alcohol and tobacco use, beginning at an early age. Owing to the several benefits provided by low to moderate drinking as discussed in this course, certain patients can be advised to drink more regularly, provided alcohol use is not contraindicated due to drug or herb interactions.
| Al-Anon Family Groups |
| The mission of Al-Anon is to provide support for friends and families of problem drinkers. |
| https://al-anon.org |
| Alcoholics Anonymous |
| https://www.aa.org |
American Society of Addiction Medicine (ASAM)
The nation's medical specialty society dedicated to educating physicians and improving the treatment of individuals suffering from alcoholism and other addictions. The mission of the ASAM is to:
Increase access to and improve the quality of addiction treatment
Educate physicians, medical and osteopathic students, other healthcare providers, and the public
Promote research and prevention
Promote the appropriate role of the physician in the care of patients with addiction
Establish addiction medicine as a specialty recognized by the American Board of Medical Specialties
| https://www.asam.org |
| 301-656-3920 |
| MedicAlert Foundation |
| https://www.medicalert.org |
1. Camargo CA Jr, Stampfer MJ, Glynn RJ, et al. Prospective study of moderate alcohol consumption and risk of peripheral arterial disease in US male physicians. Circulation. 1997;95(3):577-580.
2. Ronksley PE, Brien SE, Turner BJ, Mukamal KJ, Ghali WA. Association of alcohol consumption with selected cardiovascular disease outcomes: a systematic review and meta-analysis. BMJ. 2011;342:d671.
3. Grant KA. Emerging neurochemical concepts in the actions of ethanol at ligand-gated ion channels. Behav Pharmacol. 1994;5(4, 5):383-404.
4. Koob GF,Rassnick S, Heinrichs S, Weiss F. Alcohol, the reward system and dependence. In: Jansson B, Jörnvall H, Rydberg U, Terenius L, Vallee BL (eds). Toward a Molecular Basis of Alcohol Use and Abuse. Basel: Birkauser; 1994:103-114.
5. Tizabi Y, Copeland RL Jr, Louis VA, Taylor RE. Effects of combined systemic alcohol and central nicotine administration into ventral tegmental area on dopamine release in the nucleus accumbens. Alcohol Clin Exp Res. 2002;26(3):394-399.
6. Tizabi Y, Bai L, Copeland RL Jr, Taylor RE. Combined effects of systemic alcohol and nicotine on dopamine release in the nucleusaccumbens shell. Alcohol Alcohol. 2007;42(5):413-416.
7. Tolu S, Marti F, Morel C, et al. Nicotine enhances alcohol intake and dopaminergic responses throughβ2* andβ4* nicotinic acetylcholine receptors. Sci Rep. 2017;7:45116.
8. Nies MA, McEwen M. Community/Public Health Nursing: Promoting the Health of Populations. 5th ed. St. Louis, MO: Elsevier Saunders; 2010.
9. U.S. Department of Health and Human Services. 10th Special Report to the U.S. Congress on Alcohol and Health. Available at https://webarchive.library.unt.edu/eot2008/20090113011041/http://pubs.niaaa.nih.gov/publications/10report/intro.pdf. Last accessed April24, 2024.
10. Nelson JP. Economic and demographic factors in U.S. alcohol demand: a growth-accounting analysis. Empirical Econ. 1997;22(1):83-102.
11. Schuckit MA. Drug and Alcohol Abuse: A Clinical Guide to Diagnosis and Treatment. 6th ed. New York, NY: Springer; 2010.
12. Substance Abuse and Mental Health Services Administration. 2022 National Survey on Drug Use and Health Releases: Detailed Tables. Available at https://www.samhsa.gov/data/release/2022-national-survey-drug-use-and-health-nsduh-releases#detailed-tables. Last accessed April 24, 2024.
13. White AM, Castle JP, Powell PA, et al. Alcohol-related deaths during the COVID-19 pandemic. JAMA. 2022;327(17):1704-1706.
14. Substance Abuse and Mental Health Services Administration. Results from the 2013 National Survey on Drug Use and Health: Summary of National Findings, NSDUH Series H-48, HHS Publication No. (SMA) 14-4863. Rockville, MD: Substance Abuse and Mental Health Services Administration; 2014.
15. Knight JR, Wechsler H, Kuo M, Seibring M, Weitzman ER, Schuckit MA. Alcohol abuse and dependence among U.S. college students. J Stud Alcohol. 2002;63(3):263-270.
16. Centers for Disease Control and Prevention. Excessive Drinking is Draining the U.S. Economy. Available at https://www.cdc.gov/alcohol/features/excessive-drinking.html. Last accessed April 24, 2024.
17. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. Washington, DC: American Psychiatric Association; 2013.
18. Dufour MC. What is moderate drinking? Defining "drinks" and drinking levels. Alcohol Res Health. 1999;23(1):5-14.
19. U.S. Department of Health and Human Services. 2020-2025 Dietary Guidelines for Americans and Online Materials. Available at https://www.dietaryguidelines.gov/resources/2020-2025-dietary-guidelines-online-materials. Last accessed April 24, 2024.
20. Trevisan LA, Boutros N, Petrakis IL, Krystal JH. Compilations of alcohol withdrawal: pathophysiological insights. Alcohol Health Res World. 1998;22(1):61-66.
21. Bertrand J, Floyd RL, Weber MK, et al. Fetal Alcohol Syndrome: Guidelines for Referral and Diagnosis. Atlanta, GA: Centers for Disease Control and Prevention; 2004.
22. MukamalKJ, Rimm EB. Alcohol's effects on the risk for coronary heart disease. Alcohol Res Health. 2001;25(4):255-261.
23. Beulens JWJ, Rimm EB, Ascherio A, Spiegelman D, Hendriks HFJ, Mukamal KJ. Alcohol consumption and risk of coronary heart disease among men with hypertension. Ann Intern Med. 2007;146(1):10-19.
24. Rimm EB. Alcohol consumption and good health. In: Gold MS (ed). Addiction and Psychiatry. Gainesville, FL: University of Florida College of Medicine; 2007.
25. Kitamura A, Iso H, Sankai T, et al. Alcohol intake and premature coronary heart disease in urban Japanese men. Am J Epidemiol. 1998;147(1):59-65.
26. Klatsky AL, Armstrong MA, Friedman GD. Red wine, white wine, liquor, beer, and risk for coronary artery disease hospitalization. Am J Cardiol. 1997;80(4):416-420.
27. Rehm JT, Bondy SJ, Sempos CT, Vuong CV. Alcohol consumption and coronary heart disease morbidity and mortality. Am J Epidemiol. 1997;146(6):495-501.
28. Renaud SC, Guéguen R, Siest G, Salamon R. Wine, beer, and mortality in middle-aged men from eastern France. Arch Intern Med. 1999;159(16):1865-1870.
29. MukamalKA, Conigrave KM, Mittleman MA, et al. Roles of drinking pattern and type of alcohol consumed in coronary heart disease in men. N Engl J Med. 2003;348(2):109-118.
30. Hvidtfeldt UA, Tolstrup JS, Jakobsen MU, et al. Alcohol intake and risk of coronary heart disease in younger, middle-aged, and older adults. Circulation. 2010;121(14):1589-1597.
31. World Health Organization. Diet, Nutrition and the Prevention of Chronic Diseases: Report of the Joint WHO/FAO Expert Consultation. WHO Technical Report Series, No. 916. Available at https://www.who.int/publications/i/item/924120916X. Last accessed April 24, 2024.
32. Hines LM, Stampfer MJ, Ma J, et al. Genetic variation in alcohol dehydrogenase and the beneficial effect of moderate alcohol consumption on myocardial infarction. N Engl J Med. 2001;344(8):549-555.
33. Zahler R, Piselli C. Smoking, alcohol and drugs. In: Zaret BL, Moser M, Cohen LS (eds). Yale University School of Medicine Heart Book. New York, NY: Hearst Books; 1992.
34. Renaud SC, Beswick AD, Fehily AM, Sharp DS, Elwood PC. Alcohol and platelet aggregation: the Caerphilly Prospective Heart Disease Study. Am J Clin Nutr. 1992;55(5):1012-1017.
35. Lippi G, Franchini M, Favaloro EJ, Targher G. Moderate red wine consumption and cardiovascular disease risk: beyond the "French paradox." Semin Thromb Hemost. 2010;36(1):59-70.
36. Reding KW, Daling JR, Doody DR, O'Brien CA, Porter PL, Malone KE. Effect of prediagnostic alcohol consumption on survival after breast cancer in young women. Cancer Epidemiol Biomarkers Prev. 2008;17(8):1988-1996.
37. Franceschi S, Dal Maso L, Zucchetto A, et al. Prospective analysis of case-control studies on environmental factors and health (PACE) study group: alcohol consumption and survival after breast cancer. Cancer Epidemiol Biomarkers Prev. 2009;18(3):1011-1012.
38. Li CI, Chlebowski RT, Freiberg M, et al. Alcohol consumption and risk of postmenopausal breast cancer by subtype: the women's health initiative observational study. J Natl Cancer Inst. 2010;102(18):1422-1431.
39. Stătescu C, Clement A, Serban IL, Sascau R. Consensus and controvery in the debate over the biphasic impact of alcohol consumption on the cardiovascular system. Nutrients. 2021;13(4):1076.
40. Puddey IB, Mori TA, Barden AE, Beilin LJ. Alcohol and hypertension – new insights and lingering controversies. Curr Hypertens Rep. 2019;21(10):79.
41. Tanasescu M, Hu FB, Willett WC, Stampfer MJ, Rimm EB. Alcohol consumption and risk of coronary heart disease among men with type 2 diabetes mellitus. J Am Coll Cardiol. 2001;38(7):1836-1842.
42. Ahmed AT, Karter AJ, Liu J. Alcohol consumption is inversely associated with adherence to diabetes self-care behaviours. Diabet Med. 2006;23(7):795-802.
43. Ahmed AT, Karter AJ, Warton EM, Doan JU, Weisner CM. The relationship between alcohol consumption and glycemic control among patients with diabetes: the Kaiser Permanente Northern California Diabetes Registry. J Gen Intern Med. 2008;23(3):275-282.
45. Kendler KS, Prescott CA, Neale MC, Pedersen NL. Temperance board registration for alcohol abuse in a national sample of Swedish male twins, born 1902 to 1949. Arch Gen Psychiatry. 1997;54(2):178-184.
46. Schuckit MA. Vulnerability factors for alcoholism. In: Davis KL, Charney D, Coyle JT, Nemeroff C (eds). Neuropsychopharmacology: The Fifth Generation of Progress. An Unofficial Publication of the College of Neuropharmacology. Philadelphia, PA: Lippincott Williams & Wilkins; 2002.
47. Prescott CA. Sex differences in the genetic risk for alcoholism. Alcohol Res Health. 2002;26(4):264-273.
48. Bowers BJ, Owen EH, Collins AC, Abeliovich A, Tonegawa S, Wehner JM. Decreased ethanol sensitivity and tolerance development in gamma-protein kinase C null mutant mice is dependent on genetic background. Alcohol Clin Exp Res. 1999;23(3):387-397.
49. Engel SR, Allan AM. 5-HT3 receptor over-expression enhances ethanol sensitivity in mice. Psychopharm. 1999;144(4):411-415.
50. Hwang BH, Suzuki R, Lumeng L, Li TK, McBride WJ. Innate differences of neuropeptide Y (NPY) in hypothalamic nuclei and central nucleus of the amygdala between selectively bred rats with high and low alcohol preference. Alcohol Clin Exp Res. 1999;23(6):1023-1030.
51. Risinger FO, Doan AM, Vickrey AC. Oral operant ethanol self-administration in 5-HT1b knockout mice. Behav Brain Res. 1999;102(1-2):211-215.
52. Roberts AJ, McDonald JS, Heyser CJ, et al. mu-Opioid receptor knockout mice do not self-administer alcohol. J Pharm Exp Ther. 2000;293(3):1002-1008.
53. Thiele TE, Marsh DJ, Ste. Marie L, Bernstein IL, Palmiter RD. Ethanol consumption and resistance are inversely related to neuropeptide Y levels. Nature. 1998;396(6709):366-369.
54. Yagi T, Yasuda M, Niki H. Ethanol sensitivity and molecular function of Fyn tyrosine kinase. In: Hoek JB, Gordon AS, Mochly-Rosen D, Zakhari S (eds). Ethanol and Intracellular Signaling: From Molecules to Behavior. Bethesda, MD: NIAAA; 2000: 195-202.
55. Schuckit MA. Biological, psychological and environmental predictors of the alcoholism risk: a longitudinal study. J Stud Alcohol. 1998;59(5):485-494.
56. Krystal JH, Tabakoff B. Ethanol abuse, dependence, and withdrawal: neurobiology and clinical implications. In: Davis KL, Charney D, Coyle JT, Nemeroff C (eds). Neuropsychopharmacology: The Fifth Generation of Progress. An Unofficial Publication of the College of Neuropharmacology. New York, NY: Lippincott Williams & Wilkins; 2002.
57. Schuckit MA, Smith TL. The relationships of a family history of alcohol dependence, a low level of response to alcohol and six domains of life functioning to the development of alcohol use disorders. J Stud Alcohol. 2000;61(6):827-835.
58. Paulus MP, Schuckit MA, Tapert SF, et al. High versus low level of response to alcohol: evidence of differential reactivity to emotional stimuli. Biol Psychiatry. 2012;72(10):848-855.
59. Bates ME, Mun EY, Buckman JF, et al. Getting to the heart of low sensitivity to alcohol: context moderates low cardiovascular response to alcohol in persons with a family history of alcohol use disorder. Alcohol Clin Exp Res. 2020;44(3):589-599.
60. Schuckit MA, Smith TL, Kalmijn J, Tsuang J, Hesselbrock V, Bucholz K. Response to alcohol in daughters of alcoholics: a pilot study and a comparison with sons of alcoholics. Alcohol Alcohol. 2000;35(3):242-248.
61. Hill SY, DeBellis MD, Keshavan MS, et al. Right amygdala volume in adolescent and young adult offspring from families at high risk for developing alcoholism. Biol Psychiatry. 2001;49(11):894-905.
62. O'Brien JW, Hill SY. Neural predictors of substance use disorders in young adulthood. Psychiatry Res. 2017;268:22-26.
63. National Institute on Alcohol Abuse and Alcoholism. Alcohol Facts and Statistics. Available at https://www.niaaa.nih.gov/publications/brochures-and-fact-sheets/alcohol-facts-and-statistics. Last accessed April 24, 2024.
64. National Institute on Alcohol Abuse and Alcoholism. Alcohol-Related Deaths Increasing in the United States. Available at https://www.niaaa.nih.gov/news-events/news-releases/alcohol-related-deaths-increasing-united-states. Last accessed April 24, 2024.
65. Grossman ER, Benjamin-Neelon SE, Sonnenschein S. Alcohol consumption during the COVID-19 pandemic: a cross-sectional survey of US adults. Int J Environ Res Public Health. 2020;17(24):9189.
66. Meyers JL, McCutcheon VV, Horne-Osipenko KA, et al. COVID-19 pendemic stressors are associated with reported increases in frequency of drunkenness among individuals with a history of alcohol use disorder. Transl Psychiatry. 2023;13(1):311.
67. Cadoret RJ, Yates WR, Troughton E, Woodworth G, Stewart MA. Adoption study demonstrating two genetic pathways to drug abuse. Arch Gen Psychiatry. 1995;52(1):42-52.
68. Kendler KS, Ohlsson H, Sundquist K, Sundquist J. The rearing environment and risk for drug abuse: a Swedish national high-risk adopted and not-adopted co-sibling control study. Psychol Med. 2016;46(7):1359-1366.
69. Grant BF, Stinson FS, Harford TC. Age at onset of alcohol use and DSM-IV alcohol abuse and dependence: a 12-year follow-up. J Subst Abuse. 2001;13(4):493-504.
70. Goodwin DW,Schulsinger F, Moller N, Hermansen L, Winokur G, Guze SB. Drinking problems in adopted and nonadopted sons of alcoholics. Arch Gen Psychiatry. 1974;31(2):164-169.
71. Conrod PJ, Pihl RO, Ditto B. Autonomic reactivity and alcohol-induced dampening in men at risk for alcoholism and men at risk for hypertension. Alcohol Clin Exp Res. 1995;19(2):482-489.
72. Söderpalm AH, de Wit H. Effects of stress and alcohol on subjective state in humans. Alcohol Clin Exp Res. 2002;26(6):818-826.
73. Childs E, O'Connor S, de Wit H. Bidirectional interactions between acute psychosocial stress and acute intravenous alcohol in healthy men. Alcohol Clin Exp Res. 2011;35(10):1794-1803.
74. 74. Volavka J, Czobor P, Goodwin DW, et al. The electroencephalogram after alcohol administration in high-risk men and the development of alcohol use disorders 10 years later. Arch Gen Psychiatry. 1996;53(3):258-263.
75. Holdstock L, de Wit H. Individual differences in the biphasic effects of ethanol. Alcohol Clin Exp Res. 1998;22(9):1903-1911.
76. Holdstock L, de Wit H. Individual differences in responses to ethanol and d-amphetamine: a within-subject study. Alcohol Clin Exp Res. 2001;25(4):540-548.
77. Morean ME, Corbin WR. Subjective response to alcohol: a critical review of the literature. Alcohol Clin Exp Res. 2010;34(3):385-395.
78. Quinn PD, Fromme K. Subjective response to alcohol challenge: a quantitative review. Alcohol Clin Exp Res. 2011;35(10):1759-1770.
79. Ray LA, Bujarski S, Roche DJ. Subjective response to alcohol as a research domain criterion. Alcohol Clin Exp Res. 2016;40(1):6-17.
80. Rangaswamy M, Porjesz B. Understanding alcohol use disorders with neuroelectrophysiology. Handb Clin Neurol. 2014;125:383-414.
81. Stenberg G, Sano M, Rosen I, et al. EEG topography of acute ethanol effects in resting and activated normals. J Stud Alcohol. 1994;55:645-656.
82. 82. Ehlers CL, Phillips E, Wall TL, et al. EEG alpha and level of response to alcohol in Hispanic- and non-Hispanic-American young adults with a family history of alcoholism. J Stud Alcohol. 2004;65:301-308.
83. Kahkonen S, Kesaniemi M, Nikouline VV, et al. Ethanol modulates cortical activity: direct evidence with combined TMS and EEG. Neuroimage. 2001;14:322-332.
84. Sullivan MA, Rudnik-Levin F. Attention deficit/hyperactivity disorder and substance abuse: diagnostic and therapeutic considerations. Ann NY Acad Sci. 2001;931:251-270.
85. Schuck AM, Widom CS. Childhood victimization and alcohol symptoms in females: causal inferences and hypothesized mediators. Child Abuse Negl. 2001;25(8):1069-1092.
86. Resnick MD, Bearman PS, Blum RW, et al. Protecting adolescents from harm: findings from the National Longitudinal Study on Adolescent Health. JAMA. 1997;278(10):823-832.
87. Donovan JE. Adolescent alcohol initiation: a review of psychosocial risk factors. J Adolesc Health. 2004;35(6):e7-e18.
88. Nash SG, McQueen A, Bray JH. Pathways to adolescent alcohol use: Family environment, peer influence, and parental expectations. J Adolesc Health. 2005;37(1):19-28.
89. Galambos NL, Barker ET, Almeida DM. Parents do matter: Trajectories of change in externalizing and internalizing problems in early adolescence. Child Dev. 2003;74(2):578-594.
90. Lansford JE, Criss MM, Pettit GS, Dodge KA, Bates JE. Friendship quality, peer group affiliation, and peer antisocial behavior as moderators of the link between negative parenting and adolescent externalizing behavior. J Res Adolesc. 2003;13(2):161-184.
91. Mrug S, Windle M. Moderators of negative peer influence on early adolescent externalizing behaviors: The roles of individual behavior, parenting, and school connectedness. J Early Adolesc. 2009;29:518-540.
92. Schuckit MA, Smith TL, Landi NA. The 5-year clinical course of high-functioning men with DSM-IV alcohol abuse or dependence. Am J Psychiatry. 2000;157(12):2028-2035.
93. Schuckit MA, Smith TL, Danko GP, et al. Five-year clinical course associated with DSM-IV alcohol abuse or dependence in a large group of men and women. Am J Psychiatry. 2001;158(7):1084-1090.
94. National Institute on Alcohol Abuse and Alcoholism. Alcohol Facts and Statistics. Available at https://www.niaaa.nih.gov/alcohols-effects-health/alcohol-topics/alcohol-facts-and-statistics. Last accessed April 24, 2024.
95. Thompson W, Lande RG, Kalapatapu RJ. Alcoholism. Available at https://emedicine.medscape.com/article/285913-overview. Last accessed April 24, 2024.
96. National Institute on Alcohol Abuse and Alcoholism. Alcohol's Effects on the Body. Available at https://www.niaaa.nih.gov/alcohols-effects-health/alcohols-effects-body. Last accessed April 24, 2024.
97. Heuman DM, Mihas AA, Hung PD. Alcoholic Hepatitis. Available at https://emedicine.medscape.com/article/170539-overview. Last accessed April 24, 2024.
98. Gavaler JS, Arria AM. Increased susceptibility of women to alcoholic liver disease: artifactual or real? In: Hall P (ed). Alcoholic Liver Disease: Pathology and Pathogenesis. 2nd ed. London: Edward Arnold; 1995:123-133.
99. Hall P. Factors influencing individual susceptibility to alcoholic liver disease. In: Hall P (ed). Alcoholic Liver Disease: Pathology and Pathogenesis. 2nd ed. London: Edward Arnold; 1995: 299-316.
101. Luu L. Liver Transplants. Available at https://emedicine.medscape.com/article/776313-overview. Last accessed April 24, 2024.
102. Dhar S, Omran L, Bacon BR, Solomon H, Di Bisceglie AM. Liver transplantation in patients with chronic hepatitis C and alcoholism. Dig Dis Sci. 1999;44(10):2003-2007.
103. Gómez-Moreno G, Guardia J, Cutando A. Interaction of paracetamol in chronic alcoholic patients: importance for odontologists. Med Oral Patol Oral Cir Bucal. 2008;13(4):E235-E238.
104. Girre C, Hispard E, Palombo S, N'Guyen C, Dally S. Increased metabolism of acetaminophen in chronically alcoholic patients. Alcohol Clin Exp Res. 1993;17(1):170-173.
105. Seeff LB, Cuccherini BA, Zimmerman HJ, Adler E, Benjamin SB. Acetaminophen hepatotoxicity in alcoholics: a therapeutic misadventure. Ann Intern Med. 1986;104(3):399-404.
107. 107. Kuffner EK, Green JL, Bogdan GM, et al. The effect of acetaminophen (four grams a day for three consecutive days) on hepatic tests in alcoholic patients: a multicenter randomized study. BMC Med. 2007;30:13.
108. U.S. Food and Drug Administration. Acetaminophen Prescription Combination Drug Products with More than 325 mg: FDA Statement: Recommendation to Discontinue Prescribing and Dispensing. Available at https://wayback.archive-it.org/7993/20170406123735/https://www.fda.gov/Safety/MedWatch/SafetyInformation/SafetyAlertsforHumanMedicalProducts/ucm381650.htm. Last accessed April 24, 2024.
110. Lee DH, Ha MH, Kim JR, Gross M, Jacobs DR. Gamma-glutamyltransferase, alcohol, and blood pressure: a four year follow-up study. Ann Epidemiol. 2002;12(2):90-96.
111. Rosenthal L. Holiday Heart Syndrome. Available at https://emedicine.medscape.com/article/155050-overview. Last accessed April 24, 2024.
112. Rafie IM, Colucci WS. Alcohol-Induced Cardiomyopathy. Available at https://www.uptodate.com/contents/alcohol-induced-cardiomyopathy. Last accessed April 24, 2024.
113. Menz V, Grimm W, Hoffmann J, Maisch B. Alcohol and rhythm disturbance: the holiday heart syndrome. Herz. 1996;21(4):227-231.
114. Ettinger PO, Wu CF, De La Cruz C, et al. Arrhythmias and the "holiday heart:" alcohol-associated cardiac rhythm disorders. Am Heart J. 1978;95(5):555-562.
115. 115. Hansson A, Madsen-Härdig B, Olsson SB. Arrhythmia-provoking factors and symptoms at the onset of paroxysmal atrial fibrillation: a study based on interviews with 100 patients seeking hospital assistance. BMC Cardiovasc Disord. 2004;3:4-13.
116. Noessler N, Schweintzger S, Kurath-Koller S. Holiday heart syndrome: an upcoming tachyarrhythmia in today's youth? Cardiol Young. 2021;31(6):1054-1056.
117. Mangoni AA, Jackson SH. Homocysteine and cardiovascular disease: current evidence and future prospects. Am J Med. 2002;112(7):556-565.
118. 118. Chambers JC, Ueland PM, Obeid OA. Improved vascular endothelial function after oral B vitamins: an effect mediated through reduced concentrations of free plasma homocysteine. Circulation. 2000;102(20):2479-2483.
119. Debreceni B, Debreceni L. The role of homocysteine-lowering B-vitamins in the primary prevention of cardiovascular disease. Cardiovasc Ther. 2014;32(3):130-138.
120. Martí-Carvajal AJ, Solà I, Lathyris D, Dayer M. Homocysteine-lowering interventions for preventing cardiovascular events. Cochrane Database Syst Rev. 2017;(8):CD006612.
121. Weber A, Tannapfel A, Wittekind C, Bootz F. Carcinogen-induced site-specific mutagenesis and genetic susceptibility in squamous cell carcinoma of the head and neck. Onkologie. 2002;25(1):8-13.
122. Stinson FS, DeBakey SF. Alcohol-related mortality in the United States, 1979–1988. Br J Addict. 1992;87(5):777-783.
123. Peng Q, Chen H, Huo JR. Alcohol consumption and corresponding factors: a novel perspective on the risk factors of esophageal cancer. Oncol Lett. 2016;11(5):3231-3239.
124. International Agency for Research on Cancer. Alcohol consumption and ethyl carbamate In: IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. Lyon: IARC Press, International Agency for Research on Cancer; 2010.
125. Castellsagué X, Muñoz N, De Stefani E, et al. Independent and joint effects of tobacco smoking and alcohol drinking on the risk of esophageal cancer in men and women. Int J Cancer. 1999;82(5):654-657.
126. Zambon P, Talamini R, La Vecchia C, et al. Smoking, type of alcoholic beverage and squamous-cell oesophageal cancer in northern Italy. Int J Cancer. 2000;86(1):144-149.
127. Drummond J. Alcohol and Hormones. Available at https://integrativewomenshealthinstitute.com/alcohol-and-hormones/. Last accessed April 24, 2024.
128. Stermer E. Alcohol consumption and the gastrointestinal tract. Isr Med Assoc. 2002;4(3):200-202.
129. Hagedorn JC, Encarnacion B, Brat GA, Morton JM. Does gastric bypass alter alcohol metabolism? Surg Obes Relat Dis. 2007;3(5):543-548.
130. Bisceglie AM, Segal I. Cirrhosis and chronic pancreatitis in alcoholics. J Clin Gastroenterol. 1984;6(3):199-200.
131. Etemad B, Whitcomb DC. Chronic pancreatitis: diagnosis, classification, and new genetic developments. Gastroenterology. 2001;120(3):682-707.
132. Ferrone C, Rattner DW. Chronic pancreatitis. In: Rakel RE, Bope ET (eds). Conn's Current Therapy, 2014. Philadelphia, PA: W.B. Saunders; 2002: 515-516.
133. Gumaste VV. Alcoholic pancreatitis: unraveling the mystery. Gastroenterology. 1995;108(1):297-299.
134. Witt H, Apte MV, Keim V, Wilson JS. Chronic pancreatitis: challenges and advances in pathogenesis, genetics, diagnosis, and therapy. Gastroenterology. 2007;132:1557-1573.
136. National Institute on Alcohol Abuse and Alcoholism. Alcohol Metabolism. Available at https://www.niaaa.nih.gov/publications/alcohol-metabolism. Last accessed April 24, 2024.
137. Moses S. Lab Markers of Malnutrition. Available at https://fpnotebook.com/Pharm/Lab/LbMrkrsOfMlntrtn.htm. Last accessed April 24, 2024.
138. Breslow RA, Smothers BA. Drinking patterns and body mass index in never smokers: National Health Interview Survey, 1997–2001. Am J Epidemiol. 2005;161(4):368-376.
139. Butler L, Popkin BM, Poti JM. Associations of alcoholic beverage consumption with dietary intake, waist circumference, and body mass index in U.S. adults: National Health and Nutrition Examination Survey 2003–2012. J Acad Nutr Diet. 2018;118(3):409-420.
140. Saules KK, Wiedemann A, Ivezaj V, Hopper JA, Foster-Hartsfield J, Schwarz D. Bariatric surgery history among substance abuse treatment patients: prevalence and associated features. Surg Obes Relat Dis. 2010;6(6):615-621.
141. Kleiner KD, Gold MS, Frost-Pineda K, Lenz-Brunsman B, Perri MG, Jacobs WS. Body mass index and alcohol use. J Addict Dis. 2004;23(3):105-118.
142. Van den Berg H, van der Gaag M, Hendriks H. Influence of lifestyle on vitamin bioavailability. Int J Vitam Nutr Res. 2002;72(1):53-59.
143. Thomson AD. Mechanisms of vitamin deficiency in chronic alcohol misusers and the development of the Wernicke-Korsakoff syndrome. Alcohol Alcohol. 2000;35(1):2-7.
144. Zhang P, Bagby GJ, Happel KI, Summer WR, Nelson S. Pulmonary host defenses and alcohol. Front Biosci. 2002;7:d1314-d1330.
145. Kaphalia L, Calhoun WJ. Alcoholic lung injury: metabolic, biochemical and immunological aspects. Toxicol Lett. 2013;222(2):171-179.
146. Sarmiento X, Guardiola JJ, Soler M. Alcohol and acute respiratory distress syndrome: casuality or causality? Med Clin (Barc). 2013;140(12):546-553.
147. Joshi PC, Guidot DM. The alcoholic lung: epidemiology, pathophysiology, and potential therapies. Am J Physiol Lung Cell Mol Physiol. 2007;292(4):L813-L823.
148. Liang Y, Yeligar SM, Brown LA. Chronic-alcohol-abuse-induced oxidative stress in the development of acute respiratory distress syndrome. Scientific World J. 2012;740308.
149. Liang Y, Harris FL, Brown LA. Alcohol induced mitochondrial oxidative stress and alveolar macrophage dysfunction. Biomed Res Int. 2014;371593.
152. National Institutes for Health. Alcohol Abuse, HIV, and Infectious Disease. Available at https://grants.nih.gov/grants/guide/pa-files/PA-95-067.html. Last accessed April 24, 2024.
153. Shi X, DeLucia AL, Bao J, Zhang P. Alcohol abuse and disorder of granulopoiesis. Pharmacol Ther. 2019;198:206-219.
154. Astry CL, Warr GA, Jakab GJ. Impairment of poly-morphonuclear leukocyte immigration as a mechanism of alcohol-induced suppression of pulmonary antibacterial defenses. Am Rev Respir Dis. 1983;128(1):113-117.
155. 155. Davis CC, Mellencamp MA, Preheim LC. A model of pneumococcal pneumonia in chronically intoxicated rats. J Infect Dis. 1991;163(4):799-805.
156. Melvan JN, Siggins RW, Stanford WL, et al. Alcohol impairs the myeloid proliferative response to bacteremia in mice by inhibiting the stem cell antigen-1/ERK pathway. J Immunol. 2012;188(4):1961-1969.
158. Roehrs T, Roth T. Sleep, sleepiness, sleep disorders and alcohol use and abuse. Sleep Med Rev. 2001;5(4):287-297.
159. Thakkar MM, Sharma R, Sahota P. Alcohol disrupt sleep homeostasis. Alcohol. 2015;49(4):299-310.
160. 160. Landolt HP, Roth C, Dijk D, Borbely A. Late-afternoon ethanol intake affects nocturnal sleep and the sleep EEG in middle-aged men. J Clin Psychopharmacol. 1996;16(6):428-436.
161. Yang S, Guo X, Liu W, Li Y, Liu Y. Alcohol as an independent risk factor for obstructive sleep apnea. Ir J Med Sci. 2022;191(3):1325-1330.
162. Simou E, Britton J, Leonardi-Bee J. Alcohol and the risk of sleep apnoea: a systematic review and meta-analysis. Sleep Med. 2018;42:38-46.
163. Kolla BP, Foroughi M, Saeidifard F, Chakravorty S, Wang Z, Mansukhani MP. The impact of alcohol on breathing parameters during sleep: a systematic review and meta-analysis. Sleep Med Rev. 2018;42:59-67.
164. Taveira KVM, Kuntze MM, Berretta F, et al. Association between obstructive sleep apnea and alcohol, caffeine and tobacco: a meta-analysis. J Oral Rehabil. 2018;45(11):890-902.
165. Bassetti C, Aldrich MS. Alcohol consumption and sleep apnea in patients with TIA and ischemic stroke. Sleep Res. 1996;25:400.
166. Yaggi HK, Concato J, Kernan WN, Lichtman JH, Brass LM, Mohsenin V. Obstructive sleep apnea as a risk factor for stroke and death. N Engl J Med. 2005;353(19):2034-2041.
167. Rich J, Raviv A, Raviv N, Brietzke SE. All-cause mortality and obstructive sleep apnea severity revisited. Otolaryngol Head Neck Surg. 2012;147(3):583-587.
168. Xiong GL. Wernicke-Korsakoff Syndrome. Available at https://emedicine.medscape.com/article/288379-overview. Last accessed April 24, 2024.
169. Schuckit MA, Mazzanti C, Smith TL, et al. Selective genotyping for the role of 5-HT2A, 5-HT2C, and GABA alpha 6 receptors and the serotonin transporter in the level of response to alcohol: a pilot study. Biol Psychiatry. 1999;45(5):647-651.
170. Cope LM, Munier EC, Trucco EM, et al. Effects of the serotonin transporter gene, sensitivity of response to alcohol, and parental monitoring on risk for problem alcohol use. Alcohol. 2017;59:7-16.
171. Hommer D,Momenan R, Rawlings R, et al. Decreased corpus callosum size among alcoholic women. Arch Neurol. 1996;53(4):359-363.
172. Ruiz SM, Oscar-Berman M, Sawyer KS, Valmas MM, Urban T, Harris GJ. Drinking history associations with regional white matter volumes in alcoholic men and women. Alcohol Clin Exp Res. 2013;37(1):110-122.
173. Kisner MA, Sussman L, Manuweera T, et al. Evaluating effects of sex and age on white matter microstructural alterations in alcohol use disorder: a diffusion tensor imagins study. Alcohol Clin Exp Res. 2021;45(9):1790-1803.
174. Albayrak O, Krug S,Scherbaum N. Sex-specific aspects of addiction. MMW Fortschr Med. 2007;149(24):29-32.
175. Frezza M, di Padova C, Pozzato G, Terpin M, Baraona E, Lieber CS. High blood alcohol levels in women: the role of decreased gastric alcohol dehydrogenase activity and first-pass metabolism. N Engl J Med. 1990;322(2):95-99.
176. Glazkov VA, Sviderskaia NE. Gender differences in chronic alcoholism: EEG analysis. Zh Vyssh Nerv Deiat Im I P Pavlova. 2006;56(5):597-602.
177. Walter H, Dvorak A, Gutierrez K, Zitterl W, Lesch OM. Gender differences: does alcohol affect females more than males? Neuropsychopharmacology Hung. 2005;7(2):78-82.
178. Moos RH, Moos BS, Timko C. Gender, treatment and self-help in remission from alcohol use disorders. Clin Med Res. 2006;4(3):163-174.
179. Bloomfield K, Gmel G, Wilsnack S. Introduction to special issue: gender, culture and alcohol problems: a multi-national study. Alcohol Alcohol Suppl. 2006;41(1):i3-i7.
180. Centers for Disease Control and Prevention. Fetal Alcohol Spectrum Disorders. Available at https://www.cdc.gov/ncbddd/fasd/facts.html. Last accessed April 24, 2024.
181. Coles CD, Platzman KA, Raskind-Hood CL, et al. A comparison of children affected by prenatal alcohol exposure and attention deficit, hyperactivity disorder. Alcohol Clin Exp Res. 1997;21(1):150-161.
182. Jacobson SW, Jacobson JL, Sokol RJ. Effects of fetal alcohol exposure on infant reaction time. Alcohol Clin Exp Res. 1994;18(5):1125-1132.
183. Kodituwakku PW,Handmaker NS, Cutler SK, Weathersby EK, Handmaker SD. Specific impairments in self-regulation in children exposed to alcohol prenatally. Alcohol Clin Exp Res. 1995;19(6):1558-1564.
184. Mattson SN, Riley EP. A review of the neurobehavioral deficits in children with fetal alcohol syndrome or prenatal exposure to alcohol. Alcohol Clin Exp Res. 1998;22(2):279-294.
185. Mattson SN, Riley EP, Delis DC, Stern C, Jones KL. Verbal learning and memory in children with fetal alcohol syndrome. Alcohol Clin Exp Res. 1996;20(5):810-816.
186. Roebuck TM, Mattson SN, Riley EP. Behavioral and psychosocial profiles of alcohol-exposed children. Alcohol Clin Exp Res. 1999;23(6):1070-1076.
187. Roebuck TM, Mattson SN, Riley EP. A review of the neuro-anatomical findings in children with fetal alcohol syndrome or prenatal exposure to alcohol. Alcohol Clin Exp Res. 1998;22(2):339-344.
188. Institute of Medicine, Stratton K, Howe C (eds). Fetal Alcohol Syndrome: Diagnosis, Epidemiology, Prevention, and Treatment. Washington, DC: National Academy Press; 1996.
189. Streissguth AP, Barr HM, Sampson PD, Parrish-Johnson JC, Kirchner GL, Martin DC. Attention, distraction, and reaction time at age 7 years and prenatal alcohol exposure. Neurobehav Toxicol Teratology. 1986;8(6):717-725.
190. Uecker A, Nadel L. Spatial locations gone awry: object and spatial memory deficits in children with fetal alcohol syndrome. Neuropsychologia. 1996;34(3):209-223.
191. IkonomidouC, Bittigau P, Ishimaru MJ, et al. Ethanol-induced apoptotic neurodegeneration and fetal alcohol syndrome. Science. 2000;287(5455):1056-1060.
192. Centers for Disease Control and Prevention. Fetal Alcohol Spectrum Disorders: Data and Statistics on FASDs. Available at https://www.cdc.gov/ncbddd/fasd/data.html. Last accessed April 24, 2024.
193. Quick S. Fetal alcohol syndrome: the nurse practitioner perspective. J Am Acad Nurse Pract. 1996;8(7):343-349.
194. Carson DK, Council JR, Volk MA. Temperament, adjustment, and alcoholism in adult female incest victims. Violence Vict. 1988;3(3):205-216.
195. Rose SM, Peabody CG, Stratigeas B. Undetected abuse among intensive case management clients. Hosp Community Psychiatry. 1991;42(5):499-503.
196. Higley JD, Suomi SJ, Linnoila M. A nonhuman primate model of type II excessive alcohol consumption? Part 1. Low cerebrospinal fluid 5-hydroxyindoleacetic acid concentrations and diminished social competence correlate with excessive alcohol consumption. Alcohol Clin Exp Res. 1996;20(4):629-642.
198. Brown GL, Linnoila MI. CSF serotonin metabolite (5-HIAA) studies in depression, impulsivity, and violence. J Clin Psychiatry. 1990;51(Suppl):31-43.
199. Centers for Disease Control and Prevention. Pricing Strategies for Alcohol Products. Available at https://www.cdc.gov/policy/hi5/alcoholpricing/. Last accessed April 24, 2024.
200. Grossman M, Markowitz S. Alcohol regulation and violence on college campuses. In: Grossman M, Hsieh CR, eds. Economic Analysis of Substance Use and Abuse: The Experience of Developed Countries and Lessons for Developing Countries. Cheltenham, UK: Edward Elgar; 2001.
201. Saffer H. Substance abuse control and crime: evidence from the National Survey of Drug Abuse. In: Grossman M, Hsieh CR, eds. Economic Analysis of Substance Use and Abuse: The Experience of Developed Countries and Lessons for Developing Countries. Cheltenham, UK: Edward Elgar; 2001.
202. Hollingworth W, Ebel BE, McCarty CA, Garrison MM, Christakis DA, Rivara FP. Prevention of deaths from harmful drinking in the United States: the potential effects of tax increases and advertising bans on young drinkers. J Stud Alcohol. 2006;67(2):300-308.
203. Elder RW, Lawrence B, Ferguson A, et al. The effectiveness of tax policy interventions for reducing excessive alcohol consumption and related harms. Am J Prevent Med. 2010;38(2):217-229.
204. Wagenaar AC, Tobler AL, Kornro KA. Effects of alcohol tax and price policies on morbidity and mortality: a systematic review. Am J Public Health. 2010;100(11):2270-2278.
205. National Council on Alcoholism and Drug Dependence. Alcohol, Drugs and Crime. Available at https://ncadd.us/about-addiction/addiction-update/alcohol-drugs-and-crime. Last accessed April24, 2024.
206. American Addiction Centers. How Drugs and Alcohol Can Fuel Violent Behaviors. Available at https://americanaddictioncenters.org/rehab-guide/addiction-and-violence. Last accessed April 24, 2024.
207. U.S. Department of Transportation. National Highway Traffic Safety Administration. Traffic Safety Facts. Overview of Motor Vehicle Traffic Crashes in 2022. Available at https://crashstats.nhtsa.dot.gov/Api/Public/ViewPublication/813560. Last accessed April 24, 2024.
208. Centers for Disease Control and Prevention. Transportation Safety: Impaired Driving: Get the Facts. Available at https://www.cdc.gov/transportationsafety/impaired_driving/impaired-drv_factsheet.html. Last accessed April 24, 2024.
209. 209. Lapham SC, Smith E, C'de Baca J, et al. Prevalence of psychiatric disorders among persons convicted of driving while impaired. Arch Gen Psychiatry. 2001;58(10):943-949.
210. Nelson SE, Belkin K, LaPlante DA, Bosworth L, Shaffer HJ. A prospective study of psychiatric comorbidity and recidivism among repeat DUI offenders. Arch Sci Psychol. 2015;3(1):8-17.
211. Robbe H. Marijuana's impairing effects on driving are moderate when taken alone but severe when combined with alcohol. Hum Psychopharmacol Clin Exp. 1998;13(S2):S70-S78.
212. Lacey JH, Kelley-Baker T, Furr-Holden D, et al. National Roadside Survey of Alcohol and Drug Use By Drivers: Drug Results. Washington, DC: National Highway Traffic Safety Administration; 2007.
213. 213. Biecheler MB, Peytavin JF, Facy F, Martineau H. SAM survey on "drugs and fatal accidents:" search of substances consumed and comparison between drivers involved under the influence of alcohol or cannabis. Traffic Inj Prev. 2008;9:11-21.
214. Walsh JM, Flegel R, Atkins R, Cangianelli LA, Cooper C, Welsh C, Kerns TJ. Drug and alcohol use among drivers admitted to a level-1 trauma center. Accid Anal Prev. 2005;37:894-901.
215. Berning A, Compton R, WochingerK. Results of the 2013–2014 National Roadside Survey of Alcohol and Drug Use by Drivers. Available at https://www.nhtsa.gov/sites/nhtsa.dot.gov/files/812118-roadside_survey_2014.pdf. Last accessed April 24, 2024.
216. Thomas FD, Darrah J, Graham L, et al. Alcohol and Drug Prevalence Among Seriously or Fatally Injured Road Users (Report No. DOT HS 813 399). Washington, DC: U.S. Department of Transportation, National Highway Traffic Safety Administration; 2022.
217. Stephen RJ, Martin PR. Co-occurring psychiatric disorders and alcoholism. Handb Clin Neurol. 2014;125:573-588.
218. Regier DA, Farmer ME, Rae DS, et al. Comorbidity of mental disorders with alcohol and other drug abuse. JAMA. 1990;264(19):2511-2518.
219. Davis L,Uezato A, Newell JM, Frazier E. Major depression and comorbid substance use disorders. Curr Opin Psychiatry. 2008;21(1):14-18.
220. Nunes EV, Liu X, Samet S, et al. Independent versus substance-induced major depressive disorder in substance-dependent patients: observational study of course during follow-up. J Clin Psychiatry. 2006;67(10):1561-1567.
221. Centers for Disease Control and Prevention. WISQARS Leading Causes of Death Visualization Tool. Available at https://wisqars.cdc.gov/lcd/?. Last accessed April 24, 2024.
222. Mann JJ. A current perspective of suicide and attempted suicide. Ann Intern Med. 2002;136(4):302-311.
223. Kim J, Lee KS, Kim DJ, et al. Characteristic risk factors associated with planned versus impulsive suicide attempters. Clin Psychopharmacol Neurosci. 2015;13(3):308-315.
224. Cornelius JR, Salloum IM, Day NL, Thase ME, Mann JJ. Patterns of suicidality and alcohol use in alcoholics with major depression. Alcohol Clin Exp Res. 1996;20(8):1451-1455.
225. Murphy GE, Wetzel RD. The lifetime risk of suicide in alcoholism. Arch Gen Psychiatry. 1990;47(4):383-392.
226. 226. Pandey S, Bolstad I, Lien L, Walby FA, Myhre MO, Bramness JG. Sex-specific factors associated with lifetime suicide attempt among patients with alcohol use disorders. BJPsych Open. 2022;8(4):e135.
227. Pompili M, Serafini G, Innamorati M, et al. Suicidal behavior and alcohol abuse. Int J Environ Res Public Health. 2010;7(4):1392-1431.
228. Powell KE, Kresnow MJ, Mercy JA, et al. Alcohol consumption and nearly lethal suicide attempts. Suicide Life Threat Behav. 2001;32(1 Suppl):30-41.
229. 229. Preuss UW, Schuckit MA, Smith TL, et al. Comparison of 3190 alcohol-dependent individuals with and without suicide attempts. Alcohol Clin Exp Res. 2002;26(4):471-477.
230. Akechi T, Iwasaki M, Uchitomi Y, Tsugane S. Alcohol consumption and suicide among middle-aged men in Japan. Bri J Psychiatry. 2006;18(8):231-236.
231. Agabio R, Trogu E, Pani PP. Antidepressants for the treatment of people with co-occurring depression and alcohol dependence. Cochrane Database Syst Rev. 2018;(4):CD008581.
232. Hashimoto E, Tayama M, Ishikawa H, Yamamoto M, Saito T. Influence of comorbid alcohol use disorder on treatment response of depressive patients. J Neural Transm (Vienna). 2015;122(2):301-306.
233. Garcia-Portilla MP, Bascarán MT, Saiz PA, et al. Effectiveness of venlafaxine in the treatment of alcohol dependence with comorbid depression. Actas Esp Psiquiatr. 2005;33(1):41-45.
234. LexiComp Online. Available at https://online.lexi.com. Last accessed April 24, 2024.
235. Tang VM, Yu D, Weissman CR, et al. Treatment outcomes in major depressive disorder in patients with comorbid alcohol use disorder: a STAR*D analysis. J Affect Disord. 2023;339:691-697.
236. McHugh RK, Weiss RD. Alcohol use disorder and depressive disorders. Alcohol Res. 2019;40(1):arcr.v40.1.01.
237. Addolorato G, Leggio L, Hopf FW, Diana M, Bonci A. Novel therapeutic strategies for alcohol and drug addiction: focus on GABA, ion channels and transcranial magnetic stimulation. Neuropsychopharmacology. 2012;37(1):163-177.
238. Chengappa KN, Levine J, Gershon S, Kupfer DJ. Lifetime prevalence of substance or alcohol abuse and dependence among subjects with bipolar I and II disorders in a voluntary registry. Bipolar Disord. 2000;2(3 Pt 1):191-195.
239. Xia Y, Ma D, Perich T, Hu J, Mitchell PB. Demographic and clinical differences between bipolar disorder patients with and without alcohol use disorders. Front Psychiatry. 2020;11:570574.
240. Brennan PL, Schutte KK, Moos RH. Pain and use of alcohol to manage pain: prevalence and 3-year outcomes among older problem and non-problem drinkers. Addiction. 2005;100(6):777-786.
241. National Institute on Alcohol Abuse and Alcoholism. The Complex Relationship Between Alcohol and Pain. Available at https://niaaa.scienceblog.com/231/the-complex-relationship-between-alcohol-and-pain/. Last accessed April 24, 2024.
242. Pennings EJ, Leccese AP, Wolff FA. Effects of concurrent use of alcohol and cocaine. Addiction. 2002;97(7):773-83.
243. Fettiplace MR, Pichurko A, Ripper R, et al. Cardiac depression induced by cocaine or cocaethylene is alleviated by lipid emulsion more effectively than by sulfobutylether-β-cyclodextrin. Acad Emerg Med. 2015;22(5):508-517.
244. Patten CA, Martin JE, Owen N. Can psychiatric and chemical dependency treatment units be smoke free? J Subst Abuse Treat. 1996;13(2):107-118.
245. Hurt RD, Offord KP, Croghan IT, et al. Mortality following inpatient addictions treatment: role of tobacco use in a community-based cohort. JAMA. 1996;275(14):1097-1103.
246. Pelucchi C, Gallus S, Garavello W, Bosetti C, La Vecchia C. Cancer risk associated with alcohol and tobacco use: focus on upper aero-digestive tract and liver. Alcohol Res Health. 2006;29(3):193-198.
247. Centers for Disease Control and Prevention. 2014 Surgeon General's Report: The Health Consequences of Smoking – 50 Years of Progress. Available at https://archive.cdc.gov/#/details?url=https://www.cdc.gov/tobacco/sgr/50th-anniversary/index.htm. Last accessed April 24, 2024.
248. Ahrendt SA, Chow JT, Yang SC, et al. Alcohol consumption and cigarette smoking increase the frequency of p53 mutations in non-small cell lung cancer. Cancer Res. 2000;60(12):3155-3159.
249. Barrett SP, Tichauer M, Leyton M, Pihl RO. Nicotine increases alcohol self-administration in non-dependent male smokers. Drug Alcohol Depend. 2006;81(2):197-204.
250. Montanari C, Secci ME, Driskell A, McDonald KO, Schratz CL, Gilpin NW. Chronic nicotine increases alcohol self-administration in adult male Wistar rats. Psychopharmacology (Berl). 2021;238(1):201-213.
251. Domi E, Xu L, Pätz M, et al. Nicotine increases alcohol self-administration in male rats via a μ-opioid mechanisms within the mesolimbic pathway. Br J Pharmacol. 2020;177(19):4516-4531.
252. Walsh RA, Bowman JA, Tzelepis F, et al. Smoking cessation interventions in Australian drug treatment agencies: a national survey of attitudes and practices. Drug Alcohol Rev. 2005;24:235-244.
253. Zullino DF, Besson J, Favrat B, et al. Acceptance of an intended smoking ban on an alcohol dependence clinic. Eur Psychiatry. 2003;18:255-257.
254. Richter KP, Choi AS, Alford DP. Smoking policies in U.S. outpatient drug treatment facilities. Nicotine Tob Res. 2005;7(3):475-480.
255. 255. Ziedonis DM, Guydish JW, Steinberg M, Foulds J. Barriers and solutions to addressing tobacco dependence in addiction treatment programs. Alcohol Res Health. 2007;9(3):228-235.
256. Hughes JR, Callas PW. Past alcohol problems do not predict worse smoking cessation outcomes. Drug Alcohol Depend. 2003;71:269-273.
257. Wang R, Li B, Jiang Y, Guan Y, Wang G, Zhao G. Smoking cessation mutually facilitates alcohol drinking cessation among tobacco and alcohol co-users: a cross-sectional study in a rural area of Shanghai, China. Tob Induc Dis. 2019;17:85.
258. Bulik CM, Klump KL, Thornton L, et al. Alcohol use disorder comorbidity in eating disorders: a multicenter study. J Clin Psychiatry. 2004;65(7):1000-1006.
259. Munn-Chernoff MA, Few LR, Matherne CE, et al. Eating disorders in a community-based sample of women with alcohol use disorder and nicotine dependence. Drug Alcohol Depend. 2020;212:107981.
260. Hodgkins CC, Jacobs WS, Gold MS. Weight gain after adolescent drug addiction treatment and supervised abstinence. Psychiatric Annals. 2003;33(2):112-116.
261. Kyngdon A, Dickerson M. An experimental study of the effect of prior alcohol consumption on a simulated gambling activity. Addiction. 1999;94(5):697-707.
262. Tobias-Webb J, Limbrick-Oldfield EH, Verancombe S, Duka T, Clark L. The effects of alcohol on sequential decision-making biases during gambling. Psychopharmacology (Berl). 2020;237(2):395-407.
263. Moyer VA. Screening and behavioral counseling interventions to reduce unhealthy alcohol use in adolescents and adults: U.S. Preventive Services Task Force Recommendation Statement. JAMA. 2018;320(18):1899-1909.
264. O'Connor EA, Perdue LA, Senger CA, et al. Screening and Behavioral Counseling Interventions in Primary Care to Reduce Unhealthy Alcohol Use in Adolescents and Adults: Updated Systematic Review for the US Preventive Services Task Force: Evidence Synthesis No. 171. Rockville, MD: Agency for Healthcare Research and Quality; 2018. AHRQ publication 18-05242-EF-1.
265. Madson MB, Schutts JW, Jordan HR, Villarosa-Hurlocker MC, Whitley RB, Mohn RS. Identifying at-risk college student drinkers with the AUDIT-US: a receiver operating characteristic curve analysis. Assessment. 2020;27(6):1089-1099.
266. National Institute on Alcohol Abuse and Alcoholism. Core Resource on Alcohol. Available at https://www.niaaa.nih.gov/health-professionals-communities/core-resource-on-alcohol/screen-and-assess-use-quick-effective-methods. Last accessed April 24, 2024.
267. Thompson W, Lande RG, Kalapatapu RK. Alcoholism. Available at https://emedicine.medscape.com/article/285913-overview. Last accessed April 24, 2024.
268. U.S. Department of Health and Human Services, National Institute on Alcohol Abuse and Alcoholism. The Physicians' Guide to Helping Patients with Alcohol Problems. Rockville, MD: National Institute on Alcohol Abuse and Alcoholism; 1995.
269. Saunders JB, Aasland OG, Babor TF, de la Fuente JR, Grant M. Development of the alcohol use disorder identification test (AUDIT): WHO collaborative project on early detection of persons with harmful alcohol consumption-II. Addiction. 1993;88(6):791-804.
270. Babor TF, Biddle-Higgins JC, Saunders JB, Monteiro MG. AUDIT: The Alcohol Use Disorders Identification Test. Guidelines for Use in Primary Health Care. 2nd ed. Geneva, Switzerland: World Health Organization; 1992.
271. Reinert DF, Allen JP. The Alcohol Use Disorders Identification Test (AUDIT): a review of recent research. Alcohol Clin Exp Res. 2002;26(2):272-279.
272. 272. AUDIT. Available at https://auditscreen.org/. Last accessed April 24, 2024.
273. AUDIT-C for Alcohol Use. Available at https://www.mdcalc.com/calc/2021/audit-c-alcohol-use. Last accessed April 24, 2024.
274. Substance Abuse and Mental Health Services Administration. Treatment Improvement Protocol (TIP) Series, No. 24. Rockville (MD): Centers for Substance Abuse Treatment; 1997.
275. Barnes AJ, Moore AA, Xu H, et al. Prevalence and correlates of at-risk drinking among older adults: the Project SHARE Study. J Gen Intern Med. 2010;25(8):840-846.
276. Moore AA,Giulu L, Gould R, et al. Alcohol use, comorbidity, and mortality. J Am Geriatr Soc. 2006;54(5):757-762.
277. Lee E. Cross-cultural communication: therapeutic use of interpreters. In: Lee E (ed). Working with Asian Americans: A Guide for Clinicians. New York, NY: The Guilford Press; 1997: 477-489.
278. National Hispanic Medical Association (NHMA). NHMA Screening and Brief Intervention Toolkit for the Hispanic Patient. National Hispanic Medical Association. Washington, DC; 2008.
279. National Institute on Alcohol Abuse and Alcoholism. Treatment for Alcohol Problems: Finding and Getting Help. Available at https://www.niaaa.nih.gov/publications/brochures-and-fact-sheets/treatment-alcohol-problems-finding-and-getting-help. Last accessed April 24, 2024.
280. 280. Bell H, Tallaksen CME, Try K, Haug E. Carbohydrate deficient transferrin and other markers of high alcohol consumption: a study of 502 patients admitted consecutively to a medical department. Alcohol Clin Exp Res. 1994;18(5):1103-1108.
281. Sorvajärvi K, Blake JE, Israel Y, Niemelä O. Sensitivity and specificity of carbohydrate-deficient transferrin as a marker of alcohol abuse are significantly influenced by alterations in serum transferrin. Alcohol Clin Exp Res. 1996;20(3):449-454.
282. Cylwik B, Chrostek L, Szmitkowski M. New methods for the determination of transferrin isoforms in the diagnostics of alcohol abuse. Postepy Hig Med Dosw. 2006;60:101-112.
283. Mayo Clinic. Test ID: CDTA. Available at https://www.mayocliniclabs.com/test-catalog/Specimen/82425. Last accessed April 24, 2024.
284. Arndt T, Gilg T, Soyka M. Diagnosis of alcoholism: CDT test does help. MMW Fortschr Med. 2000;146(26-27):10-11.
285. Sillanaukee P, Massot N, Jousilahti P, et al. Dose response of laboratory markers to alcohol consumption in a general population. Am J Epidemiol. 2000;152(8):747-751.
286. Hannuksela ML, Liisanantti MK, Nissinen AE, Savolainen MJ. Biochemical markers of alcoholism. Clin Chem Lab Med. 2007;45(8):953-61.
288. Anderson G. Management of alcohol problems: the role of the general practitioner. Alcohol Alcohol. 1993;28(3):263-272.
289. Miller WR, Sanchez VC. Motivating young adults for treatment and lifestyle change. In: Howard G, Nathan P (eds). Alcohol Use and Misuse by Young Adults. Notre Dame, IN: University of Notre Dame Press; 1994.
290. Bien TH, Miller WR, Tonigan JS. Brief interventions for alcohol problems: a review. Addiction. 1993;88(3):315-335.
291. Mattoo SK, Prasad S, Ghosh A. Brief intervention in substance use disorders. Indian J Psychiatry. 2018;60(Suppl 4):S466-S472.
292. Graham AW, Fleming MS. Brief interventions. In: Graham AW, Schultz TK, Wilford BB (eds). Principles of Addiction Medicine. 3rd ed. Chevy Chase, MD: American Society of Addiction Medicine, Inc.; 2003: 361-372.
293. Henry-Edwards S, Humeniuk R, Ali R, Monteiro M, Poznyak V. Brief Intervention for Substance Use: A Manual for Use in Primary Care. Geneva: World Health Organization; 2003.
294. Carroll KM, Ball SA,Nich C, et al. Motivational interviewing to improve treatment engagement and outcome in individuals seeking treatment for substance abuse: a multisite effectiveness study. Drug Alcohol Depend. 2006;81(3):301-312.
295. Chafetz ME, Blane HT, Abram HS, et al. Establishing treatment relations with alcoholics. J Nerv Ment Dis. 1962;134(5):395-409.
296. National Institute on Alcohol Abuse and Alcoholism. Matching alcoholism treatments to client heterogeneity: Project MATCH posttreatment drinking outcomes. J Stud Alcohol. 1997;58(1):7-29.
297. Whitlock EP, Green CA, Polen MR, et al. Behavioral Counseling Interventions in Primary Care to Reduce Risky/Harmful Alcohol Use. Systematic Evidence Reviews, No. 30. Rockville, MD: Agency for Healthcare Research and Quality; 2004,
298. McCambridge J, Rollnick S. Should brief interventions in primary care address alcohol problems more strongly? Addiction. 2014;109(7):1054-1058.
300. National Institute on Alcohol Abuse and Alcoholism. Matching alcoholism treatments to client heterogeneity: Project MATCH three-year drinking outcomes. Alcohol Clin Exp Res. 1998;22(6):1300-1311.
301. Moyer A, Finney JW, Swearingen CE, Vergun P. Brief interventions for alcohol problems: a meta-analytic review of controlled investigations in treatment-seeking and non-treatment-seeking populations. Addiction. 2002;97(3):279-292.
302. Fleming MF, Mundt MP, French MT, et al. Brief physician advice for problem drinkers: long-term efficacy and benefit-cost analysis. Alcohol Clin Exp Res. 2002;26(1):36-43.
303. Tanner-Smith EE, Risser MD. A meta-analysis of brief alcohol interventions for adolescents and young adults: variability in effects across alcohol measures. Am J Drug Alcohol Abuse. 2016;42(2):140-151.
304. American Society of Addiction Medicine. About the ASAM Criteria. Available at https://www.asam.org/asam-criteria/about-the-asam-criteria. Last accessed April 24, 2024.
305. Donovan DM, Rosengren DB. Motivation for behavior change and treatment among substance abusers. In: Tucker JA, Donovan DM, Marlatt GA (eds). Changing Addictive Behavior: Bridging Clinical and Public Health Strategies. New York, NY: The Guilford Press; 1999: 127-159.
306. Prochaska JO, Norcross JC, DiClemente CC. Changing for Good: The Revolutionary New Program that Explains the Six Stages of Change and Teaches You How to Free Yourself from Bad Habits. New York, NY: William Morrow; 1994.
307. Bayard M, McIntyre J, Hill KR, Woodside J Jr. Alcohol withdrawal syndrome. Am Fam Physician. 2004;69(6):1443-1450.
308. Merck Manual. CIWA-Ar Clinical Institute Withdrawal Assessment for Alcohol Scale. Available at https://www.merckmanuals.com/professional/multimedia/clinical-calculator/ciwa-ar-clinical-institute-withdrawal-assessment-for-alcohol-scale. Last accessed April 24, 2024.
309. Kattimani S, Bharadwaj B. Clinical management of alcohol withdrawal: a systematic review. Ind Psychiatry J. 2013;22(2):100-108.
310. Sachdeva A, Choudhary M, Chandra M. Alcohol withdrawal syndrome: benzodiazepines and beyond. J Clin Diagn Res. 2015;9(9):VE01-VE07.
311. 311. Long D, Long B, Koyfman A. The emergency medicine management of severe alcohol withdrawal. Am J Emerg Med. 2017;35(7):1005-1011.
312. Modesto-Lowe V, Barron GC, Aronow B, Chaplin M. Gabapentin for alcohol use disorder: a good option, or cause for concern? Clev Clin J Med. 2019;86(12):815-823.
313. Toohey S. Delirium Tremens (DTs) Medication. Available at https://emedicine.medscape.com/article/166032-medication#. Last accessed April 24, 2024.
314. Addolorato G, Leggio L, Hopf FW, Diana M, Bonci A. Novel therapeutic strategies for alcohol and drug addiction: focus on GABA, ion channels, and transcranial magnetic stimulation. Neuropsychopharmacology. 2012;37:163-177.
315. McKay JR, Lynch KG, Shepard DS, Pettinati HM. The effectiveness of telephone-based continuing care for alcohol and cocaine dependence 24-month outcomes. Arch Gen Psychiatry. 2005;62(2):199-207.
316. National Institute on Alcohol Abuse and Alcoholism. NIAAA Reports Project MATCH Main Findings. Available at https://www.niaaa.nih.gov/news-events/news-releases/niaaa-reports-project-match-main-findings. Last accessed April 24, 2024.
317. Kelly JF, Abry A, Ferri M, Humphreys K. Alcoholics Anonymous and 12-step facilitation treatments for alcohol use disorder: a distillation of a 2020 Cochrane Review for clinicians and policy makers. Alcohol Alcohol. 2020;55(6):641-651.
318. Hathaway B. Why "One Day at a Time" Works for Recovering Alcoholics. Available at https://news.yale.edu/2020/08/27/why-one-day-time-works-recovering-alcoholics. Last accessed April 24, 2024.
319. Dutra L, Stathopoulou G, Basden SL,Leyro TM, Powers MB, Otto MW. A meta-analytic review of psychosocial interventions for substance use disorders. Am J Psychiatry. 2008;165:179-187.
320. Magill M, Ray LA. Cognitive-behavioral treatment with adult alcohol and illicit drug users: A meta-analysis of randomized controlled trials. J Stud Alcohol Drugs. 2009;70:516-527.
321. Liese BS, Beck AT. Beyond the therapeutic alliance: keeping the drug-dependent individual in treatment. In: Simon Onken L, Blaine JD, Boren JJ (eds). Back to Basics: Fundamental Cognitive Therapy Skills for Keeping Drug-Dependent Individuals in Treatment: NIDA Research Monograph 165. Rockville, MD: National Institute on Drug Abuse; 1997: 207-232.
322. Shawe-Taylor M, Rigby J. Cognitive behaviour therapy: its evolution and basic principles. J R Soc Promot Health. 1999;119(4):244-246.
323. Wright JH, Thase ME, Beck AT. Cognitive therapy. In: Hale RE, Yudofsky SC, Talbott JA (eds). The American Psychiatric Press Textbook of Psychiatry. 4th ed. Arlington, VA: American Psychiatric Publishing; 2008.
324. Kranzler HR. Pharmacotherapy of alcoholism: gaps in knowledge and opportunities for research. Alcohol Alcohol. 2000;35(6):537-547.
325. Substance Abuse and Mental Health Services Administration and National Institute on Alcohol Abuse and Alcoholism. Medication for the Treatment of Alcohol Use Disorder: A Brief Guide. HHS Publication No. (SMA) 15-4907. Rockville, MD: Substance Abuse and Mental Health Services Administration; 2015.
326. Miller NS, Gold MS. Alcohol. In: Gold MS (ed). Drugs of Abuse: A Comprehensive Series. New York, NY: Plenum Medical Book Co.; 1991.
327. 327. Garbutt JC, West SL, Carey TS, Lohr KN, Crews FT. Pharmacological treatment of alcohol dependence: a review of the evidence. JAMA. 1999;281(14):1318-1325.
328. Yoshimura A, Kimura M, Nakayama H, et al. Efficacy of disulfiram for the treatment of alcohol dependence assessed with a multicenter randomized controlled trial. Alcohol Clin Exp Res. 2014;38(2):572-578.
329. Skinner MD, Lahmek P, Pham H, Aubin HJ. Disulfiram efficacy in the treatment of alcohol dependence: a meta-analysis. PLoS One. 2014;9(2):e87366.
330. Fuller RK, Gordis E. Does disulfiram have a role in alcoholism treatment today? Addiction. 2004;99(1):21-24.
331. Agency for Healthcare Quality and Research. Pharmacotherapy for Alcohol Dependence: Evidence Report/Technology Assessment No. 3. Available athttps://archive.ahrq.gov/clinic/epcsums/alcosumm.htm. Last accessed April 24, 2024.
332. Volpicelli JR, Volpicelli LA, O'Brien CP. Medical management of alcohol dependence: clinical use and limitations of naltrexone treatment. Alcohol Alcohol. 1995;30(6):789-798.
333. Clapp P. Current progress in pharmacologic treatment strategies for alcohol dependence. Expert Rev Coin Pharmacol. 2012;5(4):427-435.
334. Altshuler HL, Phillips PE, Feinhandler DA. Alteration of ethanol self-administration by naltrexone. Life Sci. 1980;26(9):679-688.
335. Volpicelli JR, Alterman AI, Hayashida M, O'Brien CP. Naltrexone in the treatment of alcohol dependence. Arch Gen Psychiatry. 1992;49(11):876-880.
336. O'Malley SS, Jaffe AJ, Chang G, Schottenfeld RS, Meyer RE, Rounsaville B. Naltrexone and coping skills therapy for alcohol dependence: a controlled study. Arch Gen Psychiatry. 1992;49(11):881-887.
337. O'Malley SS, Jaffe AJ, Chang G, et al. Six-month follow-up of naltrexone and psychotherapy for alcohol dependence. Arch Gen Psychiatry. 1996;53(3):217-224.
338. Monterosso JR, Flannery BA, Pettinati HM, et al. Predicting treatment response to naltrexone: the influence of craving and family history. Am J Addict. 2001;10(3):258-268.
339. Capone C, Kahler CW, Swift RM, O'Malley SS. Does family history of alcoholism moderate naltrexone's effects on alcohol use? J Stud Alcohol Drugs. 2011;72(1):135-140.
340. Worley MJ, Witkiewitz K, Brown SA, Kivlahan DR, Longabaugh R. Social network moderators of naltrexone and behavioral treatment effects on heavy drinking in the COMBINE study. Alcohol Clin Exp Res. 2015;39(1):93-100.
341. National Institute on Alcohol Abuse and Alcoholism. A Multisite Trial of Combined Pharmacotherapies and Behavioral Interventions (COMBINE) for Alcohol Dependence. Available at https://www.niaaa.nih.gov/multisite-trial-combined-pharmacotherapies-and-behavioral-interventions-combine-alcohol-dependence. Last accessed April 24, 2024.
342. Witkiewitz K, Roos CR, Mann K, Kranzler HR. Advancing precision medicine for alcohol use disorder: replication and extension of reward drinking as a predictor of naltrexone response. Alcohol Clin Exp Res. 2019;43(11):2395-2405.
343. Anton RF, O'Malley SS, Ciraulo DA, et al. Combined pharmacotherapies and behavioral interventions for alcohol dependence: the COMBINE study: a randomized controlled trial. JAMA. 2006;295(17):2003-2017.
344. Pettinati HM, O'Brien CP, Rabinowitz AR, et al. The status of naltrexone in the treatment of alcohol dependence: specific effects on heavy drinking. J Clin Psychopharmacol. 2006;26(6):610-625.
345. Richardson K, Baillie A, Reid S, et al. Do acamprosate or naltrexone have an effect on daily drinking by reducing craving for alcohol? Addiction. 2008;103(6):953-959.
346. Mason BJ. Treatment of alcohol-dependent outpatients with acamprosate: a clinical review. J Clin Psychiatry. 2001;62(Suppl 20):42-48.
347. Tempesta E, JaniriL, Bignamini A, Chabac S, Potgieter A. Acamprosate and relapse prevention in the treatment of alcohol dependence: a placebo-controlled study. Alcohol Alcohol. 2000;35(2):202-209.
348. Rösner S, Hackl-Herrwerth A, Leucht S, Lehert P, Becchi S, Soyka M. Acamprosate for alcohol dependence. Cochrane Database Syst Rev. 2010;(9):CD004332.
349. Mason BJ. Acamprosate for alcohol dependence: an update of the clinician. Focus. 2006;4:505-511.
350. Flannery BA, Garbutt JC, Cody MW, et al. Baclofen for alcohol dependence: a preliminary open-label study. Alcohol Clin Exp Res. 2004;28(10):1517-1523.
351. Addolorato G, Leggio L, Ferrulli A, et al. Effectiveness and safety of baclofen for maintenance of alcohol abstinence on alcohol-dependent patients with liver cirrhosis: randomised, double-blind controlled study. Lancet. 2007;370(9603):1915-1922.
352. Rose AK, Jones A. Baclofen: its effectiveness in reducing harmful drinking, craving, and negative mood: a meta-analysis. Addiction. 2018;113(8):1396-1406.
353. Agabio R, Saulle R, Rosner S, Minozzi S. Baclofen for alcohol use disorder. Cochrane Database Syst Rev. 2023;1:CD012557.
354. Guglielmo R, Martinotti G, Quatrale M, et al. Topiramate in alcohol use disorders: review and update. CNS Drugs. 2015;29(5):383-395.
355. Johnson BA, Rosenthal N, Capece JA, et al., for the Topiramate for Alcoholism Advisory Board and the Topiramate for Alcoholism Study Group. Topiramate for treating alcohol dependence: a randomized controlled trial. JAMA. 2007;298(14):1641-1651.
356. Williams SH. Medications for treating alcohol dependence. Am Fam Physician. 2005;72(9):1775-1780.
357. Johnson BA, Ait-Daoud N, Bowden CL, et al. Oral topiramate for treatment of alcohol dependence: a randomised controlled trial. Lancet. 2003;361(9370):1677-1685.
358. Barrons R, Roberts N. The role of carbamazepine and oxcarbazepine in alcohol withdrawal syndrome. J Clin Pharm Ther. 2010;35(2):153-167.
359. Mueller TI, Stout RL, Rudden S, et al. A double-blind, placebo-controlled pilot study of carbamazepine for the treatment of alcohol dependence. Alcohol Clin Exp Res. 1997;21(1):86-92.
360. Martinotti G, Di Nicola M, Romanelli R, et al. High and low dosage oxcarbazepine versus naltrexone for the prevention of relapse in alcohol-dependent patients. Hum Psychopharmacol Clin Exp. 2007;22(3):149-156.
361. 361. Johnson BA, Roache JD, Javors MA, et al. Ondansetron for reduction of drinking among biologically predisposed alcoholic patients: a randomized controlled trial. JAMA. 2000;284(8):963-971.
362. Kenna GA, Zywiak WH, Swift RM, et al. Ondansetron reduces naturalistic drinking in nontreatment-seeking alcohol-dependent individuals with the LL 5'-HTTLPR genotype: a laboratory study. Alcohol Clin Exp Res. 2014;38(6):1567-1574.
363. Kranzler HR, Burleson JA, Del Boca FK, et al. Buspirone treatment of anxious alcoholics: a placebo-controlled trial. Arch Gen Psychiatry. 1994;51(9):720-731.
364. Gimeno C, Dorado ML, Roncero C, et al. Treatment of comorbid alcohol dependence and anxiety disorder: review of the scientific evidence and recommendations for treatment. Front Psychiatry. 2017;8:173.
365. 365. Drake RE, Xie H, McHugo GJ, Green AI. The effects of clozapine on alcohol and drug use disorders among patients with schizophrenia. Schizophr Bull. 2000;26(2):441-449.
366. Lowry F. Drug May Aid in Alcohol Dependence, but Safety Concerns Linger. Available at https://www.medscape.com/viewarticle/805427. Last accessed April 24, 2024.
367. Schacht JP, Anton RF, Randall PK, Li X, Henderson S, Myrick H. Varenicline effects on drinking, craving and neural reward processing among non-treatment-seeking alcohol-dependent individuals. Psychopharmacology (Berl). 2014;231(18):3799-3807.
368. Erwin BL, Slaton RM. Varenicline in the treatment of alcohol use disorders. Ann Pharmacother. 2014;48(11):1445-1455.
369. 369. Mitchell JM, Teague CH, Kayser AS, Bartlett SE, Fields HL. Varenicline decreases alcohol consumption in heavy-drinking smokers. Psychopharmacology (Berl). 2012;223(3):299-306.
370. McKeown NJ, West PL. Withdrawal Syndromes. Available at https://emedicine.medscape.com/article/819502-overview. Last accessed April 24, 2024.
371. Fisher CM. Prompt responses to the administration of ethanol in the treatment of the alcohol withdrawal syndrome (AWS). Neurologist. 2009;15(5):24í2-244.
372. Hayner CE, Wuestefeld NL, Bolton PJ. Phenobarbital treatment in a patient with resistant alcohol withdrawal syndrome. Pharmacotherapy. 2009;29(7):875-878.
373. Weinberg JA, Magnotti LJ, Fischer PE, et al. Comparison of intravenous ethanol versus diazepam for alcohol withdrawal prophylaxis in the trauma ICU: results of a randomized trial. J Trauma. 2008;64(1):99-104.
374. American Psychiatric Association. The American Psychiatric Association Practice Guideline for the Pharmacological Treatment of Patients with Alcohol Use Disorder. Available at https://psychiatryonline.org/doi/book/10.1176/appi.books.9781615371969. Last accessed April 24, 2024.
1. U.S. Preventive Services Task Force. Unhealthy Alcohol Use in Adolescents and Adults: Screening and Behavioral Counseling Interventions. Available at https://www.uspreventiveservicestaskforce.org/uspstf/recommendation/unhealthy-alcohol-use-in-adolescents-and-adults-screening-and-behavioral-counseling-interventions. Last accessed May 20, 2024.
2. World Health Organization. Guidelines for the Identification and Management of Substance Use and Substance Use Disorders in Pregnancy. Geneva: World Health Organization; 2014. Available at https://www.who.int/publications/i/item/9789241548731. Last accessed May 20, 2024.
3. Management of Substance Use Disorders Work Group. VA/DoD Clinical Practice Guideline for the Management of Substance Use Disorders. Washington, DC: Department of Veterans Affairs, Department of Defense; 2021. Available at https://www.healthquality.va.gov/guidelines/mh/sud. Last accessed May 20, 2024.
Mention of commercial products does not indicate endorsement.